Abstract
In the mouse Pax6 function is critical in a dose-dependent manner for proper eye development. Pax6 contiguous gene deletions were shown to be homozygous lethal at an early embryonic stage. Heterozygotes express belly spotting and extreme microphthalmia. The eye phenotype is more severe than in heterozygous Pax6 intragenic null mutants, raising the possibility that deletions are functionally different from intragenic null mutations or that a region distinct from Pax6 included in the deletions affects eye phenotype. We recovered and identified the exact regions deleted in three new Pax6 deletions. All are homozygous lethal at an early embryonic stage. None express belly spotting. One expresses extreme microphthalmia and two express the milder eye phenotype similar to Pax6 intragenic null mutants. Analysis of Pax6 expression levels and the major isoforms excluded the hypothesis that the deletions expressing extreme microphthalmia are directly due to the action of Pax6 and functionally different from intragenic null mutations. A region distinct from Pax6 containing eight genes was identified for belly spotting. A second region containing one gene (Rcn1) was identified for the extreme microphthalmia phenotype. Rcn1 is a Ca+2-binding protein, resident in the endoplasmic reticulum, participates in the secretory pathway and expressed in the eye. Our results suggest that deletion of Rcn1 directly or indirectly contributes to the eye phenotype in Pax6 contiguous gene deletions.
CONTIGUOUS gene deletions account for a significant portion of human genetic syndromes. The application of fluorescence in situ hybridization (FISH) cytogenetics and array comparative genome hybridization (array-CGH) technologies have enabled more accurate localization of deletion breakpoints. This deletion information combined with the annotation of the human genome structure provides critical information to identify genes responsible for particular phenotypes associated with a syndrome. For example, deletions of the 11p11p12 and 11p13 regions on the short arm of human chromosome (Chr) 11 have been identified in the Potocki–Shaffer syndrome (Shaffer et al. 1993; Bartsch et al. 1996; Potocki and Shaffer 1996) and the Wilm's tumor- aniridia- genitourinary abnormalities- mental retardation (WAGR) syndrome (Riccardi et al. 1978; Francke et al. 1979; Hittner et al. 1979; Fryns et al. 1981), respectively. Deletion analyses were important in identifying genes associated with clinical features of the syndromes: EXT2 for multiple exostoses and ALX4 for parietal foramina in Potocki–Shaffer syndrome (Ligon et al. 1998; Wu et al. 2000; Wakui et al. 2005), WT1 for Wilm's tumor, and PAX6 for aniridia in WAGR syndrome (van Heyningen et al. 1985; Glaser et al. 1986, 1992; Fantes et al. 1992). Deletion analyses have also defined the extent of the deleted region in patients with combined Potocki–Shaffer and WAGR syndromes (McGaughran et al. 1995; Brémond-Gignac et al. 2005) as well as microdeletions 3′ to PAX6, which prevent expression of PAX6 and cause aniridia (Lauderdale et al. 2000; D'elia et al. 2007; Davis et al. 2008).
The mouse Chr 2 region homologous to the human WAGR region contains the genes Wt1, Rcn1, Pax6, and Elp4. An extensive allelic series at Pax6 has been identified (Bult et al. 2008). Heterozygote Pax6 intragenic null mutants express microphthalmia, iris anomalies, corneal opacities, lens opacities, and lens-corneal adhesions. Homozygotes are anophthalmic and die shortly after birth (Roberts 1967; Hogan et al. 1986). Five deletions in the region have been identified: Pax6Sey-Dey, Pax6Sey-H, Pax6Sey-2H, Pax6Sey-3H, Pax6Sey-4H of which two, Pax6Sey-H (Hogan et al. 1986; Kent et al. 1997; Kleinjan et al. 2002; Webb et al. 2008) and Pax6Sey-Dey (Theiler et al. 1978; Hogan et al. 1987; Glaser et al. 1990), have been well characterized. Heterozygotes for both deletions express belly spotting and a more extreme eye phenotype than that observed for heterozygotes of intragenic Pax6 null mutations. Homozygotes for both deletions are lethal at an early embryonic stage.
We were particularly interested in the extreme eye phenotype associated with the Pax6 deletions and considered two alternative hypotheses. Either Pax6 deletions are functionally different from Pax6 intragenic null mutations or deletion of a region linked to but distinct from the Pax6 structural gene affects the eye phenotype.
In the present study we identify three new deletions encompassing the Pax6 region of the mouse. They have been assigned the mutant allele symbols Del(2)Pax611Neu/1Neu, Del(2)Pax612Neu/2Neu, and Del(2)Pax613Neu/3Neu and will be referred to throughout this publication as Pax611Neu, Pax612Neu, and Pax613Neu, respectively. All three deletions are homozygous lethal at an early embryonic stage. The deletions differentiate for the extent of the eye abnormality expressed: Pax611Neu heterozygotes express extreme microphthalmia similar to that observed in the Pax6Sey-Dey and Pax6Sey-H deletions. Pax612Neu and Pax613Neu heterozygotes express the milder eye abnormality seen in heterozygous intragenic null mutants. For all three deletions, heterozygotes do not express belly spotting. Genetic, phenotypic, and molecular characterization of the deletions allowed us to identify regions associated with the array of phenotypes in these contiguous gene deletions.
MATERIALS AND METHODS
Mutations, animals, and mapping:
The original Pax611Neu and Pax613Neu mutants were found in our breeding colonies. The original Pax612Neu mutant was recovered in a mutagenesis experiment. Ophthalmological examinations were done as previously described (Favor 1983). Congenic C3H/HeJ mutant lines were constructed prior to initiating the studies. The mapping of the mutations followed our standard laboratory protocol (Favor et al. 1997). For timed pregnancies, females were mated and checked daily for the presence of a vaginal plug. The day at which a vaginal plug was observed was defined as day 0 p.c. (E0). In matings to generate offspring, females were checked daily for newborn litters and the day of birth was defined as postnatal day 0 (P0). Animals were bred and maintained in our animal facilities according to the German law for the protection of animals. All inbred strain C3H/HeJ and C57BL/6El animals used in the present study were obtained from breeding colonies maintained by the Department of Animal Resources at Neuherberg.
Histology, gross embryo morphology, and slit lamp photography:
Pregnant females were killed by cervical dislocation. Embryos were freed from placentae and embryonic membranes in room temperature PBS, phenotyped under a dissecting microscope (MZ APO; Leica, Bensheim, Germany), and photographed. Postnatal day 1 (P1) mice were killed by decapitation and phenotyped after dissecting away the skin overlying the eyes. P21 mice were phenotyped by slit lamp examination and killed by CO2 asphyxiation. Embryos and heads of P1 or P21 mice were fixed in 10% buffered formalin. The heads from P21 mice were demineralized in EDTA. Fixed materials were embedded in paraffin, and serially sectioned (coronal) at 5 μm. Sections were stained with hematoxylin and eosin, and evaluated by light microscopy (Axioplan; Carl Zeiss, Hallbergmoos, Germany). Digital photos were acquired (Axiocam and Axiovision; Carl Zeiss, Hallbergmoos, Germany) and imported into Adobe Photoshop CS (Adobe Systems, Unterschleissheim, Germany).
P35 mice were anesthetized with 137 mg ketamine and 6.6 mg xylazine per kg body weight and quickly photographed with a slit lamp microscope (Zeiss SL 120) equipped with a compact video camera. Images were captured in Axiovision (Zeiss) and imported into Adobe Photoshop CS. After photography ophthalmic salve (Regepithel, Alcon) was applied to the eyes of the anesthetized mice to prevent eye injury due to dehydration and the animals were caged individually until fully recuperated.
Segregation analysis of embryos:
Pregnant females were killed as above between days 14 and 16 p.c. (E14 and E16 stages) of pregnancy. The entire uterus was removed, opened, and the uterine contents classified for live embryos, dead implants (implantation site with an obvious placenta, extra-embryonic membranes, and necrotic embryonic tissue), and decidua (resorption sites consisting of the remnants of the decidual reaction tissue due to implantation but subsequent early embryonic death). The live embryos were freed from the placentae and embryonic membranes and phenotyped.
Body weight and gross eye morphology:
Eye morphology was assessed as previously described (Favor et al. 2001, 2008). P35 heterozygous mutant and wild-type littermates were ophthalmologically examined by slit lamp microscopy and categorized for the degree of lens/corneal opacity and extreme microphthalmia. The animals were weighed and killed by cervical dislocation. Eyes were enucleated, washed in room temperature PBS, blotted dry on filter paper, and weighed. Data were statistically analyzed by linear mixed model ANOVA employing SAS software release 9.1 (Cary, NC). Differences in group means were assessed by applying the F-test for the contrast derived from the linear model.
