Abstract
Significant morbidity and mortality can be attributed to inflammatory diseases; therefore, a greater understanding of the mechanisms involved in the progression of inflammation is crucial. Here we demonstrate that p21(WAF1/CIP1), an established suppressor of cell cycle progression, is an inhibitor of IL-1β synthesis in macrophages. Mice deficient for p21 (p21−/−) display increased susceptibility to endotoxic shock which is associated with increased serum levels of IL-1β. Administration of IL-1 receptor antagonist reduces LPS-induced lethality in p21−/− mice. Analysis of isolated macrophages, which are one of the central producers of IL-1β, reveals that deficiency for p21 led to more IL-1β mRNA and pro-protein synthesis following toll-like receptor (TLR) ligation. The increase in IL-1β pro-protein is associated with elevated secretion of active IL-1β by p21−/− macrophages. siRNA-mediated knockdown of p21 in human macrophages results in increased IL-1β secretion as well. A peptide mapping strategy shows that the cyclin dependent kinase (CDK) binding domain of p21 is sufficient to reduce the secretion of IL-1β by p21−/− macrophages. These data suggest a novel role for p21 and specifically for the CDK binding domain of p21(WAF1/CIP1) in inhibiting inflammation.
Keywords: p21, macrophages, IL-1β, endotoxic shock, inflammation
INTRODUCTION
The pleiotropic cytokine IL-1β is required for the progression of numerous inflammatory disorders [1]. As key components of the innate immune response, monocytes and macrophages play an important role in the initiation and/or progression of inflammatory disease, largely through secretion of IL-1β, which contributes significantly to pathogenesis. Current treatments for diseases such as rheumatoid arthritis, gout, and periodic fever syndromes include the IL-1 receptor antagonist (IL-1Ra) anakinra (Kineret®, Amgen) [2], yet this therapy is ineffective in a cohort of patients [3]. Therefore, a better understanding of the mechanisms by which IL-1β production is regulated remains crucial for the development of new therapies to treat inflammatory diseases.
Here, we investigate the role that p21(WAF1/CIP1) (p21) plays in the production of IL-1β and development of inflammatory disease. As a member of the Cip/Kip family of cyclin dependent kinase (CDK) inhibitors, p21 induces blockade of cell cycle progression via inhibition of the activity of CDK/cyclin complexes as well as that of proliferating cell nuclear antigen (PCNA) [4]. p21 binds to CDKs and cyclins through two distinct domains on the N-terminus and to PCNA via its C-terminus [5]. Mice deficient for p21 show no developmental or reproductive abnormalities, although the mice display sensitivity to radiation [6] and develop a form of lupus-like disease [6–8]. Furthermore, a paucity of p21 expression is observed in synovial tissue from rheumatoid arthritis patients as compared to osteoarthritis controls [9]. Because of the implication of p21 in the pathogenesis of lupus and rheumatoid arthritis and the importance of IL-1β in these and other inflammatory diseases, we sought to investigate the role of p21 in this pathway. In this study, we identify p21 as a novel candidate for the treatment of IL-1β mediated pathologies.
