Abstract
Procyclic forms of Trypanosoma brucei isolated from the midguts of infected tsetse flies, or freshly transformed from a strain that is close to field isolates, do not use a complete Krebs cycle. Furthermore, short stumpy bloodstream forms produce acetate and are apparently metabolically preadapted to adequate functioning in the tsetse fly.
African trypanosomatids comprise various pleomorphic trypanosome species that proliferate in the bloodstream of their mammalian hosts as long slender bloodstream form (BSF) trypanosomes, and at the peak of parasitemia they differentiate into nondividing short stumpy form trypanosomes (1). After being ingested during a bloodmeal by a tsetse fly (Glossina sp.), short stumpy form trypanosomes differentiate into procyclic form (PCF) trypanosomes, which actively multiply and colonize the midgut of the fly. Subsequently, PCF Trypanosoma brucei migrates to the salivary glands while undergoing a complex differentiation (22). Here, attached epimastigote forms start multiplying, after which nondividing metacyclic trypomastigotes develop. The life cycle of T. brucei is completed when these metacyclic trypomastigotes are injected into a mammal through the bite of an infected fly, after which they transform into long slender BSF trypanosomes. During this life cycle, trypanosomes encounter different environments to which they have adapted, resulting in distinct stages, characterized by morphological as well as metabolic changes. Long slender BSF trypanosomes degrade glucose by glycolysis and excrete pyruvate as the sole metabolic end product (12, 13, 23). On the other hand, PCF trypanosomes do not excrete pyruvate but degrade glucose to acetate and succinate as main end products (25). Krebs cycle activity was thought previously to be present in trypanosomatids, at least in insect stages of some African trypanosomatids (3, 9, 10, 12, 21). However, this presumed flux through the Krebs cycle is supported only poorly by direct experimental evidence and was based mainly on the presence of certain enzyme activities. Although genes for all enzymes of this cycle are indeed present in the genome and expressed in the insect stages, recent studies revealed that at least in T. brucei, the cycle is not used for the complete oxidation of acetyl-coenzyme A (CoA) to carbon dioxide (2, 26). Instead, parts of the cycle are most likely used in anabolic pathways, such as gluconeogenesis and fatty acid formation, and also for the final steps in the degradation of amino acids (26). It is possible that the reported discrepancies on the presence or absence of full-circle Krebs cycle activity are caused by differences in the number of passages through mice after the isolation of the strain from the field. Such passages may have been ongoing for many years, during which the parasites were continuously propagated as BSF trypanosomes. Furthermore, most insect form trypanosomes that were investigated up to now have been propagated for many years as PCF trypanosomes in rich culture media. Hence, the reported discrepancies could be due to differences between freshly differentiated PCF trypanosomes and those well adapted to in vitro culture, and the absence of an active Krebs cycle in PCF trypanosomes could be the result of an adaptation caused by the prolonged in vitro culturing. To investigate these possibilities, we analyzed the glucose metabolism of PCF T. brucei directly after isolation from the midguts of tsetse flies. We also studied freshly differentiated PCF trypanosomes from the AntAR 1 strain, a T. brucei strain that has had a minor history of animal passaging since its field isolation (15, 17).