Deletion analyses at Pax6, MIT-microsatellite markers, and SNP sites:
Pax611Neu and Pax612Neu heterozygotes were mated to Pax69Neu and Pax64Neu heterozygotes. Pax613Neu heterozygotes were mated to Pax63Neu heterozygotes. Pregnant females were prepared as above. E15 Pax611Neu/Pax69Neu, Pax611Neu/Pax64Neu, Pax612Neu/Pax69Neu, Pax612Neu/Pax64Neu, and Pax613Neu/Pax63Neu compound heterozygotes were identified as anophthalmic embryos and liver tissue samples were snap frozen on dry ice for genomic DNA extractions as above. The Pax69Neu allele is a 7-bp deletion in the 5′ region of the Pax6 gene, the Pax64Neu allele is a base-pair substitution in the 3′ region of the Pax6 gene and the Pax63Neu allele is a 1-bp insert in the 5′ region of the Pax6 gene (Favor et al. 2001). The regions containing the Pax69Neu, Pax64Neu, and the Pax63Neu mutant sites were sequenced as previously described (Favor et al. 2001) in the compound heterozygotes. A region was shown to be deleted if, in the compound heterozygote, the sequence corresponded to the Pax69Neu, Pax64Neu, or the Pax63Neu sequence and not to a heterozygous sequence containing the wild-type and the mutant alleles.
Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes were created with either a wild-type Chr 2 from C3H/HeJ or C57BL/6El. Genomic DNA was extracted from liver samples as above from the six genotype constructs as well as the inbred strains C3H/HeJ and C57BL/6El. Animals were genotyped for MIT-microsatellite markers and SNP sites that are polymorphic between strains C3H/HeJ and C57BL/6El. An MIT-microsatellite or SNP site was determined to be not deleted when both MIT-microsatellite alleles or both SNP alleles were observed in the mutant heterozygotes carrying the wild-type Chr 2 from strain C57BL/6El. An MIT-microsatellite or SNP site was determined to be deleted when only a single microsatellite allele or a single SNP allele was observed in heterozygotes for both wild-type Chr 2 constructs, and the allele observed corresponded to the allele carried by the wild-type strain chromosome.
Deletion analysis by array-CGH:
Genomic DNA was extracted from liver samples of P35 Pax611Neu −/+, Pax612Neu −/+, and Pax6 +/+ mice as above. Genomic DNA from Pax6Sey-Dey −/+ was purchased from The Jackson Laboratory. Genomic DNA from Pax6Sey-H −/+ mice was kindly provided by Sally Cross (MRC, Human Genetics Unit, Edinburgh, UK). A set of 836 BAC clones covering Chr 2 from position 3 Mb to 181 Mb was analyzed for copy number variations in the deletion heterozygotes. For details see supporting information, File S1.
Deletion analysis by FISH cytogenetics:
E15 Pax611Neu −/+, Pax612Neu −/+, and Pax6 +/+ embryos were obtained from pregnant females as outlined above. Embryos were killed by decapitation, skin samples dissected and minced in the presence of trypsin/EDTA, and cells isolated by filtration through a 70-μm BD Falcon cell strainer (BD Biosciences, Munich, Germany). Cells were cultured for 3 days in GIBCO Dulbecco's modified Eagle's medium (Invitrogen, Karlsruhe, Germany) supplemented with 10% fetal calf serum after which chromosome preparations were made according to standard cytogenetic techniques. Continuous series of overlapping BACs were selected for the regions surrounding the putative deletion breakpoints and were used as probes for FISH cytogenetic analyses of the deletion-bearing chromosomes. For details see File S1.
Localization of the deletion breakpoints by DNA walking or sequencing across the deletions:
The proximal and distal breakpoints of the Pax611Neu, Pax612Neu, and Pax613Neu deletions were more precisely localized by an analysis of SNP or insert/deletion sites polymorphic between strains C3H/HeJ and C57BL/6 (File S1and Table S4, Table S5, Table S6, Table S7, Table S8, Table S9, and Table S10). We sequenced across the Pax611Neu deletion using genomic DNA from a Pax611Neu heterozygote as substrate, with the Seegene DNA walking Speedup premix kit (BioCat, Heidelberg, Germany) according to the manufacturer's protocol. The first, second and third genomic-specific primers were all within the nondeleted region defined by the BAC RP23-8C14: first specific primer, AGCCTGGCATCGTCACACTG; second specific primer, TGTGAAGGTGTGGAGAGTTGGAGG; third specific primer, TCAGTGTTCCAAGGAGGGCTGT. The chimeric sequence jumped from the sequence defined by the BAC RP23-8C14 to a distal region defined by the BAC RP23-431C3. The results were confirmed by sequencing across the presumed deletion using genome specific primers, i.e., the third specific primer from the region defined by the BAC RP23-8C14 as above, and a genome specific primer from the sequence defined by the BAC RP23-431C3 (TGCTGCAGACGTGCCAAAGAAC).
On the basis of the analysis of polymorphic sites of the Pax612Neu and the Pax613Neu deletions the adjacent proximal and distal nondeleted regions were identified. A group of genome-specific primers within each of these regions was designed, and long-range PCR was carried out to amplify across the deletions using genomic DNA from Pax612Neu or Pax613Neu heterozygotes as substrates with the Expand Long Range dNTPack (Roche Diagnostics, Mannheim, Germany) according to the manufacturer's protocol. Specific amplification products were isolated and used as substrates for sequencing.
Analysis of gene transcript levels by real-time PCR:
P1 mice were killed by decapitation. Eyes were enucleated and snap frozen on dry ice. RNA was extracted with the RNeasy kit (QIAGEN, Hilden, Germany) and concentrations adjusted to 0.25 μg/μl. Reverse transcription was carried out from 1μg of RNA as substrate using random hexamere primers and SuperScriptII polymerase (Invitrogen, Karlsruhe, Germany), following the protocol as recommended by the manufacturer. Quantitative real-time RT–PCR was done on a TaqMan 770 sequence detection system (Applied Biosystems, Foster City, CA) using the following probes: Pax6 (Mm00443072_m1), Elp4 (Mm_00517859_m1), Rcn1 (Mm_00485644_m1), and Tbp (Mm_00446973_m1) and TaqMan Universal PCR master mix (all reagents, Applied Biosystems). The Pax6 probe assays a 56-bp amplicon that spans its exon 3–exon 4 junction. The Elp4 probe assays a 101-bp amplicon spanning its exon 4–exon 5 junction. The Rcn1 probe assays a 119-bp amplicon that spans its exon 4–exon 5 junction.
Calibration curves of the real-time RT–PCR for each gene were generated using eight dilution steps (covering five orders of magnitude) of a whole-head cDNA pool from an E15 Pax6 +/+ embryo. Each dilution was measured twice and the resulting CT values were linearly fitted against the log10 of the dilution ratios.
The CT values using the cDNA samples from the mutant and wild-type eyes were measured in triplicates for each gene and each individual animal, and the relative expression values normalized to Tbp (as a housekeeping gene) and to the total-head cDNA pool using the calibration curves. Group means were compared with the Student's t-test employing the software available in Excel.
Following TaqMan real-time RT–PCR the resulting reaction products were electrophoretically separated on an agarose gel to confirm a single PCR product of the expected size.
Sequencing:
Primers (Table S11) were purchased from Metabion International AG (Martinsried, Germany) or synthesized in house. The PCR products were electrophoretically separated on 1% agarose gels, extracted with a QIAquick gel extraction kit (QIAGEN), and used as templates for sequencing in both directions with a Taq Dye-Deoxy terminator cycle sequencing kit on an ABI 3730 DNA sequencer (Applied Biosystems). BAC alignment and inspection of sequencing results of the regions utilized the Ensembl database build 44.
RESULTS
Eye morphology, belly spotting, and mapping:
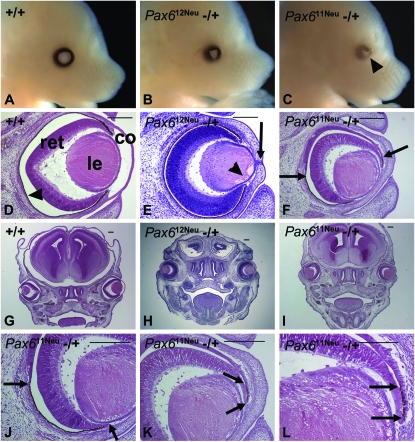
We compared the eye abnormalities associated with the Pax611Neu and Pax612Neu mutations in E15 embryos (Figure 1). Pax612Neu heterozygotes expressed microphthalmia with a triangular-shaped pupil (gross morphology) with a thickened cornea, lens-corneal adhesions, persistence of epithelial cells in the cornea, and the absence of the anterior chamber (histology). Pax611Neu heterozygotes expressed an extreme eye phenotype, not typical for Pax6 intragenic mutations, and comparable to the phenotype observed in the Pax6Sey-Dey and Pax6Sey-H deletions (Theiler et al. 1978; Hogan et al. 1986; Curto et al. 2007): microphthalmia, iris coloboma, and reduced iris pigmentation (gross morphology) with thickened cornea, lens-cornea adhesion, absence of the anterior chamber, posterior coloboma, and the orientation of the eyes was rotated such that the medial-lateral eye axis was at a 45° angle to the dorsal-ventral axis of the head (histology). However, at this embryonic stage the eye size was not extremely reduced.
