Abstract
Purpose
We assessed Tau protein expression in primary breast cancer specimens of patients included in the National Surgical Breast and Bowel Project (NSABP) -B 28 clinical trial. Expression levels were correlated with disease-free survival (DFS) and overall survival (OS). Interaction between this marker and paclitaxel efficacy was also examined.
Methods
Tissue microarrays were available for 1,942 patients (63% of all trial participants) who were randomly assigned to receive either four courses of doxorubicin and cyclophosphamide (AC) or AC followed by four courses of adjuvant paclitaxel chemotherapy. All patients who were hormone receptor positive received adjuvant endocrine therapy. Immunohistochemistry was used to measure Tau expression at M. D. Anderson Cancer Center blinded to clinical outcome. Correlation between Tau and OS and DFS was performed by the NSABP Biostatistical Center.
Results
Forty-three percent of patients were Tau positive. Tau positivity correlated with estrogen receptor (ER) -positive status (P < .0001), lower histologic grade (P < .001), and lack of human epidermal growth factor receptor 2 (HER2) expression (P < .0001). In univariate analyses, Tau- and ER-positive status were both associated with better DFS and OS (P < .0001) whereas greater tumor size, high grade, lymph node- and HER2-positive status were each associated with worse DFS and OS (P < .0001). In multivariate analysis, the same variables except HER2 remained significant. There was no significant interaction between Tau expression and benefit from paclitaxel in the total population or among ER-positive or -negative patients.
Conclusion
High Tau protein expression is associated with better prognosis in patients treated with adjuvant anthracycline and paclitaxel chemotherapy and endocrine therapy, but there was no significant interaction between Tau expression and paclitaxel benefit.
INTRODUCTION
Gene expression profiling experiments suggest that low expression of microtubule associated protein Tau mRNA in pretreatment cancer biopsies is associated with higher rates of pathologic complete response (pCR) to preoperative sequential paclitaxel, followed by fluorouracil, cyclophosphamide, and doxorubicin chemotherapy.1,2 Immunohistochemical evaluation of 122 breast cancer specimens from patients who received similar preoperative chemotherapy confirmed the inverse relationship between Tau protein expression and pCR rate.3 These studies also showed that Tau expression was higher in estrogen receptor (ER) -positive and low grade cancers compared to ER-negative, high grade tumors. However, the Tau/pCR odds ratio remained high (2.7) with borderline significance (95% CI, 0.9 to 7.9; P = .059) even after adjustment for age, tumor size, nodal status, nuclear grade, and ER and HER2 status. This suggested a possible independent predictive value for this protein.3 In vitro functional experiments indicated that downregulation of Tau expression with small interfering RNA in breast cancer cell lines increased sensitivity to paclitaxel but not to epirubicin.3,4 Further mechanistic studies suggested that Tau partially protected microtubules from paclitaxel binding in vitro and therefore low levels in a cell could render microtubules more accessible and therefore more vulnerable to this drug. Several other investigators also reported correlation between low Tau expression and greater chemotherapy sensitivity. Pancreatic cell lines with low Tau expression showed increased sensitivity to docetaxel.5 Impaired paclitaxel-induced tubulin polymerization was seen in MCF7 breast cancer cells with high Tau expression.6 Reduced Tau expression also correlated with better clinical response to paclitaxel in patients with gastric cancer.7 However, not all studies confirmed this inverse correlation between Tau and chemotherapy sensitivity. Gene expression profiling of tumor samples from breast cancer patients who received docetaxel, doxorubicin, and cyclophosphamide combination chemotherapy failed to identify Tau as a predictor of response.8 Similarly, an immunohistochemical analysis of tumor samples from patients with locally advanced or metastatic breast cancer treated with either docetaxel or doxorubicin also failed to show correlation between Tau expression and response to these drugs.9
The goal of this study was to examine the prognostic value of Tau protein expression and to assess interaction between Tau expression and benefit from paclitaxel. We used tissue specimens from the National Surgical Breast and Bowel Project (NSABP) -B28 clinical trial that randomly assigned women to either doxorubicin and cyclophosphamide (AC; n = 1,529) or doxorubicin, cyclophosphamide, and paclitaxel (AC-T; n = 1,531) adjuvant chemotherapy. Inclusion of paclitaxel improved the median 5-year disease-free survival (DFS) by approximately 15% (76% v 72%; P = .006) compared to AC alone but there was no significant difference in overall survival (OS).10 Our hypothesis was that patients with low Tau expression benefited the most from paclitaxel whereas those with high Tau expression benefited less.