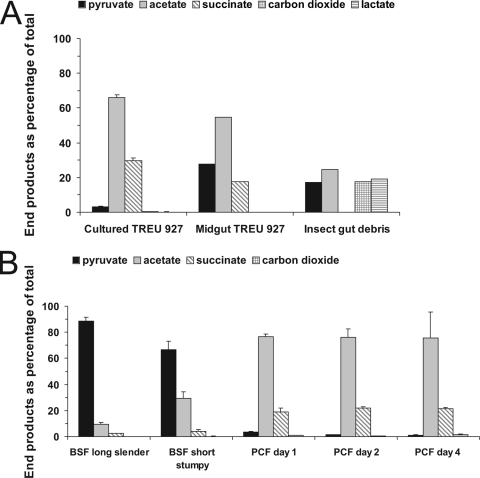
To investigate the cause of the conflicting reports on Krebs cycle activity in PCF trypanosomes, we first analyzed the effect of environmental factors by comparing the carbohydrate metabolism of PCF trypanosomes well adapted to in vitro culturing and PCF trypanosomes isolated from their natural environment, the midguts of tsetse flies. These experiments were performed with PCF TREU 927 T. brucei, a pleomorphic strain that has been thoroughly characterized and is still able to infect Glossina morsitans, performing a complete physiological life cycle (2). For the infection of tsetse flies, male G. morsitans flies originating from the colony maintained at the Institute of Tropical Medicine in Antwerp, Belgium, were infected with procyclic TREU 927 T. brucei by in vitro membrane feeding and subsequently maintained for 10 days by feeding on rabbit blood (15). Then, flies were dissected on a sterile glass slide and the infected midguts were isolated and incubated for at least 30 min at 28°C in SDM-79 medium that was gently rotated. After sedimentation of the midguts by gravity, insect gut debris was removed by centrifugation at 300 × g for 5 min. PCF trypanosomes were then isolated from the collected supernatant by centrifugation at 1,500 × g for 10 min. Since PCF trypanosomes could not be isolated from the midgut without minor amounts of contaminating insect gut material, such as gut cells and debris, we also investigated the glucose metabolism of this fraction. Analysis of metabolic end products produced from [6-14C]glucose in this control incubation of insect gut debris, which also contained minor amounts of trypanosome cells, showed the formation of 14C-labeled pyruvate, CO2, acetate, and lactate (Fig. 1A). Minor amounts of lactate were also produced in the incubations with PCF trypanosomes isolated from the midgut, which also contained minor amounts of insect gut debris. Since lactate is not an excreted end product of T. brucei, this labeled lactate is indicative for the glucose degradation activity of insect gut debris. Therefore, end product formation in the incubations with PCF trypanosomes isolated from the midgut was corrected for end products produced by the contaminating insect gut debris by subtracting all produced lactate and the calculated accompanying amounts of other end products produced in the insect gut debris incubation. The metabolic incubations with PCF trypanosomes directly after isolation from the tsetse midgut showed that these trypanosomes degrade glucose to the same metabolic end products, acetate, succinate, and pyruvate, as the in vitro culture-adapted PCF trypanosomes (Fig. 1A). Furthermore, the ratio of acetate and succinate produced by PCF trypanosomes isolated from the midgut were similar to that of in vitro-cultured PCF trypanosomes (Fig. 1A). On the other hand, a major difference was observed in the amount of glucose consumed since the PCF trypanosomes isolated from the midguts of tsetse flies consumed 16-fold less glucose than PCF trypanosomes that were derived from in vitro cultures. This difference in glucose consumption can probably be explained by our observation that both motility and especially growth of PCF trypanosomes isolated from the midgut were significantly reduced compared to the in vitro culture-derived PCF trypanosomes. Apparently, the environmental conditions in the midgut of the fly did affect the PCF trypanosomes, but they did not significantly alter the metabolic pathways used for energy metabolism. However, PCF trypanosomes isolated from the midgut of the fly excreted more pyruvate (Fig. 1A), which suggests that pyruvate is a more important metabolic end product for PCF trypanosomes under physiological conditions than acknowledged thus far. Most importantly, however, just like continuously in vitro-cultured ones, PCF trypanosomes isolated from the midgut of the fly did not degrade [6-14C]glucose to labeled CO2 (Fig. 1A), which demonstrates the absence of a functional Krebs cycle in these tsetse fly-derived PCF trypanosomes.
FIG. 1.
