Abstract
Background
Vascular localization of symptomatic large artery atherosclerotic (LAA) stroke differs for unknown reasons by race-ethnicity. The metabolic syndrome (MetSD) is associated with higher atherosclerotic stroke risk and comprises abnormal risk factors that can vary by race. Thus, we investigated whether MetSD may contribute to race-ethnic differences in LAA stroke by examining the association of MetSD with symptomatic intra- and extracranial atherosclerosis among a diverse race-ethnic group.
Method
We analyzed data prospectively collected over a 4-year period on subjects with ischemic stroke/TIA. Independent vascular risk factor associations with intracranial and extracranial LAA vs. non-LAA mechanism were evaluated in two groups stratified by race-ethnicity; whites and non-whites (Hispanics, African-American, and Asian-American).
Results
1167 patients met study criteria. Intracranial LAA was more prevalent in non-whites vs. whites (20.4% vs. 9.6%, P<0.001), while extracranial LAA had a more frequent point value in whites compared to non-whites (10.7% vs. 7.5%, P = 0.267). The presence of MetSD was more prevalent in both intracranial and extracranial LAA group than in non-LAA group: no significant differences were observed in the prevalence of MetSD between intra vs. extracranial LAA or whites vs. non-whites. However, with increasing numbers of abnormal metabolic components, whites were more likely to have experienced extracranial LAA, whereas non-whites were more likely to have experienced intracranial LAA. After adjusting for covariates, MetSD was associated with extracranial LAA in whites (OR, 1.98; 95% CI, 1.13-3.45), while there was a tendency that intracranial LAA was associated with MetSD in non-whites (OR, 1.80; 95% CI, 0.97-3.32). No association was found between MetSD and extracranial LAA in non-whites and between this syndrome with intracranial LAA in whites.
Conclusions
Our results showed that the impact of MetSD on the distribution of cervicocephalic atherosclerosis differed by race-ethnicity. This finding may in part explain the well-known differences in race-ethnic predilection to intracranial or extracranial atherosclerosis.
Keywords: Atherosclerosis, Risk factors, Metabolic syndrome, Race, Stroke, Ischemic
1. Introduction
The metabolic syndrome, a constellation of metabolic risk factors linked to insulin resistance, has been independently associated with a higher risk of symptomatic cardiovascular disease including ischemic stroke [1]. Although the guidelines in the Adult Treatment Panel III (ATP-III) [2] recognize this syndrome as a secondary target for vascular risk-reduction therapy, additional data are needed to characterize and understand the relationship between vascular disease particularly its most prevalent and burdensome manifestation, atherosclerosis, and the metabolic syndrome. For instance, while it is well known that the distribution of symptomatic cervicocephalic atherosclerosis varies by race-ethnicity, we are unaware of any prior reports detailing the impact of the metabolic syndrome on distribution of large artery atherosclerosis among diverse race-ethnic groups. Therefore, in this study we evaluated the influence of metabolic syndrome on the site of symptomatic large artery atherosclerosis (LAA) among subjects of diverse race and ethnicity.
2. Methods
2.1. Patients and workups
We retrospectively analyzed data in prospectively maintained registries of patients admitted with acute (<7 days after onset) ischemic stroke or transient ischemic attack (TIA) from September 2002 through April 2007 to UCLA medical center. We excluded patients who underwent an incomplete workup, had two or more causes of stroke identified, or for whom race-ethnicity information was not documented.
Data were collected according to stroke program protocols included demographic data, medical history, vascular risk factors, and stroke mechanism [3,4]. All patients underwent comprehensive workups including routine blood tests, neuroimaging, vascular imaging and cardiac studies [5]. All patients without contraindication underwent magnetic resonance imaging (MRI) and angiography (MRA) of the cervical and intracranial vessels; computed tomography and computed tomographic angiography was performed if MRI was contraindicated. Select patients additionally underwent digital subtraction angiography at attending physician discretion. All the patients underwent electrocardiography and cardiac telemetry for at least 24 h. Transthoracic echocardiography was obtained in all patients with a history, physical exam, or electrocardiographic evidence of coronary artery disease or left ventricular disease. Transesophageal echocardiography was performed in patients with infarcts unexplained after initial diagnostic evaluation. Arterial hemostatic markers of prothrombotic tendency, including antiphospholipid antibodies (antic-ardiolipidin antibody, diluted Russel Viper venom time, β2-glycoprotein 1 antibody) and homocysteine were measured in patients younger than 50 years old or with stroke cause undefined after initial workup. Venous hypercoagulable states were assessed in cryptogenic stroke patients with right to left shunts.
