Abstract
Eosinophils generate and store a battery of proteins, including classical cationic proteins, cytokines, chemokines, and growth factors. Rapid secretion of these active mediators by eosinophils is central to a range of inflammatory and immunoregulatory responses. Eosinophil products are packaged within a dominant population of cytoplasmic specific granules and generally secreted by piecemeal degranulation, a process mediated by transport vesicles. Large, pleiomorphic vesiculotubular carriers were identified recently as key players for moving eosinophil proteins from granules to the plasma membrane for extracellular release. During secretion, these specialized, morphologically distinct carriers, termed eosinophil sombrero vesicles, are actively formed and direct differential and rapid release of eosinophil proteins. This review highlights recent discoveries concerning the organization of the human eosinophil secretory pathway. These discoveries are defining a broader role for large vesiculotubular carriers in the intracellular trafficking and secretion of proteins, including selective receptor-mediated mobilization and transport of cytokines.
Keywords: vesicular transport, cell biology, inflammation, piecemeal degranulation, eosinophil sombrero vesicles (EoSVs)
Introduction
Eosinophils are bone marrow-derived granulocytes with functions in allergic, inflammatory, and immunoregulatory responses (reviewed in refs. [1–5]). Eosinophils generate and store a diversity of proteins, including four major cationic proteins, cytokines, chemokines, and growth factors, all packaged within a dominant population of cytoplasmic granules with a distinguishing, unique morphology, termed specific granules (Fig. 1) [4–7].
Fig. 1.
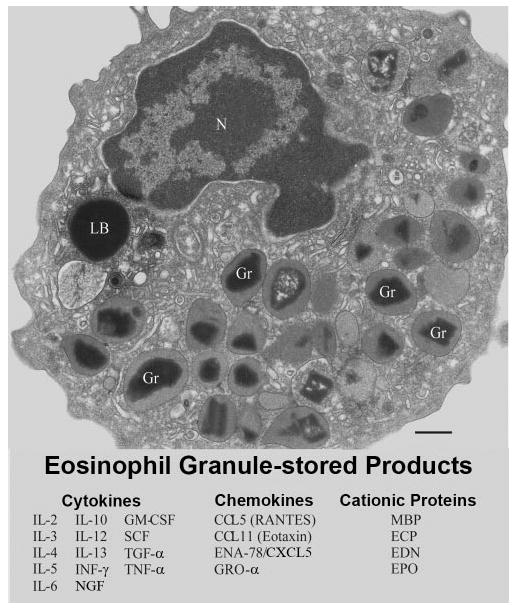
Multifunctional granule-stored products within human eosinophils. Ultrastructural image of a human eosinophil shows the cytoplasm packed with specific granules containing an internal, often electron-dense crystalline core, and cores are surrounded by an electron-lucent matrix. In response to a variety of stimuli, eosinophils secrete cytotoxic cationic proteins and an array of cytokines and chemokines. Eosinophil proteins documented within specific granules are listed. Original bar, 480 nm. Gr, Specific granules; N, nucleus; LB, lipid body. CCL5 (RANTES) [8]; CCL11 (eotaxin) [9]; ECP, eosinophil cationic protein [10]; EDN, eosinophil-derived neurotoxin [11]; ENA-78, epithelial cell-derived neutrophil-activating peptide/CXCL5 [12]; EPO, eosinophil peroxidase [10]; GM-CSF [13, 14]; GRO-α, growth-related oncogene-α [15]; IL-2 [16]; IL-3 [14]; IL-4 [17, 18]; IL-5 [14, 19]; IL-6 [20]; IL-10 (L. A. Spencer, P. F. Weller, unpublished data); IL-12 (L. A. Spencer, P. F. Weller, unpublished data); IL-13 [21]; INF-γ, (L. A. Spencer, P. F. Weller, unpublished data); MBP, major basic protein [22, 23]; NGF, nerve growth factor [24]; SCF, stem cell factor [25]; TGF-α [26]; TNF-α [27].
Secretion of these active mediators is central to eosinophil functions. In response to varied stimuli, eosinophils are recruited from the circulation into inflammatory foci, where they modulate immune responses through the release of granule-derived products (reviewed in refs. [4, 5, 28, 29]). Specific eosinophil proteins can be released rapidly and selectively when these cells are activated [30, 31].
Granule-stored products are released from eosinophils through different modes: classical exocytosis, by which granules release their entire contents following granule fusion with the plasma membrane, including compound exocytosis, also involving intracellular granule– granule fusion before extracellular release; piecemeal degranulation (PMD), a process for secretion from intracellular granules mediated by transport vesicles; and cytolysis, which involves the extracellular deposition of intact granules upon lysis of the cell (reviewed in refs. [31, 32]).
PMD is a general secretion process implicated in the release of products from activated eosinophils in a range of human diseases including allergic inflammation [33–37]. PMD has also been identified extensively in other secretory cells [38–40]. During PMD, eosinophil specific granules undergo progressive emptying of their contents, as secretory vesicles are mobilized to transfer secretory cargo from granules to the cell surface [39, 41, 42]. It is more important that PMD enables differential release of eosinophil products [30], a physiologically relevant event, which underlies distinct functions of eosinophils. How these transport vesicles are formed and mediate the rapid and specific secretion of eosinophil proteins are questions, which have been addressed recently. In addition to small, classical, round vesicles, it has now become apparent that the eosinophil secretory pathway is mediated by morphologically distinct, large pleiomorphic vesiculotubular carriers, termed eosinophil sombrero vesicles (EoSVs), which are responsible for moving proteins between granules and the plasma membrane [43– 45]. During eosinophil secretion, EoSVs are actively formed from granules and direct differential release of proteins.
In this review, we highlight the intriguing and distinct eosinophil vesicular system and the recent discoveries, based on agonist-stimulated human eosinophils, which define a broader role for large vesiculotubular carriers in intracellular trafficking and secretion of proteins, including selective receptor-mediated mobilization and transport of cytokines.
Distinct Vesicular Compartments Operate in the Eosinophil Secretory Pathway
An earlier model for the transport of secretory proteins between the eosinophil cytoplasmic granules and cell surface involves the formation and movement of small, round vesicles [41, 46, 47]. Recent data have brought conclusive evidence for the participation of morphologically distinct, large, membrane-bound, tubular compartments in the eosinophil secretory route [43–45]. Although these vesiculotubular structures have long been recognized in the cytoplasm of eosinophils [48–50] (Fig. 2A), little attention was given to them, and their functional roles remained poorly understood for 30 years. These structures were also reported previously as microgranules (reviewed in ref. [50]) or cup-shaped structures [48] in the early eosinophil literature. It is interesting that cytoplasmic vesiculotubular structures, recently referred to as EoSVs (Fig. 2A) [44], are sufficiently unique in eosinophils that their presence in the cytoplasm of granulocytes, devoid of specific granules, is useful for lineage assignment of granule-poor, activated cells [50].
Fig. 2.
