Abstract
Purpose
The authors have shown that twitching motility, a pilus-mediated form of bacterial surface movement, is required for Pseudomonas aeruginosa virulence in a murine model of keratitis. To study the role of twitching motility in virulence, Pseudomonas traversal of multilayered corneal epithelia in vitro was investigated.
Methods
Translocation of multilayered corneal epithelia was investigated with the invasive strain PAK and isogenic twitching motility mutants. Rabbit corneal epithelial cells were grown to multilayers with filters and inoculated on their apical surfaces with 106 colony-forming unit bacteria, and translocating bacteria were quantified by viable counts of the basal chamber. Transepithelial resistance (TER) was recorded. Cellular exit of P. aeruginosa after invasion was quantified with modified gentamicin survival assays, and the role of apoptosis in exit was explored.
Results
PAK translocated the epithelia as early as 1 hour after infection, and by 8 hours apical and basal numbers of bacteria were similar. Bacterial translocation did not reduce TER. Each twitching motility mutant (pilU, pilT with pili, pilA lacking pili) was defective in translocation (>2 log reduction vs. PAK; P < 0.005). All twitching mutants were competent for cell invasion but defective in cellular exit, accumulating intracellularly to numbers exceeding those of PAK. Inhibiting apoptosis reduced the cellular exit of PAK.
Conclusions
These results show that twitching motility enables P. aeruginosa to translocate corneal epithelial layers and suggest that it contributes to epithelial cell exit by a mechanism involving apoptosis. The relationship between these in vitro findings and the role of twitching motility in P. aeruginosa virulence in vivo remains to be determined.
The Gram-negative opportunistic pathogen Pseudomonas aeruginosa is capable of causing severe disease in patients with various forms of immunocompromise. In the eye, P. aeruginosa can cause an aggressive corneal infection (infectious keratitis) that is most commonly associated with contact lens wear or ocular trauma. Development of P. aeruginosa keratitis is thought to require bacterial entry into the corneal stroma underlying the multilayered epithelium. For the latter to occur, bacteria must overcome physical and biochemical defenses of the tear fluid and corneal epithelium. Although corneal epithelial defenses could be directly breached by ocular trauma, mechanisms by which P. aeruginosa traverses multilayered corneal epithelia in other situations that predispose to infectious keratitis remain unknown.
Bacteria have been shown to use various strategies to traverse epithelial cell monolayers grown in culture. For example, enteropathogenic Escherichia coli break down barrier function by directly disrupting tight junctions.1,2 In contrast, Salmonella and Yersinia spp. disturb cellular junctions indirectly after cell contraction or cytoskeleton rearrangement.3–6 Other pathogens, such as Listeria monocytogenes and Shigella spp., can travel through tissues intracellularly by invading cells and then traveling directly from one cell to the next without ever leaving the intracellular environment.7,8
We have previously shown that P. aeruginosa invades corneal epithelial cells.9–11 Some of the bacterial factors involved in invasion include lipopolysaccharide (LPS),12 pili,13 and FlhA.14 Host cell factors involved include the cystic fibrosis transmembrane conductance regulator (CFTR),15 lipid rafts,16–18 asialo-GM1,13 and intracellular signaling proteins PI3K and Akt.19,20 Although bacterial invasion has been shown to be a key component in the pathogenesis of corneal infection caused by this pathogen,21 little is known of the mechanisms by which P. aeruginosa moves from cell to cell or by which it translocates (crosses) a multilayer epithelial barrier.
P. aeruginosa traversal of epithelial cells has been quantitatively studied only with MDCK cell monolayers.22,23 Not surprisingly, cytotoxic strains that encode the ExoU phospholipase toxin, which can quickly kill cells,24 were found to cause a rapid decrease in tight-junction integrity, as measured by a decrease in transepithelial resistance (TER). As expected, bacterial traversal then occurred at the same rate as the simultaneously added E. coli, which would not penetrate when used alone. This suggested that, for cytotoxic strains, bacterial traversal occurred where cells had been killed.
Very different results were found for invasive P. aeruginosa strains, which cause approximately 50% of P. aeruginosa corneal infections.25 Invasive strains had little impact on TER (consistent with their low cytotoxic activity), but they traversed more rapidly than the cytotoxic strains or the simultaneously added control E. coli. Those findings suggested that invasive strains of P. aeruginosa were using specialized mechanism(s) to travel across cells. Further, the absence of TER loss suggested that their capacity for becoming intracellular might be involved. Although it is known that invasive strains of P. aeruginosa can invade corneal and other epithelial cell types9,26,27 and that invasion is involved in corneal disease pathogenesis,21 it is not yet known whether invasion contributes to epithelial cell traversal. In that regard, P. aeruginosa should also be capable of exiting cells it has invaded.
P. aeruginosa and some other bacterial pathogens use twitching as a form of surface-associated motility that involves the extension, tethering, and retraction of polar type IV pili.28–34 We previously showed that twitching motility mutants (pilU or pilT) of P. aeruginosa had reduced virulence in a murine model for corneal infection than did twitching motility-competent wild-type bacteria.35 In that study, twitching motility was not required for initial bacterial adherence to, or invasion of, corneal epithelial cells, suggesting that the role of twitching involved later events. Indeed, the capacity for corneal colonization by the mutants was reduced at 48 hours, but not at 4 hours, after infection, implicating a role for twitching motility in tissue penetration or bacterial dissemination. In this regard others have shown that twitching motility is required for P. aeruginosa–induced apoptosis of airway epithelial cells or cell death of MDCK cells exposed to a large inoculum of an invasive strain for extended periods of time (107–109 colony-forming units [CFU] for up to 11 hours).13,36,37
Given that many (if not most) potential sites of infection are covered with a multilayered epithelium, it is surprising that little is known about strategies used by bacteria to cross multilayers of cells. The aim of this study was to begin to study traversal mechanisms used by P. aeruginosa when it crosses multilayered epithelia and to specifically test the hypothesis that twitching motility is involved. Previously defined twitching motility mutants were compared with wild-type P. aeruginosa in several in vitro assay systems designed to quantify and locate viable bacteria over time at various stages after infection.
