Summary
The allatostatins comprise three structurally distinct peptide families that regulate juvenile hormone production by the insect corpora allata. A-type family members contain the C-terminal motif –YXFGLamide and have been found in species from numerous arthropod taxa. Members of the B-type family exhibit a –WX6Wamide C-terminus and, like the A-type peptides, appear to be broadly conserved within the Arthropoda. By contrast, members of the C-type family, typified by the unblocked C-terminus –PISCF, a pyroglutamine blocked N-terminus, and a disulfide bridge between two internal Cys residues, have only been found in holometabolous insects, i.e. lepidopterans and dipterans. Here, using transcriptomics, we have identified SYWKQCAFNAVSCFamide (disulfide bridging predicted between the two Cys residues), a known honeybee and water flea C-type-like peptide, from the American lobster Homarus americanus (infraorder Astacidea). Using matrix assisted laser desorption/ionization Fourier transform mass spectrometry (MALDI-FTMS), a mass corresponding to that of SYWKQCAFNAVSCFamide was detected in the H. americanus brain, supporting the existence of this peptide and its theorized structure. Furthermore, SYWKQCAFNAVSCFamide was detected by MALDI-FTMS in neural tissues from five additional astacideans as well as 19 members of four other decapod infraorders (i.e. Achelata, Anomura, Brachyura and Thalassinidea), suggesting that it is a broadly conserved decapod peptide. In H. americanus, SYWKQCAFNAVSCFamide is capable of modulating the output of both the pyloric circuit of the stomatogastric nervous system and the heart. This is the first demonstration of bioactivity for this peptide in any species.
Keywords: Homarus americanus, transcriptomics, expressed sequence tag (EST), matrix assisted laser desorption/ionization-Fourier transform mass spectrometry (MALDI-FTMS), stomatogastric ganglion, heart
INTRODUCTION
The allatostatins, named for their inhibition of juvenile hormone by the corpora allata in insects, are pleiotropic arthropod neuropeptides (Stay and Tobe, 2007). Three distinct families of allatostatins are currently known (Stay and Tobe, 2007). The first family identified, commonly referred to as the A-type allatostatins (A-ASTs), is typified by the carboxyl (C)-terminal motif –YXFGLamide (where X represents a variable amino acid). Although initially identified from the cockroach Diploptera punctata (Woodhead et al., 1989), A-ASTs have subsequently been found in species from a wide array of insect taxa, though they appear to possess allatostatic function in only a subset of these insects, i.e. cockroaches, crickets and termites (Stay and Tobe, 2007). A-type peptides have also been found in a number of crustaceans (Duve et al., 1997; Dircksen et al., 1999; Duve et al., 2002; Huybrechts et al., 2003; Fu et al., 2005; Yasuda-Kamatani and Yasuda, 2006; Yin et al., 2006; Christie et al., 2008a; Ma et al., 2008; Gard et al., 2009; Ma et al., 2009), where they have been shown to function as locally released and/or hormonally delivered neuro/myomodulators (Skiebe and Schneider, 1994; Jorge-Rivera and Marder, 1997; Jorge-Rivera et al., 1998; Dircksen et al., 1999; Kreissl et al., 1999; Birmingham et al., 2003; Billimoria et al., 2006; Cruz-Bermúdez and Marder, 2007). Molecular, biochemical and mass spectral analyses have shown that multiple isoforms of A-type peptides are common in both insect and crustacean species, with multiple peptides encoded by the known arthropod A-AST prepro-hormones (e.g. Duve et al., 1997; Dircksen et al., 1999; Lenz et al., 2000; Meyering-Vos et al., 2001; Duve et al., 2002; Huybrechts et al., 2003; Abdel-Latief et al., 2004; Fu et al., 2005; Yasuda-Kamatani and Yasuda, 2006; Yin et al., 2006; Bowser and Tobe, 2007; Christie et al., 2008a; Ma et al., 2008; Gard et al., 2009; Ma et al., 2009).
A second family of allatostatic peptides, originally isolated from the cricket Gryllus bimaculatus (Lorenz et al., 1995) and possessing the C-terminal motif –WX6Wamide (X6 representing six variable residues), has become known as the B-type allatostatins (B-ASTs) (Stay and Tobe, 2007). As with the A-type peptides, the B-ASTs have subsequently been found in a number of insect species, although their allatostatic potential appears to be limited to crickets (Stay and Tobe, 2007). B-type peptides have also been found in a number of crustaceans (Fu et al., 2005; Fu et al., 2007; Christie et al., 2008b; Ma et al., 2008; Gard et al., 2009; Ma et al., 2009), where they, like the A-type family members, appear to function as locally released and/or hormonally delivered neuromodulators (Fu et al., 2007). In both insect and crustacean species, multiple B-type peptides are common, with multiple isoforms encoded by the known B-AST precursors (e.g. Williamson et al., 2001a; Wang et al., 2004; Fu et al., 2005; Christie, 2008a; Christie et al., 2008b; Ma et al., 2008; Weaver and Audsley, 2008; Gard et al., 2009; Ma et al., 2009).
The final allatostatin family is commonly referred to as the C-type allatostatins (C-ASTs). Members of this peptide group, first identified from the tobacco hornworm Manduca sexta (Kramer et al., 1991), are characterized by the presence of the non-amidated C-terminal motif –PISCF, a pyroglutamine blocked amino (N)-terminus, and a disulfide bridge between the Cys residues located at positions 7 and 14 (Stay and Tobe, 2007). Thus far, C-ASTs have been isolated/predicted only from lepidopteran and dipteran species, where in at least the former, they possess allatostatic activity (Stay and Tobe, 2007). In contrast to the A- and B-ASTs, only a single C-type isoform has been identified in any given insect, and only a single C-AST is encoded by the known prepro-C-ASTs (e.g. Williamson et al., 2001b; Li et al., 2006; Sheng et al., 2007). No information is currently available on the presence or function of C-ASTs in decapod crustaceans, though a putative C-type-related peptide, SYWKQCAFNAVSCFamide, originally described from the honeybee Apis mellifera (Hummon et al., 2006), has recently been predicted via transcriptomics from the cladoceran crustacean Daphnia pulex (Gard et al., 2009).
In the study presented here, we have used a strategy combining bioinformatics, mass spectrometry and physiological studies to explore the possibility that bioactive C-AST-like peptides exist in decapod crustaceans. Specifically, functional genomics was used to search the extant decapod expressed sequence tag (EST) database for transcripts encoding putative C-AST-like peptides via queries employing the sequences of known precursor proteins, an approach that has recently proved successful for similar peptide mining in other arthropod species (Christie, 2008a; Christie, 2008b; Christie et al., 2008b; Gard et al., 2009; Ma et al., 2009). Through our in silico (i.e. computer-based) mining, a putative C-type-related peptide-encoding transcript was identified from the American lobster Homarus americanus (infraorder Astacidea). The mature structure of the encoded peptide (SYWKQCAFNAVSCFamide; disulfide bridging between the two internal Cys residues) was predicted through a combination of online peptide processing programs and by homology to known C-AST and C-AST-related peptide isoforms. Using mass spectral methods, we confirmed the existence of SYWKQCAFNAVSCFamide in H. americanus, and identified it in 24 other decapods, which included members of four additional infraorders, suggesting a broad conservation of the peptide in this taxon. In addition, physiological experiments demonstrated that SYWKQCAFNAVSCFamide modulates the neural circuitry present in the stomatogastric ganglion (STG) and is cardioactive in H. americanus. These are the first demonstrations of bioactivity for this peptide in any species. Some of these data have appeared previously in abstract form (Christie et al., 2008c).
MATERIALS AND METHODS
Animals
The animals used in our study were as follows: infraorder Penaeidae – Farfantepenaeus duorarum Burkenroad (purchased from Gulf Specimen Marine Laboratories, Panacea, FL, USA) and Litopenaeus vannamei Boone (purchased from Greene Prairie Aquafarms, Boligee, AL, USA); infraorder Caridea – Crangon septemspinosa Say (collected by hand at Mount Desert Island Biological Laboratory, Salisbury Cove, ME, USA), Pandalus danae Stimpson (collected by hand at Friday Harbor Laboratories, Friday Harbor, WA, USA) and Pandalus platyceros Brandt (collected by trap or dredge, San Juan Island area, WA, USA); infraorder Astacidea – Cherax quadricarinatus von Martens (purchased from Stick-Fins Fish Farm, Elkton, FL, USA), Homarus americanus Milne-Edwards (purchased from local suppliers; Brunswick, ME, USA), Homarus gammarus Linnaeus (purchased from Scottish Wild Harvest, Plainfield, NJ, USA), Nephrops norvegicus Linnaeus (purchased from Scottish Wild Harvest), Pacifastacus leniusculus Dana (collected by trap from Lake Washington, Seattle, WA, USA) and Procambarus clarkii Girard (purchased from Carolina Biological Supply Company, Burlington, NC, USA); infraorder Thalassinidea – Callianassa californiensis Dana (purchased from Ray's Bait Works, Snohomish, WA, USA); infraorder Achelata–Panulirus interruptus Randall (purchased from Tomlinson Commercial Fishing, San Diego, CA, USA), Panulirus versicolor Latreille (purchased from That Pet Place, Lancaster, PA, USA) and Scyllarides latus Latreille (purchased from That Pet Place); infraorder Anomura – Clibanarius vittatus Bosc (purchased from Gulf Specimens Marine Laboratories), Lithodes maja Linnaeus (purchased from local suppliers; Brunswick, ME, USA), Pachycheles rudis Stimpson (collected by hand at Neah Bay, WA, USA), Pagurus acadianus Benedict (collected by hand at Mount Desert Island Biological Laboratory), Pagurus granosimanus Stimpson (collected by hand throughout the great Puget Sound area), Pagurus pollicaris Say (collected by hand at Rocky Neck State Park, Niantic, CT, USA), Petrolisthes cinctipes Randall (collected by hand at Neah Bay) and Petrolisthes eriomerus Stimpson (collected by hand throughout the great Puget Sound area, WA, USA); infraorder Brachyura – Cancer antennarius Stimpson (collected by hand at Neah Bay), Cancer borealis Stimpson (purchased from local suppliers, Brunswick, ME, USA), Cancer gracilis Dana (collected by hand at False Bay, San Juan Island, WA, USA), Cancer irroratus Say (purchased from local suppliers, Brunswick, ME, USA), Cancer magister Dana (collected by hand or trap throughout the great Puget Sound area), Cancer productus Randall (collected by hand or trap throughout the great Puget Sound area), Carcinus maenas Linnaeus (collected by hand at Mount Desert Island Biological Laboratory), Hemigrapsus nudus Dana (collected by hand throughout the great Puget Sound area), Lophopanopeus bellus Stimpson (collected by hand throughout the great Puget Sound area), Ovalipes ocellatus Herbst (collected by hand at Rocky Neck State Park), Pugettia gracilis Dana (collected by hand at Friday Harbor Laboratories), Pugettia producta Randall (collected by hand at Friday Harbor Laboratories) and Scyra acutifrons Dana (collected by hand at Friday Harbor Laboratories).
