Abstract
The Thr54 allele of the intestinal fatty acid-binding protein Ala54Thr functional polymorphism (FABP2) is associated with increased fat oxidation and insulin resistance. We determined the cross-sectional associations of the FABP2 gene with lipid levels and insulin resistance in 2148 participants who completed the year 20 exam of the Coronary Artery Risk Development in Young Adults (CARDIA) study. No significant difference in total cholesterol, low-density or high-density lipoprotein cholesterol, triglycerides, high-density lipoprotein cholesterol to total cholesterol ratio, or HOMA-IR was found between FABP2 genotypes. However, in the presence of a high saturated fat diet (≥ 53.2 grams per day, the 90th percentile for the population), the AA/AG genotypes (carriers of the Thr54 allele) of FABP2 had statistically significantly higher levels of log(HOMA-IR) (p=0.006) and a lower high-density lipoprotein cholesterol to total cholesterol ratio (p=0.03), and borderline statistically significantly higher levels of total cholesterol, low-density lipoprotein cholesterol, and log(triglycerides) (p-values = 0.08, 0.07, and 0.05, respectively) compared to those with the GG genotype (Ala54 homozygotes). Lipid levels and log(HOMA-IR) did not vary by genotype with saturated fat intake below 53.2 grams per day. Limiting dietary saturated fat intake may be particularly important among carriers of the A allele of FABP2.
Keywords: intestinal fatty acid-binding protein, FABP2, lipids, insulin resistance, saturated fat
INTRODUCTION
The intracellular fatty acid-binding proteins (FABPs) belong to a multigene family with nearly twenty identified members that form 14–15 kDa proteins thought to participate in the uptake, intracellular metabolism, and/or transport of long-chain fatty acids. The fatty acid-binding protein 2 (FABP2) gene codes for intestinal FABP, an abundant cytosolic intracellular lipid-binding protein in small intestine epithelial cells playing a role in fat absorption and transport. The G to A mutation at codon 54 of FABP2 results in a substitution of Thr54 for Ala54. This polymorphism has been associated with dyslipidemia and insulin resistance after a high-fat meal in sedentary nondiabetic men and women. FABP2 Thr54 carriers have lower glucose tolerance and lower insulin action than do Ala54-homozygous persons.
Although some investigators have suggested that the Ala54Thr polymorphism specifically influences small intestinal lipid absorption without modifying glucose uptake or metabolism, thus influencing postprandial lipid metabolism and plasma levels of lipids, the majority of reports have linked the polymorphism to impaired glucose tolerance and Type 2 diabetes. Additional evidence of a gene-by-environmental interaction with dietary fat intake led us to explore the association of this common polymorphism in the population-based Coronary Artery Risk Development in Young Adults (CARDIA) study in order to examine dietary influence on lipid levels and insulin resistance in young men and women who are free of clinically manifested cardiovascular disease and who have a low prevalence of diabetes.
SUBJECTS AND METHODS
Study Population
The CARDIA study is a prospective study of cardiovascular risk factors in black and white men and women aged 18–30 years at baseline. The recruitment of participants, study design, and methods have been previously described. Briefly, 5115 young adults were recruited during 1985–1986 from 4 U.S. cities: Birmingham, AL, Chicago, IL, Minneapolis, MN, and Oakland, CA. The cohort was balanced within each center on gender, race, age (18–24 and 25–30), and education (high school or less and more than high school). Follow-up examinations occurred at years 2 (1987–1988), 5 (1990–1991), 7 (1992–1993), 10 (1995–1996), 15 (2000–2001), and 20 (2005–2006), with response rates of 90%, 86%, 81%, 79%, 74%, and 72%, respectively. The CARDIA study was approved by institutional review boards of the coordinating center and the 4 participating field centers, and informed consent was obtained from participants at every examination.
Laboratory Measurements
The FABP2 polymorphism was genotyped using the TaqMan assay (Applied Biosystems, Foster City, CA) as previously described. Primer and probes are available from the authors upon request. Polymorphism genotyping in the CARDIA study adheres to a rigorous quality control (QC) program, which includes barcode identification of samples, robotic sample handling, and blind replicate genotype assessment on 5% of the total sample.
