Abstract
A total of 198 sera from stray cats was assayed against Toxoplasma gondii antigen by western blot. Out of 198 sera assayed, 26 sera (13.1%) showed typical blot patterns against T. gondii. When spotted by ELISA absorbance and indirect latex agglutination test (ILAT) titer, all 26 cases were distributed over the cut-off value of ELISA whereas 24 cases (92.3%) were in the positive range of 1:32 or higher and 2 cases in negative range by ILAT. Among western blot negative 172 sera, 162 cases were negative in both ILAT and ELISA while 10 cases were reactive falsely such that three cases were ILAT positive with 1:32 titer and 9 cases were ELISA positive (2 cases overlapped). These 10 cases reacted peculiarly without typical binding pattern in Western blot. Sandwich-ELISA was performed with monoclonal antibodies (mAbs) of Tg563 (30 kDa, SAG1), Tg505 (22 kDa, SAG2), Tg605 (43 kDa, SAG3), Tg556 (28 kDa, GRA2), Tg737 (32 kDa, GRA6), Tg695 (66 kDa, ROP2), Tg786 (42 kDa, ROP6), and Tg621 (32 kDa, anonymous but cytosolic) clone, respectively. All western blot-positive cases were in the positive range and negative cases in the negative range clearly. Among the 10 false reactive cases, 3 cases were in the positive range with one or more mAbs. All mAbs used in this study were confirmed to be specific to T. gondii infection as a standardized sandwich-ELISA to differentiate it from other pathogens.
Keywords: Toxoplasma gondii, stray cat, western blot, ILAT, ELISA, sandwich-ELISA, monoclonal antibodies
INTRODUCTION
Toxoplasma gondii is an obligate intracellular protozoan parasite which causes congenital defects or abortion and fatal diseases in immune compromised patients. As many as 15-85% of individuals in adult human population show serological evidence of T. gondii infection world-widely (Jackson and Hutchison, 1989). The ingestion of tissue cysts in undercooked meat from intermediate hosts or the ingestion of food or water contaminated with oocysts from feces of definitive hosts are the two major modes of transmission (Dubey et al., 1997). Experimental transmission of T. gondii by certain Felidae (Jewell et al., 1972) and high prevalence of antibody against T. gondii in animals of Felidae (Choi et al., 1987) supported the possible candidates of these animals as the final hosts of T. gondii indirectly. But only cat has been confirmed as unique definitive host (Frenkel et al., 1970) that can excrete the environmentally resistant oocysts until now.
Pet cats recently infected may shed large numbers of oocysts in the household, thereby putting their owners at risk from the infection (Pereira et al., 1992). Cats allowed to roam free or stray cats may contaminate the environment with oocysts which may infect human or livestock (Tenter et al., 1994). Therefore, cats play a key role in the epidemiology of toxoplasmosis and the availability of a reference test for the detection of T. gondii infection in cats are needed. We demonstrate here the seroprevalence of stray cats by western blot analysis against T. gondii, comparison of western blot with ELISA and indirect latex agglutination test (ILAT), and the diagnostic availability of sandwich-ELISA with monoclonal antibodies.
MATERIALS AND METHODS
Antigen preparation
RH tachyzoites of T. gondii were maintained by a peritoneal passage in inbreeding mice (ICR strain) and purified by centrifugation over 40% Percoll (Amersham Pharmacia Biotech, Uppsala, Sweden) in PBS solution (Choi et al., 1992).
Sera
A total of 198 sera from stray cats was collected from 1996 to 1998 at suburban and rural areas near Chinju City, Gyeongsangnam-do and was freezed at -70℃ until use. Sex and age of cats were not identified.
Western blot, ELISA and ILAT
Western blot was done by the method of Towbin et al. (1979). The whole RH extracts were separated in 12% SDS-PAGE gels and transferred onto nitrocellulose (NC, Schlleicher and Shuell, Keene, NH, USA) papers. NC papers were incubated with sera of 1:200 dilution, and then with 1:5,000 diluted HRP-conjugated goat anti-cat IgG antibody (Jackson ImmunoResearch Lab, West Grove, PA, USA). They were soaked in enhanced chemiluminescence (ECL) solution (Amersham Pharmacia Biotech) for 1 min and exposed to an X-ray film (Konica, Tokyo, Japan) for 5 to 10 sec.
