In chronic lymphocytic leukemia (CLL) lenalidomide causes striking immune activation, possibly leading to clearance of tumor cells. This study shows that upregulation of CD80 on tumor cells and T-cell activation, which appears to be dispensable for the drug’s anti-tumor effects, correlate with unique toxicities of lenalidomide in CLL. This provides a rationale for combinations of lenalidomide with immunosuppressive agents. See related perspective article on page 1198.
Keywords: chronic lymphocytic leukemia, lenalidomide, tumor flare, CD80, cytokine release syndrome
Abstract
Background
In chronic lymphocytic leukemia lenalidomide causes striking immune activation, possibly leading to clearance of tumor cells. We conducted this study to investigate the mechanism of action of lenalidomide and the basis for its unique toxicities in chronic lymphocytic leukemia.
Design and Methods
Patients with relapsed chronic lymphocytic leukemia were treated with lenalidomide 20 mg (n=10) or 10 mg (n=8) daily for 3 weeks on a 6-week cycle. Correlative studies assessed expression of co-stimulatory molecules on tumor cells, T-cell activation, cytokine levels, and changes in lymphocyte subsets.
Results
Lenalidomide upregulated the co-stimulatory molecule CD80 on chronic lymphocytic leukemia and mantle cell lymphoma cells but not on normal peripheral blood B cells in vitro. T-cell activation was apparent in chronic lymphocytic leukemia, weak in mantle cell lymphoma, but absent in normal peripheral blood mononuclear cells and correlated with the upregulation of CD80 on B cells. Strong CD80 upregulation and T-cell activation predicted more severe side effects, manifesting in 83% of patients as a cytokine release syndrome within 8–72 h after the first dose of lenalidomide. Serum levels of various cytokines, including tumor necrosis factor-α, increased during treatment. CD80 upregulation on tumor cells correlated with rapid clearance of leukemic cells from the peripheral blood. In contrast, neither the severity of the cytokine release syndrome nor the degree of T-cell activation in vitro correlated with clinical response.
Conclusions
Upregulation of CD80 on tumor cells and T-cell activation correlate with unique toxicities of lenalidomide in chronic lymphocytic leukemia. However, T-cell activation appears to be dispensable for the drug’s anti-tumor effects. This provides a rationale for combinations of lenalidomide with fludarabine or alemtuzumab.
Introduction
The immunomodulating drug lenalidomide is active in chronic lymphocytic leukemia (CLL). Response rates of 47% and 32% have been reported.1,2 However, several side effects are more severe in CLL than in other diseases and have hindered the widespread use of lenalidomide in CLL. Most important among the CLL-specific side effects is the tumor flare reaction, characterized by tender lymph node swelling, low-grade fever, pain and rash.3 Significant morbidity and fatal adverse events have been attributed to tumor flare reaction, often in conjunction with tumor lysis syndrome.4,5 Based on these observations, clinical trials have been modified to include lower starting doses and close monitoring. In contrast, lenalidomide 30 mg daily in multiple myeloma and 25 mg daily in relapsed or refractory aggressive non-Hodgkin’s lymphoma has not caused tumor flare reaction or tumor lysis syndrome.6,7 The basis for these differences in the side effect profile of lenalidomide is unclear.
Multiple biological effects of lenalidomide have been described; however, the relative contributions of individual effects to this drug’s anti-cancer activity remain to be defined. Despite the rapid decrease in leukemic cell count in patients at the start of treatment,1 lenalidomide has no apparent pro-apoptotic effect on CLL cells in vitro.4,8 Lenalidomide has been thought to modulate the tumor microenvironment by downregulating cytokines including tumor necrosis factor alpha (TNFα), interleukin (IL)-6, and vascular endothelial growth factor (VEGF). However, clinical studies in CLL have reported increased serum cytokine levels during lenalidomide treatment.1,2 A postulated inhibitory effect on bone marrow angiogenesis could not be confirmed.2
Immune activation is thought to be an important effect of lenalidomide and related immunomodulating drugs. Lenalidomide can activate T cells, NK cells, or both and in some situations can lead to an expansion of immune effector cells in vivo.9–11 Lenalidomide has been found to enhance immunological synapse formation, which could increase the anti-tumor effect of immune effector cells.12 Upregulation of co-stimulatory molecules and/or surface antigens on CLL cells, which could render the leukemic cells more immunogenic, has been described.4,8 A partially overlapping mechanism of action has been attributed to DNA oligonucleotides containing CpG motifs that are also known to increase expression of co-stimulatory molecules and to cause immune activation.13,14 However, evidence linking immune activation and clinical response to lenalidomide is missing.