Radioactive end products of [6-14C]glucose metabolism of procyclic TREU 927 T. brucei cells grown in vitro or isolated from the midguts of tsetse flies (A) and that of AntAR 1 T. brucei during differentiation of BSF to PCF trypanosomes (B). (A) The results of a single experiment for PCF trypanosomes isolated from the midgut and for insect gut debris and the mean + the standard deviation (SD) of three parallel incubations for in vitro-cultured PCF trypanosomes are shown. Total end product formation from [6-14C]glucose was 2.08 ± 0.19 μmol/h per 108 cells and 0.23 μmol/h per 108 cells for in vitro-cultured and midgut-isolated PCF trypanosomes, respectively, and was calculated using the number of trypanosome cells present at the beginning of the incubation. End product formation in the incubation with PCF trypanosomes isolated from the midgut was corrected for end products produced by contaminating insect gut debris (see text for details). (B) Metabolic incubations using 6-14C-labeled glucose were performed during differentiation from short stumpy BSF trypanosomes to insect stage PCF trypanosomes. Incubations with PCF trypanosomes were started at 24, 48, and 96 h after induction of differentiation (PCF trypanosomes on day 1, PCF trypanosomes on day 2, and PCF trypanosomes on day 4, respectively); means + SDs of three parallel incubations are shown (for the short stumpy form, six incubations in two independent experiments). Total glucose consumption in incubations with long slender BSF trypanosomes, short stumpy BSF trypanosomes, PCF trypanosomes on day 1, PCF trypanosomes on day 2, and PCF trypanosomes on day 4 was 4.8, 3.4, 1.5, 1.1, and 0.79 μmol/h per 108 cells, respectively. Excreted labeled end products shown in panels A and B were analyzed as described previously (25) and are expressed as the percentage of the total amount of radioactive end products produced (in the incubation of gut debris, one other unidentified end product was produced, which explains why this total in the figure does not add up to 100%). The decrease in pyruvate production between long slender and short stumpy BSF trypanosomes as well as the increase in acetate production is significant as calculated using an unpaired t test (P < 0.01 for pyruvate and P < 0.001 for acetate).
Although TREU 927 T. brucei is a pleomorphic trypanosome strain, it cannot be excluded that these trypanosomes have adapted their energy metabolism during the substantial period that this strain has been cultured in vitro. Therefore, we also studied the carbohydrate metabolism of freshly transformed PCF of the T. brucei AntAR 1 strain, a well-characterized pleomorphic strain that is close to the wild isolate (17). To investigate the energy metabolism of these freshly differentiated PCF trypanosomes, AntAR 1 BSF trypanosomes were harvested from the blood of infected immune-suppressed NMRI mice as described previously (16) and either directly incubated with [6-14C]glucose or differentiated to PCF trypanosomes, by addition of 6 mM cis-aconitate and incubation at 27°C (7). These trypanosomes were then incubated with [6-14C]glucose at different time points after the initiation of differentiation. Our experiments (Fig. 1B) confirmed that differentiation of trypanosomes from BSF to PCF is accompanied by a metabolic shift in excreted end products from pyruvate to acetate and succinate (3, 14, 25). This metabolic shift during differentiation of BSF to PCF trypanosomes was complete after 1 to 2 days (Fig. 1B), which is in agreement with previous observations (9). A subsequent switch in medium from HMI-9, a medium used to culture BSF T. brucei, to SDM-79, a medium used for the culture of PCF T. brucei, did not result in further changes in excreted end products (data not shown).
Our experiments, however, did not show any significant production of labeled CO2 and certainly not the massive increase in CO2 formation upon differentiation of BSF into PCF trypanosomes that was reported in a comparable study by Durieux et al. (9). We cannot exclude that this difference in Krebs cycle activity between our study and that of Durieux et al. is caused by a strain difference, but since the AntAR 1 strain we used can be considered to be close to the field isolate, the results presented here are indicative of wild-type T. brucei metabolism and strongly suggest that a functional Krebs cycle is absent in PCF T. brucei cells in vivo.
