Summary
During the post-natal development, GFAP-expressing cells in the parenchyma progressively lose their neural stem cell (NSC) potential, whereas this peculiar attribute is preserved in GFAP-expressing cells of adult germinal zones. Although the relationship between astrocytes localized in the parenchyma and those in the germinal zones is elusive, many reports suggest that GFAP-expressing cells contained in germinal zones are maintained in immature developmental stage. Starting from the observation that the pan-astrocytic marker S100B is not expressed in the GFAP-expressing cells of adult germinal zones, we first investigated the relationship between S100B expression and the developmental status of these astrocytic cells. We demonstrated that long after the loss of RC2 and gain of GFAP, the onset of S100B expression characterizes a mature developmental stage in mouse telencephalic GFAP-expressing cells, both in vitro and in vivo. Using a transgenic s100b-EGFP mouse-derived culture model, we next demonstrate that in vitro, S100B expression in GFAP-expressing cells coincides with the loss of their NSC potential. Through a series of ectopic and orthotopic grafting experiments, we found that in the adult subventricular zone, S100B expression is controlled by environmental cues. Furthermore, we showed that treatment with epidermal growth factor represses S100B expression in GFAP-expressing cells in vitro as well as in mouse forebrain. Altogether, our results indicate that the S100B expression defines a late developmental stage after which GFAP-expressing cells lose their NSC potential.
Keywords: Animals; Animals, Newborn; Astrocytes; cytology; metabolism; Biological Markers; metabolism; Brain; cytology; growth & development; metabolism; Cell Communication; physiology; Cell Differentiation; drug effects; physiology; Cell Lineage; drug effects; physiology; Cells, Cultured; Corpus Striatum; cytology; growth & development; metabolism; Epidermal Growth Factor; pharmacology; Glial Fibrillary Acidic Protein; metabolism; Green Fluorescent Proteins; genetics; metabolism; Mice; Mice, Inbred C57BL; Mice, Transgenic; Nerve Growth Factors; metabolism; Neuroglia; cytology; drug effects; metabolism; Neurons; cytology; metabolism; S100 Proteins; metabolism; Spheroids, Cellular; Stem Cells; cytology; drug effects; metabolism
Keywords: neural stem cells, astrocytes, S100 proteins, SVZ, subventricular zone, adult neurogenesis, GFAP, EGF, maturation, microenvironment cues
Introduction
In the adult mammalian forebrain, neurogenesis is thought to persist throughout life in only two zones: the subventricular zone (SVZ) of the lateral ventricular wall, and the subgranular layer (SGL) of the dentate gyrus of the hippocampus (Alvarez-Buylla and Garcia-Verdugo, 2002; Palmer et al., 1999). The new neurons derive from adult neural stem cells (NSCs) (Gage, 2000). NSCs are capable of multilineage differentiation and unlimited self-renewal and have been considered as undifferentiated cells. However, this view has recently been challenged by the finding that, in the adult mammalian forebrain, the NSCs may also have phenotypical and ultrastructural characteristics of mature astrocytes including the presence of a light cytoplasm, gap junctions, glycogen granules and the intermediate filament glial fibrillary acidic protein (GFAP) (Ahn and Joyner, 2005; Doetsch et al., 1999; Filippov et al., 2003; Garcia et al., 2004; Seri et al., 2001). The astroglial nature of some NSCs is not surprising since radial glial cells (RGCs), their embryonic counterparts, have recently been demonstrated to fulfill neurogenic and gliogenic functions during the forebrain development (Anthony et al., 2004; Levitt and Rakic, 1980; Malatesta et al., 2003; Noctor et al., 2001; Voigt, 1989). During the postnatal stage, RG-derived cells progressively evolve into GFAP+ cells. In contrast to those emigrating into parenchyma, those remaining in close proximity to the ventricular surface conserve their multipotent abilities and locally evolve into adult neurogenic cells (Laywell et al., 2000; Merkle et al., 2004).
The adult neurogenesis is mainly controlled by different cues present in the germinal microenvironment (Garcion et al., 2001; Lim et al., 2000; Liu et al., 2005; Machold et al., 2003; Tropepe et al., 1997). This restricted environment might among others, also control the development of astrocytes, suggesting that multipotent GFAP-expressing cells in the adult forebrain refer to astrocytes in an immature but unidentified developmental stage (Bonaguidi et al., 2005; Garcia et al., 2004; Laywell et al., 2000; Steindler and Laywell, 2003).
A prerequisite step in categorizing immature cells within a continuum of differentiation includes finding specific molecular markers. In the case of GFAP-expressing cells, the S100B calciprotein could be such a marker since its expression correlates with the spatiotemporal maturation of the glial cells (Cicero et al., 1972; Deloulme et al., 2004; Ghandour et al., 1981). This hypothesis is strengthened by the recent observation that S100B is not expressed in the bipolar GFAP-expressing cells present in SGL and the SVZ of adult mouse brain (Deloulme et al., 2004; Filippov et al., 2003; Garcia et al., 2004). To determine whether the presence or the absence of S100B in GFAP-expressing cells is relevant to their maturation status and their NSC attributs, we have examined the expression pattern of S100B during the forebrain development. We show here, that S100B is a late marker of astrocyte development being expressed long after GFAP and characterizing a mature stage. By taking advantage of s100b-EGFP transgenic mice, we demonstrate that the onset of S100B expression in astrocytes is associated with the loss of their potential to form neurospheres. We next show that the adult subventricular environment and EGF repress S100B expression in vivo while maintaining their neurosphere forming potential in vitro. Altogether, our findings suggest that the onset of S100B expression defines a critical period in which GFAP-expressing cells acquire a more mature developmental stage and lose their neural stem cell potential.
Materials and Methods
Mice
s100b-EGFP (Vives et al., 2003) and β-actin-GFP transgenic mice (Okabe et al., 1997) were housed under standard laboratory conditions. C57/Bl6 mice were purchased from Charles River Laboratories (Arbresle, France). Experiments were performed according to the principles of laboratory animal care in compliance with European and French law. Mouse embryos and newborn mice (E16-P15) were anesthetized by hypothermia and killed by decapitation.
Primary astrocytic cultures
Hemispheres from whole forebrain of P2 newborns were separated, hippocampus, fimbria and septum were excluded and the meningeal membrane was removed. Lateral ventricular zone and cortex were carefully dissected out and treated separately. Tissues were dissociated by trypsinization and mechanical trituration in Hanks balanced saline solution containing 2.5 mg/ml trypsin (Invitrogen) and 1 μg/ml DNAse. Microglial cells were removed by preplating the cell suspension on non-coated Petri dishes for 15 min. The resulting cell suspension was then replated on non-coated Falcon Petri dishes in DMEM medium supplemented with 10% FBS. After 4 days in vitro (DIV), oligodendrocyte progenitor cells and neuroblasts growing on top of the developing astroglial layer, were selectively removed by flushing as previously described (Deloulme et al., 1990). At DIV 15 and DIV 30, non-astrocytic cell contamination was evaluated using O4 antibody, antibodies against PDGFR-α and MBP for oligodendroglial cells, β-tubulin III for neuronal cells, fibronectin (DAKO Cytomation) for fibroblastic cells and MOMA-2 monoclonal antibody (Chemicon) for macrophagic/monocytic cells. The percentage of non-astrocytic cells never exceeded 2.5% of the cells.