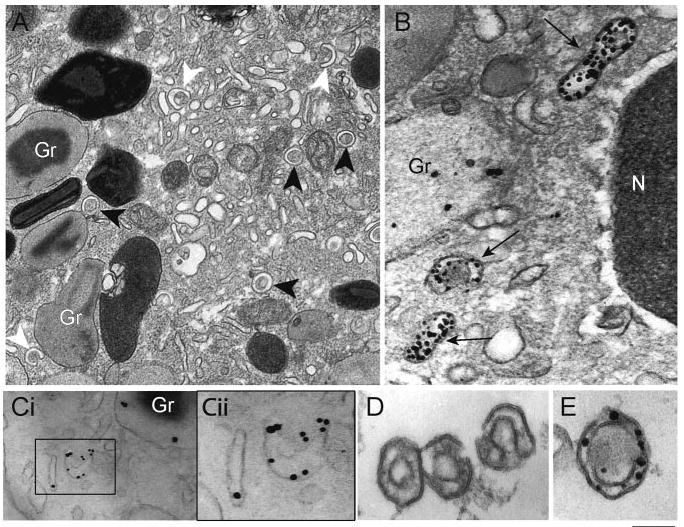
EoSVs are large, tubular carriers involved in the secretory pathway. (A) EoSVs (arrowheads) with typical morphology are observed in the cytoplasm by transmission electron microscopy (TEM). These vesicles show a “Mexican hat” (sombrero) appearance (black arrowheads) in conventional cross-thin sections (∼80 nm thickness) of eosinophils. EoSVs have a central area of cytoplasm and a brim of circular membrane-delimited vesicle. These tubular vesicles also exhibit a “C”-shaped morphology (white arrowheads). (B and C) EoSVs within activated eosinophils are immunolabeled for MBP and IL-4, respectively. Note a highly mobilized, electron lucent granule in B. (Cii) The boxed area of Ci in higher magnification. (D and E) EoSVs isolated by subcellular fractionation and visualized by, respectively, conventional TEM and after immunonanogold labeling for MBP. Note that although MBP is preferentially labeled within the vesicle lumen (B and E), IL-4 is clearly detected on vesicle membranes (C). Original bars, 400 nm (A); 230 nm (B); 250 nm (Ci); 200 nm (Cii); 180 nm (D); 150 nm (E). Gr, specific granules. (C) Reprinted from ref. [45]; (D and E) reprinted from ref. [44] with permission.
EoSVs, in conjunction with small, classical, round vesicles, represent alternative pathways for transport of granule products to the plasma membrane for extracellular release [44, 45]. Both vesicular compartments are immunolabeled positively for typical granule products [43, 44]. EPO-loaded vesicles and tubules were detected initially within eosinophils, which developed from human-cord blood mononuclear cell cultures supplemented with IL-5 [51]. Accordingly, mobilization of MBP into large tubular vesicles (Fig. 2B) was demonstrated more recently by immunonanogold EM when eosinophils were stimulated with eotaxin (CCL11), a potent CC-chemokine, which recruits and activates eosinophils [43]. MBP is one of the most abundant cationic proteins stored within and recognized as a marker of eosinophil specific granules [5, 52]. Vesicles containing MBP were identified within and extending from granules as well as around emptying granules and underneath the plasma membrane [43]. EoSVs were labeled extensively for MBP (Fig. 2A). It is interesting that the Golgi region was negative for MBP, indicating that EoSVs are not associated with a biosynthetic route from the trans-Golgi network (TGN) [43]. Another granule-derived protein, ECP, has been documented in subcellular fractionation studies to be localized in cytosolic vesicles isolated from the eosinophils of allergic patients specifically during their seasonal allergen exposures [34].
Vesicular trafficking of IL-4, a hallmark, granule-stored cytokine recognized for a long time only within cores of eosinophil granules [17, 18], was identified recently in human eosinophils using different approaches [44]. Combining pre-embedding immunonanogold EM for precise epitope preservation and subcellular localization associated with small gold particles (1.4 nm) as a probe, IL-4 was detected on cytoplasmic vesicle populations (small, spherical vesicles and EoSVs; Fig. 2C) as well as on matrices, cores, and membranes of specific granules from eotaxin-stimulated eosinophils [44]. In confirmation that vesicular compartments mediate release of IL-4 in activated eosinophils, a single probe consisting of an antibody labeled with 1.4 nm gold and a fluorochrome (fluoronanogold) was used for comparative microscopy (TEM and EliCell assay) [44]. EliCell, a sensitive immunofluorescent system [53], enabled visualization of released IL-4 at the cell surface, and immunonanogold EM showed, with the use of the same probe, direct labeling of small and large tubular vesicles [44]. It is remarkable that different TEM approaches clearly demonstrated a consistent and preferential labeling for IL-4 on vesicle membranes (Fig. 2C) and not on their internal content, as shown previously for the cytokine TGF-α in eosinophil small vesicles [26]. A functional implication of a membrane-bound vesicular transport of cytokines is that it adds support to the occurrence of selective release of products from eosinophils, as indicated previously. Moreover, as a pool of IL-4-loaded vesicles is also identified in unstimulated eosinophils, this may contribute to the rapid cytokine mobilization and release following cell activation [44].
EoSVs represent a distinct vesicle population, which can also be isolated by subcellular fractionation of human eosinophils. In contrast to small vesicles, which localize to more buoyant light fractions, EoSVs are largely localized in fractions slightly less dense than granule-containing fractions [44]. When imaged by TEM, isolated EoSVs (Fig. 2D) show the same morphology observed in conventional EM preparations of entire cells (Fig. 2A) and are positively immunolabeled for MBP (Fig. 2E) [43].
It is clear, therefore, that round vesicles and vesiculotubular structures operate in the eosinophil secretory pathway, possibly with differing contributions of each. As large tubular carriers are labeled extensively for granule products and actively formed when eosinophils are activated (see below), it seems likely that these particular vesicles are fundamental for the diversity of proteins, which needs to be transported rapidly from within eosinophils.
Intracellular Distribution and Formation of Tubular Carriers
Tubular carriers (EoSVs) are structures typically observed in mature eosinophils. Ultrastructural analysis of a large number of cultures of human umbilical cord blood cells, supplemented with a number of growth factors, has shown few numbers of EoSV-like structures within eosinophilic myelocytes. Maturation of these cells, however, is accompanied by increased numbers of EoSVs in parallel with the formation of specific granules (reviewed in ref. [7]).