Next to the absence of carbon dioxide formation via Krebs cycle activity during differentiation of BSF to PCF trypanosomes, our metabolic experiments also demonstrated that acetate accounted for 30% of the glucose-derived excreted labeled end products in freshly isolated BSF AntAR 1 T. brucei cells (Fig. 1B). This is a surprising observation since BSF trypanosomes are reported to rely on glycolysis only and to excrete pyruvate and minor amounts of glycerol (12, 13, 23). However, the BSF trypanosomes that we tested in our incubations were predominantly short stumpy BSF cells, whereas nearly all previously performed metabolic studies of BSF trypanosomes were performed with long slender BSF cells. In order to investigate whether differentiation from long slender to short stumpy form trypanosomes indeed shifts the metabolism toward acetate formation, we analyzed the energy metabolism of BSF trypanosomes harvested from mice at two different time points after infection. At day 4 after infection, predominantly long slender BSF trypanosomes were isolated (94% long slender versus 6% short stumpy), whereas at day 7 after infection, predominantly short stumpy BSF trypanosomes were isolated (92% short stumpy versus 8% long slender). Analysis of glucose-derived metabolic end products from incubations with BSF AntAR 1 trypanosomes isolated at day 4 or at day 7 after infection showed that short stumpy BSF trypanosomes indeed produce significant amounts of acetate as an end product of glucose metabolism (Fig. 1B). In the incubations with predominantly long slender BSF AntAR 1 T. brucei cells, some acetate was also produced, but this relatively small amount of acetate formation can be explained by the presence of a certain amount of short stumpy cells. Although the incubations were started with nearly 95% long slender BSF cells, BSF cells from the AntAR 1 strain are highly pleomorphic and rapidly differentiate to short stumpy forms during in vitro culture conditions. Therefore, increasing amounts of short stumpy form T. brucei were formed during our incubations (up to 40 to 50% at the end of incubation), which accounts for the amount of acetate formed during these incubations.
Since acetate production in Trypanosomatidae is catalyzed by the mitochondrial enzyme acetate:succinate CoA transferase (ASCT), which was previously shown not to be expressed in in vitro-cultured BSF T. brucei (20), we examined the ASCT enzyme activity in lysates derived from either over 92% short stumpy cells or 94% long slender cells. These experiments showed that the ASCT enzyme is present in short stumpy BSF trypanosomes in an amount equivalent to around 15% of that of PCF trypanosomes (Fig. 2). This is in agreement with the observation that acetate is a more prominent excreted end product in PCF trypanosomes than in short stumpy BSF cells. On the other hand, ASCT activity was nearly absent in long slender BSF trypanosomes (Fig. 2), which confirms the conclusion that in our incubations acetate is not produced by long slender BSF trypanosomes but by short stumpy BSF trypanosomes.
FIG. 2.
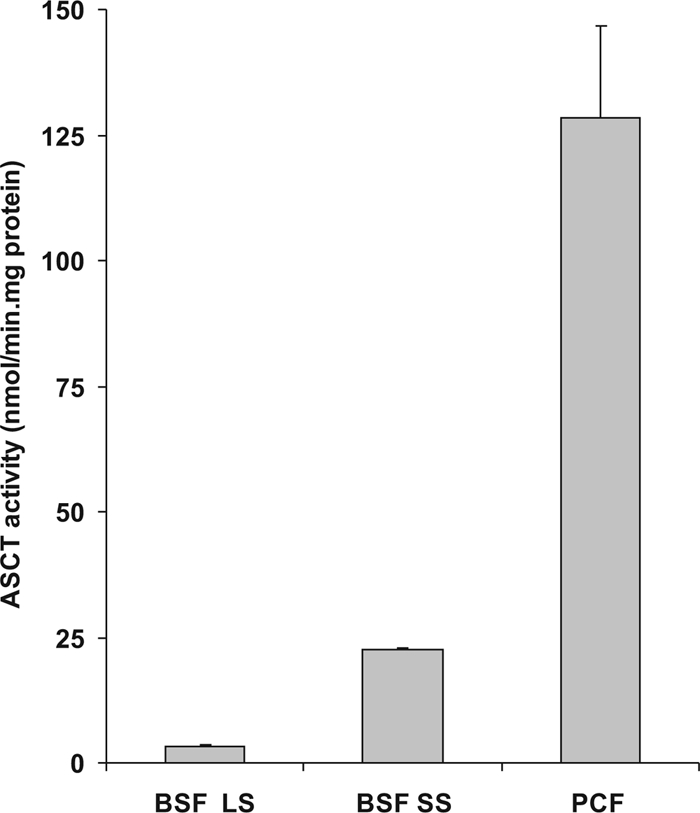
ASCT activity in total lysates of T. brucei AntAR 1. Enzymatic activity of ASCT was determined in total lysates derived from cultures containing predominantly long slender BSF trypanosomes (BSF LS; 94%), predominantly short stumpy BSF trypanosomes (BSF SS; 92%), or exclusively PCF trypanosomes (PCF). Shown are the means + standard deviations of three experiments.