Materials and Methods
Cell Culture
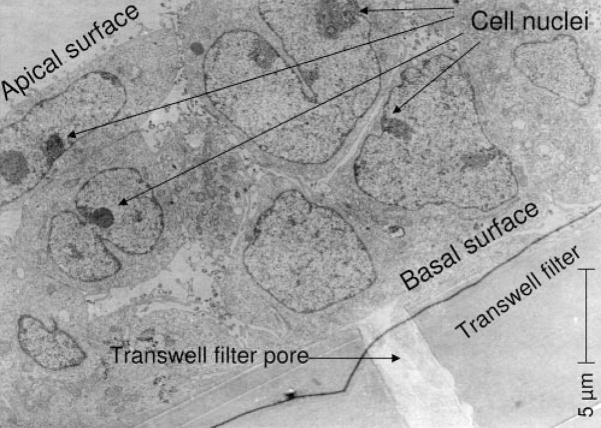
Rabbit corneal epithelial cell layers were grown to confluence on 3-μm pore size, 12-mm diameter, semipermeable filters (Transwell; Corning Costar, Lowell, MA). Cells grown on filters were cultured in supplemental hormonal epithelial medium (SHEM)38 according to the airlifted method, as previously described.39 Briefly, cells were cultured on the filters for 3 days. Apical and basal surfaces were submerged in SHEM and then air-lifted by removing media from the apical side on day 4 and feeding only the basal side until day 7, when cells were used for experiments. Cells grown in this fashion formed multilayers on the filters (Fig. 1). In other experiments, cells were grown to confluence as monolayers on 24-well tissue culture plates or on microscope glass coverslips (22cir-1D; Fisher Scientific, Pittsburgh, PA). These were used for experiments on day 4 after passage.
Figure 1.
Transmission electron micrograph of air-lifted immortalized rabbit corneal epithelial cells showing multiple (two to four) cell layers on a semipermeable filter inserts after 7 days of culture; magnification 2900×. Cells were cultured for 3 days submerged, then for 4 days air-lifted on the filter.
Transmission Electron Microscopy
Cells grown as multilayers according to the air-lifted method were prepared for transmission electron microscopy (TEM). Samples were fixed with 2% glutaraldehyde and postfixed in 1% osmium tetroxide (OsO4); both steps were performed in 0.1 M sodium cacodylate buffer (pH 7.2) at 4°C for 1 hour. Filters were precut into small strips and dehydrated using a graded acetone series (35%, 50%, 70%, 85%, 95%, and pure acetone) twice for 10 minutes each. Samples were then infiltrated sequentially in 1:3, 1:1, 3:1 Spurr resin/acetone for 1 hour each and then transferred to pure resin to polymerize at 60°C for 2 days. The samples were ultrathin-sectioned (70 nm), stained with uracyl acetate and lead tartrate, and viewed and photographed with a transmission electron microscope (Tecnai 12; FEI Company, Hillsboro, OR) at 2.9 kV.
Bacterial Strains and Mutants
P. aeruginosa strain PAK and the twitching motility mutants PAKpi-lA.Tcr (pilA), PAKpilU::Tn5 (pilU), and PAKpilT::Tn5 (pilT) were used. These mutants were previously constructed and characterized by John S. Mattick (University of Queensland, Australia).34,40 Wild-type PAK and the isogenic mutants were kindly provided by Joanne Engel (University of California at San Francisco) and were used with the permission of John Mattick. The pilA mutant is a nonpiliated, isogenic mutant (twitching motility defective by virtue of nonpiliation).40 Piliated twitching motility mutants (pilU and pilT) each originated from a transposon mutagenesis screen.34 Bacteria were cultured on trypticase soy agar plates at 37°C and resuspended in Hanks balanced salt solution (HBSS) or SHEM without antibiotics to a concentration of 108 CFU/mL (0.1 OD at 650 nm). That suspension was subsequently diluted to a final bacterial concentration of 106 CFU/mL. Bacteria were enumerated by viable count to confirm concentrations after serial dilutions.
In Vitro Bacterial Traversal (Translocation) Assay
To study the ability of bacteria to cross multilayers of corneal epithelial cells, cells grown on semipermeable filters (Transwell; Corning Costar) for 7 days were inoculated on the apical surface with invasive P. aeruginosa strain PAK or with one of the twitching motility mutants (pilA, pilU, pilT). Infected cells were then incubated for as long as 8 hours at 37°C. At hourly intervals the number of bacteria in the apical (upper) and basal (lower) chambers was determined by viable counts of aliquots collected from each chamber. The number of bacteria calculated to be in the upper chamber at each time point was used to estimate the replication rate of the inoculum (which was added to that chamber) over time. Bacteria in the lower chamber (which was not inoculated) represented bacteria that had traversed the multilayer of cells. Control experiments showed that the 3-μm pore size semipermeable filters (Transwell; Corning Costar) alone were not significant barriers to the translocation of wild-type P. aeruginosa or that of the twitching motility mutants (data not shown). To monitor tight-junction integrity, TER readings were collected with a meter (EVOM; World Precision Instruments Inc., Sarasota, FL), and results were compared with those of HBSS-treated (sham-inoculated) control cells and ethylene glycol tetraacetic acid (EGTA)-treated (100 mM; Fisher Scientific, Pittsburgh, PA) control cells. TER readings from EGTA-treated samples were used as a baseline control for TER (EGTA disrupts tight junctions).41 Finally, cells were rinsed with fresh HBSS before the addition of antibiotic-free SHEM supplemented with gentamicin (200 μg/mL) to kill extracellular bacteria for 1 hour.26 At the end of each experiment, intracellular survivors were quantified by treating the cells with 0.25% (vol/vol) nonionic surfactant (Triton X-100; Sigma, St. Louis, MO) in phosphate-buffered saline (PBS) for 15 minutes and performing viable counts on the lysate.
Bacterial Invasion and Exit Assays
Corneal epithelial cells were grown as monolayers on 24-well tissue culture plates to determine bacterial capacity for cellular invasion, cellular exit, or both. For these experiments, cells were inoculated with wild-type PAK or one of the twitching motility mutants (pilA, pilU) for 3 hours at 37°C (invasion period). Cells were then incubated with antibiotic-free SHEM supplemented with gentamicin (200 μg/mL) to kill extracellular bacteria.26 The ability of bacteria to exit cells after invasion was quantified by removing the gentamicin solution from the cells at time points between 1 hour and 6 hours after the 3-hour invasion period, adding fresh SHEM without antibiotics for 1 hour (exit period), and sampling the extracellular medium by viable counts. Viable bacteria that had exited the cells during that hour were thus enumerated. Additional samples were included to enumerate bacteria that were intracellular at various time points after inoculation. These cells were treated with gentamicin-containing media for at least 1 hour to kill extracellular bacteria,26 followed by a 15-minute treatment with 0.25% (vol/vol) nonionic surfactant (Triton X-100; Sigma, in PBS. Viable counts were then performed on aliquots of the cell lysate.
