Abstract
Study Objectives:
The goal of the study was to seek physiological correlates of lucid dreaming. Lucid dreaming is a dissociated state with aspects of waking and dreaming combined in a way so as to suggest a specific alteration in brain physiology for which we now present preliminary but intriguing evidence. We show that the unusual combination of hallucinatory dream activity and wake-like reflective awareness and agentive control experienced in lucid dreams is paralleled by significant changes in electrophysiology.
Design:
19-channel EEG was recorded on up to 5 nights for each participant. Lucid episodes occurred as a result of pre-sleep autosuggestion.
Setting:
Sleep laboratory of the Neurological Clinic, Frankfurt University.
Participants:
Six student volunteers who had been trained to become lucid and to signal lucidity through a pattern of horizontal eye movements.
Measurements and Results:
Results show lucid dreaming to have REM-like power in frequency bands δ and θ, and higher-than-REM activity in the γ band, the between-states-difference peaking around 40 Hz. Power in the 40 Hz band is strongest in the frontal and frontolateral region. Overall coherence levels are similar in waking and lucid dreaming and significantly higher than in REM sleep, throughout the entire frequency spectrum analyzed. Regarding specific frequency bands, waking is characterized by high coherence in α, and lucid dreaming by increased δ and θ band coherence. In lucid dreaming, coherence is largest in frontolateral and frontal areas.
Conclusions:
Our data show that lucid dreaming constitutes a hybrid state of consciousness with definable and measurable differences from waking and from REM sleep, particularly in frontal areas.
Citation:
Voss U; Holzmann R; Tuin I; Hobson A. Lucid dreaming: a state of consciousness with features of both waking and non-lucid dreaming. SLEEP 2009;32(9):1191-1200.
Keywords: Lucid dreaming, consciousness, REM sleep, coherence, EEG
THE GOAL OF THE STUDY WAS TO SEEK ELECTROPHYSIOLOGICAL CORRELATES OF LUCID DREAMING. OUR WORKING HYPOTHESIS WAS THAT THE BRAIN must change state if the mind changes state.
Lucid dreaming is the experience of achieving conscious awareness of dreaming while still asleep. Lucid dreams are generally thought to arise from non-lucid dreams in REM sleep.1
An obstacle to experimental studies of lucid dreams is that spontaneous lucidity is quite rare. However, subjects can be trained to become lucid via pre-sleep autosuggestion.1–5 Subjects often succeed in becoming lucid when they tell themselves, before going to sleep, to recognize that they are dreaming by noticing the bizarre events of the dream. An experimental advantage is that subjects can signal that they have become lucid by making a sequence of voluntary eye movements. In combination with retrospective reports confirming that lucidity was attained and that the eye movement signals were executed, these voluntary eye movements can be used as behavioral indication of lucidity in the sleeping, dreaming subject, as evidenced by EEG and EMG tracings of sleep. Such signal-verified lucid dreams, in which dreamers not only realize that they are currently dreaming, but are also able to deliberately control their own behavior, enabling them to signal lucidity by making prearranged patterns of eye movements, constitute lucid control dreams. The current study, thus, targets lucid control dreams.
Because lucidity can be self-induced, it constitutes not only an opportunity to study the brain basis of conscious states but also demonstrates how a voluntary intervention can change those states.
METHODS
A group of 20 undergraduate students of psychology at Bonn University took part in weekly lucidity training sessions. After 4 months, 6 subjects had claimed to be lucid more than 3 times per week. These 6 were invited to the sleep laboratory of the Frankfurt University. They gave written consent to participate and received 50 Euro per night as compensation. Participants reported to the sleep laboratory 2 hours prior to their usual bedtime. As subjects confirmed literature reports stating that lucid dreams commonly occur after several hours of sleep, during later REM periods,1,3 they were allowed to sleep in. Recordings were made on weekends, i.e. Fridays to Mondays.
Scalp EEG electrodes were placed at 19 positions (10-20 system). Electrodes were referenced to linked mastoids (bandpass filter 0.3–100 Hz, sampling rate 200 Hz). EOG was taken from the outer canthi of both eyes and supraorbitally to the left eye. Submental EMG electrodes were fixed at the chin. EEG was recorded for 2–5 nights per subject.
Data Analysis
Statistical analysis was restricted to artifact-free, continuous segments of state “wake with eyes closed” (WEC), “lucid dreaming” (lucid), and “non-lucid REM sleep” (REM) of at least 70 s duration. Epochs analyzed were controlled for time of night. All data were corrected for ocular artifacts using the Gratton et al. algorithm6 before statistical analysis. If, following this correction procedure, eye movements were still detectable upon visual inspection, these epochs were excluded from further analysis. A bandpass filter was applied (0.5–70 Hz) and a notch filter set at 50 Hz. The filtered signal was baseline corrected (range for mean value calculation = 0-4 s) and subjected to an automatic artifact rejection procedure (maximum allowed voltage step = 50 μV, maximum and minimum amplitude = ± 200 μV, maximum allowed absolute difference of values in the segment = 200 μV, lowest allowed activity = 0.5 μV).
Waking and REM sleep EEG was scored visually according to Rechtschaffen and Kales.7 Power and coherence analyses were performed using Brain Vision Analyzer software (Version 2.0, Schwarzer, Munich, Germany) and results were graphically rendered using TEMPO (http://code.google.com/p/tempo/) and ROOT (http://root.cern.ch).
Power Analysis
Power analyses based on the Fast Fourier Transform (FFT, Hanning windowing) inform about state-specific variations in activity within a given frequency band of the EEG. For data analyses, EEG records were partitioned into 4s epochs with 2s overlap. Statistical analyses were performed for standardized FFTs, to facilitate between-subject comparisons. Standardization was achieved through normalization of power over the 0.5–70 Hz range, yielding the relative distribution of activity on the individual spectral lines. Hence, for each epoch of the EEG, the sum of power values from all frequency bins equals 100%. Mean standardized power values were analyzed in the following frequency bands: δ (1–4 Hz), θ (4–8 Hz), α (8–12 Hz), β1 (12-16 Hz), β2 (16–20 Hz), γ1 (20–28 Hz), γ2 (28–36 Hz) and γ-40 Hz (36–45 Hz). Power was also analyzed for different regions of interest (ROI), namely frontal, frontolateral, central, temporal, occipital, and parietal areas.
Coherence Analysis
Interelectrode normalized cross power (coherence) provides a measure for large-scale neuronal synchronization patterns. Coherence analysis is also based on standardized FFTs (Hanning windowing) of 4s non-overlapping epochs and frequency bands δ, θ, α, β (12-20 Hz), and γ-40 Hz. Since different inter-electrode ranges may be functionally relevant with respect to levels of conscious processing,8,9 electrode couples were grouped into short (inter-electrode distance: 3–10 cm), middle (10–15 cm), and long range (> 15 cm) pairs. Inter-electrode distance was measured along the scalp. As with power, coherences were analyzed for different ROIs: frontal, frontolateral, frontocentral, temporal, temporoparietal, and occipitoparietal.
