Abstract
Dendritic cell (DC) responses to extracellular self-DNA and self-RNA are prevented by the endosomal seclusion of nucleic acid–recognizing Toll-like receptors (TLRs). In psoriasis, however, plasmacytoid DCs (pDCs) sense self-DNA that is transported to endosomal TLR9 upon forming a complex with the antimicrobial peptide LL37. Whether LL37 also interacts with extracellular self-RNA and how this may contribute to DC activation in psoriasis is not known. Here, we report that LL37 can bind self-RNA released by dying cells, protect it from extracellular degradation, and transport it into endosomal compartments of DCs. In pDC, self-RNA–LL37 complexes activate TLR7 and, like self-DNA–LL37 complexes, trigger the secretion of IFN-α without inducing maturation or the production of IL-6 and TNF-α. In contrast to self-DNA–LL37 complexes, self-RNA–LL37 complexes also trigger the activation of classical myeloid DCs (mDCs). This occurs through TLR8 and leads to the production of TNF-α and IL-6, and the differentiation of mDCs into mature DCs. We also found that self-RNA–LL37 complexes are present in psoriatic skin lesions and are associated with mature mDCs in vivo. Our results demonstrate that the cationic antimicrobial peptide LL37 converts self-RNA into a trigger of TLR7 and TLR8 in human DCs, and provide new insights into the mechanism that drives the auto-inflammatory responses in psoriasis.
Dendritic cells (DCs) sense viral infections through a subset of nucleic acid–recognizing Toll-like receptors (TLRs) expressed in endosomal compartments (Akira et al., 2006). These receptors include TLR3, which detects double-stranded viral RNA (Alexopoulou et al., 2001); TLR7 and TLR8, which recognize guanosine- and uridine-rich single-stranded RNA (ssRNA) (Diebold et al., 2004; Heil et al., 2004; Lund et al., 2004); and TLR9, which recognizes the phosphodiester backbone in natural DNA or unmethylated CpG motifs (Hemmi et al., 2000; Haas et al., 2008). TLR7 and TLR9 are selectively expressed by human plasmacytoid dendritic cells (pDCs) (Jarrossay et al., 2001; Kadowaki et al., 2001; Hornung et al., 2002), a subset of dendritic cells specialized in type I IFN production (Colonna et al., 2004; Gilliet et al., 2008). In contrast, classical human myeloid DCs (mDCs), which are potent stimulators of T cell responses by virtue of their inherent capacity to present antigens and migrate from the periphery into secondary lymphoid organs, do not express TLR7 and TLR9, but instead express TLR3 and TLR8 (Jarrossay et al., 2001; Kadowaki et al., 2001; Hornung et al., 2002).
Through endosomal TLRs, pDCs and mDCs efficiently sense viral nucleic acids but do not respond to self-nucleic acids released into the extracellular environment by dying host cells. Structural differences such as the high levels of unmethylated CpG motifs in viral DNA and clusters of U or GU-rich sequences in viral RNA have been considered a key factor in the discrimination between viral and self-nucleic acids (Krieg, 2002; Diebold et al., 2004; Heil et al., 2004). However, more recently, it has become clear that this discrimination is mainly achieved by the intracellular localization of these TLRs, which allows recognition of viral nucleic acids released into endosomal compartments by the endocytosed virus (Barton et al., 2006). In contrast, self-nucleic acids are rapidly degraded in the extracellular environment and fail to access endosomal compartments spontaneously (Barton et al., 2006). Self-DNA and self-RNA can, however, become a potent trigger of pDC activation when they are aberrantly transported into TLR-containing endosomes in the context of autoimmunity. In systemic lupus erythematosus (SLE), self-RNA and self-DNA are complexed with autoantibodies against the nucleic acid or nucleoproteins, which deliver the nucleic acids into endosomal compartments of pDCs via FcγRII-mediated endocytosis (Rönnblom et al., 2003; Barrat et al., 2005; Means et al., 2005). As a result, pDCs are continuously activated to produce type I IFNs, which drives the development of autoimmunity and disease formation. This has been demonstrated in mouse models of SLE using TLR7- or TLR9-deficient mice or inhibitors for TLR7 and TLR9 (Leadbetter et al., 2002; Lau et al., 2005; Christensen et al., 2006; Barrat et al., 2007), and by a study showing that TLR7 gene duplication in mice induces SLE-like disease (Deane et al., 2007).
In psoriasis, a chronic autoimmune-inflammatory disease of the skin, self-DNA forms complexes with the cationic antimicrobial peptide LL37. Self-DNA–LL37 complexes gain access to endosomal TLR9, leading to an aberrant activation of pDCs to produce IFN-α (Lande et al., 2007). pDC-derived IFN-α initiates the autoimmune-inflammatory cascade in psoriasis, a process characterized by the activation of mDCs and their maturation into DCs that stimulate pathogenic autoimmune T cells (Nestle et al., 2005). However, whether LL37 also interacts with extracellular self-RNA, and whether this pathway is involved in the activation of mDCs in psoriasis, is unknown.
Here, we found that LL37 also forms complexes with extracellular self-RNA. These complexes are highly protected from RNase degradation and gain access to the endosomal compartments of both pDCs and mDCs. Self-RNA–LL37 complexes induce TLR7 activation in pDCs and trigger the secretion of IFN-α. Self-RNA–LL37 complexes also trigger the direct activation of mDCs to secrete TNF-α and IL-6 and differentiate into mature DCs. This maturation of mDCs is triggered by endosomal TLR8 and is enhanced by the concomitant activation of pDCs to produce IFN-α. In-vivo, self-RNA–LL37 complexes are specifically found in psoriatic skin and the number of these complexes correlates with the presence of mature mDCs. The current findings identify self-RNA–LL37 complexes as endogenous triggers of TLR7 and TLR8 in human DCs, and provide a new link between the expression of antimicrobial peptides and DC-mediated inflammation in psoriasis.
RESULTS
LL37 converts nonstimulatory self-RNA into a trigger of pDC activation to produce IFN-α
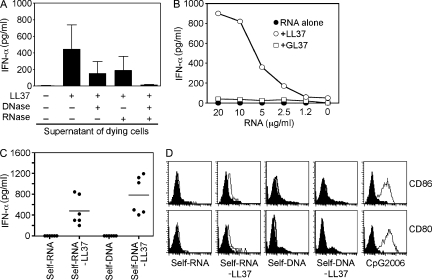
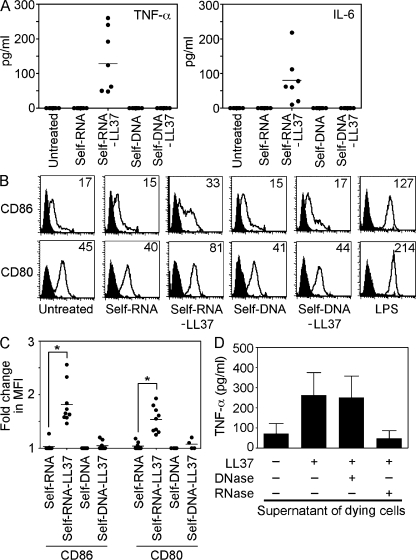
To determine whether the antimicrobial peptide LL37 enables pDCs to sense self-nucleic acids released by dying cells, we stimulated pDCs with supernatants of UV-irradiated U937 cells, which undergo apoptosis and secondary necrosis with the release of self-DNA and self-RNA (Lövgren et al., 2004). The supernatants induced high levels of IFN-α in pDCs when mixed with LL37, but not when given alone (Fig. 1 A). Depletion of DNA from the supernatants substantially inhibited IFN-α induction; however, this inhibition was partial (Fig. 1 A). Depletion of RNA from the supernatants also led to partial inhibition, whereas the combined depletion of RNA and DNA completely abolished IFN-α induction (Fig. 1 A). These results indicate that both self-DNA and self-RNA contributed to pDC activation.