Fluorescence Microscopy and Caspase Inhibition Studies
Coverslip-grown cells were rinsed once with PBS and treated with 80 μM caspase inhibitor (Caspase Inhibitor I [Calbiochem, San Diego, CA]; Z-VAD [Ome]-FMK general caspases inhibitor, Fas-mediated apoptosis inhibited) for 1 hour at 37°C. After inhibitor pretreatment, cells were inoculated with 1 mL SHEM (without antibiotics) containing bacteria (106 CFU/mL) and 80 μM caspase inhibitor (Caspase Inhibitor I; Calbiochem). Samples were incubated for 1 hour at 37°C (invasion period). Coverslips were then treated for 1 hour with gentamicin-containing media, rinsed twice with PBS, and stained for 1 hour with the use of a caspase staining kit (CaspGlow Red; Biovision, Eugene, OR) diluted 1:300 in SHEM without antibiotics to label apoptotic cells. Coverslips were rinsed and allowed to dry before mounting on a glass slide with one drop of mounting medium (Vectashield; Vector Laboratories, Burlingame, CA) for fluorescence with mounting medium (DAPI H-1200; Vector Laboratories). Slides were immediately examined with the use of fluorescence microscopy to quantify the number of cells undergoing apoptosis. Approximately 600 cells in total were quantified per coverslip. Control samples without caspase inhibitor (Caspase Inhibitor I; Calbiochem) were also included to determine the efficacy of the inhibitor. To determine whether exit involves apoptosis, additional coverslips were treated as described. After 1-hour gentamicin treatment, coverslips were rinsed and incubated for an additional 2 hours with SHEM without antibiotics (2-hour exit period). Aliquots were then collected, and bacterial exit was quantified by viable count. Subsequently, coverslips were stained and analyzed as described. The caspase inhibitor did not affect bacterial viable (data not shown).
Statistical Analysis
Statistical significance of differences between groups was determined using Student’s t-test. Data were expressed as mean ± SD. P < 0.05 was considered significant. Experiments were repeated at least three times, and at least three samples were used in each group.
Results
Translocation of Multilayers of Corneal Epithelial Cells In Vitro by P. aeruginosa
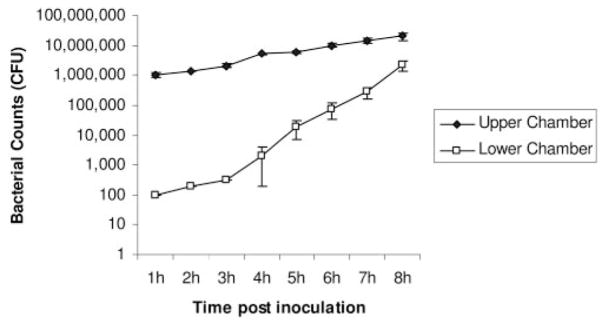
Wild-type invasive P. aeruginosa strain PAK traversed multiple layers of corneal epithelial cells (Fig. 2). Bacteria that were added to only the upper chamber of the semipermeable filter (Transwell; Corning Costar) apparatus were detected within the lower chamber of cells as early as 1 hour after inoculation (0.01% of the inoculum). By the 8-hour time point, the number of bacteria in the upper and lower chambers approached similar levels. Bacteria in the upper chamber (wild-type and mutants) increased similarly in number (by approximately 10-fold) during the 8-hour assay, reflecting their normal replication rate in that medium. In contrast, bacterial numbers in the lower chamber increased by approximately 10,000-fold over the same time period. Although it is likely that replication of previously translocated bacteria contributed to the number of bacteria in the lower chamber, the data suggest that translocation was the predominant contributor.
Figure 2.
P. aeruginosa traversal of multilayered corneal epithelial cells grown on semipermeable filters grown in vitro. P. aeruginosa strain PAK (106 CFU) was added to the upper chamber, and viable counts of the upper and lower chambers were performed hourly. Bacteria first appeared in the lower chamber (the other side of the cell layer) 1 hour after inoculation. By 8 hours, the numbers of bacteria in the upper and lower chambers were similar. Data are expressed as the mean ± SD. Three semipermeable filters were used for each sample.
There was no significant difference in TER between infected and uninfected (control) cells; it remained higher in both than in EGTA-treated baseline controls (Table 1). These data suggested that traversal could occur without functional impact on tight-junction integrity. Furthermore, LDH release assays confirmed that bacteria were not cytotoxic to the epithelia at these inocula (data not shown).
Table 1.
TER across Multilayers of Rabbit Corneal Epithelial Cells Infected with Wild-type P. aeruginosa Strain PAK
| Mean (±SD) TER Reading (Ω/cm2) at Postinoculation Time Points |
|||||||||
|---|---|---|---|---|---|---|---|---|---|
| 0 h | 1 h | 2 h | 3 h | 4 h | 5 h | 6 h | 7 h | 8 h | |
| Wild-type PAK* | 135 ± 1 | 132 ± 2 | 131 ± 2 | 130 ± 2 | 130 ± 2 | 127 ± 2 | 125 ± 1 | 123 ± 2 | 121 ± 2 |
| No bacteria control | 133 ± 2 | 132 ± 2 | 131 ± 3 | 130 ± 2 | 129 ± 2 | 128 ± 3 | 128 ± 3 | 124 ± 2 | 123 ± 2 |
| EGTA control† | 104 ± 2 | 100 ± 2 | 99 ± 3 | 100 ± 1 | 99 ± 1 | 99 ± 1 | 99 ± 1 | 92 ± 2 | 97 ± 2 |
Layers of infected cells showed TER values similar to those of uninfected (HBSS-inoculated) cells throughout the 8-hour traversal assay. Each remained significantly higher than in controls treated with 100 mM EGTA (P < 0.05, t-test).
No significant difference versus uninfected cells at each time point (P < 0.05, t-test).
Similar to baseline control (submerged filter without epithelial cells).
Twitching Motility Required for Bacterial Traversal of Corneal Epithelia In Vitro
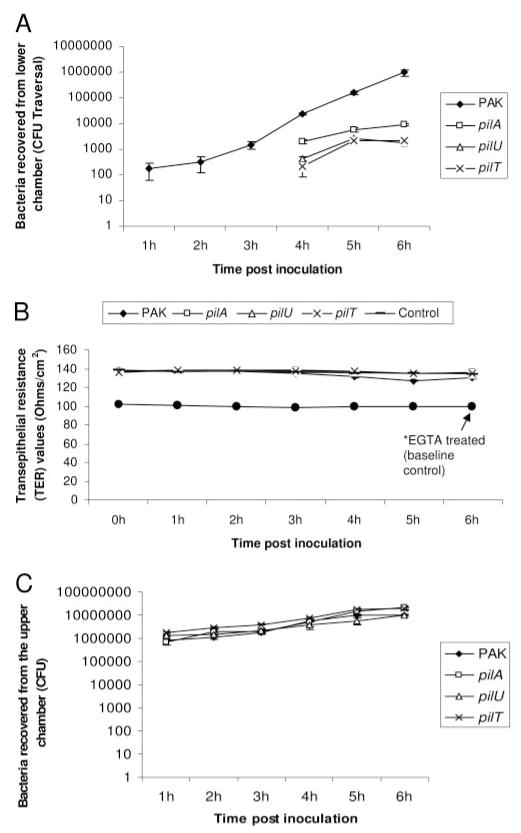
Each twitching motility mutant (pilU, pilT, pilA) was compared with wild-type PAK for its ability to translocate rabbit corneal epithelial cells. All three twitching mutants were significantly reduced in their ability to traverse these epithelial multilayers (Fig. 3A). Twitching motility mutants showed a delay in the initiation of traversal (level of detection of 100 CFU reached at 4 hours after inoculation compared with 1 hour for wild-type), and numbers remained lower than for wild-type at all time points, (>2 log reduction at the end of the 6-hour assay; Fig. 3A). However, there was an approximately fivefold greater traversal by the pilA mutant than by the pilU or pilT mutant (Fig 3A; P < 0.001, t-test).