Coherence effects were investigated on coherence values corrected for volume conduction effects, using the procedure proposed by Nunez et al.10,11 This correction mainly readjusts the short-range coherences among neighboring electrodes inflated by volume conduction.
Power and Coherences of Current Source Densities
Recent studies suggest that event-related EEG activity in the 40-Hz frequency band in waking is strongly influenced by ocular micro-saccades12,13 and EMG activity.14,15 Although it is presently not yet clear if this finding can be generalized to steady-state EEG recordings, in particular during sleep, caution dictates to explore other more robust analysis methods. Besides the EEG scalp potentials, we have, therefore, repeated our analyses on the current source densities (CSD).16 Extensive experimental and theoretical investigations of CSD, also known as scalp current densities (SCD), have demonstrated that this quantity is, by construction, free of reference artifacts,10,17 suppresses volume conduction very effectively for electrode spacings larger than about 3 cm,18,19 and has better localization than the raw EEG potentials.20,21 At present, CSD are considered to be largely immune to artifacts due to micro-saccades.22 CSD were derived from the measured EEG electrode potentials by first applying a spherical spline interpolation (order of splines = 4, max. degree of Legendre polynomials = 10, λ = 10-5) followed by the calculation of the Laplacian, which, being based on 2nd-order spatial derivatives, naturally takes out any reference electrode dependence.17 Furthermore, it has been shown that this method is applicable even with a small number of electrodes.23 The CSD were Fourier transformed, like the scalp potentials. Both, average power and coherence of the CSD signal were then calculated for specified frequency bands. This implies that an additional correction for volume conduction is not necessary.
In the following, we will discuss results based on the scalp potential (POT) and support them with the corresponding results from CSD analyses.
RESULTS
Lucidity Induction and Detection
Of the 6 participants tested, 3 subjects (1 m age 22, 2 f ages 23 years) were each able to become lucid once in the laboratory setting. All 6 subjects were very sensitive to sound and light. This heightened sensitivity may have been a characteristic of our subjects but unrelated to lucid dreaming. As a result of it, however, it was not possible to induce lucidity with dedicated devices, either those which are commercially available (e.g., the REM dreamer), or those of our own design. These devices rely on emitting specific light or sound signals, and only led to arousals and awakenings but not to lucidity in our subjects. The 3 recorded lucid episodes in our sample refer to spontaneous lucid episodes that occurred as a result of autosuggestion but not custom-made induction devices. Two of the participants used ear plugs (Ohropax).
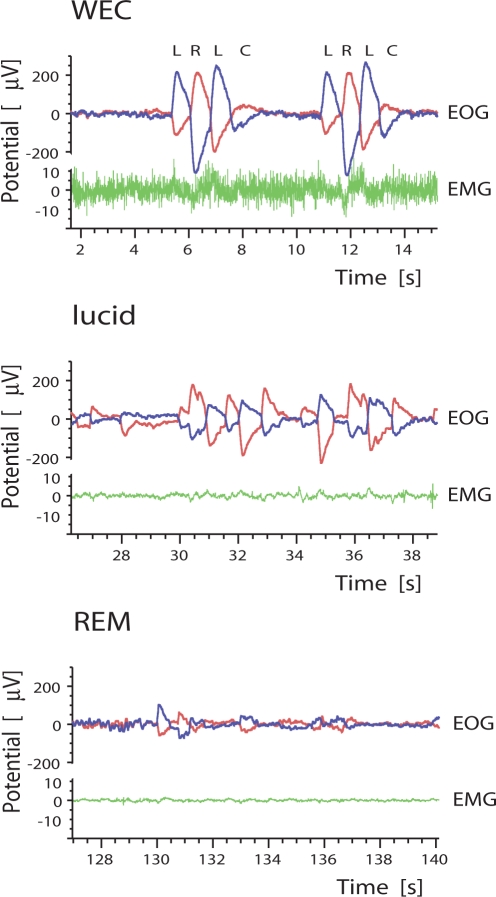
Subjects were trained to signal lucidity by horizontal eye movements. Since the eye signal (L-R-L) recommended in the literature1 resulted in frequent false positive events in the first subject tested (data not analyzed), participants were trained to signal lucidity by a more reliable pattern of sequential horizontal eye movements, consisting of ≥ 2 sets of eye movements separated by a brief pause. Subjects were asked to repeat this pattern several times during the lucid episode. Figure 1 shows recordings of typical repetitive eye movements carried out in waking and lucid dreaming. By contrast, the involuntary eye movements characteristic of REM sleep are of much lesser amplitude and more random in pattern. The amplitude of the REM sleep eye movements in Figure 1 appears relatively low only as a result of scaling to accommodate the larger amplitude of voluntary eye movements in lucid dreaming.
Figure 1.
Eye movement signals (EOG) and electromyographic activity (EMG) in waking with eyes closed (WEC), during lucid dreaming, and during REM sleep. EOG refers to 2 channels, one from each eye, as indicated by separate colors. Eyes are moved to the left (L), right (R), left (L), and back to a central position (C). Eye movements in lucid dreaming are systematic, repetitive, and more pronounced than in REM sleep. Low EMG tracings are found in lucid dreaming and REM sleep, highlighting the muscle relaxation common to both states. Mean EMG amplitude for lucid dreaming and REM sleep showed no systematic variability between the 2 states. Subject 1 gave 3 repetitive eye signals. Subjects 2 signalled 4 times and subject 3 three times repetitively.
In all 3 subjects who achieved lucidity in the laboratory, this occurred in the morning hours during one of the first 2 nights and not thereafter. Subjects who were able to achieve lucid control dreams either woke spontaneously (subjects 1 and 3) or were awakened at the termination of the REM period (subject 2). In the latter case, the REM period terminated within 1 min after the subject gave her last lucid eye signal. Lucidity was confirmed subjectively by a free report upon awakening. It is difficult (impossible) to indicate the duration of lucidity with precision. In future studies, it might be useful to instruct subjects to signal out as soon as they become lucid and again at time-estimated one minute intervals thereafter.
Power
Figure 2 shows power averaged across all electrodes as a function of frequency for the 3 states wake with eyes closed (WEC), lucid, and REM. The graph illustrates that power in lucid dreaming is REM-like in lower frequencies and rises above REM sleep at higher frequencies, commencing at around 28 Hz and peaking at 40 Hz. This effect is evident with both, POT and CSD. Compared to waking, power in frequencies 1–8 Hz, i.e., in frequency bands δ and θ, is increased in lucid dreaming and REM sleep. By contrast, power in the α band (8–12 Hz) is distinctively and selectively elevated in waking. This increase in α power seen clearly in Figure 2 is typical for the state of waking with eyes closed. Both, the higher δ and θ activity and the lower α power are evidence that lucid dreaming occurs in a state of sleep. The increase in higher frequency power during lucid dreaming compared to REM sleep shows that lucid dreaming differs from REM sleep and that lucid dreaming constitutes a unique, hybrid state of sleep.