RESULTS AND DISCUSSION
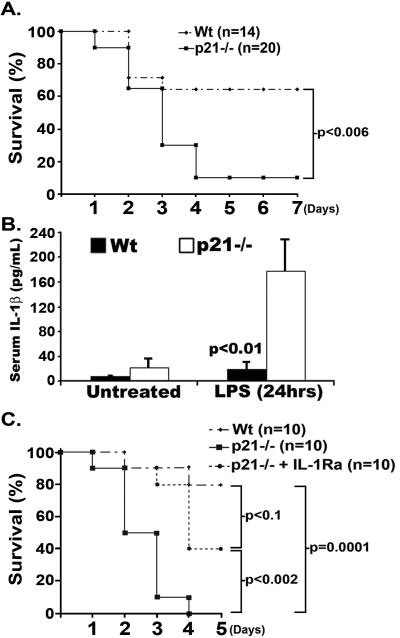
p21−/−mice are more susceptible to endotoxic shock
We examined the effect of p21 deficiency on inflammatory disease, using the LPS-induced endotoxic shock model. Mice were challenged intraperitoneally with LPS (10 mg/kg of body weight) and a 53% and 85% decrease in survival was observed in p21−/− mice at day 3 and at days 4–7 (p<0.006), respectively, as compared to WT mice (Figure 1A). Because previous studies demonstrate that release of active IL-1β is required for LPS-induced endotoxic shock [10], we examined the in vivo production of IL-1β. A 9.5-fold (p<0.01) increase in serum IL-1β was detected in p21−/− mice as compared to WT mice at 24 hours post-injection (Figure 1B). IL-6 and TNFα levels were also elevated in p21−/− mice as compared to WT mice (Supporting Information Figure S1). Administration of anakinra significantly increased the survival of p21−/− mice following endotoxic shock as compared to mock-treated p21−/− mice (Figure 1C), consistent with the concept that IL-1β plays a pivotal role in LPS-induced shock. However, since a subset of these mice did not survive, these data suggest that anakinra incompletely blocks IL-1β signaling or that other cytokines such as TNFα may contribute to lethality. Our data showing elevated TNFα in serum from LPS-treated p21−/− mice (Supporting Information Figure S1) support this notion and corroborate previous studies [11].
Figure 1. p21−/− mice develop severe LPS-induced endotoxic shock.
(A) Survival was monitored over seven days in Wt (n=14) or p21−/− (n=20) mice intraperitoneally injected with LPS (10mg/kg of body weight). Data shown are representative of two independent combined experiments. Values represent the percent survival compared by chi square analysis. (B) Circulating levels of IL-1β were measured in serum from Wt or p21−/− mice following LPS injection as in (A). Serum was isolated at 0 (WT, n=12; p21−/−, n=12) and 24 (WT, n=6; p21−/−, n=13) hours post injection and IL-1β levels were analyzed using a Luminex based assay. Data shown are representative of two combined independent experiments. Values show mean ± SE (Student's t-test). (C) Survival following treatment with LPS as in (A) was monitored over five days in Wt (n=10) or p21−/− (n=10) mice or a cohort of p21−/− mice (n=10) treated with anakinra as described in the Materials and methods. Data shown are representative of an individual experiment. Values represent the percent survival compared by chi square analysis.
Deficiency for p21 leads to enhanced expression of IL-1β by macrophages
Because macrophages are one of the major producers of IL-1β, we examined whether deficiency for p21 affects IL-1β production in these cells. The expression of IL-1β message was 3.4-fold higher (p<0.04) in p21−/− bone marrow derived macrophages (BMDM) as compared to WT cells at 360 minutes following stimulation with LPS (Figure 2A). Immunoblot analysis revealed an increase in pro-IL-1β protein levels in p21−/− BMDM as compared to WT cells at 12 hours post-LPS stimulation (Figure 2B). Further, IL-6 and TNFα synthesis were increased in p21−/− macrophages as compared to WT cells (Supporting Information Figure S2). Secretion of IL-1β requires cleavage of the pro-protein by caspase-1 which is activated by the inflammasome [10]. BMDM were stimulated with TLR agonists and then activated with ATP, which has been shown to activate the P2X7 receptor and pannexin channel leading to inflammasome assembly [12]. Upon incubation with PAM3CSK4 (data not shown) or LPS, p21−/− BMDM secreted mature IL-1β as early as 7.5 minutes post-ATP stimulation (35.0-fold increase, p<0.04) and levels remained substantially higher as compared to WT macrophages (Figure 2C). The secreted IL-1β was biologically active, as there was a substantial increase of bioactive IL-1β in supernatants from p21−/− BMDM as compared to WT cells (Figure 2D). The P2X7 receptor and pannexin channel functioned similarly regardless of the status of p21 (Supporting Information Figure S3). Gout crystals, which do not require ATP or the P2X7 receptor for IL-1β release [13], led to increased secretion of IL-1β from LPS primed p21−/− macrophages as compared to WT cells (data not shown). Further, there were no differences in mRNA levels in any members of the inflammasome in WT or p21−/− macrophages (data not shown). Taken together, these data suggest that the increased kinetics of IL-1β release may reflect a role for p21 in suppressing the inflammasome, though not at the early stages of P2X7-mediated activation of the complex or at the expression level of individual components.