Figure 3.
Traversal of multilayered corneal epithelial cells grown on semipermeable filters in vitro by P. aeruginosa strain PAK and its twitching motility mutants (pilA, pilU, and pilT) after inoculation of the upper chamber with 106 CFU bacteria. (A) Each of the three twitching motility mutants (pilA, pilU, and pilT) was defective in its ability to traverse cells over 6 hours compared with wild-type PAK (P < 0.05, t-test). (B) Neither wild-type nor twitching motility mutants impacted TER during the 6-hour assay. EGTA-treated samples significantly reduced TER to baseline values (*P < 0.05, t-test, vs. untreated samples at each time point). (C) Growth rates of the twitching mutants were similar to wild-type PAK, as shown by the number of bacteria in the upper chamber over the 6-hour assay. Data are expressed as the mean ± SD. Three semipermeable filters were used for each sample.
No significant differences in TER were found between any of the infected cells, for which TER values remained similar to uninfected cells at all time points, and significantly higher than EGTA-treated baseline controls (Fig. 3B).
The deficiency in traversal of twitching motility mutants did not relate to differences in extracellular growth rates. Viable counts performed on aliquots from the upper chamber, where inocula had been added, showed that mutants and wild-type grew at similar rates (Fig. 3C).
Accumulation of Twitching Motility Mutants within Cells over Time
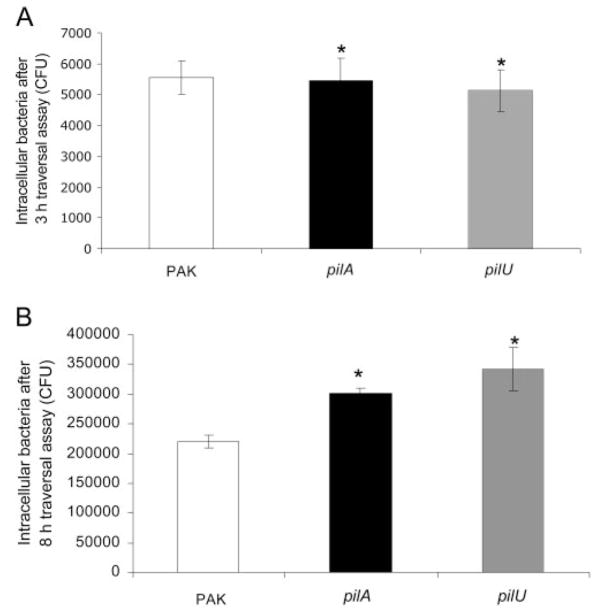
To understand why twitching mutants were deficient in traversal, the location of bacteria within the traversal assay system was explored. Although our previously published data showed that twitching mutants were as competent as wild-type in their ability to enter corneal epithelial cells grown as monolayers, the traversal assay system involved infection of multilayers. Thus, twitching motility mutants (pilA and pilU) were compared with wild-type for internalization capacity in the semipermeable filter (Transwell; Corning Costar) system. Comparison of pilU and pilA mutants to wild-type at the 3-hour traversal period showed no significant differences in invasion (Fig. 4A).
Figure 4.
Quantification of bacteria within corneal epithelial cells (intracellular bacteria) during traversal assay at 3 hours (A) and 8 hours (B) after inoculation. Cells were grown as multilayers on semipermeable filters and incubated with 106 CFU of P. aeruginosa strain PAK or its pilA or pilU mutants. (A) Invasion of twitching motility mutants (pilA and pilU) was similar to that of wild-type PAK after 3 hours (*P > 0.05, t-test, for each comparison). (B) After 8 hours, more intracellular bacteria were recovered from within corneal epithelial cells infected with twitching motility mutants (pilA and pilU) than from cells infected with wild-type PAK (*P < 0.01, t-test, for each mutant vs. wild-type PAK). Data are expressed as mean ± SD. Three semipermeable filters were used for each sample.
However, another potential mechanism for reduced traversal of twitching mutants could be defects in their ability to survive intracellularly after invasion. Hence, the numbers of bacteria recovered intracellularly from wild-type and mutant-infected cells were compared at the end of the 8-hour traversal assay. Surprisingly, twitching mutants demonstrated a significant increase in intracellular numbers compared with wild-type (Fig. 4B).
Contribution of Twitching Motility to Bacterial Exit from Epithelial Cells after Invasion
Increased intracellular numbers of twitching motility mutants compared with wild-type over time suggested either an increased intracellular replication rate or a greater accumulation of twitching motility mutants inside epithelial cells compared with wild-type P. aeruginosa. Given that intracellular accumulation could also explain their reduced ability to traverse cell multilayers, we focused on that possibility.
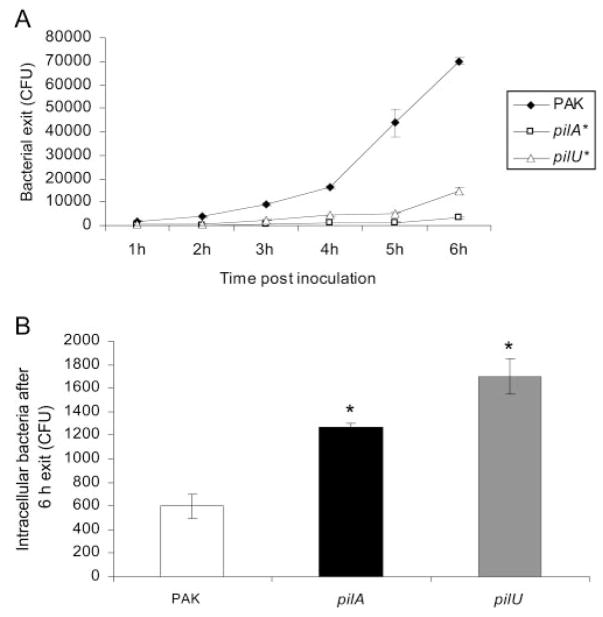
The ability of wild-type PAK to exit cells was tested first because that had not been previously explored. Bacteria were allowed to invade cells for 3 hours before gentamicin-containing media (antibiotic-free SHEM supplemented with 200 μg/mL gentamicin) were added to kill extracellular bacteria.26 After 1 hour, the gentamicin-containing medium was replaced with SHEM (without antibiotics), and the extracellular media were monitored for the appearance of extracellular bacteria. By removing gentamicin solution at different hourly intervals in different samples, the exit profile over time was “mapped,” and the short 1-hour window for allowing exit eliminated the possibility of significant replication by extracellular bacteria. The results showed that wild-type bacteria were capable of exiting cells after internalization and that the number of exiting bacteria increased with time after inoculation (Fig. 5A). However, pilA and pilU twitching motility mutants showed significantly reduced capacity to exit epithelial cells after invasion compared with wild-type bacteria (approximately sevenfold reduction at 6 hours after inoculation; Fig. 5A). Reduced ability to exit cells correlated with intracellular accumulation in the same experiments (Fig. 5B).
