Abstract
IgG complexes bind to Fc receptor family members FcγRI (CD64), FcγRII (CD32) and FcγRIII (CD16), activating cell MAPK and PI3K resulting in increased cytokine production from particular leukocytes. The signaling molecules involved in cytokine production after cross-linking CD16 have not been determined in monocytes. To address this question, TNF-α, IL-1β and IL-6 were measured in activated monocytes after inhibiting mitogen-activated protein kinase kinase 1/2 (MEK1/2), PI3K and glycogen synthase kinase-β (GSK-3β). The roles of GSK-3β and NF-κB were then determined using reporter assays and siRNA treatment. The data suggested that a MAPK pathway stimulated TNF-α release but that active PI3K limited TNF-α, IL-1β and IL-6 cytokine production after cross-linking CD16. PI3K was also shown to limit nuclear translocation of NF-κB. The limiting effect of PI3K on TNF-α production from activated monocytes depended on the decrease of GSK-3β activity, which significantly reduced the transactivation of NF-κB. Moreover, the TNF-α production induced by CD16 cross-linking was reduced in monocytes after treatment with siRNA against NF-κB, implying that this transcription factor functioned in TNF-α production. The results suggest that CD16 cross-linking activated PI3K and that active PI3K limited TNF-α production by inhibiting GSK-3β activity, in part, by blocking the action of NF-κB.
Keywords: Fc receptor, FcγRIII, IgG, monocytes
Introduction
CD16, also termed “FcγRIII”, is a member of the Fc receptor family [1;2]. CD16 is expressed on multiple hematopoietic cell types, and binding is preferential for small IgG dimer or trimer complexes [3] that can include IgG anti-IgG antibody complexes [4]. These complexes are important components of auto-antigens and rheumatoid factors that potentially trigger the onset or maintenance of autoimmune diseases such as rheumatoid arthritis [5;6;7;8]. Furthermore, the expression of CD16 on monocytes/macrophages is restricted to tissues, such as synovial tissue and the pericardium, that are impacted by rheumatoid arthritis [9].
Structural components of the CD16 receptor include an α subunit that is primarily extracellular and functions in binding antigen. Additional associated components include a cytoplasmic signaling protein that is a homo- or heterodimer made up of a ζ or γ subunit [10]. These subunits have been shown to be necessary for receptor assembly and signal transduction of the complete receptor in human cells [11]. The ζ subunit has not been detected in monocytes, and thus, the active CD16 receptor in monocytes likely consists of an α subunit associated with a homodimer of the γ subunit [10].
TNF-α and IL-1β production can be induced by an antibody binding and cross-linking the CD16 receptor expressed on the surface of the monocytes; this production requires de novo transcript synthesis and not simply the release of stored TNF-α [3]. In contrast to antibodies that cross-link the CD16 receptor, the primary antibodies to CD32 (FcγRII) and CD64 (FcγRI) alone do not stimulate TNF-α production from monocytes [3]. A secondary antibody is required to stimulate TNF-α production, suggesting that these receptors need to be associated in larger clusters than are characteristic of CD16 to activate the signaling pathways [12]. Our previous studies have contributed to this body of knowledge by demonstrating that IL-6 production can also be stimulated by CD16 cross-linking [13].
Fc receptors utilize MAPK and PI3K pathways to activate leukocytes. It was found that in primary mouse macrophages, MAPK was necessary to signal increased TNF-α production after CD32 and CD16 cross-linking [14], and in monocytic cell lines, the cross-linking of CD16, CD32 or CD64 activated MAPK pathways [15;16]. MAPK and PI3K pathways were activated in natural killer cells after stimulation of CD16 [15;17;18] and in monocytic U937 cells PI3K signaled cellular activation after CD32 and CD64 cross-linking [19]. Upon the addition of IgG complexes, IL-6 production was shown to be partially dependent on PI3K in primary bone marrow-derived macrophages [20] but the function of PI3K in monocyte cytokine production has not been determined after specifically cross-linking the CD16 receptor.
In this study, we examined the role of PI3K in modulating cytokine production from primary human monocytes after cross-linking the CD16 receptor. Moreover, the role that glycogen synthase kinase-β (GSK-3β) and NF-κB have modulating TNF-α production from activated monocytes was explored.
Results
TNF-α, IL-1β and IL-6 production can be induced by an antibody binding and cross-linking the CD16 receptor expressed on the surface of the monocytes [3;13]. The signaling molecules involved in cytokine production after cross-linking CD16 have not been determined in monocytes. To address this question, TNF-α, IL-1β and IL-6 were measured in activated monocytes after treatment with various kinase inhibitors. The roles of GSK-3β and NF-κB in signaling cytokine production after CD16 activation were then determined using reporter assays and siRNA treatment.
MAPK pathways are stimulated by CD16 activation
The transcript levels for TNF-α were significantly (P<0.05) increased 3 fold after treatment with anti-CD16 versus treatment with an IgG isotype control. Our results also showed that anti-CD16 antibodies stimulated increased TNF-α protein production from monocytes (Fig. 1), which was consistent with previous results [3;13]. In peripheral blood monocytes, the use of PD98059 to block mitogen-activated protein kinase kinase (MEK) (Fig. 1A) significantly decreased the TNF-α production. The inhibition of TNF-α production after treatment with 20 µM PD98059 was likely due to the inhibition of MEK1 because the inhibition of MEK2 requires a higher concentration of this antagonist [21]. PD98059 can also inhibit prostaglandins and leukotrienes to effect the immune function [22]; thus, we also treated the monocytes with the MEK inhibitor U0126. Treatment with U0126 inhibited TNF-α production from activated monocytes (Fig. 1B). Cell counts before and after treatment indicated that the cell number and viability did not significantly decrease with treatment (data not shown) consistent with the idea that CD16 stimulation activates MEK and signals cytokine production from the monocytes.
Figure 1. A MAPK pathway regulated TNF-α release from monocytes after CD16 stimulation.
Primary human monocytes were pretreated with kinase inhibitors PD98059 (A) and U0126 (B) at two concentrations for 1 hour prior to the addition of anti-CD16 antibody (clone 3G8) or an IgG isotype control. Supernatants were collected 48 hours later and the concentration of TNF-α was measured by ELISA. Total protein from the cells was then measured. Y-axis shows the concentration of TNF-α expressed as pg of cytokine per mg of total cellular protein. *p<0.05, as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
PI3K pathway limits cytokine production after CD16 activation
Surprisingly, after activating the CD16 receptor with the anti-CD16 antibodies and treating with the PI3K inhibitor LY294002, the amounts of TNF-α (Fig. 2A), IL-1β (Fig. 2B) and IL-6 (Fig. 2C) significantly increased. The transcript levels for TNF-α were shown to increase 6 fold after treatment with anti-CD16 and LY294002 versus the IgG isotype control and LY294002 treatment. Cell counts did not significantly increase during the 48 hour treatment with LY294002. At this point in the study, we focused on dissecting the mechanism by which PI3K limits cytokine production. In dissecting the mechanism it was advantageous to focus on one cytokine. We chose to focus on the TNF-α expression because previous studies in other laboratories have demonstrated a similar phenomenon after LPS stimulation, showing that the TNF-α expression significantly increases after the inhibition of PI3K [23]. One potential problem with using LY294002 is that this chemical inhibits both PI3K and casein kinase 2 (CK2) [24]. Thus, to show specificity only for PI3K, we treated the monocytes with a second PI3K inhibitor, wortmannin and consistent with the previous results, the TNF-α cytokine levels significantly increased in a dose dependent manner (Fig. 3).