For this study, only lipid levels and HOMA-IR measured at the year 20 exam were used. Participants were asked to fast for 12 hours before the examination and blood draw. Lipoprotein assays were conducted by Northwest Lipids Research Laboratory (Seattle, WA). Enzymatic methods were used to measure total cholesterol (TC) and triglyceride (TG) levels. High-density lipoprotein cholesterol (HDL-c) levels were determined enzymatically after dextran sulfate-Mg2+ precipitation of other lipoproteins. Low-density lipoprotein cholesterol (LDL-c) levels were estimated with the Friedewald formula for individuals with TG levels < 400 mg/dL.
Fasting insulin and fasting glucose assays were performed at Linco Research, Inc (St. Charles, MO). An immunoassay technique was used to measure serum insulin. Serum glucose was measured using the hexokinase method with a Cobas Mira Plus chemistry analyzer (Roche Diagnostic Systems). We calculated HOMA-IRby dividing the product of fasting insulin (μU/mL) and fasting glucose (mmol/L) by 22.5.
Additional Measurements
During the year 20 exam, each participant completed an interviewer-administered diet history questionnaire, designed specifically for the CARDIA study. Participants were asked to report details about foods consumed in the past 28 days, including the amount and method used to prepare the food. Total kilocalories and grams of saturated fat consumed per day were calculated from responses to the diet history questionnaire using the nutrient database (version 36) developed by the Nutrition Coordinating Center at the University of Minnesota.
Waist circumference was measured to the nearest 0.5 cm at the smallest abdominal girth. Standardized questionnaires were used to determine years of education and smoking status. Milliliters of ethanol per day was calculated from the quantity and type of alcoholic beverages consumed. Amount of moderate and heavy physical activity during the past year was assessed using an interviewer-administered questionnaire. A score of 100 exercise units (EU) is roughly equivalent to 2–3 hours per week of vigorous physical activity for 6 months of the year.
Statistical Analysis
All analyses were conducted using SAS version 8.2 (SAS Institute, Cary, NC). Of the 5115 participants originally enrolled in CARDIA, 3549 participants completed the year 20 exam. Those not attending the year 20 exam differed significantly from those who were present in regard to demographic, and some, but not all, major cardiovascular risk factors. For example, those who dropped out before year 20 were less educated, and more likely to be male, black, current smokers, and have greater alcohol intake at baseline than those who completed the year 20 exam. After excluding participants with TG >400 mg/dL (n=33), those on cholesterol medications at year 20 (n=314), 1 participant who had a saturated fat intake of 363.2 grams per day, and those who were not genotyped for FABP2 or who did not consent for DNA analysis (n=504), there were 2697 participants in our dataset for analysis.
Agreement of the FABP2 genotype frequencies with Hardy Weinberg equilibrium expectations was tested using a chi-square goodness-of-fit test. The FABP2 polymorphism was tested for allelic dominance or co-dominance by comparing statistical significance of least-squares means of lipid values between genotypes. Based on the results of the dominance or co-dominance evaluations, the AA and AG genotypes were grouped together for comparison with the GG genotype.
Participant characteristics at year 20 by FABP2 genotype group (AA/AG vs. GG) were compared using chi-squared tests for categorical variables and t-tests for continuous variables. General linear models were used to estimate the mean differences in lipid levels and HOMA-IR by genotype group and saturated fat intake after adjustment for race, sex, center, educational attainment and the following covariates at year 20: age, waist circumference, alcohol intake, smoking status, and daily total calorie intake. We chose to use waist instead of body mass index (BMI) because waist circumference is more strongly related to insulin resistance. However, we also conducted identical analyses using BMI instead of waist circumference and results were not materially different. Saturated fat intake was dichotomized at the 90th percentile (53.2 grams per day) to assess the additive interaction on 1 degree of freedom between high versus low/normal saturated fat intake and genotype on levels of TC, LDL-c, HDL-c, TG, HDL/TC ratio, and HOMA-IR. The 90th percentile of saturated fat intake was chosen as a cut-point in order to minimize misclassification of individuals with low/normal saturated fat intake into the high saturated fat intake category. TG and HOMA-IR were natural log transformed in the general linear models and interaction analyses because of non-normal distributions. We additionally tested for effect modification by race. We did not find evidence that the association between FABP2 genotype and saturated fat intake with lipid levels or HOMA-IR differed in blacks and whites. Therefore, all models were run in the entire cohort adjusting for race.