With the following exceptions, animals were maintained in aerated natural seawater aquaria at 8–12°C. For F. duorarum, L. vannamei, P. versicolor, S. latus and C. vittatus, aerated natural seawater aquaria were held at 18–20°C, and those for P. interruptus were maintained at approximately 15°C. C. californiensis were maintained in seawater-moistened wood shavings at 10°C. C. quadricarinatus and P. clarkii were held in aerated tanks of aged tap water at 18–20°C, and P. leniusculus were maintained in aged tap water tanks at 10°C.
All animals were anesthetized by packing in ice for 30–60 min prior to dissections. Tissues were removed in cold (approximately 10°C) physiological saline appropriate to the species (compositions in mmol l–1): for Penaeidea, Caridea and Achelata, 479 NaCl, 12.8 KCl, 13.7 CaCl2, 3.9 Na2SO4, 10 MgSO4, 11 Trizma base, and 4.8 maleic acid; pH 7.5–7.6; for salt water Astacidea (Homarus and Nephrops species) and Anomura, 479 NaCl, 12.8 KCl, 13.7 CaCl2, 20 MgSO4, 3.9 Na2SO4, and 4.8 Hepes (pH 7.4–7.5); for freshwater Astacidea (crayfish species), 200 NaCl, 5.4 KCl, 17.2 CaCl2, 5.5 MgCl2, 22 Tris base and 4.7 maleic acid, pH 7.2–7.4; for Thalassinidea and Brachyura, 442 NaCl, 11 KCl, 13 CaCl2, 26 MgCl2, 12 Trizma base, and 1.2 maleic acid (pH 7.4–7.6).
Functional genomics
Database searches
Transcriptome searches were conducted using methods modified from several recent publications (Christie, 2008a; Christie, 2008b; Christie et al., 2008b; Gard et al., 2009; Ma et al., 2009). Specifically, the online program tblastn (NCBI; http://www.ncbi.nlm.nih.gov/BLAST/) was used to mine for ESTs encoding putative crustacean C-AST and C-AST-related peptide precursors via queries using known arthropod prepro-hormone sequences. For all searches, the program database was set to non-human, non-mouse ESTs (EST_others) and was restricted to crustacean sequences (taxid:6657). All hits were fully translated and checked manually for homology to the target query (see below).
Peptide prediction
Translation of the nucleotide sequence of the identified ESTs was performed using the Translate tool of ExPASy (Swiss Institute of Bioinformatics, Basel, Switzerland; http://www.expasy.ch/tools/dna.html). Signal peptide prediction was done via the online program SignalP 3.0, using both the Neural Networks and the Hidden Markov Models algorithms (Center for Biological Sequence Analysis, Technical University of Denmark, Lyngby, Denmark; http://www.cbs.dtu.dk/services/SignalP/ (Bendtsen et al., 2004). Pro-hormone convertase cleavage sites were predicted based on the information presented in Veenstra (Veenstra, 2000). Post-translational modifications, e.g. cyclization of N-terminal Gln/Glu residues, C-terminal amidation at Gly residues and disulfide bridging between Cys residues, were predicted by homology to known C-type and/or C-type-like peptides.
Mass spectrometry
Sample preparation
Direct tissue analyses
To prepare samples for direct tissue matrix-assisted laser desorption/ionization-Fourier transform mass spectrometry (MALDI-FTMS), we first isolated either small pieces of a larger tissue sample, i.e. the supraoesophageal ganglion (brain), posterior midgut caecum (PMC) or pericardial organ (PO), or the entire tissue, i.e. the commissural ganglion (CoG), stomatogastric ganglion (STG) or sinus gland (SG), using manual micro-dissection techniques. The ganglionic sheath surrounding the brain, CoGs and STG was removed with further manual microdissection. The isolated tissue was then removed from the saline with fine forceps, rinsed sequentially in two 20 μl droplets of 0.75 mol l–1 fructose (Sigma-Aldrich, St Louis, MO, USA; 99%) and placed on a face of a ten-faceted stainless steel probe tip, minimizing co-transfers of solution. The tissue was then sliced 10–20 times with a 0.2 mm needle, gathered together and covered with a 0.5 μl droplet of 1.0 mol l–1 2,5-dihydroxybenzoic acid (DHB; Sigma-Aldrich; 98%, sublimed prior to use) prepared in 1:1 acetonitrile (Fisher Scientific, Pittsburg, PA, USA; HPLC grade):water containing 2% (v/v) phosphoric acid (Sigma-Aldrich, 99%).
Analyses of tissue extracts
To prepare tissue extracts for mass spectral analyses, paired, desheathed CoGs, an entire eyestalk, brain or SG were removed from saline, rinsed sequentially in two 20 μl droplets of 0.75 mol l–1 fructose and placed in a 0.6 ml tube with 30 μl of extraction solvent (7% acetic acid, 64% methanol, 29% deionized H2O). The tissues were homogenized by cutting with spring scissors. The homogenate was sonicated for 2 min and centrifuged at 2200 g for 5 min in a microcentrifuge (Fischer Scientific). The supernatant was saved, and the pellet resuspended with 5 μl of deionized water. The sonication, centrifugation and resuspension steps were repeated two additional times. The supernatants of all cycles were combined. Deionized H2O (20μl) and CDCl3 (25μl, Sigma-Aldrich) were added to the solution. The organic layer was removed, and the aqueous layer evaporated to dryness. For most samples, the resultant extracts were desalted using C18 ZipTip pipette tips (Millipore, Billerica, MA, USA). After their preparation, 0.5 μl of extract was mixed with 0.5 μl of DHB matrix on one face of the MALDI probe and the extract-matrix mixture was allowed to co-crystallize.
Instrumentation
Samples were analyzed using a HiResMALDI Fourier transform mass spectrometer (IonSpec, Lake Forest, CA, USA) equipped with a Cryomagnetics (Oak Ridge, TN, USA) 4.7 Tesla actively-shielded superconducting magnet. Ions were generated using a pulsed nitrogen laser (337 nm) and were transported from the external ion source to the closed cylindrical ICR cell using a quadrupole ion guide. The ion guide radio frequency potential and trapping delay time were optimized to transmit and trap ions of a selected mass range (optimized for m/z 1,500 or 2,500). A pulse of argon was introduced to the vacuum system during trapping to elevate the system pressure transiently for collisional cooling. All spectra were measured using ion accumulation techniques, where ions from seven to 30 successive laser shots were accumulated in the cell. Exact mass measurements were calibrated using either the internal calibration on adjacent samples (InCAS) technique (O'Connor and Costello, 2000), modified to include the accumulation of mass-selected calibrant ions (Stemmler et al., 2007) or calibration with previously identified peptides (GYRKPPFNGSIFamide, VYRKPPFNGSIFamide, APSGFLGMRamide and pQDLDHVFLRFamide). For calibration of samples from brachyurans and anomurans, NFDEIDRSGFGFA and its fragment ions were also used for calibration. A delay of 5–10 s preceded ion detection, which occurred with analyzer pressures of 1–7×10–8 Pa. Transients from direct tissue spectra were apodized using a Blackman function and zero-filled prior to fast Fourier transformation.
Electrophysiology
To assess the effects of SYWKQCAFNAVSCFamide on the H. americanus neurogenic heart and on the neural circuits contained within the stomatogastric nervous system (STNS), individual animals were cold-anaesthetized by packing them in ice for 30–60 min, after which the posterior dorsal region of the thoracic carapace that lies directly over the heart, as well as the underlying cardiac tissue, was removed. The foregut, over which the STNS lies, was also removed at this time.
Stomatogastric physiology
To assess the actions of SYWKQCAFNAVSCFamide on the motor output of the stomatogastric neural circuits, the STNS was isolated from the foregut by manual micro-dissection. Specifically, the foregut was flattened by making a longitudinal cut on its ventral side from the oesophagus to the pylorus, followed by a pair of medial cuts directed along the ossicles of the cardiac sac/gastric mill. The foregut was flattened and pinned inside down in a Sylgard 170 (KR Anderson)-lined dish containing chilled physiological saline (see above). The STNS, including the nerves that interconnect the ganglia and the major motor nerves, was then dissected free of the foregut musculature.