Figure 1.—
Eye morphology and histology in E15 embryos. (A–C) Gross morphology. (A) Pax6 +/+ with well-developed eye. (B) Pax612Neu −/+ with the typical eye phenotype associated with Pax6 null mutations; microphthalmia and triangular shaped pupil. Iris pigmentation is normal. (C) Pax611Neu −/+ expressing microphthalmia, reduced iris pigmentation and iris coloboma (arrowhead). (D–F) Eye histology. (D) Pax6 +/+ with well-developed cornea (co), lens (le), retina (ret), and an intact retinal pigmented epithelium (arrowhead). There is a distinct anterior chamber separating the cornea and the anterior surface of the lens. (E) Pax612Neu −/+ with a thickened cornea, adhesion of the lens to the cornea resulting in the absence of an anterior chamber, remnants of epithelial cells in the cornea (arrow) and vacuoles in the anterior region of the lens (arrowhead). The retinal pigmented epithelium is normal. (F) Pax611Neu −/+ with a thickened cornea, adhesion of the lens to the cornea, and absence of the anterior chamber. Posterior coloboma is present, indicated by an interruption of the retinal pigmented epithelium in the region between the arrows. The orientation of the eye is rotated ∼45° ventrally. (G–I) Head overview documenting the eye orientation. (G) Pax6 +/+ and (H) Pax612Neu −/+ in which the medial-lateral eye axes are perpendicular to the dorsal-ventral axis of the head. (I) Pax611Neu −/+ in which the medial-lateral axes of both eyes are orientated at a 45° angle to the dorsal-ventral axis of the head. (J–L) Higher magnification of F. (J) The extent of the retinal pigmented epithelium in the ventral region is indicated by the arrows. (K and L) The retinal pigmented epithelium in the dorsal region is indicated by the arrows. Bars in D–K, 200 μm; bar in L, 100 μm.
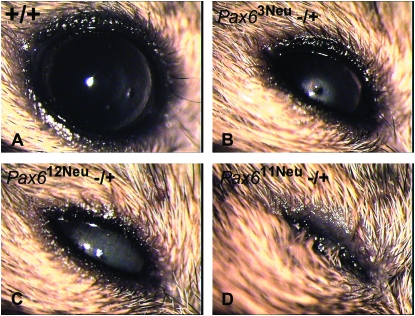
At P35 the differences in the eye phenotypes observed in the Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes were more extreme (Figure 2, Table 1). Pax611Neu heterozygotes were clearly associated with extreme microphthalmia. By contrast, the eye phenotype in Pax612Neu and Pax613Neu heterozygotes was similar to that of heterozygote intragenic Pax6 null mutations (Favor et al. 2001) with a median eye opacity of 75%. For all three mutant lines the eye weight in heterozygotes was significantly lower than that of wild-type littermates: Pax611Neu: −/+, 4.61 mg ± 0.15, n = 125; +/+, 18.27 mg ± 0.16, n = 120; t279 = 61.97, P < 0.0001. Pax612Neu: −/+, 14.59 mg ± 0.20, n = 78; +/+, 18.08 mg ± 0.21, n = 68, t279 = 12.20, P < 0.0001. Pax613Neu: −/+, 15.25 mg ± 0.20, n = 76; +/+, 18.92 mg ± 0.18, n = 94; t279 = 13.76, P < 0.0001. The eye weight of Pax611Neu heterozygotes was significantly less than the eye weights in Pax612Neu (t279 = 40.06, P < 0.0001) and Pax613Neu (t279 = 42.34, P < 0.0001) heterozygotes. Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes expressed a slight and significant reduction in body weight as compared to their wild-type littermates. However, there were no significant differences in body weight among the Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes, indicating that the extreme microphthalmia observed in Pax611Neu heterozygotes is not due to a reduction in general growth (data not shown).
Figure 2.—
Slit lamp microscopy documenting eye phenotypes in P35 mice. (A) Pax6 +/+. (B) Pax63Neu −/+, an intragenic null mutation expressing microphthalmia, a central opacity, lens-corneal adhesion and corneal opacity. (C) Pax612Neu −/+ expressing microphthalmia and total lens opacity. The degree of eye abnormality is similar to that observed in Pax63Neu heterozygotes. (D) Pax611Neu −/+ expressing extreme microphthalmia. All eyes were photographed at 32× magnification.
TABLE 1.
Degree of lens/corneal opacity in eyes of P35 Pax611Neu −/+, Pax612Neu −/+, Pax613Neu −/+, and +/+ littermates
| Phenotype class
|
|||||||
|---|---|---|---|---|---|---|---|
| Line | Genotype | 0% | 25% | 50% | 75% | 100% | Extreme microphthalmia |
| Pax611Neu | +/+ | 119 | 0 | 0 | 1 | 0 | 0 |
| Pax611Neu | −/+ | 0 | 0 | 1 | 3 | 21 | 101 |
| Pax612Neu | +/+ | 70 | 1 | 3 | 0 | 0 | 0 |
| Pax612Neu | −/+ | 0 | 10 | 11 | 31 | 36 | 0 |
| Pax613Neu | +/+ | 94 | 0 | 0 | 0 | 0 | 0 |
| Pax613Neu | −/+ | 0 | 6 | 30 | 29 | 11 | 0 |
Since Pax6 function is also critical for brain morphogenesis (Schmahl et al. 1993), we evaluated brain morphology in Pax611Neu −/+, Pax612Neu −/+, Pax63Neu −/+, and Pax6 +/+ P1 and P21 mice. Heterozygotes of all three Pax6 mutations expressed hypoplasia of the telencephalic frontal area, increased diameters of the ventricular and subventricular zones, and reduced diameter of the marginal zone in the dorsal pallial region of the forebrain. However, there were no differences in the degree of abnormality expressed in Pax611Neu heterozygotes as compared to the phenotypes expressed by Pax612Neu or Pax63Neu heterozygotes (data not shown).
Heterozygotes were examined at weaning for the presence of belly spotting: 542 Pax611Neu −/+, 256 Pax612Neu −/+, and 381 Pax613Neu −/+. No animals expressed belly spotting. All mutations mapped to Chr 2 with the locus order (frequency of recombinants between adjacent loci in parentheses) from the combined results: D2Mit249–(4/308)-Mut-(62/308)-agouti.
Segregation analyses in embryonic stages and complementation tests among Pax611Neu, Pax612Neu, Pax613Neu, and Pax6 intragenic mutant lines:
On the basis of genomic position and the eye phenotype associated with the mutations, we hypothesized Pax6 to be the affected gene. We established inter se matings of heterozygotes in the Pax611Neu, Pax612Neu, and Pax613Neu presumed Pax6 mutant lines to generate homozygotes for sequence analysis. Anophthalmic homozygous Pax6 null mutants are easily identified and survive to the perinatal stage. However, from the inter se matings in all three mutant lines no anophthalmic embryos were recovered. Only wild type and embryos expressing microphthalmia were observed in an ∼1:2 ratio (Pax611Neu χ2 = 0.56, 0.50 > P > 0.10; Pax612Neu χ2 = 0.15, 0.90 > P > 0.50; Pax613Neu χ2 = 0.02, 0.975 > P > 0.90), and there was an increase in the number of decidua (Table 2). Thus we hypothesized the mutations to be homozygous lethal at an early postimplantation stage. To determine if the Pax6 gene was affected, we crossed Pax611Neu, Pax612Neu, or Pax613Neu heterozygotes with heterozygotes for Pax6 intragenic mutations. In these complementation tests with Pax6, embryos with the typical anophthalmic phenotype were observed (Table 2), which indicates that (a) the Pax611Neu, Pax612Neu, and Pax613Neu mutations do not complement the Pax6 intragenic null mutations for the homozygous anophthalmia phenotype, and (b) the intragenic Pax6 null mutations complement the Pax611Neu, Pax612Neu, and Pax613Neu mutations for early embryonic lethality. Finally, we crossed Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes with each other. We observed wild-type and microphthalmic embryos in an ∼1:2 ratio (Pax611Neu × Pax612Neu χ2 = 0.11, 0.90 > P > 0.50; Pax611Neu × Pax613Neu χ2 = 0.21, 0.90 > P > 0.50; Pax612Neu × Pax613Neu χ2 = 0.01, 0.975 > P > 0.90). There were no anophthalmic embryos and there was an increase in the number of decidua (Table 2). Taken together, the results suggest that the Pax611Neu, Pax612Neu, and Pax613Neu mutations are multilocus deletions affecting Pax6 and a linked gene or genes responsible for early embryonic lethality.