Figure 1.
LL37 converts self-RNA into a trigger of pDCs to produce IFN-α. (A) IFN-α produced by pDCs after stimulation with supernatants from dying (UV-irradiated) U937 cells, either alone or premixed with LL37. In some experiments, DNA and/or RNA was depleted from supernatants of dying cells by pretreatment with DNase I (2,000 U/ml) and/or RNase A (50 µg/ml). Data are the mean ± SEM of four independent experiments. (B) IFN-α produced by pDCs after stimulation with increasing concentrations of total human RNA purified from U937 cells (self-RNA), either alone (closed circles) or premixed with LL37 (self-RNA–LL37; open circles) or the scrambled peptide GL37 (self-RNA+GL37; open squares). (C) IFN-α produced by pDCs after stimulation with self-RNA or self-DNA (both at 5 µg/ml) alone or in complex with LL37 (self-RNA–LL37 and self-DNA–LL37). Each symbol represents an independent experiment; horizontal bars represent the mean. (D) Flow cytometric analysis of stimulated pDCs for CD80 and CD86 surface expression. CpG-2006 (1 µM) was used as a control to induce pDC maturation. Data are representative of at least three independent experiments.
We have previously demonstrated that LL37 can convert self-DNA into a trigger of IFN-α induction in pDCs (Lande et al., 2007). To confirm that LL37 can also interact with self-RNA and convert it into a trigger of IFN-α production, pDCs were stimulated with LL37 premixed with total human RNA extracted from U937 cells. U937-derived self-RNA induced dose-dependent IFN-α production when mixed with LL37, but not when given alone or mixed with the scrambled peptide GL37 (Fig. 1 B). Similar to self-DNA (Lande et al., 2007), pDCs activated by self-RNA mixed with LL37 produced high levels of IFN-α (Fig. 1 C), but did not produce TNF-α or IL-6 (Fig. S1) or undergo maturation as assessed by measuring the expression of costimulatory molecules CD80 and CD86 (Fig. 1 D). Importantly, self-RNA isolated from a variety of cell types and tissue samples from various types of skin pathologies induced similar levels of IFN-α when mixed with LL37, indicating that cellular- or disease-dependent variations in RNA composition do not play a role in the responses to self-RNA (Fig. S2). These data demonstrate that LL37 can convert otherwise nonstimulatory self-RNA into a trigger of pDC activation to produce IFN-α, and thus enable the RNA released during cell death to induce innate immune activation.
LL37 binds self-RNA to form aggregated complexes that are protected from extracellular degradation
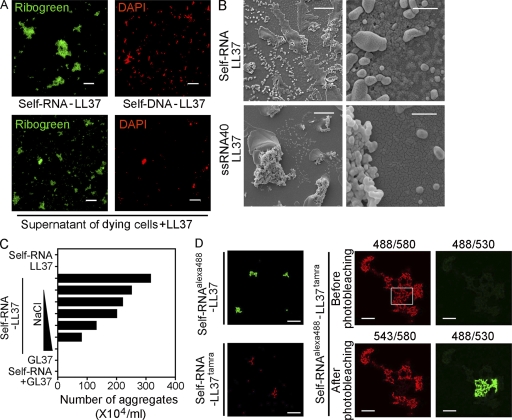
Interestingly, when total human RNA purified from U937 cells was mixed with LL37 (but not GL37), we observed formation of particulate complexes that ranged in size from 1 to 30 µm that precipitated to the bottom of the culture plate. Fluorescence microscopy revealed that the visible complexes contained RNA, as they fluoresced when stained with Ribogreen (which stains RNA preferentially, but also stains DNA) but not with DAPI (which stains DNA exclusively) (Fig. 2 A). At high magnification, the complexes had a characteristic bead-like branched morphology (Fig. S3). Similarly, supernatants of dying U937 cells formed particulate complexes when mixed with LL37 but not with GL37. A large number of these complexes contained RNA given that they stained Ribogreen+/DAPI− (Fig. 2 A) and their formation was inhibited by RNase pretreatment of the supernatants (Fig. S4). Interestingly, visible RNA particles were more abundant than were DNA complexes identified as DAPI+ particles (Fig. 2 A) that failed to form upon DNase pretreatment of the supernatants (Fig. S4). Further analysis of the RNA complexes by electron microscopy revealed that the complexes consisted of aggregates of smaller electron-dense nanoscale particles with sizes ranging from 100 to 700 nm when total human RNA was used, and ranging from 100 to 300 nm when short ssRNA sequences were used (Fig. 2 B). RNA–LL37 complexes were rapidly dissolved by addition of sodium chloride, indicating that ionic interactions between the cationic peptide and the negatively charged phosphate groups of the nucleic acid were involved in the formation of the complexes (Fig. 2 C).
Figure 2.
LL37 binds self-RNA and forms aggregated particles. (A) Confocal microscopy images of self-RNA–LL37 and self-DNA–LL37 complexes generated in vitro (top panels) or formed by mixing supernatant of dying U937 cells with LL37 (bottom panels) and stained with Ribogreen (which stains both RNA and DNA) and DAPI (which stains DNA exclusively). RNA and DNA complexes were detected as Ribogreen+/DAPI− and Ribogreen+/DAPI+ complexes, respectively. Bar, 20 µm. (B) Scanning electron microscopy of self-RNA–LL37 complexes formed by self-RNA fragments (top panels) or the short ssRNA sequence ssRNA40 (bottom panels). Bars: (left panels) 10 µm; (right panels) 1 µm. (C) Number of self-RNA–LL37 complexes counted as visible precipitates by phase-contrast microscopy. Increasing concentrations of NaCl (10, 50, 100, 200, 500, and 1,000 mM) were used to dissolve the complexes. Data are representative of three independent experiments. (D) Confocal microscopy of self-RNAAlexa488–LL37 (top left panel) and self-RNA–LL37Tamra (bottom left panel), or self-RNAAlexa488–LL37Tamra complexes (right panels). Self-RNAAlexa488–LL37Tamra complexes did not appear in the green channel after excitation at 488 nm (top right quadrant) but did appear in the red channel (top left quadrant), suggesting FRET. Photobleaching of the red fluorochrome TAMRA with the 543 laser (bottom left quadrant) resulted in recovery of green fluorescence of the Alexa Fluor 488 fluorochrome in response to excitation at 488 nm (bottom right quadrant). Numbers on the top of the right panels indicate excitation/emission wavelength. Bars: (left panels) 20 µm; (right panels) 10 µm.
A molecular interaction between RNA and LL37 was confirmed by fluorescence resonance energy transfer (FRET) between human RNA labeled with fluorescent dye Alexa 488 (self-RNAAlexa488) and Tamra-labeled LL37 (LL37Tamra). Excitation of self-RNAAlexa488/LL37Tamra complexes at 488 nm did not elicit green fluorescence, but did result in red fluorescence of the complex owing to efficient energy transfer to Tamra. In contrast, after photobleaching of the Tamra fluorophore at 543 nm, excitation at 488 nm could induce green fluorescence of the complexes, with a FRET efficiency of >90%, indicating a close molecular proximity between RNA and LL37 (Fig. 2 D).