For physiological recordings, the STNS was pinned out in a Sylgard 184-lined Petri dish containing chilled physiological saline, and the sheath over the STG was removed to provide the superfused SYWKQCAFNAVSCFamide (see above) with access to the somata and the neuropil present within the ganglion. In addition, the sheath surrounding the stomatogastric nerve (stn) was removed so that action potential conduction could be reversibly blocked using isotonic (750 mmol l–1) sucrose in a petroleum jelly well surrounding the desheathed portion of the nerve. This sucrose block eliminated the majority of the modulatory inputs to the STG, as the stn is the only nerve that carries the axons of the modulatory projection neurons located in the commissural (CoG) and oesophageal (OG) ganglia to the STG. During recordings, the dish containing the STNS was continuously superfused with chilled (10–12°C) physiological saline. A petroleum jelly wall was constructed across the dish so that the STG could be superfused with SYWKQCAFNAVSCFamide-containing saline while superfusing the remainder of the dish with normal saline. The peptide was superfused over the preparation for 10 min, and then replaced with control saline. In all preparations in which cycle frequency changed, it returned to baseline levels after the peptide was washed out.
SYWKQCAFNAVSCFamide (including a disulfide bridge between Cys residues 6 and 13; custom synthesized by GenScript Corporation, Piscataway, NJ, USA) was dissolved in deionized water at a concentration of 10–3 mol l–1, and kept as a frozen stock solution at –20°C for use in physiological experiments. This peptide stock was diluted in chilled physiological saline to a final concentration of 10–6 mol l–1 just prior to use.
Neuronal activity was recorded extracellularly on the motor nerves of the STNS with an A-M Systems Model 1700AC amplifier (A-M Systems, Inc., Carlsborg, WA, USA) using stainless steel pin electrodes, which were isolated from the bath with petroleum jelly wells. The electrical activity in the stomatogastric neurons was further processed with a Brownlee 410 instrumentation amplifier and recorded directly onto a computer using a Micro 1401 data acquisition board and Spike2 version 5 or 6 software. Data were analyzed using the built-in functions of Spike2 and scripts available on the Bucher lab website (http://www.whitney.ufl.edu/BucherLab/Spike2_Scripts2_box.htm). Measurements of control cycle frequency are averages of the 100 s just before peptide application; measurements of cycle frequency in SYWKQCAFNAVSCFamide are the averages of cycle frequency recorded for 100 s beginning 8 min after the onset of peptide application. Data were further analyzed and graphed using Prism 5 software (GraphPad Software, San Diego, CA, USA).
Cardiac physiology
For assessment of the cardiotropic actions of SYWKQCAFNAVSCFamide, the dissected region containing the heart was pinned through the carapace to the bottom of a small Sylgard 184 (KR Anderson, Santa Clara, CA, USA)-lined dish. The dorsal part of the heart remained attached to the carapace, so that the extent to which it was stretched was identical to that in the intact animal. The posterior artery was cannulated with a short piece of polyethylene tubing drawn out to fit the artery, and was continuously perfused with chilled physiological saline at a flow rate of 2.5 to 3.0 ml m–1. A second perfusion line was directed across the top of the heart to help maintain temperature, which was monitored continuously and kept between 10–12°C. In all experiments, preparations were allowed to stabilize for 1–2 h before the first application of peptide. To examine the effects of SYWKQCAFNAVSCFamide on heartbeat amplitude and frequency, 10–6 mol l–1 SYWKQCAFNAVSCFamide was applied through the heart via the cannula for 8 min, after which the perfusion was switched back to control saline. Both frequency and amplitude of heart contractions returned to baseline levels within 30 min in all preparations.
To record heart contractions, the anterior arteries were tied off with 6–0 suture silk and attached to a Grass FT03 force-displacement transducer (Astro-Med, West Warwick, RI, USA) at an angle of approximately 30°, with an initial tension of 2 g. The output of the transducer was amplified via a Brownlee 410 instrumentation amplifier (San Jose, CA, USA), and recorded onto a PC computer using a Micro 1401 data acquisition board and Spike2 version 5 or 6 software (Cambridge Electronic Design Limited, Cambridge, UK). Both heart rate and contraction amplitude were measured using the built-in functions of Spike2 and a script written for this purpose. To calculate percentage change, average heartbeat amplitudes and frequencies at the peak of the peptide effect, 5–6 min after the onset of peptide application, were measured for a 200 s period and compared with the 200 s just before application of the peptide. Data were further analyzed and graphed using Prism4 software (GraphPad Software, San Diego, CA, USA).
RESULTS
Identification of SYWKQCAFNAVSCFamide by functional genomics
Recently, extensive collections of ESTs have been generated for a number of crustacean species and deposited in publicly accessible databases. This public deposition of sequence data has created a rich resource from which to mine potential proteins of interest using bioinformatics, including neuropeptide precursor proteins (e.g. Christie, 2008a; Christie, 2008b; Christie et al., 2008b; Gard et al., 2009; Ma et al., 2009). Here, using the program tblastn (search of translated nucleotide databases using a protein query), we have searched for putative decapod C-AST-related peptide-encoding transcripts via queries using known insect/crustacean precursors. Moreover, we have predicted the structures of the mature peptides contained within the deduced prepro-hormones via on-line protein processing programs and by homology to known C-AST and C-type-related peptide isoforms.
Three H. americanus ESTs were identified as encoding putative C-AST-related peptide precursors (Table 1) via queries using the deduced sequence of a D. pulex C-AST-related peptide-containing prepro-hormone (accession no. FE277533) (Gard et al., 2009). Translation of these transcripts revealed EY291152 to encode a 105 amino acid, putative full-length prepro-hormone, with CN852636 and CN852647 encoding identical 81 amino acid, C-terminal, partial prepro-hormones (Fig. 1). The two partial prepro-hormones overlap with the C-terminal portion of the full-length prepro-hormone, with the sequences of the overlapping portions being identical (Fig. 1). SignalP analysis of the full-length sequence identified the first 25 amino acids as a signal peptide, with the cleavage locus predicted between Ala25 and Lys26 (Fig. 1). Within the remaining pro-hormone, a single Lys-Arg prohormone convertase processing site was identified (Fig. 1), cleavage at which, followed by carboxypeptidase activity and α-amidation at an exposed C-terminal Gly residue, is predicted to produce two peptides (listed in their order of appearance in the pro-hormone): KALPDQDPQVYGQMPHMLDPAGNHLIDDDGSLDAVLINYLFAKQMVER LRNNADIKDLQR and the C-AST-related peptide SYWKQCAFNAVSCFamide (disulfide bridging predicted between Cys6 and Cys13).
Table 1.
Bioinformatics of putative decapod C-type allatostatin-related peptide encoding expressed sequence tags (ESTs)
The Homarus americanus cDNA library from which the ESTs were derived was generated using multiple tissues (Towle and Smith, 2006). The only neural tissue present was the supraoesophageal ganglion, commonly referred to as the brain
ESTs identified using the deduced amino acid sequence of accession no. FE277533 (Gard et al., 2009) as a query
Fig. 1.
Deduced amino acid sequence of a Homarus americanus C-AST-like peptide-containing prepro-hormone. Accession numbers of the ESTs from which the prepro-hormone was predicted are shown on the left with the deduced amino acid sequences of the precursor protein shown on the right. Signal peptides (when present) are shown in grey, with prohormone convertase cleavage loci shown in black. The C-AST-like peptide isoform is shown in red, with an additional precursor-related peptide shown in blue. Asterisks indicate the position of the stop codon.
MALDI-FTMS analyses support the existence of SYWKQCAFNAVSCFamide in Homarus americanus neural tissues
To determine if the predicted C-AST-related peptide, SYWKQCAFNAVSCFamide (with a disulfide bridge), was present in neural and gut (PMC) tissues, we directly analyzed freshly dissected tissue samples from H. americanus. For larger tissue samples, we surveyed smaller pieces of the larger tissue from the brain, PMC and PO. For smaller tissues, including the CoG, STG and SG, the entire tissue was analyzed directly. For some tissues [eyestalk (including the SG), brain and CoG] we also analyzed tissue extracts generated from either a single tissue (eyestalk, SG and brain) or from two paired tissues that originated from one individual (CoGs).
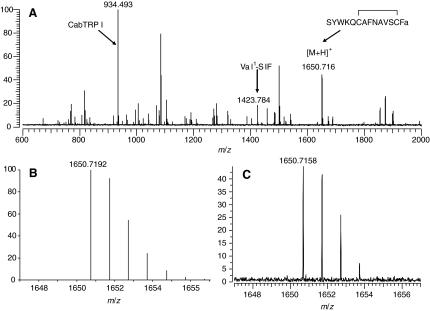
As shown in Fig. 2, we found evidence to support the detection of SYWKQCAFNAVSCFamide (with a disulfide bridge) in direct tissue samples from the brain of H. americanus. We detected a peak showing good agreement (–2.0 p.p.m.) with the predicted m/z value of 1650.7192 (see Fig. 2A), and an expansion of the measured isotopic distribution (Fig. 2C) was consistent with the distribution predicted by the peptide elemental composition (Fig. 2B). The intensity of the putative SYWKQCAFNAVSCFamide peak shown in Fig. 2A was replicated in other measurements made from this small brain tissue sample; however, the relative intensity of this peak was generally found to be much lower in other samples regardless of whether brain tissues were analyzed directly or as extracts. Low intensity putative SYWKQCAFNAVSCFamide peptide peaks were also commonly observed for our analyses of CoG tissues (for H. americanus and other species). This low response may reflect low peptide abundance relative to other peptides in the sample. It may also be the consequence of the lower ionization efficiency for SYWKQCAFNAVSCFamide, which does not contain a basic arginine residue in the peptide sequence. The absence of an arginine residue has been correlated with a lower MALDI response for tryptic peptides (Krause et al., 1999). Because of low signal intensities, we have relied upon exact mass measurements for our identification of SYWKQCAFNAVSCFamide (with a disulfide bond) in different tissue samples.