In activated human eosinophils, EoSVs undergo a remarkable formation and redistribution. When eosinophils are stimulated with classical eosinophil agonists, such as eotaxin, there is an increase of the total number of cytoplasmic EoSVs [44]. In addition, EoSVs are observed more frequently surrounding and/or in contact with secretory granules [44] (Fig. 3A). By quantitative TEM, it was demonstrated that activation induces a significant increase in the numbers of granule-attached EoSVs (Fig. 3B). It is interesting that the majority of these EoSVs (90%) is associated with granules showing ultrastructural changes typical of PMD, i.e., granules with lucent areas in their cores, matrices, or both, reduced electron density, disassembled matrices and cores, residual cores, or membrane empty chambers (Fig. 3, A, C) [44]. These gross alterations within secretory granules, indicative of progressive release of their products, are described in a diversity of cell types [7, 39, 40, 54]. In human eosinophils, it is recognized that the number of emptying granules increases in activated cells, in vivo and in vitro, in different conditions [33–36, 43]. Inflammatory stimuli, such as chemokines (eotaxin and RANTES) or platelet-activating factor, trigger PMD, and pretreatment with BFA, a potential inhibitor of vesicular transport [55], inhibits agonist-induced, granule emptying [43].
Fig. 3.
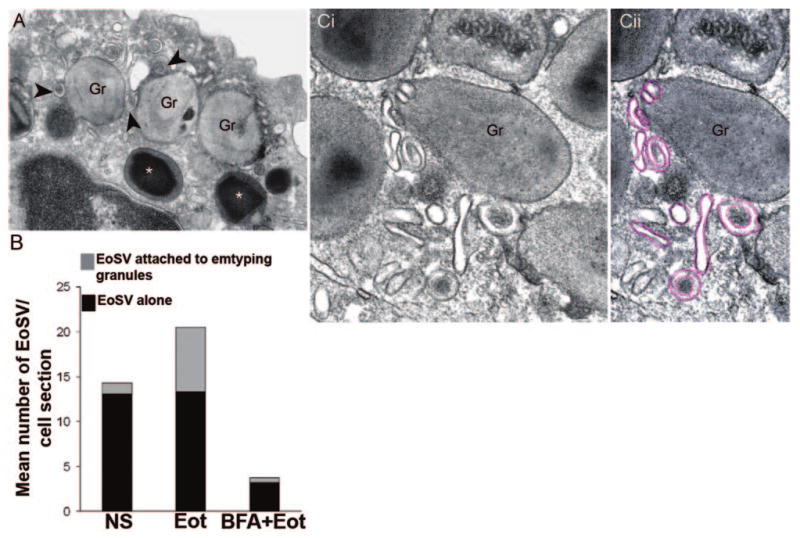
EoSVs associate with specific granules for protein transport. (A) EoSVs (arrowheads), within an eotaxin-stimulated eosinophil, are in contact with enlarged emptying granules (Gr) exhibiting reduced electron-density. Intact, nonemptying granules (*), with typical morphology, are seen close to emptying granules. (B) Quantification of EoSV numbers by TEM. Eotaxin (Eot) induced significant formation of EoSVs and association of these vesicular compartments with granules undergoing release of their products, as described in ref. [44]. Brefeldin-A (BFA) pretreatment suppressed all EoSV numbers dramatically (P<0.05). NS, Not stimulated. (C) Different profiles of EoSVs (highlighted in pink; Cii) in high magnification in contact and surrounding mobilized specific granules (Gr). Note the trilaminar unit membrane of vesicles and granules. Original bars, 400 nm (A); 250 nm (C).
Attempts to characterize the origin of EoSVs revealed that eosinophil secretory granules are able to generate these vesicles. There are several evidences for this. First, eosinophil specific granules are not merely storage stations but are elaborate and compartmentalized organelles with internal, CD63 (a transmembrane tetraspanin protein [56])-positive, membranous vesiculotubular domains [43]. These intragranular membranes are able to sequester and relocate granule products upon stimulation with eotaxin and can collapse under BFA pretreatment [43]. In parallel with the BFA-induced collapse of intragranular membranes, there was a reduction of the total number of cytoplasmic EoSVs [44] (Fig. 3B). Second, conventional TEM images strongly indicated a structural connection between EoSVs and emptying granules. EoSVs were seen attached and apparently budding from specific granules in stimulated cells (Figs. 3, A and C, and 4, A and B) [44]. Eosinophil granules can also show peroxidase-positive tubular extensions from their surfaces [42] and IL-4-loaded tubules [44]. Third, tracking of vesicle formation using 4 nm thickness digital sections by electron tomography (Fig. 4C) revealed that EoSVs can indeed emerge from mobilized granules through a tubulation process [44]. Electron tomography also showed that small, round vesicles bud from eosinophil specific granules. These findings provide direct evidence for the origin of vesicular compartments from granules undergoing release of their products by PMD.
Fig. 4.
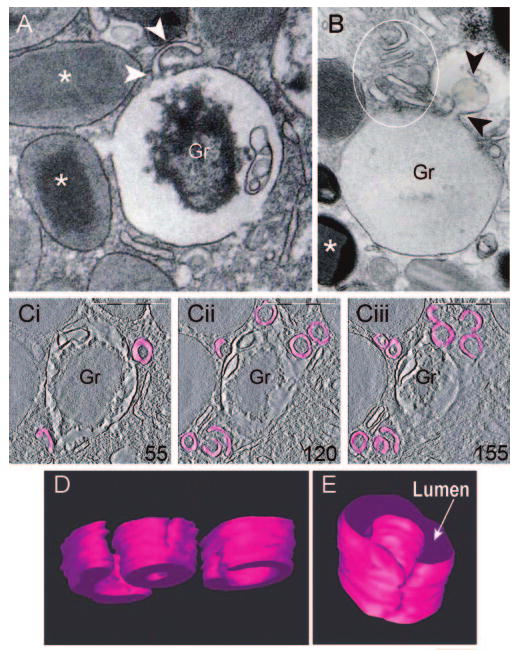
EoSVs are formed from mobilized eosinophil granules. (A and B) Images from conventional TEM (80 nm thickness sections), showing progressive stages of granule emptying. (A) A clear, tubular extension (arrowheads) is seen on the granule surface. Note the disarranged core within the granule. (B) Several EoSV profiles (circle) are in close apposition to a virtually empty granule. Arrowheads point to a large vesicle, apparently formed from the granule surface. *, Full granules. (C) Representative serial tomographic slices (4 nm thickness), obtained from electron tomography of a mobilized granule, show substantial changes associated with EoSV formation. EoSVs, the lumens of which are highlighted in pink, are imaged as open, tubular-shaped structures [44]. Numbers on the lower right corners of the panels indicate slice numbers through the tomographic volume. Cells were stimulated with eotaxin as described [44]. Original bars, 300 nm (A, B); 500 nm (C); 150 nm (D); 100 nm (E). Gr, specific granules. C–E was reprinted from Traffic [44] with permission.
