Abstract
To determine a suitable condition for in vitro infection model of Cryptosporidium parvum, four different cell lines, AGS, MDCK, HCT-8 and Caco-2, were used as host cell lines which were cultured at various concentrations of added supplements. These supplement include fetal bovine serum (FBS), sodium choleate, ascorbic acid, folic acid, calcium pantothenate, para-aminobenzoic acid and pyruvate and their effects on the cell lines which were infected with C. parvum were evaluated. The results of this study showed that the AGS cell line was most susceptible to C. parvum whereas the Caco-2 cells appeared to be least susceptible to C. parvum. In regards to the serum condition, 10% FBS was suitable for the growth of AGS and HCT-8 cells, and 1% FBS was good for the growth of the MDCK cells when they were inoculated with C. parvum. Vitamines had a positive effect on the AGS cells, and pyruvate also showed positive effects on all of the cell lines except for Caco-2. Modified medium for each cell line was prepared by adding appropriate amounts of each supplement which resulted in the highest parasite infection number. Modified media increased the number of parasites infected on AGS cells to 2.3-fold higher when compared to the control media. In this study, we found that the AGS cell line was a suitable host model for evaluating C. parvum in vitro study and the media contents for the optimal infection conditions were suggested.
Keywords: Cryptosporidium parvum, in vitro study, AGS
INTRODUCTION
Cryptosporidium parvum is a well-known opportunistic protozoa inducing diarrheal disease from immunosuppressed patients. About 24 years have been passed since the first human case was reported in 1976, but no specific anti-cryptosporidial agent has been discovered yet (Nime et al., 1976). In vitro system is considered to be less expensive and more convenient to study the biology of Cryptosporidium and to screen the anti-cryptosporidial chemotherapeutic agents. Although numerous in vitro cell culture systems have been devised (Buraud et al., 1991; Gut et al., 1991; Rasmussen et al., 1993; Upton et al., 1994), they were not always reproducible. In this study, we compared four different cell lines as the hosts to design a suitable in vitro system for C. parvum infection. Also, a total of seven supplements was used independently or in conjunction with one another to determine the optimal condition for each cell line.
MATERIALS AND METHODS
Oocyst preparation
Oocysts of C. parvum were isolated from the laboratory mice (C57BL/6J) at the laboratory of Parasitology at Konkuk University in 1997 and maintained through C57BL/6J mice. The 5 week-old mice were immunosuppressed with intramuscular injection of Depomedrol® (0.1 mg/g, Korea Upjohn Ltd.) one week before infecting them with C. parvum. The oocyst expulsion was detected by stool examination using modified acid fast staining from 3 days after infection. The feces containing oocysts were collected in a solution containing 2.5% potassium dichromate and kept at 4℃ until further purification. The oocyst purification was carried out according to a method described by Petry et al. (1995). Briefly, the feces were passed through a metal sieve (pore size, 500 µm) and resuspended in 15 to 20 volumes of water. After standing at the room temperature for 10 min, the supernatant was decanted and centrifuged at 1,600 g for 10 min. The pellet was resuspended in saturated NaCl and centrifuged at 1,600 g for 10 min. The uppermost layer containing oocysts was obtained and washed three times with 10 volumes of water. The oocyst pellet was surface sterilized by standing for 10 min in 10% sodium hypochlorite solution and washed three times in ice cold sterile distilled water. Oocysts were excysted by incubation in 1% trypsin in PBS at 37℃ for 1 hr.
Preparation of host cell lines
The host cell lines used were human stomach adenocarcinoma (AGS), Madin-Darby Canine kidney cell (MDCK), human ileocecal adenocarcinoma (HCT-8), (Korean Cell Line Bank, Seoul, Korea) and Caco-2 (American Type Culture Collection, Rockville, MD, USA). All of these cell lines have the epithelial cell nature. The three host cell lines except Coco-2 were incubated in RPMI 1640 with L-glutamine (Sigma) containing 15 mM N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid (HEPES), 14 mM sodium bicarbonate, 100 U of penicillin per ml, 100 µg of streptomycin per ml, and 0.25 µg of amphotericin B per ml. The Caco-2 cells were maintained in an Eagle's minimum essential medium. For a routine cell passage, 10% fetal bovine serum (FBS) (Gibco BRL, Gaithersburg, MD, USA) were used; however, after worm inoculation, 10% FBS was used for AGS and HCT-8 whereas 1% FBS was used for the MDCK cells. The host cells for infection were plated onto coverglass 22 mm2 placed in 6 well plates and incubated at 37℃ in a 6% CO2-94% air humidified incubator.