Figure 2. PI3K limits cytokine release from monocytes after CD16 stimulation.
Cultured human primary monocytes were pretreated with the PI3K inhibitor LY294002 one hour prior to the addition of anti-CD16 antibody (clone 3G8) or an IgG isotype control antibody. Supernatants were collected 48 hours after the addition of the antibodies. Secreted TNF-α (A), IL-1β (B) and IL-6 (C) were measured by ELISA. Total protein from the cells was then quantitated. Y-axis shows the concentration of cytokines as pg of cytokine per mg of total cellular protein. *p<0.05, as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
Figure 3. PI3K inhibitor wortmannin increased TNF- α from activated monocytes.
Cultured human primary monocytes were pretreated with the PI3K inhibitor wortmannin one hour prior to the addition of anti-CD16 antibody (clone 3G8) or an IgG isotype control. Supernatants were collected 48 hours after the addition of the antibodies. Secreted TNF-α was measured by ELISA and the concentration of TNF-α was expressed as pg of cytokine per mg of total tissue protein. *p<0.05, as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
Inhibition by PI3K is not primarily mediated through MAPK pathways
PI3K modulates TNF-α production through a mechanism exclusive of the ERK1/2 MAPK pathway. Using the PI3K inhibitor LY294002, a continued increase in TNF-α was observed in the presence of inhibitor PD98059 after cross-linking CD16 (Fig. 4A). Moreover, the addition of MAPK inhibitors PD98059 had no significant effect on the increase in TNF-α (Fig. 4A) compared to the treatment with LY294002 alone (Fig. 2A). To substantiate this result, we treated the primary monocytes with anti-CD16 and 1 µM of MAPK inhibitor U0126, as well as, 10 µM of the PI3K inhibitor wortmannin (Fig. 4B). The results showed that the TNF-α expression in the activated monocytes significantly increased (P<0.05) after pretreatment with wortmannin and U0126. The combined results suggested that PI3K did not limit the cytokine expression through a MAPK pathway.
Figure 4. PI3K and MAPK pathway interactions.
Cultured human primary monocytes were pretreated with the MEK1/2 inhibitor PD98059 (A) or U0126 (B) and the PI3K inhibitor LY294002 (A) or wortmannin (B) one hour prior to the addition of anti-CD16 antibody (3G8) or an IgG isotype control antibody. Supernatants were collected 48 hours after the addition of the antibodies and secreted TNF-α was measured by ELISA. TNF-α concentration is expressed as pg of cytokine per mg of total tissue protein. *p<0.05, as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
Inhibition by PI3K involves GSK-3β and NF-κB
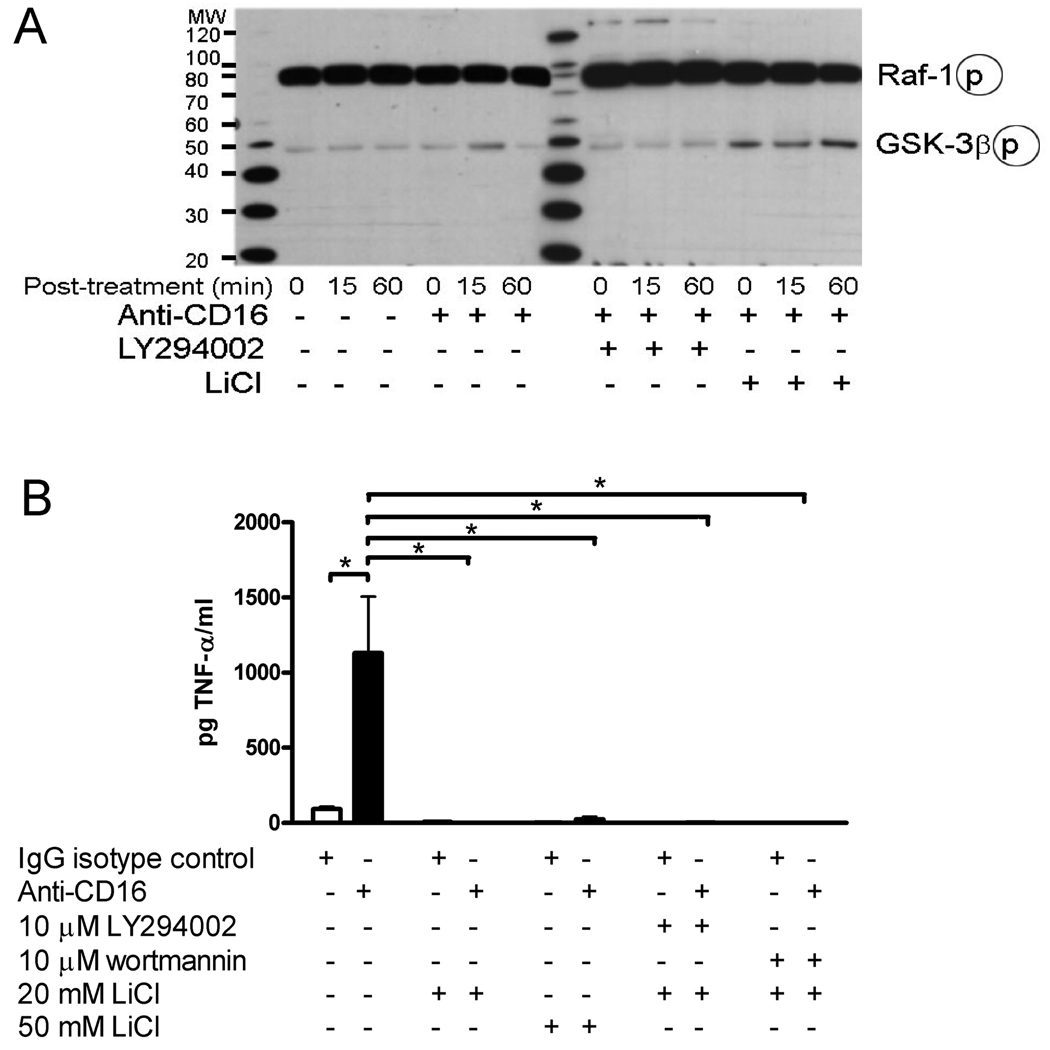
An additional mechanism by which PI3K can limit the TNF-α production is through GSK-3β modulating the NF-κB activity [25]. PI3K can inactivate GSK-3β, an enzyme shown to stimulate nuclear localization and transactivation of NF-κB [23;26]. Western blot analysis indicated that the treatment of monocytes with anti-CD16 for 15 minutes increased the level of inactive phospho-GSK-3β and that the treatment with the PI3K inhibitor LY294002 blocked this inactivation (Fig. 5A). The treatment with LiCl resulted in a rapid rise in the level of the phosphorylated (i.e., inactive) GSK-3β (Fig. 5A). The inhibition of GSK-3β with 20 and 50 mM concentrations of LiCl completely blocked TNF-α expression after treatment with anti-CD16 (Fig. 5B). Moreover, LiCl blocked the enhancement of the TNF-α expression resulting from the treatment with the PI3K inhibitor LY2984002 and wortmannin (Fig. 5B)
Figure 5. PI3K and GSK-3β function to inhibit TNF-α release.