Figure 2.
Grand averages for standardized power across the analyzed frequency bands, based on scalp potentials (POT, left frame) and CSD (right frame). Power values are shown for all 3 states, WEC (blue), lucid dreaming (red) and REM sleep (black). Frequency resolution is 4 Hz.
For statistical analysis, single-subject MANOVAs were calculated on mean power values averaged across all electrodes and epochs for each frequency band. STATE (WEC, lucid, and REM) was the independent variable. Statistical analysis was based on equal sample sizes in each state (subject 1: 374 s = 186 epochs, subject 2: 94 s = 46 epochs, subject 3: 70 s = 34 epochs). Between-state effects were further analyzed with Bonferroni post hoc procedures or t-tests.
Main effects for STATE with large effect sizes were found for all subjects, both with POT and CSD (see Table 1). Furthermore, analyses on both signals showed lucid dreaming to have REM-like power in lower frequency bands δ and θ, and significantly increased activity in high frequency bands γ1 (P < 0.05 in 2 subjects), γ2 (P < 0.01 in 2 subjects), γ-40 Hz (P < 0.05 in all subjects). CSD slightly differed from POT analysis in significance levels for δ and θ, with θ being REM-like for all subjects in CSD power vs. only 2 subjects in POT power. The reverse was true for δ power. However, we attribute this discrepancy to the large power values in these frequency bands, where relatively small differences lead to significant statistical effects. As is evident from Figure 2, power values in the respective frequency bands are very much alike in lucid dreaming and REM sleep (for individual values, see Table 2).
Table 1.
Effect Sizes and α Error Probabilities for MANOVAs on Power and Coherences
| POT |
CSD |
|||
|---|---|---|---|---|
| Effect | P | ηp2 | P | ηp2 |
| Power | ||||
| State | ||||
| subject 1 | <0.001 | 0.50 | <0.001 | 0.52 |
| subject 2 | <0.001 | 0.55 | <0.001 | 0.63 |
| subject 3 | <0.001 | 0.50 | <0.001 | 0.51 |
| Coherences | ||||
| State | ||||
| subject 1 | <0.001 | 0.41 | <0.05 | 0.16 |
| subject 2 | <0.001 | 0.32 | <0.05 | 0.14 |
| subject 3 | <0.001 | 0.35 | <0.001 | 0.60 |
| Range | ||||
| subject 1 | <0.001 | 0.24 | <0.01 | 0.03 |
| subject 2 | <0.001 | 0.17 | <0.001 | 0.04 |
| subject 3 | <0.001 | 0.14 | <0.001 | 0.04 |
| State × Range | ||||
| subject 1 | <0.01 | 0.03 | n.s. | n.a. |
| subject 2 | n.s. | n.a. | n.s. | n.a. |
| subject 3 | n.s. | n.a. | n.s. | n.a. |
| Site (selected electrode pairs) | ||||
| subject 1 | <0.001 | 0.23 | <0.001 | 0.13 |
| subject 2 | <0.001 | 0.23 | <0.001 | 0.07 |
| subject 3 | <0.001 | 0.18 | <0.001 | 0.15 |
| State × Site | ||||
| subject 1 | <0.001 | 0.21 | <0.001 | 0.14 |
| subject 2 | <0.001 | 0.25 | <0.001 | 0.13 |
| subject 3 | <0.001 | 0.14 | <0.001 | 0.12 |
Since most contrasts were significant on the P <0.001 level, effect sizes are reported (ηp2) instead of F values.
Note: SITE was based on selected electrode pairs in the following regions of interest: frontal (Fp1-Fp2, Fp1-F3, Fp1-F4, Fp2-F3, Fp2-F4, Fp1-Fz, Fp2-Fz, F3-Fz, F4-Fz, F3-F4); frontolateral (F7-Fz, F8-Fz, F7-F3, F8-F4, F7-Fp1, F8-Fp2); frontocentral (Fp1-Cz, Fp2-Cz, F3-Cz, F4-Cz, Fp1-C3, Fp2-C4, F3-C3, F4-C4); temporal (T3-T4, T3-T5, T3-T6, T4-T5, T4-T6, T5-T6); temporoparietal (T3-P3, T4-P4, T5-P3, T6-P4, T3-Pz, T4-Pz, T5-Pz, T6-Pz); and occipitoparietal (O1-P3, O2-P4, O1-O2, P3-P4, O1-Pz, O2-Pz). n.s. = not significant, n.a. = not applicable.
Table 2.
Standardized Power Values for Each Subject in the Relevant Frequency Bands Averaged Across EEG Electrodes
| Frequency band | WEC | Lucid | REM | ||
|---|---|---|---|---|---|
| Mean (s.e.) | Mean (s.e.) | Mean (s.e.) | |||
| δ POT | |||||
| subject 1 | 32.82 (0.64) | <** | 48.17 (0.67) | = | 48.88 (0.46) |
| subject 2 | 11.46 (0.60) | <** | 52.76 (1.25) | = | 53.04 (0.79) |
| subject 3 | 29.89 (1.28) | <** | 52.24 (2.05) | = | 55.13 (0.75) |
| δ CSD | |||||
| subject 1 | 19.83 (0.23) | <** | 37.85 (0.34) | <** | 39.35 (0.44) |
| subject 2 | 10.90 (0.41) | <** | 43.60 (0.59) | = | 42.06 (0.65) |
| subject 3 | 23.07 (0.45) | <** | 42.14 (1.48) | = | 36.62 (0.34) |
| θ POT | |||||
| subject 1 | 15.36 (0.43) | <** | 23.69 (0.50) | <** | 26.65 (0.31) |
| subject 2 | 14.02 (0.78) | <** | 30.89 (0.94) | = | 33.89 (0.63) |
| subject 3 | 13.41 (0.81) | <** | 27.90 (1.94) | = | 27.84 (0.54) |
| θ CSD | |||||
| subject 1 | 13.56 (0.18) | <** | 24.47 (0.34) | = | 24.49 (0.26) |
| subject 2 | 12.23 (0.95) | <** | 27.53 (0.53) | = | 28.28 (0.60) |
| subject 3 | 10.53 (0.39) | <** | 23.54 (0.91) | = | 23.86 (0.34) |
| γ-40 Hz POT | |||||
| subject 1 | 2.24 (0.12) | > ** | 1.18 (0.06) | > ** | 0.77 (0.03) |
| subject 2 | 5.77 (0.17) | > ** | 0.81 (0.04) | > ** | 0.40 (0.01) |
| subject 3 | 1.91 (0.26) | > ** | 1.18 (0.06) | > * | 0.76 (0.01) |
| γ-40 Hz CSD | |||||
| subject 1 | 0.49 (0.01) | > ** | 0.40 (0.01) | > ** | 0.35 (0.01) |
| subject 2 | 3.94 (0.11) | > ** | 0.66 (0.02) | > * | 0.38 (0.01) |
| subject 3 | 1.67 (0.12) | > ** | 0.87 (0.08) | > * | 0.64 (0.07) |
> value to the left is significantly bigger or smaller (<) than value to the right.