TABLE 2.
Segregation analysis in E14–E16 embryos in crosses of Pax6 mutant heterozygotes
| Implantation sites
|
Phenotype classes of live embryos
|
||||||
|---|---|---|---|---|---|---|---|
| Mating | Litters (n) | Live | Dead | Decidua | Wild type | Microphthalmia | Anophthalmia |
| Pax611Neu−/+ × Pax611Neu−/+ | 14 | 89 | 2 | 45 | 33 | 56 | 0 |
| Pax612Neu−/+ × Pax612Neu−/+ | 8 | 52 | 0 | 16 | 16 | 36 | 0 |
| Pax613Neu−/+ × Pax613Neu−/+ | 7 | 31 | 0 | 17 | 10 | 21 | 0 |
| Pax611Neu−/+ × Pax6−/+a | 12 | 94 | 0 | 11 | 31 | 48 | 15 |
| Pax612Neu−/+ × Pax62Neu−/+ | 2 | 16 | 0 | 2 | 5 | 6 | 5 |
| Pax613Neu−/+ × Pax63Neu−/+ | 2 | 17 | 0 | 4 | 6 | 7 | 4 |
| Pax611Neu−/+ × Pax612Neu−/+ | 7 | 39 | 0 | 21 | 14 | 25 | 0 |
| Pax611Neu−/+ × Pax613Neu−/+ | 5 | 38 | 0 | 13 | 14 | 24 | 0 |
| Pax612Neu−/+ × Pax613Neu−/+ | 10 | 55 | 0 | 35 | 18 | 37 | 0 |
| Pax63Neu−/+ × Pax63Neu−/+ | 6 | 64 | 0 | 8 | 15 | 36 | 13 |
The complementation crosses of Pax611Neu heterozygotes were with Pax62Neu, Pax63Neu, and Pax6Sey-Neu.
Microdeletion analysis within the Pax6 region:
To PCR amplify and sequence across the deletions we first needed to more accurately localize the deletion breakpoints. Analyses to define the Pax611Neu, Pax612Neu, and Pax613Neu deleted regions were carried out with polymorphic microsatellite marker sites and the Pax6 gene, FISH cytogenetics, array-CGH, and polymorphic SNP or insert/deletion sites (Table S1, Table S2, Table S3, Table S4, Table S5, Table S6, Table S7, Table S8, Table S9, Table S10, Figure S1). The results were collated to give the most accurate localization of the deletion breakpoints (Table 3). On the basis of these results we were able to design primers outside of but close to the deletion breakpoints to amplify and sequence across the deletions.
TABLE 3.
Localization of Chr 2 deleted regions in the mouse
|
Pax6 deletions
|
||||||
|---|---|---|---|---|---|---|
| BAC | Position (Mb)a | Pax6Sey-Dey | Pax6Sey-H | Pax611Neu | Pax612Neu | Pax613Neu |
| RP24-334I20 | 104.37 | |||||
| RP23-124B20 | 104.40 | Δ | ||||
| RP23-189F6 | 104.51 | Δ | ||||
| RP23198B6 | 104.65 | Δ | Δ | |||
| RP23-86J23 | 104.84 | Δ | Δ | |||
| RP23-8C14 | 104.93 | Δ | Δ | Δ | ||
| RP23-247F16 | 105.09 | Δ | Δ | Δ | ||
| RP24-182O5 | 105.23 | Δ | Δ | Δ | Δ | |
| RP23-403K1 | 105.44 | Δ | Δ | Δ | Δ | |
| RP23-431C3 | 105.45 | Δ | Δ | Δ | Δ | Δ |
| Pax6 | 105.47 | Δ | Δ | Δ | Δ | Δ |
| RP23-146D23 | 105.58 | Δ | Δ | Δ | Δ | |
| RP24-244B3 | 105.59 | Δ | Δ | |||
| RP24-483E9 | 107.38 | Δ | Δ | |||
| RP23-35J22 | 107.41 | Δ | ||||
| RP23-336F11 | 111.12 | Δ | ||||
| RP23-35G10 | 111.30 | |||||
Based on analyses of microsatellite markers, FISH cytogenetics, array CGH and SNPs (Table S1, Table S2, Table S3, Table S4, Table S5, Table S6, Table S7, Table S8, Table S9, Table S10, Figure S1). Triangles indicate a partially or fully deleted BAC.
Proximal end position of the BAC or gene sequence.
Localization of deletion breakpoints by sequencing:
We designed a DNA walking strategy to sequence across the Pax611Neu deletion. A chimeric sequence that jumped from the region defined by BAC RP23-8C14 to the distal region defined by the BAC RP23-431C3 was obtained (Figure 3). The DNA walking results were confirmed by PCR amplification and sequencing across the Pax611Neu deletion using genome-specific primers contained in the RP23-8C14 and RP23-431C3 BACs. By BAC alignment and inspection of the region in the Ensembl database the proximal and distal breakpoints were localized to Chr 2 105.001 and 105.541 Mb, respectively. The Pax611Neu deletion is 540,470 bp long, starts proximal to the Rcn1 gene, includes the entire Rcn1 and Pax6 genes, and ends in intron 9–10 of the Elp4 gene. Since the Elp4 gene is oriented tail to tail with the Pax6 gene, the deletion within the Elp4 gene results in the loss of the 3′ end of intron 9–10 and exon 10.
Figure 3.—
Chr 2 sequences flanking the Pax611Neu, Pax612Neu, and Pax613Neu deletions. The deleted regions are depicted by the black triangles. Pax611Neu: The chimeric sequence across the Pax611Neu deletion contained in the 5′ end a portion of genomic sequence defined by the BAC RP23-8C14 joined in the 3′ end to a portion of genomic sequence defined by the BAC RP23-431C3. The proximal deletion breakpoint is after the BAC RP23-8C14 position 20,431, and the distal breakpoint is after the BAC RP23-431C3 position 132,356. Pax612Neu: The chimeric sequence across the Pax612Neu deletion contained in the 5′ end a portion of genomic sequence defined by the BAC RP23-290H11 joined in the 3′ end to a portion of genomic sequence defined by the BAC RP23-35G10. The proximal deletion breakpoint is after the BAC RP23-290H11 position 2388, and the distal breakpoint is after the BAC RP23-35G10 position 5858. Pax613Neu: The chimeric sequence across the Pax613Neu deletion contained in the 5′ end a portion of genomic sequence defined by the BAC RP23-431C3 joined in the 3′ end to a portion of genomic sequence defined by the BAC RP23-146D23. The proximal deletion breakpoint is after the BAC RP23-431C3 position 78,814, and the distal breakpoint is after the BAC RP23-146D23 position 148,187.
For the Pax612Neu deletion, we designed a group of 5′ and 3′ specific primers in nondeleted regions flanking the deletion to amplify across the deletion breakpoints. We obtained a PCR amplification product ∼8000 bp in length, which was used as substrate to sequence. A chimeric sequence with a 5′ region contained within the sequence defined by BAC RP23-290H11 and a 3′ region contained within the sequence defined by the BAC RP23-35G10 was observed (Figure 3). By BAC alignment and inspection in the Ensembl database the Pax612Neu deletion could be identified to be 6.08 Mb in length with the proximal and distal breakpoints at Chr 2 105.299 and 111.380 Mb, respectively. The deletion begins distal to the Rcn1 gene, includes the Pax6 and Elp4 genes, as well as 17 genes distal to Elp4 (Immp1L, Dph4, Dcdc5, Mppede2, Fshb, Kcna4, Rpl35a, Hadhb, Mett5d1, Kif18a, Bdnf, Lin7c, Lgr4, Cdc34, Bbox1, Slc5a12, and Muc15) and 18 genes within the Olfr gene cluster at Chr 2 111.06–111.90 Mb (Olfr1275–Olfr1281, Olfr1283–Olfr1291, Olfr1294, and Olfr1295).
A series of 5′ and 3′ primers in nondeleted regions flanking Pax613Neu were designed to amplify across the deletion breakpoints. We obtained a specific PCR amplification product of ∼3700 bp in length, which we used as a substrate to sequence. A chimeric sequence with a 5′ region contained within the sequence defined by BAC RP23-431C3 and a 3′ region contained within the sequence defined by the BAC RP23-146D23 was observed (Figure 3). By BAC alignment and inspection in the Ensembl database the Pax613Neu deletion could be identified to be 237,725 bp long with the proximal and distal breakpoints at Chr 2 105.488 and 105.726 Mb, respectively. The deletion begins within the Pax6 gene in intron 6–7 (canonical isoform, ENSMUST00000090397), includes the entire Elp4 gene, and ends in intron 2–3 of the Immp1L gene.