2.2. Conventional risk factors and metabolic syndrome
Socio-demographic, self-reported medical history, and vascular risk biomarker data were assessed and included: age, sex, race, ethnicity, histories of hypertension, diabetes, smoking, hypercholesterolemia, previous stroke or TIA, and coronary (CAD) and peripheral arterial disease (PAD) [3,4]. CAD was defined as history of coronary heart disease, physician diagnosed myocardial infarct or angina pectoris, or EKG evidence of an old myocardial infarct prior to index stroke or TIA. Smoking history was further divided into: non-smoker, ex smoker, and current smoker at the time of symptom onset.
To define the metabolic syndrome, we used the ATP-III guidelines [2] with some modification. Individuals were considered to have the metabolic syndrome if they met abnormality criteria for at least three of the following five factors at entry into the study: (1) obesity, (2) elevated fasting serum triglyceride levels (≥150 mg/dl), (3) low high-density lipoprotein (HDL) cholesterol levels (<40 mg/dl for men, <50 mg/dl for women), (4) hypertension (systolic blood pressure ≥130 mmHg or diastolic blood pressure ≥85 mmHg obtained at least 48 h after symptom onset), and (5) impaired fasting serum glucose levels (≥110 mg/dl). Instead of the waist circumference category, we used body mass index (BMI) ≥30 kg/m2, which is the index of obesity used in the WHO metabolic syndrome definition [6].
2.3. Patient grouping
Based on their clinical syndromes, diffusion-weighted magnetic resonance imaging (DWI) data, and the results of vascular, cardiologic, and laboratory studies, we divided the patients into two stroke etiologic groups: LAA and non-LAA group [5]. Patients of the LAA group were further subdivided according to their vascular status: (a) those with occlusive lesions (>50% stenosis or occlusion) of the extracranial portion of carotid or vertebral artery (the extracranial LAA group), and (b) those with occlusive lesions of the intracranial portion of internal carotid artery (ICA) or vertebral artery (VA), the proximal portion of the middle cerebral artery (MCA), or basilar artery (BA) (the intracranial LAA group). The degree of extracranial stenosis was measured per the methods of the North American Symptomatic Carotid Endarterectomy trial (NASCET) [7] and intracranial stenosis per the methods of the Warfarin-Aspirin Symptomatic Intracranial Disease (WASID) trial [8]. Patients with symptomatically relevant stenoses of MCA branch (M2) or proximal segment of anterior (A1) or posterior (P1) cerebral artery were classified as the intracranial LAA group when there were coexistence of stenoses on non-relevant large intracranial vessels (intracranial portion of ICA or vertebral artery, basilar artery, or M1), supporting intrinsic atherosclerosis rather than recanalized emboli. Isolated stenosis on the M2, A1, or P1 segment was classified as cryptogenic cause (cryptogenic embolism). The `other' group included cardioembolism, small arterial occlusion, other cause, and no determined cause [5].
2.4. Statistical analysis
Differences between groups were examined using the χ2 test, student t test, or one-way analysis of variance with post hoc tests for significance. We performed multiple logistic regression analysis to evaluate the independent association between intra- and extracranial LAA and metabolic syndrome with the non-LAA group as reference. All risk factors were entered into a stepwise logistic regression model, and potential confounding factors considered for inclusion in the model were (a) age, sex, race-ethnicity (Non-Hispanic White vs. African-American vs. Asian-American vs. Hispanic White), (b) history of hypertension, diabetes, dyslipidemia, smoking habits (non-smoker vs. ex smoker vs. current smoker), metabolic syndrome, previous stroke or TIA, CAD, and PAD, (c) laboratory findings including fasting serum levels of total cholesterol and low-density lipoprotein (LDL) cholesterol. To evaluate the contribution of each constituent variable of the metabolic syndrome to the prediction of intra- and extracranial LAA, independent associations of each metabolic component were also evaluated. Potential factors that were not significant (P<0.2) were sequentially deleted from the full model. Excluded variable were reintroduced at various stages of model development until only significant factors remained. Results are given as odds ratio (OR), as estimates of relative risk with 95% CI. Significance was established at the P<0.05 level.
3. Results
3.1. Characteristics of patients
Of 1221 patients, 1167 met full study entry criteria. Reasons for exclusion were: incomplete workup (n = 38), two or more causes of stroke (n = 8), and race-ethnicity not documented (n = 8). Among the analyzed 1167 patients, 152 (13.0%) patients were classified in the intracranial LAA group, 113 (9.7%) in the extracranial LAA, and 902 (77.3%) in the non-LAA group. The non-LAA group included 448 patients with cardioembolism, 238 patients with lacunar stroke, 114 patients with other determined etiology, and 102 patients with cryptogenic stroke. Male sex was 54% with the average age 67 years (range, 15 to 101); 794 patients were non-Hispanic white (NHW) and 373 were Hispanic or racially non-white (HRNW) (166 were Hispanic, 101 were African-American, and 106 were Asian).