Immunohisto/cytochemistry
Cryostat brain tissue sections and cell preparations were fixed with 4% PFA as detailed previously (Deloulme et al., 2004). The following primary antibodies were used: mouse hybridoma supernatants RC2 and O4 (Developmental Studies Hybridoma Bank, University of Iowa), rat monoclonal antibody (mAb) anti-GFAP (1:500, USB biological), rat mAb anti PDGFR-α (1:100, Pharmingen), mouse mAb anti-S100B (1:1500, (Takahashi et al., 1999), mouse mAb anti-β-tubulin III (1:1000, Berkley company), rabbit polyclonal antibodies (pAb) anti-human S100B (1:700, DAKO Cytomation), rabbit pAb anti-Olig 2 (1:1000), rabbit pAb anti-proteoglycan NG2 (1:200, Chemicon), guinea pig pAb anti-GLAST (1:8000, Chemicon). The specificity of anti-S100B antibodies was controlled on S100B−/− brain sections as previously described (Deloulme et al., 2004). All sections and cell preparations were permeabilized and blocked in TBS containing 1 mM CaCl2, 0.2% Triton X-100 and 5% normal goat serum (NGS) for 25 min. Embryonic tissue preparations were further treated with Mouse-on-Mouse blocking IgG reagent as described by the manufacturer (VectorShield). Finally, the primary antibodies were applied overnight at 4°C followed an incubation of 45 min with an appropriate secondary antibodies conjugated with cyanin3, cyanin5 (Jackson immunoresearch Laboratories) or Alexa 488 (Molecular Probes Inc.). Nuclear counterstaining was performed with Hoechst 33258 (1 μg/ml) or To-pro3 (2 μM, Molecular Probes). Fluorescent images were obtained with a Zeiss fluorescent microscope (Axiovert 200M) or with a Leica (TCS SP2) confocal microscope.
Neurospheres from astrocytic cultures
7, 15 or 30 DIV primary astrocytic cultures from cortex and lateral ventricle SVZ were prepared as described above. Astrocytic cells were dissociated by incubation at 37°C with cell dissociation reagent (Sigma, C5782) for 20 min at 37°C and then trypsin was added to a final dilution of 1:100 (Gibco) for 2 min. Cells were gently triturated, pelletted for 5 min at 1000 rpm, resuspended in cell dissociation reagent, filtered through a 40 μm-mesh nylon cell strainer (Falcon). Dissociated cells were resuspended at a density of 5000 cells/ml in DMEM/F12 (Invitrogen) supplemented with B-27 complement (1:50, Invitrogen) and 10 ng/ml each of human recombinant EGF (Peprotech) and bFGF (our laboratory). Growth factors were added every second day. In this model, only astrocytic cells and GFAP-expressing cells are able to generate primary neurospheres (Imura et al., 2003; Laywell et al., 2000). After 12 DIV, spheres were plated on PLL-coated coverslips and either counted or induced to differentiate for 4 days by growth factor withdrawal and addition of N2 and B27 complements (1:100 and 1:200, respectively, Invitrogen) and 3% FBS.
FACS Sorting
15 and 30 DIV primary astrocytic cultures obtained from the forebrain of P2 newborn s100b-EGFP mice were prepared and dissociated as described above. Dissociated astrocytic cells were sorted using a MoFlo machine (Dako Cytomation). Electronic gates were defined using wild-type and GFP-astrocytic cultures derived from GFP transgenic mice. EGFP-positive and negative cells sorted from s100b-EGFP brain tissue were counted and resuspended in neurosphere medium. In some experiments, secondary astrocytic cultures were generated from sorted s100b-EGFP negative cells. s100b-EGFP negative cells were plated at high density (1.105 cells/cm2) on PLL-coated coverslips and treated every second day with 10 ng/ml EGF.
Astrocytic cell transplantation
SVZ astrocytic cultures derived from β-actin-GFP mice were dissociated at 15 DIV and then injected stereotaxically (1.104 cells/2 μl) into the striatum or the SVZ of adult (8-week old) C57/Bl6 hosts at the following coordinates (mm) anteroposterior (AP), mediolateral (ML) and dorsoventral (DV) relative to Bregma AP/ML/DV: 0.6/2/−3 or 0.6/1.1/−3 respectively. After 1 or 4 weeks, brains were removed after intracardial perfusion of 4% PFA and stained as described above. In some experiments, grafted mice have been given an i.v. injection of EGF (400 ng/40 μl PBS) every second day, for 4 weeks.
Western blot-analysis
The analysis of S100B, GFAP and α-tubulin in astrocytic cell extracts was carried out using 11% SDS-Tris-Tricine polyacrylamide gels as previously described (Deloulme et al., 2000). The commercial secondary antibodies used in Western blot were conjugated with horseradish peroxidase and proteins were visualized using ECL kit (NEN).
Cell counts and statistics
For counting, astrocytic cells were plated in 4-well Permanox slides and treated as described above. Quantitative analyses were performed by counting at least 500 cells per well. Two or 3 wells were counted for each combination of double or triple staining. The number of independent culture is indicated in legends.
In situ, the number of counted cells and the number of mice are indicated in figure legends. For each combination staining, the counting was performed on 2 to 5 slice preparations using a confocal microscope.
In neurosphere experiments, astrocytic cells were resuspended at a density of 5000 cells/ml of DMEM in 35 mm Falcon Petri dishes containing 2 ml culture medium supplemented as described above. After 12 DIV, neurospheres were sedimented, resuspended in 100 μl of DMEM and plated in 96 well Falcon micro-plates. Neurospheres were photographed and spheres larger than 50 μm were counted. Three dishes were plated for each point. The number of independent cultures is indicated in legends.
Statistical analysis of the raw data was performed by analysis of variance followed by Student’s t test using Excel software. The values are considered as significant when p is <0.05.
Results
In this study, we designated astrocytes, cells that express the GFAP.
S100B expression characterizes a mature developmental stage of astrocytic lineage
During astrogliogenesis, RGCs progressively lose the nestin epitope recognized by the RC2 monoclonal antibody and acquire GFAP staining and a stellate morphology. To determine when S100B expression appears during astrogliogenesis, we analyzed and quantified its expression from after birth until adulthood in different zones of the telencephalon including the striatum, the cortex and the corpus callosum (Fig. 1). At postnatal day 2 (P2), S100B is absent from RC2+ transforming RGCs which begin to express GFAP in their thin radial processes (Fig. 1A and B). As previously reported, (Deloulme et al., 2004; Hachem et al., 2005), most S100B+ cells present at P2 express the proteglycan NG2, the PDGF-α receptor and the Olig2 transcriptional factor and likely correspond to oligodendroglial cells (supplemental figure 1). At P8, the percentage of GFAP+ cells expressing S100B varies between 10 to 30 % depending on the region of the telencephalon (Fig. 1C and H). At this stage, differentiating astroglial cells harbor a more ramified morphology, a strong GFAP immunostaining, and progressively lose their RC2+ processes (Fig. 1D). By P14, RC2 staining has totally disappeared (data not show) and the astrocytic morphological maturation is almost completed. More than 67% at P14, and virtually all highly ramified GFAP+ cells in the adult parenchyma, express the S100B protein (Fig 1E–F and H). The late acquisition of S100B by multipolar astrocytic cells of the forebrain, long after the gain of GFAP and loss of RC2 staining, is also evident on Western blots of brain extracts (Fig. 1G). Together, these results indicate that in astrocytic cells, S100B expression characterizes a mature developmental stage.
Figure 1.
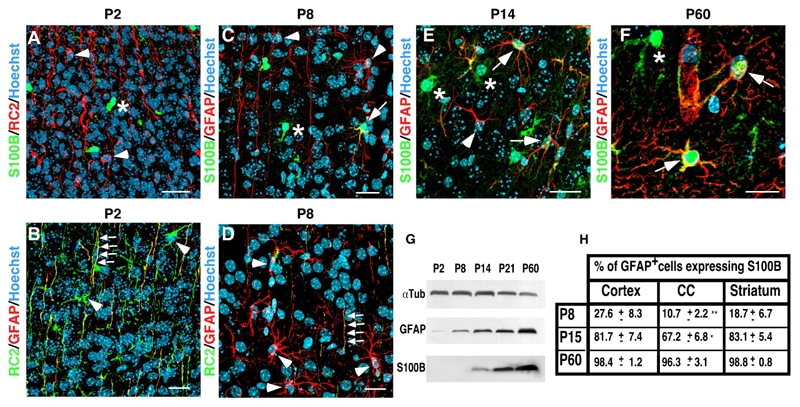
S100B is expressed long after the gain of GFAP and the loss of RC2 and characterizes a mature developmental stage of the astrocytic lineage. A–E, coronal sections of developing post-natal telencephalon of mice at P2, P8, P14 and P60 were subjected to double immunolabeling as indicated. A–B, at P2, RC2+ transforming RGCs do not yet express S100B (A, arrowheads) but start to express GFAP in their radial processes (B, arrows). In B, arrowheads point to RC2+ RGC bodies which are still negative for GFAP staining. C–D, at P7, RG transforming cells are more stellate and express GFAP in their cell bodies (C and D, arrowheads). Some of these cells start to express S100B (C, arrow). In D, arrows point to a radial process with residual RC2 staining. E, at P14, most of GFAP expressing cells express S100B (arrows). The arrow points to a GFAP+ cell that does not yet express S100B. F, at P60, all GFAP+ cells express S100B (arrows). In A, C, E and F, asterisks indicate S100B expressing cells that do not express RC2 or GFAP. These cells likely are oligodendroglial cells as indicated in supplemental figure 1. G, Western blot analysis of GFAP and S100B expression during the forebrain postnatal development. α-tubulin (αTub) was used as loading control. H, quantitative analysis of GFAP+ cells expressing S100B in different telencephal regions including the cortex, the corpus callosum (CC) and the striatum during postnatal development. 3 mice were analyzed at P8 and P14 and 5 at P60. Errors are ± SD. ** p<0.001, * p<0,05. Scale bars: 20 μm.