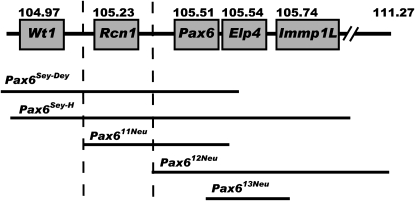
The extent of the deleted regions relative to Pax6 and closely linked genes for the Pax611Neu, Pax612Neu, and Pax613Neu as well as the previously characterized Pax6Sey-Dey and Pax6Sey-H mutations is schematically depicted in Figure 4.
Figure 4.—
Schematic overview of the deleted regions in the Pax6Sey-Dey, Pax6Sey-H, Pax611Neu, Pax612Neu, and the Pax613Neu deletions. Pax6 and the proximal genes Wt1 and Rcn1 as well as the distal genes Elp4 and Immp1L are shown with their proximal end position in Mb (Ensembl build 51). The Pax6Sey-Dey deletion begins most proximal, includes Wt1, Rcn1, Pax6, and Elp4, and is estimated to be 1.2 Mb. The Pax6Sey-H deletion includes Wt1, Rcn1, Pax6, Elp4, and Immp1L, extends much further distally, and is estimated to be 2.9 Mb. The Pax611Neu deletion begins distal to Wt1 and proximal to Rcn1, extends into Elp4, and is 540 kb. Since the Elp4 gene is orientated tail to tail to the Pax6 gene the 3′ end of the Elp4 gene is deleted. The Pax612Neu deletion begins distal to Rcn1 and proximal to Pax6, extends furthest distally, and is 6.08 Mb long. The Pax613Neu deletion is 238 kb, begins within intron 6-7 of Pax6, extends through Elp4, and ends within the 5′ region of Immp1L. The critical region responsible for the extreme eye phenotype is marked by the broken lines.
Transcript levels of Rcn1, Pax6, and Elp4, alternative Pax6 isoforms, and Pax6-Immp1L fusion transcript in the eyes of Pax6 deletion or intragenic mutant heterozygotes:
We next measured the transcript levels of Pax6 and affected genes closely linked to Pax6 in the eyes of Pax611Neu, Pax612Neu, Pax613Neu, and Pax63Neu heterozygotes by quantitative real-time RT–PCR (Table 4). The level of Pax6 transcript was significantly reduced in both Pax611Neu (t6 = 5.69, P = 0.001) and Pax612Neu (t5 = 4.87, P = 0.002) heterozygotes when compared to homozygous wild type. The level of Pax6 transcript in Pax613Neu and Pax63Neu heterozygotes was similar to wild type. The Pax6 probe used for the quantitative real-time RT–PCR assay is based on an amplicon spanning the Pax6 exon 3–exon 4 junction. This site within the Pax6 gene is not deleted in Pax613Neu. The observation that the level of Pax6 transcript in Pax613Neu heterozygotes was similar to wild type suggests that a stable transcript encoded by the 5′ region of Pax6 from the Pax613Neu partial Pax6 deletion was present (see below). The Rcn1 transcript level in Pax611Neu heterozygotes was significantly less than that observed in homozygous wild types (t4 = 2.78, P = 0.025). The levels of Rcn1 transcript in Pax612Neu, Pax613Neu, and Pax63Neu heterozygotes were not different from homozygous wild types. The Elp4 transcript levels were significantly reduced in Pax611Neu (t4 = 5.57, P = 0.002), Pax612Neu (t4 = 13.38, P = 0.002), and Pax613Neu (t4 = 5.71, P = 0.002) heterozygotes and not different in Pax63Neu heterozygotes, as compared to homozygous wild types. The Elp4 probe used for the quantitative real-time RT–PCR assay is based on an amplicon spanning the Elp4 exon 4–exon 5 junction. This region of Elp4 is not deleted in Pax611Neu. The observation that the transcript level of Elp4 in the Pax611Neu heterozygotes was reduced suggests that a transcript encoded by the nondeleted portion of Elp4 in Pax611Neu was not produced or was unstable.
TABLE 4.
Transcription levels of Pax6, Rcn1, and Elp4 in eyes of P1 mice relative to the expression of Tbp and to total head mRNA
|
Pax6
|
Rcn1
|
Elp4
|
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Genotype | Mean | SD | n | Mean | SD | n | Mean | SD | n |
| Pax611Neu −/+ | 4.34** | 0.07 | 4 | 3.10* | 0.31 | 3 | 2.91** | 0.75 | 3 |
| Pax612Neu −/+ | 4.25** | 0.24 | 3 | 4.29 | 0.44 | 3 | 3.94** | 0.27 | 3 |
| Pax613Neu −/+ | 6.38 | 1.62 | 3 | 4.95 | 0.61 | 3 | 3.03** | 0.21 | 3 |
| Pax63Neu −/+ | 6.12 | 0.14 | 3 | 4.39 | 0.35 | 3 | 6.12 | 0.77 | 3 |
| +/+ | 6.15 | 0.63 | 3 | 4.58 | 0.87 | 3 | 5.80 | 0.49 | 3 |
Significantly different from +/+: *P < 0.05; **P < 0.01.
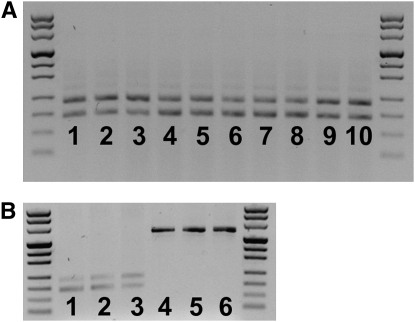
We assayed for the presence of the canonical Pax6 transcript (does not contain exon 5a) and the alternatively spliced Pax6 isoform (containing exon 5a) in Pax611Neu, Pax612Neu, Pax613Neu, and Pax63Neu heterozygotes, and in homozygous wild types. For all genotypes, both Pax6 isoforms were present, which suggests that the mutations did not affect alternative splicing (Figure 5, A and B).
Figure 5.—
Pax6 isoforms and Pax6-Immp1L fusion transcript in mutant and wild-type eyes of P1 mice. (A) PCR amplification products of a region of the Pax6 transcript spanning exon 5a. In all samples both a shorter 182-bp band, which represents the amplification of the canonical Pax6 transcript, and a longer 224-bp band, which represents the amplification of the alternatively spliced Pax6(5a) isoform, are present, indicating that alternative splicing was not affected by the deletions. (Lanes 1–3) Pax611Neu −/+. (Lanes 4 and 5) Pax612Neu −/+. (Lanes 6–8) Pax6 +/+. (Lanes 9 and 10) Pax63Neu −/+. (B) Pax6 canonical, Pax6(5a) and Pax6-Immp1L fusion transcripts in Pax613Neu heterozygotes. (Lanes 1–3) Pax613Neu −/+ amplified for the region of Pax6 spanning exon 5a. (Lanes 4–6) Pax613Neu −/+ amplified for the predicted Pax6-Immp1L fusion transcript.
The Pax613Neu deletion begins in intron 6–7 of Pax6 and ends in intron 2–3 of Immp1L. The resulting genomic rearrangement juxtaposes the Pax6 exon–intron structure up to exon 6 to the Immp1L exon–intron structure starting at exon 3, with an intervening fusion intron consisting of the 5′ region of Pax6 intron 6–7 and the 3′ region of Immp1L intron 2–3. To determine whether a fusion transcript was expressed, we used a 5′ primer specific for Pax6 and a 3′ primer specific for Immp1L to assay by PCR amplification of cDNA from P1 eyes of Pax613Neu heterozygotes. A 618-bp product was obtained (Figure 5B), indicating that a Pax6–Immp1L fusion transcript was present. We designed a series of overlapping primer pairs to amplify and sequence across the predicted fusion transcript. Two isoforms were confirmed to be present. The most abundant form corresponded to Pax6 exons 1–6 without exon 5a fused to Immp1L exons 3–7. Low levels of Pax6 exons 1–6 including exon 5a fused to Immp1L exons 3–7 were also observed. The fusion results in an out-of-frame transcript. Translation is predicted to proceed through Pax6 exon 6, followed by two tryptophans and a stop codon. Since normal function of Pax6 requires intact paired-, homeo-, and transactivation domains, we conclude that the Pax6 activity is abolished in the Pax613Neu mutant gene product. It should be noted that the entire ORF of Immp1L is contained within the fusion transcript, although 5′ nontranslated sequences contained within Immp1L exons 1 and 2 are deleted. However, we do not know if translation of Immp1L from the fusion transcript occurs.