Age and sex ratio did not differ between the groups (Table 1). History of hypertension, hypercholesterolemia, obesity (BMI≥30 kg/m2) were more frequently observed in the extracranial LAA group. In contrast, levels of fasting glucose, total cholesterol, and triglyceride were higher in the intracranial atherosclerosis group than other groups. The presence of metabolic syndrome and current smoker were more prevalent in both intra- and extracranial LAA groups than in the non-LAA group.
Table 1.
Demographics and risk factors in each stroke subtype
| Non-atherosclerosis |
Extracranial atherosclerosis |
Intracranial atherosclerosis |
P | |
|---|---|---|---|---|
| (n = 902) | (n = 113) | (n = 152) | ||
| Male sex | 484 (53.7) | 60 (53.1) | 86 (56.6) | .784 |
| Age | 67.0 ± 17.5 | 69.0 ± 13.1 | 65.3 ± 15.2 | .210 |
| Race/ethnicity (%) | <.001 | |||
| Non-Hispanic White (n = 794) | 633 (79.7) | 85 (10.7) | 76 (9.6) | |
| Hispanic (n = 166) | 127 (76.5) | 12 (7.2) | 27 (16.3) | |
| African-American (n = 101) | 68 (67.3) | 7 (6.9) | 26 (25.7) | |
| Asian-American (n = 106) | 74 (69.8) | 9 (8.5) | 23 (21.7) | |
| Risk factor history (%) | ||||
| Hypertension | 595 (66.3) | 95 (84.1) | 105 (69.1) | .001 |
| Diabetes | 221 (24.6) | 33 (29.2) | 51 (33.6) | .052 |
| Hypercholesterolemia | 297 (34.0) | 57 (50.9) | 70 (46.7) | <.001 |
| Metabolic syndrome | 265 (37.4) | 46 (52.3) | 65 (52.0) | .001 |
| Smoking habits | .009 | |||
| Ex smoker | 187 (20.8) | 33 (29.2) | 28 (18.5) | |
| Current smoker | 116 (12.9) | 22 (19.5) | 29 (19.2) | |
| Previous stroke or TIA | 257 (28.7) | 44 (38.9) | 51 (33.6) | .052 |
| Coronary heart disease | 210 (23.4) | 30 (26.5) | 26 (17.1) | .144 |
| Peripheral artery disease | 29 (3.4) | 9 (8.2) | 4 (2.7) | .055 |
| Metabolic status on admission | ||||
| BMI | 26.3 ± 6.2 | 28.0 ± 6.1 | 26.3 ± 4.8 | .100 |
| Systolic BP | 152.9 ± 29.7 | 151.5 ± 30.5 | 156.7 ± 32.1 | .302 |
| Diastolic BP | 81.0 ± 16.4 | 76.7 ± 15.2 | 81.7 ± 15.1 | .026 |
| Fasting glucose | 127.6 ± 54.3 | 142.4 ± 84.0 | 142.8 ± 73.1 | .002 |
| Triglyceride | 124.9 ± 81.3 | 149.8 ± 93.8 | 150.3 ± 116.8 | <.001 |
| HDL-cholesterol | 44.6 ± 15.2 | 42.2 ± 11.3 | 46.3 ± 17.0 | .118 |
| Total cholesterol | 172.6 ± 45.3 | 176.4 ± 47.3 | 182.0 ± 53.3 | .077 |
| LDL cholesterol | 103.8 ± 36.9 | 104.7 ± 39.5 | 111.6 ± 47.3 | .089 |
| Vascular territory | ||||
| Carotid | 600 (66.5) | 93 (82.3) | 91 (59.9) | |
| Vertebrobasilar | 246 (27.3) | 18 (15.9) | 57 (37.5) | |
| Both | 18 (2.0) | - | - | |
| Unknown | 38 (4.2) | 2 (1.8) | 4 (2.6) | |
| NIH stroke scale score | 7.8 ± 8.1 | 7.2 ± 8.1 | 7.0 ± 7.4 | .558 |
| Medications prior to admission | ||||
| Thiazide diuretics | 106 (13.5) | 23 (20.9) | 20 (13.9) | .114 |
| Beta-blocker | 205 (26.1) | 29 (26.4) | 31 (21.5) | .502 |
| Calcium channel blocker | 50 (6.4) | 13 (11.8) | 14 (9.7) | .064 |
| ACEI or ARB | 236 (30.1) | 42 (38.2) | 49 (34.0) | .678 |
| Statin | 244 (28.3) | 50 (44.2) | 47 (30.9) | .002 |
| Antithrombotic agents | .348 | |||
| Antiplatelet | 331 (38.1) | 49 (43.4) | 51 (33.6) | |
| Anticoagulant | 87 (10.0) | 8 (7.1) | 9 (5.9) |
Numbers in parenthesis are percentages.