RESULTS
After exclusions, 2697 participants remained in our dataset for analysis. However, due to missing values for the covariates in our linear regression models, the sample size was 2148 for the models examining the association of FABP2 genotype and saturated fat intake on lipid levels and 2145 for the model examining this effect on HOMA-IR. The biggest contributors to missing values were from questionnaire data on education (n=185), and daily calories and saturated fat intake (n=299).
Table 1 shows the genotype frequencies in all participants, as well as separately by race group. Individuals with the AA and AG genotypes had similar mean lipid and HOMA-IR levels (data not shown). Thus, the AA and AG genotypes were pooled and compared to the GG genotype in further analysis. The FABP2 gene was in Hardy-Weinberg equilibrium in all participants and within race groups. Participant characteristics at year 20 by FABP2 genotype are shown in Table 2. Most participant characteristics did not vary by genotype. However, there was a greater percentage of individuals with a graduate school education in the AA/AG genotype group compared to the GG group.
Table 1.
FABP2 Genotype Frequencies by Race Group, The CARDIA Study, 2005–2006
| AA | AG | GG | |
|---|---|---|---|
| All | 163 (6.0) | 982 (36.4) | 1552 (57.6) |
| Blacks | 78 (6.2) | 444 (35.2) | 738 (58.6) |
| Whites | 85 (5.9) | 538 (37.4) | 814 (56.7) |
Values are N (%)
Table 2.
Participant Characteristics at Year 20 by FABP2 genotype, The CARDIA Study, 2005–2006
| AA/AG (n=1145) | GG (n=1552) | P-value | |
|---|---|---|---|
| Race | |||
| Black | 522 (45.6) | 738 (47.6) | 0.32 |
| White | 623 (54.4) | 814 (52.5) | |
| Sex | |||
| Male | 465 (40.6) | 680 (59.4) | 0.24 |
| Female | 665 (42.8) | 887 (57.2) | |
| Age | 45.2 ± 0.1 | 45.0 ± 0.1 | 0.32 |
| Center | |||
| Birmingham | 270 (23.6) | 338 (21.8) | 0.62 |
| Chicago | 268 (23.4) | 355 (22.9) | |
| Minneapolis | 300 (26.2) | 418 (26.9) | |
| Oakland | 307 (26.8) | 441 (28.4) | |
| Education | |||
| ≤ High School | 222 (20.9) | 298 (20.5) | 0.01 |
| Some College | 289 (27.2) | 468 (32.3) | |
| College | 286 (27.0) | 390 (26.9) | |
| Graduate School | 264 (24.9) | 295 (20.3) | |
| BMI, kg/m2 | |||
| <25 | 327 (28.6) | 489 (31.7) | 0.23 |
| 25 to <30 | 389 (34.1) | 504 (32.7) | |
| ≥ 30 | 426 (37.3) | 548 (35.6) | |
| Waist, cm | 91.1 ± 0.5 | 90.8 ± 0.4 | 0.66 |
| Smoking Status | |||
| Never | 703 (62.1) | 963 (62.6) | 0.39 |
| Former | 228 (20.1) | 281 (18.3) | |