GFAP-expressing cells never express S100B protein in adult germinal zones
In contrast to parenchymal astrocytes, we found that astrocytes of the germinal zones do not express S100B. This is true from the onset of postnatal development (Fig 2A–C) until adulthood (Fig. 2D and E), whereas the level of S100B expression progressively increases with time in ependymocytes (Fig 2B and C). Because of their bipolar morphology, the lack of S100B coexpression in GFAP+ astrocytes of adult germinal zones could be interpreted as these cells being locked in an immature developmental stage. However, one could not exclude the possibility that these GFAP-expressing cells constitute a specific subpopulation of cells unable to express S100B.
Figure 2.
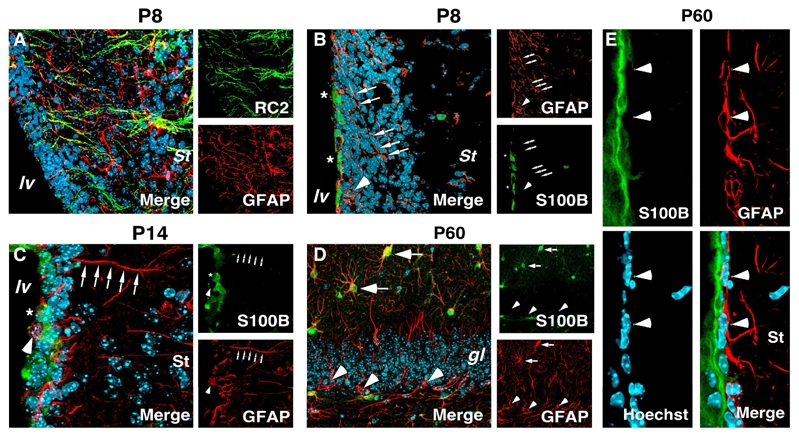
S100B expression pattern in developing and adult germinal zones A–E, coronal sections of developing and adult SVZ (A–C and E) and adult hippocampus (D) were subjected to double immunolabeling as indicated. A, at P8, RC2+ RG transforming cells express GFAP in radial processes. B–C, cell bodies (arrowhead) and radial process (arrows) of transforming astrocytic cells at P7 (B) and P14 (C) express GFAP but are devoid of S100B staining. In developing SVZ, S100B is expressed in ependymocytes (asterisks). D, in adult dentate gyrus, radial GFAP+ cells of the subgranular layer never express S100B (arrowheads). Arrows point to stellate GFAP+/S100B+ cells. E, in adult SVZ, S100B expression is restricted to ependymal cells. The subependymal GFAP+ cells with bipolar morphology are devoid of S100B staining (arrowheads). lv, lateral ventricle; St, striatum; gl, granular layer. Scale bars: 20 μm.
Subventricular GFAP-expressing cells have the potential to mature into S100B+ astrocytes in vitro
To test whether the S100B expression onset correlates with terminal development of GFAP-expressing cells or whether GFAP+ cells of SVZ constitute a specific subpopulation of astrocytic cells that are intrinsically unable to express S100B, we investigated their behavior in culture. To this end, we compared the pattern of S100B and other astroglial markers in astrocytic monolayer cultures derived from the cortex or the lateral ventricular wall of P2 mice. After 7 days in culture (DIV 7), the persistence of immature astroglial characteristics in SVZ astrocytes was evidenced by the high expression of embryonic or astroglial progenitor markers such as RC2 (Fig. 3A), vimentin, GLAST or nestin (data not shown). The same was found with whole cortex-derived cultures (data not shown). At this stage, only 20% of GFAP+ cells express S100B (Fig 3C). Then, a phenotypical transition occurs in both types of culture that is accompanied by a progressive and drastic down regulation of RC2 (Fig. 3B and E) and nestin expression (data not shown), the persistence of GLAST expression (data not shown) and the generalization of GFAP and S100B expression between DIV 15 and DIV 30 (Fig. 3D and E). At 30 DIV, more than 90% of GFAP-expressing cells express S100B (Fig. 3D). There is no significant difference in the percentages of S100B+, GFAP+ or RC2+ cells in astrocytic cultures derived from the SVZ or the entire cortex after 30 DIV (Fig. 3E). The time course of GFAP and S100B expression analyzed by Western blot confirmed that S100B is detected later than GFAP in crude protein extracts of astrocytic cultures (Fig. 3F).
Figure 3.
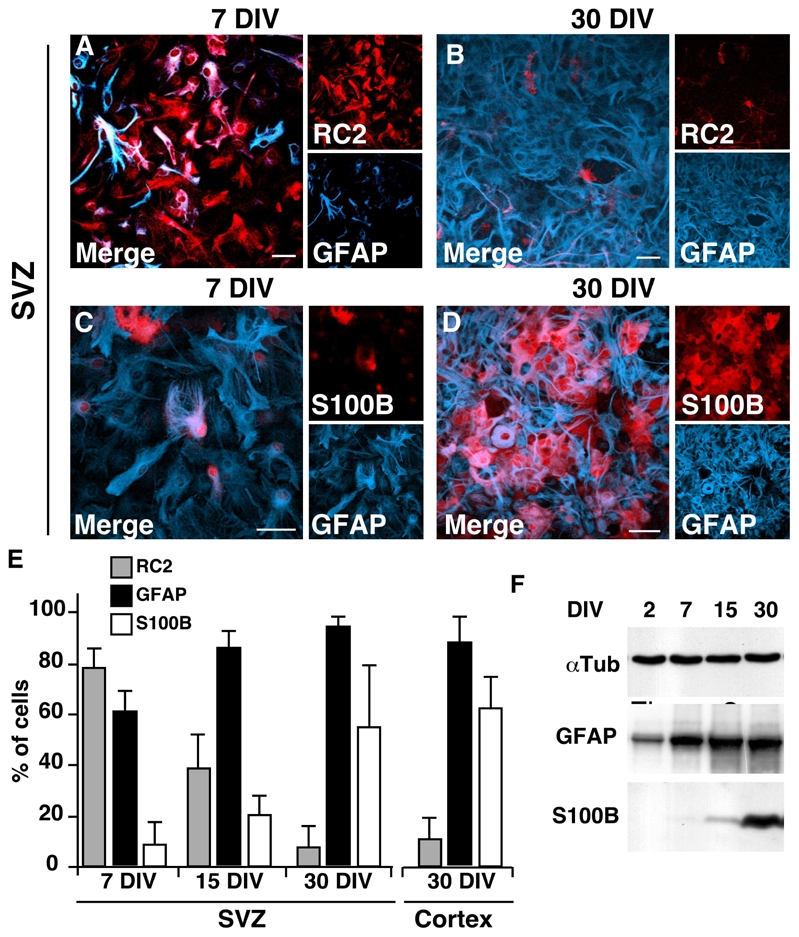
Cultured subventricular and cortical astrocytes mature into S100B+ cells. Astrocytic cultures derived from the SVZ (A–D) and the neocortex (not shown) of newborn mice were immunolabeled at 7, 15 and 30 DIV as indicated. The percentages of RC2+, GFAP+ and S100B+ cells in the SVZ and the cortical cultures are represented in E. Western blot analysis of GFAP and S100B expression during astrocytic development in culture is presented in F. α-tubulin (αTub) was used as loading control. Each bar represents the average of 2 to 4 independent cultures. Errors are ± SD. DIV, days in vitro. Scale bars: 20 μm.