Design and Methods
Patients, clinical samples, flow cytometry and in vitro assays
CLL patients treated with lenalidomide were enrolled on the institutional review board-approved NHLBI protocol 07-H-0104 (ClinicalTrials.gov Identifier: NCT00465127). The patients’ baseline characteristics are given in Table 1. In this study lenalidomide was given in cycles: 3 weeks on, 3 weeks off. The starting dose for the first 10 patients was 20 mg, whereas that for subsequent patients was 10 mg. Allopurinol was given for 14 days in cycles 1 – 3. Side effects were classified according to the NCI common toxicity criteria (version 3.0). Control peripheral blood mononuclear cells (PBMC) from healthy volunteers (n=12) were obtained through the Department of Transfusion Medicine, NIH. Cells from patients with mantle cell lymphoma (MCL) (n=12, Online Supplementary Table S1) were collected under NHLBI protocol 04-H-0012 (ClinicalTrials.gov Identifier: NCT0071045). PBMC isolated by gradient centrifugation (Lymphocyte Separation Media, MP Biomedicals, Irvine, CA, USA) were adjusted to 5×106/mL and cultured in AIM V serum-free media (Invitrogen, Carlsbad, CA, USA). Lenalidomide (Sequoia Research Products, Berkshire, UK) and CpG oligonucleotides (Integrated DNA Technologies, Coralville, IA, USA) were used at doses of 2 μM and 1 μM, respectively. The mean fluorescence intensity (MFI) of surface antigens was measured by flow cytometry on BD FACSCanto II equipment and analyzed with BD FACS-Diva software (BD BioSciences, San Jose, CA, USA). Mouse anti-human antibodies were: fluorescein isothiocyanate (FITC)-conjugated CD2 and CD19, phycoerythrin (PE)-conjugated CD25, CD69, CD80, CD86, and CD95, CD3 allophycocyanin (APC), mouse isotype G1 FITC, and mouse isotype G2a PE (BD BioScience, San Jose, CA, USA).
Table 1.
Patients’ characteristics at the start of lenalidomide treatment.
Serum cytokines
Serum samples obtained from study subjects prior to treatment (n=13), on day 4 (n=4) and on day 8 (n=13) of the first cycle of treatment were stored at −80°C. Samples from patient 13 were collected on days 1, 2, 3, 4, and 8. The cytokines and chemokines IFN-γ, TNF-α, IL-1α, IL-1β, IL-1Rα, IL-2, IL4, IL-5, IL-6, IL-10, IL-17, CXCL5, CXCL8/IL-8, CCL2, CCL3, and CCL4 were measured using Fluorokine MultiAnalyte Profiling (F-MAP) kits from R&D systems (Minneapolis, MN, USA) on a Luminex IS100 instrument (Luminex Corp. Austin, TX, USA) and analyzed using Masterplex software (Hitachi Software Engineering America, South San Francisco, CA, USA) (Online Supplementary Table S2).
Peripheral blood lymphocyte subsets and lymph node analysis
T, B and NK cells stained with CD4 APC, CD3 FITC, CD8 PE, CD45 PerCP, CD16, and CD56 PE and CD 19 APC using the BD FACS Sample Prep Assistant IVD (Becton Dickinson, Franklin Lakes, NJ, USA) were quantified at the indicated times on a FACS Caliber using FACS multiSET software (Becton Dickinson, Franklin Lakes, NJ, USA). Core biopsies from superficial lymph nodes obtained prior to lenalidomide treatment and on day 8 of cycle 1 of treatment in 11 patients were stained for CD3, CD68 and CD56. The number of CD3+ cells was scored in five representative high-power fields. Images were captured at 400x fold magnification on an Olympus Bx41 microscope (Center Valley, PA, USA).