ELISA was performed according to the method described previously (Choi et al., 1992) except the serum dilution of 1:500 and HRP-conjugate dilution of 1:10,000. ILAT was done by the method described in a kit manual (Eiken Toxo-kit, Tokyo, Japan) and titers of 1:32 or higher were regarded as positives.
Sandwich-ELISA with monoclonal antibodies
For the selection of monoclonal antibodies (mAbs) to be used in sandwich-ELISA, mAbs were confirmed by immunofluorescence assay (IFA) and western blot. Freshly prepared RH tachyzoites cytospinned onto 18-mm coverslips and Vero cells (CRL 6318, American Type Culture Collection, Rockville, MD, USA) cultured on coverslips infected with RH tachyzoites for 24 hr were fixed either with cold absolute methanol for 5 min or with paraformaldehyde for 10 min and then permeabilized with Triton X-100 for 5 min, separately. mAbs were applied with 1:100 dilution in incubation solution (3% BSA in PBS) and coverslips were incubated with 1:500 diluted FITC-conjugated goat anti-mouse IgG antibody (Sigma Chem Co., St. Louis, MO, USA). Fluorescence was observed under a fluorescence microscopy (Axiophot, Carl Zeiss Co., Oberkochen, Germany).
Sandwich-ELISA was done as follows. Subcellular organelle (surface membrane, dense granule, rhoptry, and cytosolic) specific mAbs were coated onto a 96-well EIA plate (Costar, Dover, NH, USA) with 1:100 dilution in coating buffer (50 mM carbonate buffer, pH 9.6) overnight at 4℃. After blocking with 5% skim milk in PBS/0.05% Tween-20 (PBS/T) for 1 hr, RH tachyzoite extracts of 200 µl were incubated with 10 µg/ml in blocking solution for 2 hr. After then conventional ELISA procedure was adapted as described above except serum dilution of 1:200 and HRP-conjugate dilution of 1:10,000.
RESULTS
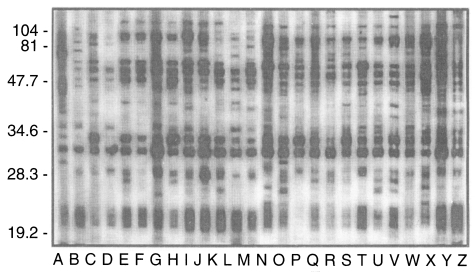
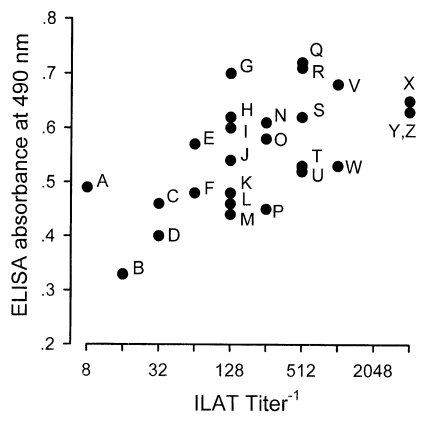
Typical western blot patterns against T. gondii were found in 26 sera (13.1%) out of 198 stray cat sera. The patterns revealed a set of major surface protein reacting bands with molecular weights of 30 kDa (SAG1, Mineo et al., 1993) and 22 kDa (SAG2, Parmley et al., 1992), 22-30 kDa dense granular proteins (Cesbron-Delauw, 1994), and 50-70 kDa rhoptry proteins (Sam-Yellowe, 1996) with some minor bands (Fig. 1). When spotted by ILAT titer and ELISA absorbance as shown in Fig. 2, 24 cases (92.3%) were in the positive range of 1:32 or higher and 2 cases (lane A and B) in reactive but negative range by ILAT, and all 26 cases were distributed over cut-off value of 0.275 calculated as mean+3 SD of 124 ILAT-not-reactive cases (mean, 0.140l; SD, 0.045) by ELISA.
Fig. 1.
Western blot positive sera of 26 cases (A to Z) showing typical binding patterns of Toxoplasma gondii infection. Numerals indicat MW markers in kDa.
Fig. 2.
Distribution of western blot positive sera by ILAT and ELISA. Each serum was from the same lane in Fig. 1 as labelled alphabetically.