Hence, our experiments show that short stumpy BSF trypanosomes do not only degrade glucose by glycolysis but additionally produce acetate. Acetate formation in trypanosomes occurs via the mitochondrial enzyme ASCT and involves transfer of a CoA moiety from acetyl-CoA to succinate, yielding succinyl-CoA (24). This succinyl-CoA can then be converted back into succinate by succinyl-CoA synthetase, a reaction concomitantly converting ADP in ATP (6, 24). Therefore, our observations that short stumpy BSF trypanosomes produce acetate and express ASCT demonstrate that these stages in addition to glycolysis also use a mitochondrial pathway for the degradation of glucose and production of ATP.
Multiple mitochondrial adaptations have been reported to occur during the transition from long slender BSF to short stumpy BSF T. brucei. Differential gene expression and the formation of cristea in the inner mitochondrial membrane have been shown to occur during this transition (8, 11, 19). Furthermore, the trypanosomal homologue of complex I of the respiratory chain is expressed in short stumpy BSF trypanosomes (4, 5, 18). Our experiments show that this more elaborate composition of the electron transport chain is also used by this stage, as the production of acetate implies that acetyl-CoA is formed, which is catalyzed by the pyruvate dehydrogenase complex and results in the production of NADH inside the mitochondrion. This means that either complex I or the alternative NADH dehydrogenase is active in this stage (18). Moreover, our experiments show that the previously reported mitochondrial adaptations in short stumpy BSF trypanosomes are not restricted to morphological changes and to changes in the composition of the electron transport chain but also result in a functionally altered energy metabolism.
In conclusion, the data described in this paper demonstrate the absence of a functional Krebs cycle in the mitochondria of PCF T. brucei, isolated from the tsetse midgut or freshly differentiated from BSF trypanosomes. Furthermore, we show that short stumpy BSF T. brucei cells produce large amounts of acetate. Therefore, the mitochondria of short stumpy trypanosomes are metabolically divergent from the mitochondria in long slender BSF T. brucei cells. These results are consistent with prior work (4, 5, 8, 11). The functional changes might be a preadaptation that allows short stumpy BSF T. brucei to function in the intestines of infected tsetse flies and enables them to differentiate further into PCF trypanosomes.
Footnotes
Published ahead of print on 19 June 2009.
REFERENCES
- 1.Barrett, M. P., R. J. Burchmore, A. Stich, J. O. Lazzari, A. C. Frasch, J. J. Cazzulo, and S. Krishna. 2003. The trypanosomiases. Lancet 3621469-1480. [DOI] [PubMed] [Google Scholar]
- 2.Berriman, M., E. Ghedin, C. Hertz-Fowler, G. Blandin, H. Renauld, D. C. Bartholomeu, N. J. Lennard, E. Caler, N. E. Hamlin, B. Haas, U. Bohme, L. Hannick, M. A. Aslett, J. Shallom, L. Marcello, L. Hou, B. Wickstead, U. C. Alsmark, C. Arrowsmith, R. J. Atkin, A. J. Barron, F. Bringaud, K. Brooks, M. Carrington, I. Cherevach, T. J. Chillingworth, C. Churcher, L. N. Clark, C. H. Corton, A. Cronin, R. M. Davies, J. Doggett, A. Djikeng, T. Feldblyum, M. C. Field, A. Fraser, I. Goodhead, Z. Hance, D