** = P <0.01;
= P <0.05,
Bonferroni post hoc procedures. First rows (POT) refer to power values extracted from the raw signal, rows labelled “CSD” list power values of the current source densities. POT = scalp potentials; CSD = current source densities; WED = wake with eyes closed
Localization of Effects
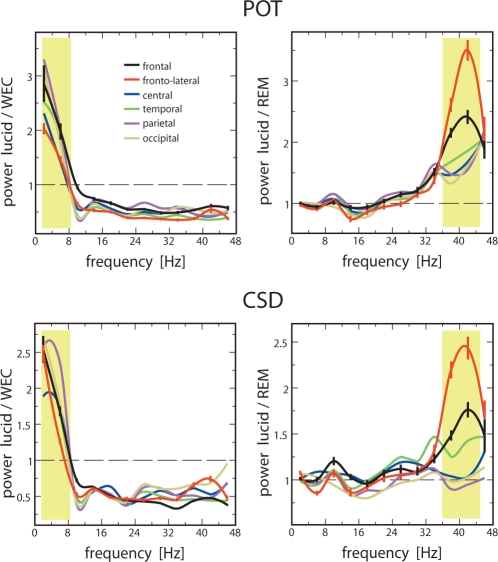
For a demonstration of state-related differences at specific ROIs on the scalp, the complete frequency spectrum at each ROI is plotted as power ratios between wake and lucid dreaming (Figure 3, left frames) and between lucid dreaming and REM sleep (Figure 3, right frames), averaged across all 3 subjects. ROIs are grouped into frontal (electrodes Fp1, Fp2, F3, F4, Fz), frontolateral (F7, F8), central (C3, C4, Cz), temporal (T3, T4, T5, T6), parietal (P3, P4, Pz), and occipital (O1, O2) areas.
Figure 3.
Regions of interest (ROIs): Grand averages for the ratio of mean FFT power in lucid dreaming vs. wake with eyes closed (WEC) (left frames) and lucid dreaming vs. REM sleep (right frames) for the analyzed frequency bands. The yellow shaded areas indicate the frequency bands of relevance, i.e., increased power in lower frequencies in lucid dreaming compared to waking (left frames) and increased 40-Hz band activity in lucid dreaming compared to REM (right frames). Power based on scalp potentials (POT) is plotted in the upper frames and CSD in the lower frames. Standard errors are indicated by vertical bars for frontal and frontolateral ROIs to facilitate interpretation of the relevant results. Frequency resolution is 4 Hz. Statistics for the contrasts between lucid dreaming and REM for frontal and frontolateral ROIs, listed in succession for subjects 1, 2, and 3: frontolateral POT power: P < 0.01, t = 11.86, 27.86, 7.02, dfcorr = 234.46, df = 90, dfcorr = 57,44; frontolateral CSD power: P < 0.01, t = 17.31, 15.59, 9.07, dfcorr = 280, df = 90, dfcorr = 53.69. Frontal POT power: P < 0.01, t = 12.54, 24.71, 9.17, dfcorr = 307, df = 90, dfcorr = 59.11; frontal CSD power: P < 0.05, t = 11.25, 8.27, 2.41, df = 370, dfcorr = 61.14, df = 66.
Besides the already mentioned finding (see Figure 2) that lucid dreaming is higher in lower-frequency and lower in high-frequency power than waking, Figure 3 (left frames) shows that for lucid dreaming and waking, all ROIs in the relevant frequency bands δ, θ, and γ-40 Hz are similarly activated, displaying no state-specific pattern. Accordingly, contrasts calculated for each ROI between the 2 states (t-tests) show no effects that are consistent for all 3 subjects. By contrast, when we compare lucid dreaming and REM sleep (Figure 3, right frames), we see a diverging pattern in the 40-Hz band (yellow shading), both, for POT and CSD ratios. The largest state-related difference in ROIs occurs frontolaterally and frontally, showing significantly elevated power in lucid dreaming (see caption Figure 3).
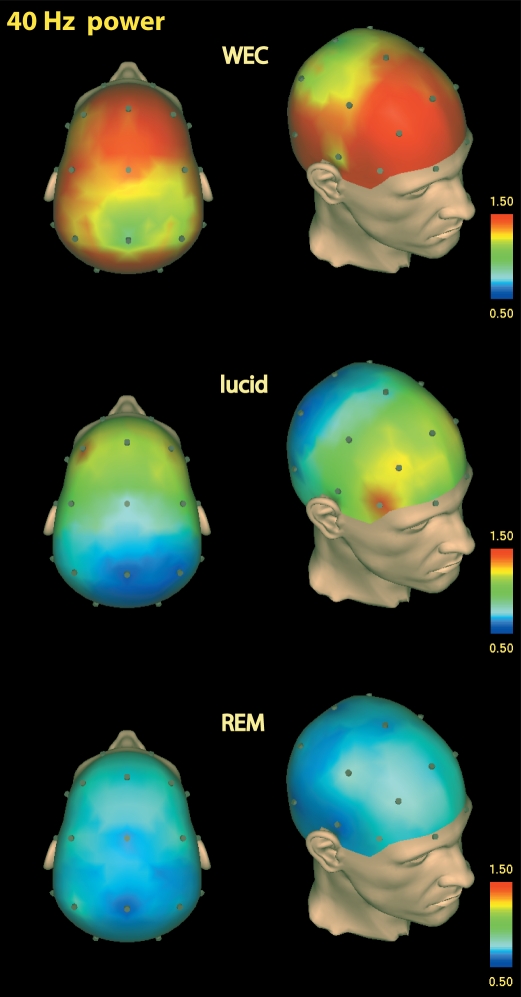
Since the most uniform increase in upper frequency activity was observed for γ-40 Hz, Figure 4 illustrates the topographic representation of the overall increase in γ-40 Hz activity in lucid dreaming compared to non-lucid REM sleep (P < 0.001) in a single subject.
Figure 4.
Single subject γ40-Hz standardized CSD power during WEC (top), lucid dreaming (middle), and REM sleep (bottom). Topographic images are based on movement-free EEG episodes and are corrected for ocular artifacts. For each state, power values are averaged across the respective episode.
To summarize, the findings on power suggest that lucidity occurs in a hybrid state with some features of REM (δ and θ) and some features of waking (γ) and that the frontal and frontolateral regions of the brain play a key role in gaining lucid insight into the dream state and agentive control.