These results first indicate that the succession of the various astrocytic developmental stages observed in vitro recapitulates that seen in vivo. S100B appears in GFAP-expressing cells and its onset of expression correlates with terminal astrocytic developmental stage. Second, these results indicate that, provided they are removed from their original SVZ microenvironment, perinatal SVZ astrocytic cells can complete the same program of differentiation along the astroglial lineage as their parenchymal counterparts.
S100B− astrocytic cells are multipotent whereas S100B+ astrocytes are unipotent
To determine whether the S100B expression onset correlates with the lost of their NSC potential, we took advantage of transgenic mice expressing EGFP under the control of the s100b promoter (Vives et al., 2003). Since S100B expression in brain was not restricted to astrocytic lineage (Deloulme et al., 2004; Vives et al., 2003) and supplemental Figure 1), we generated primary cortical astrocytic cultures from P2 s100b-EGFP newborn mice. As expected, the number of EGFP+ cells increased with time spent in culture (Fig. 4A). We then ensured that variations in the level of EGFP expression faithfully reflected variations in the level of the endogenous S100B protein. At 15 and 30 DIV, approximately 15% and 60% of the cells (15%±6 and 57%± 8, n=2 independent cultures) expressed EGFP respectively, and virtually all EGFP+ cells expressed endogenous S100B (Fig. 4B). We then sorted EGFP− from EGFP+ astrocytic cells by flow cytometry after 15 or 30 DIV and tested their capacity to form multipotent self-renewing neurospheres in vitro. As expected, both cell populations were GLAST+ and GFAP+ (Fig. 4D and E) and most of EGFP− sorted cells expressed RC2 (Fig. 4C). When placed in neurosphere culture conditions, EGFP− astrocytic cells generated numerous spheres devoid of EGFP expression (Fig. 4G–G1) and capable of generating secondary self-renewing neurospheres (data not shown). In differentiating medium, neurospheres from EGFP− astrocytes generated O4+ oligodendroglial cells, β-tubulin III+ neuroblasts and GFAP+ astrocytic cells, attesting to their in vitro multipotentiality (Fig. 4I, J). In contrast to EGFP− cells, EGFP+ sorted cells generated only a few, small EGFP+ spheres (Fig. 4H–H1), with no self-renewal potential (data not shown). Most of these spheres were unipotent and gave rise only to GFAP+ astrocytes under differentiating conditions (Fig. 4K). Interestingly, an equivalent amount of neurospheres was generated from EGFP− cells derived from either 15 or 30 DIV cultures (Fig. 4F). This indicates that “old” cultured astrocytes have the same differentiation potential as “young” astrocytes provided that they are in a S100Bimmature state. The converse is true with EGFP+ mature astrocytes lacking the ability to generate multipotent neurospheres regardless of the time spent in culture (Fig. 4F). Interestingly, after 4 days spent in differentiating medium, neurospheres derived from EGFP− astrocytic cells gave rise to EGFP+/GFAP+ astrocytes and EGFP+/O4+ oligodendrocyte progenitors (Fig. 4I and J). Furthermore, when sorted EGFP− cells were grown straight away in astroglial differentiating medium, the level of EGFP expression progressively increased with time (compare Fig. 6A to Fig. 6C). These results obtained in vitro confirm that EGFP− cells are genuine astrocytes capable of progressing toward a higher level of maturation. In addition, EGFP+ derived neurospheres gave rise to cells that continuously expressed EGFP in the presence of EGF/bFGF, even after 12 DIV (Fig. 4H–H1). This strongly suggests that the growth factors, at least in our paradigm, cannot induce the down-regulation of S100B expression nor de novo acquisition of a multipotent stem/progenitor cell fate by mature astrocytes in culture. Taken together, these results show that the onset of S100B expression defines a critical period after which the multipotent state of murine astrocytic cells is irreversibly lost.
Figure 4.
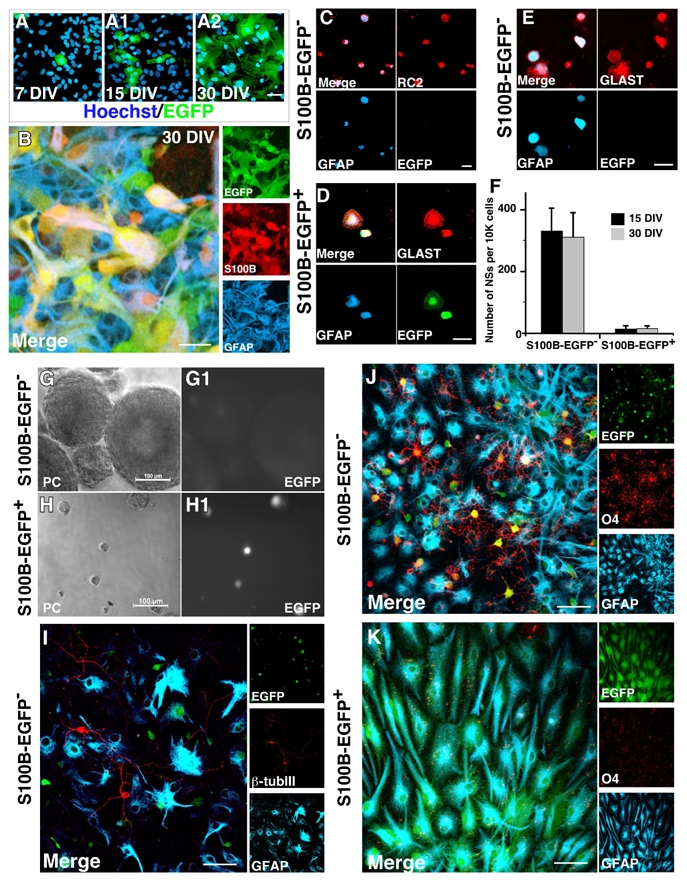
In astrocytes, activation of s100b gene is linked to the loss of NSC potential. A and B, cultured astrocytic cells were obtained from whole forebrains of newborn transgenic s100b-EGFP mice and were characterized at 7, 15 and 30 DIV as indicated. A–A2, EGFP expression increases with the time spent in culture. B, at 30 DIV, nearly all EGFP+ cells express both endogenous S100B and GFAP. C–E, EGFP+ and EGFP− cells derived from 15 and 30 DIV cultures were sorted by FACS and double immunolabeled as indicated. Most sorted EGFP− cells are in a RC2+/GFAP− or a RC2+/GFAP+ developmental stage, all of them express GLAST cells (C,E), while EGFP+ cells are GFAP+/GLAST+ cells (D). F–H, unlike sorted EGFP− cells, most sorted EGFP+ cells are unable to generate neurospheres (NSs) with a size higher than 50 μm (H). EGFP expression is maintained in small NSs derived from sorted EGFP+ cells even after 12 days of EGF/bFGF treatment (H1). In F, each bar represents the average of 4 independent cultures, each counted in triplicate. Errors are ± SD. I–K, NSs derived from EGFP− cells are multipotent and give rise to β-tubulin III+ neuroblasts (I), O4+ oligodendroglial cells (J) and GFAP+ astrocytic cells (I and J), whereas the small NSs derived from sorted EGFP+ cells are mostly unipotent and give rise to GFAP+ astrocytes (K). A and G, Zeiss conventional microscopy. B–F and H–J, stack of 5 to 8 optical sections captured with Leica confocal microscope. Scale bars: 20 μm in A–E and H–J; 100 μm in G.
Figure 6.