Statistics
Changes in surface antigen expression and lymphocyte subsets were examined by paired t tests. MFI measurements of surface antigens were compared using an unpaired t test, assuming unequal variances. Pearson’s correlation coefficient was used to describe correlations. All tests were two-tailed and a p value of less than 0.05 was used as the criterion for statistical significance.
Results
Lenalidomide induces expression of co-stimulatory molecules preferentially on malignant B cells but responses are weaker than those induced by immune stimulatory CpG oligonucleotides
Lenalidomide and CpG oligonucleotides can increase the expression of co-stimulatory molecules on tumor cells. The effect of CpG differs between various B-cell malignancies.15 Whether the response to lenalidomide also differs between different types of B cells has not been determined. We measured the effect of lenalidomide on the expression of CD80, CD86 and CD95 on tumor cells in vitro by flow cytometry. Seventeen of the 18 patients with CLL enrolled on a phase II clinical trial using lenalidomide were studied. For comparison, we included peripheral blood B cells from healthy volunteers (n=12), and from patients with leukemic MCL (n=12). After 48 h of in vitro exposure to lenalidomide, CD80 or CD95 expression on normal B cells was unchanged. In contrast, average expression of CD80 and CD95 increased almost 2-fold on CLL B cells (Figure 1A). CD86 was upregulated on normal B cells (average increase 85±30%) and more strongly on CLL cells (average increase 125±81%). Leukemic MCL cells upregulated all three markers in the same way as CLL cells (Figure 1A). For comparison we determined the effect of CpG oligonucleotides on the same samples (Figure 1B). Upregulation of CD80 was again stronger on CLL and MCL cells than on normal B cells, but CpG increased expression of CD86 and CD95 on all three types of B cells equally and was at least twice as potent as lenalidomide in inducing CD80 and CD86. Thus, while lenalidomide preferentially upregulated co-stimulatory molecules on malignant B cells, CpG oligonucleotides were less selective but more potent stimulators.
Figure 1.
Upregulation of co-stimulatory molecules and T-cell activation. Upregulation of CD80, CD86 and CD95 on malignant B cells from patients with CLL, MCL and on normal B cells with (A) lenalidomide or (B) CpG. CD69 upregulation on T cells in the same samples: (C) lenalidomide, (D) CpG. (E) correlation between B-cell and T-cell responses to lenalidomide: CLL (open squares), MCL (gray diamond), normal donors (black triangles).
In the presence of chronic lymphocytic leukemia cells, lenalidomide induces T-cell activation
To assess T-cell activation in response to lenalidomide, we measured expression of CD69 on T cells in the same PBMC samples used above for B cells (Figure 1C). In normal PBMC, we found only minimal T-cell activation (average increase in CD69 8±22%, p=0.1). In contrast, lenalidomide induced T-cell activation in CLL (average increase in CD69 95±95%, p=0.01). T cells from most patients with MCL (n=12) showed a weak response (average increase in CD69 25±27%, p=0.03). Consistent with the stronger effect of CpG oligonucleotides on B cells, we also found more pronounced T-cell activation in response to CpG than to lenalidomide (Figure 1D). T-cell activation in response to lenalidomide correlated strongly with CD80 upregulation on B cells (r=0.57, p=0.0001, Figure 1E) but not with upregulation of CD86 (r=0.1, p=0.53, data not shown).