(A) Cultured human primary monocytes were pretreated with PI3K kinase inhibitor LY294002 or GSK-3β kinase inhibitor LiCl one hour prior to the addition of anti-CD16 antibody (clone 3G8) or vehicle. Monocytes were collected 0, 15 or 60 minutes after the addition of the CD16 antibody or vehicle, and 10 µg of total protein was loaded in each lane for western blot analysis. The western blot was probed with an antibody against Ser388 phosphorylated c-Raf antibody (73 kDa) or a Ser9 phosphorylated GSK-3β (46KDa). (B) Cultured human primary monocytes were pretreated with kinase inhibitors one hour prior to the addition of anti-CD16 antibody (clone 3G8) or an IgG isotype control. Supernatants were collected 48 hours after the addition of the antibodies. TNF-α concentration was determined by ELISA and expressed as pg of cytokine per mg of total tissue protein. *p<0.05 as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
PI3K activation has been shown to inhibit Raf-1 activity and the downstream enzymes MEK1/2 and ERK1/2 [27;28]. PI3K has also been shown to limit cytokine production after LPS stimulation, in part, by inhibiting Raf-1 in the Raf/MEK/ERK pathway [23]. In our experiment phosphorylated Raf-1 levels were unchanged after treatment with LY294002 (Fig. 5A) consistent with the data suggesting PI3K does not limit the cytokine expression through a Raf/MEK/ERK pathway.
GSK-3β can modulate the localization of NF-κB to the nucleus and effect expression of genes dependent on NF-κB activation [26]. Monocytes treated with LY294002 after cross-linking the CD16 receptor showed a significant increase in the level of p65 localized in the nucleus (Fig. 6a). No significant change in the cytoplasmic, NFκB pool was detected (data not shown). To test our nuclear localization experiment we used a positive control; which was treatment with LPS. We observed that LPS stimulated a significant increase in the nuclear fraction of NF-κB (LPS = 0.302 ± 0.137 relative expression; no treatment = 0.130 ± 0.019 relative expression). Interestingly, the amount of p65 with a phosphorylated Ser536 did not increase in the nucleus or cytoplasm after the treatment of active monocytes with LY294002 (Fig. 6b and data not shown).
Figure 6. PI3K effects nuclear localization of p65 and phosphorylated p65.
Human primary monocytes were pretreated with vehicle or PI3K kinase inhibitor LY294002 one hour prior to the addition of anti-CD16 antibody (3G8). Monocytes were collected 0, 15 or 60 minutes after the addition of the CD16 antibody. Nuclear extracts were prepared, and the amounts of p65 (A) and p65 phosphorylated at Ser536 (B) were determined by ELISA. *p<0.05 as determined by two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
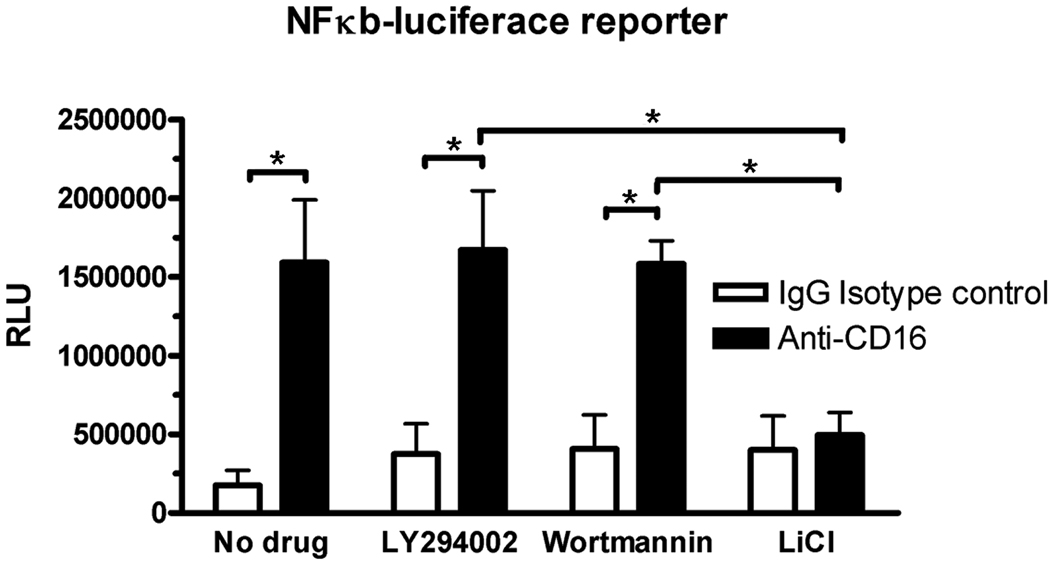
Using a luciferase reporter construct with a minimal promoter containing five tandem NF-κB binding sites, we demonstrated that transactivation by NF-κB significantly increased after cross-linking CD16 (Fig. 7). LY294002 did not significantly enhance transactivation (Fig. 7). Importantly, the inactivation of GSK-3β by treatment with LiCl completely blocked the transactivation that occurs after cross-linking CD16 (Fig. 7). Treatment with LiCl did not affect the number of live cells as assayed with trypan blue.
Figure 7. NF-κβ transactivation was regulated by GSK-3β.
Human monocytes were transfected with a luciferase construct containing five 5’ NF-κβ sites (GGGGACTTTCC). A SV40 Renilla luciferase construct was co-transfected to control for variability in transfection efficiencies. 24 h after transfection, monocytes were treated with the PI3K kinase inhibitor LY294002 or wortmannin or the GSK-3β kinase inhibitor LiCl. One hour after the addition of the kinase inhibitors, monocytes were treated with an anti-CD16 antibody (3G8) or an IgG isotype control antibody. Luciferase activity was measured 5 hours after the addition of the antibody and reported as relative light units (RLU), which is the luminescent signal from the firefly luciferase normalized to an internal control Renilla luciferase construct. *p<0.05, as determined by a two-way ANOVA and a Duncan’s post-hoc test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
To directly test the function of NF-κB on TNF-α production, NF-κB expression was reduced using siRNA with homology to the NF-κB gene. Transfection with NF-κB siRNA significantly decreased the amount of total NF-κB in monocytes after 24 hours (Fig. 8A) and significantly decreased transactivation by NF-κB (Fig. 8B). Also, treatment with NF-κB siRNA significantly reduced TNF-α production from monocytes stimulated with anti-CD16 compared to cells transfected with control siRNA (Fig. 8C). Thus, a decrease in NF-κB expression and transactivation was associated with decreased TNF-α expression in the monocytes activated by treatment with anti-CD16.