The distribution of the race-ethnic group (NHW vs. HRNW) among the extracranial LAA group was similar to that of the non-LAA group, while intracranial LAA was more frequent in HRNW than in NHW (Table 1). Intracranial LAA was more prevalent than extracranial LAA in African-American, Asian-American, and Hispanics (Table 1 and Fig. 1).
Fig. 1.
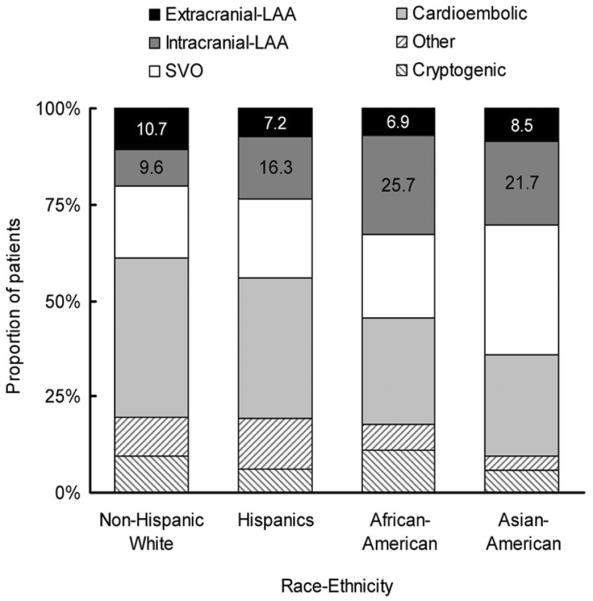
Stroke subtype by race-ethnic group.
3.2. Race-ethnic specific impact of metabolic syndrome on the distribution of atherosclerosis
Racial-ethnic differences in risk factors and metabolic status were noted. Atrial fibrillation, CAD and PAD were more frequently observed in NHWs, whereas history of diabetes and fasting hyperglycemia were more prevalent in HRNWs (Supplementary Table 1). No significant difference was found in the prevalence of metabolic syndrome between NHWs (38.7%) and HRNWs (45.1%; African-American-38.0%, Asian-American-45.7%, and Hispanics-48.6%). However, as shown in Fig. 2, the metabolic status on admission and the impact of metabolic syndrome on the distribution of atherosclerosis differed depending on race-ethnicity. With increasing numbers of abnormal metabolic components, NHW patients were more likely to have experienced an extracranial atherosclerotic stroke, whereas HRNW patients were more likely to have experienced an intracranial atherosclerotic stroke (Fig. 2).
Fig. 2.
Difference in the impact of number of metabolic abnormalities on the distribution of atherosclerosis by race-ethnicity.
After adjustment of age, sex, risk factors, and total and LDL cholesterol, metabolic syndrome was independently associated with LAA. Independent factors associated with intracranial LAA were the presence of metabolic syndrome, hypercholesterolemia and HRNW status; compared to NHW, African-American, Asian-American, and Hispanics were 2.7, 2.5, and 1.6 times more likely to have intracranial LAA, respectively (Table 2). Hypertension and presence of metabolic syndrome were independently associated with extracranial atherosclerotic stroke (Table 3).
Table 2.
Logistic regression analysis: intracranial LAA vs. non-LAA stroke
| Results of 773 NHW and 290 HRNW patients |
Multivariate OR (95% CI) analyzed separately in NHW and HRNW |
||||
|---|---|---|---|---|---|
| Crude | Multivariate OR (95% CI) | P | NHW | HRNW | |
| Age | 1.01 | 1.00 | 1.01 | ||
| Male sex | 1.21 | 1.54 | 0.94 | ||
| Hypertension | 1.11 | 1.54 | 0.73 | ||
| Diabetes | 1.25 | 0.89 | 2.00 (1.06-3.74) | ||
| Hyperlipidemia | 1.51 | 1.61 (1.07-2.41) | .022 | 1.44 | 1.58 |
| Smoking | |||||
| Ex smoking | 0.88 | 0.77 | 1.22 (0.57-2.60) | ||
| Current smoking | 1.28 | 0.75 | 2.65 (1.19-5.89) | ||
| Total cholesterol, mg/dl | 1.00 | 0.99 | 1.00 | ||
| LDL cholesterol, mg/dl | 1.01 | 1.01 | 1.00 | ||
| Previous stroke or TIA | 1.36 | 2.10 (1.21-3.67)* | 0.91 | ||
| History of CAD | 0.76 | 0.70 | 0.89 | ||
| History of PAD | 0.67 | 0.63 | 1.27 | ||
| Metabolic syndrome | 1.44 | 1.50 (1.01-2.24) | .047 | 1.14 | 1.80 (0.97-3.32)* |
| Ethnicity | N/A | N/A | |||
| Non-Hispanic | Reference | Reference | |||
| White | |||||
| African- | 2.69 | 2.80 (1.53-5.14) | .001 | ||
| American | |||||
| Asian- | 2.52 | 2.74 (1.52-4.92) | .001 | ||
| American | |||||
| Hispanics | 1.59 | 1.68 (0.99-2.84) | .054 | ||
NHW: non-Hispanic white patients, HRNW: Hispanic and racially non-White patients.