| Current | 201 (17.8) | 294 (19.1) | |
| Alcohol, ml/day | 10.4 ± 0.6 | 11.0 ± 0.6 | 0.46 |
| Physical Activity, EU | 339.9 ± 8.3 | 345.0 ± 7.1 | 0.64 |
| Saturated Fat, g/day | 31.3 ± 0.6 | 31.1 ± 0.5 | 0.83 |
| Energy Intake, kcal/day | 2415.7 ± 41.0 | 2389.0 ± 35.1 | 0.62 |
| Total Cholesterol, mg/dL | 186.7 ± 1.0 | 185.4 ± 0.9 | 0.32 |
| LDL Cholesterol, mg/dL | 111.1 ± 0.9 | 110.2 ± 0.8 | 0.49 |
| HDL Cholesterol, mg/dL | 55.3 ± 0.5 | 54.6 ± 0.4 | 0.33 |
| Triglycerides, mg/dL | 101.8 ± 1.8 | 102.7 ± 1.5 | 0.73 |
| HDL/TC ratio | 0.3 ± 0.003 | 0.3 ± 0.003 | 0.74 |
| Diabetes | |||
| No | 1061 (93.8) | 1464 (94.8) | 0.29 |
| Yes | 70 (6.2) | 81 (5.2) | |
| HOMA-IR | 4.0 ± 0.1 | 3.9 ± 0.1 | 0.46 |
Values are N (%) for categorical variables, and mean ± SEM for continuous variables
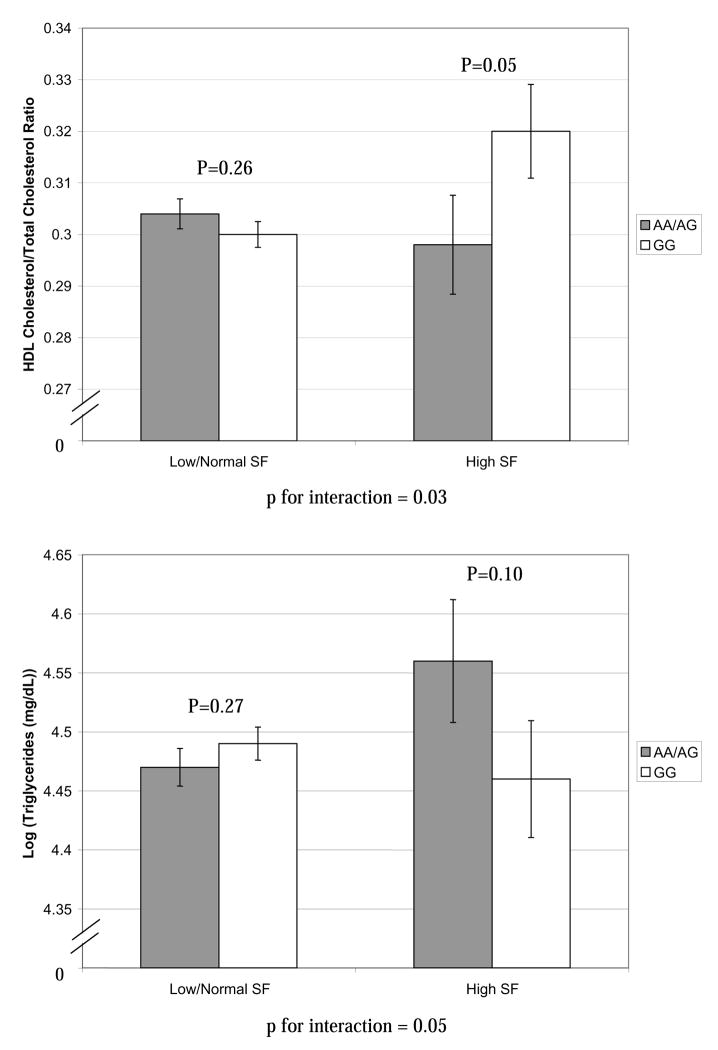
There were no significant differences in mean levels of TC, LDL-c, HDL-c, TG, HDL/TC ratio, or HOMA-IR by FABP2 genotype group (Table 2). Likewise, there was no association of daily saturated fat intake with levels of TC, LDL-c, HDL-c, TG, HDL/TC ratio, or HOMA-IR (data not shown). However, a significant FABP2 gene by saturated fat intake interaction on log(HOMA-IR) was found when saturated fat was modeled continuously (p=0.03). In addition, a borderline significant interaction between the FABP2 gene and saturated fat intake on TC, LDL-c, and HDL/TC ratio was found (p-values = 0.06, 0.05, and 0.06, respectively). When saturated fat intake was stratified at the 90th percentile (corresponding to an intake of 53.2 grams/day), the effect of high saturated fat intake on the association of FABP2 genotype with HDL/TC ratio and log(HOMA-IR) was significant (p-values = 0.03 and 0.006, respectively) compared to low/normal saturated fat intake (Figure 1). Borderline statistically significant genotype-by-saturated fat interactions on TC, LDL-c, and log(TG) were also found when saturated fat was stratified at the 90th percentile (p-values = 0.08, 0.07, and 0.05, respectively). Those with the A allele of the FABP2 gene and saturated fat intake at or above the 90th percentile had marginally higher TC, LDL-c, log(TG) and log(HOMA-IR) levels and a lower HDL/TC ratio than those with the GG genotype and similar saturated fat intake. Individuals with saturated fat intake below the 90th percentile had similar levels of lipids and log(HOMA-IR) regardless of genotype. Results were comparable when saturated fat intake was stratified at the 75th percentile (data not shown).