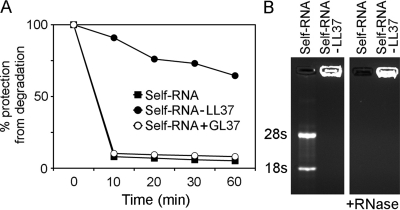
Because free RNA is exquisitely sensitive to degradation by extracellular RNases, which may limit its intracellular delivery, we tested whether LL37 can protect self-RNA from nuclease activity. Self-RNA–LL37 complexes, self-RNA alone, or self-RNA mixed with the control peptide GL37 were treated with RNase A and RNA was quantitated by a fluorometric assay at different time points. Whereas RNA alone and RNA mixed with GL37 were rapidly degraded, self-RNA–LL37 complexes were highly resistant to degradation (Fig. 3 A). The ability of LL37 to protect RNA from RNase degradation was confirmed by agarose gel electrophoresis wherein we found that self-RNA in complex with LL37 was protected from digestion by RNase and was retained in the loading wells (Fig. 3 B). Thus, LL37 forms compacted self-RNA particles that are highly protected from extracellular degradation.
Figure 3.
Self-RNA–LL37 complexes are protected from enzymatic degradation. (A) Self-RNA alone, self-RNA–LL37 complexes, or self-RNA mixed with the control peptide GL37 were treated with RNase A and quantified over 60 min by a fluorometric assay. The percentage of protection was calculated as the ratio of remaining RNA over the initial RNA input. Data are representative of at least three experiments. (B) Agarose gel electrophoresis of self-RNA alone and self-RNA–LL37 complexes before and after RNase A treatment visualized by ethidium bromide staining.
Self-RNA–LL37 complexes trigger endosomal TLR7 in pDC
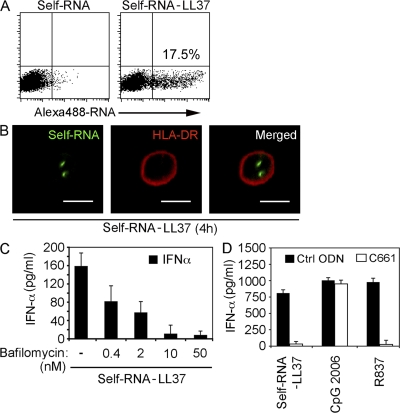
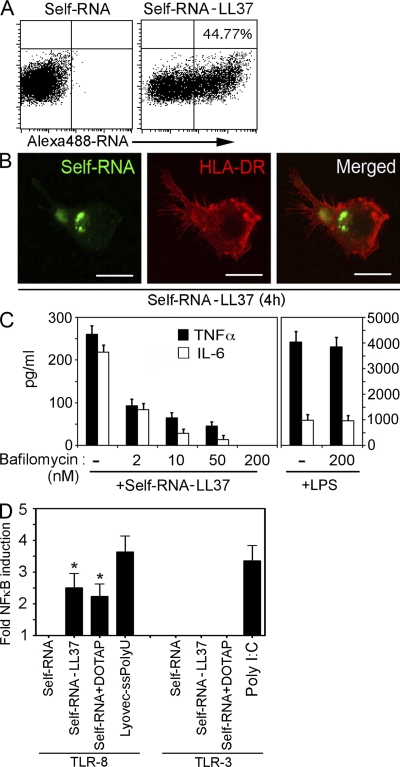
To test whether LL37 can deliver self-RNA particles to TLR-containing endosomes, we stimulated pDCs with self-RNAAlexa488 alone or in complex with LL37. After 4 h of stimulation, self-RNAAlexa488 was found to associate with pDCs when in complex with LL37 but not when given alone (Fig. 4 A). The self-RNA–LL37 complexes were internalized as indicated by confocal microscopy showing the intracellular location of the self-RNAAlexa488 (Fig. 4 B).
Figure 4.
LL37 transports self-RNA into pDCs to trigger endosomal TLR-7. (A) pDCs were stimulated for 4 h with self-RNAAlexa488 alone or self-RNAAlexa488–LL37 and analyzed by flow cytometry for self-RNAAlexa488–containing pDCs. (B) Confocal microscopy of pDCs stimulated for 4 h with self-RNAAlexa488–LL37 complexes and stained with Alexa 647–labeled anti–HLA-DR antibody to visualize the contour of the cell. (C) IFN-α produced by pDCs stimulated with self-RNA–LL37 complexes after pretreatment with increasing concentrations of bafilomycin. (D) IFN-α produced by pDCs after stimulation with self-RNA–LL37, CpG-2006 (1 µM), or R837 (10 µg/ml) after pretreatment with 1 µM TLR7 inhibitor C661 or a control oligonucleotide (ctrl-ODN). Data in A–D are representative of at least three independent experiments; error bars in C and D represent the SD of triplicate wells.
Because pDCs sense RNA through endosomal TLR7 (Diebold et al., 2004; Heil et al., 2004; Lund et al., 2004), we examined whether TLR7 is involved in the recognition of self-RNA–LL37 complexes. IFN-α induced in pDCs by self-RNA–LL37 complexes was inhibited in a dose-dependent-manner by bafilomycin, which blocks endosomal acidification and TLR signaling (Fig. 4 C). To specifically inhibit TLR7, we used the short oligonucleotide C661, which selectively blocks TLR7 (Barrat et al., 2005), as shown by the inhibition of IFN-α induction by the synthetic TLR7 agonist R837 but not the TLR9 agonist CpG2006 (Fig. 4 D). Pretreatment of pDCs with C661 specifically blocked the IFN-α induction by self-RNA–LL37 complexes (Fig. 4 D), indicating that pDC activation by self-RNA–LL37 complexes occurs through TLR7. In support of these data, we also found that short synthetic ssRNA sequences that activate TLR7 in pDCs upon liposomal transfection (Diebold et al., 2004; Heil et al., 2004) were able to trigger IFN-α production in pDCs when complexed with LL37 (Fig. S5).
Self-RNA–LL37 complexes trigger maturation and production of proinflammatory cytokines in mDCs
Human mDCs do not express any TLR that can recognize DNA, but they do express TLRs that recognize RNA (Kadowaki et al., 2001; Jarrossay et al., 2001; Hornung et al., 2002). To test whether mDCs acquire the ability to respond to self-RNA when complexed with LL37, we stimulated immature monocyte-derived mDCs or mDCs isolated directly from human peripheral blood with self-RNA or self-RNA–LL37 complexes. Self-RNA–LL37 complexes but not self-RNA alone activated mDCs to produce the proinflammatory cytokines TNF-α and IL-6 (Fig. 5 A and Fig. S5), but not IFN-α (Fig. S1). Self-RNA–LL37 complexes also activated mDCs to undergo maturation as shown by the up-regulation of CD80 and CD86 expression (Fig. 5, B and C, and Fig. S6). mDC activation by self-RNA–LL37 complexes was entirely dependent on self-RNA, given that these responses were abrogated by decreasing the amount of self-RNA in the complexes (unpublished data). In contrast to self-RNA–LL37 complexes, self-DNA–LL37 complexes were unable to activate mDCs (Fig. 5, A and B). In accordance with these findings, stimulation of mDCs with supernatants of apoptotic cells combined with LL37 induced the secretion of proinflammatory cytokines, and this secretion was entirely dependent on self-RNA because activity was abolished by depletion of self-RNA but not self-DNA (Fig. 5 D).
Figure 5.