PATIENTS AND METHODS
Tumor Samples and Tissue Microarrays
Tissue microarrays (TMA) were constructed from formaldehyde-fixed, paraffin-embedded primary breast cancer tissues of patients who received therapy on the NSABP-B28 randomized clinical trial. Specimens on the TMA represent tissues from the original diagnostic specimen. Patients assigned to the AC arm received doxorubicin 60 mg/m2 and cyclophosphamide 600 mg/m2 every 21 days for four cycles. Patients assigned to the AC-T arm also received four additional cycles of paclitaxel 225 mg/m2 as a 3-hour infusion every 21 days after completion of the four courses of AC. All patients who had hormone receptor–positive tumors and those who were 50 years or older also received tamoxifen 20 mg daily for 5 years concomitant with chemotherapy beginning on day 1 of the first course of AC. All patients treated with lumpectomy also received whole-breast irradiation. This correlative science study was approved by the institutional review board of the University of Texas M. D. Anderson Cancer Center and by the NSABP.
Immunohistochemistry for Tau
The TMAs were constructed by the NSABP Pathology Department and were mailed to investigators at M. D. Anderson Cancer Center for immunohistochemical analysis who were blinded to clinical outcome. Immunohistochemistry and scoring were performed as described previously.3 Cytoplasmic expression of Tau protein in normal breast epithelium and blood vessels were considered as internal positive controls and as reference for scoring. Tumors with no Tau staining were scored as immunohistochemistry (IHC) 0, tumors with Tau expression less than in normal breast epithelial cells were scored as IHC 1+. Cancers with staining intensity that was similar to normal epithelium were scored as IHC 2+, and tumors with uniformly high cytoplasmic staining greater than normal cells were scored as IHC 3+. Tumors with IHC scores of 0 or 1 were considered to be Tau negative (ie, showed no or lower Tau expression than normal breast epithelium) and cases with scores 2+ or 3+ were considered to be Tau positive. The TMA slides were scored independently by two pathologists (Y.G., J.S.R.). Discrepant cases were resolved in consultation with a third pathologist (W.F.S.) and final results were returned to the NSABP Biostatistical Center for correlation with clinical outcome.
Statistical Analysis
Two survival end points were used in this analysis including DFS and OS that were nonparametrically estimated by the Kaplan-Meier method. The DFS was calculated as time from study entry to either local, regional, or any distant recurrence, or death from any cause before recurrence, or second primary cancer. The OS end point was calculated from study entry to death from any cause. The results presented here are based on follow-up data with the cutoff date of December 31, 2007. The mean time on study was 131 months. Differences in survival end points between the two treatment groups were assessed by the two-sided log-rank test. The χ2 test was used to compare associations between Tau expression and routine clinical variables. Both univariate and multivariate proportional hazards models were developed to relate Tau expression, age, tumor size, histologic grade, lymph node status, ER and HER2 expression with DFS and OS. The interaction between Tau expression and paclitaxel efficacy was performed by testing the logarithm of the ratio of two hazard ratios. All reported P values are two sided and P < .05 was considered significant.