DISCUSSION
We provide genetic, phenotypic, and molecular characterizations of three new Pax6 deletions of the mouse. We were able to sequence across all three deletions and could exactly identify the deleted regions. These results extend the allelic series for Pax6 deletions to include Pax6Sey-Dey, Pax6Sey-H, Pax6Sey-2H, Pax6Sey-3H, Pax6Sey-4H, Pax611Neu, Pax612Neu, and Pax613Neu. More importantly, we introduce the first two deletions that are not associated with extreme microphthalmia and the first three deletions that do not express belly spotting. With the available panel of five Pax6 deletions (the previously described Pax6Sey-Dey and Pax6Sey-H, as well as the three deletions from the present study) we were able to define the Chr 2 regions associated with the extreme eye phenotype and belly spotting.
Extreme eye phenotype in Pax6 microdeletions:
Since Pax6 is critical for eye formation, our initial hypothesis considered the differences observed in the eye phenotypes expressed in the deletions and the intragenic null mutations to be directly due to the action of Pax6 and that Pax6 deletions are functionally different from Pax6 intragenic null mutations. Our observations that the Pax612Neu and Pax613Neu deletions do not express extreme microphthalmia and the levels of Pax6 transcription are similarly reduced in the heterozygous Pax611Neu (extreme microphthalmia) and Pax612Neu (no extreme eye phenotype) deletions, allowed us to reject the hypothesis.
We also considered the possibility that the deletions may affect alternative splicing of Pax6. There are two major isoforms expressed in the eye, Pax6 (canonical) and Pax6 (5a), and a correct ratio of these isoforms is critical for normal eye development (Epstein et al. 1994; Duncan et al. 2000; Singh et al. 2002). However, we did not observe a disturbance in the canonical/5a isoform ratios in the Pax611Neu heterozygotes expressing extreme microphthalmia as compared to the ratios seen in the Pax612Neu, Pax613Neu, and Pax63Neu heterozygotes expressing the milder eye phenotype.
Since the levels of Pax6 expression do not correlate with the extent of eye phenotype expressed by the Pax6 deletion heterozygotes, we considered our alternative initial hypothesis, i.e., that a region linked to but distinct from the Pax6 gene is responsible for the extreme eye phenotype observed in the deletions. Figure 4 depicts the extent of the regions surrounding Pax6 affected in five different deletions. Pax6Sey-Dey, Pax6Sey-H, and Pax611Neu heterozygotes express extreme microphthalmia, while Pax612Neu and Pax613Neu heterozygotes do not. By alignment of the five deletions, we may exclude the region proximal to the proximal breakpoint in the Pax611Neu deletion, included in the Pax6Sey-Dey and Pax6Sey-H deletions, as responsible for extreme microphthalmia. Similarly, we may exclude the extensive region included in the Pax612Neu and Pax613Neu deletions as responsible for extreme microphthalmia. Thus, on the basis of the analysis of these five deletions, the region defined by the proximal breakpoint of the Pax611Neu deletion up to the proximal breakpoint of the Pax612Neu deletion is responsible for extreme microphthalmia. The region contains one gene, reticulocalbin 1 (Rcn1). Rcn1 is a Ca2+-binding protein, resident in the endoplasmic reticulum and implicated in the secretory pathway (Ozawa and Muramatsu 1993; Weis et al. 1994; Ozawa 1995a,b; Tachikui et al. 1997). Rcn1 has been shown to be expressed in a number of tissues (Fukuda et al. 2007), including the eye (present study). Linkage between Pax6 and Rcn1 has been conserved among mouse, man, and fish (Kent et al. 1997; Miles et al. 1998; Kleinjan et al. 2008). Unfortunately there are no known intragenic mutations of the Rcn1/RCN1 genes in mouse or man. We are currently in the process of generating a mutation of Rcn1 to directly test the hypothesis that Rcn1, when mutated, is either directly or in conjunction with a Pax6 mutation responsible for the extreme eye phenotype observed in mouse Pax6 deletions that include the Rcn1 gene.
An extensive series of PAX6 intragenic mutations has been identified in humans (http://pax6.hgu.mrc.ac.uk) and a number of contiguous gene deletions encompassing the PAX6 region have been characterized (van Heyningen et al. 1985; Fantes et al. 1992; Drechsler et al. 1994; Crolla et al. 1997; Chao et al. 2000; Grønskov et al. 2001; Crolla and van Heyningen 2002; Robinson et al. 2008). We are unaware of any studies which have compared the extent of the eye abnormalities expressed by carriers of deletions vs. intragenic mutant alleles. Such information would be extremely valuable to further test if the region containing the RCN1 gene is associated with a more extreme eye abnormality in PAX6 multilocus deletions.
The Pax613Neu partial deletion of Pax6 results in the expression of a Pax6–Immp1L chimera transcript. The predicted translation product is a truncated Pax6 protein, and indeed the eye phenotype associated with Pax613Neu heterozygotes is similar to heterozygotes for intragenic Pax6 mutations leading to premature termination of translation (Hill et al. 1991; Lyon et al. 2000; Favor et al. 2001; Graw et al. 2005). Since we did not observe any unusual phenotypes associated with the Pax613Neu deletion, we conclude that the expressed Pax6–Immp1L chimera transcript is not acting as a dominant negative.
Early embryonic lethality:
The five well-characterized mouse Pax6 deletions (Figure 4) are all homozygous lethal at an early embryonic stage (Varnum and Stevens 1974; Theiler et al. 1978; Hogan et al. 1986; Hogan et al. 1987; present study). There are probably a number of genes within the region which, if their function were ablated, would lead to lethality of the affected animal. Rcn1 has been previously suggested (Kent et al. 1997). Homozygous Wt1 mutant embryos die between E13 and E15 (Kreidberg et al. 1993). Dph4 homozygotes die prior to E14.5 when carried on a C3H/HeH genetic background (Webb et al. 2008). Homozygous Pax6 intragenic null mutations are lethal shortly after birth. With the available panel of characterized mouse deletions we cannot exclude any genes within the region responsible for early embryonic lethality. However, our complementation tests between the Pax611Neu, Pax612Neu, and Pax613Neu deletions show that loss of Elp4 alone in the Pax611Neu/Pax613Neu compound heterozygotes results in early embryonic lethality. Elp4 is located adjacent to Pax6 with conserved linkage from mammals to fish (Kleinjan et al. 2002, 2008). Elp4 is one subunit within the elongator complex that functions in transcript elongation (Winkler et al. 2001) and ELP4/Elp4 has been shown to be ubiquitously expressed in human and mouse tissues (Winkler et al. 2001; Kleinjan et al. 2002). As differentiation progresses, proper embryonic development becomes more dependent on embryonic-derived transcripts. The function and expression pattern of Elp4 would be consistent with our observation that loss of Elp4 leads to early embryonic lethality.
Belly spotting:
The five Pax6 deletions included in our comparisons (Figure 4) differentiate for the belly-spotting trait. Pax6Sey-Dey and Pax6Sey-H heterozygotes express belly spotting, while the Pax611Neu, Pax612Neu, and Pax613Neu heterozygotes do not. By alignment of the deleted regions from the five mutations we may exclude the extensive region between the proximal breakpoint of the Pax611Neu deletion and the distal breakpoint of the Pax612Neu deletion to be responsible for belly spotting. Similarly, we may exclude the region defined by the proximal breakpoint of the Pax6Sey-Dey deletion through to the proximal breakpoint of the Pax6Sey-H deletion. Thus, the region responsible for belly spotting is between the proximal breakpoint of the Pax6Sey-H deletion and the proximal breakpoint of the Pax611Neu deletion. Consistent with this conclusion is the observation that a yeast artificial chromosome containing the human PAX6 gene as well as the genomic regions 200 kb upstream and 200 kb downstream of PAX6 rescues the small-eye phenotype but not the belly-spotting phenotype of Pax6Sey-H heterozygotes (Kleinjan et al. 2001, 2002). Inspection of the genome organization in the human PAX6 region indicates that the Y593-1 YAC extends upstream of PAX6 to the vicinity of the RCN1 gene. The eye phenotype in Pax6Sey-H heterozygotes was rescued by the PAX6 gene contained within the YAC. The belly-spotting phenotype was not rescued in Pax6Sey-H heterozygotes because the YAC does not contain the homologs of the mouse genes proximal to Rcn1, which are deleted in the Pax6Sey-H deletion.
Belly spotting is an easily identifiable trait, and numerous independent mutations have been recovered in breeding colonies as well as in radiation or chemical mutagenesis studies (Bult et al. 2008). The fact that none were mapped to this region of Chr 2 would imply that either a mutation at a potential single gene target within this region has not yet been recovered or that a potential single gene target does not exist. Cattanach et al. (1993) have shown that chromosomal imbalance due to large multilocus deletions are often associated with belly spotting and growth retardation. This may imply that a multilocus deletion within the Chr 2 region and not the deletion of a single gene results in belly spotting.