Fig. 2.
(A) Direct MALDI-FTMS spectrum of a small piece of freshly dissected Homarus americanus supraoesophageal ganglion (brain). This spectrum was measured using DHB as the matrix, with the conditions optimized for accumulation of m/z 2500. Calibration was done using the known peptide peaks, APSGFLGMRamide (CabTRP I) and VYRKPPFNGSIFamide (Val1-SIF) at m/z 934.4927 and m/z 1423.7845, respectively. As can be seen from this spectrum, a peak corresponding to that of the [M+H]+ ion for SYWKQCAFNAVSCFamide (a disulfide bridge present between the cysteine residues) was present at m/z 1650.7158 (–2.0 p.p.m. error from the theoretical m/z of 1650.7192). (B) Predicted mass and isotopic distribution for the [M+H]+ ion for SYWKQCAFNAVSCFamide (a disulfide bridge present between the cysteine residues). (C) An expansion of the measured isotopic distribution for putative SYWKQCAFNAVSCFamide, showing that the measured mass and isotopic distribution strongly support the existence of this peptide in the lobster brain.
Table 2 summarizes the results of our survey of tissues from H. americanus. We found evidence, based upon exact mass measurements, to support the detection of SYWKQCAFNAVSCFamide (with a disulfide bond) in the brain, CoG and eyestalk tissues. Although SYWKQCAFNAVSCFamide was detected in the analysis of an extract of an entire eyestalk tissue, the peptide was not detected in the analysis of single SGs (either direct tissue or tissue extracts). We also did not detect the peptide in the POs or PMC. Interestingly, we did detect a peak at m/z 1652.7310 in the direct analysis of tissues from the POs, which shows good agreement (–2.4 p.p.m.) with the mass for the reduced form of SYWKQCAFNAVSCFamide (m/z 1652.7348, calculated).
Table 2.
Exact mass measurements for SYWKQCAFNAVSCFamide (with a disulfide bridge) from the analysis of freshly dissected tissue samples or tissue extracts using MALDI-FTMS
| Tissue | Sample type* | Measured m/z† | Error (p.p.m.)‡ |
|---|---|---|---|
| Supraoesophageal ganglion (brain) | DT and E | 1650.7160 | –1.9 |
| Eyestalk | E | 1650.7220 | 1.3 |
| Sinus gland (SG) | DT and E | ND | |
| Commisural ganglion (CoG) | DT and E | 1650.7205 | 0.8 |
| Stomatogastric ganglion (STG) | DT | ND | |
| Pericardial organ (PO) | DT | ND§ | |
| Posterior midgut caecum (PMC) | DT | ND |
Peptide identity, SYWKQCAFNAVSCFamide (with disulfide bridge); ion identity, [M+H]+; calculated m/z, m/z 1650.71918 (monoisotopic)
ND, not detected
DT, direct tissue; E, extract
Average of three or more calibrated mass measurements. Spectra were calibrated using either poly(propylene glycol) or VYRKPPFNGSIFamide (m/z 1423.7845), APSGFLGMRamide (m/z 934.4927), and pQDLDHVFLRFamide (m/z 1271.6531 and m/z 817.4832)
Mass measurement error in p.p.m.
A peak detected at m/z 1652.7309, which may correspond to reduced SYWKQCAFNAVSCFamide (calculated m/z 1652.7348)
MALDI-FTMS suggests that SYWKQCAFNAVSCFamide is a broadly conserved decapod neuropeptide
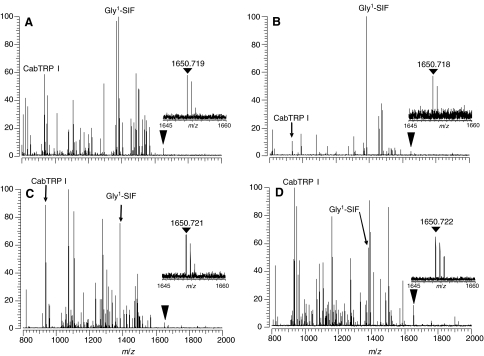
To determine whether SYWKQCAFNAVSCFamide (with a disulfide bond) is a broadly conserved decapod neuropeptide, we searched for this peptide in MALDI-FTMS spectra of directly analyzed CoG tissues from 36 species, spanning seven infraorders. MALDI-FTMS spectra from four species representing four crustacean infraorders are shown in Fig. 3. The majority of the collection of spectra that were analyzed were acquired as part of a prior study that involved a broad survey to detect highly conserved neuropeptides (Stemmler et al., 2007). The spectra were collected using conditions optimized for the detection of m/z 1500 and peptide masses were established using internal calibration with a polymer calibrant. Table 3 contains a summary of species in which we found evidence for SYWKQCAFNAVSCFamide (with a disulfide bond). In many cases, the putative peptide was not detected in spectra calibrated with the polymer calibrant, but peaks were detected in other spectra in the data set. These spectra were then internally calibrated using the masses of peptides that had been established using the polymer calibrant. Because of the low response we generally observed for the putative peptide in CoG tissue samples, and because the spectra were acquired using conditions that were not optimized for the detection of SYWKQCAFNAVSCFamide, it is important to recognize that the peptide may be present in species in which it was not detected in our study. Regardless, our results provide evidence to support SYWKQCAFNAVSCFamide (with a disulfide bond) as a peptide found in five infraorders of decapod crustaceans.
Fig. 3.
Direct tissue MALDI-FTMS spectra of freshly dissected commissural ganglia from (A) Petrolisthes cinctipes (infraorder Anomura), (B) Cancer magister (infraorder Brachyura), (C) Panulirus versicolor (infraorder Achelata) and (D) Callianassa californiensis (infraorder Thalassinidea). All spectra were measured using DHB as the matrix, with conditions optimized for m/z 1500. The inverted triangle shows the location of the m/z of the [M+H]+ ion for SYWKQCAFNAVSCFamide (disulfide bridge present between the cysteine residues; calculated m/z=1650.7192). The inserts show an expansion of the [M+H]+ peak region to show the measured mass and the isotopic distributions. Spectra were calibrated using known peptide peaks, including APSGFLGMRamide (CabTRP I) and GYRKPPFNGSIFamide (Gly1-SIF) at m/z 934.4927 and m/z 1381.7375, respectively.
Table 3.
Exact mass measurements for SYWKQCAFNAVSCFamide (with a disulfide bridge) from the analysis of freshly dissected and desheathed commissural ganglia tissue samples using MALDI-FTMS
| Infraorder | Species | Measured m/z* | Error (p.p.m.)† |
|---|---|---|---|
| Penaeidae | Farfantepenaeus duorarum | ND | |
| Litopenaeus vannamei | ND | ||
| Caridea | Crangon septemspinosa | ND | |
| Pandalus danae | ND | ||
| Pandalus platyceros | ND | ||
| Astacidea | Cherax quadricarinatus | 1650.7149 | –2.6 |
| Homarus americanus | 1650.7205 | 0.8 | |
| Homarus gammarus | 1650.7196 | 0.2 | |
| Nephrops norvegicus | 1650.7188 | –0.2 | |
| Pacifastacus leniusculus | 1650.7117 | –4.5 | |
| Procambarus clarkii | 1650.7169 | –1.4 | |
| Thalassinidea | Callianassa californiensis | 1650.7199 | 0.4 |
| Achelata | Panulirus interruptus | 1650.7113‡ | –4.8 |
| Panulirus versicolor | 1650.7228 | 2.2 | |
| Scyllarides latus | 1650.7222 | 1.8 | |
| Anomura | Clibanarius vittatus | ND | |
| Lithodes maja | 1650.7202 | 0.6 | |
| Pachycheles rudis | ND | ||
| Petrolisthes cinctipes | 1650.7185 | –0.4 | |
| Petrolisthes eriomerus | 1650.7213 | 1.3 | |
| Pagurus acadianus | 1650.7242 | 3.0 | |
| Pagurus granosimanus | ND | ||
| Pagurus pollicaris | 1650.7266 | 4.5 | |
| Brachyura | Cancer antennarius | 1650.7165 | –1.6 |
| Cancer borealis | 1650.7118 | –4.5 | |
| Cancer gracilis | 1650.7146 | –2.8 | |
| Cancer irroratus | 1650.7213 | 1.3 | |
| Cancer magister | 1650.7208 | 1.0 | |
| Cancer productus | 1650.7262 | 4.3 | |
| Carcinus maenas | 1650.7099 | –5.6 | |
| Hemigrapsus nudus | ND | ||
| Lophopanopeus bellus | 1650.7135 | –3.5 | |
| Ovalipes ocellatus | 1650.7234 | 2.6 | |
| Pugettia gracilis | ND | ||
| Pugettia producta | 1650.7203‡ | 0.7 | |
| Scyra acutifrons | ND |
Peptide identity, SYWKQCAFNAVSCFamide (with disulfide bridge); ion identity, [M+H]+; Calculated m/z, m/z 1650.71918 (monoisotopic)
ND, not detected
Average of calibrated mass measurements. Spectra were calibrated using either poly(propylene glycol) or NFDEIDRSGFGFA (m/z 1474.6597) (Anomura and Brachyura) and APSGFLGMRamide (m/z 934.4927), pQDLDHVFLRFamide (m/z 1271.6531 and m/z 817.4832), and GYRKPPFNGSIFamide (m/z 1381.7738) or VYRKPPFNGSIFamide (m/z 1423.7845)
Mass measurement error in p.p.m.