N/A: not assessed.
Significant independent predictors in multivariate test.
Table 3.
Logistic regression analysis: extracranial LAA vs. non-LAA stroke
| Results of 773 NHW and 290 HRNW patients |
Multivariate OR (95% CI) analyzed separately in NHW and HRNW |
||||
|---|---|---|---|---|---|
| Crude | Multivariate OR (95% CI) | P | NHW | HRNW | |
| Age | 1.00 | 1.00 | 1.02 | ||
| Male sex | 0.97 | 0.99 | 0.69 | ||
| Hypertension | 2.57 | 2.57 (1.34-4.94) | .005 | 2.18 (1.05-4.52)* | 4.04 (0.91-17.97)* |
| Diabetes | 0.77 | 0.90 | 0.54 | ||
| Hyperlipidemia | 1.59 | 1.93 (1.09-3.40)* | 1.00 | ||
| Smoking | |||||
| Ex smoking | 1.41 | 1.08 | 2.58 (0.95-7.05)* | ||
| Current smoking | 1.60 | 1.10 | 2.91 (0.90-9.37)* | ||
| Total cholesterol, mg/dl | 1.00 | 1.00 | 1.00 | ||
| LDL cholesterol, mg/dl | 1.00 | 1.01 | 1.00 | ||
| Previous stroke or TIA | 1.21 | 1.39 | 0.75 | ||
| History of CAD | 0.82 | 0.94 | 0.39 | ||
| History of PAD | 1.90 | 1.68 | 3.14 | ||
| Metabolic syndrome | 1.65 | 1.51 (0.95-2.42) | .084 | 1.98 (1.13-3.45)* | 1.10 |
| Ethnicity | N/A | N/A | |||
| Non-Hispanic | Reference | ||||
| White | |||||
| African- | 0.92 | ||||
| American | |||||
| Asian- | 0.76 | ||||
| American | |||||
| Hispanics | 0.85 | ||||
NHW: non-Hispanic white patients, HRNW: Hispanic and racially non-White patients.
N/A: not assessed.
Significant independent predictors in multivariate test.
Factors independently associated with intra- and extracranial LAA were analyzed separately in NHW and HRNW subjects. There was a trend that metabolic syndrome was associated with intracranial LAA in HRNWs, although statistically insignificant probably due to small sample size (P = 0.062) (Table 2). On the contrary, the presence of metabolic syndrome was independently associated with extracranial LAA in NHWs (Table 3). No association was found between metabolic syndrome and extracranial LAA in HRNWs and between this syndrome with intracranial LAA in NHWs. For NHW patients, previous stroke or TIA was also associated with intracranial LAA, and hypertension and dyslipidemia were associated with extracranial LAA. For HRNW patients, smoking status was independently associated with both intra- and extracranial LAA. In addition, diabetes was associated with intracranial LAA, whereas hypertension was associated with extracranial LAA in HRNW patients.
4. Discussion
African-Americans, Hispanics and Asians have been found to have a higher frequency of intracranial rather than extracranial atherosclerosis, while non-Hispanic whites tend to have more extracranial vs. intracranial atherosclerosis [9-11]. Factors affecting the severity and distribution of atherosclerosis are variably reported among studies; the severity and distribution of atherosclerosis is reported to be influenced by race [12], vascular risk factors [13,14], or both [14].