Acknowledgments
We thank Brigitta May, Elenore Samson, and Sylvia Wolf for expert technical assistance; Bahar Sanli-Bonazzi for quantitative real-time PCR analyses; Utz Linzner (Institute of Pathology, Helmholtz Zentrum München, National Research Center for Environmental Health, Neuherberg, Germany) for the synthesis of primers; and Laure Bally-Cuif for critically reading and making valuable suggestions to the manuscript. Research was partially supported by National Institutes of Health grant R0-1EY10321 and contract no. CHRX-CT93-0181 from the Commission of the European Communities.
The mutant allele symbols Del(2)Pax611Neu/1Neu, Del(2)Pax612Neu/2Neu and Del(2)Pax613Neu/3Neu were submitted to and approved by the Mouse Genetic Nomenclature Committee, and assigned the MGI accession ID nos. 3698295, 3698297, and 3710946, respectively.
Supporting information is available online at http://www.genetics.org/cgi/content/full/genetics.109.104562/DC1.
References
- Bartsch, O., W. Wuyts, W. Van Hul, J. T. Hecht, P. Meinecke et al., 1996. Delineation of a contiguous gene syndrome with multiple exostoses, enlarged parietal foramina, craniofacial dysostosis, and mental retardation, caused by deletions in the short arm of chromosome 11. Am. J. Hum. Genet. 58 734–742. [PMC free article] [PubMed] [Google Scholar]
- Brémond-Gignac, D., J. A. Crolla, H. Copin, A. Guichet, D. Bonneau et al., 2005. Combination of WAGR and Potocki-Shaffer contiguous deletion syndromes in a patient with an 11p11.2-p14 deletion. Eur. J. Hum. Genet. 13 409–413. [DOI] [PubMed] [Google Scholar]
- Bult, C. J., J. T. Eppig, J. A. Kadin, J. E. Richardson, J. A. Blake et al., 2008. The Mouse Genome Database (MGD): mouse biology and model systems. Nucleic Acids Res. 36 D724–D728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattanach, B. M., M. D. Burtenshaw, C. Rasberry and E. P. Evans, 1993. Large deletions and other gross forms of chromosome imbalance compatible with viability and fertility in the mouse. Nat. Genet. 3 56–61. [DOI] [PubMed] [Google Scholar]
- Chao, L. Y., V. Huff, L. C. Strong and G. F. Saunders, 2000. Mutation in the PAX6 gene in twenty patients with aniridia. Hum. Mutat. 15 332–339. [DOI] [PubMed] [Google Scholar]
- Crolla, J. A., and V. van Heyningen, 2002. Frequent chromosome aberrations revealed by molecular cytogenetic studies in patients with aniridia. Am. J. Hum. Genet. 71 1138–1149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crolla, J. A., J. E. Cawdery, C. A. Oley, I. D. Young, J. Gray et al., 1997. A FISH approach to defining the extent and possible clinical significance of deletions at the WAGR locus. J. Med. Genet. 34 207–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curto, G. G., J. M. Lara, M. Parrilla, J. Aijón and A. Velasco, 2007. Modifications of the retina neuronal populations of the heterozygous mutant small eye mouse, the SeyDey. Brain Res. 1127 163–176. [DOI] [PubMed] [Google Scholar]
- D'Elia, A. V., L. Pellizzari, D. Fabbro, A. Pianta, M. T. Divizia et al., 2007. A deletion 3′ to the PAX6 gene in familial aniridia cases. Mol. Vis. 13 1245–1250. [PubMed] [Google Scholar]
- Davis, L. K., K. J. Meyer, D. S. Rudd, A. L. Librant, E. A. Epping et al., 2008. Pax6 3′ deletion results in aniridia, autism and mental retardation. Hum. Genet. 123 371–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drechsler, M., E. J. Meijers-Heijboer, S. Schneider, B. Schurich, C. Grond-Ginsbach et al., 1994. Molecular analysis of aniridia patients for deletions involving the Wilms‘ tumor gene. Hum. Genet. 94 331–338. [DOI] [PubMed] [Google Scholar]
- Duncan, M. K., Z. Kozmik, K. Cveklova, J. Piatigorsky and A. Cvekl, 2000. Overexpression of PAX6(5a) in lens fiber cells results in cataract and upregulation of α5β1 integrin expression. J. Cell Sci. 113(Pt 18): 3173–3185. [DOI] [PubMed] [Google Scholar]
- Epstein, J. A., T. Glaser, J. Cai, L. Jepeal, D. S. Walton et al., 1994. Two independent and interactive DNA-binding subdomains of the Pax6 paired domain are regulated by alternative splicing. Genes Dev. 8 2022–2034. [DOI] [PubMed] [Google Scholar]
- Fantes, J. A., W. A. Bickmore, J. M. Fletcher, F. Ballesta, I. M. Hanson et al., 1992. Submicroscopic deletions at the WAGR locus, revealed by nonradioactive in situ hybridization. Am. J. Hum. Genet. 51 1286–1294. [PMC free article] [PubMed] [Google Scholar]
- Favor, J., 1983. A comparison of the dominant cataract and recessive specific-locus mutation rates induced by treatment of male mice with ethylnitrosourea. Mutat. Res. 110 367–382. [DOI] [PubMed] [Google Scholar]
- Favor, J., P. Grimes, A. Neuhäuser-Klaus, W. Pretsch and D. Stambolian, 1997. The mouse Cat4 locus maps to chromosome 8 and mutants express lens-corneal adhesion. Mamm. Genome 8 403–406. [DOI] [PubMed] [Google Scholar]
- Favor, J., H. Peters, T. Hermann, W. Schmahl, B. Chatterjee et al., 2001. Molecular characterization of Pax62Neu through Pax610Neu: an extension of the Pax6 allelic series and the identification of two possible hypomorph alleles in the mouse Mus musculus. Genetics 159 1689–1700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favor, J., C. J. Gloeckner, A. Neuhäuser-Klaus, W. Pretsch, R. Sandulache et al., 2008. Relationship of Pax6 activity levels to the extent of eye development in the mouse, Mus musculus. Genetics 179 1345–1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francke, U., L. B. Holmes, L. Atkins and V. M. Riccardi, 1979. Aniridia-Wilms' tumor association: evidence for specific deletion of 11p13. Cytogenet. Cell Genet. 24 185–192. [DOI] [PubMed] [Google Scholar]
- Fryns, J. P., J. Beirinckx, E. De Sutter, J. Derluyn, J. Francois et al., 1981. Aniridia-Wilms' tumor association and 11p interstitial deletion. Eur. J. Pediatr. 136 91–92. [DOI] [PubMed] [Google Scholar]
- Fukuda, T., H. Oyamada, T. Isshiki, M. Maeda, T. Kusakabe et al., 2007. Distribution and variable expression of secretory pathway protein reticulocalbin in normal human organs and non-neoplastic pathological conditions. J. Histochem. Cytochem. 55 335–345. [DOI] [PubMed] [Google Scholar]
- Glaser, T., W. H. Lewis, G. A. Bruns, P. C. Watkins, C. E. Rogler et al., 1986. The beta-subunit of follicle-stimulating hormone is deleted in patients with aniridia and Wilms' tumour, allowing a further definition of the WAGR locus. Nature 321 882–887. [DOI] [PubMed] [Google Scholar]
- Glaser, T., J. Lane and D. Housman, 1990. A mouse model of the aniridia-Wilms tumor deletion syndrome. Science 250 823–827. [DOI] [PubMed] [Google Scholar]
- Glaser, T., D. S. Walton and R. L. Maas, 1992. Genomic structure, evolutionary conservation and aniridia mutations in the human PAX6 gene. Nat. Genet. 2 232–239. [DOI] [PubMed] [Google Scholar]
- Graw, J., J. Löster, O. Puk, D. Munster, N. Haubst et al., 2005. Three novel Pax6 alleles in the mouse leading to the same small-eye phenotype caused by different consequences at target promoters. Invest. Ophthalmol. Vis. Sci. 46 4671–4683. [DOI] [PubMed] [Google Scholar]
- Grønskov, K., J. H. Olsen, A. Sand, W. Pedersen, N. Carlsen et al., 2001. Population-based risk estimates of Wilms tumor in sporadic aniridia. A comprehensive mutation screening procedure of PAX6 identifies 80% of mutations in aniridia. Hum. Genet. 109 11–18. [DOI] [PubMed] [Google Scholar]
- Hill, R. E., J. Favor, B. L. Hogan, C. C. Ton, G. F. Saunders et al., 1991. Mouse Small eye results from mutations in a paired-like homeobox-containing gene. Nature 354 522–525. [DOI] [PubMed] [Google Scholar]