Figure 2. Increased production of IL-1β in p21−/− macrophages.
(A) IL-1β mRNA levels were analyzed by RT-PCR (performed in duplicate and normalized to GAPDH) following LPS (10 ng/mL) stimulation and RNA harvest from triplicate BMDM cultures per genotype. Shown is the fold increase for IL-1β expression normalized to unstimulated expression levels. (B) Pro-IL-1β protein and GAPDH levels were assayed in protein extracts isolated 12 hours following LPS stimulation (10 ng/mL) by immunoblot. (C) IL-1β secretion by BMDM from WT or p21−/− mice stimulated with LPS (10 ng/mL) and ATP (5 mM) as described in the Materials and methods was measured by ELISA. Supernatants were collected from at least three individual wells per genotype per timepoint and data were normalized to the number of cells per well. (D) Secretion of biologically active IL-1β by Wt or p21−/− macrophages stimulated as in (C) was assayed as described in the Materials and methods. (E) IL-1β secretion by Wt or p21−/− peritoneal macrophages treated as described in (C) was analyzed by ELISA. (F) IL-1β secretion by human macrophages transfected with non-specific or p21 siRNA as described in the Materials and methods and stimulated with LPS and ATP as in (C) was measured by ELISA. Knockdown of p21 was confirmed by western blot analysis (insert). (A-F) Data shown are representative of at least two independent experiments. Values represent the mean ± SE per time point, compared by Student's t-test (*p<0.04).
The reduction in IL-1β production in WT cells is not dependent on p21-mediated inhibition of the cell cycle. Peritoneal macrophages, which have terminally withdrawn from the cell cycle, showed a 2.4-fold increase in secreted IL-1β by p21−/− macrophages as compared to WT cells (Figure 2E). In addition, primary human macrophages, which are also non-proliferative, secreted increased amounts of IL-1β after LPS/ATP stimulation when transfected with p21 siRNA, as compared to cells treated with non-specific siRNA (Figure 2F). Transfection of p21 siRNA resulted in a 3.4-fold (p<0.01) reduction in p21 mRNA (data not shown) which correlated with lower p21 protein levels (Figure 2F, insert). Taken together, these data suggest that deficiency for p21 results in enhanced IL-1β secretion irrespective of its proliferative capacity or origin of the macrophage.
p21-mediated suppression of IL-1β requires the CDK binding domain of p21
We next sought to elucidate which domain of p21 is important in the suppression of IL-1β production. Similar to previous studies, we generated peptides corresponding to the cyclin, CDK, and PCNA binding domains of p21 (Figure 3A) [5], which were conjugated to a polycationic peptide derived from HIV-1 TAT to allow entry into macrophages [14]. p21−/− BMDM treated with cyclin, CDK, or PCNA binding domain peptides all displayed a similar reduction in cell number as compared to untreated cells, while viability remained unaffected (data not shown). In contrast, only the p21-peptide mimetic to the CDK binding domain (aa 46–65) was sufficient to suppress IL-1β secretion, with 59% (p<0.08) and 79% (p<0.01) reduction at 30 and 120 minutes post-ATP treatment, respectively, as compared to LPS/ATP-treated p21−/− macrophages without peptide treatment (untreated) (Figure 3B). These data demonstrate for the first time a novel role for the CDK domain of p21 as an inhibitor of IL-1β production and suggest a potential therapeutic use for p21 CDK domain peptides. Future studies will be necessary to identify the factors which interact with the CDK domain of p21 and mediate this function. Interestingly, small molecule CDK inhibitors were recently shown to have efficacy in the treatment of collagen-induced arthritis [15], a model of inflammation which requires IL-1β secretion [16]. Furthermore, injection of adenovirus expressing p21 into the joints of mice ameliorates collagen-induced arthritis [17]. These data, combined with the data presented here, suggest that p21 mimetics may be beneficial in the treatment of inflammatory diseases.
Figure 3. The CDK binding domain of p21 is required to suppress IL-1β synthesis.