Clinical presentation of immune stimulation in chronic lymphocytic leukemia: a potentially life-threatening cytokine release syndrome
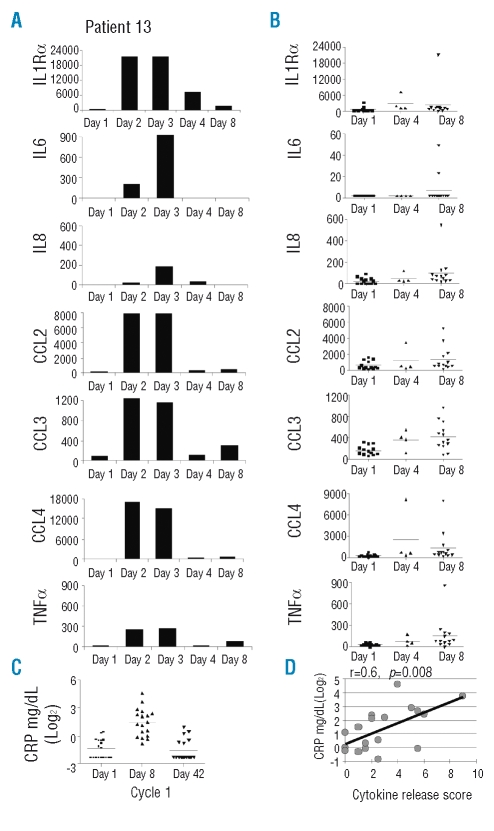
The unique side effects of lenalidomide in CLL appear to be related to immune activation. This is exemplified by patient 13, who 16 h after his first dose of 10 mg developed fatigue and back pain. On hospital admission he was febrile to 40.1°C, slightly hypotensive, without a rash or change in lymphadenopathy. Lenalidomide was withheld and intravenous hydration was started. Within hours the fever rose to 41°C and the patient developed hypotensive shock that was controlled with 8 L of intravenous fluids and methylprednisolone. There was no increase in white cell count, no evidence of tumor lysis syndrome and a work-up for infection was negative. All symptoms resolved over 3 days. This clinical presentation is reminiscent of the acute cytokine release syndrome experienced by CLL patients within hours of the first administration of rituximab16 or with the administration of oblimersen,17 a CpG antisense nucleotide. We, therefore, measured the cytokine serum levels in patient 13 during the acute presentation, finding a striking increase in inflammatory cytokines, most notably TNFα and IL6 (Figure 2A). We then measured cytokine levels in prospectively collected serum from day 8, cycle 1 in an additional 12 patients (Figure 2B). The levels of the cytokines and chemokines, IL8/CXCL8, TNFα, CCL2, CCL3, and CCL4 and the soluble receptor IL-1Rα showed statistically significant increases on day 8 compared to pretreatment levels (p <0.05, Figure 2B). IFNγ, IL-1α, IL-1β, IL-2, IL-4, IL-5, and IL-17 were below the limit of detection in all samples (Online Supplementary Table S2). An increase in cytokines was already apparent on day 4 in the four patients available for analysis, suggesting that cytokine release within the first few days of treatment is a general reaction to lenalidomide. This interpretation is consistent with the rapid onset of clinical symptoms (see below).
Figure 2.
Cytokine release in CLL patients treated with lenalidomide: (A) daily after the initiation of lenalidomide in patient L13 and (B) in patients pre-treatment (squares, n=13), on day 4 (triangle, n=4) and on day 8 (diamonds, n=13) of lenalidomide treatment. (C) CRP serum levels. (D) Correlation of CRP (maximal value week 1) and cytokine release score.
While patient 13 had an exceptionally severe presentation, similar symptoms were noted in the majority of patients. To standardize the clinical presentation of the cytokine release syndrome, we uniformly recorded and graded the main symptoms and findings in all patients (Table 2). Among the 18 patients enrolled in the study, the following symptoms were observed within 8 to 72 h (average 38 h) of starting lenalidomide: fatigue (72%), pain (61%), a greater than 25% increase in lymph node size and/or in absolute lymphocyte count (ALC) over baseline (50%), fever exceeding 38°C in the absence of signs of infection (44%), dehydration (39%), chills (33%) and a rise in creatinine (33%). Three out of 18 patients (17%) did not experience any of these side effects. In order to correlate the clinical presentation with laboratory and biological features we summarized the number and clinical grade of the individual manifestations into a score (Table 2). This score captures the degree of an inflammatory state induced by lenalidomide well, as shown by its close correlation with C-reactive protein (CRP) levels, a laboratory marker of inflammation that was rapidly induced in most patients (Figure 2C and D). Both the clinical score and CRP levels illustrate the considerable variability in lenalidomide-induced immune activation between different patients.
Table 2.
Adverse events during cycle 1 of lenalidomide therapy define a clinical cytokine release syndrome.