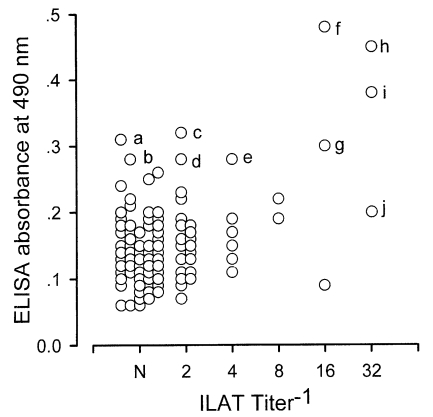
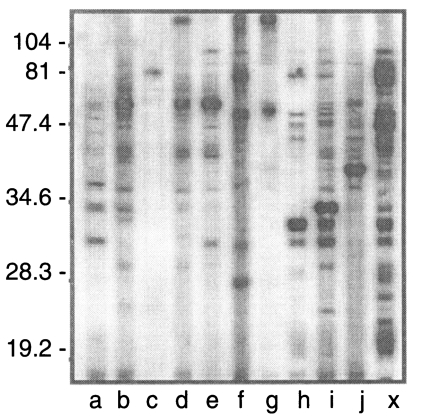
Western blot negative 172 sera were distributed as shown in Fig. 3. Three cases (h, i, and j) were ILAT positive (1:32) and 9 cases (a to i) were ELISA positive (over 0.275) with 2 cases positive by both ILAT and ELISA (h and i). Western blot of these 10 cases were presented in Fig. 4 with one positive serum (lane X) in addition. Each serum showed various binding patterns with RH antigens but not typical as described above.
Fig. 3.
Distribution of western blot negative sera by ILAT and ELISA. Ten cases (a to j) were located in positive ranges of ILAT titers over 1:32 or ELISA absorbance over 0.275.
Fig. 4.
Western blot patterns of 10 cases (a to j) described in Fig. 3. Lane X shows positive bands as a standard.
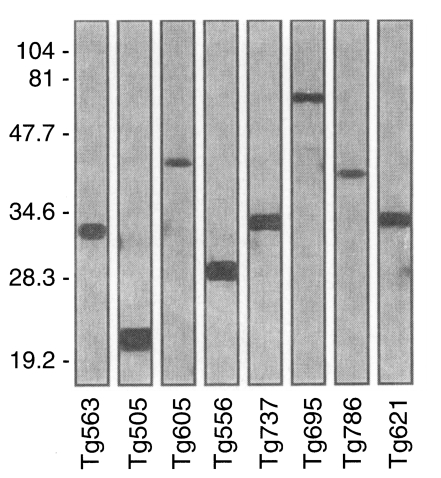
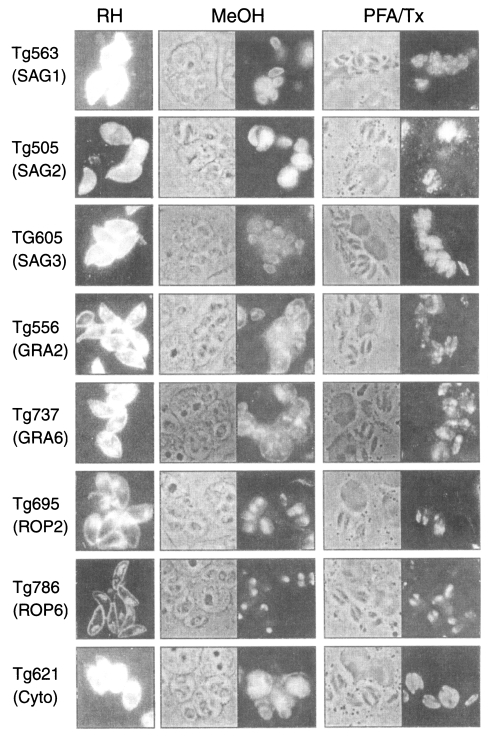
To clarify the reactivity of these sera in western blot, ELISA or ILAT, we performed sandwich-ELISA with mAbs which were confirmed by western blot (Fig. 5) and IFA (Fig. 6). Tg563 clone detected 30 kDa protein in western blot and surface membrane in extracellular RH (designated by RH in Fig. 5) and both in methanol fixed intracellular RH (designated by MeOH) and PFA/Tx, which represented SAG1. Tg505 and Tg605 clones also showed similar IFA patterns with Tg563 clone except for positive reaction with 22 kDa protein (SAG2) and 43 kDa (SAG3) by western blot, respectively. Both Tg556 and Tg737 clones labelled extracellular and intracellular granules with parasitophorous vacuolar membrane (PVM), but immunoblotting detection of Tg556 was 28 kDa (GRA2) and that of Tg737 was 32 kDa (GRA6), respectively. Tg695 and Tg786 clones labelled rhoptry proteins of 66 kDa ROP2 and 42 kDa ROP6. And Tg621 clone labelled unidentified cytosolic 32 kDa protein which was secreted into PVM.