(A) Schematic of the cyclin, CDK, and PCNA binding domains on p21 protein. (B) Secretion of IL-1β by p21−/− BMDM pretreated with saline or p21-mimetic peptide (10 μM) for two hours, stimulated with LPS (10 ng/mL) in media containing saline or p21-mimietic peptide for 12 hours, and activated with ATP as described in the Materials and methods was measured by ELISA. Data shown are representative of two combined independent experiments. Values represent the mean ± SE per time point normalized by cell count (Student's t-test).
CONCLUDING REMARKS
Here, we are the first to show that p21 functions to limit the innate immune response to inflammatory stimuli in mice and in isolated macrophages. We demonstrate that p21−/− mice are more susceptible to death and express higher levels of IL-1β, TNFα, and IL-6 in serum following injection with LPS, that p21 deficient macrophages secrete elevated levels of these cytokines after TLR ligation, and that a peptidomimetic to the CDK domain of p21 reduces IL-1β secretion. These data suggest that p21 mimetic therapy may have therapeutic value in the treatment of inflammatory disease.
MATERIALS AND METHODS
Mice
B6129S2-Cdkn1atm1Tyj/J (p21−/−) and B6129SF2/J (congenic control, WT) mice were purchased from Jackson Laboratory (Bar Harbor, ME). p21−/− mice on a C57Bl/6 background were provided by Dr. Dimtrios Balomenos [8]. Six week old mice were injected intraperitoneally with LPS (10mg/kg of body weight) from E. coli 0111:B4 (Sigma; St. Louis, MO) and survival was monitored. An additional cohort of mice was euthanized at 24 hours following LPS injection and serum was collected. Anakinra (15mg/kg) or saline was injected intraperitoneally 20 minutes prior to LPS and every 6 hours for a total of 5 doses. All experiments on mice were approved by the Animal Care and Use Committee at Saint Louis University.
Cell Culture
Bone marrow cells were isolated from 6–8 week old mice by tibia flush and grown in complete DMEM with 20% L-cell supernatant containing macrophage-colony stimulating factor. Greater than 95% of the cells were F4/80+, CD45+, CD11b+, and CD16/32+. Peritoneal cells were harvested via lavage from 6–8 week old mice 3 days following intraperitoneal injection of 4% aged thioglycollate, adhered for 1 hour in serum-free media, then maintained in complete DMEM. Human monocytes were isolated from the buffy coat (Lifesource; Chicago, IL) of healthy donors by countercurrent centrifugal elutriation (JE-6B; Beckman Coulter; Fullerton, CA), and then differentiated in vitro for 7 days in complete RPMI. These studies were approved by the Office for the Protection of Research Subjects at Northwestern University. LPS (Sigma) was used at 10ng/mL for TLR stimulation experiments. For secretion assays, macrophages were further treated with ATP (5mM) for up to 30 minutes to induce IL-1β release.
Real Time PCR
Total RNA was isolated from cell pellets using the RNeasy Mini Kit (Qiagen; Valencia, CA), reverse transcribed into cDNA (Promega; Madison, WI), and assayed in duplicate using SYBR Green as the reporter (Applied Biosystems; Foster City, CA). Primers for IL-1β and GAPDH were previously described [18]. Data were normalized to GAPDH and analyzed using the ΔΔCT method.
Immunoblot analysis
Whole-cell extracts were analyzed by immunoblot analysis using rat anti-mouse IL-1β (R&D Systems; Minneapolis, MN), mouse anti-human p21 (BD Pharmingen, San Jose, CA), or mouse anti-rabbit GAPDH (US Biological, Swampscott, MA) antibodies as previously described [19].
ELISA
For detection of IL-1β from cell supernatants, sandwich ELISAs were performed according to the manufacturer's instructions (R & D Systems, sensitivity: 15.6pg/mL). ELISAs were quantitated by absorbance at 450nm on a microplate reader (BioRad; Hercules, CA) and normalized by number of cells per well. The levels of IL-1β in serum were determined using a Luminex based assay according to manufacturer's specifications (Linco Research; Earth City, MO).