Figure 8. Reducing NF-κB expression decreased NF-κβ transactivation and CD16-induced TNF-α production in activated monocytes.
Human primary monocytes were transfected with siRNA bearing homology to the NF-κB gene or a control siRNA construct. (A) Monocytes were isolated 48 hours following transfection, and the total amount of cellular NF-κB was measured by ELISA. (B) 24 h after transfection, monocytes were again transfected with a luciferase construct containing five 5’ NF-κB sites and a SV40 Renilla luciferase construct. 24 h following the second transfection the monocytes were treated with an anti-CD16 antibody (clone 3G8) or an IgG isotype control antibody. Luciferase activity was measured 5 hours after the addition of the antibody and reported as relative light units (RLU), which is the luminescent signal from the firefly luciferase normalized to an internal control Renilla luciferase construct. (C) Monocytes were treated with an anti-CD16 antibody (clone 3G8) or an IgG isotype control antibody 48 hours after transfection. Supernatants were collected 48 hours after the addition of the antibodies and TNF-α concentration was measured by ELISA and expressed as pg of cytokine per mg of total tissue protein. *p<0.05, as determined by the Student’s t-test. Each experiment was repeated three times with monocytes from different donors. Graphs show the mean + SEM.
Discussion
MAPK pathways and PI3K pathways function to signal cytokine production in leukocytes [15;17;18] but the function of PI3K in monocyte cytokine production after CD16 stimulation has not been determined. The blockade of MEK1/2 reduced TNF-α expression in human peripheral blood monocytes. In contrast to the stimulatory action of the MAPK pathway, PI3K limited the expression of TNF-α, IL-1β and IL-6 after cross-linking with CD16. This limiting effect of PI3K did not depend primarily on MAPK pathways but was contingent upon the activity of GSK-3β. The blockade of PI3K increased the NF-κB nuclear localization and the transactivation by NF-κB required GSK-3β activity. The siRNA inhibition of NF-κB reduced the TNF-α production, suggesting that the limiting effect of PI3K was due, in part, to the inhibition of NF-κB. In summary, our results are consistent with those showing that PI3K limits TNF-α production through increasing phosphorylation and inactivation of GSK-3β in the monocytes after CD16 cross-linking. The inactivation of GSK-3β inhibited the NF-κB activity, a transcription factor that functions in TNF-α expression.
Our results demonstrated that the inhibition of a MAPK pathway reduced TNF-α, IL-1β and IL-6 expression in the monocytes that were activated by cross-linking CD16. In leukocytes, MAPK pathways have been shown to signal cytokine production [14–16;29;30]. Citing just two examples one, the activation of Fc receptors CD32 and CD16 increased TNF-α production but the blockade of MAPK activity reduced the TNF-α production [14]. Two, other studies showed that the treatment of the monocytic cell lines J774 and THP-1 with antibodies or IgG complexes, which cross-link Fc receptors, activated the MAPK pathways and stimulated cytokine production [15;16].
After receptor binding, PI3K signaling can be inhibitory or stimulatory, depending on the type of receptor activated. IgG complexes bind and cross-link multiple types of Fc receptors (e.g., CD16, CD32, CD64) on macrophages, and this cross-linking event has been shown to activate PI3K, which was then necessary for signaling increased IL-6 expression [20]. In another study, just the opposite was observed: 1) the expression of the gene inositol 3-phosphatase and tensin homologue deleted on chromosome 10 (PTEN) produced a molecule that inhibited Fc receptor-induced production of TNF-α, IL-6, and IL-10 through activation of the PI3K pathway, and 2) the deletion of PTEN reduced the activity of the PI3K pathway and increased the TNF-α production in macrophages after the activation of Fc receptors [31]. Researchers in two other studies found that PI3K limited the cytokine expression. In the first study, the blockade of PI3K increased the IL-1 expression in the promyelocytic cell line HL-60 [32], and in the second studies, blockade of PI3K during the activation of Toll-like receptor 4 increased the TNF-α production in monocytes and macrophages [23;33]. Even though much work has concentrated on PI3K signaling and cytokine expression, no study has focused on PI3K signaling after the activation of CD16. The present results are consistent with reports that PI3K limits cytokine production but our work adds to the current body of knowledge that such signaling occurs after specific cross-linking of CD16.
An increased amount of inactive or phosphorylated GSK-3β was concurrent with the limiting action of PI3K after the addition of anti-CD16. LY294002 has been shown to cause the accumulation of endogenous GSK-3β in its active form [26]. The blockade of PI3K with LY294002 prevented the inactivation of GSK-3β after the addition of anti-CD16. Also, the level of inactive or phosphorylated GSK-3β increased using the GSK-3β inhibitor LiCl in activated monocytes. The addition of LiCl also prevented any increase in the TNF-α production resulting from the addition of anti-CD16. Moreover, LiCl blocked any effect of LY294002, suggesting that PI3K inhibits the TNF-α expression by inhibiting GSK-3β activity, a pathway similar to that observed after LPS stimulation of cytokine production in human monocytes [23].
PI3K limited NF-κB nuclear localization but the role that GSK-3β plays in this process is unclear. The amount of NF-κB localized in the nucleus increased in activated monocytes after treatment with LY294002, implying that PI3K inhibits nuclear localization, which is consistent with previous studies [25]. Previous reports show that PI3K inhibits GSK-3β through phosphorylation at Ser9 [34;35] and that inactivation of GSK-3β can either increase NF-κB activity [26;36] or decrease NF-κB activity [37]. We suggest from our work that inactivation of GSK-3β will decrease NF-κB activity by preventing nuclear localization of NF-κB. The expression of the luciferase reporter construct containing a promoter with five NF-κB binding sites increased upon treatment with anti-CD16, indicating transactivation by NF-κB upon cross-linking of this receptor. The treatment of these monocytes with LiCl significantly reduced the NF-κB transactivation of the reporter. Because LiCl inhibits GSK-3β [37–39], the data suggested that active GSK-3β increases NF-κB transactivation. GSK-3β has been shown to increase NF-κB-dependent transactivation through direct phosphorylation of the p65 subunit [40;41]. Even though changes in phosphorylation at Ser536 was not observed in this study, previous results demonstrated that transactivation can occur at other amino acids in NF-κB [41–44] to alter NF-κB activity and further studies will focus on this possibility.