Three-Dimensional (3D) Structure of EoSVs
As EoSVs were implicated directly in the secretory pathway [44], their morphology was delineated recently in more detail in human cells activated by inflammatory stimuli [43, 44, 57]. To define the spatial organization of EoSVs, they were evaluated by automated electron tomography [44, 57], a robust tool to generate 3D images of subcellular structures, which have been used increasingly in the membrane-traffic field [58–60]. Electron tomography provided new insights into the intriguing structure of EoSVs. 3D reconstructions and models generated from digital serial sections revealed that individual EoSVs are curved, tubular structures with cross-sectional diameters of 150–300 nm (Fig. 4D). Along the length of EoSVs, continuous, fully connected, cylindrical and circumferential domains and incompletely connected and only partially circumferential, curved domains were identified [44] (Fig. 4, D and E). These two domains explain the C-shaped morphology of these vesicles and the presence of elongated, tubular profiles close to typical EoSV, as frequently seen in 2D cross-sectional images of eosinophils (Fig. 2A). Electron tomography revealed therefore that EoSVs present substantial membrane surfaces and are larger and more pleiomorphic than the small, spherical vesicles (∼50 nm in diameter) classically involved in intracellular transport [44, 57]. In fact, the findings using electron tomography highlight EoSVs as a dynamic system with a remarkable ability to change their shape and to interact with secretory granules [44, 57].
The morphology of EoSVs is therefore quite distinct from that of conventional, small transport vesicles and might offer several advantages to eosinophil secretion. First, it would provide a higher surface:volume ratio system for specific transport of membrane-bound proteins. Second, tubular carriers are more effective in dealing with the long distances, which must be traversed in the cytoplasm until the cell surface is reached [61]. This fact might be particularly relevant for rapid delivery of eosinophil-preformed cytokines or other proteins. Third, EoSVs might serve as storage pools of specific proteins for rapid mobilization under stimulation and/or provide a more effective means to recycle granule membrane after the mobilization of granule products [44].
Mechanisms of Cytokine Vesicular Transport and Release from Eosinophils
Stimulus-induced release of cytokines from human eosinophils is a tightly regulated, highly selective process, occurring within minutes of agonist stimulation and raising several mechanistic questions. Upon cell stimulation, specific cytokines must be selectively mobilized, from among over two dozen other preformed, granule-stored proteins, into tubular vesicles. These tubular carriers then need to disengage from granules, traverse the cytoplasm, and dock at the plasma membrane to release their specific cargo. Paralleling recent, novel, ultrastructural analyses described above, molecular approaches have begun to uncover key mechanisms governing these processes.
ImmunoEM images clearly demonstrate association of IL-4 [44], as noted above, and TGF-α [26] with membranes of vesicular carriers, in contrast to the predominantly luminal association of MBP within vesicles [44]. Thus, the hypothesis was generated that specific membrane-associated carriers participate in the selective mobilization and trafficking of cytokines into and through vesicular compartments. In fact, intracellular, granule-associated pools of cytokine receptor chains quantitatively exceed that expressed on the cell surface, and a recent study combining immunoEM and molecular approaches demonstrated eotaxin-induced association of IL-4Rα chains with intragranular membrane reorganization, preceded by mobilization of IL-4Rα, bearing bound IL-4, from granules into secretory vesicles [45]. It is important that the signal-transducing, accessory chain of the IL-4R complex (γc chain) did not exhibit eotaxin-induced mobilization. Thus, trafficking of IL-4Rα-chaperoned IL-4 within tubular carriers may be accomplished without initiating an IL-4R-mediated signaling cascade. Intracellular stores of CCR3 and IL-6Rα were also expressed within human eosinophils, and intracellular detection of the former increased upon stimulus-induced release of RANTES, a known CCR3 ligand [45]. Receptor-mediated trafficking of cytokines, a mechanism ideally suited to the large surface area inherent in tubular carriers, is thus a likely mechanism governing the selectivity and rapid transit of cytokines for secretion.
Once loaded, granule-derived vesicles dock at appropriate membrane locales, fuse, and release their specific cargo. A series of elegant studies has demonstrated the relevance of soluble N-ethylmaleimide-sensitive factor attachment protein (SNAP) receptor (SNARE; SNAP receptors) complexes, comprised of v (vesicle) and t (target) SNARES, to this process (reviewed in ref. [31]). Specifically, eosinophil secretory vesicles, but not granules, express the v-SNARE vesicle-associated membrane protein 2, which colocalized with RANTES throughout IFN-γ-induced PMD of RANTES [62], and likely mediates specific membrane docking through interaction with plasma membrane t-SNARES, SNAP-23, and syntaxin-4 [63]. Figure 5 shows a model for mobilization and transport of cytokines from secretory granules to the plasma membrane in the human eosinophil.
Fig. 5.
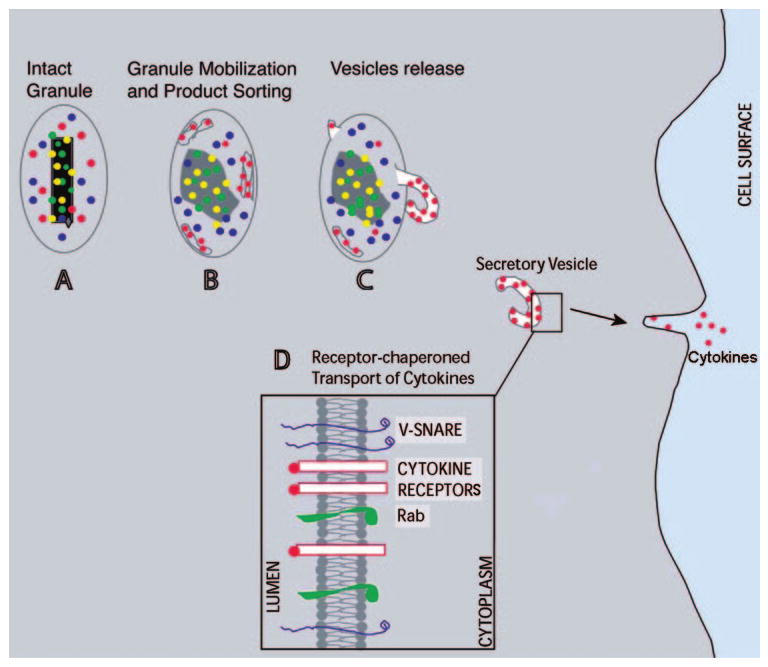
A model for mobilization and transport of cytokines from secretory granules to the plasma membrane in human eosinophils. (A) An intact granule with its electron-dense crystalline core and secretory products. (B and C) Sequential stages of structural changes related to granule mobilization and formation of tubular carriers (EoSVs), which transport a specific cytokine to the plasma membrane. (D) The differential sorting of cytokines is based on the presence of cytokine receptors in granule and vesicle membranes [45]. Rab-SNAREs mediate vesicle movement and plasma membrane fusion, resulting in cytokine secretion [31].
Large Tubular Carriers Mediate Transport in Different Cell Secretory Pathways
The identification of large tubular carriers within different cell types transferring secretory cargo signals a departure from models, which are based on small, round vesicles, as the only mediators of vesicular transport. Emerging evidence has pointed to the participation of vesiculotubular carriers in different cell secretory pathways. Well-documented examples are the transport system between the endoplasmic reticulum (ER) and Golgi complex [64–67], from the endosomal system [68] or the TGN [69, 70] to the plasma membrane, and along axons [71–72]. These carriers appear as vesiculotubular structures of various shapes and sizes. They show complex plasticity, often changing shapes or dividing during transport [73]. Moreover, it has been suggested that large transport carriers could form by fusion of small vesicles or by direct budding from donor organelles (reviewed in refs. [61, 66, 74]).