In vivo effects of lenalidomide on lymphocyte subsets in chronic lymphocytic leukemia
Recently, Ferrajoli and colleagues described that overall CD3 T-cell numbers remained stable at 3 and 6 months in 19 patients receiving continuous administration of lenalidomide.2 However, there are no reports on early changes during the time of the most pronounced clinical side effects and an analysis of lymphocyte subsets is not available. We, therefore, monitored B, T, and NK cell numbers during the first cycle of lenalidomide treatment (Figure 3). In the first week, almost all patients had a rapid reduction in circulating lymphocytes averaging 30–40% for B, T, and NK cells (p<0.01, Figure 3A). By the end of week 3 the normal lymphocyte subsets started to recover and after the 3-week period off drug had reached pre-treatment levels (Figure 3B). In contrast, the CLL cell count continued to decrease and by day 21, ten of 12 patients who completed 3 weeks of treatment had an average reduction of 62% (p<0.001). After the 3-week drug-free period the leukemic cell count exceeded pre-treatment levels in two patients, while the remaining patients maintained an average reduction of 38% (p=0.002).
Figure 3.
Effect of lenalidomide on lymphocytes: (A) percent change compared to pre-treatment cycle 1 day 8. Patient 11: no data. (B) Mean and standard deviation of cell counts. (C) CD3 T-cell content in lymph node biopsies pre-treatment (white columns) and on day 8 of cycle 1 (black columns). (D) CD3 staining in lymph node core biopsies.
The tender lymph node swelling commonly observed with lenalidomide in CLL could reflect recruitment of immune effector cells into tumor sites or cell proliferation. To address this issue, we obtained lymph node biopsies pre-treatment and on day 8 of cycle 1 of treatment in 11 patients. T cells (CD3) were counted in all biopsies and were slightly increased in two patients, while there was no change or even a slight decrease in nine patients (Figure 3C). CD56 and CD68 stained few NK cells and macrophages, respectively, but the numbers of these cells did not change in response to lenalidomide (data not shown). Collectively, our analysis of paired lymph node biopsies provides little evidence that tumor-infiltrating T cells are responsible for lymph node swelling in the context of flare reactions.
T-cell activation and leukemic cell clearance are distinct effects of lenalidomide
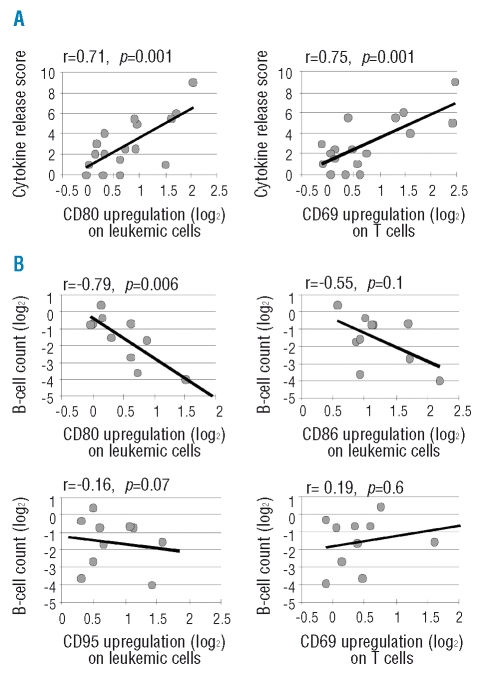
The severity of clinical side effects summarized in the cytokine release score correlated with the in vitro response to lenalidomide (Figure 4A) measured by upregulation of CD80 on CLL cells (r=0.71, p=0.001), and CD69 on T cells (r=0.75, p=0.001), and inversely with treatment-induced changes in T-cell numbers (r =−0.64; p=0.006, data not shown). There was no correlation between the clinical score and the decrease in leukemic cell count, age, RAI stage, number of prior therapies, renal impairment, or bulky disease. The average score in all patients was 2.89 (range, 0–9) with no difference between the 20 mg and the 10 mg cohort (mean scores of 2.95 and 2.81, respectively, p=0.9).
Figure 4.
Pearson’s correlation between the upregulation of cell surface markers in response to lenalidomide in vitro and clinical outcome: (A) for the cytokine release score (n=17), and (B) for peripheral blood response of patients measured as reduction in B-cell count on day 21 compared to pre-treatment (n=10).