Detected in the stomatogastric ganglion (STG), not commissural ganglion (CoG)
Physiological actions of SYWKQCAFNAVSCFamide in Homarus americanus
In decapod crustaceans, two common targets of neuropeptides, including circulating hormones as well as locally released transmitters, are the STNS, which generates the rhythmic motor patterns that drive the gastric mill and pyloric filter of the foregut, and the neurogenic heart. We thus examined the effects of SYWKQCAFNAVSCFamide (disulfide bridging between the two internal Cys residues) on the rhythmic output of the gastric mill and pyloric neural networks and on the activity of the whole heart preparation.
Effects on the stomatogastric motor patterns
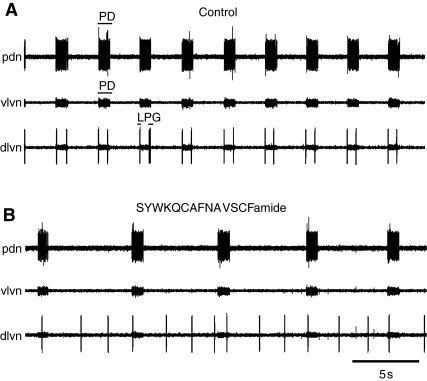
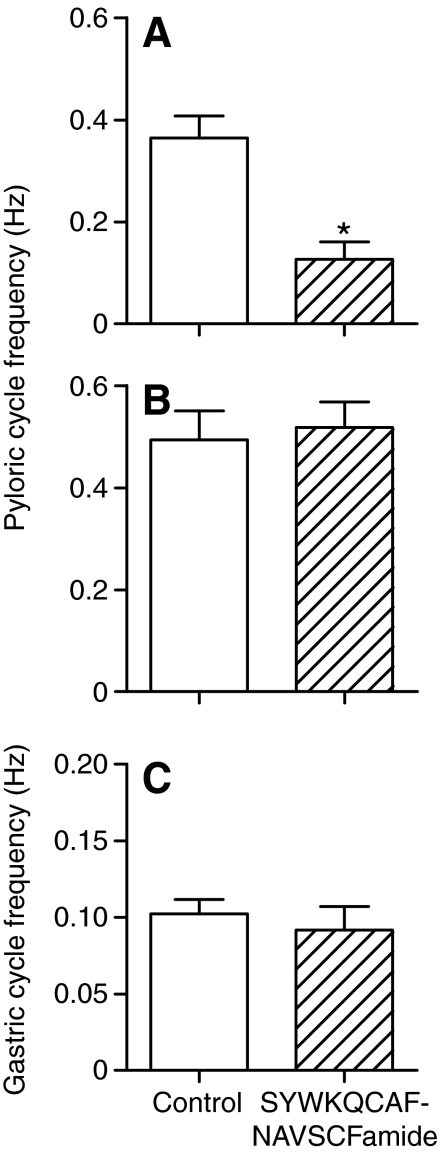
To examine the effects of SYWKQCAFNAVSCFamide on the pattern generators of the stomatogastric ganglion, we initially blocked action potential activity in the single input nerve to the STG, the stomatogastric nerve (stn). This results in a preparation in which other modulatory inputs have been removed, so that the effects of a single modulator on the underlying neurons and circuitry are more readily visible. Under these conditions in the Homarus STG, nearly all rhythmic activity except regular bursting in the electrically coupled pyloric dilator (PD) and anterior burster (AB) neurons was eliminated. These neurons continued to burst in regular and strong bursts of action potentials, as seen in extracellular recordings of PD neuron activity (Fig. 4A). In addition, the lateral posterior gastric (LPG) neurons fired a few action potentials with each PD neuron burst, as a result of the weak electrical coupling between these neurons (Kotecha, 2008). Superfusion of 10–6 mol l–1 SYWKQCAFNAVSCFamide over such a preparation caused a reliable decrease in PD neuron cycle frequency (from an average of 0.36±0.04 Hz to 0.13±0.03 Hz; paired t-test, P=0.015, N=4; Fig. 4, Fig. 5A). PD neuron burst duration remained unchanged (P>0.05, N=4). At the same time, firing in the LPG neurons became tonic rather than linked to the PD neuron bursts (Fig. 4), suggesting that the bursts in the PD neurons were less able to entrain firing in the LPG neurons.
Fig. 4.
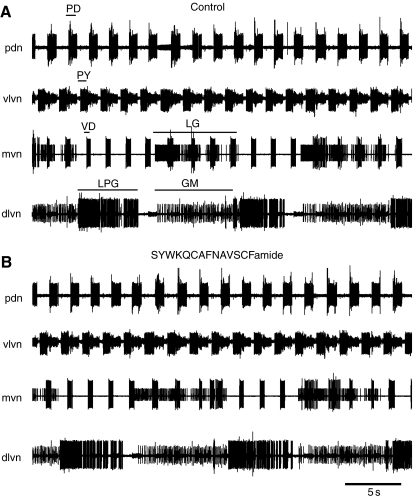
SYWKQCAFNAVSCFamide decreased the frequency of the pyloric motor pattern in preparations in which all modulatory input to the stomatogastric ganglion (STG) was eliminated by blocking conduction in the single input nerve to the STG, the stomatogastric nerve (stn). (A) In control saline, the pattern consisted of bursts in the pyloric dilator (PD) neurons (seen here on the pyloric dilator nerve (pdn) and the ventral lateral ventricular nerve (vlvn). The lateral posterior gastric (LPG) neurons [recorded on the dorsal lateral ventricular nerve (dlvn)] fired several spikes with each PD neuron burst. (B) During bath application of 10–6 mol l–1 SYWKQCAFNAVSCFamide, cycle frequency decreased, and firing in the LPG neurons became tonic rather than linked to the PD neuron bursts.
Fig. 5.
Bath application of 10–6 mol l–1 SYWKQCAFNAVSCFamide significantly decreased pyloric cycle frequency when conduction in the single input nerve to the stomatogastric ganglion, the stomatogastric nerve (stn), was blocked and cycle frequency was relatively low (average 0.34 Hz) (A), but had no effect on pyloric cycle frequency (B) or gastric cycle frequency (C) when the stn was intact and both patterns were spontaneously active.
Because modulators frequently show state-dependent effects, we also tested the effects of SYWKQCAFNAVSCFamide on the intact stomatogastric nervous system, in which other modulators are present, and both pyloric and gastric patterns were fully active. Interestingly, under these conditions, SYWKQCAFNAVSCFamide had no significant effect on either the pyloric or gastric motor patterns (Fig. 6). As shown in Fig. 5B,C, both gastric and pyloric frequency remained constant.
Fig. 6.
Bath application of 10–6 mol l–1 SYWKQCAFNAVSCFamide had no apparent effects on the activity of either the pyloric or gastric motor patterns when modulatory inputs from the anterior ganglia [commissural ganglia (CoGs) and oesophageal ganglion (OG)] were intact and the patterns were both active. (A) Control; (B) in the presence of SYWKQCAFNAVSCFamide. Pyloric pattern seen in the firing of the pyloric dilator (PD), pyloric (PY) and ventricular dilator (VD) neurons; gastric activity is recorded in the firing of the lateral gastric (LG), lateral posterior gastric (LPG) and gastric mill (GM) neurons. Activity was recorded on the pyloric dilator (pdn), ventral lateral ventricular (vlvn), medial ventricular (mvn) and dorsal lateral gastric (dlvn) nerves.
Effects on the heart
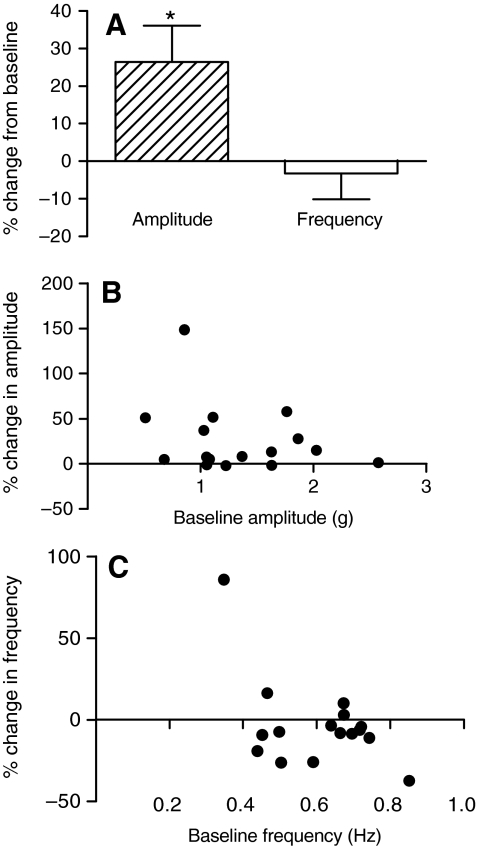
The isolated lobster heart preparation includes the cardiac ganglion, which generates the rhythmic bursts of action potentials that drive the rhythm, as well as the muscles that transform that activity into movement. Perfusion of 10–6 mol l–1 SYWKQCAFNAVSCFamide through this preparation caused an average increase in heartbeat amplitude of 26.5±9.6% (±s.e.m.) across all preparations tested, a value significantly greater than 0 (one sample two-tailed t-test against a hypothetical value of 0, P<0.05, N=16; Fig. 7A). However, perfusion of the peptide through the heart resulted in a clear and consistent increase in heartbeat amplitude in only 75% of preparations (12 of 16); it had relatively little effect on this parameter in others (four of 16, or 25% of preparations). To enable us to compare preparations with different baseline contraction parameters, we calculated the percentage change from control levels for hearts that showed noticeable changes in amplitude in response to SYWKQCAFNAVSCFamide perfusion separately from those that showed no obvious changes. Among the `high response' preparations, the increase in amplitude averaged 35.6±11.7%, a value that was significantly different from 0 (one sample two-tailed t-test against a hypothetical value of 0, P=0.01, N=12). Average change in the `low-response preparations' (N=4) was not significantly different from 0 (average change=–0.09±0.7%).