Coherence: Short- vs. Middle- vs. Long-Range Effects
Coherences were analyzed to test if synchronization in waking, lucid dreaming and REM sleep differed in the degree of inter-scalp networking.8
As can be seen from Figure 5, short range coherences were larger than middle and long range values in all three states. The absence of state-related differences suggests that differences in long range coherences compared to medium and short range coherences cannot account for the change in consciousness.
Figure 5.
Grand averages of short, middle and long range coherences obtained for POT (top) and CSD (bottom), respectively, for states WEC, lucid dreaming and REM sleep. Coherences are averaged across electrode pairs in 4-s epochs. Short-range (55 pairs) was defined as 3 - 10 cm, mid range (51 pairs) as 10–15 cm, and long-range (65 pairs) as > 15-cm interelectrode distance. Standard errors are indicated by vertical bars for short range coherences to facilitate data interpretation. Frequency resolution is 4 Hz. POT = scalp potentials; CSD = current source densities; WED = wake with eyes closed
Coherences in waking were elevated in the α frequency band. For CSD, coherences were even highest for the α band. A peak in this frequency band is typical for WEC21 and was not present in either lucid dreaming or non-lucid REM sleep. This finding is consistent with the results for power and clearly distinguishes lucid dreaming from waking.
Compared to lucid dreaming, REM sleep is characterized by a broad decrease of coherences in all frequency bands analyzed. As can be seen from Figure 5, different results were obtained for high frequency coherences with the POT and the CSD method. In WEC and lucid dreaming, POT data show a steady increase of coherences throughout the γ band. By contrast, coherence values based on CSD remain at a uniform level with the exception of elevated coherences in the δ and θ bands.
For statistical analysis, MANOVAS were conducted for each subject with STATE (WEC, lucid, REM) and RANGE (short, middle, and long range) as independent variables and frequency band as dependent measures. With both analysis methods, POT and CSD, the main effects for STATE and RANGE were significant for all 3 subjects (see Table 1). Although in Figure 5, coherences in waking and lucid dreaming appear very similar, single-subject statistical analysis shows waking coherences to be higher than in lucid dreaming for α, β, and the 40-Hz band with both, POT and the CSD method. Coherences in the δ frequency band are wake-like in lucid dreaming.
With regard to RANGE, post hoc comparisons revealed significantly higher values for short range than middle and long range coherences in all frequency bands with both, POT and the CSD method, for all three subjects. Long and middle ranges were not significantly different.
The STATE × RANGE interaction was significant for POT in one subject but not for CSD. The effect size for the single effect established with the POT analysis was negligible, confirming the descriptive finding that short range coherences were uniformly increased across all states (see Figure 5).
Regional Differences
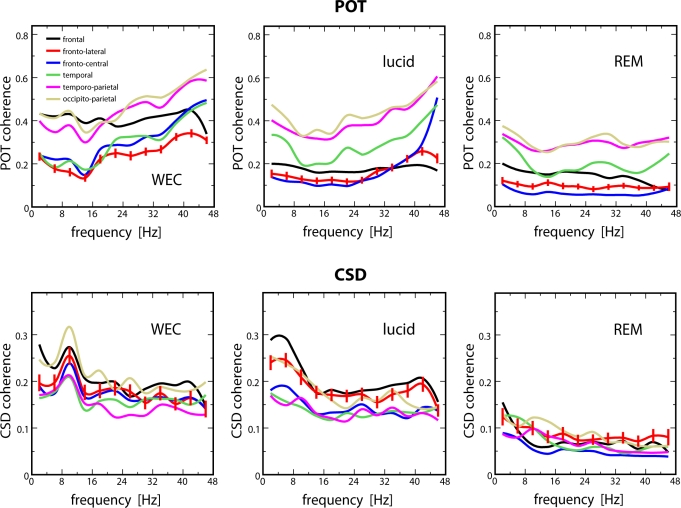
Similar to the results from general coherence analysis, splitting selected electrode pairs into ROIs (frontal, frontolateral, frontocentral, temporal, temporoparietal, occipitoparietal) yields a global difference between states, especially between lucid dreaming and REM sleep. As Figure 6 shows, coherences in lucid dreaming are strongest in frontal and frontolateral ROIs. This is similar to waking, except that in waking, we also see a strong occipitoparietal synchronicity which is strongest in the alpha band. By contrast, we cannot discern any ROI differentiation in REM sleep coherences.
Figure 6.
Grand averages for coherences at selected regions of interest (ROIs) based on POT (top) and CSD (bottom). F = frontal (10 electrode pairs), FL = frontolateral (6 pairs), FC = frontocentral (8 pairs), T = temporal (6 pairs), TP = temporoparietal (8 pairs), OP = occipitoparietal (6 pairs). Statistics for the larger-than-waking CSD coherences at the frontolateral ROI: P < 0.05, t = 6.20, 2.90, 2.18, df = 18 for subjects 1, 2, and 3. Statistics for the larger-than-REM CSD coherences in lucid dreaming at the frontal ROI: P < 0.05, t = 9.12, 2.96, 6.53, df = 14. Note that only CSD was analyzed statistically (see text). Standard errors are indicated by vertical bars for the frontolateral ROI. Frequency resolution is 4-Hz. POT = scalp potentials; CSD = current source densities; WED = wake with eyes closed
Single subject MANOVAs with STATE (3) and SITE (6) as independent variables and mean coherence values in each frequency band as dependent variables confirm the described state-dependent differences in overall coherence levels at distinct ROIs. Besides the already established effect for STATE, we found significant effects for SITE and the STATE × SITE interaction (see Table 1). In light of our observation that between-state differences in coherences occur on a global level and do not appear frequency specific, we looked at differences in ROIs across all frequency bands. Since coherences based on POT and CSD differ substantially, we will report only the relevant statistics on CSD because they represent the more reliable signal. Consistent for all subjects were the larger-than-waking coherences in lucid dreaming at the frontolateral ROI and the larger-than-REM coherences in lucid dreaming at the frontal ROI.
In conclusion, lucid dreaming coherences are quite similar to waking without the waking peak in the α frequency band. In REM sleep there was no increase in δ or θ coherences relative to waking as there was for power. Instead, across all frequencies, REM sleep coherences were decreased, evidence of a large scale desynchronization that is consistent with literature reports.24,25 By contrast, the high level of synchronization observed in lucid dreaming shows that this state is characterized by wake-like inter-scalp networking, including high-frequency bands. The high synchronization in the α band observed only in waking, clearly distinguishes lucid dreaming from waking, however, and marks lucid dreaming as a hybrid state. As with power values, the hybridicity of lucid dreaming is most pronounced in frontal and frontolateral coherences.
DISCUSSION
Methodological Issues
We were surprised to find that while dream lucidity may commonly occur in home settings, it is not easily transferable to the sleep laboratory. Of our initial group of 20 subjects who claimed that they experienced lucidity at least twice a week, only three achieved lucidity in the laboratory, and they achieved it only once. Since our subjects were highly motivated and carefully monitored, we are reluctantly forced to conclude that lucid dreaming is fragile and not easy to study in the laboratory. This makes it all the more important to evaluate our hard-won data critically.