Figure 1. Multivariable-Adjusted* Plots of Association between Genotype and Endpoints Stratified on the 90th Percentile of Saturated Fat Intake, The CARDIA Study, 2005–2006.
*Models are adjusted for race, sex, center, educational attainment, and the following at year 20: age, waist circumference, alcohol intake, smoking status, and daily total caloric intake.
SF refers to daily saturated fat intake.
N=2148 for the lipid models. Among those with saturated fat intake above the 90th percentile, 98 were AA/AG and 121 were GG for FABP2. Among those with low/normal saturated fat intake, 813 were AA/AG and 1116 were GG.
N=2145 for the HOMA-IR model. Among those with saturated fat intake above the 90th percentile, 97 were AA/AG and 121 were GG for FABP2. Among those with low/normal saturated fat intake, 813 were AA/AG and 1114 were GG.
DISCUSSION
In this cross-sectional analysis at year 20 of the CARDIA exam, we found evidence that the association of FABP2 genotype with TC, LDL-c, HDL/TC ratio, log(TG), and log(HOMA-IR) varied by level of saturated fat consumed. Among individuals with a high saturated fat intake (above the 90th percentile or ≥ 53.2 grams per day), the AA/AG genotypes of FABP2 had marginally higher levels of TC, LDL-c, log(TG), and log(HOMA-IR) and a lower HDL/TC ratio than those with the GG genotype. Lipid levels and log(HOMA-IR) did not vary by genotype with low to moderate saturated fat intake below 53.2 grams per day. This is analogous to saying that there is no difference in lipid levels or log(HOMA-IR) among individuals with the GG genotype regardless of saturated fat intake, whereas those who carry the A allele of FABP2 have marginally higher levels of TC, LDL-c, log(TG), and log(HOMA-IR) and a lower HDL/TC ratio with high saturated fat intake compared to low/normal saturated fat intake.
Potential mechanisms behind these findings have been reported. In vitro experiments showed that a recombinant Thr54-containing protein had a 2-fold greater affinity for long chain fatty acids than the recombinant Ala54-containing protein, and that transformed human colonic carcinoma cell line (Caco-2) cells expressing Thr54 transport long-chain fatty acids secrete triglycerides to a greater degree than Caco-2 cells expressing Ala54. Additionally, Thr54-containing fetal jejunal explants had increased synthesis and secretion of TG and increased secretion of chylomicrons compared to Ala54 explants. Furthermore, the Thr54 allele of the FABP2 gene was associated with higher LDL-c and apolipoprotein B in men and higher TC and LDL-c in women compared to the Ala54 allele in the Framingham Offspring Study. Postprandial TG concentrations were 3-fold higher among women carriers of the Thr54 allele compared to the wild type, but this association was not found in men. Although there is conflicting literature that suggest the Ala54Thr polymorphism is not associated with lipid metabolism, these findings suggest that those with the A allele of the FABP2 gene may have increased absorption and processing of dietary fatty acids possibly resulting in increased plasma lipid levels.
In addition to increased metabolism of long-chain fatty acids and increased plasma lipid levels, results from some studies suggest that the FABP2 gene is associated with insulin resistance. For example, in the Framingham Offspring Study, women carriers of the Thr54 allele had higher 2-hour postchallenge insulin levels than women homozygous for the Ala54 allele. Additionally, among older (aged 40 to 65 years) normoglycemic Japanese men, the FABP2 Thr/Thr genotype was associated with higher fasting plasma glucose compared to men carrying the Ala54 allele. Among Pima Indians, carriers of the Thr54 allele had higher mean fat oxidation rates, higher fasting plasma insulin and higher insulin resistance in response to oral glucose overload than those homozygous for the Ala54 allele. In a review by Weiss, et al, approximately half of the studies reviewed found an association of the Thr54 allele with lower glucose tolerance or insulin action but findings in Japanese that used HOMA-IR found an association of increased insulin resistance with the Thr54 allele in only 1 of 4 studies. Although the results of previous studies are conflicting, an increase in absorption and processing of fatty acids may increase fat oxidation, and thus increased insulin resistance among individuals with the Thr54 allele of FABP2 is biologically plausible.