Consistent with the findings within eosinophils described above, it was demonstrated recently that the abundance of tubular carriers operating in the ER-Golgi interface, within a population of cells and in individual cells themselves, can be increased dramatically compared with steady-state conditions [65].
Large transport compartments could explain, for instance, the export of large macromolecular cargo such as procollagen from the ER or the secretion of large lipoprotein particles such as chylomicrons, which would be too large to be accommodated in 60–90 nm, small vesicles (reviewed in ref. [66]). Indeed, the transport of specific proteins within large tubular carriers has increasingly been documented. E-cadherin, a cell–cell adhesion protein, is transported from the TGN to the recycling endosome on its way to the cell surface in vesiculotubular carriers [75]. EM studies also describe an assortment of convoluted tubular-vesicular structures as vehicles for the delivery of receptor-hydrolase complexes from the TGN to the endosomal system [76]. It was demonstrated recently that IL-6 is loaded into vesiculotubular structures budding from the TGN in live macrophages [77], a finding, which coupled with our previous results [44, 45], adds support to a broader role for these large carriers in the intracellular trafficking and release of cytokines.
It is believed that large tubular carriers could provide an additional mechanism to transport material rapidly between membranes in different secretory pathways [44, 65]. The dissection of these carriers and the understanding of their intrinsic complexity are beginning to emerge.
Concluding Remarks and Questions for the Future
The classical picture of the cell secretory pathway includes protein and lipid synthesis within the ER, transport of cargo inwards toward the Golgi apparatus and then through the Golgi, and TGN en route to the plasma membrane, all carried by transport vesicles [78]. Eosinophils show a novel, secretory pathway organization, which may underlie their remarkable ability to rapidly release preformed proteins involved in immune responses. Eosinophils contain morphologically unique secretory organelles, cytoplasmic specific granules, and a distinct population of large secretory vesicles (EoSVs). During secretion, these specialized tubular carriers are actively formed, and in parallel, specific granules undergo highly dynamic changes related to the progressive release of their contents. EoSVs bud from mobilized granules and in conjunction with small, round vesicles, transport granule products to the plasma membrane for extracellular release [44, 45, 57]. Important questions remain about these secretory components and in understanding secretory trafficking events within eosinophils, such as whether proteins can be synthesized within specific granules or if there is traffic between the primary secretory organelles (ER and Golgi) and granules. Although recent findings have provided a new view of eosinophil specific granules as highly elaborate organelles with internal membranes [43], their potential capacity to synthesize proteins remains to be investigated. Although receptor-mediated recruitment of cytokines to secretory vesicles seems to be a common mechanism for differential secretion in eosinophils, other segments of the molecular pathways, controlling, for instance, sorting of intracellular receptors, await further investigation. Other issues still need to be addressed: Is there a retrograde trafficking of tubular carrier membranes to specific granules after delivery? Answers to basic cell biological questions about eosinophil secretory trafficking are critical, not only to understand normal leukocyte function but also to understand the pathological basis of allergic and inflammatory diseases, including asthma, often linked intimately to eosinophils.
Acknowledgments
The work of the authors is supported by National Institutes of Health grants AI33372, AI20241, AI051645, AI22571, and HL70270 (Bethesda, MD, USA) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil). We thank Rita Monahan-Earley, Tracey Sciuto, and Ellen Morgan (Electron Microscopy Unit, Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA) and Wim Voorhout of FEI Company (Eindhoven, The Netherlands) for previous electron microscopy assistance.
References
- 1.Rosenberg HF, Phipps S, Foster PS. Eosinophil trafficking in allergy and asthma. J Allergy Clin Immunol. 2007;119:1303–1310. doi: 10.1016/j.jaci.2007.03.048. [DOI] [PubMed] [Google Scholar]
- 2.Trivedi SG, Lloyd CM. Eosinophils in the pathogenesis of allergic airways disease. Cell Mol Life Sci. 2007;64:1269–1289. doi: 10.1007/s00018-007-6527-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jacobsen EA, Taranova AG, Lee NA, Lee JJ. Eosinophils: singularly destructive effector cells or purveyors of immunoregulation? J Allergy Clin Immunol. 2007;119:1313–1320. doi: 10.1016/j.jaci.2007.03.043. [DOI] [PubMed] [Google Scholar]
- 4.Rothenberg ME, Hogan SP. The eosinophil. Annu Rev Immunol. 2006;24:147–174. doi: 10.1146/annurev.immunol.24.021605.090720. [DOI] [PubMed] [Google Scholar]
- 5.Gleich GJ. Mechanisms of eosinophil-associated inflammation. J Allergy Clin Immunol. 2000;105:651–663. doi: 10.1067/mai.2000.105712. [DOI] [PubMed] [Google Scholar]
- 6.Melo RCN, Weller PF, Dvorak AM. Activated human eosinophils. Int Arch Allergy Immunol. 2005;138:347–349. doi: 10.1159/000089189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dvorak AM, Ishizaka T. Human eosinophils in vitro. An ultrastructural morphology primer. Histol Histopathol. 1994;9:339–374. [PubMed] [Google Scholar]
- 8.Lacy P, Mahmudi-Azer S, Bablitz B, Hagen SC, Velazquez JR, Man SF, Moqbel R. Rapid mobilization of intracellularly stored RANTES in response to interferon-γ in human eosinophils. Blood. 1999;94:23–32. [PubMed] [Google Scholar]