Were our subjects really lucid? The eye movement sequence adopted by us to detect voluntary signals is more complex a code than that which was previously described. We changed our criteria to avoid what appeared to be false positive patterns in our first subject (not analyzed here).
To assure ourselves that our subjects really were making voluntary signals, we adjusted the amplitude of our EOG recordings to reveal the high voltage deflections associated with intentional horizontal movements. This fact, in addition to the post-awakening confirmation of subjective experience, strengthens the interpretation of our findings. The persistence of low EMG voltage, the diminished α–power and coherence, and the REM-like power in lower frequency bands δ and θ are evidence that our subjects were still asleep when they became lucid.
When we compare results for power and coherences, we find consistent effects for the α band in waking and for the θ band in lucid dreaming. The increased activity in higher frequency power observed during waking and lucid dreaming was also observed for coherences based on POT but not on those derived from CSD. Since, at this point, we do not know whether the discrepancies between results based on POT and CSD are related to over- or undercorrection of one of these methods, we chose to discuss only those results that are evident from both procedures. With both methods, we observe a substantial increase of coherence levels in lucid dreaming compared to REM sleep.
Lucid Dreaming as a Hybrid State
Our new data help us to resolve a controversy regarding the relationship of lucidity to REM sleep. In previous work, it has been asserted that because lucidity usually emerged from REM sleep dreaming, that lucidity was a REM sleep phenomenon. Our results suggest, instead, that lucidity occurs in a state with features of both REM sleep and waking. In order to move from non-lucid REM sleep dreaming to lucid REM sleep dreaming, there must be a shift in brain activity in the direction of waking.
This is what we mean by “hybrid” and this interpretation is of crucial importance to our hypothesis that lucid dreaming is of particular importance to studies of consciousness.
We assert that REM sleep dreaming is non-lucid in that the dreamer mistakenly concludes that he or she is awake whereas in fact, he or she is asleep. The reason for this delusional error could be the persistent inactivation of frontal and parietal cortical circuits necessary for waking memory, self-reflective awareness, and insight. In lucid dreaming, self-reflection arises and augments so that the dreamer recognizes that he is not awake but asleep. In our view, something must have changed the underlying brain physiology such that self-reflection, voluntary control and other characteristics of waking come to be associated with the subjective experience of dreaming. We submit that the observed increases in frontal lobe 40-Hz power and the global increase in EEG coherence compared to REM sleep may be signs of a distinctive neurophysiology.
Lucidity and the AIM Model
This hybridicity interpretation relates to our 3D AIM model of brain-mind-state which explains state differences dimensionally and categorically.26 The AIM model plots Activation (A) Input-output gating (I) and chemical Modulation (M) as the x, y, and z dimensions of a 3D state space. Our underlying assumption is that brain-mind-state is never static. Instead, it is dynamic and the state of the brain-mind is constantly changing. Another assumption is that the state space described by AIM has an infinite number of subregions which far exceeds what we now consider to be cardinal states like waking, NREM and REM sleep. Lucid dreaming is one of them. Others include diminished levels of waking consciousness including coma and the so-called mental illnesses, especially those mood disorders already known to be characterized by changes in sleep.
Lucidity and Consciousness
Differences between REM sleep and lucid dreaming were most prominent in the 40-Hz frequency band. The increase in 40-Hz power was especially strong at frontolateral and frontal sites. These results suggest that 40-Hz activity holds a functional role in the modulation of conscious awareness across different conscious states.
We also found evidence of heightened cortical networking, across short as well as middle and long ranges for all frequency bands, suggesting a large increase in global networking. A selective rise in 40-Hz binding (based on EEG) was observed for POT but not for CSD. Although coherences in β and 40-Hz frequency bands reached waking levels in two subjects for CSD, however, inspection of the complete frequency spectrum suggests a uniform increase in networking rather than a frequency-specific one. Such a specific increase would support theories linking 40-Hz synchronization with perceptual binding27–29 and the integration of cognitive processes.30–32 By restricting the discussion to the CSD results, we may be disregarding an existing effect (β-error). However, it may also be that previously published results on 40-Hz binding were influenced by artifacts from concurrent noncortical activity as, for example, micro-saccades or EMG. Regardless of a possible frequency-specific effect in the 40-Hz band, the finding that networking in lucid dreaming differs distinctly from that in REM sleep further documents the hybrid character of lucid dreaming, with REM-like features in lower frequency band power and wake-like synchronization across all frequency bands. Table 3.
Table 3.
Coherence Values Averaged Across All EEG Electrode Pairs for Single subjects
| Frequency band | WEC | Lucid | REM | ||
|---|---|---|---|---|---|
| Mean (s.e.) | Mean (s.e.) | Mean (s.e.) | |||
| δ POT | |||||
| subject 1 | 0.28 (0.02) | = | 0.23 (0.02) | > ** | 0.17 (0.02) |
| subject 2 | 0.15 (0.02) | = | 0.20 (0.02) | = | 0.20 (0.02) |
| subject 3 | 0.28 (0.01) | = | 0.25 (0.01) | > ** | 0.11 (0.01) |
| δ CSD | |||||
| subject 1 | 0.08 (0.01) | <** | 0.15 (0.01) | > ** | 0.09 (0.01) |
| subject 2 | 0.13 (0.01) | = | 0.14 (0.01) | > ** | 0.07 (0.01) |
| subject 3 | 0.24 (0.01) | = | 0.24 (0.01) | > ** | 0.05 (<0.01) |
| θ POT | |||||
| subject 1 | 0.23 (0.02) | > ** | 0.15 (0.02) | = | 0.12 (0.02) |
| subject 2 | 0.16 (0.02) | = | 0.19 (0.02) | = | 0.21 (0.02) |
| subject 3 | 0.31 (0.01) | > ** | 0.24 (0.01) | > ** | 0.10 (0.01) |
| θ CSD | |||||
| subject 1 | 0.08 (<0.01) | <** | 0.15 (0.01) | > ** | 0.08 (0.01) |
| subject 2 | 0.14 (0.01) | = | 0.14 (0.01) | > ** | 0.07 (0.01) |
| subject 3 | 0.30 (0.01) | > ** | 0.24 (0.01) | > ** | 0.05 (<0.01) |
| α POT | |||||
| subject 1 | 0.21 (0.02) | > ** | 0.13 (0.02) | = | 0.10 (0.02) |
| subject 2 | 0.17 (0.02) | = | 0.18 (0.01) | = | 0.13 (0.02) |
| subject 3 | 0.27 (0.01) | > ** | 0.19 (0.01) | > ** | 0.09 (0.01) |
| α CSD | |||||
| subject 1 | 0.12 (0.01) | = | 0.12 (0.01) | > ** | 0.07 (0.01) |
| subject 2 | 0.16 (0.01) | > ** | 0.11 (0.01) | > ** | 0.07 (0.01) |
| subject 3 | 0.39 (0.01) | > ** | 0.23 (<0.01) | > ** | 0.04 (<0.01) |
| β POT | |||||
| subject 1 | 0.17 (0.01) | = | 0.15 (0.02) | = | 0.12 (0.02) |
| subject 2 | 0.15 (0.02) | = | 0.17 (0.01) | > ** | 0.11 (0.01) |
| subject 3 | 0.28 (0.01) | > ** | 0.20 (0.01) | > ** | 0.09 (0.01) |
| β CSD | |||||
| subject 1 | 0.08 (<0.01) | = | 0.09 (0.01) | > ** | 0.06 (0.01) |
| subject 2 | 0.09 (0.01) | = | 0.09 (<0.01) | > ** | 0.06 (<0.01) |
| subject 3 | 0.26 (0.01) | > ** | 0.22 (<0.01) | > ** | 0.04 (<0.01) |
| γ-40 Hz POT | |||||
| subject 1 | 0.56 (0.01) | > ** | 0.43 (0.02) | > ** | 0.30 (0.02) |
| subject 2 | 0.44 (0.02) | > ** | 0.26 (0.01) | > ** | 0.06 (0.01) |
| subject 3 | 0.41 (0.02) | > * | 0.37 (0.01) | > ** | 0.11 (<0.01) |
| γ-40 Hz CSD | |||||
| subject 1 | 0.09 (<0.01) | = | 0.08 (<0.01) | > * | 0.05 (<0.01) |
| subject 2 | 0.13 (<0.01) | > ** | 0.10 (<0.01) | > ** | 0.05 (<0.01) |
| subject 3 | 0.27 (<0.03) | = | 0.24 (<0.04) | > ** | 0.03 (<0.01) |
> value to the left is significantly bigger or smaller (<) than value to the right.