Our present findings raise the possibility that vascular response to the same risk factor may differ depending on the race-ethnicity. Metabolic syndrome has been reported to be associated with atherosclerotic stroke across in individual race-ethnicity categories [15-17]. Our study with multiple race-ethnic groups showed the prevalence of metabolic syndrome to be similar between intra- and extracranial atherosclerotic stroke; it was higher in both intra- and extracranial LAA groups than in the non-LAA group. However, our data showed that the presence of the metabolic syndrome may differentially afflict large vessels in the cervicocephalic arterial tree (extracranial and intracranial) by race-ethnicity. We noted that the increasing abnormal components of the metabolic syndrome among non-Hispanic white patients was associated with a greater likelihood of extracranial LAA, but such association was not observed in Hispanics and non-whites. On the contrary, there was tendency that rising metabolic abnormalities in Hispanics and non-whites was associated with intracranial LAA. Metabolic syndrome has been reported to be an independent factor for extracranial carotid atherosclerosis, in populations of predominantly white patients [18,19], but not in Japanese [20] and Koreans [21,22]. On the contrary, it was reported that the metabolic syndrome was associated with intracranial atherosclerotic stroke in Asian patients [21,22].
Why might the metabolic syndrome act differently based on the ethnicity? A potential explanation could be the marked differences in the types and degree of abnormal components of the metabolic syndrome across racial and ethnic groups [23]. Compared to non-Hispanic whites, higher rates of obesity and hyperglycemia as well as lower rates of physical activity in Hispanics have been reported [23,24]. On the contrary, African-Americans have lesser vasodilator capacity in the microcirculation as well as higher prevalence of hypertension and elevated HDL levels, whereas lower LDL levels but higher prevalence of hypertension and smoking in Asians have been reported [23,24]. Most importantly, race-ethnic groups have shown differences in levels of visceral adiposity: for example, there is a strong tendency for insulin resistance even in apparently lean Asian Indians suggesting a primary metabolic defect [23,25]. The recent International Diabetes Federation definition of the metabolic syndrome acknowledges race-ethnic differences in fat distribution [26]. In addition, race-ethnic differences (White vs. Indo-Asians [27] and white vs. black [28]) in the secretion of adiponectin, a protein secreted by adipose tissue, have been reported. Findings from the Bogalusa Heart Study specifically suggested that race-specific physiological and metabolic characteristics may contribute to the observed white vs. non-white divergences [28-30].
Recently, race and gender-based differences in the mechanisms of metabolic syndrome-induced carotid atherosclerotic progression were reported [18,29]. In the Los Angeles Atherosclerosis Study, atherogenic effects of metabolic syndrome were mediated differently in the sexes, through central obesity in men and triglycerides in women [18]. In the Bogalusa Heart study, the predictability of childhood cardiovascular risk factors for increased intimal media thickness in adulthood varied by race and gender [29]. Thus, it is also conceivable that the race-specific impact of metabolic syndrome on the distribution of atherosclerosis may be caused by racial-specific differences in (a) the prevalence of metabolic components and (b) in the host-response to the presence of specific metabolic component (i. e. obesity, plasma lipid). Our present data suggested that single components of the metabolic syndrome may modify the site of symptomatic large vessel atherosclerosis as evidenced by our finding that increasing degree of obesity strongly predicted the occurrence of extracranial LAA in non-Hispanic white patients.
Strengths of our study included the consecutive, prospective recruitment of a well phenotyped group of ischemic stroke patients. All subjects underwent comprehensive workups, including vascular, laboratory and cardiologic studies. However, the results of this study should be interpreted with the caution because this is a hospital-based cohort and a relatively small number of non-white groups were included. While the combination of Hispanics, African-Americans, and Asian-Americans together helps the power of the study, it may obscure some differences among these groups. Larger, multicenter studies analyzing each race-ethnic group separately are needed. In addition, the race-ethnic groups `Hispanic' and `Asian' are rather heterogeneous. Second, general obesity (increased BMI) instead of abdominal obesity was used for diagnosing metabolic syndrome. The impact of general obesity on the distribution of atherosclerosis might differ from that of abdominal obesity. Further studies concerning race-ethnic differences on fat distribution and measuring of serologic markers such as s-adiponectin are needed. Lastly, the control population was not included in this study. Thus, our results in a cohort that includes only stroke patients can not be generalized to a population without stroke, and our analysis could be underestimating the true effects of these factors on the risk of LAA stroke subtypes.
In conclusion, our study suggests that metabolic abnormalities may differentially affect the cerebrovascular arterial beds by race-ethnicity, which may partly explain the previously known difference in frequency of intracranial and extracranial atherosclerosis among race-ethnic groups. Given the impact of metabolic syndrome on both types of atherosclerotic stroke, identifying and treating the metabolic syndrome should remain an important management goal in symptomatic atherosclerotic stroke patients.
Supplementary Material
Acknowledgements
This study was supported by a grant from NIH/National Institute of Neurological Disorders and Stroke 1 K23NS054084-01A1 and Korean Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A080044).
Footnotes
Appendix A. Supplementary data Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.jns.2009.03.033.