Our study did not find evidence of an association between FABP2 genotype (AA/AG vs. GG) alone on levels of TC, LDL-c, HDL-c, TG, HDL/TC ratio, or HOMA-IR. However, we did find evidence of interactions between saturated fat intake and FABP2 on TC, LDL-c, HDL/TC ratio, log(TG), and log(HOMA-IR) levels. Among those with high saturated fat intake, at or above 53.2 grams per day, individuals with the AA and AG genotypes (carriers of the Thr54 allele) had marginally higher TC, LDL-c, log(TG) and log(HOMA-IR) levels and a lower HDL/TC ratio than those with the GG genotype (Ala54 homozygotes), while there was no difference among those consuming a lower fat diet. Our results are consistent with some studies that examined the effect of the Thr54 allele of the FABP2 gene and dietary fat intake on plasma lipid levels and insulin resistance. Among individuals following a low-fat diet in another study, fasting insulin and fasting glucose were higher in Thr54 carriers and, in response to a high fat meal, postprandial lipid oxidation rates were higher in Thr54 carriers compared to Ala54 homozygotes, suggesting that augmented lipid absorption among Thr54 carriers may cause insulin resistance. In a study examining 3 different diets, 1) a high saturated fat diet, 2) a Mediterranean diet high in monounsaturated fat, and 3) a low-fat and high-carbohydrate diet, individuals with the Thr54 allele of FABP2 had less insulin sensitivity and increased free fatty acid concentrations after consumption of the high saturated fat diet only. Another study found a gene-by-diet interaction that is somewhat inconsistent with the previous study where Thr54 carriers had increased chylomicron cholesterol after consumption of olive oil only and not butter or safflower oil compared to Ala54 homozygotes.
Our study analyzed data from a large community-based ethnically diverse cohort. However, there are some limitations in our study. First, food frequency questionnaires generally result in underestimations of nutrient intake, so individual levels of saturated fat intake among participants are likely underestimated. However, the rank ordering of fat intake is appropriate for cross-sectional dietary analysis. Further, dichotomizing the saturated fat intake at the 90th percentile likely captured very high intake of saturated fat as a comparison with to low/moderate saturated fat intake. Lastly, because these data are cross-sectional, results must be viewed as hypothesis generating only, and caution should be used in making causal interpretations.
In conclusion, there was no effect of the FABP2 polymorphism alone on levels of TC, LDL-c, HDL-c, TG, HDL/TC ratio, or HOMA-IR in this large population-based study. However, in the presence of a high saturated fat diet, the AA/AG genotypes of FABP2 showed evidence of a borderline association with adverse lipids and increased insulin resistance compared to the GG genotype. The FABP2 polymorphism is common, with approximately 43% of our study population carrying at least one copy of the A allele. According to our findings, given that individuals who are carriers of the A allele of FABP2 with high saturated fat intake have somewhat more adverse lipid levels and insulin resistance, it may be beneficial for these individuals to follow a low-fat diet. Therefore, limiting dietary intake of saturated fat to less than 7% of total calories, as recommended by the American Heart Association, may be particularly important among the large proportion of the population who are carriers of the A allele of FABP2.
Acknowledgments
Work on this manuscript was supported (or partially supported) by contracts: University of Alabama at Birmingham, Coordinating Center, N01-HC-95095; University of Alabama at Birmingham, Field Center, N01-HC-48047; University of Minnesota, Field Center and Diet Reading Center (Year 20 Exam), N01-HC-48048; Northwestern University, Field Center, N01-HC-48049; Kaiser Foundation Research Institute, N01-HC-48050; University of California, Irvine, Echocardiography Reading Center (Year 5 & 10), N01-HC-45134; Harbor-UCLA Research Education Institute, Computed Tomography Reading Center (Year 15 Exam), N01-HC-05187; Wake Forest University (Year 20 Exam), N01-HC-45205; New England Medical Center (Year 20 Exam), N01-HC-45204 from the National Heart, Lung and Blood Institute.