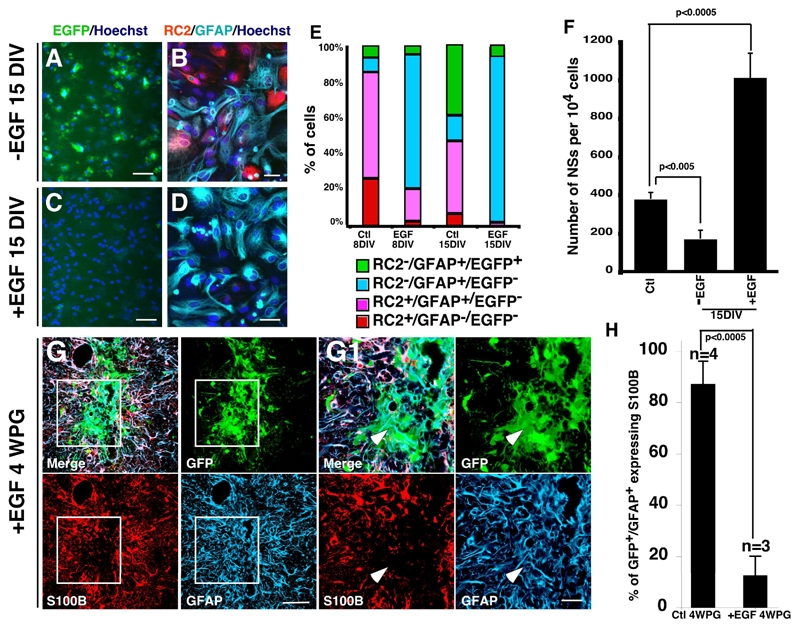
EGF prevents the activation of the s100b gene and maintains astrocytic cells in S100B− immature state in vitro and in vivo. A–D, sorted s100b-EGFP− cells (EGFP−) from 15 DIV primary astrocytic cultures were replated in medium with serum in the absence (−EGF) or in the presence of EGF (+EGF), and double-immununolabeled for the presence of RC2 and GFAP at 8 DIV and 15 DIV as indicated. EGF treatment stimulated the transition from RC2+/GFAP−/EGFP− and RC2+/GFAP+/EGFP− stages to RC2−/GFAP+/EGFP− stage and maintained the cells in this stage (C–D). E, percentages of different cell populations. F, EGF treatment increases the number of NSs generated from cultured astrocytic cells after 15 DIV. NSs are generated from 15 DIV secondary astrocytic culture in the absence (−EGF) or in the presence of EGF (+EGF) and from sorted s100b-EGFP− cells (Ctl) derived from 15 DIV primary culture. Each bar represents the average of 3 independent cultures, each counted in triplicate. Errors are ± SD, Scale bars are 50 μm in A and C and 20 μm in B and D. G–G1, 15 DIV cultured astrocytic cells derived from transgenic newborn mice expressing GFP under the ubiquitous β-actin promoter were grafted stereotaxically in the striatum of adult mice as described in figure 4. Multiple i.v. injections of EGF (400 ng/40 μl every second day for 4 weeks) prevent the expression of S100B in GFP+/GFAP+ grafted cells compared to control mice (see fig. 4B–B1). H, Quantitative analysis of GFP+/GAFP+ cells expressing S100B. At least 400 GFP+/GFAP+ cells were counted and n the number of transplanted mice. Errors are ± SD. Scale bars: 50 μm in G and 20 μm in G1.
The adult SVZ microenvironment represses the S100B expression in transplanted astrocytic cells
To determine the role of microenvironmental cues in controlling the expression of S100B in vivo, primary astrocytic cells obtained from the SVZ of β-actin-GFP mice (Okabe et al., 1997) were grafted into the adult striatum or the SVZ of C57/BL6 hosts after 15 DIV (Fig. 5A–B). One week after striatal grafting, 17% (SD ±6%, n=2 mice) of GFP+/GFAP+ cells expressed S100B (Fig. 5A–A1). This value increased to 87% (SD±9.5%, n=4 mice) three weeks later (Fig. 5A–A1). In addition, many of these astrocytes were highly ramified, completely integrated into the host brain and contacted blood vessels, confirming the functional maturation of these cells (supplemental Figure 5). Furthermore, 3 weeks after transplantation into the striatum, FACS sorted s100b-EGFP− became EGFP+ and contacted blood vessel (data not shown). In contrast, when β-actin-GFP astrocytic cells were orthotopically grafted into the SVZ, only 30% of GFAP+/GFP+ cells (28±11%, n=3 mice, p<0.0005) became S100B+ 4 weeks after implantation (Fig. 5C–C1) and they were mainly localized in the striatal part of the graft (Fig. 5C1, arrowheads).
Figure 5.
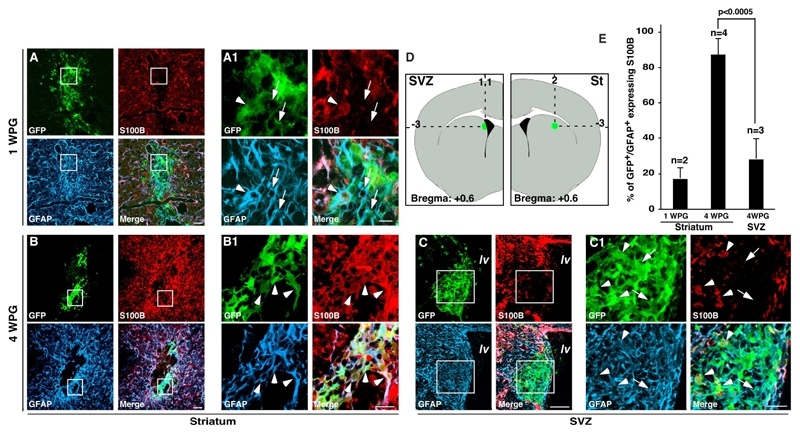
Maturation of grafted astrocytic cells is controlled by micro-environmental factors. A–C, 15 DIV cultured astrocytic cells derived from transgenic newborn mice expressing GFP under the ubiquitous β-actin promoter were grafted stereotaxically in the striatum (A–A1, B–B1) or in the SVZ (C–C1) of adult mice as indicated in D. GFP+ cells grafted in the striatum progressively expressed S100B after 1 and 4 weeks (A–A1 and B–B1 respectively) whereas most of GFAP+/GFP+ cells grafted in SVZ were maintained in S100B− immature state even after 4 weeks (C–C1). Arrows and arrowheads point to GFP+/GFAP+/S100B+ and GFP+/GFAP+/S100B− respectively. E, Quantitative analysis of GFP+/GAFP+ expressing S100B. At least 400 GFP+/GFAP+ cells were counted and n is the number of transplanted mice. Errors are ± SD. lv, lateral ventricle; St, striatum, WPG, Week Post Grafting. Scale bars are 50 μm in A, B and C and 20 μm in A1, B1 and C1.
Together, these observations confirm that SVZ astrocytic cells have the potential to mature towards fully differentiated S100B+ astrocytes. They also clearly demonstrate the major role played by the adult SVZ microenvironment in preventing the onset of S100B expression and differentiation along the astrocyte lineage.
EGF blocks the S100B expression in GFAP-expressing cells and enhances their multipotent abilities in vitro
Many reports have shown that the EGF receptor (EGF-R) signaling pathway, activated by EGF and/or TGF-α, stimulates astrocyte development in vitro and in vivo (Burrows et al., 1997; Kornblum et al., 1998; Sibilia et al., 1998). We therefore wondered whether EGF could be part of the microenvironemental factors acting on S100B expression and astrocyte development. To this end, we first generated, from the SVZ of s100b-EGFP mice, 15 DIV astrocytic primary cultures. Then EGFP− cells were sorted by flow cytometry, and platted at high density in astrocytic secondary culture medium. After 8 and 15 DIV in the absence or presence of EGF, we analyzed and quantified the respective proportions of RC2, GFAP and EGFP positive cells (Fig. 6A–E). As expected, culturing the cells in the absence of EGF for 8–15 days substantially increased the proportion of mature RC2−/GFAP+/EGFP+ (11±2% and 42±5%, respectively, n=3 independent cultures) and this was done mainly at the expense of immature RC2+ populations (i.e. RC2+/GFAP−/EGFP− and RC2+/GFAP+/EFGP− cells) (Fig. 6E). In contrast, in the presence of EGF, only few RC2−/GFAP+/EGFP+ cells could be detected after 8 and 15 DIV (4.6±0.9% and 5.6±1.1 respectively, n=4 independent cultures), whereas the RC2−/GFAP+/EGFP− population was drastically expanded after 8 and 15 days of treatment (Fig. 6E). This expansion was done at the expense of RC2+ populations which nearly disappeared at 15 DIV (3,8±1%, n= 4) (Fig. 6B and C). It is important to note that the repression of S100B expression is reversible. Indeed, and in agreement with our neurospheres differentiation data (Fig. 4I and J), EGF withdrawal induces a progressive expression of EGFP in GFAP expressing cells (data not show). Together, these results show the dual effect of EGF signaling on the maturation of astrocytic cells. It favors the RC2+/GFAP− to RC2−/GFAP+ transition, but it also blocks the progression to the mature GFAP+/S100B+ stage.