Fig. 5.
Western blot of monoclonal antibodies against RH antigen chosen for sandwich-ELISA. Numerals indicate MW markers in kDa.
Fig. 6.
Immunofluorescence images of monoclonal antibodies of Fig. 5 on the extracellular RH (RH column) and the intracellular RH (MeOH and PFA/Tx column). × 1,000.
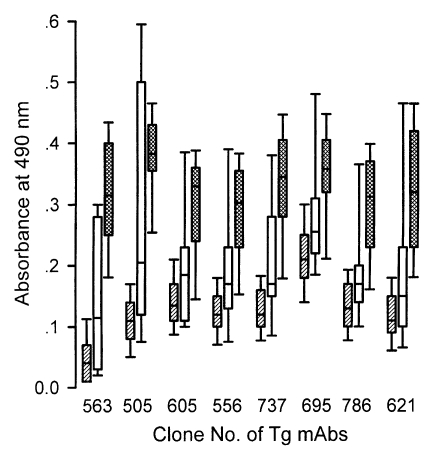
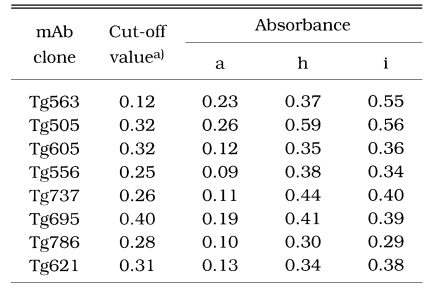
By the sandwich ELISA with mAbs (Fig. 7), 124 ILAT-not-reactive cases were distributed as negative and all 26 western blot-positive cases were in the positive range clearly with cut-off value calculated by the absorbance using each mAb. Among the 10 false reactive cases, 7 cases were revealed to be in the negative range but 3 cases (lane a, h, and i) were in the positive range with one or more mAbs showing high absorbance as summarized in Table 1.
Fig. 7.
Distributional pattern of sandwich-ELISA with various subcellular organelle specific monoclonal antibodies to cat sera. Monoclonal clones were derived from Fig. 5. Hatched boxes indicate the negative sera, blank boxes, false reactive sera of Fig. 4, and cross hatched boxes, positive sera, respectively.
Table 1.
Sandwich-ELISA absorbance of 3 false reactive cases (lane a, h and i) in Fig. 4.
a)Cut-off value of each clone was calculated as mean + 3SD of negative sera.
DISCUSSION
A total of 26 cases (13.1%) out of 198 stray cats was detected to be seropositive against T. gondii by western blot. This low seropositive rate was comparable to the European and American regions with high seroprevalence rate approximately over 40% in cats (Frenkel et al., 1995; Hill et al., 1998). Since cats as the final host of T. gondii are related directly with T. gondii transmission to human by shedding of oocysts (Choi et al., 1997), low seroprevalence rate among cats in this study may reflect low seroprevalence rate of T. gondii antibodies in Korean people of 2 to 10% according to the group tested (Choi et al, 1989, 1992). The prevalence of T. gondii infection varies according to the life style of cats such that it is higher in feral cats than those in domestic cats (Dubey, 1973; Nogami et al., 1998). But pet cats are exposed to be free-ranging stage easily, it is suggested that pet cats are not much less infected with T. gondii than stray cats in Korea.
The western blot patterns of T. gondii infection has been established well in such reactions with major surface membrane proteins, dense granular proteins, and rhoptry proteins. Therefore, it is a method of choice to detect antibodies in the sera of infected individuals. All 26 cases of western blot positive were detected as positive in ELISA, but 2 cases were missed by ILAT (Fig. 2). However, out of 172 western blot negative sera, 9 cases were in the range of positive by ELISA and 3 cases were positive ILAT titer of 1:32 (two cases were positive by both ELISA and ILAT). Western blot of these 10 cases revealed peculiar patterns not comparable to each other. Irregular reactivities might be resulted from non-specific bindings or the epitopic similarity of T. gondii antigens with other pathogenic infections common to cats such as Isospora felis (Omata et al., 1991), Hammondia sp. (Hendricks et al., 1979), and feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) antigens (Svobodova et al., 1998) especially.