RESULTS
Patient Characteristics and Tau Expression
Tumor tissue was available for TMA construction from 1,942 patients that represented 63% of all clinical trial participants. Clinical and biomarker characteristics of these patients were similar to the whole trial population (Appendix Table A1). Tau staining was mostly homogenous among specimens from the same cases. Eight percent (n = 228) of all IHC scores determined independently by two pathologists were discordant for Tau status (ie, Tau negative by one and Tau positive by the other). A third pathologist reviewed all discrepant cases in order to obtain a consensus. After the final scoring, 43% of all cancers (n = 833) were Tau positive (ie, ≥ Tau expression than normal breast epithelium). Tau expression was more frequent among ER-positive cancers, 57% of these tumors were Tau positive compared with 15% of ER-negative tumors (P < .0001). Tau expression was also associated with lower grade, close to 60% of low grade cancers (n = 103 of 182) were Tau positive compared to only 30% of high grade tumors (P < .0001). We also observed an inverse correlation between Tau and HER2 HER-2 expression, only 20% of HER2-positive patients were Tau positive (P < .001). Tau expression did not correlate with tumor size, nodal status, or patient age (Table 1).
Table 1.
Correlation Between Tau Expression and Clinical Variables
| Variable | Tau |
P | |
|---|---|---|---|
| Negative | Positive | ||
| ER | |||
| Negative | 557 | 98 | |
| Positive | 551 | 736 | < .0001 |
| PR | |||
| Negative | 615 | 146 | |
| Positive | 493 | 688 | < .0001 |
| Lymph nodes | |||
| 1-3 | 777 | 572 | |
| 4-9 | 288 | 225 | |
| 10+ | 43 | 37 | .71 |
| Age | |||
| ≤ 49 | 559 | 421 | |
| 50-59 | 351 | 245 | |
| ≥ 60 | 198 | 168 | .35 |
| Tumor size | |||
| 2.0 | 605 | 493 | |
| 2.1-4.0 | 399 | 280 | |
| > 4 | 103 | 60 | .08 |
| HER2 | |||
| Negative | 718 | 619 | |
| Positive | 224 | 55 | < .0001 |
| Grade | |||
| Low | 79 | 103 | |
| Intermediate | 330 | 407 | |
| High | 653 | 284 | < .0001 |
Abbreviations: ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2.
Tau Expression and DFS and OS
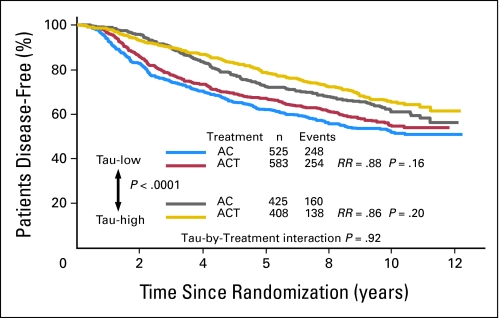
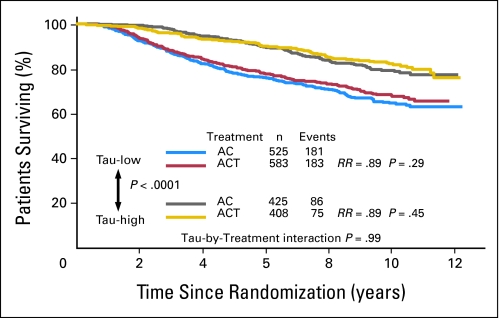
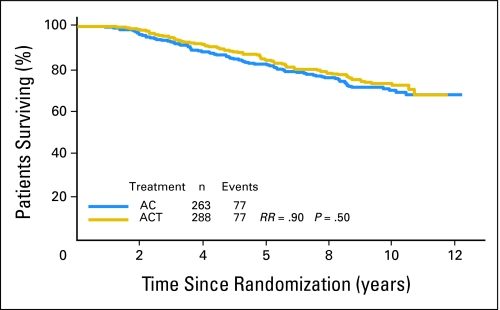
The median DFS has not been reached yet but there was a highly significant difference in DFS between Tau-negative and Tau-positive patients (P < .0001; Fig 1). Tau-positive tumors had better DFS. However, there was no significant interaction between Tau expression and paclitaxel effect (P = .92). The DFS of patients with Tau-negative tumors was not statistically different when treated with AC-T compared to AC (risk ratio [RR], 0.88 favoring paclitaxel; P = .16). Similar results were observed among the Tau-positive patients (RR, 0.86; P = .20). When OS was analyzed in a similar manner, the same results were observed; patients with Tau-positive tumors had better OS (Fig 2).