We next asked whether the arrest of astrocytic cells in a RC2−/GFAP+/EGFP− stage by EGF influence their neurosphere neurosphere-forming capacity. We compared the percentages of multipotent neurosphere-forming cells immediately following the sorting, or after 15 days of culture in medium supplemented or not with EGF (Fig. 6F). Immediately after sorting, 3.7% of EGFP− cells generated multipotent neurospheres NSs (373±38 NSs per 104 cells). The neurosphere-forming capacity dropped by 50% when sorted EGFP− cells were cultured for 15 days in the absence of EGF (166±47 NSs per 104 cells, P<0,005). By contrast, after 15 days of EGF pretreatment the neurosphere-forming capacity was tripled (1005±131 NSs per 104 cells, P<0.0005). Hence, it appears that a chronic treatment of astrocytic cells by EGF stimulates the progression from the RC2+/GFAP− to the RC2−/GFAP+ stage, but arrests GFAP-expressing cells in an S100B− immature stage. In addition to maintaining cells in an immature state, the 15 DIV pretreatment with EGF also strongly increases the multipotent neurosphere-forming capacity of sorted EGFP− astrocytic cells.
Circulating EGF had previously been shown to pass easily through the blood brain barrier and to diffuse into the host brain (Pan and Kastin, 1999). We then wondered if EGF could modulate S100B expression and astrocyte maturation in vivo. Astrocytic cells grown in secondary culture obtain from β-actin-GFP mouse brain were grafted into the striatum. After 4 weeks, 87% (SD±9.5%, n=4, p<0,0005) of GFP+/GFAP+ cells expressed S100B (Fig. 4B–B1 and Fig. 6H). EGF treatment reduced the amount of S100B+ cells to 12% (SD±8%, n=3) (Fig. 6G–H). This indicates, that as in vitro, EGF has the potential to prevent the evolution of astrocytic cells to an S100B-expressing stage in vivo. Taken together, these results suggest that EGF is a microenvironmental factor which maintains the SVZ GFAP-expressing cells into the immature S100B-developmental stage.
Discussion
The aim of the present study was to determine whether the heterogeneity of S100B expression within the GFAP expressing population might be related to their developmental status and their NSC potential. Our results can be summarized as follows. First, the onset of S100B expression in GFAP expressing astrocytic cells defines a terminal developmental stage in vivo and in vitro. Second, cultured astrocytic cells lose their NSC abilities when they express S100B. Third, expression of S100B in transplanted astrocytic cells is controlled by adult microenvironmental cues. Forth, chronic exposure to EGF maintains astrocytic cells in a S100B− immature state, both in vitro and in vivo. Our data collectively support the idea that in adult germinal zones of the mammalian brain, GFAP-expressing astrocytes are maintained in an immature S100B− developmental stage through the action of microenvironmental cues.
In the developing mouse brain, S100B expression defines a new mature developmental stage and discriminates astrocytes present in adult germinal zones from parenchymal astrocytes
During neocortical gliogenesis, astrocytic cells derived from RC2+ radial glia progressively lose their elongated aspect to become highly ramified GFAP+ astrocytes. The acquisition of GFAP together with a complex morphology has led researchers in the field to consider all GFAP-expressing cells as mature astrocytes. However, this view has recently been challenged by the discovery that some GFAP+ cells located in adult germinal zones are neural stem cells (Doetsch et al., 1999; Garcia et al., 2004; Laywell et al., 2000; Seri et al., 2001) displaying features of fully differentiated astrocytes like GFAP expression, and immature characteristics like bipolarity (Garcia et al., 2004). In this study, we show that unlike GFAP immunochemistry which does not allow to clearly determine the number of processes of a given cell, and thus its maturation state, S100B reliably defines a late stage of GFAP+ cell maturation. Based on the combined use of S100B, GFAP and RC2, one can thus distinguish three successive stages of murine astrocytic development (Fig. 7). The RC2+/GFAP+ stage (Stage I) coincides with the beginning of astrogliogenesis near the wall of the lateral ventricle and in the neocortex. The second stage (Stage II) correlates with the lost of RC2 staining in transforming RGCs. Finally, the third stage (Stage III) corresponds to the onset of S100B expression in neocortical GFAP+ astrocytes. This stage correlates with their terminal morphological maturation and defines the more mature state of the neocortical astrocytes in vivo as well as in vitro. However, one should be aware that this characterization might not hold true for every species. Indeed, our preliminary results showed that the spatiotemporal pattern of forebrain S100B expression differs between mouse and rat forebrains in both the astrocytic and oligodendroglial lineages (ER and JCD, unpublished data). In agreement with a recent report describing the post-natal development of rat and mouse SVZ (Peretto et al., 2005), we found that the onset of S100B expression occurs before the onset of GFAP expression in the astrocytic lineage of rat telencephalon (our unpublished results). Therefore, in adult and developing rat brain, S100B does not discriminate immature from mature GFAP-expressing cells and might explain the presence of S100B in GFAP-expressing cell of adult rat germinal zones (Namba et al., 2005) and our unpublished data).
Figure 7.
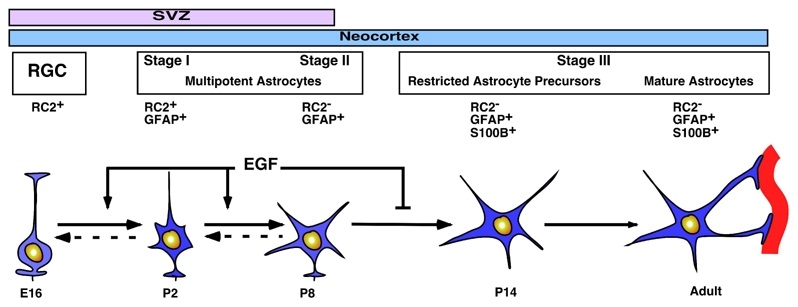
Model of GFAP+ astrocytic development. The onset of GFAP expression in parallel with the progressive down-regulation of RC2 characterizes the stage I and the stage II respectively. Next, GFAP+ astrocytes continue their developmental program and the onset of S100B expression defines the stage III. This stage includes restricted astrocyte precursors and terminally differentiated astrocytes. EGF signaling exerts a double effect on GFAP+ astrocytic development. It activates the transition from stage I to the stage II and blocks the transition from stage II to stage III.
In the adult mouse germinal zones, GFAP+ cells are S100B−. These cells have the potential to become S100B+ when cultured. This is in agreement with previous studies suggesting that germinal GFAP+ cells belong to the astroglial lineage. Moreover, it strongly suggests that the main differences (functional and phenotypical) between germinal zone and parenchymal astrocytes relate to their developmental stages rather than to their belonging to different astrocytic subtypes. However, we cannot exclude the existence in vivo of a subpopulation of GFAP-expressing cells harboring multiple differentiation traits except S100B expression.
Astrocytic S100B expression and neural stem cell potentials
The results of our in vitro investigations support a recent model proposing that the degree of maturation of germinal zone astrocytes is lower than that of their parenchymal counterparts (Bonaguidi et al., 2005; Garcia et al., 2004; Laywell et al., 2000). Astrocytic cultures neonatally derived from the SVZ or the neocortex conserve their stem/progenitor cell potential as long as they remain S100B−, and lose it when they are S100B+ (Fig. 4). Furthermore, the expression pattern of S100B in developing neocortical astrocytic lineage (Fig. 1) coincides with the loss of their multipotentiality which occurs during the second postnatal week (Laywell et al., 2000).