Treatment with LY294002 did not significantly increase NF-κB transactivation in our reporter construct but LY294002 did significantly increase the TNF-α expression. Also, treatment with NF-κB siRNA inhibited largely inhibited NF-κB expression but only partially blocked TNF-α expression. A possible explanation for these two results is that the promoter in the reporter construct has a different transcription binding site(s) from the endogenous TNF-α promoter. In the event that several transcription factors are acted upon by PI3K to limit the cytokine expression, we may not see an increase in our NF-κB reporter after treating the monocytes with LY294002. Also, if full TNF-α expression requires multiple transcription factors complete suppression would not be induced by blocking only NF-κB. As one example, AP-1 has been associated with Fc receptor signaling in macrophages [16;45], and AP-1 can interact with NF-κB to signal efficient transactivation [46;47]. Thus, if PI3K reduces both AP-1 and NF-κB activity, and the endogenous TNF-α promoter requires both AP-1 and NF-κB, then inhibiting PI3K with LY294002 would not lead to a dramatic increase in a NF-κB-dependent reporter. Nor would inhibition of only NF-κB activity completely block expression from the TNF-α promoter. Alternatively, the reporter construct does not have the same chromatin structure found in the endogenous TNF-α promoter. PI3K has been found to limit TNF-α expression through a GSK-3β-dependent mechanism in which active GSK-3β modified the chromatin structure of the genes and altered the NF-κB transactivation of these genes [45]. Thus, our reporter would not give the same response to treatment with LY294002 as that observed in the endogenous TNF-α gene.
In summary, we demonstrated for the first time that PI3K limits the TNF-α expression from human primary monocytes after specifically cross-linking CD16. PI3K limits TNF-α expression through a GSK-3β-dependent pathway. GSK-3β modulates the activity of NF-κB, which is a known regulator of TNF-α expression. This relationship is consistent with a mechanism where cross-linking CD16 activates PI3K, inactivating GSK-3β and reducing NF-κB activity to limit TNF-α expression.
Materials and Methods
Primary monocyte isolation and culture
Primary monocyte isolation occurred in three stages. First, the buffy coats from healthy volunteers between the ages of 20 and 45 years were obtained from the Carter Blood Care Center following IRB-approved protocols. Second, the blood products were diluted in 0.15 M NaCl/2 mM EDTA; 31 ml of this mixture was layered on 18 ml of Ficoll-hypaque (Sigma) solution in a 50 ml polypropylene tube and separated by density gradient centrifugation. The banded peripheral blood mononuclear cell fraction (PBMC) (monocytes, T and B cells) was removed from the gradient, red blood cells and polymorphonuclear cells form a pellet that is discarded. Third, the PBMC fraction was suspended in RPMI media with 10% fetal bovine serum (FBS) and 10% DMSO and frozen in liquid nitrogen (2×107 cells/vial).
A frozen vial of human PBMCs was thawed quickly at 37°C, the cells pelleted and mixed with 80 µl of buffer (1 X PBS, 2mM EDTA and 0.5% bovine serum albumin) and 20 µl of anti-CD14 antibody-conjugated magnetic beads (MicroBeads, Miltenyi Biotech, Auburn, CA). After 15 minutes at 6–12˚C, the cells were gently washed with 2 mls of buffer and pelleted. The cell pellets were resuspended in 0.5 ml of buffer and the CD14 positive cells separated on a LS column following the manufacturer’s directions (Miltenyi Biotech). The positive cells were resuspended in media (RMPI, 5% charcoal-stripped FBS and 10 ng/ml transforming growth factor-β) [48]. Using FACS, 95% of the cells expressed CD14 after column purification. FACS was completed on a FACSCalibur flow cytometer using BD CellQuest Pro software. The expression of the CD16 receptor was observed in 10–20% of the purified monocytic population as determined by FACS analysis.
Monocyte activation and treatment
After column separation, approximately 2×105 CD14+ monocytic cells with 95% viability (based on trypan blue staining) in 0.5 ml of media were placed in the individual wells of a 24-well plate. To analyze the signaling pathways that regulate cytokine expression, several inhibitors or vehicles were added to a portion of the cultures. Mitogen-activated protein/extracellular signal-regulated kinase 1/2 (MEK1/2) inhibitors U0126 and PD98059 and PI3K inhibitors LY294002 and wortmannin [24] were all dissolved in DMSO (EMD Biosciences, San Diego, CA) and added to the culture media. Lithium chloride is an inhibitor of GSK-3 β [37–39] and was dissolved in RPMI. One hour following the addition of the vehicle or inhibitor, 25 µg/ml of anti-CD16 [49] human monoclonal antibody (clone 3G8, Ancell Immunology Research Products) or 25 µg/ml of an IgG1 isotype control antibody (Ancell Immunology Research Products) was added to the cultures. Antibody clone 3G8 specifically cross-links the CD16 receptor, inducing TNF-α, IL-1β and IL-6 cytokine release [3;13]. The monocytes were isolated after various time points, cells number and viability (based on trypan blue staining) was counted and the media collected. In an additional experiment, the monocytes were treated with 10 µg/ml lipopolysaccharide (LPS) 026:B6 (Sigma L-2654), incubated for 1 hour and harvested for analysis of NF-κB by ELISA.
Western blot
After treatment with kinase inhibitors, the monocytes were isolated and mixed with T-Per tissue protein extraction reagent (Pierce). The total protein in the T-Per reagent was quantitated using the BCA protein assay (Thermo Scientific, Rockford, IL). Ten µg of total protein was electrophoresed through a 8% tris-gylcine acrylamide gel and transferred onto a PVDF membrane in 25 mM Tris, 192 mM glycine, 0.1% SDS, pH 8.3 (150 mA for 5 hours). The membrane was blocked (1 hr in TBST buffer-100 mM Tris-HCl, 150 mM NaCl, 0.1% Tween-20, pH 7.4) plus 5% BSA (w/v), and then a polyclonal antibody (diluted in the same blocking buffer plus 5% milk) was added. Antibodies against phosphorylated c-Raf (Ser338) and phosphorylated GSK-3 β (Ser9) (Cell Signaling Technologies, Boston, MA) were used in this study at a ratio of 1:1000. The membranes were incubated overnight in the primary antibody at 4°C. The next day the membranes were washed in TBST buffer, incubated for 1 hour in goat anti-rabbit secondary antibody (1:1000) conjugated to horseradish peroxidase (BioRad, Hercules, CA), washed in TBST buffer, reacted with ECL plus reagent (Pierce) and exposed to film.
Nuclear Isolation
After treatment, the isolated monocytes were rinsed twice in 1 X PBS at 4˚C and resuspended in buffer A (10mM HEPES pH 7.9, 1.5 mM MgCl2, 10 mM KCl 300 mM Sucrose, 0.5 mM DTT) containing a protease inhibitor cocktail (Complete mini, Roche Scientific). After a 15-minute incubation on ice, the cell membrane was disrupted by dousing the cells 10 times. The cell nuclei were isolated by centrifugation at 500 g, rinsed once in buffer A and centrifuged at 500 g. The nuclear fraction was resuspended in the cell lysis buffer provided in the PathScan ELISA kit (Cell Signaling Technology, Boston, MA).