- Hittner, H. M., V. M. Riccardi and U. Francke, 1979. Aniridia caused by a heritable chromosome 11 deletion. Ophthalmology 86 1173–1183. [DOI] [PubMed] [Google Scholar]
- Hogan, B. L., G. Horsburgh, J. Cohen, C. M. Hetherington, G. Fisher et al., 1986. Small eyes (Sey): a homozygous lethal mutation on chromosome 2 which affects the differentiation of both lens and nasal placodes in the mouse. J. Embryol. Exp. Morphol. 97 95–110. [PubMed] [Google Scholar]
- Hogan, B. L., C. M. Hetherington and M. F. Lyon, 1987. Allelism of small eyes (Sey) with Dickie's small eye (Dey) on Chromosome 2. Mouse News Lett. 77 135–138. [Google Scholar]
- Kent, J., M. Lee, A. Schedl, S. Boyle, J. Fantes et al., 1997. The reticulocalbin gene maps to the WAGR region in human and to the Small eye Harwell deletion in mouse. Genomics 42 260–267. [DOI] [PubMed] [Google Scholar]
- Kleinjan, D. A., A. Seawright, A. Schedl, R. A. Quinlan, S. Danes et al., 2001. Aniridia-associated translocations, DNase hypersensitivity, sequence comparison and transgenic analysis redefine the functional domain of PAX6. Hum. Mol. Genet. 10 2049–2059. [DOI] [PubMed] [Google Scholar]
- Kleinjan, D. A., A. Seawright, G. Elgar and V. van Heyningen, 2002. Characterization of a novel gene adjacent to PAX6, revealing synteny conservation with functional significance. Mamm. Genome 13 102–107. [DOI] [PubMed] [Google Scholar]
- Kleinjan, D. A., R. M. Bancewicz, P. Gautier, R. Dahm, H. B. Schonthaler et al., 2008. Subfunctionalization of duplicated zebrafish pax6 genes by cis-regulatory divergence. PLoS Genet. 4 e29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreidberg, J. A., H. Sariola, J. M. Loring, M. Maeda, J. Pelletier et al., 1993. WT-1 is required for early kidney development. Cell 74 679–691. [DOI] [PubMed] [Google Scholar]
- Lauderdale, J. D., J. S. Wilensky, E. R. Oliver, D. S. Walton and T. Glaser, 2000. 3′ deletions cause aniridia by preventing PAX6 gene expression. Proc. Natl. Acad. Sci. USA 97 13755–13759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ligon, A. H., L. Potocki, L. G. Shaffer, D. Stickens and G. A. Evans, 1998. Gene for multiple exostoses (EXT2) maps to 11(p11.2p12) and is deleted in patients with a contiguous gene syndrome. Am. J. Med. Genet. 75 538–540. [DOI] [PubMed] [Google Scholar]
- Lyon, M. F., D. Bogani, Y. Boyd, P. Guillot and J. Favor, 2000. Further genetic analysis of two autosomal dominant mouse eye defects, Ccw and Pax6coop. Mol. Vis. 6 199–203. [PubMed] [Google Scholar]
- McGaughran, J. M., H. B. Ward and D. G. Evans, 1995. WAGR syndrome and multiple exostoses in a patient with del(11)(p11.2p14.2). J. Med. Genet. 32 823–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles, C., G. Elgar, E. Coles, D. J. Kleinjan, V. van Heyningen et al., 1998. Complete sequencing of the Fugu WAGR region from WT1 to PAX6: dramatic compaction and conservation of synteny with human chromosome 11p13. Proc. Natl. Acad. Sci. USA 95 13068–13072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozawa, M., 1995. a Cloning of a human homologue of mouse reticulocalbin reveals conservation of structural domains in the novel endoplasmic reticulum resident Ca2+-binding protein with multiple EF-hand motifs. J. Biochem. 117 1113–1119. [DOI] [PubMed] [Google Scholar]
- Ozawa, M., 1995. b Structure of the gene encoding mouse reticulocalbin, a novel endoplasmic reticulum-resident Ca2+-binding protein with multiple EF-hand motifs. J. Biochem. 118 154–160. [DOI] [PubMed] [Google Scholar]
- Ozawa, M., and T. Muramatsu, 1993. Reticulocalbin, a novel endoplasmic reticulum resident Ca2+-binding protein with multiple EF-hand motifs and a carboxyl-terminal HDEL sequence. J. Biol. Chem. 268 699–705. [PubMed] [Google Scholar]
- Potocki, L., and L. G. Shaffer, 1996. Interstitial deletion of 11(p11.2p12): a newly described contiguous gene deletion syndrome involving the gene for hereditary multiple exostoses (EXT2). Am. J. Med. Genet. 62 319–325. [DOI] [PubMed] [Google Scholar]
- Riccardi, V. M., E. Sujansky, A. C. Smith and U. Francke, 1978. Chromosomal imbalance in the Aniridia-Wilms' tumor association: 11p interstitial deletion. Pediatrics 61 604–610. [PubMed] [Google Scholar]
- Roberts, R. C., 1967. Small eyes—a new dominant eye mutant in the mouse. Genet. Res. 9 121–122. [Google Scholar]
- Robinson, D. O., R. J. Howarth, K. A. Williamson, V. van Heyningen, S. J. Beal et al., 2008. Genetic analysis of chromosome 11p13 and the PAX6 gene in a series of 125 cases referred with aniridia. Am. J. Med. Genet. 146A 558–569. [DOI] [PubMed] [Google Scholar]
- Schmahl, W., M. Knoedlseder, J. Favor and D. Davidson, 1993. Defects of neuronal migration and the pathogenesis of cortical malformations are associated with Small eye (Sey) in the mouse, a point mutation at the Pax-6-locus. Acta Neuropathol. 86 126–135. [DOI] [PubMed] [Google Scholar]
- Shaffer, L. G., J. T. Hecht, D. H. Ledbetter and F. Greenberg, 1993. Familial interstitial deletion 11(p11.12p12) associated with parietal foramina, brachymicrocephaly, and mental retardation. Am. J. Med. Genet. 45 581–583. [DOI] [PubMed] [Google Scholar]
- Singh, S., R. Mishra, N. A. Arango, J. M. Deng, R. R. Behringer et al., 2002. Iris hypoplasia in mice that lack the alternatively spliced Pax6(5a) isoform. Proc. Natl. Acad. Sci. USA 99 6812–6815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tachikui, H., A. F. Navet and M. Ozawa, 1997. Identification of the Ca2+-binding domains in reticulocalbin, an endoplasmic reticulum resident Ca2+-binding protein with multiple EF-hand motifs. J. Biochem. 121 145–149. [DOI] [PubMed] [Google Scholar]
- Theiler, K., D. S. Varnum and L. C. Stevens, 1978. Development of Dickie's small eye, a mutation in the house mouse. Anat. Embryol. 155 81–86. [DOI] [PubMed] [Google Scholar]
- van Heyningen, V., P. A. Boyd, A. Seawright, J. M. Fletcher, J. A. Fantes et al., 1985. Molecular analysis of chromosome 11 deletions in aniridia-Wilms tumor syndrome. Proc. Natl. Acad. Sci. USA 82 8592–8596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varnum, D., and L. C. Stevens, 1974. Dickie's small eye (Dey). Mouse News Lett. 50 43. [Google Scholar]
- Wakui, K., G. Gregato, B. C. Ballif, C. D. Glotzbach, K. A. Bailey et al., 2005. Construction of a natural panel of 11p11.2 deletions and further delineation of the critical region involved in Potocki-Shaffer syndrome. Eur. J. Hum. Genet. 13 528–540. [DOI] [PubMed] [Google Scholar]
- Webb, T. R., S. H. Cross, L. McKie, R. Edgar, L. Vizor et al., 2008. Diphthamide modification of eEF2 requires a J-domain protein and is essential for normal development. J. Cell. Sci. 121 3140–3145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weis, K., G. Griffiths and A. I. Lamond, 1994. The endoplasmic reticulum calcium-binding protein of 55 kDa is a novel EF-hand protein retained in the endoplasmic reticulum by a carboxyl-terminal His-Asp-Glu-Leu motif. J. Biol. Chem. 269 19142–19150. [PubMed] [Google Scholar]
- Winkler, G. S., T. G. Petrakis, S. Ethelberg, M. Tokunaga, H. Erdjument-Bromage et al., 2001. RNA polymerase II elongator holoenzyme is composed of two discrete subcomplexes. J. Biol. Chem. 276 32743–32749. [DOI] [PubMed] [Google Scholar]
- Wu, Y. Q., J. L. Badano, C. McCaskill, H. Vogel, L. Potocki et al., 2000. Haploinsufficiency of ALX4 as a potential cause of parietal foramina in the 11p11.2 contiguous gene-deletion syndrome. Am. J. Hum. Genet. 67 1327–1332. [DOI] [PMC free article] [PubMed] [Google Scholar]