Figure 5.
(A) Exit of P. aeruginosa strain PAK and its pilA or pilU mutant from corneal epithelial cells after invasion. Cells were grown as monolayers on tissue culture plates and incubated with 106 CFU bacteria for 3 hours to allow invasion to occur. After extracellular bacteria were killed with gentamicin (1 hour), fresh media were placed on the cells, and viable counts were performed from 1 hour to 6 hours. Twitching motility mutants (pilA and pilU) showed reduced ability to exit corneal epithelial cells after invasion (P < 0.005, t-test, vs. wild-type PAK). (B) Numbers of intracellular bacteria at the conclusion of the 6-hour exit assay. Cells were treated with gentamicin for 1 hour and, after washing, were lysed with 0.25% (vol/vol) nonionic surfactant. More intracellular bacteria were recovered from within corneal epithelial cells infected with twitching motility mutants than from cells infected with wild-type bacteria (*P < 0.005, t-test, for each mutant vs. wild-type PAK). Data are expressed as mean ± SD. Three wells were used per sample.
Bacterial Exit from Epithelial Cells Involving Apoptotic Processes
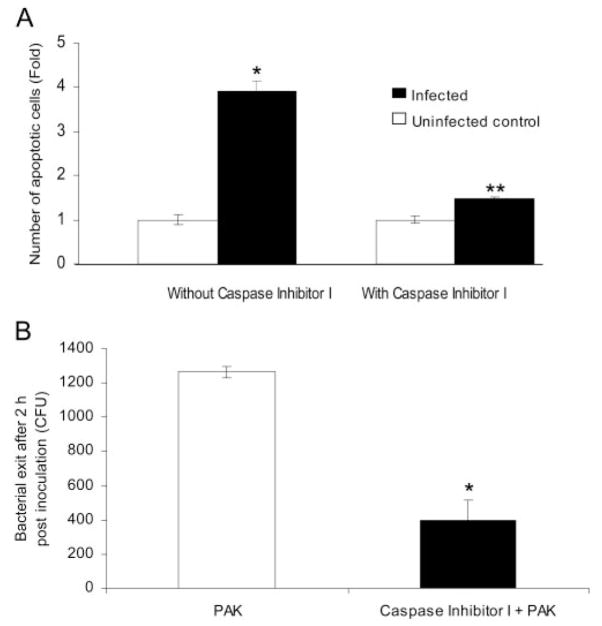
Twitching motility is required for P. aeruginosa-induced apoptosis in conjunctival epithelial cells.18 To determine whether apoptosis in turn provided a mechanism for bacterial exit from corneal epithelial cells, we studied the effect of an apoptosis caspase inhibitor (Caspase Inhibitor I; Calbiochem) on bacterial exit. Control experiments showed that PAK-induced apoptosis of corneal epithelial cells was effectively inhibited by treatment with the caspase inhibitor (Fig. 6A). This coincided with a greater than threefold reduction in bacterial exit from inhibitor-treated cells (Fig. 6B).
Figure 6.
(A) Effect of caspase inhibitor on bacterial-induced apoptosis in corneal epithelial cells grown as monolayers on glass coverslips. Corneal cells were pretreated with caspase inhibitor (80 μM for 1 hour) or cell culture media (controls). Cells were then inoculated with 106 CFU P. aeruginosa strain PAK and incubated for 1 hour in the continuous presence of the inhibitor. After another hour of gentamicin treatment, apoptotic cells were detected using a caspase staining kit. Without inhibitor, PAK significantly increased the number of apoptotic corneal epithelial cells (*P < 0.001 vs. uninfected cells, t-test). This induction was significantly reduced by the caspase inhibitor (**P < 0.005 vs. untreated infected cells, t-test). (B) Effect of caspase inhibitor on PAK exit after invasion of corneal epithelial cells grown on glass coverslips. Cells were pretreated with inhibitor, inoculated with bacteria, and treated with gentamicin as described, followed by a 2-hour exit assay. The caspase inhibitor decreased bacterial exit from these cells (*P < 0.001 vs. infected without inhibitor, t-test). Data are expressed as mean ± SD. Three wells were used per sample.
Discussion
We have shown that P. aeruginosa invades epithelial cells9,26 and can replicate intracellularly.26 The results of this study show that the twitching motility of P. aeruginosa is involved in bacterial translocation of corneal epithelial cell multilayers and in bacterial exit from these cells after internalization (the latter involves a caspase-dependent mechanism).
The data provide some insight into the mechanisms by which P. aeruginosa can traverse multilayers of corneal epithelial cells. For example, traversal did not significantly disrupt TER. This is consistent with previous findings with epithelial cell monolayers (MDCK cells) showing that invasive strains of P. aeruginosa traverse more efficiently than cytotoxic strains that are more disruptive to TER.22,23 The absence of TER disruption suggests that invasive P. aeruginosa travel between cells without disrupting the integrity of the tight junctions that maintain TER, or they traffic through cells intracellularly, or they temporarily disrupt tight junctions to travel between cells. The TER effects of temporarily disrupting tight junctions to travel between cells are masked by deeper layer junctional complexes.42 However, further studies are needed to determine the pathways of traversal.
We previously reported that twitching motility is required for virulence to be established in vivo in a corneal infection model, in which bacteria first penetrate a multilayered epithelium so that disease can result in the underlying stroma.10,35,43 Others have found that though twitching motility mutants retain the capacity to cause pathologic conditions in the lung, they have a reduced capacity to disseminate to the liver,36 a process likely to require bacterial penetration through epithelial barriers. Our previously published data show that twitching motility is not required for adherence to, or invasion of, corneal epithelial cells.35 The data presented here show that twitching is, however, required for efficient bacterial traversal of epithelial cell multilayers, at least in vitro. Whether this will translate in vivo to explain the loss of virulence for twitching mutants is to be determined.