Fig. 7.
SYWKQCAFNAVSCFamide significantly increase the amplitude, but not the frequency, of spontaneous heartbeat in Homarus americanus. (A) Pooled data from 16 preparations show that SYWKQCAFNAVSCFamide significantly increased the amplitude of heart contractions (t-test, P=0.015). However, it had no consistent effect on heartbeat frequency. (B) The extent of increase was larger in preparations with initially low amplitudes, suggesting that the effect of the peptide is to some extent state dependent. (C) It is probable that the lack of significance for frequency change is due, at least in part, to a state-dependent effect of the peptide, where the rate of heartbeat was increased when the baseline frequency was low, but decreased when baseline rate was high.
One potential explanation for the differential responsiveness of different preparations is the starting amplitude, as there is a clear maximum level to which any muscle can contract. Thus, preparations in which baseline contraction amplitudes are very high have a more limited range over which they can increase. To determine whether starting amplitude was responsible for the differential responsiveness, we plotted percentage change as a function of initial contraction amplitude. As is shown in Fig. 7B, preparations with the highest initial amplitudes showed very little change; however, there was no clear correlation (r2<0.1 for linear or non-linear regressions) between these two parameters, suggesting that other factors are probably involved as well.
In contrast to amplitude, in which all preparations showed either an increased amplitude or were unaffected, heartbeat frequency was increased in a few preparations (greater than 10% change in three of 16, i.e. 18.8% of preparations), but decreased by five to 40% in others (10 of 16, i.e. 62.5%), with very little effect in the remaining few (three of 16). Because of the wide variety of responses, there was no significant change in heartbeat frequency when the effects on all preparations were averaged together.
To determine whether the changes in frequency were a function of the starting frequency, we plotted percentage change in frequency as a function of starting frequency, and found that initial frequency might account for some of the difference in response. The preparations with the lowest initial frequencies showed the largest increases, whereas preparations with the highest starting frequencies showed the largest decreases (Fig. 7C); the slope of a linear regression analysis was significantly different from 0 (P<0.05.) However, there was considerable variation between preparations; consequently, the correlation coefficient was very low (r2=0.25), suggesting that starting frequency alone does not account for the differences between responses to the peptide.
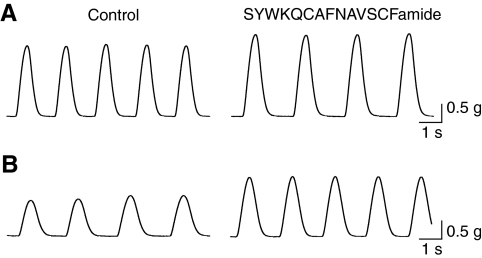
Interestingly, since most preparations responded with an increase in amplitude, but effects on frequency were mixed, there was not a one-to-one correlation between the changes in heartbeat amplitude and in frequency induced by SYWKQCAFNAVSCFamide. A number of preparations, for example, responded to the peptide with an increase in amplitude and a decrease in frequency (Fig. 8A), whereas others showed increases in both amplitude and frequency (Fig. 8B).
Fig. 8.
SYWKQCAFNAVSCFamide increased the amplitude and altered the frequency of heartbeat in Homarus americanus. Heartbeat was recorded with a force transducer in control saline and during perfusion with 10–6 mol l–1 SYWKQCAFNAVSCFamide. (A) In some preparations, the frequency of heartbeat decreased, whereas the amplitude of contractions increased in the presence of SYWKQCAFNAVSCFamide. (B) In other preparations, both the frequency and the amplitude of the heartbeat increased in the presence of the peptide.
DISCUSSION
C-type allatostatin-like peptides are not limited to holometabolous insects
In insects, three structurally distinct families of allatostatins are known: the A-ASTs, exhibiting a –YXFGLamide C-terminus, the B-ASTs, possessing a –WX6Wamide C-terminus, and the C-ASTs, typified by an unblocked –PISCF C-terminus (Stay and Tobe, 2007). In crustaceans, multiple isoforms of the A- and B-type peptides have been identified, with members of both families shown to serve neuro/myomodulatory roles. By contrast, C-ASTs have been identified only in insects, and there, only in holometabolous species, leading to the question of whether or not they are unique to members of this insect grouping (Stay and Tobe, 2007). Here, using functional genomics and mass spectrometry, we identified the C-AST-like peptide SYWKQCAFNAVSCFamide in the astacidean lobster H. americanus. Specifically, this peptide and the C-ASTs share the C-terminal sequence –QCXFN/IXV/ISCF (X indicating a variable residue), with, probably, disulfide bridging between the two internal Cys residues in both peptide groups. Moreover, both SYWKQCAFNAVSCFamide and the holometabolous insect C-type peptides are the only C-AST or C-AST-like peptide isoforms encoded within their respective precursors, and are the C-terminal most peptide encoded within their prepro-hormones. By contrast, the lobster peptide is not N-terminally blocked by pyroglutamine as are the C-ASTs; moreover, it is C-terminally amidated, whereas the known C-type isoforms all possess free C-termini.
It is interesting to note that, at least for species studied to date, the animals shown to possess the C-AST-like peptide SYWKQCAFNAVSCFamide are distinct from those that have been demonstrated to possess C-ASTs. For example, in the honeybee A. mellifera, where SYWKQCAFNAVSCFamide was first predicted (Hummon et al., 2006), no C-AST has been found, even though the complete genome of this species has been sequenced and annotated. Similarly, preliminary transcriptomics of insect ESTs suggest that SYWKQCAFNAVSCFamide is absent in other holometabolous species, whereas C-type isoforms are often predicted (A.E.C., unpublished). The converse appears to be the case for hemimetabolous insects, where C-ASTs are absent from the EST database, but SYWKQCAFNAVSCFamide is found, for example in the cricket Laupala kohalensis (accession nos. EH629527, EH633454, EH632422 and EH631480; A.E.C., unpublished) and the migratory locust Locusta migratoria (accession no. CO820847; A.E.C., unpublished). Moreover, although it does not possess a C-AST peptide, A. mellifera does appear to possess a receptor for C-type peptide, i.e. XP_396335. Collectively, these data raise the intriguing possibility that SYWKQCAFNAVSCFamide may be the honeybee and hemimetabolous insect equivalent of C-AST. Clearly additional studies will be needed to determine if SYWKQCAFNAVSCFamide is evolutionarily related to the C-ASTs, as well as to assess what effects the structural differences between SYWKQCAFNAVSCFamide and the C-ASTs may have on their relative bioactivities and/or potencies. However, structure–function studies done using M. sexta C-AST suggest that the presence of the disulfide bridge and its internal sequence are far more critical for maintaining the bioactivity of this peptide than are either a blocked N- or C-terminus (Kramer et al., 1991; Matthews et al., 2006), and it is the internal sequence and disulfide bridging that are shared by SYWKQCAFNAVSCFamide and the C-type isoforms.
Is SYWKQCAFNAVSCFamide a broadly conserved neuropeptide?
As stated earlier, the first description of SYWKQCAFNAVSCFamide was from the honeybee A. mellifera (Hummon et al., 2006). Recently, the same peptide was predicted via transcriptomics from the cladoceran crustacean D. pulex (Gard et al., 2009). Here, we report the in silico identification of SYWKQCAFNAVSCFamide from the astacidean lobster H. americanus, confirming the existence of it in this species via MALDI-FTMS. In addition, mass spectral analyses conducted on other decapods suggest that this peptide is broadly conserved in this taxon, being detected in members of five of its eight extant infraorders: Achelata, Astacidea, Thalassinidea, Anomura and Brachyura. The detection of the same peptide in the honeybee and from a wide variety of crustaceans shows that SYWKQCAFNAVSCFamide is a broadly conserved peptide, at least among arthropods. Further supporting this hypothesis are functional genomic data suggesting the presence of SYWKQCAFNAVSCFamide in the cricket L. kohalensis and the locust L. migratoria (see above), both hemimetabolous insects. In fact, an EST encoding SYWKQCAFNAVSCFamide was also identified from the Atlantic halibut Hippoglossus hippoglossus (accession no. FD698509; A.E.C., unpublished), a teleost chordate, suggesting that this peptide may be broadly conserved in a general sense.
A physiological role for SYWKQCAFNAVSCFamide
The apparent large-scale phylogenetic conservation of SYWKQCAFNAVSCFamide suggests that this peptide may play important, and possibly similar, physiological roles in a variety of animals. Here we have demonstrated one function for this peptide in crustaceans, namely the modulation of rhythmic pattern generation. This AST-C-like peptide modulates the activity of both the neurogenic heart and the pyloric pattern generated by the stomatogastric nervous system in H. americanus.
Numerous neuropeptides have been shown to modulate the output of the pyloric pattern generator, but the vast majority of these are excitatory (reviewed by Skiebe, 2001; Nusbaum and Beenhaker, 2002). Interestingly, however, previous studies in crabs have shown that two other allatostatins, an A-type and a B-type allatostatin, inhibit the activity of the pyloric motor pattern in the crab Cancer borealis (Skiebe and Schneider, 1994; Fu et al., 2007). In both of these cases, the effects of AST are state dependent: cycle frequency decreases substantially in weakly active preparations, but changes minimally in rapidly cycling preparations. Because all pyloric neurons except the pacemaker ensemble (i.e. the PD and AB neurons) cease firing when the stn is blocked in H. americanus, we were unable to examine the effects of SYWKQCAFNAVSCFamide over a range of activity levels, as was done in C. borealis. We did find, however, that it inhibited the pyloric motor pattern only when other modulatory inputs to the ganglion were blocked. Under these conditions, the pyloric frequency was relatively low and only the pacemaker neurons were active. In this respect, the effects of this structurally unrelated peptide are strikingly similar to those of the other ASTs in another decapod crustacean species. The fact that the inhibitory effects of SYWKQCAFNAVSCFamide were seen only when the pyloric pattern consisted solely of regular bursts in the pacemaker neurons suggests not only that the peptide exerts an inhibitory effect on the pacemaker neuron ensemble itself, but also that this inhibition can be overridden by other neuromodulators or the downstream effects of other modulators that are normally released from neurons in the more anterior ganglia (i.e. either the CoGs or the OG). No effect was seen on the gastric mill pattern, but this pattern is not active when inputs from the anterior ganglia are blocked, so we were unable to test the effects of the peptide under the conditions in which it affects the pyloric pattern.