Fig 1.
Disease-free survival of all patients by treatment arm and Tau status. Kaplan-Meier survival curves are shown. Tau-low cases correspond to Tau-negative immunohistochemistry results. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Fig 2.
Overall survival of all patients by treatment arm and Tau status. Kaplan-Meier survival curves are shown. Tau-low cases correspond to Tau-negative immunohistochemistry results. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
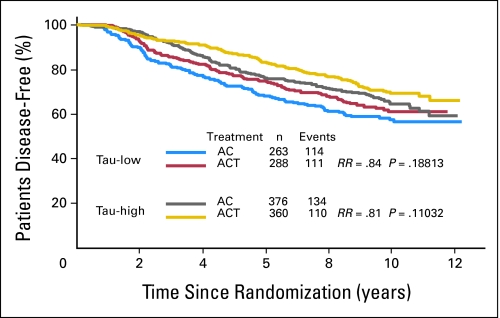
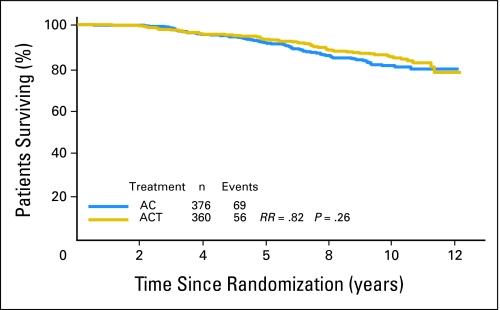
Tau is an ER-regulated gene and its expression can be induced by both estrogen and tamoxifen in vitro.11–13 High Tau expression in ER-positive cancers was also associated with better survival when patients received 5 years of adjuvant tamoxifen therapy without chemotherapy.14 These observations suggested high Tau levels are associated with increased endocrine sensitivity, but at the same time predict for lower chemotherapy sensitivity in ER-positive tumors. This opposing bifunctional predictive role could complicate interpretation of survival curves in patients who receive both treatment modalities. Therefore, we examined the interaction between Tau and survival in ER-negative and -positive cases separately. Among the ER-positive patients (n = 1,287), patients with Tau-positive cancers (n = 736) had better DFS (Fig 3) and OS (Appendix Figs A1, A2, online only) compared to Tau-negative tumors, but there was no significant interaction between Tau expression and benefit from paclitaxel. Both Tau categories showed a nonsignificant trend for better survival with AC-T compared to AC. It is important to recognize that in the B-28 clinical trial all ER-positive patients received tamoxifen concomitant with chemotherapy, therefore Tau levels during treatment could have been higher (due to tamoxifen) than at baseline measured by the tissue arrays.
Fig 3.
Disease-free survival of estrogen receptor–positive patients by treatment arm and Tau status. Kaplan-Meier survival curves are shown. Tau-low cases correspond to Tau-negative immunohistochemistry results. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
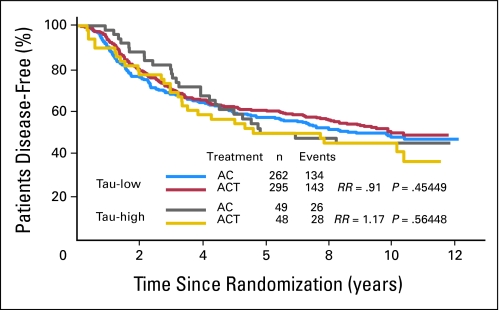
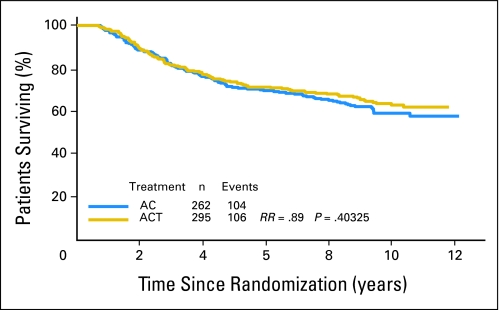
Among the ER-negative patients (n = 655), Tau expression had no prognostic value. Patients with ER-negative but Tau-positive tumors (n = 97) had similar outcome as Tau-negative tumors (n = 557). There was a small, nonsignificant trend among the ER- and Tau-negative patients for better DFS (RR, 0.91; P = .45; Fig 4) and OS (RR, 0.89; P = .40) (Appendix Fig A3, online only) with AC-T relative to AC. In contrast, in the ER-negative and Tau-positive patients, there was an nonsignificant advantage in DFS (RR, 1.17; P = .56) and OS (RR, 1.24; P = .5) when treated with AC alone (Appendix Fig A4, online only). Taken together, these data do not support that Tau immunohistochemistry is clinically useful to determine which patient will benefit from paclitaxel adjuvant therapy.