Parasite inoculation
Oocysts were washed once with RPMI 1640 medium after excystation and inoculated 106 per well when the host cell made 70% confluency. Seven kinds of supplements such as FBS, sodium choleate (Sigma), L-ascorbic acid (Sigma), folic acid (Sigma), calcium pantothenate (CAPN, Sigma), para-aminobenzoic acid (PABA, Sigma), pyruvate (Sigma) were tested to evaluate the proper media condition for parasite infection. The tested concentrations of supplements were 1%, 5%, and 10% of FBS; 0.2 mg of sodium choleate per ml; 8.75 and 35 µg of ascorbic acid per ml; 0.25 and 1 µg of folic acid per ml; 0.5, 1, 2, and 4 µg of CAPN per ml; 1, 4, and 8 µg of PABA per ml; 0, 5, 1, and 2.5 mM pyruvate.
Infection evaluation
The cover glass was taken out from the well after 24 hr of incubation at 37℃ in a 6% CO2-94% air humidified incubator, washed in PBS, pH 7.4 and fixed in methanol for 1 min. The cover glass was stained by hematoxylin and eosin method and then mounted on the slide glass. The total number of parasites infected in host cells was counted from 25 fields under the light microscope with a resoultion of 1,000 × (oil) lens (Olympus). The experimental data of each group were compared with those of control groups using student t-test.
RESULTS
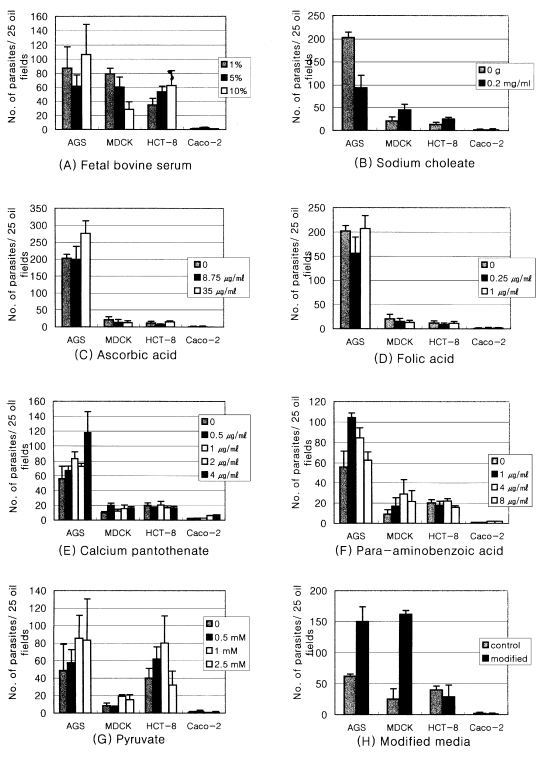
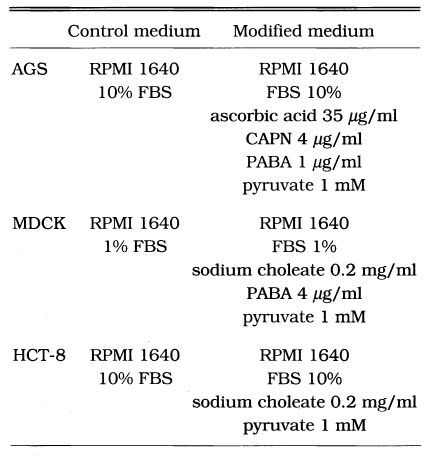
The AGS cell line always showed a higher rate of infection when compared to those of other cell lines (Fig. 1). However, the infected parasite number in Caco-2 cells was significantly lower than those of any other cell lines regardless of the kind of media supplement. The optimal serum concentration leading to the highest infection number varied according the cell line used. The highest number was reached by using 10% FBS in AGS and HCT-8 cell lines and by 1% in MDCK (Fig. 2A). From this result, the FBS concentrations of the media for further supplement experiments were maintained at 10% for AGS and HCT-8 whereas the concentration of FBS was kept at 1% for the MDCK cells. AGS showed a significantly higher number of infection rate (p<0.05) when the medium was deprived of the bile; however, MDCK and HCT-8 showed increased rate of infection when 0.2 mg/ml of bile was added to the media (Fig. 2B). The infected parasite number increased (p<0.05) with 35 µg/ml of ascorbic acid in AGS cell line only (Fig. 2C). The other cells were not affected by ascorbic acid. Folic acid did not make any difference on the infection of C. parvum to all of host cells tested (Fig. 2D). The parasite number also increased with the addition of 4 µg/ml of CAPN in AGS (Fig. 2E). Paraaminobenzoic acid had a positive effect on the number of parasite infected in AGS and MDCK cells (Fig. 2F). One mM pyruvate also made a positive effect on increasing the number of parasite infected in three cell lines (Fig. 2G). Modifed media were prepared based on the above results (Table 1). The number of infected parasite increased 2.3-fold in AGS and 4.7-fold in MDCK cells when the modified media were applied rather than the use of control media (Fig. 2H).