Next we tested the association of clinical and biological characteristics with leukemic cell clearance. We found a strong correlation between the in vitro response of CLL B cells to lenalidomide, measured by CD80 upregulation, and the reduction in leukemic cells during the first cycle of treatment (Figure 4B). In contrast, upregulation of CD86 and CD95 was not correlated with treatment response. Metrics of immune activation, including CD69 upregulation on T cells (Figure 4B), the cytokine release score, individual cytokine serum levels, or CRP did not correlate with leukemic cell clearance in vivo, suggesting that T-cell activation and anti-leukemic activity of lenalidomide are separate effects.
Discussion
The clinical use of lenalidomide in CLL has been complicated by unexpected toxicities at doses that are well tolerated in multiple myeloma and aggressive non-Hodgkin’s lymphomas.4–7,17 A major concern in CLL has been the occurrence of tumor flare reactions, the etiology of which is not understood. Here, we provide an explanation for the exacerbated reaction to lenalidomide in CLL. We identify the tumor flare reaction as part of a cytokine release syndrome, which is the main immediate toxicity of lenalidomide in CLL patients, and which was severe enough to cause hospital admissions and/or dose interruptions in 28% of our patients during cycle 1. The 83% incidence of the cytokine release syndrome in our study was higher than the 30%2 to 58%1 described for the tumor flare reaction and was present in all our patients manifesting with flares. Consistent with clinical impressions, serum measurements confirmed the release of several pro-inflammatory cytokines and chemokines, including TNFα, IL-6, CCL2, CCL3 and CCL4. The source of these cytokines and the interactions leading to their release remain to be defined. CLL cells are an unlikely source for many of them, and in vitro studies failed to detect IL-6 or TNFα production by leukemic cells.4
The in vitro response to lenalidomide, measured by CD80 expression on CLL B cells and CD69 expression on T cells, was strongly correlated with the occurrence and severity of the cytokine release syndrome. The predictive value of these markers for severe clinical reactions will have to be validated in an independent cohort. Given the severity of side effects, it is conceivable that a test based on the in vitro response to lenalidomide could be used to stratify patients; such a strategy could reduce the need for low starting doses that is currently pursued. Our first ten patients started treatment with 20 mg daily before the dose was changed to 10 mg. This dose reduction has not had any noticeable impact on the cytokine release syndrome and two of the most severe reactions occurred in the 10 mg cohort. Because lenalidomide induces expression of co-stimulatory molecules on responsive cells over a wide concentration range,4 it is likely that doses much lower than 10 mg have to be used to avoid reactions in patients at risk.
Whether or not immune activation is required for the clinical activity of lenalidomide is controversial. In support is the observation that flare reactions may herald better clinical responses.3 However, other investigators have not made this observation.2 A recent report suggested that tumor-infiltrating immune cells may be responsible for the lymph node swelling and, by extension, for anti-tumor activity.4 In contrast, our analysis of matched lymph node biopsies obtained pre-treatment and on day 8 of cycle 1 from 11 patients, i.e. during the peak of the flare reaction, did not confirm increased T-cell infiltration. Differences between our study and that Andritsos et al. include the type of biopsies and their timing. Andritsos and colleagues compared a tonsil that had been surgically removed on day 28, a week after lenalidomide had been stopped, to a pre-treatment lymph node biopsy. Thus the observed differences in T-cell content could be related to the different tissues analyzed or to a rebound of T cells in the period off lenalidomide. Consistent with earlier reports,1 we observed a rapid reduction in ALC even in the first cycle. However, T-cell activation, the presence of a cytokine release syndrome or tumor flare reaction, and upregulation of FAS (CD95) on tumor cells did not correlate with the decrease in leukemic cells. Thus, we found no evidence to link T-cell activation or the occurrence of a cytokine release syndrome to clinical responses in CLL. Also, our finding that MCL tumor cells responded to lenalidomide in a manner similar to CLL cells but only induced minimal activation of autologous T cells in vitro is consistent with the notable absence of flare reactions in this disease. These observations, in light of a 50% response rate to lenalidomide in MCL, support the notion that anti-tumor effects may be independent of T-cell activation. The severity of the cytokine release syndrome in CLL appears to be determined by tumor characteristics, which raises the possibility of prospectively identifying patients at risk of severe reactions.