= P <0.01;
= P <0.05,
Bonferroni post hoc procedures. POT = scalp potentials; CSD = current source densities; WED = wake with eyes closed
Regional Differences
Our new results beg comparison with recent imaging studies which show that compared to waking, REM sleep is characterized by diminished activation of the dorsolateral prefrontal cortex (DLPFC).33–36 Since the DLPFC is thought to be the site of executive ego function, it has been suggested that the loss of volition, self-reflective awareness, and insight that is typical of normal dreaming, may be related to DLPFC inactivation. In lucid dreaming, these psychological functions return leading to the prediction that lucid dreaming will involve reactivation of the DLPFC.37 Our EEG data do not permit us to test this hypothesis because EEG measures are not highly localized. We consider it significant, however, that the coherence increases we observed are clearly frontal and look forward to putting our subjects in the PET scanner to obtain data regarding the DLPFC hypothesis.
Since submission of this manuscript we have become aware of an fMRI study of lucid dreaming35 which reports that in addition to the DLPFC activation predicted by us,34 a widely distributed cortical network including frontal, parietal, and temporal zones, corresponding to the uniquely human expansion over the macaque, was activated when subjects became lucid.
We are tempted to interpret this finding as evidence that the substrate of what Edelman38 calls secondary consciousness is turned on and conveys the many aspects of waking consciousness that characterize lucidity.
Conclusion
Our findings indicate that when subjects become lucid, they shift their EEG power, especially in the 40-Hz range and especially in frontal regions of the brain. We emphasize that this shift is, in part, a consequence of pre-sleep autosuggestion indicating that REM dream consciousness, which is largely automatic, i.e., spontaneous, involuntary, and intrinsic, is partially subject to volitional force. Our speculative hypothesis is that dream lucidity arises when wake-like frontal lobe activation is associated with REM-like activity in posterior structures.
DISCLOSURE STATEMENT
This was not an industry supported study. The authors have indicated no financial conflicts of interest.
ACKNOWLEDGMENTS
We thank H. Steinmetz for the use of his laboratory; U. Ermis for assisting with the EEG, J. Bredenkamp, K. Schermelleh-Engel and A. Bröoder for useful discussion of statistical procedures.
Abbreviations
- WEC
wake with eyes closed
- REM
rapid eye movements
- POT
scalp potentials
- CSD
current source densities
- SCD
scalp current densities
- ROI
regions of interest
- EEG
electroencephalography
- EMG
electromyography
- EOG
electrooculogram
- FFT
fast Fourier transform
- MANOVA
multivariate analysis of variance
REFERENCES
- 1.LaBerge S, Levitan L, Dement WC. Lucid dreaming: Physiological correlates of consciousness during REM sleep. J Mind Behav. 1986;7:251–58. [Google Scholar]
- 2.Arnold-Forster M. Studies in dreams. New York: McMillan; 1921. [Google Scholar]
- 3.Brooks JE, Vogelsong J. The conscious exploration of dreaming: discovering how we create and control our dreams. Bloomington: 1stBooks Library; 2000. [Google Scholar]
- 4.D'Hervey de Saint-Denis JML, Marquis . In: Dreams and the means of directing them. van Schatzman M, trans, Fry N, editors. London: Gerald Duckworth; 1982. [Google Scholar]
- 5.Holzinger B, LaBerge S, Levitan L. Psychophysiological correlates of lucid dreaming. Dreaming. 2006;6:88–95. [Google Scholar]
- 6.Gratton G, Coles MG, Donchin EA. A new method for off-line removal of ocular artefact. Electroenceph Clin Neurophysiol. 1983;55:468–84. doi: 10.1016/0013-4694(83)90135-9. [DOI] [PubMed] [Google Scholar]
- 7.Rechtschaffen A, Kales A. A manual of standardized terminology, techniques, and scoring system for sleep stages of human subjects (NIH publ. No. 204) Washington, DC: U.S. Government Printing Office; 1968. [Google Scholar]
- 8.Melloni L, Molina C, Pena M, et al. Synchronization of neural activity across cortical areas correlates with conscious perception. J Neurosci. 2007;27:2858–65. doi: 10.1523/JNEUROSCI.4623-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dehaene S, Changeux JP, Naccache L, et al. Conscious, preconscious, and subliminal processing: a testable taxonomy. Trends Cogn Sci. 2006;10:204–11. doi: 10.1016/j.tics.2006.03.007. [DOI] [PubMed] [Google Scholar]
- 10.Nunez PL, Srinivasan R, Westdorp AF, et al. EEG coherence I: statistics, reference electrode, volume conduction, Laplacians, cortical imaging, and interpretation at multiple scales. Electroenceph Clin Neurophysiol. 1997;103:499–515. doi: 10.1016/s0013-4694(97)00066-7. [DOI] [PubMed] [Google Scholar]
- 11.Nunez PL, Silberstein RB, Shi ZP, et al. EEG coherence II: experimental comparisons of multiple measures. Clin Neurophysiol. 1999;110:469–86. doi: 10.1016/s1388-2457(98)00043-1. [DOI] [PubMed] [Google Scholar]
- 12.Yuval-Greenberg S, Tomer O, Keren AS, et al. Transient induced gamma-band response in EEG as a manifestation of miniature saccades. Neuron. 2008;58:429–41. doi: 10.1016/j.neuron.2008.03.027. [DOI] [PubMed] [Google Scholar]
