Abstract
Background: Breast cancer central nervous system (CNS) metastases are an increasingly important problem because of high CNS relapse rates in patients treated with trastuzumab and/or taxanes.
Patients and methods: We evaluated data from 2887 node-positive breast cancer patients randomised in the BIG 02-98 trial comparing anthracycline-based adjuvant chemotherapy (control arms) to anthracycline–docetaxel-based sequential or concurrent chemotherapy (experimental arms). After a median follow-up of 5 years, 403 patients had died and detailed information on CNS relapse was collected for these patients.
Results: CNS relapse occurred in 4.0% of control patients and 3.7% of docetaxel-treated patients. CNS relapse occurred in 27% of deceased patients in both treatment groups. CNS relapse was usually accompanied by neurologic symptoms (90%), and 25% of patients with CNS relapse died without evidence of extra-CNS relapse. Only 20% of patients survived 1 year from the diagnosis of CNS relapse. Prognosis of CNS relapse was worse for patients with meningeal carcinomatosis when compared with brain metastases. Unexpected findings included a higher rate of positive cerebrospinal fluid cytology (8% versus 3%) and more frequent use of magnetic resonance imaging for diagnosis (47% versus 30%) in the docetaxel-treated patients.
Conclusion: There is no evidence that adjuvant docetaxel treatment is associated with an increased frequency of CNS relapse.
Keywords: adjuvant chemotherapy, breast cancer, central nervous system, meningeal carcinomatosis, taxane, trastuzumab
introduction
Node-positive breast cancer is a disease with a significant risk of relapse and death. Anthracycline-containing adjuvant chemotherapy has been shown to increase disease-free survival (DFS) and overall survival [1–3]. Adjuvant chemotherapy and/or endocrine therapy (for hormone receptor-positive patients) are recommended to women with node-positive breast cancer after surgery on the basis of a multitude of adjuvant trials summarised in meta-analyses [1] and guidelines [2, 3]. The activity of docetaxel in metastatic disease mandated its evaluation in the adjuvant setting. In addition, the relative merits of combination versus sequential anthracycline–taxane adjuvant chemotherapy remain undefined. Similarly to other trials initiated in the 1990s, the BIG 02-98 trial randomised 2887 patients with node-positive breast cancer into experimental taxane-containing arms versus control arms. First efficacy results of the BIG 02-98 trial have been reported [4]. At database cut-off in March 2006, median follow-up was just >5 years and 732 events had occurred. The addition of docetaxel resulted in a borderline improvement in DFS {hazard ratio (HR) of 0.86 [95% confidence interval (CI) = 0.74–1.00; P = 0.051]}. However, the sequential docetaxel arm A (doxorubicin) → T (docetaxel) → cyclophosphamide, methotrexate, fluorouracil (CMF) produced superior DFS over both the concurrent docetaxel arm AT → CMF (HR = 0.83; 95% CI = 0.69–1.00) and a sequential control arm A → CMF (HR = 0.79; 95% CI = 0.64–0.98).
The objective of the present analysis was to evaluate the frequency of CNS relapse in patients treated with or without adjuvant taxanes. This question was studied because taxane treatment in advanced breast cancer was associated with high CNS relapse rates in some studies (up to 30%) [5, 6].
patients and methods
The Breast International Group initiated the BIG 02-98 trial with the participation of eight cooperative groups in 22 countries. The trial was open to accrual from June 1998 to June 2001.
Patients with histologically proven node-positive breast cancer staged T1–3, aged 18–70 years, resected with clear margins and containing at least eight resected axillary lymph nodes were required to give written informed consent before entering the study. Mastectomy could be followed by radiotherapy while the latter was mandatory after breast-conservation surgery. Endocrine therapy was given to all patients with oestrogen receptor (ER) and/or progesterone receptor (PgR)-positive tumours after chemotherapy. Exclusion criteria included evidence of metastatic breast cancer assessed by chest X-ray, abdominal ultrasound or computed tomography (CT) scan and bone scan; abnormal cardiac function assessed by multiple uptake gated acquisition scan or echocardiography and a prior cancer or major comorbidities.
The BIG 02-98 trial randomised 2887 node-positive breast cancer patients into four arms of adjuvant chemotherapy: docetaxel given either sequentially (A–T) or in combination (AT) with doxorubicin, followed by CMF (experimental arms), in comparison to doxorubicin alone (A) or in combination with cyclophosphamide (AC), followed by CMF (control arms). Twice as many patients were randomly assigned into the experimental arms (A–T or AT) than into the control arms (A or AC). Randomisation was stratified by the following factors: centre, number of positive nodes (1–3 versus 4+) and age (<50 versus ≥50).