Twitching motility mutants were found to accumulate inside cells, correlating with a loss of ability to exit from invaded cells. Cellular exit by most pathogens, including P. aeruginosa, is poorly understood. Shigella and Listeria are among the best-characterized bacterial pathogens known to mediate their exit from cells in a two-step process involving escape from the phagosome44–46 and then cell-to-cell spread involving actin polymerization.47,48 Salmonella has also been reported to exit cells involving the induction of oncosis, an irreversible swelling of cells that leads to cell death. This follows an accumulation of intracellular bacteria and flagellum-dependent exit.49 In the case of Chlamydia, two mutually exclusive mechanisms for exit have been described: cellular lysis involving membrane permeabilization and packaged release or extrusion involving pinching of the inclusion, protrusion, and subsequent detachment from cells.50 Others, such as the malaria parasite Plasmodium falciparum, are able to escape cells through the action of cysteine proteases, which cause the rupture of vacuoles and cells.51 Our data provide evidence that P. aeruginosa can also exit cells they have invaded. They also suggest that exit may be required for pathogenesis because the lack of cell exit correlated with a diminished ability to traverse cells and reduced in vivo virulence with mutants that displayed normal cell adherence and invasion capacity.
The mechanism for exit is likely to require apoptosis because a caspase inhibitor reduced bacterial-induced apoptosis and exit. This follows a previous report showing that twitching motility is required for apoptosis caused by invasive strains of P. aeruginosa.37 In that study, it was hypothesized that apoptosis of P. aeruginosa-infected cells served as a host defense mechanism Whether apoptosis is involved because it is directly or indirectly involved in the mechanism for escape is yet to be determined. Other cytotoxic effects of strain PAK have been reported with the use of MDCK cells.4 These were not observed in the present study, possibly reflecting the significantly lower (~100-fold) bacterial numbers involved. Nevertheless it remains possible that invasive P. aeruginosa uses multiple cytotoxic mechanisms to facilitate the translocation of epithelial barriers.
The pilA (pilin) mutant, which lacks pili altogether, was more effective at traversal than the two twitching motility mutants (pilU and pilT), which have pili. One speculation about pili is that its presence on twitching mutants physically hinders traversal so that it is reduced below the level of the nonpiliated mutants. Alternatively, the absence of pili might compromise host cell defenses against traversal if pilin is involved in recognition of the pathogen by the host, thereby enabling the nonpiliated mutants to traverse more efficiently. Whatever the mechanism for differences between piliated and nonpiliated twitching mutants, the fact that all were compromised compared with wild-type confirmed a role for twitching motility in traversal. Further studies are needed to determine the relative contribution(s) of these mutations to P. aeruginosa translocation of multilayered epithelium.
A model for wild-type traversal suggested by the data is that it involves intracellular travel requiring cellular entry and exit. This would explain why twitching mutants that are defective in exit and accumulate inside cells are also defective in traversal capacity. Yet it remains possible that traversal is reduced for twitching mutants for other reasons. For example, if traversal involves an extracellular component, twitching motility, which is a form of surface-associated motility, could very well be involved by providing a means for locomotion between cells.
In summary, the data presented in this study show that invasive P. aeruginosa can traverse multilayers of epithelial cell without disrupting TER. Given that, and considering that invasive P. aeruginosa strains have the capacity to enter and exit cells, an intracellular pathway may be involved. Twitching motility, previously determined to be an important virulence factor for P. aeruginosa in multiple in vivo models, was found to be required for bacterial traversal in vitro and for bacterial exit from invaded cells, which was in turn reduced by a caspase inhibitor. Apoptosis has previously been shown to require twitching. One possible explanation for the contribution of twitching to virulence in vivo is that after bacteria adhere/invade, twitching facilitates the traversal of epithelial cell layers by enabling bacteria to exit the cells they have invaded through a process involving apoptosis. However, P. aeruginosa–induced apoptosis has also been hypothesized to serve as a host defense mechanism.52 Further studies will help clarify the role of apoptosis in P. aeruginosa pathogenesis and host defense. In addition, it will be important to explore the various in vitro identified roles for twitching to establish cause-and-effect relationships and to prove their relevance to virulence through in vivo methods.
Once initiated, disease caused by P. aeruginosa is notoriously difficult to treat. Studies aimed at understanding how P. aeruginosa penetrates epithelial cell barriers to initiate disease and how host defenses normally prevent bacterial penetration could lead to new approaches to prevent P. aeruginosa–induced disease. Studies of the mechanisms involved in bacterial traversal of epithelia are therefore warranted. The data presented here showing the involvement of twitching motility in traversal provide new directions for future research in this area.
Acknowledgments
The authors thank Alex Carrasco for technical assistance.
Supported by National Institutes of Health Grant EY11221 (SMJF), National Eye Institute Fellowship F31 EY016328-02, and an ASM Robert D. Watkins Graduate Research Fellowship (IA).