- 9.Nakajima T, Yamada H, Iikura M, Miyamasu M, Izumi S, Shida H, Ohta K, Imai T, Yoshie O, Mochizuki M, Schroder JM, Morita Y, Yamamoto K, Kirai K. Intracellular localization and release of eotaxin from normal eosinophils. FEBS Lett. 1998;434:226–230. doi: 10.1016/s0014-5793(98)00863-1. [DOI] [PubMed] [Google Scholar]
- 10.Egesten A, Alumets J, von Mecklenburg C, Palmegren M, Olsson I. Localization of eosinophil cationic protein, major basic protein, and eosinophil peroxidase in human eosinophils by immunoelectron microscopic technique. J Histochem Cytochem. 1986;34:1399–1403. doi: 10.1177/34.11.3772075. [DOI] [PubMed] [Google Scholar]
- 11.Peters MS, Rodriguez M, Gleich GJ. Localization of human eosinophil granule major basic protein, eosinophil cationic protein, and eosinophil-derived neurotoxin by immunoelectron microscopy. Lab Invest. 1986;54:656–662. [PubMed] [Google Scholar]
- 12.Persson T, Monsef N, Andersson P, Bjartell A, Malm J, Calafat J, Egesten A. Expression of the neutrophil-activating CXC chemokine ENA-78/CXCL5 by human eosinophils. Clin Exp Allergy. 2003;33:531–537. doi: 10.1046/j.1365-2222.2003.01609.x. [DOI] [PubMed] [Google Scholar]
- 13.Levi-Schaffer F, Lacy P, Severs NJ, Newman TM, North J, Gomperts B, Kay AB, Moqbel R. Association of granulocyte-macrophage colony-stimulating factor with the crystalloid granules of human eosinophils. Blood. 1995;85:2579–2586. [PubMed] [Google Scholar]
- 14.Desreumaux P, Delaporte E, Colombel JF, Capron M, Cortot A, Janin A. Similar IL-5, IL-3, and GM-CSF syntheses by eosinophils in the jejunal mucosa of patients with celiac disease and dermatitis herpetiformis. Clin Immunol Immunopathol. 1998;88:14–21. doi: 10.1006/clin.1997.4494. [DOI] [PubMed] [Google Scholar]
- 15.Persson-Dajotoy T, Andersson P, Bjartell A, Calafat J, Egesten A. Expression and production of the CXC chemokine growth-related oncogene-α by human eosinophils. J Immunol. 2003;170:5309–5316. doi: 10.4049/jimmunol.170.10.5309. [DOI] [PubMed] [Google Scholar]
- 16.Levi-Schaffer F, Barkans J, Newman TM, Ying S, Wakelin M, Hohenstein R, Barak V, Lacy P, Kay AB, Moqbel R. Identification of interleukin-2 in human peripheral blood eosinophils. Immunology. 1996;87:155–161. [PMC free article] [PubMed] [Google Scholar]
- 17.Moqbel R, Ying S, Barkans J, Newman TM, Kimmitt P, Wakelin M, Taborda-Barata L, Meng Q, Corrigan CJ, Durham SR, Kay AB. Identification of messenger RNA for IL-4 in human eosinophils with granule localization and release of the translated product. J Immunol. 1995;155:4939–4947. [PubMed] [Google Scholar]
- 18.Moller GM, de Jong TA, van der Kwast TH, Overbeek SE, Wierenga-Wolf AF, Thepen T, Hoogsteden HC. Immunolocalization of interleukin-4 in eosinophils in the bronchial mucosa of atopic asthmatics. Am J Respir Cell Mol Biol. 1996;14:439–443. doi: 10.1165/ajrcmb.14.5.8624248. [DOI] [PubMed] [Google Scholar]
- 19.Moller GM, de Jong TA, Overbeek SE, van der Kwast TH, Postma DS, Hoogsteden HC. Ultrastructural immunogold localization of interleukin 5 to the crystalloid core compartment of eosinophil secondary granules in patients with atopic asthma. J Histochem Cytochem. 1996;44:67–69. doi: 10.1177/44.1.8543784. [DOI] [PubMed] [Google Scholar]
- 20.Lacy P, Levi-Schaffer F, Mahmudi-Azer S, Bablitz B, Hagen SC, Velazquez J, Kay AB, Moqbel R. Intracellular localization of interleukin-6 in eosinophils from atopic asthmatics and effects of interferon γ. Blood. 1998;91:2508–2516. [PubMed] [Google Scholar]
- 21.Woerly G, Lacy P, Younes AB, Roger N, Loiseau S, Moqbel R, Capron M. Human eosinophils express and release IL-13 following CD28-dependent activation. J Leukoc Biol. 2002;72:769–779. [PubMed] [Google Scholar]
- 22.Gleich GJ, Loegering DA, Maldonado JE. Identification of a major basic protein in guinea pig eosinophil granules. J Exp Med. 1973;137:1459–1471. doi: 10.1084/jem.137.6.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lewis DM, Lewis JC, Loegering DA, Gleich GJ. Localization of the guinea pig eosinophil major basic protein to the core of the granule. J Cell Biol. 1978;77:702–713. doi: 10.1083/jcb.77.3.702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Toyoda M, Nakamura M, Makino T, Morohashi M. Localization and content of nerve growth factor in peripheral blood eosinophils of atopic dermatitis patients. Clin Exp Allergy. 2003;33:950–955. doi: 10.1046/j.1365-2222.2003.01719.x. [DOI] [PubMed] [Google Scholar]
- 25.Hartman M, Piliponsky AM, Temkin V, Levi-Schaffer F. Human peripheral blood eosinophils express stem cell factor. Blood. 2001;97:1086–1091. doi: 10.1182/blood.v97.4.1086. [DOI] [PubMed] [Google Scholar]
- 26.Egesten A, Calafat J, Knol EF, Janssen H, Walz TM. Subcellular localization of transforming growth factor-α in human eosinophil granulocytes. Blood. 1996;87:3910–3918. [PubMed] [Google Scholar]
- 27.Beil WJ, Weller PF, Tzizik DM, Galli SJ, Dvorak AM. Ultrastructural immunogold localization of tumor necrosis factor- α to the matrix compartment of eosinophil secondary granules in patients with idiopathic hypereosinophilic syndrome. J Histochem Cytochem. 1993;41:1611–1615. doi: 10.1177/41.11.8409368. [DOI] [PubMed] [Google Scholar]
- 28.Munitz A, Levi-Schaffer F. Eosinophils: ‘new’ roles for ‘old’ cells. Allergy. 2004;59:268–275. doi: 10.1111/j.1398-9995.2003.00442.x. [DOI] [PubMed] [Google Scholar]
- 29.Adamko DJ, Odemuyiwa SO, Vethanayagam D, Moqbel R. The rise of the phoenix: the expanding role of the eosinophil in health and disease. Allergy. 2005;60:13–22. doi: 10.1111/j.1398-9995.2005.00676.x. [DOI] [PubMed] [Google Scholar]
- 30.Bandeira-Melo C, Sugiyama K, Woods LJ, Weller PF. Cutting edge: eotaxin elicits rapid vesicular transport-mediated release of preformed IL-4 from human eosinophils. J Immunol. 2001;166:4813–4817. doi: 10.4049/jimmunol.166.8.4813. [DOI] [PubMed] [Google Scholar]
- 31.Moqbel R, Coughlin JJ. Differential secretion of cytokines. Sci STKE. 2006;2006:pe26. doi: 10.1126/stke.3382006pe26. [DOI] [PubMed] [Google Scholar]