Although we have demonstrated that the multipotentiality is lost when astrocytes reach the S100B+ stage III, our results do not allow us to assign a specific pattern of molecular markers stage to multipotent astrocytes. It is important to note that the time spent in culture does not impact on the percentage of multipotent neurospheres generated from EGFP−/S100B− sorted cells (Fig. 4). This percentage, ranging between 2.5 to 4%, is low and similar to that reported previously (Garcia et al., 2004; Laywell et al., 2000). Altogether, these observations suggest that remaining in an immature S100B− state is a sine qua non condition for GFAP-expressing cells to fulfill their stem/progenitor cell function.
Nevertheless, in s100b knock-out mice (Xiong et al., 2000), we were unable to observe morphological or functional differences between SVZ and parenchymal astrocytes (ER and JCD, unpublished data). This suggests that S100B may not control stem/progenitor attributes of GFAP expressing cells. However, we and others have shown that S100B can form heterodimers with S100A1 and S100A6, two isoformes expressed in GFAP-expressing cells of the SVZ (Deloulme et al., 2000; Yamashita et al., 1997; Yang et al., 1999). Therefore, functional redundancy or compensatory mechanisms might explain the lack of obvious phenotype in GFAP expressing cells of s100b null mice.
Environmental cues and astrocytic development
Adult SVZ astrocytes reside in a particular and complex microenvironment called “niche” which regulates inherent NSC features (Doetsch, 2003; Spradling et al., 2001). Vasculogenesis, multiple diffusible signaling factors (TGFα, BMP2, Noggin, GABA, Shh), components of the extracellular matrix (Tenacin-C) are some of the actors present in these “niches” and implicated in self-renewal and differentiation of adult NSCs (Garcion et al., 2001; Lim et al., 2000; Liu et al., 2005; Tropepe et al., 1997). We have shown that, whereas cultured SVZ astrocytes have the potential to mature into S100B+ cells, they are arrested at developmental stage II in vivo (i.e. RC2−/GFAP+/S100B−) (Fig. 5). Furthermore, grafting experiments show that in contrast to the parenchyma that allows maturation of S100B− into S100B+ astrocytes, the SVZ environment prevents it. We have shown that EGF treatment blocks astrocytes at stage II and increases their capacity to generate multipotent neurospheres in culture. When injected in vivo, EGF also prevents the onset of S100B expression in grafted β-actin-GFP and s100b-EGFP astrocytic cells (Fig. 6 and data not shown).
The physiological relevance of these results is validated by the fact that in the adult SVZ, the EGF-R is expressed in dividing progenitors as well as in some astrocytes (Doetsch et al., 2002) and EGF-R signaling has been implicated in astrocytic development and in the control of adult neurogenesis (Burrows et al., 1997; Kornblum et al., 1998; Sibilia et al., 1998). Although exogenously added EGF acts to maintain astrocytes in a relatively immature state, it probably is not the sole factor implicated in this process in vivo (Bonaguidi et al., 2005). we can however propose that EGF-R signaling is a key element of the SVZ niches allowing GFAP-expressing cells to remain in developmental stage II which is a necessary condition for the adequate functioning of this germinal zone.
Supplementary Material
Acknowledgments
We thank N. Bertacchi and A. Schweitzer for excellent technical assistance; N. Chaumontel, and S. Fremy for animal care; A. Depaulis for stereotaxic expertise, J. P. Onave for chirurgical materials, V. Collin and D. Grunwald from DRDC for assistance in Moflo sorting, and confocal scanning microscopy, respectively; J. Ciesielski-Treska; H. Chneiweiss, and M. Takahashi for the gift of anti-vimentin, antiolig 2 and S16 antibodies, respectively; the antibody against RC2 has been obtained from the Developmental Studies Hybridoma Bank (Iowa DSHB). We gratefully thank L. Aubry, C. Benaud and G. Labourdette for their critical reading and writing of the manuscript. E.R. obtained a fellowship from the Commissariat à l’Energie Atomique. The authors declare that they have no competing financial interests.
References
- Ahn S, Joyner AL. In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature. 2005;437:894–7. doi: 10.1038/nature03994. [DOI] [PubMed] [Google Scholar]
- Alvarez-Buylla A, Garcia-Verdugo JM. Neurogenesis in adult subventricular zone. J Neurosci. 2002;22:629–34. doi: 10.1523/JNEUROSCI.22-03-00629.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anthony TE, Klein C, Fishell G, Heintz N. Radial glia serve as neuronal progenitors in all regions of the central nervous system. Neuron. 2004;41:881–90. doi: 10.1016/s0896-6273(04)00140-0. [DOI] [PubMed] [Google Scholar]
- Bonaguidi MA, McGuire T, Hu M, Kan L, Samanta J, Kessler JA. LIF and BMP signaling generate separate and discrete types of GFAP-expressing cells. Development. 2005;132:5503–14. doi: 10.1242/dev.02166. [DOI] [PubMed] [Google Scholar]
- Burrows RC, Wancio D, Levitt P, Lillien L. Response diversity and the timing of progenitor cell maturation are regulated by developmental changes in EGFR expression in the cortex. Neuron. 1997;19:251–67. doi: 10.1016/s0896-6273(00)80937-x. [DOI] [PubMed] [Google Scholar]
- Cicero TJ, Ferrendelli JA, Suntzeff V, Moore BW. Regional changes in CNS levels of the S-100 and 14-3-2 proteins during development and aging of the mouse. J Neurochem. 1972;19:2119–25. doi: 10.1111/j.1471-4159.1972.tb05121.x. [DOI] [PubMed] [Google Scholar]
- Deloulme JC, Assard N, Mbele GO, Mangin C, Kuwano R, Baudier J. S100A6 and S100A11 are specific targets of the calcium- and zinc-binding S100B protein in vivo. J Biol Chem. 2000;275:35302–10. doi: 10.1074/jbc.M003943200. [DOI] [PubMed] [Google Scholar]
- Deloulme JC, Janet T, Au D, Storm DR, Sensenbrenner M, Baudier J. Neuromodulin (GAP43): a neuronal protein kinase C substrate is also present in 0–2A glial cell lineage. Characterization of neuromodulin in secondary cultures of oligodendrocytes and comparison with the neuronal antigen. J Cell Biol. 1990;111:1559–69. doi: 10.1083/jcb.111.4.1559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deloulme JC, Raponi E, Gentil BJ, Bertacchi N, Marks A, Labourdette G, Baudier J. Nuclear expression of S100B in oligodendrocyte progenitor cells correlates with differentiation toward the oligodendroglial lineage and modulates oligodendrocytes maturation. Mol Cell Neurosci. 2004;27:453–65. doi: 10.1016/j.mcn.2004.07.008. [DOI] [PubMed] [Google Scholar]
- Doetsch F. A niche for adult neural stem cells. Curr Opin Genet Dev. 2003;13:543–50. doi: 10.1016/j.gde.2003.08.012. [DOI] [PubMed] [Google Scholar]
- Doetsch F, Caille I, Lim DA, Garcia-Verdugo JM, Alvarez-Buylla A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell. 1999;97:703–16. doi: 10.1016/s0092-8674(00)80783-7. [DOI] [PubMed] [Google Scholar]
- Doetsch F, Petreanu L, Caille I, Garcia-Verdugo JM, Alvarez-Buylla A. EGF converts transit-amplifying neurogenic precursors in the adult brain into multipotent stem cells. Neuron. 2002;36:1021–34. doi: 10.1016/s0896-6273(02)01133-9. [DOI] [PubMed] [Google Scholar]
- Filippov V, Kronenberg G, Pivneva T, Reuter K, Steiner B, Wang LP, Yamaguchi M, Kettenmann H, Kempermann G. Subpopulation of nestin-expressing progenitor cells in the adult murine hippocampus shows electrophysiological and morphological characteristics of astrocytes. Mol Cell Neurosci. 2003;23:373–82. doi: 10.1016/s1044-7431(03)00060-5. [DOI] [PubMed] [Google Scholar]