Self-RNA but not self-DNA in complex with LL37 activates mDCs to secrete TNF-α and IL-6 and undergo maturation. Immature monocyte-derived mDCs were stimulated with self-RNA, self-DNA, self-RNA–LL37, or self-DNA–LL37. (A) TNF-α and IL-6 secretion was measured after overnight culture. Each symbol represents an independent experiment, and horizontal bars represent the mean. (B) Flow cytometry for surface expression of CD86 and CD80 on stimulated mDCs. LPS was used as a control to induce mDC maturation. (C) Mean fluorescence intensity (MFI) changes for CD86 and CD80 expression on mDCs in response to stimulation with self-RNA–LL37 complexes. Each symbol represents an independent experiment, and horizontal bars represent the mean. *, P < 0.0005; paired Student's t test. (D) TNF-α production by mDCs stimulated with supernatants of dying U937 cells, either alone or premixed with LL37. In some experiments, DNA and/or RNA was depleted from supernatants of dying cells by pretreatment with DNase I and/or RNase A. Data indicate the mean ± SEM of three independent experiments.
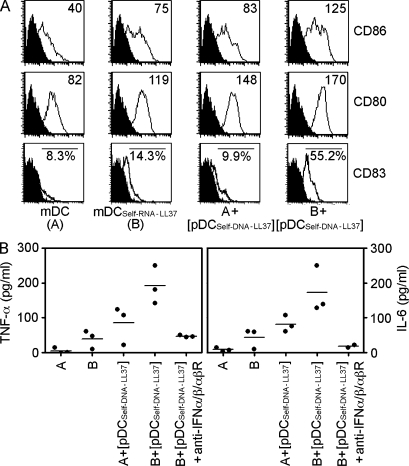
Because type I IFNs have been reported to directly stimulate mDCs (Luft et al., 1998; Santini et al., 2000), we next investigated whether pDC-derived type I IFNs could enhance the activation and maturation of mDCs stimulated by self-RNA–LL37 complexes. Compared with stimulation with either supernatant of activated pDCs or self-RNA–LL37 alone, the combination of both significantly enhanced the activation of mDCs to secrete IL-6 and TNF-α and enhanced their differentiation into mature CD83+ DCs (Fig. 6, A and B). This activity was completely blocked by antibodies against IFN-α, IFN-β and IFN-αβR (Fig. 6 B). Thus, self-RNA–LL37 complexes can trigger mDC activation and maturation, and this process is enhanced by the concomitant activation of pDCs to produce IFN-α.
Figure 6.
IFN-α induced by concomitant pDC activation enhances maturation and cytokine production by mDCs. (A) Flow cytometry for CD86, CD80, and CD83 surface expression. Data are representative of at least three experiments. (B) TNF-α and IL-6 secretion measured by ELISA. Each symbol represents an independent experiment, and horizontal bars represent the mean.
Self-RNA–LL37 complexes enter mDCs and trigger endosomal TLR8
As seen in pDCs, self-RNAAlexa488 was associated with mDCs when complexed with LL37 but not when given alone (Fig. 7 A). The self-RNA–LL37 complexes were internalized (Fig. 7 B). The cytokine production and the maturation of mDCs induced by self-RNA–LL37 complexes but not by the TLR4 agonist LPS was completely inhibited by bafilomycin in a dose-dependent manner (Fig. 7 C), demonstrating that mDC activation by self-RNA–LL37 complexes involved endosomal TLR activation. Because mDCs express endosomal TLR3 and TLR8 that recognize RNA, we investigated whether these TLRs are involved in sensing self-RNA–LL37 complexes. Using 293T cells transfected with TLR8 and TLR3 expression vectors along with a NF-κB luciferase reporter plasmid, we found that self-RNA–LL37 complexes activated TLR8 but not TLR3 (Fig. 7 D). In support of this finding, we also found that synthetic short ssRNA sequences that activate TLR8 in human mDCs (Diebold et al., 2004; Heil et al., 2004) also activated mDCs when complexed with LL37 but not when given alone (Fig. S5).
Figure 7.
LL37 transports self-RNA into mDCs and activates endosomal TLR-8. (A) mDCs were stimulated for 4 h with self-RNAAlexa488 alone or self-RNAAlexa488–LL37 and analyzed by flow cytometry for self-RNAAlexa488–containing mDCs. (B) Confocal microscopy of mDCs stimulated for 4 h with self-RNAAlexa488–LL37 complexes and stained with Alexa 647–labeled anti–HLA-DR antibody to visualize the contour of the cell. Data in A and B are representative of at least five independent experiments. (C) TNF-α and IL-6 secretion by mDCs stimulated with self-RNA–LL37 complexes after pretreatment with increasing concentrations of bafilomycin. Stimulation of the mDCs with LPS was used as negative control. Data are representative of three independent experiments. (D) NF-κB promoter activity of TLR8- or TLR3-transfected HEK293 cells measured by luciferase reporter assay after stimulation at the indicated conditions. Data in D are the mean ± SEM of five independent experiments. *, P < 0.05; paired Student's t test.
Self-RNA complexes are present in psoriatic skin and are associated with mature mDCs
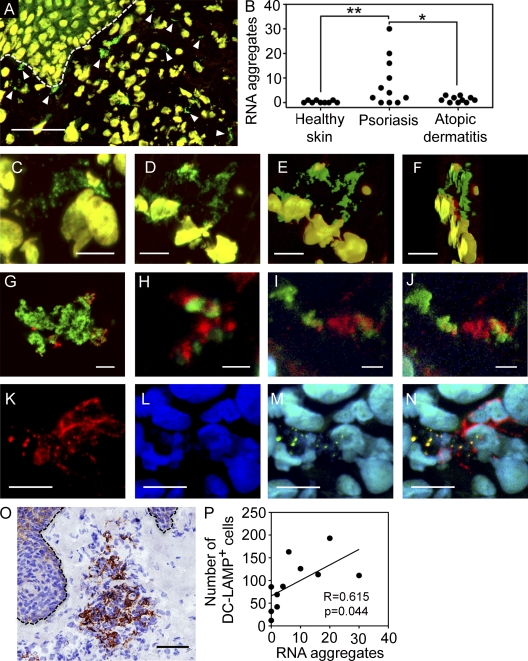
We have previously demonstrated that LL37 is highly expressed in the inflamed skin of psoriasis but is undetectable in inflamed skin of atopic dermatitis or in healthy skin (Lande et al., 2007). To determine whether extracellular self-RNA–LL37 complexes form in vivo, we stained skin sections with Ribogreen and DAPI. We found numerous extracellular Ribogreen+/DAPI− complexes in the dermal compartment of psoriatic skin lesions, but not in skin of atopic dermatitis or healthy skin (Fig. 8, A and B). These tissue RNA complexes presented several features of self-RNA–LL37 complexes generated in vitro, including the size and bead-like branched structures resulting from the aggregation of smaller particles (Fig. 8, C–F).
Figure 8.
Extracellular RNA complexes are present in psoriatic skin and associated with mDC activation. (A) Confocal microscopy image of a representative psoriatic skin lesion stained with DAPI (red) and Ribogreen (green). DNA in cell nuclei appears yellow (Ribogreen+/DAPI+), and arrowheads indicate extracellular RNA aggregates appearing green (Ribogreen+/DAPI−). Bar, 50 µm. (B) Numbers of visible extracellular RNA aggregates in the dermal compartment of healthy skin (n = 9), psoriatic skin (n = 11), and atopic dermatitis (n = 10). **, P = 0.0295; *, P = 0.042; unpaired Student's t test (both two-tailed). (C–F) High magnification images of extracellular RNA aggregates in the dermal compartment of psoriatic skin lesions. Bar, 10 µm. (E and F) SFP projection of panel D. (G-I) Representative confocal microscopy images of in vitro–generated self-RNA–LL37 complexes (G), and tissue self-RNA–LL37 aggregates stained with Ribogreen (green) and an anti-LL37 antibody (red) (H-I). Bars, (G) 10 µm; (H and I) 2 µm. (J) SFP projection of panel I. (K-N) Confocal microscopy images of DC-LAMP+ mature mDCs (red) in the dermal compartment of a representative psoriatic skin lesion co-stained with DAPI (blue) and Ribogreen (green). The images depict DC-LAMP (K), DAPI (L), and the merged image of DAPI and Ribogreen (M). (N) Merged image showing the colocalization of RNA complexes (Ribogreen+/DAPI−; green) with DC-LAMP–positive organelles (red) appearing yellow (N). Bar, 10 µm. (O) Immunohistochemical results for DC-LAMP in a representative psoriatic skin lesion. Bar, 50 µm. (P) Correlation between the numbers of DC-LAMP+ mDCs and the numbers of visible extracellular RNA aggregates in multiple psoriatic skin lesions (n = 11). Pearson R = 0.615, two-tailed P = 0.044. SFP, simulated fluorescence projection.