- 32.Erjefalt JS, Persson CG. New aspects of degranulation and fates of airway mucosal eosinophils. Am J Respir Crit Care Med. 2000;161:2074–2085. doi: 10.1164/ajrccm.161.6.9906085. [DOI] [PubMed] [Google Scholar]
- 33.Dvorak AM, Monahan RA, Osage JE, Dickersin GR. Crohn's disease: transmission electron microscopic studies. II. Immunologic inflammatory response Alterations of mast cells, basophils, eosinophils, and the microvasculature. Hum Pathol. 1980;11:606–619. doi: 10.1016/s0046-8177(80)80072-4. [DOI] [PubMed] [Google Scholar]
- 34.Karawajczyk M, Seveus L, Garcia R, Bjornsson E, Peterson CG, Roomans GM, Venge P. Piecemeal degranulation of peripheral blood eosinophils: a study of allergic subjects during and out of the pollen season. Am J Respir Cell Mol Biol. 2000;23:521–529. doi: 10.1165/ajrcmb.23.4.4025. [DOI] [PubMed] [Google Scholar]
- 35.Erjefalt JS, Greiff L, Andersson M, Adelroth E, Jeffery PK, Persson CG. Degranulation patterns of eosinophil granulocytes as determinants of eosinophil driven disease. Thorax. 2001;56:341–344. doi: 10.1136/thorax.56.5.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ahlstrom-Emanuelsson CA, Greiff L, Andersson M, Persson CG, Erjefalt JS. Eosinophil degranulation status in allergic rhinitis: observations before and during seasonal allergen exposure. Eur Respir J. 2004;24:750–757. doi: 10.1183/09031936.04.00133603. [DOI] [PubMed] [Google Scholar]
- 37.Caruso RA, Ieni A, Fedele F, Zuccala V, Riccardo M, Parisi E, Parisi A. Degranulation patterns of eosinophils in advanced gastric carcinoma: an electron microscopic study. Ultrastruct Pathol. 2005;29:29–36. doi: 10.1080/019131290882303. [DOI] [PubMed] [Google Scholar]
- 38.Dvorak AM. Basophils and mast cells: piecemeal degranulation in situ and ex vivo: a possible mechanism for cytokine-induced function in disease. In: Coffey RG, editor. Granulocyte Responses to Cytokines Basic and Clinical Research. New York: Marcel Dekker; 1992. pp. 169–271. [PubMed] [Google Scholar]
- 39.Dvorak AM. Ultrastructure of Mast Cells and Basophils Chem Immunol Allergy. Vol. 85. Basel: Karger; 2005. pp. 1–351. [Google Scholar]
- 40.Crivellato E, Nico B, Mallardi F, Beltrami CA, Ribatti D. Piecemeal degranulation as a general secretory mechanism? Anat Rec A Discov Mol Cell Evol Biol. 2003;274:778–784. doi: 10.1002/ar.a.10095. [DOI] [PubMed] [Google Scholar]
- 41.Dvorak AM, Furitsu T, Letourneau L, Ishizaka T, Ackerman SJ. Mature eosinophils stimulated to develop in human cord blood mononuclear cell cultures supplemented with recombinant human interleukin-5. I. Piecemeal degranulation of specific granules and distribution of Charcot-Leyden crystal protein. Am J Pathol. 1991;138:69–82. [PMC free article] [PubMed] [Google Scholar]
- 42.Dvorak AM, Ackerman SJ, Furitsu T, Estrella P, Letourneau L, Ishizaka T. Mature eosinophils stimulated to develop in human-cord blood mononuclear cell cultures supplemented with recombinant human interleukin-5. II. Vesicular transport of specific granule matrix peroxidase, a mechanism for effecting piecemeal degranulation. Am J Pathol. 1992;140:795–807. [PMC free article] [PubMed] [Google Scholar]
- 43.Melo RCN, Perez SAC, Spencer LA, Dvorak AM, Weller PF. Intragranular vesiculotubular compartments are involved in piecemeal degranulation by activated human eosinophils. Traffic. 2005;6:866–879. doi: 10.1111/j.1600-0854.2005.00322.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Melo RCN, Spencer LA, Perez SAC, Ghiran I, Dvorak AM, Weller PF. Human eosinophils secrete preformed, granule-stored interleukin-4 (IL-4) through distinct vesicular compartments. Traffic. 2005;6:1047–1057. doi: 10.1111/j.1600-0854.2005.00344.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Spencer LA, Melo RCN, Perez SA, Bafford SP, Dvorak AM, Weller PF. Cytokine receptor-mediated trafficking of preformed IL-4 in eosinophils identifies an innate immune mechanism of cytokine secretion. Proc Natl Acad Sci USA. 2006;103:3333–3338. doi: 10.1073/pnas.0508946103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dvorak HF, Dvorak AM. Basophilic leucocytes: structure, function and role in disease. Clin Haematol. 1975;4:651–683. [PubMed] [Google Scholar]
- 47.Dvorak AM. Ultrastructural observations of in situ, ex vivo and in vitro sites, sources, and systems. In: Kaliner MA, Metcalfe DD, editors. The Mast Cell in Health and Disease. 1992. [Google Scholar]; Lenfant C, editor. Lung Biology in Health and Disease Series. Vol. 62. New York: Marcel Dekker; pp. 1–90. [Google Scholar]
- 48.Komiyama A, Spicer SS. Microendocytosis in eosinophilic leukocytes. J Cell Biol. 1975;64:622–635. doi: 10.1083/jcb.64.3.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Okuda M, Takenaka T, Kawabori S, Ogami Y. Ultrastructural study of the specific granule of the human eosinophil. J Submicrosc Cytol. 1981;13:465–471. [PubMed] [Google Scholar]
- 50.Dvorak AM, Weller PF. Ultrastructural analysis of human eosinophils. In: Marone G, editor. Human Eosinophils: Biological and Chemical Aspects. Vol. 76. Basel, Switzerland: S. Karger; 2000. pp. 1–28. [DOI] [PubMed] [Google Scholar]
- 51.Dvorak AM, Estrella P, Ishizaka T. Vesicular transport of peroxidase in human eosinophilic myelocytes. Clin Exp Allergy. 1994;24:10–18. doi: 10.1111/j.1365-2222.1994.tb00910.x. [DOI] [PubMed] [Google Scholar]
- 52.Lacy P, Moqbel R. Eosinophil cytokines. Chem Immunol. 2000;76:134–155. doi: 10.1159/000058782. [DOI] [PubMed] [Google Scholar]
- 53.Bandeira-Melo C, Perez SAC, Melo RCN, Ghiran I, Weller PF. EliCell assay for the detection of released cytokines from eosinophils. J Immunol Methods. 2003;276:227–237. doi: 10.1016/s0022-1759(03)00076-0. [DOI] [PubMed] [Google Scholar]
- 54.Dvorak AM, Morgan ES, Lichtenstein LM, MacGlashan DW., Jr Activated human basophils contain histamine in cytoplasmic vesicles. Int Arch Allergy Immunol. 1994;105:8–11. doi: 10.1159/000236796. [DOI] [PubMed] [Google Scholar]