- Garcia AD, Doan NB, Imura T, Bush TG, Sofroniew MV. GFAP-expressing progenitors are the principal source of constitutive neurogenesis in adult mouse forebrain. Nat Neurosci. 2004;7:1233–41. doi: 10.1038/nn1340. [DOI] [PubMed] [Google Scholar]
- Garcion E, Faissner A, ffrench-Constant C. Knockout mice reveal a contribution of the extracellular matrix molecule tenascin-C to neural precursor proliferation and migration. Development. 2001;128:2485–96. doi: 10.1242/dev.128.13.2485. [DOI] [PubMed] [Google Scholar]
- Ghandour MS, Labourdette G, Vincendon G, Gombos G. A biochemical and immunohistological study of S100 protein in developing rat cerebellum. Dev Neurosci. 1981;4:98–109. doi: 10.1159/000112745. [DOI] [PubMed] [Google Scholar]
- Hachem S, Aguirre A, Vives V, Marks A, Gallo V, Legraverend C. Spatial and temporal expression of S100B in cells of oligodendrocyte lineage. Glia. 2005;51:81–97. doi: 10.1002/glia.20184. [DOI] [PubMed] [Google Scholar]
- Imura T, Kornblum HI, Sofroniew MV. The predominant neural stem cell isolated from postnatal and adult forebrain but not early embryonic forebrain expresses GFAP. J Neurosci. 2003;23:2824–32. doi: 10.1523/JNEUROSCI.23-07-02824.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornblum HI, Hussain R, Wiesen J, Miettinen P, Zurcher SD, Chow K, Derynck R, Werb Z. Abnormal astrocyte development and neuronal death in mice lacking the epidermal growth factor receptor. J Neurosci Res. 1998;53:697–717. doi: 10.1002/(SICI)1097-4547(19980915)53:6<697::AID-JNR8>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Laywell ED, Rakic P, Kukekov VG, Holland EC, Steindler DA. Identification of a multipotent astrocytic stem cell in the immature and adult mouse brain. Proc Natl Acad Sci U S A. 2000;97:13883–8. doi: 10.1073/pnas.250471697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levitt P, Rakic P. Immunoperoxidase localization of glial fibrillary acidic protein in radial glial cells and astrocytes of the developing rhesus monkey brain. J Comp Neurol. 1980;193:815–40. doi: 10.1002/cne.901930316. [DOI] [PubMed] [Google Scholar]
- Lim DA, Tramontin AD, Trevejo JM, Herrera DG, Garcia-Verdugo JM, Alvarez-Buylla A. Noggin antagonizes BMP signaling to create a niche for adult neurogenesis. Neuron. 2000;28:713–26. doi: 10.1016/s0896-6273(00)00148-3. [DOI] [PubMed] [Google Scholar]
- Liu X, Wang Q, Haydar TF, Bordey A. Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nat Neurosci. 2005;8:1179–87. doi: 10.1038/nn1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machold R, Hayashi S, Rutlin M, Muzumdar MD, Nery S, Corbin JG, Gritli-Linde A, Dellovade T, Porter JA, Rubin LL, Dudek H, McMahon AP, Fishell G. Sonic hedgehog is required for progenitor cell maintenance in telencephalic stem cell niches. Neuron. 2003;39:937–50. doi: 10.1016/s0896-6273(03)00561-0. [DOI] [PubMed] [Google Scholar]
- Malatesta P, Hack MA, Hartfuss E, Kettenmann H, Klinkert W, Kirchhoff F, Gotz M. Neuronal or glial progeny: regional differences in radial glia fate. Neuron. 2003;37:751–64. doi: 10.1016/s0896-6273(03)00116-8. [DOI] [PubMed] [Google Scholar]
- Merkle FT, Tramontin AD, Garcia-Verdugo JM, Alvarez-Buylla A. Radial glia give rise to adult neural stem cells in the subventricular zone. Proc Natl Acad Sci U S A. 2004;101:17528–32. doi: 10.1073/pnas.0407893101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Namba T, Mochizuki H, Onodera M, Mizuno Y, Namiki H, Seki T. The fate of neural progenitor cells expressing astrocytic and radial glial markers in the postnatal rat dentate gyrus. Eur J Neurosci. 2005;22:1928–41. doi: 10.1111/j.1460-9568.2005.04396.x. [DOI] [PubMed] [Google Scholar]
- Noctor SC, Flint AC, Weissman TA, Dammerman RS, Kriegstein AR. Neurons derived from radial glial cells establish radial units in neocortex. Nature. 2001;409:714–20. doi: 10.1038/35055553. [DOI] [PubMed] [Google Scholar]
- Okabe M, Ikawa M, Kominami K, Nakanishi T, Nishimune Y. ‘Green mice’ as a source of ubiquitous green cells. FEBS Lett. 1997;407:313–9. doi: 10.1016/s0014-5793(97)00313-x. [DOI] [PubMed] [Google Scholar]
- Palmer TD, Markakis EA, Willhoite AR, Safar F, Gage FH. Fibroblast growth factor-2 activates a latent neurogenic program in neural stem cells from diverse regions of the adult CNS. J Neurosci. 1999;19:8487–97. doi: 10.1523/JNEUROSCI.19-19-08487.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan W, Kastin AJ. Entry of EGF into brain is rapid and saturable. Peptides. 1999;20:1091–8. doi: 10.1016/s0196-9781(99)00094-7. [DOI] [PubMed] [Google Scholar]
- Peretto P, Giachino C, Aimar P, Fasolo A, Bonfanti L. Chain formation and glial tube assembly in the shift from neonatal to adult subventricular zone of the rodent forebrain. J Comp Neurol. 2005;487:407–27. doi: 10.1002/cne.20576. [DOI] [PubMed] [Google Scholar]
- Seri B, Garcia-Verdugo JM, McEwen BS, Alvarez-Buylla A. Astrocytes give rise to new neurons in the adult mammalian hippocampus. J Neurosci. 2001;21:7153–60. doi: 10.1523/JNEUROSCI.21-18-07153.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibilia M, Steinbach JP, Stingl L, Aguzzi A, Wagner EF. A strain-independent postnatal neurodegeneration in mice lacking the EGF receptor. Embo J. 1998;17:719–31. doi: 10.1093/emboj/17.3.719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spradling A, Drummond-Barbosa D, Kai T. Stem cells find their niche. Nature. 2001;414:98–104. doi: 10.1038/35102160. [DOI] [PubMed] [Google Scholar]
- Steindler DA, Laywell ED. Astrocytes as stem cells: nomenclature, phenotype, and translation. Glia. 2003;43:62–9. doi: 10.1002/glia.10242. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Chamczuk A, Hong Y, Jackowski G. Rapid and sensitive immunoassay for the measurement of serum S100B using isoform-specific monoclonal antibody. Clin Chem. 1999;45:1307–11. [PubMed] [Google Scholar]
- Tropepe V, Craig CG, Morshead CM, van der Kooy D. Transforming growth factor-alpha null and senescent mice show decreased neural progenitor cell proliferation in the forebrain subependyma. J Neurosci. 1997;17:7850–9. doi: 10.1523/JNEUROSCI.17-20-07850.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vives V, Alonso G, Solal AC, Joubert D, Legraverend C. Visualization of S100B-positive neurons and glia in the central nervous system of EGFP transgenic mice. J Comp Neurol. 2003;457:404–19. doi: 10.1002/cne.10552. [DOI] [PubMed] [Google Scholar]
- Voigt T. Development of glial cells in the cerebral wall of ferrets: direct tracing of their transformation from radial glia into astrocytes. J Comp Neurol. 1989;289:74–88. doi: 10.1002/cne.902890106. [DOI] [PubMed] [Google Scholar]
- Xiong Z, O’Hanlon D, Becker LE, Roder J, MacDonald JF, Marks A. Enhanced calcium transients in glial cells in neonatal cerebellar cultures derived from S100B null mice. Exp Cell Res. 2000;257:281–9. doi: 10.1006/excr.2000.4902. [DOI] [PubMed] [Google Scholar]
- Yamashita N, Kosaka K, Ilg EC, Schafer BW, Heizmann CW, Kosaka T. Selective association of S100A6 (calcyclin)-immunoreactive astrocytes with the tangential migration pathway of subventricular zone cells in the rat. Brain Res. 1997;778:388–92. doi: 10.1016/s0006-8993(97)01025-1. [DOI] [PubMed] [Google Scholar]
- Yang Q, O’Hanlon D, Heizmann CW, Marks A. Demonstration of heterodimer formation between S100B and S100A6 in the yeast two-hybrid system and human melanoma. Exp Cell Res. 1999;246:501–9. doi: 10.1006/excr.1998.4314. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