ELISA analysis of cytokines and NF-κB
The quantitative measurements of TNF-α, IL-1β and IL-6 (Invitrogen, Carlsbad, CA) in the media and of NF-κB subunit p65 or phosphorylated p65 (Ser536) (Cell Signaling Technologies) in the isolated cells or cell nuclei were performed using commercially available sandwich ELISA kits following the manufacturer’s directions. The exact amounts of cytokines within the samples were determined by comparing them to the appropriate standards generated from recombinant TNF-α, IL-1β or IL-6 protein. The total protein in the tissue sample was determined by a BCA assay (Pierce, IL), and the values of each protein determined by ELISA was expressed pg cytokine per mg of total tissue protein. The amount of NF-κB was expressed as the OD values obtained from 2×105 monocytes.
Real time RT-PCR analysis of gene transcript
To analyze the transcript levels, total RNA was isolated from the treated cells using the RNeasy RNA isolation kit (Qiagen, Valencia, CA). The total RNA was quantitated and the quality determined using an Agilent 2100 Bioanalyzer (Agilent Technologies, Palo Alto, CA) following the manufacturer’s directions. Reverse transcription was completed using the RETROscript First Strand Synthesis Kit (Ambion, Austin, TX) according to the manufacturer’s directions by isolating the total RNA from the cells as a template. The quantitation of the gene expression was completed using real-time PCR by incorporating Syber green I dye. The real-time PCR reaction mixture contained 1X Brilliant SYBR Green QPCR Master Mix (Stratagene, La Jolla, CA), 0.3 µM of primers for TNF-α (5’-tcaacctcctctctgccatcaaga-3’, 5’-atgatcccaaagtagacctgccca-3’) or primers for 18S (5′- ccgaagcgtttactttga-3′, 5′-gccgtccctcttaatcat-3′), and 2 µl of the reverse transcriptase reaction. The PCR reactions were completed on a MX4000 multiplex quantitative PCR system (Stratagene) and consisted of a 10 min denaturation step at 95°C, followed by 40 cycles of 95°C for 30 seconds, 56°C for 1 minute and 72°C for 1.5 minutes and finally, a 10-minute 72°C extension. The data were gathered with MX4000 software. Control RNA samples containing a mock reverse transcription reaction (no reverse transcriptase added) were used to verify the absence of contamination in the solutions. Relative expression was calculated according to the delta delta Ct method.
siRNA transfection
After column purification, the human monocytes were cultured in RPMI and 10% FBS. NF-κB p65 siRNA (Cell Signaling Technologies, Boston, MA) or control non-targeting siRNA #1 (Dharmacon, Chicago, IL) was transfected with TransIT-siQuest transfection reagent according to the manufacturer’s directions. In brief, 25 nM of siRNA and 3 µl of siQuest transfection reagent was mixed and added to a 50 µl volume of Opti-MEM containing approximately 1х105 monocytes. Forty-eight hours after siRNA transfection, the anti-CD16 antibody or IgG isotype control antibody was added, and the monocytes were isolated for ELISA. Or the monocytes were transfected a second time twenty-four hours later with the reporter construct pNifty-Luc followed by addition of anti-CD16 antibody or IgG isotype control antibody.
Reporter construct transfection
Transfections with the reporter construct were completed by first mixing 3 × 106 - 1 × 107 of column-purified or siRNA transfected monocytes in 100 µl of Human Monocyte Nucleofector Medium supplemented with 2 mM glutamine and 10% FCS, according to the manufacturer’s directions (Amaxa, Gaithersburg, MD). One µg of pNifty-Luc (InvivoGen, San Diego, CA) and 0.5 µg phRL-SV40 (Renilla luciferase plasmid with the SV40 promoter; Promega, Madison ,WI) were then added to the solution and mixed. The cells mixed with DNA were transferred to an Amaxa cuvette and electroporated on program Y-001. They were immediately removed from the cuvette and placed in 1 ml of RPMI plus 10% FBS for 24 hours. One hour following the addition of a vehicle or inhibitor, 25 µg/ml of the anti-CD16 antibody or 25 µg/ml of an IgG1 isotype control antibody was added to the cultures. After a 5-hour incubation at 37°C, the cells were harvested and the luciferase activity measured using the Dual-Luciferase® Reporter Assay System (Promega). Luminescent photon measurements were made on a liquid scintillation analyzer (Tricarb 2900TR, Packard BioScience Company, Meriden, CT) with the coincidence function disabled. Relative firefly luciferase activity was normalized to co-transfected Renilla luciferase activity to obtain luciferase activity plotted in arbitrary units termed “relative light units” (RLU).
Statistics
The statistical analysis was completed using two-way ANOVA with the factors drug or drug concentration (kinase inhibitor) and treatment (anti-CD16 and IgG isotype control antibody). The Duncan’s post-hoc test was utilized to determine the significant changes between treatment groups. For the siRNA treatment groups and for the real time RT-PCR experiment, an unpaired t-test was used. Each treatment group was repeated in three separate experiments with monocytes isolated from three different donors. All values are given as the mean ± SEM. A significant difference was stated when P<0.05.
Acknowledgements
This work was supported by a grant DE015372-01 from the NIH institutes National Institute of Dental and Craniofacial Research (NIDCR) and Office of Research on Women’s Health (ORWH).
Abbreviations
- MEK
mitogen-activated protein kinase kinase
- GSK-3β
glycogen synthase kinase-β
- CK2
casein kinase 2
- FBS
fetal bovine serum
- PTEN
inositol 3-phosphatase and tensin homologue deleted on chromosome 10
- RLU
relative light unit
Footnotes
Conflict of interest: The authors declare no financial or commercial conflict of interest.