Footnotes
The authors have no conflicts of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Li Y, Fisher E, Klapper M, Boeing H, Pfeiffer A, Hampe J, et al. Association between functional FABP2 promoter haplotype and type 2 diabetes. Hormone & Metabolic Research. 2006 May;38(5):300–307. doi: 10.1055/s-2006-925405. [DOI] [PubMed] [Google Scholar]
- 2.Weiss EP, Brandauer J, Kulaputana O, Ghiu IA, Wohn CR, Phares DA, et al. FABP2 Ala54Thr genotype is associated with glucoregulatory function and lipid oxidation after a high-fat meal in sedentary nondiabetic men and women. Am J Clin Nutr. 2007 January 1;85(1):102–108. doi: 10.1093/ajcn/85.1.102. [DOI] [PubMed] [Google Scholar]
- 3.Levy E, Menard D, Delvin E, Stan S, Mitchell G, Lambert M, et al. The polymorphism at codon 54 of the FABP2 gene increases fat absorption in human intestinal explants. J Biol Chem. 2001 Oct 26;276(43):39679–39684. doi: 10.1074/jbc.M105713200. [DOI] [PubMed] [Google Scholar]
- 4.Galluzzi JR, Cupples LA, Meigs JB, Wilson PW, Schaefer EJ, Ordovas JM, et al. Association of the Ala54-Thr polymorphism in the intestinal fatty acid-binding protein with 2-h postchallenge insulin levels in the Framingham Offspring Study. Diabetes Care. 2001 Jul;24(7):1161–1166. doi: 10.2337/diacare.24.7.1161. [DOI] [PubMed] [Google Scholar]
- 5.Marin C, Perez-Jimenez F, Gomez P, Delgado J, Paniagua JA, Lozano A, et al. The Ala54Thr polymorphism of the fatty acid-binding protein 2 gene is associated with a change in insulin sensitivity after a change in the type of dietary fat. Am J Clin Nutr. 2005 Jul;82(1):196–200. doi: 10.1093/ajcn.82.1.196. [DOI] [PubMed] [Google Scholar]
- 6.Friedman GD, Cutter GR, Donahue RP, Hughes GH, Hulley SB, Jacobs DR, Jr, et al. CARDIA: study design, recruitment, and some characteristics of the examined subjects. J Clin Epidemiol. 1988;41(11):1105–1116. doi: 10.1016/0895-4356(88)90080-7. [DOI] [PubMed] [Google Scholar]
- 7.Fornage M, Doris PA. Single-Nucleotide Polymorphism Genotyping for Association Studies. In: Fennell JP, Baker AH, editors. Hypertension Methods and Protocols. Totowa, NJ: Humana Press; 2004. pp. 159–172. [Google Scholar]
- 8.Warnick GR. Enzymatic methods for quantification of lipoprotein lipids. Meth Enzymol. 1986;129:101–123. doi: 10.1016/0076-6879(86)29064-3. [DOI] [PubMed] [Google Scholar]
- 9.Warnick GR, Benderson J, Albers JJ. Dextran sulfate-Mg2+ precipitation procedure for quantitation of high-density-lipoprotein cholesterol. Clin Chem. 1982 Jun;28(6):1379–1388. [PubMed] [Google Scholar]
- 10.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, Without Use of the Preparative Ultracentrifuge. Clin Chem. 1972 June 1;18(6):499–502. [PubMed] [Google Scholar]
- 11.Herbert V, Lau KS, Gottlieb CW, Bleicher SJ. Coated charcoal immunoassay of insulin. Journal of Clinical Endocrinology & Metabolism. 1965 Oct;25(10):1375–1384. doi: 10.1210/jcem-25-10-1375. [DOI] [PubMed] [Google Scholar]
- 12.McDonald A, Van Horn L, Slattery M, Hilner J, Bragg C, Caan B, et al. The CARDIA dietary history: development, implementation, and evaluation. J Am Diet Assoc. 1991 Sep;91(9):1104–1112. [PubMed] [Google Scholar]
- 13.Jacobs DRJ, Hahn LP, Haskell WL, Pirie P, Sidney S. Validity and Reliability of Short Physical Activity History: Cardia and the Minnesota Heart Health Program. J Cardiopulm Rehabil. 1989 November;9(11):448–459. doi: 10.1097/00008483-198911000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sidney S, Jacobs DR, Jr, Haskell WL, Armstrong MA, Dimicco A, Oberman A, et al. Comparison of Two Methods of Assessing Physical Activity in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Am J Epidemiol; Am J Epidemiol. 1991 June 15;133(12):1231–1245. doi: 10.1093/oxfordjournals.aje.a115835. [DOI] [PubMed] [Google Scholar]