The objective of the present report is to evaluate the frequency of CNS relapse in patients treated with adjuvant docetaxel in comparison to patients not treated with adjuvant docetaxel. We collected information on CNS relapse in patients at the time of death. With a median follow-up of 62.5 months, patients who had died by March 2006 were included in this analysis. An unknown number of patients who are still alive may have experienced CNS relapse. These patients are not included in the current analysis. The number of such patients is likely to be small, however, given the median follow-up of 5 years and the dismal prognosis of patients who experience a CNS relapse.
results
The characteristics of the patient population included in the BIG 02-98 trial are summarised in Table 1 (intention-to-treat population). The patients randomised to control (A or AC) and experimental arms (A–T or AT) were well balanced with respect to age, menopausal status, nodal status, tumour grade, tumour size, as well as hormonal status and HER2 status.
Table 1.
Patient population enrolled in the BIG 02-98 trial (N = 2887) (intention-to-treat)
| Treatment arm | Control arms (no T) |
Experimental arms (T) |
||
| Treatment | A or AC |
A–T or AT |
||
| n | % | n | % | |
| Patients | 968 | 100 | 1919 | 100 |
| Age | ||||
| <50 years | 520 | 54 | 1022 | 53 |
| ≥50 years | 448 | 46 | 897 | 47 |
| Menopausal status | ||||
| Pre | 518 | 53 | 1034 | 54 |
| Post | 393 | 41 | 780 | 41 |
| Unknown | 57 | 6 | 105 | 5 |
| Nodal status | ||||
| 1–3 LNs | 527 | 54 | 1045 | 54 |
| 4 or more | 441 | 46 | 874 | 46 |
| Tumour grade | ||||
| Grade 1 | 90 | 9 | 161 | 8 |
| Grade 2 | 403 | 42 | 855 | 45 |
| Grade 3 | 432 | 45 | 832 | 43 |
| Unknown | 43 | 4 | 71 | 4 |
| Tumour size | ||||
| pT1 | 367 | 38 | 776 | 40 |
| pT2 | 529 | 55 | 994 | 52 |
| pT3 | 66 | 7 | 141 | 7 |
| pTX | 6 | 0.6 | 8 | 0.4 |
| Hormone receptors | ||||
| ER+/PgR+ | 488 | 50 | 994 | 52 |
| ER+/PgR− | 112 | 12 | 230 | 12 |
| ER−/PgR+ | 44 | 4 | 73 | 4 |
| ER−/PgR− | 238 | 25 | 447 | 23 |
| ER+/PgR unknown | 80 | 8 | 158 | 8 |
| ER−/PgR unknown | 5 | 0.5 | 14 | 0.7 |
| ER unknown | 1 | 0.1 | 3 | 0.2 |
| HER2 available | 374 | 38.6 | 731 | 38.1 |
| HER2+ | 95 | 25.4 | 189 | 25.9 |
| HER2− | 279 | 74.6 | 542 | 74.1 |
A, doxorubicin; AC, doxorubicin/cyclophosphamide; A–T, doxorubicin followed by docetaxel; AT, doxorubicin combined with docetaxel; ER, oestrogen receptor; PgR, progesterone receptor; HER2, human epithelial (growth factor) receptor 2; LNs, lymph nodes; T, docetaxel.
The characteristics of the patient population analysed for CNS relapse at the time of death are shown in Table 2. In the control arms (A or AC), 143 patients (14.8%) had died, while in the experimental arms (A–T or AT), 260 (13.5%) patients had died. The rate of CNS relapse was 4.0% in the nondocetaxel-treated (control) patients and 3.7% in the docetaxel-treated (experimental) patients, with CNS being the first site of relapse in 13 (1.3%) and 35 (1.8%), respectively. Among patients deceased at the median follow-up of 5 years, the rate of CNS relapse was 27% for both groups. Information on CNS relapse was available for 393 (97.5%) of the 403 deceased patients.
Table 2.