References
- [1].Girman CJ, Rhodes T, Mercuri M, Pyorala K, Kjekshus J, Pedersen TR, et al. The metabolic syndrome and risk of major coronary events in the Scandinavian Simvastatin Survival Study (4 s) and the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TEXCAPS) Am J Cardiol. 2004;93:136–41. doi: 10.1016/j.amjcard.2003.09.028. [DOI] [PubMed] [Google Scholar]
- [2].Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) JAMA. 2001;285:2486–97. doi: 10.1001/jama.285.19.2486. [DOI] [PubMed] [Google Scholar]
- [3].Ovbiagele B, Saver JL, Fredieu A, Suzuki S, McNair N, Dandekar A, et al. Protect: A coordinated stroke treatment program to prevent recurrent thromboembolic events. Neurology. 2004;63:1217–22. doi: 10.1212/01.wnl.0000140493.83607.f1. [DOI] [PubMed] [Google Scholar]
- [4].Smaha LA. The American Heart Association Get with the Guidelines Program. Am Heart J. 2004;148:S46–48. doi: 10.1016/j.ahj.2004.09.015. [DOI] [PubMed] [Google Scholar]
- [5].Lee LJ, Kidwell CS, Alger J, Starkman S, Saver JL. Impact on stroke subtype diagnosis of early diffusion-weighted magnetic resonance imaging and magnetic resonance angiography. Stroke. 2000;31:1081–9. doi: 10.1161/01.str.31.5.1081. [DOI] [PubMed] [Google Scholar]
- [6].Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a who consultation. Diabet Med. 1998;15:539–53. doi: 10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- [7].Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. North American Symptomatic Carotid Endarterectomy Trial collaborators. N Engl J Med. 1991;325:445–53. doi: 10.1056/NEJM199108153250701. [DOI] [PubMed] [Google Scholar]
- [8].Samuels OB, Joseph GJ, Lynn MJ, Smith HA, Chimowitz MI. A standardized method for measuring intracranial arterial stenosis. AJNR Am J Neuroradiol. 2000;21:643–6. [PMC free article] [PubMed] [Google Scholar]
- [9].Sacco RL, Roberts JK, Boden-Albala B, Gu Q, Lin IF, Kargman DE, et al. Race-ethnicity and determinants of carotid atherosclerosis in a multiethnic population. The Northern Manhattan Stroke Study. Stroke. 1997;28:929–35. doi: 10.1161/01.str.28.5.929. [DOI] [PubMed] [Google Scholar]
- [10].Kittner SJ, White LR, Losonczy KG, Wolf PA, Hebel JR. Black-white differences in stroke incidence in a national sample. The contribution of hypertension and diabetes mellitus. JAMA. 1990;264:1267–70. [PubMed] [Google Scholar]
- [11].Mast H, Thompson JL, Lin IF, Hofmeister C, Hartmann A, Marx P, et al. Cigarette smoking as a determinant of high-grade carotid artery stenosis in Hispanic, Black, and White patients with stroke or transient ischemic attack. Stroke. 1998;29:908–12. doi: 10.1161/01.str.29.5.908. [DOI] [PubMed] [Google Scholar]
- [12].Wityk RJ, Lehman D, Klag M, Coresh J, Ahn H, Litt B. Race and sex differences in the distribution of cerebral atherosclerosis. Stroke. 1996;27:1974–80. doi: 10.1161/01.str.27.11.1974. [DOI] [PubMed] [Google Scholar]
- [13].Yasaka M, Yamaguchi T, Shichiri M. Distribution of atherosclerosis and risk factors in atherothrombotic occlusion. Stroke. 1993;24:206–11. doi: 10.1161/01.str.24.2.206. [DOI] [PubMed] [Google Scholar]
- [14].Sacco RL, Kargman DE, Gu Q, Zamanillo MC. Race-ethnicity and determinants of intracranial atherosclerotic cerebral infarction. The Northern Manhattan Stroke Study. Stroke. 1995;26:14–20. doi: 10.1161/01.str.26.1.14. [DOI] [PubMed] [Google Scholar]
- [15].Ninomiya JK, L'Italien G, Criqui MH, Whyte JL, Gamst A, Chen RS. Association of the metabolic syndrome with history of myocardial infarction and stroke in the Third National Health and Nutrition Examination Survey. Circulation. 2004;109:42–6. doi: 10.1161/01.CIR.0000108926.04022.0C. [DOI] [PubMed] [Google Scholar]