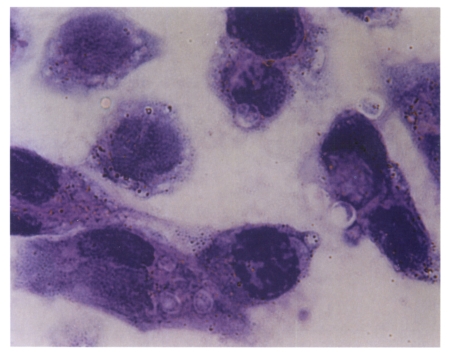
Fig. 1.
Cryptosporidium parvum infected in AGS cells. H&E stain, ×1,000.
Fig. 2.
The effects of seven supplements on Cryptosporidium parvum in vitro infection to four host cell lines.
Table 1.
Contents of the modified media based on the individual supplement experiment
DISCUSSION
Previously, Caco-2, RL 95-2, and MDCK cells have been reported as suitable in vitro systems for the complete development of C. parvum (Buraud et al., 1991; Gut et al., 1991; Rasmussen et al., 1993). It was reported that HCT-8 was the most susceptible host cell line for C. parvum in vitro (Upton et al., 1994), and 10 nutrient additives including insulin and vitamins were helpful in increasing the number of parasite in in vitro infection (Upton et al., 1995). The AGS cell line has not been investigated as a host cell line; however, in this experiment, AGS was found to be the most susceptible cell line for the in vitro culture of C. parvum isolated in our laboratory when compared to those of the others including MDCK, HCT-8 or Caco-2 cell lines. In terms of the habitat of C. parvum, eventhough the intestinal epithelium is known as the most common area, the epithelial cells of other internal organs such as the gall bladder, liver, pancreas, and respiratory tract were also reported as common habitats as well (O'Donoghue, 1995). The AGS cell line is originated from human stomach adenocarcinoma which has the epithelial cell nature. Therefore, it is not a strange result that C. parvum could infect stomach origin epithelial cells. However, in vivo status, C. muris, another mammalian Cryptosporidium species, has been reported to infect the stomach instead of C. parvum. Through this result, it can be speculated that the pH difference of infection environment between in vivo and in vitro may be the reason for possible infection of C. parvum to the stomach epithelial cells in vitro. The pH of medium already has been suggested to be an important factor to induce better condition of sporozoite infectivity, and C. parvum was thought to favor neutral pH (Upton et al., 1995).
Although the Caco-2 cell line has been known as a good model for C. parvum infection for its similarity with in vivo system, we could not find any apparent advantage for the following reasons: 1) the growth of the cell takes longer than the other cells, 2) they are very easy to be detached from the bottom of culture plate, 3) there are too many cytoplasmic vacuoles enough to be misunder-stood as parasitophorous vacuoles, and 4) the infected worm numbers are too low regardless of any kind of supplement conditions.