In addition to effects on the pyloric motor pattern, we have shown that, in the lobster H. americanus, SYWKQCAFNAVSCFamide causes clear and dramatic increases in amplitude of heartbeat in a subset of preparations. Changes in heartbeat frequency were smaller and more variable. The factor or factors that determine the extent to which a given heart is sensitive to the peptide are less clear. Possible factors include stage of the molt cycle, age and exposure to stress. Our current data do not allow us to distinguish between these and other possibilities. Although the effects of the allatostatins on heart function have been examined in relatively few preparations, in all of those cases, they have been inhibitory. Thus, A-type ASTs inhibit the heart in the cockroach Blattella germanica (Vilaplana et al., 1999) and counter the cardioexcitatory actions of proctolin on the antennal heart in the cockroach Periplaneta americana (Hertel and Penzlin, 1992). In the decapod crustacean C. borealis, a B-type allatostatin dramatically inhibits the output of the isolated cardiac ganglion (Cruz-Bermúdez and Marder, 2007). By contrast, whereas SYWKQCAFNAVSCFamide does decrease heart rate in many preparations with high heart rates, it increases frequency in at least some preparations, notably those in which initial heart rate is very low, and it increases amplitude in virtually all preparations. One notable difference between the experiments reported here and those described in Cruz-Bermúdez and Marder (Cruz-Bermúdez and Marder, 2007) is that our experiments used whole hearts, whereas the effects of AST-B on Cancer were tested on the isolated cardiac ganglion. Thus, it is possible that the excitatory effects of SYWKQCAFNAVSCFamide require feedback from heart muscle tissue. Alternatively, because neither A- nor B-type AST has been tested in Homarus, it is possible that this reflects a difference between A- and B-ASTs versus SYWKQCAFNAVSCFamide or a difference between species. It should be noted that the largest excitatory effect of SYWKQCAFNAVSCFamide reported here was on the amplitude of contraction, a parameter that cannot be measured in the isolated cardiac ganglion.
Whether or not SYWKQCAFNAVSCFamide serves additional functions in crustaceans remains an open question. Likewise, the functional roles played by this peptide in other species remain to be determined. Clearly, as additional studies are conducted, it will be interesting to see what roles are played by SYWKQCAFNAVSCFamide, including whether or not this peptide is indeed allatostatic in the insects that do not possess C-ASTs.
LIST OF ABBREVIATIONS
- A-AST
A-type allatostatin
- B-AST
B-type allatostatin
- C-AST
C-type allatostatin
- AB
anterior burstor neuron
- CoG
commissural ganglion
- DHB
2,5-dihydroxybenzoic acid
- EST
expressed sequence tag
- LPG
lateral posterior gastric neuron
- OG
esophageal ganglion
- PD
pyloric dilator neuron
- PMC
posterior midgut caecum
- PO
pericardial organ
- SG
sinus gland
- STG
stomatogastric ganglion
- stn
stomatogastric nerve
- STNS
stomatogastric nervous system
This study was supported by grants from the National Center for Research Resources' INBRE Program (NIH P20 RR-016463), NSF (IBN 01140 and MRI-0116416), the Undergraduate Science Program of the Howard Hughes Medical Institute, the Paller Fund of Bowdoin College, and an MDIBL New Investigator Award (Salisbury Cove Research Fund provided by the Thomas H. Maren Foundation) to A.E.C. A.E.C. also acknowledges support from institutional funds provided by MDIBL. Deposited in PMC for release after 12 months.
References
- Abdel-Latief, M., Meyering-Vos, M. and Hoffmann, K. H. (2004). Type-A allatostatins from the fall armyworm, Spodoptera frugiperda: molecular cloning, expression and tissue-specific localization. Arch. Insect Biochem. Physiol. 56, 120-132. [DOI] [PubMed] [Google Scholar]
- Bendtsen, J. D., Nielsen, H., von Heijne, G. and Brunak, S. (2004). Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 340, 783-795. [DOI] [PubMed] [Google Scholar]
- Billimoria, C. P., DiCaprio, R. A., Birmingham, J. T., Abbott, L. F. and Marder, E. (2006). Neuromodulation of spike-timing precision in sensory neurons. J. Neurosci. 26, 5910-5919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birmingham, J. T., Billimoria, C. P., DeKlotz, T. R., Stewart, R. A. and Marder, E. (2003). Differential and history-dependent modulation of a stretch receptor in the stomatogastric system of the crab, Cancer borealis. J. Neurophysiol. 90, 3608-3616. [DOI] [PubMed] [Google Scholar]
- Bowser, P. R. and Tobe, S. S. (2007). Comparative genomic analysis of allatostatin-encoding (Ast) genes in Drosophila species and prediction of regulatory elements by phylogenetic footprinting. Peptides 28, 83-93. [DOI] [PubMed] [Google Scholar]
- Christie, A. E. (2008a). Neuropeptide discovery in Ixodoidea: an in silico investigation using publicly accessible expressed sequence tags. Gen. Comp. Endocrinol. 157, 174-185. [DOI] [PubMed] [Google Scholar]
- Christie, A. E. (2008b). In silico analyses of peptide paracrines/hormones in Aphidoidea. Gen. Comp. Endocrinol. 159, 67-79. [DOI] [PubMed] [Google Scholar]
- Christie, A. E., Sousa, G. L., Rus, S., Smith, C. M., Towle, D. W., Hartline, D. K. and Dickinson, P. S. (2008a). Identification of A-type allatostatins possessing - YXFGI/Vamide carboxy-termini from the nervous system of the copepod crustacean Calanus finmarchicus. Gen. Comp. Endocrinol. 155, 526-533. [DOI] [PubMed] [Google Scholar]
- Christie, A. E., Cashman, C. R., Brennan, H. R., Ma, M., Sousa, G. L., Li, L., Stemmler, E. A. and Dickinson, P. S. (2008b). Identification of putative crustacean neuropeptides using in silico analyses of publicly accessible expressed sequence tags. Gen. Comp. Endocrinol. 156, 246-264. [DOI] [PubMed] [Google Scholar]
- Christie, A. E., Ackerman, R. J., Barton, E. E., Stevens, J. S., Gabranski, E. R., Stemmler, E. A. and Dickinson, P. S. (2008c). SYWKQCAFNAVSCFamide: a broadly conserved crustacean C-type allatostatin. Program No. 30.13. 2008 Neuroscience Meeting Planner. Washington, DC: Society for Neuroscience, 2008. Online.
- Cruz-Bermúdez, N. D. and Marder, E. (2007). Multiple modulators act on the cardiac ganglion of the crab, Cancer borealis. J. Exp. Biol. 210, 2873-2884. [DOI] [PubMed] [Google Scholar]
- Dircksen, H., Skiebe, P., Abel, B., Agricola, H., Buchner, K., Muren, J. E. and Nassel, D. R. (1999). Structure, distribution, and biological activity of novel members of the allatostatin family in the crayfish Orconectes limosus. Peptides 20, 695-712. [DOI] [PubMed] [Google Scholar]
- Duve, H., Johnsen, A. H., Maestro, J. L., Scott, A. G., Jaros, P. P. and Thorpe, A. (1997). Isolation and identification of multiple neuropeptides of the allatostatin superfamily in the shore crab Carcinus maenas. Eur. J. Biochem. 250, 727-734. [DOI] [PubMed] [Google Scholar]
- Duve, H., Johnsen, A. H., Scott, A. G. and Thorpe, A. (2002). Allatostatins of the tiger prawn, Penaeus monodon (Crustacea: Penaeidea). Peptides 23, 1039-1051. [DOI] [PubMed] [Google Scholar]
- Fu, Q., Kutz, K. K., Schmidt, J. J., Hsu, Y. W., Messinger, D. I., Cain, S. D., de la Iglesia, H. O., Christie, A. E. and Li, L. (2005). Hormone complement of the Cancer productus sinus gland and pericardial organ: an anatomical and mass spectrometric investigation. J. Comp. Neurol. 493, 607-626. [DOI] [PubMed] [Google Scholar]
- Fu, Q., Tang, L. S., Marder, E. and Li, L. (2007). Mass spectrometric characterization and physiological actions of VPNDWAHFRGSWamide, a novel B type allatostatin in the crab, Cancer borealis. J. Neurochem. 101, 1099-1107. [DOI] [PubMed] [Google Scholar]
- Gard, A. L., Lenz, P. H., Shaw, J. R. and Christie, A. E. (2009). Identification of putative peptide paracrines/hormones in the water flea Daphnia pulex (Crustacea; Branchiopoda; Cladocera) using transcriptomics and immunohistochemistry. Gen. Comp. Endocrinol. 156, 246-264. [DOI] [PubMed] [Google Scholar]
- Hertel, W. and Penzlin, H. (1992). Function and modulation of the antennal heart of Periplaneta americana (L.). Acta Biol. Hung. 43, 113-125. [PubMed] [Google Scholar]
- Hummon, A. B., Richmond, T. A., Verleyen, P., Baggerman, G., Huybrechts, J., Ewing, M. A., Vierstraete, E., Rodriguez-Zas, S. L., Schoofs, L., Robinson, G. E. et al. (2006). From the genome to the proteome: uncovering peptides in the Apis brain. Science 314, 647-649. [DOI] [PubMed] [Google Scholar]
- Huybrechts, J., Nusbaum, M. P., Bosch, L. V., Baggerman, G., De Loof, A. and Schoofs, L. (2003). Neuropeptidomic analysis of the brain and thoracic ganglion from the Jonah crab, Cancer borealis. Biochem. Biophys. Res. Commun. 308, 535-344. [DOI] [PubMed] [Google Scholar]
- Jorge-Rivera, J. C. and Marder, E. (1997). Allatostatin decreases stomatogastric neuromuscular transmission in the crab Cancer borealis. J. Exp. Biol. 200, 2937-2946. [DOI] [PubMed] [Google Scholar]
- Jorge-Rivera, J. C., Sen, K., Birmingham, J. T., Abbott, L. F. and Marder, E. (1998). Temporal dynamics of convergent modulation at a crustacean neuromuscular junction. J. Neurophysiol. 80, 2559-2570. [DOI] [PubMed] [Google Scholar]
- Kotecha, V. R. (2008). Modulation of a fast neural rhythm by a slower rhythm occurs through electrical coupling in the lobster Homarus americanus. Bowdoin College Honors Thesis.