Footnotes
Disclosure: I. Alarcon, None; D.J. Evans, None; S.M.J. Fleiszig, None
References
- 1.Simonovic I, Rosenberg J, Koutsouris A, Hecht G. Enteropathogenic Escherichia coli dephosphorylates and dissociates occludin from intestinal epithelial tight junctions. Cell Microbiol. 2000;2:305–315. doi: 10.1046/j.1462-5822.2000.00055.x. [DOI] [PubMed] [Google Scholar]
- 2.Sears CL. Molecular physiology and pathophysiology of tight junctions, V: assault of the tight junction by enteric pathogens. Am J Physiol Gastrointest Liver Physiol. 2000;279:G1129–G11234. doi: 10.1152/ajpgi.2000.279.6.G1129. [DOI] [PubMed] [Google Scholar]
- 3.Jepson MA, Schlecht HB, Collares-Buzato CB. Localization of dysfunctional tight junctions in Salmonella enterica serovar typhimurium-infected epithelial layers. Infect Immun. 2000;68:7202–7208. doi: 10.1128/iai.68.12.7202-7208.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cywes C, Wessels MR. Group A Streptococcus tissue invasion by CD44-mediated cell signalling. Nature. 2001;414:648–652. doi: 10.1038/414648a. [DOI] [PubMed] [Google Scholar]
- 5.Tafazoli F, Magnusson KE, Zheng L. Disruption of epithelial barrier integrity by Salmonella enterica serovar typhimurium requires geranylgeranylated proteins. Infect Immun. 2003;71:872–881. doi: 10.1128/IAI.71.2.872-881.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tafazoli F, Holmstrom A, Forsberg A, Magnusson KE. Apically exposed, tight junction-associated beta1-integrins allow binding and YopE-mediated perturbation of epithelial barriers by wild-type Yersinia bacteria. Infect Immun. 2000;68:5335–5343. doi: 10.1128/iai.68.9.5335-5343.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gedde MM, Higgins DE, Tilney LG, Portnoy DA. Role of listeriolysin O in cell-to-cell spread of Listeria monocytogenes. Infect Immun. 2000;68:999–1003. doi: 10.1128/iai.68.2.999-1003.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yu J, Edwards-Jones B, Neyrolles O, Kroll JS. Key role for DsbA in cell-to-cell spread of Shigella flexneri, permitting secretion of Ipa proteins into interepithelial protrusions. Infect Immun. 2000;68:6449–6456. doi: 10.1128/iai.68.11.6449-6456.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fleiszig SM, Zaidi TS, Fletcher EL, Preston MJ, Pier GB. Pseudomonas aeruginosa invades corneal epithelial cells during experimental infection. Infect Immun. 1994;62:3485–3493. doi: 10.1128/iai.62.8.3485-3493.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee EJ, Evans DJ, Fleiszig SM. Role of Pseudomonas aeruginosa ExsA in penetration through corneal epithelium in a novel in vivo model. Invest Ophthalmol Vis Sci. 2003;44:5220–5227. doi: 10.1167/iovs.03-0229. [DOI] [PubMed] [Google Scholar]
- 11.Angus AA, Lee AA, Augustin DK, Lee EJ, Evans DJ, Fleiszig SM. Pseudomonas aeruginosa induces membrane blebs in epithelial cells, which are utilized as a niche for intracellular replication and motility. Infect Immun. 2008;76:1992–2001. doi: 10.1128/IAI.01221-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zaidi TS, Fleiszig SM, Preston MJ, Goldberg JB, Pier GB. Lipopolysaccharide outer core is a ligand for corneal cell binding and ingestion of Pseudomonas aeruginosa. Invest Ophthalmol Vis Sci. 1996;37:976–986. [PubMed] [Google Scholar]
- 13.Comolli JC, Waite LL, Mostov KE, Engel JN. Pili binding to asialo-GM1 on epithelial cells can mediate cytotoxicity or bacterial internalization by Pseudomonas aeruginosa. Infect Immun. 1999;67:3207–3214. doi: 10.1128/iai.67.7.3207-3214.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fleiszig SM, Arora SK, Van R, Ramphal R. FlhA, a component of the flagellum assembly apparatus of Pseudomonas aeruginosa, plays a role in internalization by corneal epithelial cells. Infect Immun. 2001;69:4931–4937. doi: 10.1128/IAI.69.8.4931-4937.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kowalski MP, Dubouix-Bourandy A, Bajmoczi M, et al. Host resistance to lung infection mediated by major vault protein in epithelial cells. Science. 2007;317:130–132. doi: 10.1126/science.1142311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kowalski MP, Pier GB. Localization of cystic fibrosis transmembrane conductance regulator to lipid rafts of epithelial cells is required for Pseudomonas aeruginosa-induced cellular activation. J Immunol. 2004;172:418–425. doi: 10.4049/jimmunol.172.1.418. [DOI] [PubMed] [Google Scholar]
- 17.Yamamoto N, Jester JV, Petroll WM, Cavanagh HD. Prolonged hypoxia induces lipid raft formation and increases Pseudomonas internalization in vivo after contact lens wear and lid closure. Eye Contact Lens. 2006;32:114–120. doi: 10.1097/01.icl.0000177384.27778.4c. [DOI] [PubMed] [Google Scholar]
- 18.Yamamoto N, Petroll MW, Cavanagh HD, Jester JV. Internalization of Pseudomonas aeruginosa is mediated by lipid rafts in contact lens-wearing rabbit and cultured human corneal epithelial cells. Invest Ophthalmol Vis Sci. 2005;46:1348–1355. doi: 10.1167/iovs.04-0542. [DOI] [PubMed] [Google Scholar]
- 19.Kierbel A, Gassama-Diagne A, Mostov K, Engel JN. The phosphoinositol-3-kinase-protein kinase B/Akt pathway is critical for Pseudomonas aeruginosa strain PAK internalization. Mol Biol Cell. 2005;16:2577–2585. doi: 10.1091/mbc.E04-08-0717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kierbel A, Gassama-Diagne A, Rocha C, et al. Pseudomonas aeruginosa exploits a PIP3-dependent pathway to transform apical into basolateral membrane. J Cell Biol. 2007;177:21–27. doi: 10.1083/jcb.200605142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zaidi TS, Lyczak J, Preston M, Pier GB. Cystic fibrosis transmembrane conductance regulator-mediated corneal epithelial cell ingestion of Pseudomonas aeruginosa is a key component in the pathogenesis of experimental murine keratitis. Infect Immun. 1999;67:1481–1492. doi: 10.1128/iai.67.3.1481-1492.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hirakata Y, Finlay BB, Simpson DA, Kohno S, Kamihira S, Speert DP. Penetration of clinical isolates of Pseudomonas aeruginosa through MDCK epithelial cell monolayers. J Infect Dis. 2000;181:765–769. doi: 10.1086/315276. [DOI] [PubMed] [Google Scholar]
- 23.Hirakata Y, Srikumar R, Poole K, et al. Multidrug efflux systems play an important role in the invasiveness of Pseudomonas aeruginosa. J Exp Med. 2002;196:109–118. doi: 10.1084/jem.20020005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Finck-Barbancon V, Goranson J, Zhu L, et al. ExoU expression by Pseudomonas aeruginosa correlates with acute cytotoxicity and epithelial injury. Mol Microbiol. 1997;25:547–557. doi: 10.1046/j.1365-2958.1997.4891851.x. [DOI] [PubMed] [Google Scholar]
- 25.Cowell BA, Twining SS, Hobden JA, Kwong MS, Fleiszig SM. Mutation of lasA and lasB reduces Pseudomonas aeruginosa invasion of epithelial cells. Microbiology. 2003;149:2291. doi: 10.1099/mic.0.26280-0. [DOI] [PubMed] [Google Scholar]