The main predictor of leukemic cell clearance, which reached an average of 62% by day 21, was lenalidomide-induced upregulation of CD80 on CLL cells in vitro (Figure 4B). Response data are currently not mature enough to test whether CD80 could serve as a predictor of overall response, and this should be addressed prospectively. It will also be interesting to test whether CD80 upregulation on tumor cells correlates with clinical responses in other B-cell malignancies. Whether CD80 expression is just a marker of unidentified biological effects of lenalidomide on CLL cell biology or whether it has a functional role in mediating leukemic cell clearance remains to be determined. In this regard it is interesting to note that CD80 is a signaling molecule that has been reported to transmit growth inhibitory and pro-apoptotic signals in B-cell lymphoma cells.18,19
While we cannot exclude that lenalidomide could have different mechanisms of action in different diseases, we favor the hypothesis that T-cell activation in CLL causes increased toxicity independently of anti-tumor effects. This provides a rationale to test the combination of lenalidomide with immunosuppressive agents such as glucocorticoids, purine analogs or alemtuzumab.
Acknowledgments
we are indebted to our patients, whose participation made this research possible. We thank the Department of Laboratory Medicine, Immunology Section, for lymphocyte phenotyping analyses, the Interventional Radiology team for conducting lymph node biopsies, and Ms Theresa Davies-Hill for processing clinical specimens.
Footnotes
The online version of this article contains a supplementary appendix.
Authorship and Disclosures
This work was presented in part at the annual American Society of Hematology meeting December 6th to 9th 2008 in San Francisco, CA, USA.
GA, NN, XT, SS, TH, JV, CB, WHW and AW were investigators of the study. GA, NN, BV, KK, FG, LS, JPM, and SP performed the laboratory work for this study. GA, NN, XT and AW participated in the statistical analysis. GA, JPM, and AW coordinated the research. GA, NN, XT, SP and AW wrote the manuscript. The authors report no potential conflicts of interest.
Funding: this work was supported by the NIH Intramural Research Program.
References
- 1.Chanan-Khan A, Miller KC, Musial L, Lawrence D, Padmanabhan S, Takeshita K, et al. Clinical efficacy of lenalidomide in patients with relapsed or refractory chronic lymphocytic leukemia: results of a phase II study. J Clin Oncol. 2006;24:5343–9. doi: 10.1200/JCO.2005.05.0401. [DOI] [PubMed] [Google Scholar]
- 2.Ferrajoli A, Lee BN, Schlette EJ, O’Brien SM, Gao H, Wen S, et al. Lenalidomide induces complete and partial remissions in patients with relapsed and refractory chronic lymphocytic leukemia. Blood. 2008;111:5291–7. doi: 10.1182/blood-2007-12-130120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chanan-Khan AA, Cheson BD. Lenalidomide for the treatment of B-cell malignancies. J Clin Oncol. 2008;26:1544–52. doi: 10.1200/JCO.2007.14.5367. [DOI] [PubMed] [Google Scholar]
- 4.Andritsos LA, Johnson AJ, Lozanski G, Blum W, Kefauver C, Awan F, et al. Higher doses of lenalidomide are associated with unacceptable toxicity including life-threatening tumor flare in patients with chronic lymphocytic leukemia. J Clin Oncol. 2008;26:2519–25. doi: 10.1200/JCO.2007.13.9709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moutouh-de Parseval LA, Weiss L, DeLap RJ, Knight RD, Zeldis JB. Tumor lysis syndrome/tumor flare reaction in lenalidomide-treated chronic lymphocytic leukemia. J Clin Oncol. 2007;25:5047. doi: 10.1200/JCO.2007.14.2141. [DOI] [PubMed] [Google Scholar]