To determine whether the self-RNA complexes in the tissues contained LL37, we first stained in vitro–generated self-RNA–LL37 complexes with an anti-LL37 antibody and Ribogreen (Fig. 8 G). Interestingly, we found that the antibody was able to stain these complexes but did not colocalize with the RNA stain, suggesting competition between binding of the antibody and staining with Ribogreen. Then, we stained self-RNA complexes in psoriatic tissues and found that the majority of these complexes contained LL37, although the sites of antibody staining and the RNA stain did not colocalize (Fig. 8, H–J), as seen in the case of in vitro–generated self-RNA–LL37 complexes. Importantly, psoriatic skin also contained substantial numbers of particulate self-DNA–LL37 complexes (Fig. S7).
To determine whether the presence of tissue self-RNA complexes is associated with the presence of activated DCs in psoriatic skin, we stained serial sections of lesional psoriatic skin for RNA complexes and DC-LAMP, a lysosomal marker specific for mature mDCs. Consistent with previous reports (Lowes et al., 2005), we found large clusters of DC-LAMP–positive mature mDCs (Fig. 8 O). We also found tissue self-RNA–LL37 complexes within these clusters, and, occasionally, even inside the DCs as shown by the colocalization with endolysosomal compartments stained with DC-LAMP (Fig. 8, K–N). The number of tissue self-RNA complexes significantly correlated with the numbers of DC-LAMP–positive mDCs in psoriatic skin (Fig. 8 P). Together, these findings strongly support our in vitro data that self-RNA complexes can activate mDCs and suggest that this pathway is operational in psoriasis.
DISCUSSION
Dying host cells are a rich source of extracellular self-RNA. Yet, extracellular self-RNA normally does not lead to innate immune activation because it is rapidly degraded by RNases and fails to access endosomal compartments of DCs where TLR7 and TLR8 are located. Here, we found that extracellular self-RNA acquires the ability to trigger activation of TLR7 and TLR8 in human DCs by forming a complex with the endogenous antimicrobial peptide LL37. Previously, we had demonstrated that LL37 can form a complex with self-DNA, leading to TLR9 activation of pDCs to produce IFN-α (Lande et al., 2007). The present study shows that LL37 and the formation of RNA complexes also allow activation of classical mDCs, leading to the production of TNF-α and IL-6 and differentiation into mature DCs with T cell stimulatory activity.
The formation of complexes between LL37 and RNA is driven by ionic interactions between the cationic residues of the peptide and the anionic phosphate backbone of RNA, similar to the phenomenon described for DNA (Lande et al., 2007). In self-DNA–LL37 complexes, the α-helical structure of LL37 appears to stabilize the interactions with the double-stranded DNA (Lande et al., 2007). Because vertebrate RNA is single stranded, it is unclear whether the α-helical structure of LL37 also plays a role in the formation and stabilization of complexes with RNA. It may be possible that LL37 preferentially binds to regions where RNA folds into complex structures containing double-helical regions capped by hairpin or internal loops.
The mechanism by which LL37 converts nonstimulatory self-RNA into a trigger of TLR7 and TLR8 in DC involves two distinct steps. First, the binding of LL37 condenses free RNA into compacted structures that are highly protected from enzymatic degradation, a phenomenon that is critical for subsequent internalization of the complex. Second, by forming these complexes, LL37 allows the association of RNA with cellular membranes and the transport across these membranes into endocytic compartments. A previous study showed that DNA–LL37 complexes acquire net positive charges that allow binding to anionic proteoglycans in the membrane of CHO cells followed by lipid raft–mediated endocytosis (Sandgren et al., 2004). Similarly, RNA–LL37 complexes interact with heparan-sulfate–containing proteoglycans in the membranes of DCs, as indicated by the finding that heparinase pretreatment significantly reduced the uptake of the RNA–LL37 complexes into both pDCs and mDCs (unpublished data).
Internalized self-RNA triggers the activation of TLR7 and TLR8, which recognize uridine moieties common to both viral RNA and self-RNA (Diebold et al., 2006). Compared with viral RNA, self-RNA is highly methylated and contains high numbers of pseudouridines, which have been shown to lack the ability to activate and rather inhibit TLR7 and TLR8 activation (Karikó et al., 2005; Robbins et al., 2007). Our finding that total cellular RNA isolated from cells or released by dying cells can trigger TLR7 and TLR8 activation indicates that non-modified sequences in mammalian RNA are sufficient to induce immune activation if RNA is protected from extracellular degradation and transported into the endosomal compartments. Accordingly, self-RNA has been previously found to trigger TLR7 activation if encapsulated into liposomes and transfected into DCs (Diebold et al., 2006).
Our study identifies self-RNA–LL37 complexes as small extracellular particles in inflamed skin of psoriatic patients, demonstrating that these complexes can form in vivo in the context of immunopathology. These tissue RNA particles were associated with the presence of mature mDCs, providing evidence that self-RNA–LL37 particles are linked to mDC activation in vivo. Psoriasis is characterized by an early skin infiltration of pDC and their aberrant activation to produce IFN-α (Nestle et al., 2005). During later stages of the disease, however, psoriatic lesions are mainly characterized by large numbers of mature mDCs, which stimulate pathogenic T cells, whereas pDCs are absent (Albanesi et al., 2009). Based on our findings, we propose that self-RNA–LL37 complexes may trigger a concerted activation of pDCs and mDCs, which initiate the inflammatory process during early psoriasis. During later stages of the disease, inflammation may be exclusively maintained by mDCs that are activated by self-RNA–LL37 complexes.
LL37 expression is typically induced in epithelial cells or released by neutrophils in the context of tissue injury or infection. Although the mechanisms controlling LL37 expression are still poorly understood, recent studies revealed the key involvement of the vitamin D3 and hypoxia response pathways, as well as the effect of various microorganisms on the production of LL37 (Liu et al., 2006; Schauber et al., 2007; Peyssonnaux et al., 2008). In psoriasis, LL37 appears to be continuously overexpressed by lesional keratinocytes, leading to sustained formation of nucleic acid complexes that trigger TLR7, 8, and 9 in DCs. As a result, the uncontrolled activation of pDCs and mDCs may induce high levels of self-antigen–specific T cells and the development of overt autoimmunity. In contrast, LL37 expression in common skin wounds is short lived and may drive transient DC activation and inflammatory responses that participate in wound repair. It is possible that the transient LL37 expression and DC activation in skin wounds may induce low levels of self-antigen–specific T cells; however, they are not sufficient to drive overt autoimmunity. Indeed, a recent report has shown that T cells are activated during tissue damage and participate in wound repair (Toulon et al., 2009). It is also possible that the short-lived DC activation allows only activation of T cells that recognize high avidity antigens, such as those derived from pathogens invading the wounded tissue.