The role of FBS in motility and in the development of some coccidia in vitro is known as an important factor (Upton and Tilley, 1992). In this study, the optimal FBS concentration was different according to different cell lines. AGS and HCT-8 cells required 10% FBS, but MDCK needed only 1% FBS. Generally 10% FBS is acceptable for in vitro cell cultivation for various cell lines, but high concentrations of FBS could be an inhibitory factor to C. parvum infection as in MDCK cells, so the FBS concentration of infection medium should be readjusted when we inoculate Cryptosporidium in the medium. While the bile gave an negative effect on the AGS cell line, vitamin additives gave a positive effect except folic acid.
Pyruvate is known to enhance the development of some eimerians in cell cultures (Schmatz, 1987), but for a C. parvum culture in HCT-8, it was thought to be unnecessary as reported by Upton et al. (1995) since C. parvum did not possess mitochondria. However, in this study, 1 mM pyruvate enhanced the number of parasites in all three cell lines. The difference of pyruvate requirement is not clear between two studies, but the parasite strain difference could explain the difference partially. The modified medium prepared based on the individual supplement data is found to be effective to enhance the infection of parasite compared to the control medium which did not include other supplements but FBS in AGS and MDCK cell lines.
From this study, we found that AGS cell line can be a suitable in vitro model system for C. parvum infection. With the modified media including vitamins and pyruvate, the infection efficacy could be increased in AGS and MDCK cell lines.
Footnotes
This study was partly supported by the grant of Science and Technology Policy Institute, the Ministry of Science and Technology, Korea, 1998-1999.
References
- 1.Buroud M, Forget E, Favennec L, Bizet J, Gobert JG, Deluol AM. Sexual stage development of Cryptosporidia in the Caco-2 cell line. Infect Immun. 1991;59:4610–4613. doi: 10.1128/iai.59.12.4610-4613.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gut J, Petersen C, Nelson R, Leech J. Cryptosporidium parvum: in vitro cultivation in Madin-Darby Canine Kidney cells. J Protozool. 1991;38:72S–73S. [PubMed] [Google Scholar]
- 3.Nime FA, Burek JD, Page DL, Holscher MA, Yardley JH. Acute enterocolitis in a human being infected with the protozoan Cryptosporidium. Gastroenterology. 1976;70:592–598. [PubMed] [Google Scholar]
- 4.O'Donoghue PJ. Cryptosporidium and cryptosporidiosis in man and animals. Int J Parasitol. 1995;25:139–195. doi: 10.1016/0020-7519(94)e0059-v. [DOI] [PubMed] [Google Scholar]
- 5.Petry F, Robinson HA, McDonald V. Murine infection model for maintenance and amplification of Cryptosporidium parvum oocysts. J Clin Microbiol. 1995;33:1922–1924. doi: 10.1128/jcm.33.7.1922-1924.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rasmussen KR, Larsen NC, Healey MC. Complete development of Cryptosporidium parvum in a human endometrial carcinoma cell line. Infect Immun. 1993;61:1482–1485. doi: 10.1128/iai.61.4.1482-1485.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schmatz D. In vitro cultivation of the avian coccidia. Adv Cell Culture. 1987;5:241–266. [Google Scholar]
- 8.Upton SJ, Tilley M. Effect of select media supplements on motility and development of Eimeria nieschulzi in vitro. J Parasitol. 1992;78:329–333. [PubMed] [Google Scholar]
- 9.Upton SJ, Tilley M, Brillhart DB. Comparative development of Cryptosporidium parvum (Apicomplexa) in 11 continuous hostcell lines. FEMS Microbiol Letters. 1994;118:223–236. doi: 10.1111/j.1574-6968.1994.tb06833.x. [DOI] [PubMed] [Google Scholar]
- 10.Upton SJ, Tilley M, Brillhart DB. Effects of select medium supplements on in vitro development of Cryptosporidium parvum in HCT-8 cells. J Clin Microbiol. 1995;33:371–375. doi: 10.1128/jcm.33.2.371-375.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]