. Harper, B. R. Harris, H. Hauser, J. Hostetler, A. Ivens, K. Jagels, D. Johnson, J. Johnson, K. Jones, A. X. Kerhornou, H. Koo, N. Larke, S. Landfear, C. Larkin, V. Leech, A. Line, A. Lord, A. Macleod, P. J. Mooney, S. Moule, D. M. Martin, G. W. Morgan, K. Mungall, H. Norbertczak, D. Ormond, G. Pai, C. S. Peacock, J. Peterson, M. A. Quail, E. Rabbinowitsch, M. A. Rajandream, C. Reitter, S. L. Salzberg, M. Sanders, S. Schobel, S. Sharp, M. Simmonds, A. J. Simpson, L. Tallon, C. M. Turner, A. Tait, A. R. Tivey, S. Van Aken, D. Walker, D. Wanless, S. Wang, B. White, O. White, S. Whitehead, J. Woodward, J. Wortman, M. D. Adams, T. M. Embley, K. Gull, E. Ullu, J. D. Barry, A. H. Fairlamb, F. Opperdoes, B. G. Barrell, J. E. Donelson, N. Hall, C. M. Fraser, S. E. Melville, and N. M. El-Sayed. 2005. The genome of the African trypanosome Trypanosoma brucei. Science 309416-422. [DOI] [PubMed] [Google Scholar]
- 3.Besteiro, S., M. P. Barrett, L. Rivière, and F. Bringaud. 2005. Energy generation in insect stages of Trypanosoma brucei: metabolism in flux. Trends Parasitol. 21185-191. [DOI] [PubMed] [Google Scholar]
- 4.Bienen, E. J., R. K. Maturi, G. Pollakis, and A. B. Clarkson, Jr. 1993. Non-cytochrome mediated mitochondrial ATP production in bloodstream form Trypanosoma brucei brucei. Eur. J. Biochem. 21675-80. [DOI] [PubMed] [Google Scholar]
- 5.Bienen, E. J., M. Saric, G. Pollakis, R. W. Grady, and A. B. Clarkson, Jr. 1991. Mitochondrial development in Trypanosoma brucei brucei transitional bloodstream forms. Mol. Biochem. Parasitol. 45185-192. [DOI] [PubMed] [Google Scholar]
- 6.Bochud-Allemann, N., and A. Schneider. 2002. Mitochondrial substrate level phosphorylation is essential for growth of procyclic Trypanosoma brucei. J. Biol. Chem. 27732849-32854. [DOI] [PubMed] [Google Scholar]
- 7.Brun, R., and M. Schönenberger. 1981. Stimulating effect of citrate and cis-aconitate on the transformation of Trypanosoma brucei bloodstream forms to procyclic forms in vitro. Z. Parasitenkd. 6617-24. [DOI] [PubMed] [Google Scholar]
- 8.Denninger, V., K. Figarella, C. Schonfeld, S. Brems, C. Busold, F. Lang, J. Hoheisel, and M. Duszenko. 2007. Troglitazone induces differentiation in Trypanosoma brucei. Exp. Cell Res. 3131805-1819. [DOI] [PubMed] [Google Scholar]
- 9.Durieux, P. O., P. Schutz, R. Brun, and P. Köhler. 1991. Alterations in Krebs cycle enzyme activities and carbohydrate catabolism in two strains of Trypanosoma brucei during in vitro differentiation of their bloodstream to procyclic stages. Mol. Biochem. Parasitol. 4519-27. [DOI] [PubMed] [Google Scholar]
- 10.Evans, D. A., and R. C. Brown. 1972. The utilization of glucose and proline by culture forms of Trypanosoma brucei. J. Protozool. 19686-690. [DOI] [PubMed] [Google Scholar]
- 11.Feagin, J. E., D. P. Jasmer, and K. Stuart. 1986. Differential mitochondrial gene expression between slender and stumpy bloodforms of Trypanosoma brucei. Mol. Biochem. Parasitol. 20207-214. [DOI] [PubMed] [Google Scholar]
- 12.Flynn, I. W., and I. B. Bowman. 1973. The metabolism of carbohydrate by pleomorphic African trypanosomes. Comp. Biochem. Physiol. B 4525-42. [DOI] [PubMed] [Google Scholar]