- 6.Wiernik PH, Lossos IS, Tuscano JM, Justice G, Vose JM, Cole CE, et al. Lenalidomide monotherapy in relapsed or refractory aggressive non-Hodgkin’s lymphoma. J Clin Oncol. 2008;26:4952–7. doi: 10.1200/JCO.2007.15.3429. [DOI] [PubMed] [Google Scholar]
- 7.Richardson PG, Blood E, Mitsiades CS, Jagannath S, Zeldenrust SR, Alsina M, et al. A randomized phase 2 study of lenalidomide therapy for patients with relapsed or relapsed and refractory multiple myeloma. Blood. 2006;108:3458–64. doi: 10.1182/blood-2006-04-015909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chanan-Khan A, Porter CW. Immunomodulating drugs for chronic lymphocytic leukaemia. Lancet Oncol. 2006;7:480–8. doi: 10.1016/S1470-2045(06)70723-9. [DOI] [PubMed] [Google Scholar]
- 9.Corral LG, Haslett PA, Muller GW, Chen R, Wong LM, Ocampo CJ, et al. Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-α. J Immunol. 1999;163:380–6. [PubMed] [Google Scholar]
- 10.Davies FE, Raje N, Hideshima T, Lentsch S, Young G, Tai YT, et al. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood. 2001;98:210–6. doi: 10.1182/blood.v98.1.210. [DOI] [PubMed] [Google Scholar]
- 11.LeBlanc R, Hideshima T, Catley LP, Shringarpure R, Burger R, Mitsiades N, et al. Immunomodulatory drug costimulates T cells via the B7-CD28 pathway. Blood. 2004;103:1787–90. doi: 10.1182/blood-2003-02-0361. [DOI] [PubMed] [Google Scholar]
- 12.Ramsay AG, Johnson AJ, Lee AM, Gorgun G, Le Dieu R, Blum W, et al. Chronic lymphocytic leukemia T cells show impaired immunological synapse formation that can be reversed with an immunomodulating drug. J Clin Invest. 2008;118:2427–37. doi: 10.1172/JCI35017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jahrsdorfer B, Hartmann G, Racila E, Jackson W, Muhlenhoff L, Meinhardt G, et al. CpG DNA increases primary malignant B cell expression of costimulatory molecules and target antigens. J Leukoc Biol. 2001;69:81–8. [PubMed] [Google Scholar]
- 14.Leonard JP, Link BK, Emmanouilides C, Gregory SA, Wiesdorf D, Andrey J, et al. Phase I trial of toll-like receptor 9 agonist PF-3512676 with and following rituximab in patients with recurrent indolent and aggressive non Hodgkin’s lymphoma. Clin Cancer Res. 2007;13:6168–74. doi: 10.1158/1078-0432.CCR-07-0815. [DOI] [PubMed] [Google Scholar]
- 15.Jahrsdorfer B, Muhlenhoff L, Blackwell SE, Wagner M, Poeck H, Hartmann E, et al. B-cell lymphomas differ in their responsiveness to CpG oligodeoxynucleotides. Clin Cancer Res. 2005;11:1490–9. doi: 10.1158/1078-0432.CCR-04-1890. [DOI] [PubMed] [Google Scholar]
- 16.Byrd JC, Waselenko JK, Maneatis TJ, Murphy T, Ward FT, Monahan BP, et al. Rituximab therapy in hematologic malignancy patients with circulating blood tumor cells: association with increased infusion-related side effects and rapid blood tumor clearance. J Clin Oncol. 1999;17:791–5. doi: 10.1200/JCO.1999.17.3.791. [DOI] [PubMed] [Google Scholar]
- 17.O’Brien SM, Cunningham CC, Golenkov AK, Turkina AG, Novick SC, Rai KR. Phase I to II multicenter study of oblimersen sodium, a Bcl-2 antisense oligonucleotide, in patients with advanced chronic lymphocytic leukemia. J Clin Oncol. 2005;23:7697–702. doi: 10.1200/JCO.2005.02.4364. [DOI] [PubMed] [Google Scholar]
- 18.Suvas S, Singh V, Sahdev S, Vohra H, Agrewala JN. Distinct role of CD80 and CD86 in the regulation of the activation of B cell and B cell lymphoma. J Biol Chem. 2002;277:7765–75. doi: 10.1074/jbc.M105902200. [DOI] [PubMed] [Google Scholar]
- 19.Hirokawa M, Kuroki J, Kitabayashi A, Miura AB. Transmembrane signaling through CD80 (B7-1) induces growth arrest and cell spreading of human B lymphocytes accompanied by protein tyrosine phosphorylation. Immunol Lett. 1996;50:95–8. doi: 10.1016/0165-2478(96)02526-6. [DOI] [PubMed] [Google Scholar]