- 26.Fleiszig SM, Zaidi TS, Pier GB. Pseudomonas aeruginosa invasion of and multiplication within corneal epithelial cells in vitro. Infect Immun. 1995;63:4072–4077. doi: 10.1128/iai.63.10.4072-4077.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fleiszig SM, Wiener-Kronish JP, Miyazaki H, et al. Pseudomonas aeruginosa-mediated cytotoxicity and invasion correlate with distinct genotypes at the loci encoding exoenzyme S. Infect Immun. 1997;65:579–586. doi: 10.1128/iai.65.2.579-586.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bakaletz LO, Baker BD, Jurcisek JA, et al. Demonstration of type IV pilus expression and a twitching phenotype by Haemophilus influenzae. Infect Immun. 2005;73:1635–1643. doi: 10.1128/IAI.73.3.1635-1643.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bradley DE. A function of Pseudomonas aeruginosa PAO polar pili: twitching motility. Can J Microbiol. 1980;26:146–154. doi: 10.1139/m80-022. [DOI] [PubMed] [Google Scholar]
- 30.Mattick JS. Type IV pili and twitching motility. Annu Rev Microbiol. 2002;56:289–314. doi: 10.1146/annurev.micro.56.012302.160938. [DOI] [PubMed] [Google Scholar]
- 31.Merz AJ, So M, Sheetz MP. Pilus retraction powers bacterial twitching motility. Nature. 2000;407:98–102. doi: 10.1038/35024105. [DOI] [PubMed] [Google Scholar]
- 32.Varga JJ, Nguyen V, O’Brien DK, Rodgers K, Walker RA, Melville SB. Type IV pili-dependent gliding motility in the Gram-positive pathogen Clostridium perfringens and other Clostridia. Mol Microbiol. 2006;62:680–694. doi: 10.1111/j.1365-2958.2006.05414.x. [DOI] [PubMed] [Google Scholar]
- 33.Whitchurch CB, Hobbs M, Livingston SP, Krishnapillai V, Mattick JS. Characterisation of a Pseudomonas aeruginosa twitching motility gene and evidence for a specialised protein export system widespread in eubacteria. Gene. 1991;101:33–44. doi: 10.1016/0378-1119(91)90221-v. [DOI] [PubMed] [Google Scholar]
- 34.Whitchurch CB, Mattick JS. Characterization of a gene, pilU, required for twitching motility but not phage sensitivity in Pseudomonas aeruginosa. Mol Microbiol. 1994;13:1079–1091. doi: 10.1111/j.1365-2958.1994.tb00499.x. [DOI] [PubMed] [Google Scholar]
- 35.Zolfaghar I, Evans DJ, Fleiszig SM. Twitching motility contributes to the role of pili in corneal infection caused by Pseudomonas aeruginosa. Infect Immun. 2003;71:5389–5393. doi: 10.1128/IAI.71.9.5389-5393.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Comolli JC, Hauser AR, Waite L, Whitchurch CB, Mattick JS, Engel JN. Pseudomonas aeruginosa gene products PilT and PilU are required for cytotoxicity in vitro and virulence in a mouse model of acute pneumonia. Infect Immun. 1999;67:3625–3630. doi: 10.1128/iai.67.7.3625-3630.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jendrossek V, Fillon S, Belka C, Muller I, Puttkammer B, Lang F. Apoptotic response of Chang cells to infection with Pseudomonas aeruginosa strains PAK and PAO-I: molecular ordering of the apoptosis signaling cascade and role of type IV pili. Infect Immun. 2003;71:2665–2673. doi: 10.1128/IAI.71.5.2665-2673.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jumblatt MM, Neufeld AH. Beta-adrenergic and serotonergic responsiveness of rabbit corneal epithelial cells in culture. Invest Ophthalmol Vis Sci. 1983;24:1139–1143. [PubMed] [Google Scholar]
- 39.Kruszewski FH, Walker TL, DiPasquale LC. Evaluation of a human corneal epithelial cell line as an in vitro model for assessing ocular irritation. Fundam Appl Toxicol. 1997;36:130–140. [PubMed] [Google Scholar]
- 40.Watson AA, Mattick JS, Alm RA. Functional expression of heterologous type 4 fimbriae in Pseudomonas aeruginosa. Gene. 1996;175:143–150. doi: 10.1016/0378-1119(96)00140-0. [DOI] [PubMed] [Google Scholar]
- 41.Fleiszig SM, Evans DJ, Do N, Vallas V, Shin S, Mostov KE. Epithelial cell polarity affects susceptibility to Pseudomonas aeruginosa invasion and cytotoxicity. Infect Immun. 1997;65:2861–2867. doi: 10.1128/iai.65.7.2861-2867.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sosnova-Netukova M, Kuchynka P, Forrester JV. The suprabasal layer of corneal epithelial cells represents the major barrier site to the passive movement of small molecules and trafficking leukocytes. Br J Ophthalmol. 2007;91:372–378. doi: 10.1136/bjo.2006.097188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zolfaghar I, Angus AA, Kang PJ, To A, Evans DJ, Fleiszig SM. Mutation of retS, encoding a putative hybrid two-component regulatory protein in Pseudomonas aeruginosa, attenuates multiple virulence mechanisms. Microbes Infect. 2005;7:1305–1316. doi: 10.1016/j.micinf.2005.04.017. [DOI] [PubMed] [Google Scholar]
- 44.High N, Mounier J, Prevost MC, Sansonetti PJ. IpaB of Shigella flexneri causes entry into epithelial cells and escape from the phagocytic vacuole. EMBO J. 1992;11:1991–1999. doi: 10.1002/j.1460-2075.1992.tb05253.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gaillard JL, Berche P, Mounier J, Richard S, Sansonetti P. In vitro model of penetration and intracellular growth of Listeria monocytogenes in the human enterocyte-like cell line Caco-2. Infect Immun. 1987;55:2822–2829. doi: 10.1128/iai.55.11.2822-2829.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Smith GA, Marquis H, Jones S, Johnston NC, Portnoy DA, Goldfine H. The two distinct phospholipases C of Listeria monocytogenes have overlapping roles in escape from a vacuole and cell-to-cell spread. Infect Immun. 1995;63:4231–4237. doi: 10.1128/iai.63.11.4231-4237.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sansonetti PJ, Mounier J, Prevost MC, Mege RM. Cadherin expression is required for the spread of Shigella flexneri between epithelial cells. Cell. 1994;76:829–839. doi: 10.1016/0092-8674(94)90358-1. [DOI] [PubMed] [Google Scholar]
- 48.Tilney LG, Portnoy DA. Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes. J Cell Biol. 1989;109:1597–1608. doi: 10.1083/jcb.109.4.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sano G, Takada Y, Goto S, et al. Flagella facilitate escape of Salmonella from oncotic macrophages. J Bacteriol. 2007;189:8224–8232. doi: 10.1128/JB.00898-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hybiske K, Stephens RS. Mechanisms of host cell exit by the intracellular bacterium Chlamydia. Proc Natl Acad Sci U S A. 2007;104:11430–11435. doi: 10.1073/pnas.0703218104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rosenthal PJ. Cysteine proteases of malaria parasites. Int J Parasitol. 2004;34:1489–1499. doi: 10.1016/j.ijpara.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 52.Grassme H, Kirschnek S, Riethmueller J, et al. CD95/CD95 ligand interactions on epithelial cells in host defense to Pseudomonas aeruginosa. Science. 2000;290:527–530. doi: 10.1126/science.290.5491.527. [DOI] [PubMed] [Google Scholar]