References
- 1.Nimmerjahn F, Ravetch JV. Fc-receptors as regulators of immunity. Adv.Immunol. 2007;96:179–204. doi: 10.1016/S0065-2776(07)96005-8. 179–204. [DOI] [PubMed] [Google Scholar]
- 2.Takai T. Fc receptors and their role in immune regulation and autoimmunity. J Clin.Immunol. 2005;25:1–18. doi: 10.1007/s10875-005-0353-8. [DOI] [PubMed] [Google Scholar]
- 3.Abrahams VM, Cambridge G, Lydyard PM, Edwards JC. Induction of tumor necrosis factor alpha production by adhered human monocytes: a key role for Fcgamma receptor type IIIa in rheumatoid arthritis. Arthritis Rheum. 2000;43:608–616. doi: 10.1002/1529-0131(200003)43:3<608::AID-ANR18>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 4.Klaassen RJ, Goldschmeding R, Tetteroo PA, Von dem Borne AE. The Fc valency of an immune complex is the decisive factor for binding to low-affinity Fc gamma receptors. Eur.J Immunol. 1988;18:1373–1377. doi: 10.1002/eji.1830180911. [DOI] [PubMed] [Google Scholar]
- 5.Pope RM, Teller DC, Mannik M. The molecular basis of self-association of antibodies to IgG (rheumatoid factors) in rheumatoid arthritis. Proc.Natl.Acad.Sci.U.S.A. 1974;71:517–521. doi: 10.1073/pnas.71.2.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nardella FA, Teller DC, Mannik M. Studies on the antigenic determinants in the self-association of IgG rheumatoid factor. J.Exp.Med. 1981;154:112–125. doi: 10.1084/jem.154.1.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Klaassen RJ, Goldschmeding R, Tetteroo PA, Von dem Borne AE. The Fc valency of an immune complex is the decisive factor for binding to low-affinity Fc gamma receptors. Eur.J.Immunol. 1988;18:1373–1377. doi: 10.1002/eji.1830180911. [DOI] [PubMed] [Google Scholar]
- 8.Edwards JC, Cambridge G. Rheumatoid arthritis: the predictable effect of small immune complexes in which antibody is also antigen. Br.J.Rheumatol. 1998;37:126–130. doi: 10.1093/rheumatology/37.2.126. [DOI] [PubMed] [Google Scholar]
- 9.Bhatia A, Blades S, Cambridge G, Edwards JC. Differential distribution of Fc gamma RIIIa in normal human tissues and co-localization with DAF and fibrillin-1: implications for immunological microenvironments. Immunology. 1998;94:56–63. doi: 10.1046/j.1365-2567.1998.00491.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ravetch JV, Bolland S. IgG Fc receptors. Annu.Rev.Immunol. 2001;19:275–290. doi: 10.1146/annurev.immunol.19.1.275. [DOI] [PubMed] [Google Scholar]
- 11.Wirthmueller U, Kurosaki T, Murakami MS, Ravetch JV. Signal transduction by Fc gamma RIII (CD16) is mediated through the gamma chain. J.Exp.Med. 1992;175:1381–1390. doi: 10.1084/jem.175.5.1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Debets JM, van de Winkel JG, Ceuppens JL, Dieteren IE, Buurman WA. Cross-linking of both Fc gamma RI and Fc gamma RII induces secretion of tumor necrosis factor by human monocytes, requiring high affinity Fc-Fc gamma R interactions. Functional activation of Fc gamma RII by treatment with proteases or neuraminidase. J Immunol. 1990;144:1304–1310. [PubMed] [Google Scholar]
- 13.Kramer PR, Kramer SF, Guan G. 17 beta-estradiol regulates cytokine release through modulation of CD16 expression in monocytes and monocyte-derived macrophages. Arthritis Rheum. 2004;50:1967–1975. doi: 10.1002/art.20309. [DOI] [PubMed] [Google Scholar]
- 14.Rose DM, Winston BW, Chan ED, Riches DW, Gerwins P, Johnson GL, Henson PM. Fc gamma receptor cross-linking activates p42, p38, and JNK/SAPK mitogen-activated protein kinases in murine macrophages: role for p42MAPK in Fc gamma receptor-stimulated TNF-alpha synthesis. J.Immunol. 1997;158:3433–3438. [PubMed] [Google Scholar]
- 15.Trotta R, Kanakaraj P, Perussia B. Fc gamma R-dependent mitogen-activated protein kinase activation in leukocytes: a common signal transduction event necessary for expression of TNF-alpha and early activation genes. J.Exp.Med. 1996;184:1027–1035. doi: 10.1084/jem.184.3.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.M J, Ortega E, Ortega A. Low affinity Fc gamma receptors on murine macrophages: mitogen-activated protein kinase activation and AP-1 DNA binding activity. Immunol.Lett. 1999;67:251–255. doi: 10.1016/s0165-2478(99)00021-8. [DOI] [PubMed] [Google Scholar]
- 17.Kanakaraj P, Duckworth B, Azzoni L, Kamoun M, Cantley LC, Perussia B. Phosphatidylinositol-3 kinase activation induced upon Fc gamma RIIIA-ligand interaction. J.Exp.Med. 1994;179:551–558. doi: 10.1084/jem.179.2.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Trotta R, Fettucciari K, Azzoni L, Abebe B, Puorro KA, Eisenlohr LC, Perussia B. Differential role of p38 and c-Jun N-terminal kinase 1 mitogen-activated protein kinases in NK cell cytotoxicity. J Immunol. 2000;165:1782–1789. doi: 10.4049/jimmunol.165.4.1782. [DOI] [PubMed] [Google Scholar]
- 19.Ninomiya N, Hazeki K, Fukui Y, Seya T, Okada T, Hazeki O, Ui M. Involvement of phosphatidylinositol 3-kinase in Fc gamma receptor signaling. J Biol.Chem. 1994;269:22732–22737. [PubMed] [Google Scholar]
- 20.Ganesan LP, Joshi T, Fang H, Kutala VK, Roda J, Trotta R, Lehman A, Kuppusamy P, Byrd JC, Carson WE, Caligiuri MA, Tridandapani S. FcgammaR-induced production of superoxide and inflammatory cytokines is differentially regulated by SHIP through its influence on PI3K and/or Ras/Erk pathways. Blood. 2006;108:718–725. doi: 10.1182/blood-2005-09-3889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR. PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J Biol.Chem. 1995;270:27489–27494. doi: 10.1074/jbc.270.46.27489. [DOI] [PubMed] [Google Scholar]
- 22.Borsch-Haubold AG, Pasquet S, Watson SP. Direct inhibition of cyclooxygenase-1 and −2 by the kinase inhibitors SB 203580 and PD 98059. SB 203580 also inhibits thromboxane synthase. J Biol.Chem. 1998;273:28766–28772. doi: 10.1074/jbc.273.44.28766. [DOI] [PubMed] [Google Scholar]
- 23.Guha M, Mackman N. The Phosphatidylinositol 3-Kinase-Akt Pathway Limits Lipopolysaccharide Activation of Signaling Pathways and Expression of Inflammatory Mediators in Human Monocytic Cells. J.Biol.Chem. 2002;277:32124–32132. doi: 10.1074/jbc.M203298200. [DOI] [PubMed] [Google Scholar]
- 24.Davies SP, Reddy H, Caivano M, Cohen P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem.J. 2000;351:95–105. doi: 10.1042/0264-6021:3510095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ziegler-Heitbrock HW, Sternsdorf T, Liese J, Belohradsky B, Weber C, Wedel A, Schreck R, Bauerle P, Strobel M. Pyrrolidine dithiocarbamate inhibits NF-kappa B mobilization and TNF production in human monocytes. J Immunol. 1993;151:6986–6993. [PubMed] [Google Scholar]