Patient population analysed for CNS relapse (N = 403)
| Treatment arm | Control arms (no T) |
Experimental arms (T) |
||
| Treatment | A or AC |
A–T or AT |
||
| n | % | n | % | |
| Patients | 968 | 100 | 1919 | 100 |
| Deceased patients | 143 | 14.8 | 260 | 13.5 |
| CNS relapse at death | 39 | 4.0 | 71 | 3.7 |
| CNS relapse as first site | 13 | 1.3 | 35 | 1.8 |
| No CNS relapse at death | 99 | 10.2 | 184 | 9.6 |
| CNS relapse of all deaths | 39/143 | 27.2 | 71/260 | 27.3 |
| CNS relapse unknown | 5/143 | 4.2 | 5/260 | 1.9 |
| CNS relapse in HR+ | 15/81 | 18.5 | 29/141 | 20.6 |
| CNS relapse in HR− | 24/62 | 38.7 | 42/119 | 35.3 |
| Deceased, HER2 result available | 78/143 | 54.5 | 124/260 | 47.7 |
| Deceased, HER2 available, CNS-R unknown | 2/78 | 2.6 | 3/124 | 2.4 |
| CNS relapse in HER2+ | 15/31 | 48.4 | 18/33 | 54.5 |
| CNS relapse in HER2− | 15/45 | 33.3 | 33/88 | 37.5 |
| CNS relapse in triple-negative disease | 11/19 | 57.9 | 18/39 | 46.2 |
HER2 positivity was defined as a 3+ intensity staining in at least 30% of malignant cells by immunohistochemistry. Triple negative disease, ER negative, PgR negative and HER2 negative. HR positivity was defined as ER positive or PgR negative.
CNS, central nervous system; T, docetaxel; A, doxorubicin; AC, doxorubicin plus cyclophosphamide; HR, hormone receptor; ER, oestrogen receptor; PgR, progesterone receptor.
Endocrine responsiveness is known to be inversely related to the rate of CNS relapse [7]. Similarly, in our series among all patients entered in the trial, 44 of 2180 (2%) with hormone receptor-positive tumours were documented with CNS relapse compared with 66 of 707 (9%) of those with hormone receptor-negative tumours. Among deceased patients, the overall rate of CNS relapse was 66 of 181 (36.4%) with hormone receptor-negative tumours compared with 44 of 222 (19.8%) with hormone receptor-positive tumours, while the rates were not different between the treatment arms (Table 2). The rates of CNS relapse were not different between premenopausal patients (57 of 198, 28.8%) and postmenopausal patients (46 of 178, 25.8%).
In recent years, high rates of CNS relapse have been described in HER2-positive breast cancer patients treated with trastuzumab [8–10]. Therefore, we analysed CNS relapse in relation to HER2 status. HER2 analysis was carried out centrally by immunohistochemistry after prospective collection of tissue in the BIG 02-98 trial. Overall HER2 status is currently available for 1105 (38.3%) of the 2887 study patients (Table 1). HER2 positivity was defined as a 3+ intensity staining in at least 30% of malignant cells. HER2 positivity was distributed equally across the treatment arms, with HER2-positive tumours in 25.4% of patients in the control arms and 25.9% in the experimental arms (Table 1). In the patient population analysed for CNS relapse (n = 403), HER2 results were available in half of the patients (50.1%) (Table 2). CNS relapse was significantly more frequent in patients with HER2-positive tumours (33 of 64 = 51.6%) compared with patients with HER2-negative tumours (48 of 133 = 36.1%, P = 0.039). However, there were no differences in CNS relapse between the standard arms and the experimental (docetaxel-containing) arms according to HER2 status. In triple-negative disease (ER negative, PgR negative and HER2 negative), the frequency of CNS relapse overall was 50% (29 of 58), quite similar to HER2 positive disease, again with no difference between the arms (Table 2).
Results on the 110 patients who experienced CNS relapse are shown in Table 3. In both groups, almost all (90%) of these patients had neurological symptoms. As expected, the vast majority of CNS relapses were due to brain metastases (90% in each group). In addition, a minority was diagnosed with meningeal carcinomatosis: 20% in the docetaxel-containing experimental arms and 18% in the control arms. Roughly, one quarter of patients with CNS relapse had CNS as the only site of disease at the time of death: 26% in the experimental arms and 21% in the control arms, further attesting to the considerable clinical importance of CNS relapse. In 54% and 63% of patients who died and had a prior CNS relapse, the CNS relapse was considered the main cause of death. Unexpectedly, the proportion of patients in whom the diagnosis of CNS relapse was made by magnetic resonance imaging (MRI) as compared with computed tomography (CT) scan was somewhat higher in the docetaxel-containing arms (47% by MRI and 66% by CT) than in the control arms (30% MRI, 83% CT). This higher use of MRI in the docetaxel-containing arms would be expected to potentially increase the detection of CNS relapse in this subpopulation, thus strengthening the data showing that there was no increased rate of CNS relapse in the experimental arms. In addition, there was an unexpected increase in positive cerebrospinal fluid cytology in the docetaxel-treated patients 8.5% compared with 3.2% in the control patients. To further elucidate the latter finding, we reviewed the data on 19 patients in whom a diagnosis of meningeal carcinomatosis was made (Table 4). This diagnosis was proven by cytology or histology in six in the taxane-treated patients compared with two in the control patients.