- 13.Weinstein JM, Balaban CD, Verl-Hoeve JN. Directional tuning of the human presaccadic spike potential. Brain Res. 1991;543:243–50. doi: 10.1016/0006-8993(91)90034-s. [DOI] [PubMed] [Google Scholar]
- 14.Whitham EM, Lewis T, Pope KJ, et al. Thinking activates EMG in scalp electrical recordings. Clin Neurophysiol. 2008;119:1166–75. doi: 10.1016/j.clinph.2008.01.024. [DOI] [PubMed] [Google Scholar]
- 15.Whitham EM, Pope KJ, Fitzgibbon SP, et al. Scalp electrical recording during paralysis: Quantitative evidence that EEG frequencies above 20 Hz are contaminated by EMG. Clin Neurophysiol. 2007;118:1877–88. doi: 10.1016/j.clinph.2007.04.027. [DOI] [PubMed] [Google Scholar]
- 16.Hjorth B. Online transformation of EEG scalp potentials into orthogonal source derivations. Electroenceph Clin Neurophysiol. 1975;39:526–30. doi: 10.1016/0013-4694(75)90056-5. [DOI] [PubMed] [Google Scholar]
- 17.Lagerlund TD, Sharbrough FW, Busacker NE, et al. Interelectrode coherences from nearest-neighbor and spherical harmonic expansion computation of Laplacian scalp potential. Electroenceph Clin Neurophysiol. 1995;95:178–88. doi: 10.1016/0013-4694(95)00025-t. [DOI] [PubMed] [Google Scholar]
- 18.Winter WR, Nunez P, Ding J, et al. Comparison of the effect of volume conduction on EEG coherence with the effect of field spread on MEG coherence. Statist Med. 2007;26:3946–57. doi: 10.1002/sim.2978. [DOI] [PubMed] [Google Scholar]
- 19.Srinivasan R, Winter WR, Ding J, et al. EEG and MEG coherence: Measures of functional connectivity at distinct spatial scales of neocortical dynamics. J Neurosci Methods. 2007;166:41–52. doi: 10.1016/j.jneumeth.2007.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Srinivasan R. Methods to improve the spatial resolution of EEG. Int J Bioelectromagnetism. 1999;1:102–11. [Google Scholar]
- 21.Nunez PL, Srinivasan R. The Neurophysics of EEG. 2nd ed. New York: Oxford University Press; 2006. Electric fields of the brain. [Google Scholar]
- 22.Trujillo LT, Peterson MA, Kaszniak AW, et al. EEG phase synchrony differences across visual perception conditions may depend on recording and analysis methods. Clin Neurophysiol. 2005;116:172–89. doi: 10.1016/j.clinph.2004.07.025. [DOI] [PubMed] [Google Scholar]
- 23.Kayser J, Tenke CE. Principal components analysis of Laplacian waveforms as a generic method for identifying ERP generator patterns: II. Adequacy of low-density estimates. Clin Neurophysiol. 2006;117:369–80. doi: 10.1016/j.clinph.2005.08.033. [DOI] [PubMed] [Google Scholar]
- 24.Cantero JL, Atienza M, Madsen JR, Stickgold R. Gamma EEG dynamics in neocortex and hippocampus during human wakefulness and sleep. NeuroImage. 2004;22:1271–80. doi: 10.1016/j.neuroimage.2004.03.014. [DOI] [PubMed] [Google Scholar]
- 25.Fell J, Staedtgen M, Burr W, et al. Rhinal-hippocampal EEG coherence is reduced during human sleep. Eur J Neurosci. 2003;18:1711–6. doi: 10.1046/j.1460-9568.2003.02934.x. [DOI] [PubMed] [Google Scholar]
- 26.Hobson JA, Pace-Schott EF, Stickgold R. Dreaming and the brain: toward a cognitive neuroscience of conscious states. Behav Brain Sci. 2000;23:793–1121. doi: 10.1017/s0140525x00003976. [DOI] [PubMed] [Google Scholar]
- 27.Singer W. Striving for coherence. Nature. 1999;397:391–2. doi: 10.1038/17021. [DOI] [PubMed] [Google Scholar]
- 28.Singer W. Consciousness and the binding problem. Ann NY Acad Sci. 2001;929:123–46. doi: 10.1111/j.1749-6632.2001.tb05712.x. [DOI] [PubMed] [Google Scholar]
- 29.Fries P, Nikolic D, Singer W. The gamma cycle. Trends Neurosci. 2007;30:309–16. doi: 10.1016/j.tins.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 30.Miltner WH, Braun C, Arnold M, Witte H, Taub E. Coherence of gamma-band EEG activity as a basis for associative learning. Nature. 1999;37:434–36. doi: 10.1038/17126. [DOI] [PubMed] [Google Scholar]
- 31.Rodriguez E, George N, Lachaux JP, Martinerie J, Renault B, Varela F. Perception’s shadow: long-distance synchronization of human brain activity. Nature. 1999;397:430–3. doi: 10.1038/17120. [DOI] [PubMed] [Google Scholar]
- 32.Varela F, Lachaux JP, Rodriguez E, Martinerie J. The brainweb: phase synchronization and large-scale integration. Nat Rev Neurosci. 2001;2:229–39. doi: 10.1038/35067550. [DOI] [PubMed] [Google Scholar]
- 33.Maquet P, Peters JM, Aerts J, et al. Functional neuroanatomy of human rapid-eye-movement sleep and dreaming. Nature. 1996;383:163–6. doi: 10.1038/383163a0. [DOI] [PubMed] [Google Scholar]
- 34.Muzur A, Pace-Schott EF, Hobson JA. The prefrontal cortex in sleep. Trends Cogn Sci. 2002;6:475–81. doi: 10.1016/s1364-6613(02)01992-7. [DOI] [PubMed] [Google Scholar]
- 35.Nofzinger EA, Buysse DJ, Miewald JM, et al. Forebrain activation in REM sleep: an FDG PET study. Brain Res. 1997;770:192–201. doi: 10.1016/s0006-8993(97)00807-x. [DOI] [PubMed] [Google Scholar]
- 36.Hobson JA. Finally Some One: Reflections on Thomas Metzinger’s “Being No One”. Psyche. 2005;11:1–8. [Google Scholar]
- 37.Dresler M, Koch S, Wehrle R, et al. Neural correlates of consciousness visualized by sleep imaging. personal communication, November 10, 2008.
- 38.Edelman GM. Wider than the sky: a revolutionary view of consciousness. London: Penguin Press Science; 2005. [Google Scholar]