- 13.Hannaert, V., F. Bringaud, F. R. Opperdoes, and P. A. Michels. 2003. Evolution of energy metabolism and its compartmentation in Kinetoplastida. Kinetoplastid Biol. Dis. 211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Herman, M., D. Perez-Morga, N. Schtickzelle, and P. A. Michels. 2008. Turnover of glycosomes during life-cycle differentiation of Trypanosoma brucei. Autophagy 4294-308. [DOI] [PubMed] [Google Scholar]
- 15.Kubi, C., J. Van Den Abbeele, R. De Deken, T. Marcotty, P. Dorny, and P. Van den Bossche. 2006. The effect of starvation on the susceptibility of teneral and non-teneral tsetse flies to trypanosome infection. Med. Vet. Entomol. 20388-392. [DOI] [PubMed] [Google Scholar]
- 16.Lanham, S. M., and D. G. Godfrey. 1970. Isolation of salivarian trypanosomes from man and other mammals using DEAE-cellulose. Exp. Parasitol. 28521-534. [DOI] [PubMed] [Google Scholar]
- 17.Le Ray, D., J. D. Barry, C. Easton, and K. Vickerman. 1977. First tsetse fly transmission of the “AnTat” serodeme of Trypanosoma brucei. Ann. Soc. Belg. Med. Trop. 57369-381. [PubMed] [Google Scholar]
- 18.Opperdoes, F. R., and P. A. Michels. 2008. Complex I of Trypanosomatidae: does it exist? Trends Parasitol. 24310-317. [DOI] [PubMed] [Google Scholar]
- 19.Penketh, P. G., A. A. Divo, K. Shyam, C. L. Patton, and A. C. Sartorelli. 1991. The effects of the methylating agent 1,2-bis(methylsulfonyl)-1-methylhydrazine on morphology, DNA content and mitochondrial function of Trypanosoma brucei subspecies. J. Protozool. 38172-177. [DOI] [PubMed] [Google Scholar]
- 20.Rivière, L., S. W. H. van Weelden, P. Glass, P. Vegh, V. Coustou, M. Biran, J. J. van Hellemond, F. Bringaud, A. G. M. Tielens, and M. Boshart. 2004. Acetyl:succinate CoA-transferase in procyclic Trypanosoma brucei. Gene identification and role in carbohydrate metabolism. J. Biol. Chem. 27945337-45346. [DOI] [PubMed] [Google Scholar]
- 21.Ryley, J. F. 1962. Studies on the metabolism of the protozoa. 9. Comparative metabolism of blood-stream and culture forms of Trypanosoma rhodesiense. Biochem. J. 85211-223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Van Den Abbeele, J., Y. Claes, D. van Bockstaele, D. Le Ray, and M. Coosemans. 1999. Trypanosoma brucei spp. development in the tsetse fly: characterization of the post-mesocyclic stages in the foregut and proboscis. Parasitology 118469-478. [DOI] [PubMed] [Google Scholar]
- 23.van Hellemond, J. J., B. M. Bakker, and A. G. M. Tielens. 2005. Energy metabolism and its compartmentation in Trypanosoma brucei. Adv. Microb. Physiol. 50199-226. [DOI] [PubMed] [Google Scholar]
- 24.van Hellemond, J. J., F. R. Opperdoes, and A. G. M. Tielens. 1998. Trypanosomatidae produce acetate via a mitochondrial acetate:succinate CoA transferase. Proc. Natl. Acad. Sci. USA 953036-3041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van Weelden, S. W. H., B. Fast, A. Vogt, P. van der Meer, J. Saas, J. J. van Hellemond, A. G. M. Tielens, and M. Boshart. 2003. Procyclic Trypanosoma brucei do not use Krebs cycle activity for energy generation. J. Biol. Chem. 27812854-12863. [DOI] [PubMed] [Google Scholar]
- 26.van Weelden, S. W. H., J. J. van Hellemond, F. R. Opperdoes, and A. G. M. Tielens. 2005. New functions for parts of the Krebs cycle in procyclic Trypanosoma brucei, a cycle not operating as a cycle. J. Biol. Chem. 28012451-12460. [DOI] [PubMed] [Google Scholar]