Table 3.
Patients with CNS relapse (N = 110)a
| Treatment arms | Control (no T) |
Experimental (T) |
||
| Treatment | A or AC |
A–T or AT |
||
| n | % | n | % | |
| CNS relapse | 39 | 100 | 71 | 100 |
| Neurological symptoms | 29/32 | 91 | 58/65 | 90 |
| Brain metastases | 34/38 | 90 | 61/67 | 91 |
| Meningeal carcinomatosis | 6/34 | 18 | 13/64 | 20 |
| CNS only disease at death | 8/38 | 21 | 18/68 | 26 |
| CNS-R main cause of death | 24/38 | 63 | 37/68 | 54 |
| CT scan abnormal due to CNS-R | 30/35 | 86 | 39/59 | 66 |
| MRI abnormal due to CNS-R | 10/33 | 30 | 28/60 | 47 |
| CSF abnormal due to CNS-R | 1/31 | 3.2 | 6/59 | 8.5 |
Denominators indicate numbers of patients with information available.
CNS-R, central nervous system relapse; CSF, cerebrospinal fluid; CT, computed tomography; MRI, magnetic resonance imaging; T, docetaxel; A, doxorubicin; AC, doxorubicin plus cyclophosphamide.
Table 4.
Patients with a diagnosis of meningeal carcinomatosis (N = 19)
| Control arms (no T) | Experimental arms (T) | |
| Number of patients | 6 | 13 |
| Method of diagnosis | ||
| By CSF cytology ± MRI ± CT | 1 | 5 |
| By histology ± MRI ± CT | 1 | 1 |
| By MRI ± CT | 2 | 4 |
| By CT scan only | 2 | 2 |
| By physical exam only | 0 | 1 |
| Concurrent brain metastases | 2 | 5 |
CSF, cerebrospinal fluid, CT, computed tomography; MRI, magnetic resonance imaging; T, docetaxel.
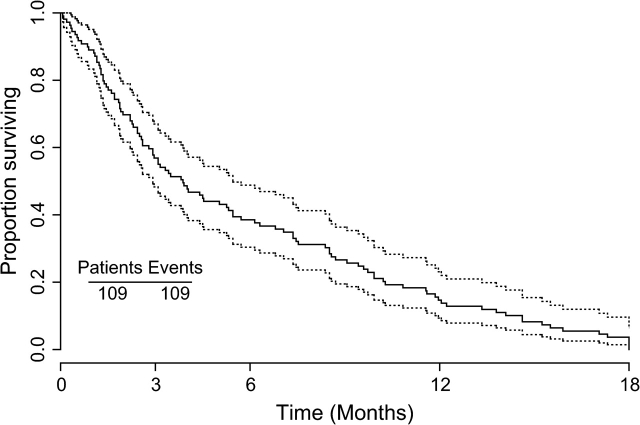
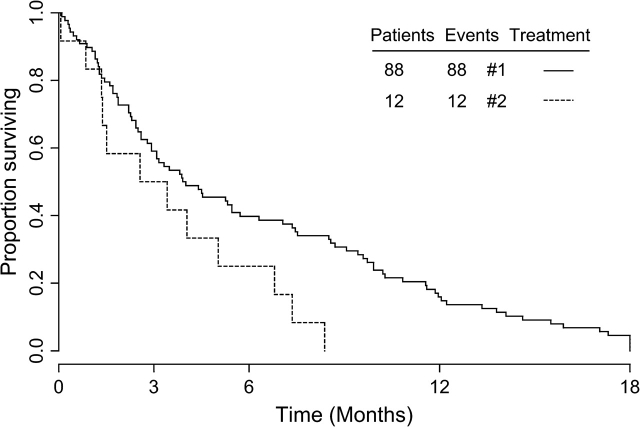
The median survival time for patients after CNS relapse was 4 months, and >80% had died at 1 year (Figure 1). When comparing survival time after CNS relapse of the patients with parenchymal brain metastases only (n = 88, 4.0 months) to patients with meningeal carcinomatosis only (n = 12, 2.6 months), the latter had a significantly worse survival time (Figure 2) (P = 0.0345). (From this analysis, seven patients with both brain metastases and meningeal carcinomatosis were excluded.) Survival for >1 year after CNS relapse was only observed in the subgroup with parenchymal brain metastases.