Persistent high levels of LL37 have also been reported in other chronic inflammatory diseases such as rosacea (Yamasaki et al., 2007), rheumatoid arthritis (Paulsen et al., 2002), colitis ulcerosa (Schauber et al., 2006), chronic nasal inflammatory disease (Kim et al., 2003), sarcoidosis (Agerberth et al., 1999), and cystic fibrosis (Bucki et al., 2007). Self-RNA–LL37 and self-DNA–LL37 complexes may also sustain the pathogenic inflammatory responses through TLR-mediated activation of DCs in these diseases, as we found in psoriasis. Thus, strategies to inhibit the expression of LL37 or the formation of LL37–nucleic acid complexes should be explored for the treatment of these diseases.
MATERIALS AND METHODS
Collection of human samples.
Studies were approved by the Institutional Review Board for human research at the University of Texas M.D. Anderson Cancer Center in Houston and the Heinrich-Heine University in Düsseldorf. Skin specimens were obtained by 6-mm punch biopsies from normal skin of healthy subjects and from lesional skin of patients with chronic plaque psoriasis and atopic dermatitis, defined according to standard clinical and histopathological criteria. Cryosections were prepared from the biopsy specimens and total RNA from homogenized skin specimen was extracted using standard protocols.
Reagents.
The synthetic peptide LL37 (LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES) and the corresponding scrambled control peptide GL37 (GLKLRFESKIKGEFLKTPEVRFRDIKLKDNRISVQR) were purchased from Innovagen. LyoVec-free ssRNA sequences ssRNA40 (5′-GsCsCsCsGsUsCsUsGsUsUsGsUsGsUsGsAsCsUsC-3′ [“s” denotes phosphothioate linkage]) and ssPolyU (single-stranded poly-uridine) were from InvivoGen and used at a final concentration of 5 µg/ml. The TLR9 agonist CpG-2006 (5′-tcgtcgttttgtcgttttgtcgtt-3′) was produced by Trilink and used at 1 μM. Synthetic TLR7 agonist R837 (10 µM) and TLR8 agonist R848 (100 ng/ml) were both from InvivoGen. The specific human TLR7 inhibitor C661 and the control oligonucleotide sequence (ctrl-ODN, 5′-tcctgcaggttaagt-3′) were from Dynavax and used at 1 µM to pretreat pDCs for 30 min at 37°C before stimulation. Bafilomycin was obtained from Sigma-Aldrich.
Isolation and generation of pDCs and mDCs.
pDCs were isolated from buffy coats from healthy volunteers with anti–BDCA-4 microbeads (Miltenyi Biotec) and were sorted by FACS Aria (BD Biosciences) according to their lineage-negative, CD11c− CD4+ phenotype. Immature mDCs were generated from blood monocytes obtained from peripheral blood mononuclear cells (PBMCs) using anti-CD14 microbeads (Miltenyi Biotec) and cultured for 5 d with GM-CSF and IL-4 (both from R&D Systems). Blood mDCs were isolated from PBMCs using anti-CD1c microbeads (Miltenyi Biotec).
In vitro generation and characterization of RNA–LL37 and DNA–LL37 complexes.
Total human RNA (self-RNA) and genomic DNA (self-DNA) were extracted from cell lines or inflammatory skin samples using standard procedures. Skin samples used were 6-mm punch biopsies previously described (Lande et al., 2007). For the generation of complexes, 10 µg of LL37 was mixed with 1 µg of self-RNA or self-DNA in 20 µl of PBS. Culture supernatant of necrotic cells was generated by an overnight culture of UV-irradiated (480 mJ/cm2 for 30 min) U937 cells (50 × 106/ml). For the generation of complexes, 100 µl of these supernatants was mixed with 10 µg of LL37. RNA and DNA complexes were visualized by confocal microscopy after staining with Ribogreen (1:100,000; Invitrogen) and DAPI (0.1 ng/ml; Sigma-Aldrich). In some experiments the supernatants were treated with DNase I (2,000 U/ml), RNase A (50 µg/ml), or both.
To detect LL37, suspensions containing precipitating complexes were stained with 10 µg/ml mouse anti-LL37 antibody (clone 3d11; HyCult Biotechnology) for 2 h, followed by careful washes and incubation with Alexa 647–labeled anti–mouse secondary antibody for 1 h. After further washes, Ribogreen was added and the particulate complexes were visualized by confocal microscopy.
For FRET analysis, complexes were generated using self-RNA labeled with Alexa 488 (self-RNAAlexa488) using the Ulysis Nucleic Acid Labeling kit (Invitrogen) and Tamra-labeled LL37 (LL37Tamra; Innovagen). For scanning electron microscopy of the complexes, suspensions were air-dried overnight on poly-l-lysine–coated coverslips and then mounted onto double-stick carbon tabs (Ted Pella, Inc.) that had been previously mounted on to aluminum specimen mounts (Electron Microscopy Sciences). The samples were then coated under vacuum using an evaporator (Balzer MED 010; Technotrade International) with platinum alloy to a thickness of 25 nm followed by flash carbon coating. Samples were examined in a scanning electron microscope (model JSM-5910; JEOL USA, Inc.) at an accelerating voltage of 5 kV.
Stimulation and analysis of pDCs and mDCs.
Suspensions (20 µl) containing RNA–LL37 and DNA–LL37 complexes, or 100-µl supernatants of dying cells were added to pDC and mDC cultures in a final volume of 200 µl. After overnight culture, supernatants of pDCs and mDCs were collected and IFN-α (PBL Biomedical Laboratories), IL-6, and TNF-α (both from R&D Systems) were measured by ELISA. pDCs and mDCs were also stained with fluorochrome-labeled anti-CD80, anti-CD86, and anti-CD83 antibodies (BD Biosciences) and analyzed by flow cytometry. mDCs were also cultured with supernatant of pDCs stimulated for 24 h with self-DNA–LL37. In some cases, pDC supernatants were treated with a combination of anti–human IFN-α (104 U/ml) antibodies, anti–human IFN-β (2 × 103 U/ml) antibodies, and anti–human IFN-αβ receptor antibodies (clone: MMHAR-2; 10 µg/ml) to block type I IFN activity (all from PBL Biomedical Laboratories).
Nuclease protection assays.
The suspension containing RNA–LL37 complexes were incubated with 50 µg/ml RNase A at 37°C. Then RNA in the suspensions was stained with Ribogreen (Invitrogen) and quantitated by fluorimetry at different time points. In other experiments the suspensions, either RNase treated or left untreated, were run on a 1% agarose gel (with ethidium bromide) and imaged in a UV transilluminator gel imager (Bio-Rad Laboratories).
Uptake of self-RNA–LL37 complexes in pDCs and mDCs.
pDCs or mDCs were stimulated with self-RNAAlexa488 alone or with self-RNAAlexa488–LL37 complexes. After 4 h, cells were analyzed by flow cytometry. For confocal microscopy, pDCs and mDCs were stimulated with self-RNAAlexa488–LL37Tamra complexes. After 4 h of stimulation, cells were stained with Alexa 647-conjugated anti–HLA-DR (BioLegend) and the slides were mounted in Prolong Gold Antifade mounting medium (Invitrogen).
Luciferase reporter assay.
HEK293 cells were transiently transfected with 500 ng of TLR3-GFP or TLR8-HA expression vectors (both from Invivogen) and 100 ng of NF-κB luciferase reporter plasmid. The Renilla luciferase reporter gene (pRL-TK) was simultaneously transfected (5 ng) as an internal control for transfection efficiency. 24 h later, cells were collected and stimulated as indicated for 24 h. Poly I:C and ssPolyU complexed with cationic lipids (DOTAP; Roche) were used as positive controls for the activation of TLR3 and TLR8, respectively. Luciferase activity was measured with a dual-luciferase reporter assay system (Promega).