- 26.Sanchez JF, Sniderhan LF, Williamson AL, Fan S, Chakraborty-Sett S, Maggirwar SB. Glycogen Synthase Kinase 3{beta}-Mediated Apoptosis of Primary Cortical Astrocytes Involves Inhibition of Nuclear Factor {kappa}B Signaling. Mol.Cell.Biol. 2003;23:4649–4662. doi: 10.1128/MCB.23.13.4649-4662.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rommel C, Clarke BA, Zimmermann S, Nunez L, Rossman R, Reid K, Moelling K, Yancopoulos GD, Glass DJ. Differentiation stage-specific inhibition of the Raf-MEK-ERK pathway by Akt. Science. 1999;286:1738–1741. doi: 10.1126/science.286.5445.1738. [DOI] [PubMed] [Google Scholar]
- 28.Zimmermann S, Moelling K. Phosphorylation and regulation of Raf by Akt (protein kinase B) Science. 1999;286:1741–1744. doi: 10.1126/science.286.5445.1741. [DOI] [PubMed] [Google Scholar]
- 29.Rose DM, Winston BW, Chan ED, Riches DW, Henson PM. Interferon-gamma and transforming growth factor-beta modulate the activation of mitogen-activated protein kinases and tumor necrosis factor-alpha production induced by Fc gamma-receptor stimulation in murine macrophages. Biochem.Biophys.Res.Commun. 1997;238:256–260. doi: 10.1006/bbrc.1997.7271. [DOI] [PubMed] [Google Scholar]
- 30.Durden DL, Kim HM, Calore B, Liu Y. The Fc gamma RI receptor signals through the activation of hck and MAP kinase. J.Immunol. 1995;154:4039–4047. [PubMed] [Google Scholar]
- 31.Cao X, Wei G, Fang H, Guo J, Weinstein M, Marsh CB, Ostrowski MC, Tridandapani S. The inositol 3-phosphatase PTEN negatively regulates Fc gamma receptor signaling, but supports Toll-like receptor 4 signaling in murine peritoneal macrophages. J Immunol. 2004;172:4851–4857. doi: 10.4049/jimmunol.172.8.4851. [DOI] [PubMed] [Google Scholar]
- 32.Choi EK, Jang HC, Kim JH, Kim HJ, Kang HC, Paek YW, Lee HC, Lee SH, Oh WM, Kang IC. Enhancement of cytokine-mediated NF-kappaB activation by phosphatidylinositol 3-kinase inhibitors in monocytic cells. Int.Immunopharmacol. 2006;6:908–915. doi: 10.1016/j.intimp.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 33.da Silva FP, Aloulou M, Skurnik D, Benhamou M, Andremont A, Velasco IT, Chiamolera M, Verbeek JS, Launay P, Monteiro RC. CD16 promotes Escherichia coli sepsis through an FcR[gamma] inhibitory pathway that prevents phagocytosis and facilitates inflammation. Nat Med. 2007;13:1368–1374. doi: 10.1038/nm1665. [DOI] [PubMed] [Google Scholar]
- 34.Srivastava AK, Pandey SK. Potential mechanism(s) involved in the regulation of glycogen synthesis by insulin. Mol Cell Biochem. 1998;182:135–141. [PubMed] [Google Scholar]
- 35.Cross DA, Culbert AA, Chalmers KA, Facci L, Skaper SD, Reith AD. Selective small-molecule inhibitors of glycogen synthase kinase-3 activity protect primary neurones from death. J Neurochem. 2001;77:94–102. doi: 10.1046/j.1471-4159.2001.t01-1-00251.x. [DOI] [PubMed] [Google Scholar]
- 36.Bournat JC, Brown AM, Soler AP. Wnt-1 dependent activation of the survival factor NF-kappaB in PC12 cells. J Neurosci.Res. 2000;61:21–32. doi: 10.1002/1097-4547(20000701)61:1<21::AID-JNR3>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 37.Hoeflich KP, Luo J, Rubie EA, Tsao MS, Jin O, Woodgett JR. Requirement for glycogen synthase kinase-3beta in cell survival and NF-kappaB activation. Nature. 2000;406:86–90. doi: 10.1038/35017574. [DOI] [PubMed] [Google Scholar]
- 38.Klein PS, Melton DA. A molecular mechanism for the effect of lithium on development. Proc.Natl.Acad.Sci.U.S.A. 1996;93:8455–8459. doi: 10.1073/pnas.93.16.8455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Stambolic V, Ruel L, Woodgett JR. Lithium inhibits glycogen synthase kinase-3 activity and mimics wingless signalling in intact cells. Curr.Biol. 1996;6:1664–1668. doi: 10.1016/s0960-9822(02)70790-2. [DOI] [PubMed] [Google Scholar]
- 40.Buss H, Dorrie A, Schmitz ML, Frank R, Livingstone M, Resch K, Kracht M. Phosphorylation of Serine 468 by GSK-3{beta} Negatively Regulates Basal p65 NF-{kappa}B Activity. J.Biol.Chem. 2004;279:49571–49574. doi: 10.1074/jbc.C400442200. [DOI] [PubMed] [Google Scholar]
- 41.Schwabe RF, Brenner DA. Role of glycogen synthase kinase-3 in TNF-alpha-induced NF-kappaB activation and apoptosis in hepatocytes. Am.J Physiol Gastrointest.Liver Physiol. 2002;283:G204–G211. doi: 10.1152/ajpgi.00016.2002. [DOI] [PubMed] [Google Scholar]
- 42.Steinbrecher KA, Wilson W, III, Cogswell PC, Baldwin AS. Glycogen synthase kinase 3beta functions to specify gene-specific NF-kappaB-dependent transcription. Mol Cell Biol. 2005;25:8444–8455. doi: 10.1128/MCB.25.19.8444-8455.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schmitz ML, Stelzer G, Altmann H, Meisterernst M, Baeuerle PA. Interaction of the COOH-terminal transactivation domain of p65 NF-kappa B with TATA-binding protein, transcription factor IIB, and coactivators. J Biol.Chem. 1995;270:7219–7226. doi: 10.1074/jbc.270.13.7219. [DOI] [PubMed] [Google Scholar]
- 44.Wang D, Baldwin AS., Jr Activation of nuclear factor-kappaB-dependent transcription by tumor necrosis factor-alpha is mediated through phosphorylation of RelA/p65 on serine 529. J Biol.Chem. 1998;273:29411–29416. doi: 10.1074/jbc.273.45.29411. [DOI] [PubMed] [Google Scholar]
- 45.Saffell JL, Doherty P, Tiveron MC, Morris RJ, Walsh FS. NCAM requires a cytoplasmic domain to function as a neurite outgrowth- promoting neuronal receptor. Mol.Cell Neurosci. 1995;6:521–531. doi: 10.1006/mcne.1995.0004. [DOI] [PubMed] [Google Scholar]
- 46.Rahmani M, Peron P, Weitzman J, Bakiri L, Lardeux B, Bernuau D. Functional cooperation between JunD and NF-kappaB in rat hepatocytes. Oncogene. 2001;20:5132–5142. doi: 10.1038/sj.onc.1204678. [DOI] [PubMed] [Google Scholar]
- 47.Stein B, Baldwin AS, Jr, Ballard DW, Greene WC, Angel P, Herrlich P. Cross-coupling of the NF-kappa B p65 and Fos/Jun transcription factors produces potentiated biological function. EMBO J. 1993;12:3879–3891. doi: 10.1002/j.1460-2075.1993.tb06066.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Welch GR, Wong HL, Wahl SM. Selective induction of Fc gamma RIII on human monocytes by transforming growth factor-beta. J.Immunol. 1990;144:3444–3448. [PubMed] [Google Scholar]
- 49.Perussia B, Trinchieri G. Antibody 3G8, specific for the human neutrophil Fc receptor, reacts with natural killer cells. J Immunol. 1984;132:1410–1415. [PubMed] [Google Scholar]