Active IL-1β Bioassay
Biological activity of IL-1β was measured by addition of supernatants or standard curve control to RINm5F cells and analysis of nitrate levels using Greiss reagent as previously described [20].
siRNA
Human macrophages were transfected with 200nM of CDKN1A (p21) SMARTpool or a non-specific siRNA (Dharmacon Inc./Thermo Fisher, Lafayette, CO) using Lipofectamine (Invitrogen) according to manufacturer specifications. After 4 hours, complete media was added and cells were cultured for 48 hours. Cells were then stimulated with LPS/ATP and supernatants were analyzed for IL-1β secretion by ELISA.
Peptides
A polycationic peptide derived from HIV-1 TAT [14] was fused to the p21-mimetic peptides, which were synthesized by and purchased from the Peptide Synthesis group at Tufts University. The CDK domain on p21 is aa 46–65: (Ac-RKKRR-Orn-RRR-RERWNFDFVTETPLEGDFAW-OH). Previous studies have shown that 10μM is the optimal dose to inhibit cell cycle for p21-mimetic peptides [21].
Supplementary Material
ACKNOWLEDGEMENTS
This work is supported by predoctoral American Heart Association grants to JCS (0515499Z) and JH (0710060Z), ACR REF/Abbott Medical Student Research Preceptorship to MM, and grants from the National Institutes of Health to JC (AI44458), TE (GM070925, HL056236), RP (AR048269, AR049217), and HP (AR050250, AR054796, AI067590).
Abbreviations used
- BMDM
bone marrow derived macrophages
- CDK
cyclin dependent kinase
- PCNA
proliferating cell nuclear antigen
- IL-1Ra
interleukin-1 receptor antagonist
Footnotes
CONFLICT OF INTEREST The authors report no conflicts of interest.
REFERENCES
- 1.Dinarello CA. Interleukin-1beta. Crit Care Med. 2005;33:S460–462. doi: 10.1097/01.ccm.0000185500.11080.91. [DOI] [PubMed] [Google Scholar]
- 2.Furst DE, Breedveld FC, Kalden JR, Smolen JS, Burmester GR, Bijlsma JWJ, Dougados M, et al. Updated consensus statement on biological agents, specifically tumour necrosis factor {alpha} (TNF{alpha}) blocking agents and interleukin-1 receptor antagonist (IL-1ra), for the treatment of rheumatic diseases, 2005. Ann Rheum Dis. 2005;64:iv2–14. doi: 10.1136/ard.2005.044941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Callen JP. Complications and adverse reactions in the use of newer biologic agents. Semin Cutan Med Surg. 2007;26:6–14. doi: 10.1016/j.sder.2006.12.002. [DOI] [PubMed] [Google Scholar]
- 4.Sherr CJ, Roberts JM. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 1999;13:1501–1512. doi: 10.1101/gad.13.12.1501. [DOI] [PubMed] [Google Scholar]
- 5.Dotto GP. p21(WAF1/Cip1): more than a break to the cell cycle? Biochim Biophys Acta. 2000;1471:M43–56. doi: 10.1016/s0304-419x(00)00019-6. [DOI] [PubMed] [Google Scholar]
- 6.Balomenos D, Martin-Caballero J, Garcia MI, Prieto I, Flores JM, Serrano M, Martinez AC. The cell cycle inhibitor p21 controls T-cell proliferation and sex-linked lupus development. Nat Med. 2000;6:171–176. doi: 10.1038/72272. [DOI] [PubMed] [Google Scholar]
- 7.Santiago-Raber ML, Lawson BR, Dummer W, Barnhouse M, Koundouris S, Wilson CB, Kono DH, Theofilopoulos AN. Role of cyclin kinase inhibitor p21 in systemic autoimmunity. J Immunol. 2001;167:4067–4074. doi: 10.4049/jimmunol.167.7.4067. [DOI] [PubMed] [Google Scholar]