- 15.Sweetser D, Birkenmeier E, Klisak I, Zollman S, Sparkes R, Mohandas T, et al. The human and rodent intestinal fatty acid binding protein genes. A comparative analysis of their structure, expression, and linkage relationships. J Biol Chem. 1987 November 25;262(33):16060–16071. [PubMed] [Google Scholar]
- 16.Cohn S, Simon T, Roth K, Birkenmeier E, Gordon J. Use of transgenic mice to map cis-acting elements in the intestinal fatty acid binding protein gene (Fabpi) that control its cell lineage- specific and regional patterns of expression along the duodenal-colonic and crypt-villus axes of the gut epithelium. J Cell Biol. 1992 October 1;119(1):27–44. doi: 10.1083/jcb.119.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baier LJ, Sacchettini JC, Knowler WC, Eads J, Paolisso G, Tataranni PA, et al. An amino acid substitution in the human intestinal fatty acid binding protein is associated with increased fatty acid binding, increased fat oxidation, and insulin resistance. J Clin Invest. 1995 Mar;95(3):1281–1287. doi: 10.1172/JCI117778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Baier LJ, Bogardus C, Sacchettini JC. A Polymorphism in the Human Intestinal Fatty Acid Binding Protein Alters Fatty Acid Transport across Caco-2 Cells. J Biol Chem. 1996 May 3;271(18):10892–10896. doi: 10.1074/jbc.271.18.10892. [DOI] [PubMed] [Google Scholar]
- 19.Ribalta J, Halkes CJ, Salazar J, Masana L, Cabezas MC. Additive effects of the PPARgamma, APOE, and FABP-2 genes in increasing daylong triglycerides of normolipidemic women to concentrations comparable to those in men. Clin Chem. 2005 May;51(5):864–871. doi: 10.1373/clinchem.2004.044347. [DOI] [PubMed] [Google Scholar]
- 20.Tahvanainen E, Molin M, Vainio S, Tiret L, Nicaud V, Farinaro E, et al. Intestinal fatty acid binding protein polymorphism at codon 54 is not associated with postprandial responses to fat and glucose tolerance tests in healthy young Europeans. Results from EARS II participants. Atherosclerosis. 2000 Oct;152(2):317–325. doi: 10.1016/s0021-9150(99)00488-8. [DOI] [PubMed] [Google Scholar]
- 21.Ishii T, Hirose H, Kawai T, Hayashi K, Maruyama H, Saito I, et al. Effects of intestinal fatty acid-binding protein gene Ala54Thr polymorphism and beta3-adrenergic receptor gene Trp64Arg polymorphism on insulin resistance and fasting plasma glucose in young to older Japanese men. Metabolism: Clinical & Experimental. 2001 Nov;50(11):1301–1307. doi: 10.1053/meta.2001.27228. [DOI] [PubMed] [Google Scholar]
- 22.Weiss EP, Brown MD, Shuldiner AR, Hagberg JM. Fatty acid binding protein-2 gene variants and insulin resistance: gene and gene-environment interaction effects. Physiol Genomics. 2002 September 3;10(3):145–157. doi: 10.1152/physiolgenomics.00070.2001. [DOI] [PubMed] [Google Scholar]
- 23.Dworatzek PD, Hegele RA, Wolever TM. Postprandial lipemia in subjects with the threonine 54 variant of the fatty acid-binding protein 2 gene is dependent on the type of fat ingested. Am J Clin Nutr. 2004 June 1;79(6):1110–1117. doi: 10.1093/ajcn/79.6.1110. [DOI] [PubMed] [Google Scholar]
- 24.Lichtenstein AH, Appel LJ, Brands M, Carnethon M, Daniels S, Franch HA, et al. Diet and Lifestyle Recommendations Revision 2006: A Scientific Statement From the American Heart Association Nutrition Committee. Circulation. 2006 July 4;114(1):82–96. doi: 10.1161/CIRCULATIONAHA.106.176158. [DOI] [PubMed] [Google Scholar]