Figure 1.
Kaplan-Meier curve with 95% confidence band. This figure was constructed from 109 of the 110 patients, because for one patient the date of diagnosis of CNS relapse is unknown.
Figure 2.
Kaplan-Meier curves of time from diagnosis of CNS-relapse to death in patients with brain metastases only (continuous line, N = 88) compared with patients with meningeal carcinomatosis only (dotted line, N = 12). The prognosis of the latter group is significantly worse (p = 0.0451).
discussion
While metastatic breast cancer is usually an incurable disease regardless of the sites involved with distant metastases, CNS relapse is a particularly feared scenario. CNS relapse is almost always accompanied by neurologic symptoms (90% in the present study). In this study, 25% of patients with CNS relapse have died without evidence of extra-CNS metastases. Of all the 403 patients who died within 5 years of adjuvant trial entry, 26 (6.8%) had CNS disease as the only site of relapse. Measured from the time of diagnosis of CNS relapse only, about 20% of patients survived 1 year and were from the subgroup with parenchymal brain metastases only. In this regard, our findings are very consistent with previously published large trials [7]. Similarly, the overall incidence of 4% CNS relapse at 5 years are consistent with published rates of 3.3% at 5 years and 5.4% at 13 years [7]. In large trials, increased rates of CNS relapse were seen in node-positive, ER-negative, high-grade, HER2-positive disease as well as in young patients [7]. Similarly, in our node-positive population, we have found increased rates of CNS relapse in ER-negative, HER2-positive and triple-negative patients. Not surprisingly, after primary chemotherapy for locally advanced or inflammatory breast cancer, CNS relapse has been observed somewhat more frequently: in 61 of 769 (7.9%) patients after a 9.5-year follow-up [8] and in 13 of 140 (9.3%) after a 5-year follow-up [9]. In these single-centre series, similar risk factors for CNS relapse have been described (e.g. young age, ER-negative tumours). In contradistinction to the present study, no comparisons between regimens were possible since a variety of chemotherapy regimens had been employed in these retrospective studies [8,9].
In recent years, high rates of CNS relapse have been observed in smaller studies of patients treated with taxane-based (14% and 30%) [5,6] or with trastuzumab-based (34% and 39%) [10,11,12] therapy for metastatic breast cancer. The reason for these higher rates are unknown, although efficacy in the systemic ‘compartment’ in conjunction with restricted efficacy of these treatments in the CNS ‘compartment’ [11], heterogeneity in tumour cell biology [13] and/or drug interactions with the blood brain barrier can be considered. Therefore, we decided to analyse CNS relapse in the well-defined cohorts of the adjuvant BIG 2-98 trial involving treatment groups with and without a taxane (docetaxel). Overall, we were unable to identify an association of adjuvant docetaxel treatment with an increased frequency of CNS relapse. Around 27% of patients in both groups had CNS relapse at the time of death. With respect to the HER2 status, our results support the findings from the literature: CNS relapse was more frequent in deceased patients with HER2-positive tumours (51.6%) compared with HER2-negative patients (36.1%). However, in neither HER2 subgroup was there a difference in CNS relapse rate between the treatment arms. Therefore, rather than adjuvant docetaxel treatment, it seems to be the biology of HER2-positive tumours that is associated with an increased rate of CNS relapse [13]. On the other hand, it is conceivable that in the future, adjuvant trastuzumab may influence CNS relapse rates in HER2-positive breast cancer since trastuzumab does not cross the blood–brain barrier due to its size [11, 14].
In conclusion, there is no evidence that adjuvant taxane treatment is associated with an increased frequency of CNS relapse. A higher incidence of CNS relapse is confirmed for HER2-positive breast cancer by this prospective study. CNS relapse continues to have a poor prognosis and more so in case of meningeal carcinomatosis than brain metastases.
funding
Australian New Zealand Breast Cancer Trials Group from National Health and Medical Research Council (100925 and 351164); U.S. NCI (CA-73362) to RDG; Swiss Group for Clinical Cancer Research.