- Kramer, S. J., Toschi, A., Miller, C. A., Kataoka, H., Quistad, G. B., Li, J. P., Carney, R. L. and Schooley, D. A. (1991). Identification of an allatostatin from the tobacco hornworm Manduca sexta. Proc. Natl. Acad. Sci. USA 88, 9458-9462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause, E., Wenschuh, H. and Jungblut, P. R. (1999). The dominance of arginine-containing peptides in MALDI-derived tryptic mass fingerprints of proteins. Anal. Chem. 71, 4160-4165. [DOI] [PubMed] [Google Scholar]
- Kreissl, S., Weiss, T., Djokaj, S., Balezina, O. and Rathmayer, W. (1999). Allatostatin modulates skeletal muscle performance in crustaceans through pre- and postsynaptic effects. Eur. J. Neurosci. 11, 2519-2530. [DOI] [PubMed] [Google Scholar]
- Lenz, C., Williamson, M. and Grimmelikhuijzen, C. J. (2000). Molecular cloning and genomic organization of an allatostatin preprohormone from Drosophila melanogaster. Biochem. Biophys. Res. Commun. 273, 1126-1131. [DOI] [PubMed] [Google Scholar]
- Li, Y., Hernandez-Martinez, S., Fernandez, F., Mayoral, J. G., Topalis, P., Priestap, H., Perez, M., Navare, A. and Noriega, F. G. (2006). Biochemical, molecular, and functional characterization of PISCF-allatostatin, a regulator of juvenile hormone biosynthesis in the mosquito Aedes aegypti. J. Biol. Chem. 281, 34048-34055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenz, M. W., Kellner, R. and Hoffmann, K. H. (1995). A family of neuropeptides that inhibit juvenile hormone biosynthesis in the cricket, Gryllus bimaculatus. J. Biol. Chem. 270, 21103-21108. [DOI] [PubMed] [Google Scholar]
- Ma, M., Chen, R., Sousa, G. L., Bors, E. K., Kwiatkowski, M., Goiney, C. C., Goy, M. F., Christie, A. E. and Li, L. (2008). Mass spectral characterization of peptide transmitters/hormones in the nervous system and neuroendocrine organs of the American lobster Homarus americanus. Gen. Comp. Endocrinol. 156, 395-409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma, M., Bors, E. K., Dickinson, E. S., Kwiatkowski, M. A., Sousa, G. L., Henry, R. P., Smith, C. M., Towle, D. W., Christie, A. E. and Li, L. (2009). Characterization of the Carcinus maenas neuropeptidome using mass spectrometry and functional genomics. Gen. Comp. Endocrinol. (in press). [DOI] [PMC free article] [PubMed]
- Matthews, H. J., Audsley, N. and Weaver, R. J. (2006). Alanine substitutions and deletion analogues of Manduca sexta allatostatin: structure-activity relationship on the spontaneous contractions of the foregut of larval Lacanobia oleracea. J. Insect Physiol. 52, 128-135. [DOI] [PubMed] [Google Scholar]
- Meyering-Vos, M., Wu, X., Huang, J., Jindra, M., Hoffmann, K. H. and Sehnal, F. (2001). The allatostatin gene of the cricket Gryllus bimaculatus (Ensifera, Gryllidae). Mol. Cell. Endocrinol. 184, 103-114. [DOI] [PubMed] [Google Scholar]
- Nusbaum, M. P. and Beenhakker, M. P. (2002). A small-systems approach to motor pattern generation. Nature 417, 343-350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connor, P. B. and Costello, C. E. (2000). Internal calibration on adjacent samples (InCAS) with Fourier transform mass spectrometry. Anal. Chem. 72, 5881-5885. [DOI] [PubMed] [Google Scholar]
- Sheng, Z., Ma, L., Cao, M. X., Li, S. and Jiang, R. J. (2007). Biochemical and molecular characterization of allatotropin and allatostatin from the Eri silkworm, Samia cynthia ricini. Insect Biochem. Mol. Biol. 37, 90-96. [DOI] [PubMed] [Google Scholar]
- Skiebe, P. (2001). Neuropeptides are ubiquitous chemical mediators: using the stomatogastric nervous system as a model system. J. Exp. Biol. 204, 2035-2048. [DOI] [PubMed] [Google Scholar]
- Skiebe, P. and Schneider, H. (1994). Allatostatin peptides in the crab stomatogastric nervous system: inhibition of the pyloric motor pattern and distribution of allatostatin-like immunoreactivity. J. Exp. Biol. 194, 195-208. [DOI] [PubMed] [Google Scholar]
- Stay, B. and Tobe, S. S. (2007). The role of allatostatins in juvenile hormone synthesis in insects and crustaceans. Annu. Rev. Entomol. 52, 277-299. [DOI] [PubMed] [Google Scholar]
- Stemmler, E. A., Cashman, C. R., Messinger, D. I., Gardner, N. P., Dickinson, P. S. and Christie, A. E. (2007). High mass resolution direct-tissue MALDI-FTMS reveals broad conservation of three neuropeptides (APSGFLGMRamide, GYRKPPFNGSIFamide and pQDLDHVFLRFamide) across members of seven decapod crustaean infraorders. Peptides 28, 2104-2115. [DOI] [PubMed] [Google Scholar]
- Towle, D. W. and Smith, C. M. (2006). Gene discovery in Carcinus maenas and Homarus americanus via expressed sequence tags. Integr. Comp. Biol. 46, 912-918. [DOI] [PubMed] [Google Scholar]
- Veenstra, J. A. (2000). Mono- and dibasic proteolytic cleavage sites in insect neuroendocrine peptide precursors. Arch. Insect Biochem. Physiol. 43, 49-63. [DOI] [PubMed] [Google Scholar]
- Vilaplana, L., Maestro, J. L., Piulachs, M. and Bellés, X. (1999). Modulation of cardiac rhythm by allatostatins in the cockroach Blattella germanica (L.) (Dictyoptera, Blattellidae). J. Insect Physiol. 45, 1057-1064. [DOI] [PubMed] [Google Scholar]
- Wang, J., Meyering-Vos, M. and Hoffmann, K. H. (2004). Cloning and tissue-specific localization of cricket-type allatostatins from Gryllus bimaculatus. Mol. Cell. Endocrinol. 227, 41-51. [DOI] [PubMed] [Google Scholar]
- Weaver, R. J. and Audsley, N. (2008). Neuropeptides of the beetle, Tenebrio molitor identified using MALDI-TOF mass spectrometry and deduced sequences from the Tribolium castaneum genome. Peptides 29, 168-178. [DOI] [PubMed] [Google Scholar]
- Williamson, M., Lenz, C., Winther, A. M., Nässel, D. R. and Grimmelikhuijzen, C. J. (2001a). Molecular cloning, genomic organization, and expression of a B-type (cricket-type) allatostatin preprohormone from Drosophila melanogaster. Biochem. Biophys. Res. Commun. 281, 544-550. [DOI] [PubMed] [Google Scholar]
- Williamson, M., Lenz, C., Winther, A. M., Nässel, D. R. and Grimmelikhuijzen, C. J. (2001b). Molecular cloning, genomic organization, and expression of a C-type (Manduca sexta-type) allatostatin preprohormone from Drosophila melanogaster. Biochem. Biophys. Res. Commun. 282, 124-130. [DOI] [PubMed] [Google Scholar]
- Woodhead, A. P., Stay, B., Seidel, S. L., Khan, M. A. and Tobe, S. S. (1989). Primary structure of four allatostatins: neuropeptide inhibitors of juvenile hormone synthesis. Proc. Natl. Acad. Sci. USA 86, 5997-6001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuda-Kamatani, Y. and Yasuda, A. (2006). Characteristic expression patterns of allatostatin-like peptide, FMRFamide-related peptide, orcokinin, tachykinin-related peptide, and SIFamide in the olfactory system of crayfish Procambarus clarkii. J. Comp. Neurol. 496, 135-147. [DOI] [PubMed] [Google Scholar]
- Yin, G. L., Yang, J. S., Cao, J. X. and Yang, W. J. (2006). Molecular cloning and characterization of FGLamide allatostatin gene from the prawn, Macrobrachium rosenbergii. Peptides 27, 1241-1250. [DOI] [PubMed] [Google Scholar]