- 8.Arias CF, Ballesteros-Tato A, Garcia MI, Martin-Caballero J, Flores JM, Martinez AC, Balomenos D. p21CIP1/WAF1 Controls Proliferation of Activated/Memory T Cells and Affects Homeostasis and Memory T Cell Responses. J Immunol. 2007;178:2296–2306. doi: 10.4049/jimmunol.178.4.2296. [DOI] [PubMed] [Google Scholar]
- 9.Perlman H, Bradley K, Liu H, Cole S, Shamiyeh E, Smith RC, Walsh K, et al. IL-6 and matrix metalloproteinase-1 are regulated by the cyclin-dependent kinase inhibitor p21 in synovial fibroblasts. J Immunol. 2003;170:838–845. doi: 10.4049/jimmunol.170.2.838. [DOI] [PubMed] [Google Scholar]
- 10.Sutterwala FS, Ogura Y, Flavell RA. The inflammasome in pathogen recognition and inflammation. J Leukoc Biol. 2007 doi: 10.1189/jlb.1206755. [DOI] [PubMed] [Google Scholar]
- 11.Beutler B, Milsark IW, Cerami AC. Passive Immunization against Cachectin/Tumor Necrosis Factor Protects Mice from Lethal Effect of Endotoxin. Science. 1985;229:869–871. doi: 10.1126/science.3895437. [DOI] [PubMed] [Google Scholar]
- 12.Pelegrin P, Surprenant A. Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. Embo J. 2006;25:5071–5082. doi: 10.1038/sj.emboj.7601378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–241. doi: 10.1038/nature04516. [DOI] [PubMed] [Google Scholar]
- 14.Gammon ST, Villalobos VM, Prior JL, Sharma V, Piwnica-Worms D. Quantitative analysis of permeation peptide complexes labeled with Technetium-99m: chiral and sequence-specific effects on net cell uptake. Bioconjug Chem. 2003;14:368–376. doi: 10.1021/bc0256291. [DOI] [PubMed] [Google Scholar]
- 15.Sekine C, Sugihara T, Miyake S, Hirai H, Yoshida M, Miyasaka N, Kohsaka H. Successful treatment of animal models of rheumatoid arthritis with small-molecule cyclin-dependent kinase inhibitors. J Immunol. 2008;180:1954–1961. doi: 10.4049/jimmunol.180.3.1954. [DOI] [PubMed] [Google Scholar]
- 16.Joosten LA, Helsen MM, van de Loo FA, van den Berg WB. Anticytokine treatment of established type II collagen-induced arthritis in DBA/1 mice. A comparative study using anti-TNF alpha, anti- IL-1 alpha/beta, and IL-1Ra. Arthritis Rheum. 1996;39:797–809. doi: 10.1002/art.1780390513. [DOI] [PubMed] [Google Scholar]
- 17.Nasu K, Kohsaka H, Nonomura Y, Terada Y, Ito H, Hirokawa K, Miyasaka N. Adenoviral transfer of cyclin-dependent kinase inhibitor genes suppresses collagen-induced arthritis in mice. J Immunol. 2000;165:7246–7252. doi: 10.4049/jimmunol.165.12.7246. [DOI] [PubMed] [Google Scholar]
- 18.Savard CE, Blinman TA, Choi HS, Lee SK, Pandol SJ, Lee SP. Expression of cytokine and chemokine mRNA and secretion of tumor necrosis factor-alpha by gallbladder epithelial cells: response to bacterial lipopolysaccharides. BMC Gastroenterol. 2002;2:23. doi: 10.1186/1471-230X-2-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Albee L, Perlman H. E. coli infection induces caspase dependent degradation of NF-kappaB and reduces the inflammatory response in macrophages. Inflamm Res. 2006;55:2–9. doi: 10.1007/s00011-005-0001-9. [DOI] [PubMed] [Google Scholar]
- 20.Hill JR, Corbett JA, Baldwin AC, McDaniel ML. Nitric oxide production by the rat insulinoma cell line, RINm5F, Is specific for IL-1: a spectrophotometric IL-1 bioassay. Anal Biochem. 1996;236:14–19. doi: 10.1006/abio.1996.0125. [DOI] [PubMed] [Google Scholar]
- 21.Goulvestre C, Chereau C, Nicco C, Mouthon L, Weill B, Batteux F. A mimic of p21WAF1/CIP1 ameliorates murine lupus. J Immunol. 2005;175:6959–6967. doi: 10.4049/jimmunol.175.10.6959. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.