- 55.Nebenfuhr A, Ritzenthaler C, Robinson DG. Brefeldin A: deciphering an enigmatic inhibitor of secretion. Plant Physiol. 2002;130:1102–1108. doi: 10.1104/pp.011569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mahmudi-Azer S, Downey GP, Moqbel R. Translocation of the tetraspanin CD63 in association with human eosinophil mediator release. Blood. 2002;99:4039–4047. doi: 10.1182/blood.v99.11.4039. [DOI] [PubMed] [Google Scholar]
- 57.Melo RCN, Dvorak AM, Weller PF. Electron tomography reveals the 3D structure of secretory organelles in eosinophils. Microscopy & Analysis. 2007;21:15–17. [Google Scholar]
- 58.McEwen BF, Marko M. The emergence of electron tomography as an important tool for investigating cellular ultrastructure. J Histochem Cytochem. 2001;49:553–564. doi: 10.1177/002215540104900502. [DOI] [PubMed] [Google Scholar]
- 59.Koster AJ, Klumperman J. Electron microscopy in cell biology: integrating structure and function. Nat Rev Mol Cell Biol. 2003;(Suppl):SS6–SS10. [PubMed] [Google Scholar]
- 60.Trucco A, Polishchuk RS, Martella O, Di Pentima A, Fusella A, Di Giandomenico D, San Pietro E, Beznoussenko GV, Polishchuk EV, Baldassarre M, Buccione R, Geerts WJ, Koster AJ, Burger KN, Mironov AA, Luini A. Secretory traffic triggers the formation of tubular continuities across Golgi sub-compartments. Nat Cell Biol. 2004;6:1071–1081. doi: 10.1038/ncb1180. [DOI] [PubMed] [Google Scholar]
- 61.Bonifacino JS, Lippincott-Schwartz J. Coat proteins: shaping membrane transport. Nat Rev Mol Cell Biol. 2003;4:409–414. doi: 10.1038/nrm1099. [DOI] [PubMed] [Google Scholar]
- 62.Lacy P, Logan MR, Bablitz B, Moqbel R. Fusion protein vesicle-associated membrane protein 2 is implicated in IFN-γ-induced piecemeal degranulation in human eosinophils from atopic individuals. J Allergy Clin Immunol. 2001;107:671–678. doi: 10.1067/mai.2001.113562. [DOI] [PubMed] [Google Scholar]
- 63.Logan MR, Lacy P, Bablitz B, Moqbel R. Expression of eosinophil target SNAREs as potential cognate receptors for vesicle-associated membrane protein-2 in exocytosis. J Allergy Clin Immunol. 2002;109:299–306. doi: 10.1067/mai.2002.121453. [DOI] [PubMed] [Google Scholar]
- 64.Mironov AA, Mironov AA, Jr, Beznoussenko GV, Trucco A, Lupetti P, Smith JD, Geerts WJ, Koster AJ, Burger KN, Martone ME, Deerinck TJ, Ellisman MH, Luini A. ER-to-Golgi carriers arise through direct en bloc protrusion and multistage maturation of specialized ER exit domains. Dev Cell. 2003;5:583–594. doi: 10.1016/s1534-5807(03)00294-6. [DOI] [PubMed] [Google Scholar]
- 65.Simpson JC, Nilsson T, Pepperkok R. Biogenesis of tubular ER-to-Golgi transport intermediates. Mol Biol Cell. 2006;17:723–737. doi: 10.1091/mbc.E05-06-0580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Watson P, Stephens DJ. ER-to-Golgi transport: form and formation of vesicular and tubular carriers. Biochim Biophys Acta. 2005;1744:304–315. doi: 10.1016/j.bbamcr.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 67.Martinez-Menarguez JA, Geuze HJ, Slot JW, Klumperman J. Vesicular tubular clusters between the ER and Golgi mediate concentration of soluble secretory proteins by exclusion from COPI-coated vesicles. Cell. 1999;98:81–90. doi: 10.1016/S0092-8674(00)80608-X. [DOI] [PubMed] [Google Scholar]
- 68.Peden AA, Oorschot V, Hesser BA, Austin CD, Scheller RH, Klumperman J. Localization of the AP-3 adaptor complex defines a novel endosomal exit site for lysosomal membrane proteins. J Cell Biol. 2004;164:1065–1076. doi: 10.1083/jcb.200311064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hirschberg K, Miller CM, Ellenberg J, Presley JF, Siggia ED, Phair RD, Lippincott-Schwartz J. Kinetic analysis of secretory protein traffic and characterization of Golgi to plasma membrane transport intermediates in living cells. J Cell Biol. 1998;143:1485–1503. doi: 10.1083/jcb.143.6.1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rustom A, Bajohrs M, Kaether C, Keller P, Toomre D, Corbeil D, Gerdes HH. Selective delivery of secretory cargo in Golgi-derived carriers of nonepithelial cells. Traffic. 2002;3:279–288. doi: 10.1034/j.1600-0854.2002.030405.x. [DOI] [PubMed] [Google Scholar]
- 71.Horton AC, Ehlers MD. Secretory trafficking in neuronal dendrites. Nat Cell Biol. 2004;6:585–591. doi: 10.1038/ncb0704-585. [DOI] [PubMed] [Google Scholar]
- 72.Ahmari SE, Buchanan J, Smith SJ. Assembly of presynaptic active zones from cytoplasmic transport packets. Nat Neurosci. 2000;3:445–451. doi: 10.1038/74814. [DOI] [PubMed] [Google Scholar]
- 73.Puertollano R, van der Wel NN, Greene LE, Eisenberg E, Peters PJ, Bonifacino JS. Morphology and dynamics of clathrin/GGA1-coated carriers budding from the trans-Golgi network. Mol Biol Cell. 2003;14:1545–1557. doi: 10.1091/mbc.02-07-0109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Luini A, Ragnini-Wilson A, Polishchuck RS, De Matteis MA. Large pleiomorphic traffic intermediates in the secretory pathway. Curr Opin Cell Biol. 2005;17:353–361. doi: 10.1016/j.ceb.2005.06.012. [DOI] [PubMed] [Google Scholar]
- 75.Lock JG, Hammond LA, Houghton F, Gleeson PA, Stow JL. E-cadherin transport from the trans-Golgi network in tubulovesicular carriers is selectively regulated by golgin-97. Traffic. 2005;6:1142–1156. doi: 10.1111/j.1600-0854.2005.00349.x. [DOI] [PubMed] [Google Scholar]
- 76.Polishchuk RS, San Pietro E, Di Pentima A, Tete S, Bonifacino JS. Ultrastructure of long-range transport carriers moving from the trans Golgi network to peripheral endosomes. Traffic. 2006;7:1092–1103. doi: 10.1111/j.1600-0854.2006.00453.x. [DOI] [PubMed] [Google Scholar]
- 77.Manderson AP, Kay JG, Hammond LA, Brown DL, Stow JL. Subcompartments of the macrophage recycling endosome direct the differential secretion of IL-6 and TNF{α} J Cell Biol. 2007;178:57–69. doi: 10.1083/jcb.200612131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Derby MC, Gleeson PA. New insights into membrane trafficking and protein sorting. Int Rev Cytol. 2007;261:47–116. doi: 10.1016/S0074-7696(07)61002-X. [DOI] [PubMed] [Google Scholar]