- [16].Ohira T, Shahar E, Chambless LE, Rosamond WD, Mosley TH, Jr, Folsom AR. Risk factors for ischemic stroke subtypes: The Atherosclerosis Risk in Communities Study. Stroke. 2006;37:2493–8. doi: 10.1161/01.STR.0000239694.19359.88. [DOI] [PubMed] [Google Scholar]
- [17].Chen HJ, Bai CH, Yeh WT, Chiu HC, Pan WH. Influence of metabolic syndrome and general obesity on the risk of ischemic stroke. Stroke. 2006;37:1060–4. doi: 10.1161/01.STR.0000206458.58142.f3. [DOI] [PubMed] [Google Scholar]
- [18].Fan AZ. Metabolic syndrome and progression of atherosclerosis among middle-aged us adults. J Atheroscler Thromb. 2006;13:46–54. doi: 10.5551/jat.13.46. [DOI] [PubMed] [Google Scholar]
- [19].Montalcini T, Gorgone G, Gazzaruso C, Sesti G, Perticone F, Pujia A. Carotid atherosclerosis associated to metabolic syndrome but not BMI in healthy menopausal women. Diabetes Res Clin Pract. 2007;76:378–82. doi: 10.1016/j.diabres.2006.09.014. [DOI] [PubMed] [Google Scholar]
- [20].Ishizaka N, Ishizaka Y, Hashimoto H, Toda E, Nagai R, Yamakado M. Metabolic syndrome may not associate with carotid plaque in subjects with optimal, normal, or high-normal blood pressure. Hypertension. 2006;48:411–7. doi: 10.1161/01.HYP.0000233466.24345.2e. [DOI] [PubMed] [Google Scholar]
- [21].Bang OY, Kim JW, Lee JH, Lee MA, Lee PH, Joo IS, et al. Association of the metabolic syndrome with intracranial atherosclerotic stroke. Neurology. 2005;65:296–8. doi: 10.1212/01.wnl.0000168862.09764.9f. [DOI] [PubMed] [Google Scholar]
- [22].Park JH, Kwon HM, Roh JK. Metabolic syndrome is more associated with intracranial atherosclerosis than extracranial atherosclerosis. Eur J Neurol. 2007;14:379–86. doi: 10.1111/j.1468-1331.2007.01682.x. [DOI] [PubMed] [Google Scholar]
- [23].Forouhi NG, Sattar N. CVD risk factors and ethnicity—a homogeneous relationship? Atheroscler Suppl. 2006;7:11–9. doi: 10.1016/j.atherosclerosissup.2006.01.003. [DOI] [PubMed] [Google Scholar]
- [24].McGruder HF, Malarcher AM, Antoine TL, Greenlund KJ, Croft JB. Racial and ethnic disparities in cardiovascular risk factors among stroke survivors: United States 1999 to 2001. Stroke. 2004;35:1557–61. doi: 10.1161/01.STR.0000130427.84114.50. [DOI] [PubMed] [Google Scholar]
- [25].Chandalia M, Abate N, Garg A, Stray-Gundersen J, Grundy SM. Relationship between generalized and upper body obesity to insulin resistance in Asian Indian men. J Clin Endocrinol Metab. 1999;84:2329–35. doi: 10.1210/jcem.84.7.5817. [DOI] [PubMed] [Google Scholar]
- [26].Ford ES. Prevalence of the metabolic syndrome defined by the International Diabetes Federation among adults in the U.S. Diabetes Care. 2005;28:2745–9. doi: 10.2337/diacare.28.11.2745. [DOI] [PubMed] [Google Scholar]
- [27].Valsamakis G, Chetty R, McTernan PG, Al-Daghri NM, Barnett AH, Kumar S. Fasting serum adiponectin concentration is reduced in Indo-Asian subjects and is related to HDL cholesterol. Diabetes Obes Metab. 2003;5:131–5. doi: 10.1046/j.1463-1326.2003.00254.x. [DOI] [PubMed] [Google Scholar]
- [28].Patel DA, Srinivasan SR, Xu JH, Chen W, Berenson GS. Adiponectin and its correlates of cardiovascular risk in young adults: The Bogalusa Heart Study. Metabolism. 2006;55:1551–7. doi: 10.1016/j.metabol.2006.06.028. [DOI] [PubMed] [Google Scholar]
- [29].Li S, Chen W, Srinivasan SR, Tang R, Bond MG, Berenson GS. Race (black-white) and gender divergences in the relationship of childhood cardiovascular risk factors to carotid artery intima-media thickness in adulthood: The Bogalusa Heart Study. Atherosclerosis. 2006 doi: 10.1016/j.atherosclerosis.2006.08.026. [DOI] [PubMed] [Google Scholar]
- [30].Patel DA, Srinivasan SR, Xu JH, Li S, Chen W, Berenson GS. Distribution and metabolic syndrome correlates of plasma C-reactive protein in biracial (black- white) younger adults: The Bogalusa Heart Study. Metabolism. 2006;55:699–705. doi: 10.1016/j.metabol.2005.07.015. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