Tissue immunofluorescence and immunochemistry.
Immunofluorescence of DNA and RNA complexes was done on frozen sections of skin specimens after a 5-min stain with DAPI (0.1 ng/ml) and Ribogreen (1:100,000) followed by washes and mounting with the Prolong Gold mounting medium (Invitrogen). In some cases, sections were first incubated with mouse anti-LL37 (clone 3d11; HyCult Biotechnology) or isotype control overnight, followed by incubation with Alexa 647–conjugated anti–mouse IgG secondary antibody. Ribogreen and DAPI were added after the antibody incubations to achieve the best staining efficiency. The images were acquired using a confocal microscope (model TCS SP2; Leica). Immunohistochemistry for mature mDCs was performed on frozen sections of human psoriasis skin specimens using mouse anti–DC-LAMP antibodies (clone 104.G4; Coulter Immunotech). Images were acquired using a microscope (model DP70; Olympus). Extracellular RNA complexes and DC-LAMP+ DCs in tissue sections were counted in three best fields per section by two independent sample-blind investigators.
Online supplemental material.
Fig. S1 shows the differential pattern of cytokine production by pDCs and mDCs in response to self-RNA–LL37 complexes. Fig. S2 shows that self-RNA isolated from different cell lines or from tissues of different chronic inflammatory skin diseases induces comparable levels of IFN-α production in pDCs when combined with LL37. Fig. S3 shows characteristic microscopic morphology of self-RNA–LL37 complexes. Fig. S4 shows the quantitation of self-RNA–LL37 and self-DNA–LL37 complexes in supernatants of dying cells. Fig. S5 shows that pDCs and mDCs respond to RNA sequences that are bona fide TLR7 and TLR8 ligands when complexed with LL37 but not when given alone. Fig. S6 shows that self-RNA–LL37 complexes trigger the maturation and secretion of TNF-α and IL-6 by circulating CD1c+ blood DCs. Fig. S7 shows DNA–LL37 complexes in psoriatic skin. Online supplemental material is available at http://www.jem.org/cgi/content/full/jem.20090480/DC1.
Acknowledgments
We thank Kenneth Dunner for scanning electron microscopy at the HREM Facility, M.D. Anderson Cancer Center, and Yi-Hong Wang and Pamela Grant for preparation of cryosections and immunohistochemistry at the Histology core facility, M.D. Anderson. We also thank Yong-Jun Liu and Vassili Soumelis for critical reading of the manuscript and Roland L. Bassett for help with statistical analysis.
This work was supported by grants from the DANA Foundation and the National Institutes of Health to M. Gilliet, and grants from the Deutsche Forschungsgemeinschaft to B. Homey and to S. Meller.
The authors have no conflicting financial interests in this work.
Footnotes
Abbreviations used: AMP, antimicrobial peptide; FRET, fluorescence resonance energy transfer; mDC, myeloid dendritic cell; pDC, plasmacytoid dendritic cell; TLR, Toll-like receptor.
References
- Agerberth B., Grunewald J., Castaños-Velez E., Olsson B., Jörnvall H., Wigzell H., Eklund A., Gudmundsson G.H. 1999. Antibacterial components in bronchoalveolar lavage fluid from healthy individuals and sarcoidosis patients.Am. J. Respir. Crit. Care Med. 160:283–290 [DOI] [PubMed] [Google Scholar]
- Akira S., Uematsu S., Takeuchi O. 2006. Pathogen recognition and innate immunity.Cell. 124:783–801 [DOI] [PubMed] [Google Scholar]
- Albanesi C., Scarponi C., Pallotta S., Daniele R., Bosisio D., Madonna S., Fortugno P., Gonzalvo-Feo S., Franssen J.D., Parmentier M., et al. 2009. Chemerin expression marks early psoriatic skin lesions and correlates with plasmacytoid dendritic cell recruitment.J. Exp. Med. 206:249–258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexopoulou L., Holt A.C., Medzhitov R., Flavell R.A. 2001. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3.Nature. 413:732–738 [DOI] [PubMed] [Google Scholar]
- Barrat F.J., Meeker T., Gregorio J., Chan J.H., Uematsu S., Akira S., Chang B., Duramad O., Coffman R.L. 2005. Nucleic acids of mammalian origin can act as endogenous ligands for Toll-like receptors and may promote systemic lupus erythematosus.J. Exp. Med. 202:1131–1139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrat F.J., Meeker T., Chan J.H., Guiducci C., Coffman R.L. 2007. Treatment of lupus-prone mice with a dual inhibitor of TLR7 and TLR9 leads to reduction of autoantibody production and amelioration of disease symptoms.Eur. J. Immunol. 37:3582–3586 [DOI] [PubMed] [Google Scholar]
- Barton G.M., Kagan J.C., Medzhitov R. 2006. Intracellular localization of Toll-like receptor 9 prevents recognition of self DNA but facilitates access to viral DNA.Nat. Immunol. 7:49–56 [DOI] [PubMed] [Google Scholar]
- Bucki R., Byfield F.J., Janmey P.A. 2007. Release of the antimicrobial peptide LL-37 from DNA/F-actin bundles in cystic fibrosis sputum.Eur. Respir. J. 29:624–632 (see comment) [DOI] [PubMed] [Google Scholar]
- Christensen S.R., Shupe J., Nickerson K., Kashgarian M., Flavell R.A., Shlomchik M.J. 2006. Toll-like receptor 7 and TLR9 dictate autoantibody specificity and have opposing inflammatory and regulatory roles in a murine model of lupus.Immunity. 25:417–428 [DOI] [PubMed] [Google Scholar]
- Colonna M., Trinchieri G., Liu Y.J. 2004. Plasmacytoid dendritic cells in immunity.Nat. Immunol. 5:1219–1226 [DOI] [PubMed] [Google Scholar]
- Deane J.A., Pisitkun P., Barrett R.S., Feigenbaum L., Town T., Ward J.M., Flavell R.A., Bolland S. 2007. Control of toll-like receptor 7 expression is essential to restrict autoimmunity and dendritic cell proliferation.Immunity. 27:801–810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diebold S.S., Kaisho T., Hemmi H., Akira S., Reis e Sousa C. 2004. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA.Science. 303:1529–1531 [DOI] [PubMed] [Google Scholar]
- Diebold S.S., Massacrier C., Akira S., Paturel C., Morel Y., Reis e Sousa C. 2006. Nucleic acid agonists for Toll-like receptor 7 are defined by the presence of uridine ribonucleotides.Eur. J. Immunol. 36:3256–3267 [DOI] [PubMed] [Google Scholar]
- Gilliet M., Cao W., Liu Y.J. 2008. Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases.Nat. Rev. Immunol. 8:594–606 [DOI] [PubMed] [Google Scholar]
- Haas T., Metzger J., Schmitz F., Heit A., Müller T., Latz E., Wagner H. 2008. The DNA sugar backbone 2′ deoxyribose determines toll-like receptor 9 activation.Immunity. 28:315–323 (see comment) [DOI] [PubMed] [Google Scholar]
- Heil F., Hemmi H., Hochrein H., Ampenberger F., Kirschning C., Akira S., Lipford G., Wagner H., Bauer S. 2004. Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8.Science. 303:1526–1529 (see comment) [DOI] [PubMed] [Google Scholar]
- Hemmi H., Takeuchi O., Kawai T., Kaisho T., Sato S., Sanjo H., Matsumoto M., Hoshino K., Wagner H., Takeda K., Akira S. 2000. A Toll-like receptor recognizes bacterial DNA.Nature. 408:740–745 [DOI] [PubMed] [Google Scholar]