Indeed, mAbs used in sandwich ELISA seemed to have equivalent sensitivity to the conventional ELISA using specific antigens purified or prepared by recombinant expression of p30 (Lunden et al., 1993; Nam et al., 1996), p22 (Parmley et al., 1992), and GRA4 (Mevelec et al., 1992). All mAbs specifically labelled subcellular organelles each other and clearly discriminated negatives from positives as depicted in Fig. 7. When applied to the 10 false reactive sera by western blot, 7 cases were defined as negative and 3 cases were still positive (Table 1). In case of lane a of Fig. 4, only Tg506 clone reacted in sandwich-ELISA, which suggested the serum could detect p30 with several other bands. And in cases of h and i of Fig. 4, all the mAbs reacted strongly despite they did not react in western blot clearly. Therefore, it is suggested to be elicited during the course of infection because there are no differences in binding patterns between surface membrane proteins and subcellular organelles.
All mAbs used in this study need further investigations to be more specific to T. gondii infection as a standardized sandwich-ELISA to differentiate it from other pathogens.
ACKNOWLEDGEMENT
We thank Mr. Nam-Seok Lee and Ms. Anna Lee of Department of Parasitology, Catholic University of Korea, College of Medicine for their skillful assistances.
Footnotes
This study was supported by the grant from Korea Science and Engineering Foundation (KOSEF-961-0716-101-1).
References
- 1.Cesbron-Delauw MF. Dense granule organelles of Toxoplasma gondii: their role in the host-parasite relationship. Parasitol Today. 1994;10:293–296. doi: 10.1016/0169-4758(94)90078-7. [DOI] [PubMed] [Google Scholar]
- 2.Choi WY, Nam HW, Youn JH, Kim WS, Kim WK. Toxoplasma antibody titers by indirect latex agglutination test in patients of Kangnam St. Mary's Hospital and Cheju Medical Center. Korean J Parasitol. 1989;27:171–175. doi: 10.3347/kjp.1989.27.3.171. [DOI] [PubMed] [Google Scholar]
- 3.Choi WY, Nam HW, Kwak NH, et al. Foodborne outbreaks of human toxoplasmosis. J Inf Dis. 1997;175:1280–1282. doi: 10.1086/593702. [DOI] [PubMed] [Google Scholar]
- 4.Choi WY, Nam HW, Youn JH, et al. Detection of antibodies in serum and cerebrospinal fluid to Toxoplasma gondii by indirect latex agglutination test and enzyme-linked immunosorbent assay. Korean J Parasitol. 1992;30:83–90. doi: 10.3347/kjp.1992.30.2.83. [DOI] [PubMed] [Google Scholar]
- 5.Choi WY, Yoo JE, Nam HW, et al. Toxoplasma antibodies by indirect latex agglutination tests in zoo animals. Korean J Parasitol. 1987;25:13–23. doi: 10.3347/kjp.1987.25.1.13. [DOI] [PubMed] [Google Scholar]
- 6.Dubey JP. Feline toxoplasmosis and coccidiosis: a survey of domiciled and stray cats. J Am Vet Med Assoc. 1973;162:873–878. [PubMed] [Google Scholar]
- 7.Dubey JP, Speer CA, Shen SK, Kwok OCH, Blixt JA. Oocyst-induced murine toxoplasmosis: life cycle, pathogenicity, and stage conversion in mice fed Toxoplasma gondii oocysts. J Parasitol. 1997;83:870–882. [PubMed] [Google Scholar]
- 8.Frenkel JK, Dubey JP, Miller NL. Toxoplasma gondii in cats: fecal stages identified as coccidian oocysts. Science. 1970;167:893–896. doi: 10.1126/science.167.3919.893. [DOI] [PubMed] [Google Scholar]
- 9.Frenkel JK, Hassanein KM, Hassanein RS, Brown E, Thulleiz P, Quintero-Nunez R. Transmission of Toxoplasma gondii in Panama City, Panama: a five-year prospective cohort study of children, cats, rodents, birds, and soil. Am J Trop Med Hyg. 1995;53:458–468. doi: 10.4269/ajtmh.1995.53.458. [DOI] [PubMed] [Google Scholar]