Acknowledgments
We thank the patients, physicians, nurses and data managers who participated in BIG 02-98 and the staff at the central offices of the co-operative groups. The following cooperative groups participated: Breast European Adjuvant Studies Team, International Breast Cancer Study Group (including Australian New Zealand Breast Cancer Trials Group), Irish Clinical Oncology Research Group, Grupo Español de Investigacion en Cancer de Mama, Danish Breast Cancer Cooperative Group, Swedish Breast Cancer Group, Austrian Breast and Colorectal Cancer Study Group and Grupo Oncologico Cooperative Chileno De Investigacion.
References
- 1.Early Breast Cancer Trialists Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–1717. doi: 10.1016/S0140-6736(05)66544-0. [DOI] [PubMed] [Google Scholar]
- 2.Goldhirsch A, Wood WC, Gelber RD, et al. Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann Oncol. 2007;18:1133–1144. doi: 10.1093/annonc/mdm271. [DOI] [PubMed] [Google Scholar]
- 3.National Comprehensive Cancer Network. www.nccn.org (5 June 2008, date last accessed) [Google Scholar]
- 4.Francis P, Crown J, Di Leo A, et al. on behalf of the BIG 02-98 Collaborative Group Adjuvant chemotherapy with sequential or concurrent anthracycline and docetaxel: Breast International Group 02-98 randomized trial. J Natl Cancer Inst. 2008;100:121–133. doi: 10.1093/jnci/djm287. [DOI] [PubMed] [Google Scholar]
- 5.Freilich RJ, Seidman AD, DeAngelis LM. Central nervous system progression of metastatic breast cancer in patients treated with paclitaxel. Cancer. 1995;76:232–236. doi: 10.1002/1097-0142(19950715)76:2<232::aid-cncr2820760212>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- 6.Crivellari D, Pagani O, Veronesi A, et al. High incidence of central nervous system involvement in patients with metastatic or locally advanced breast cancer treated with epirubicin and docetaxel. Ann Oncol. 2001;12:353–356. doi: 10.1023/a:1011132609055. [DOI] [PubMed] [Google Scholar]
- 7.Pestalozzi BC, Zahrieh D, Price KN, et al. for the International Breast Cancer Study Group (IBCSG) Identifying breast cancer patients at risk for central nervous system (CNS) metastases in trials of the International Breast Cancer Study Group (IBCSG) Ann Oncol. 2006;17:935–944. doi: 10.1093/annonc/mdl064. [DOI] [PubMed] [Google Scholar]
- 8.Gonzales-Angulo AM, Cristofanilli M, Strom EA, et al. Central nervous system metastases in patients with high-risk breast carcinoma after multimodality treatment. Cancer. 2004;101:1760–1766. doi: 10.1002/cncr.20530. [DOI] [PubMed] [Google Scholar]
- 9.Carey LA, Ewend MG, Metzger R, et al. Central nervous system metastases in women after multimodality therapy for high risk breast cancer. Breast Cancer Res Treat. 2004;88:273–280. doi: 10.1007/s10549-004-0999-3. [DOI] [PubMed] [Google Scholar]
- 10.Bendell JC, Domchek S, Burstein HJ, et al. Central nervous system metastases in women who receive trastuzumab-based therapy for metastatic breast carcinoma. Cancer. 2003;97:2972–2977. doi: 10.1002/cncr.11436. [DOI] [PubMed] [Google Scholar]
- 11.Burstein HJ, Lieberman G, Slamon DJ, et al. Isolated central nervous system metastases in patients with HER2-overexpressing advanced breast cancer treated with first-line trastuzumab-based therapy. Ann Oncol. 2005;16(11):1772–1777. doi: 10.1093/annonc/mdi371. [DOI] [PubMed] [Google Scholar]
- 12.Dawson SJ, Ranieri NF, Snyder RD, et al. Central nervous system metastases in women with HER-2 positive metastatic breast cancer after treatment with trastuzumab. Asia Pac J Clin Oncol. 2006;2:50–56. [Google Scholar]
- 13.Minn AJ, Kang Y, Serganova I, et al. Distinct organ-specific metastatic potential of individual breast cancer cells and primary tumors. J Clin Invest. 2005;115:44–55. doi: 10.1172/JCI22320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pestalozzi BC, Brignoli S. Trastuzumab in CSF. J Clin Oncol. 2000;18:2350–2351. doi: 10.1200/JCO.2000.18.11.2349. [DOI] [PubMed] [Google Scholar]