- Hornung V., Rothenfusser S., Britsch S., Krug A., Jahrsdörfer B., Giese T., Endres S., Hartmann G. 2002. Quantitative expression of toll-like receptor 1-10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides.J. Immunol. 168:4531–4537 [DOI] [PubMed] [Google Scholar]
- Jarrossay D., Napolitani G., Colonna M., Sallusto F., Lanzavecchia A. 2001. Specialization and complementarity in microbial molecule recognition by human myeloid and plasmacytoid dendritic cells.Eur. J. Immunol. 31:3388–3393 [DOI] [PubMed] [Google Scholar]
- Kadowaki N., Ho S., Antonenko S., Malefyt R.W., Kastelein R.A., Bazan F., Liu Y.J. 2001. Subsets of human dendritic cell precursors express different toll-like receptors and respond to different microbial antigens.J. Exp. Med. 194:863–869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karikó K., Buckstein M., Ni H., Weissman D. 2005. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA.Immunity. 23:165–175 (see comment) [DOI] [PubMed] [Google Scholar]
- Kim S.T., Cha H.E., Kim D.Y., Han G.C., Chung Y.S., Lee Y.J., Hwang Y.J., Lee H.M. 2003. Antimicrobial peptide LL-37 is upregulated in chronic nasal inflammatory disease.Acta Otolaryngol. 123:81–85 [DOI] [PubMed] [Google Scholar]
- Krieg A.M. 2002. CpG motifs in bacterial DNA and their immune effects.Annu. Rev. Immunol. 20:709–760 [DOI] [PubMed] [Google Scholar]
- Lande R., Gregorio J., Facchinetti V., Chatterjee B., Wang Y.H., Homey B., Cao W., Wang Y.H., Su B., Nestle F.O., et al. 2007. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide.Nature. 449:564–569 (see comment) [DOI] [PubMed] [Google Scholar]
- Lau C.M., Broughton C., Tabor A.S., Akira S., Flavell R.A., Mamula M.J., Christensen S.R., Shlomchik M.J., Viglianti G.A., Rifkin I.R., Marshak-Rothstein A. 2005. RNA-associated autoantigens activate B cells by combined B cell antigen receptor/Toll-like receptor 7 engagement.J. Exp. Med. 202:1171–1177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leadbetter E.A., Rifkin I.R., Hohlbaum A.M., Beaudette B.C., Shlomchik M.J., Marshak-Rothstein A. 2002. Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors.Nature. 416:603–607 (see comment) [DOI] [PubMed] [Google Scholar]
- Liu P.T., Stenger S., Li H., Wenzel L., Tan B.H., Krutzik S.R., Ochoa M.T., Schauber J., Wu K., Meinken C., et al. 2006. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response.Science. 311:1770–1773 (see comment) [DOI] [PubMed] [Google Scholar]
- Lövgren T., Eloranta M.L., Båve U., Alm G.V., Rönnblom L. 2004. Induction of interferon-alpha production in plasmacytoid dendritic cells by immune complexes containing nucleic acid released by necrotic or late apoptotic cells and lupus IgG.Arthritis Rheum. 50:1861–1872 [DOI] [PubMed] [Google Scholar]
- Lowes M.A., Chamian F., Abello M.V., Fuentes-Duculan J., Lin S.L., Nussbaum R., Novitskaya I., Carbonaro H., Cardinale I., Kikuchi T., et al. 2005. Increase in TNF-alpha and inducible nitric oxide synthase-expressing dendritic cells in psoriasis and reduction with efalizumab (anti-CD11a).Proc. Natl. Acad. Sci. USA. 102:19057–19062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luft T., Pang K.C., Thomas E., Hertzog P., Hart D.N., Trapani J., Cebon J. 1998. Type I IFNs enhance the terminal differentiation of dendritic cells.J. Immunol. 161:1947–1953 [PubMed] [Google Scholar]
- Lund J.M., Alexopoulou L., Sato A., Karow M., Adams N.C., Gale N.W., Iwasaki A., Flavell R.A. 2004. Recognition of single-stranded RNA viruses by Toll-like receptor 7.Proc. Natl. Acad. Sci. USA. 101:5598–5603 (see comment) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Means T.K., Latz E., Hayashi F., Murali M.R., Golenbock D.T., Luster A.D. 2005. Human lupus autoantibody-DNA complexes activate DCs through cooperation of CD32 and TLR9.J. Clin. Invest. 115:407–417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nestle F.O., Conrad C., Tun-Kyi A., Homey B., Gombert M., Boyman O., Burg G., Liu Y.J., Gilliet M. 2005. Plasmacytoid predendritic cells initiate psoriasis through interferon-alpha production.J. Exp. Med. 202:135–143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulsen F., Pufe T., Conradi L., Varoga D., Tsokos M., Papendieck J., Petersen W. 2002. Antimicrobial peptides are expressed and produced in healthy and inflamed human synovial membranes.J. Pathol. 198:369–377 [DOI] [PubMed] [Google Scholar]
- Peyssonnaux C., Boutin A.T., Zinkernagel A.S., Datta V., Nizet V., Johnson R.S. 2008. Critical role of HIF-1alpha in keratinocyte defense against bacterial infection.J. Invest. Dermatol. 128:1964–1968 [DOI] [PubMed] [Google Scholar]
- Robbins M., Judge A., Liang L., McClintock K., Yaworski E., MacLachlan I. 2007. 2′-O-methyl-modified RNAs act as TLR7 antagonists.Mol. Ther. 15:1663–1669 [DOI] [PubMed] [Google Scholar]
- Rönnblom L., Eloranta M.L., Alm G.V. 2003. Role of natural interferon-alpha producing cells (plasmacytoid dendritic cells) in autoimmunity.Autoimmunity. 36:463–472 [DOI] [PubMed] [Google Scholar]
- Sandgren S., Wittrup A., Cheng F., Jönsson M., Eklund E., Busch S., Belting M. 2004. The human antimicrobial peptide LL-37 transfers extracellular DNA plasmid to the nuclear compartment of mammalian cells via lipid rafts and proteoglycan-dependent endocytosis.J. Biol. Chem. 279:17951–17956 [DOI] [PubMed] [Google Scholar]
- Santini S.M., Lapenta C., Logozzi M., Parlato S., Spada M., Di Pucchio T., Belardelli F. 2000. Type I interferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice.J. Exp. Med. 191:1777–1788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schauber J., Rieger D., Weiler F., Wehkamp J., Eck M., Fellermann K., Scheppach W., Gallo R.L., Stange E.F. 2006. Heterogeneous expression of human cathelicidin hCAP18/LL-37 in inflammatory bowel diseases.Eur. J. Gastroenterol. Hepatol. 18:615–621 [DOI] [PubMed] [Google Scholar]
- Schauber J., Dorschner R.A., Coda A.B., Büchau A.S., Liu P.T., Kiken D., Helfrich Y.R., Kang S., Elalieh H.Z., Steinmeyer A., et al. 2007. Injury enhances TLR2 function and antimicrobial peptide expression through a vitamin D-dependent mechanism.J. Clin. Invest. 117:803–811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toulon A., Breton L., Taylor K.R., Tenenhaus M., Bhavsar D., Lanigan C., Rudolph R., Jameson J., Havran W.L. 2009. A role for human skin-resident T cells in wound healing.J. Exp. Med. 206:743–750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamasaki K., Di Nardo A., Bardan A., Murakami M., Ohtake T., Coda A., Dorschner R.A., Bonnart C., Descargues P., Hovnanian A., et al. 2007. Increased serine protease activity and cathelicidin promotes skin inflammation in rosacea.Nat. Med. 13:975–980 (see comment) [DOI] [PubMed] [Google Scholar]