- 10.Hendricks LD, Ernst JV, Courtney CH, Speer CA. Hammondia parsalis sp. n. (Sarcocystidae) from the ocelot, Felis parsalis, and experimental infection of other felines. J Protozool. 1979;26:39–43. [Google Scholar]
- 11.Hill RE, Zimmerman JJ, Wills RW, Patton S, Clark WR. Seroprevalence of antibodies against Toxoplasma gondii in free-ranging mammals in Iowa. J Wildl Dis. 1998;34:811–815. doi: 10.7589/0090-3558-34.4.811. [DOI] [PubMed] [Google Scholar]
- 12.Jackson MH, Hutchison WM. The prevalence and source of Toxoplasma infection in the environment. Adv Parasitol. 1989;28:55–105. doi: 10.1016/s0065-308x(08)60331-0. [DOI] [PubMed] [Google Scholar]
- 13.Jewell ML, Frenkel JK, Johnson KM, Reed V, Ruiz A. Development of Toxoplasma oocysts in neotropical felidae. Am J Trop Med Hyg. 1972;21:512–517. doi: 10.4269/ajtmh.1972.21.512. [DOI] [PubMed] [Google Scholar]
- 14.Lunden A, Lovgren K, Uggla A, Araujo FG. Immune responses and resistance to Toxoplasma gondii in mice immunized with antigens of the parasite incorporated into immunostimulating complexes. Infect Immun. 1993;61:2639–2643. doi: 10.1128/iai.61.6.2639-2643.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mevelec MN, Chardes T, Mercereau-Puijalon O, et al. Molecular cloning of GRA4, a Toxoplasma gondii dense granule protein, recognized by mucosal IgA antibodies. Mol Biochem Parasitol. 1992;56:227–238. doi: 10.1016/0166-6851(92)90172-g. [DOI] [PubMed] [Google Scholar]
- 16.Mineo JR, McLeod R, Mack D, et al. Antibodies to Toxoplasma gondii major surface protein (SAG1, p30) inhibit infection of host cells and are produced in murine intestine after peroral infection. J Immunol. 1993;150:3951–3964. [PubMed] [Google Scholar]
- 17.Nam HW, Im KS, Baek EJ, Choi WY, Cho SY. Analysis of antigenic domain of GST fused major surface protein (p30) fragments of Toxoplasma gondii. Korean J Parasitol. 1996;34:135–141. doi: 10.3347/kjp.1996.34.2.135. [DOI] [PubMed] [Google Scholar]
- 18.Nogami S, Moritomo T, Kamata H, et al. Seroprevalence against Toxoplasma gondii in domiciled cats in Japan. J Vet Med Sci. 1998;60:1001–1004. doi: 10.1292/jvms.60.1001. [DOI] [PubMed] [Google Scholar]
- 19.Omata Y, Oikawa H, Kanda M, et al. Humoral immune response to Isospora felis and Toxoplasma gondii in cats experimentally inoculated with Isospora felis. J Vet Med Sci. 1991;53:1071–1073. doi: 10.1292/jvms.53.1071. [DOI] [PubMed] [Google Scholar]
- 20.Parmley SF, Sgarlato GD, Mark J, Prince JB, Remington JS. Expression, characterization, and serologic reactivity of recombinant surface antigen p22 ofToxoplasma gondii. J Clin Microbiol. 1992;30:1127–1133. doi: 10.1128/jcm.30.5.1127-1133.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pereira LH, Staudt M, Tanner CE, Embil JA. Exposure to Toxoplasma gondii and cat ownership in Nova Scotia. Pediatrics. 1992;89:1169–1172. [PubMed] [Google Scholar]
- 22.Sam-Yellowe TY. Rhoptry organelles of the Apicomplexa: their role in host cell invasion and intracellular survival. Parasitol Today. 1996;12:308–316. doi: 10.1016/0169-4758(96)10030-2. [DOI] [PubMed] [Google Scholar]
- 23.Svobodova V, Knotek Z, Svoboda M. Prevalence of IgG and IgM antibodies specific to Toxoplasma gondii in cats. Vet Parasitol. 1998;80:173–176. doi: 10.1016/s0304-4017(98)00201-5. [DOI] [PubMed] [Google Scholar]
- 24.Tenter AM, Vietmeyer C, Johnson AM, Janitschke K, Rommel M, Lehmacher W. ELISAs based on recombinant antigens for seroepidemiological studies on Toxoplasma gondii infections in cats. Parasitology. 1994;109:29–36. doi: 10.1017/s0031182000077738. [DOI] [PubMed] [Google Scholar]
- 25.Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA. 1979;76:4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]