Fig 4.
Disease-free survival of estrogen receptor–negative patients by treatment arm and Tau status. Kaplan-Meier survival curves are shown. Tau-low cases correspond to Tau negative immunohistochemistry results. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Univariate and Multivariate Analyses
Results from the univariate proportional hazards analyses for DFS and OS including all cases are presented in Appendix Table A2 (online only). Tau- and ER-positive statuses were both associated with significantly (P < .0001) better DFS and OS. In contrast, greater tumor size, higher grade, lymph node involvement, and HER2-positive status were associated with worse DFS and OS (P < .0001). In multivariate analysis, the same variables except HER2 remained significant as presented in Table 2. We also performed these analyses separately for ER-positive and ER-negative cancers. Among ER-positive patients, Tau-positive status was associated with significantly better DFS and OS in both univariate (Appendix Table A3, online only) and multivariate (Table 2) analyses. Tau had no prognostic value in ER-negative patients. These results indicate that Tau-positive status is an independent good prognostic marker in ER-positive breast cancer treated with endocrine therapy and chemotherapy.
Table 2.
Results of Multivariate Proportional Hazards Models for DFS and OS for All Patients and for ER-Positive Patients
| Parameter | All DFS |
All OS |
ER+ |
|||||
|---|---|---|---|---|---|---|---|---|
| DFS |
OS |
|||||||
| HR | P | HR | P | HR | P | HR | P | |
| Treatment | 0.85 | .032 | 0.87 | .16 | 0.90 | .033 | 0.95 | .68 |
| Age, years | ||||||||
| ≤ 49 | Reference | Reference | ||||||
| 50-59 | 0.93 | .41 | 0.91 | .43 | 1.05 | 0.66 | 1.13 | .44 |
| > 60 | 1.04 | .75 | 1.41 | .0046 | 1.10 | 0.52 | 1.73 | .0014 |
| Tumor size, cm | ||||||||
| < 2.0 | Reference | Reference | ||||||
| 2.1-4.0 | 1.17 | .066 | 1.29 | .014 | 1.32 | 0.015 | 1.41 | .017 |
| > 4.0 | 1.40 | .011 | 1.38 | .044 | 2.31 | < 0.0001 | 2.21 | .00016 |
| Histologic grade | ||||||||
| Low | Reference | Reference | ||||||
| Intermediate | 1.54 | .015 | 1.83 | .016 | 1.52 | 0.032 | 1.82 | .039 |
| High | 1.61 | .0077 | 1.99 | .0064 | 1.84 | 0.0022 | 2.62 | .00077 |
| Lymph nodes, No. | ||||||||
| 0-3 | Reference | Reference | ||||||
| 4-9 | 1.75 | < .0001 | 1.95 | < .0001 | 1.89 | < 0.0001 | 2.10 | < .0001 |
| > 10 | 3.07 | < .0001 | 3.23 | < .0001 | 2.32 | 0.00025 | 1.84 | < .040 |
| ER | 0.73 | .00047 | 0.65 | < .0001 | NA | NA | NA | NA |
| HER2 | 1.21 | .055 | 1.21 | .10 | 1.45 | .0069 | 1.59 | .0053 |
| Tau | 0.80 | .018 | 0.63 | < .0001 | 0.78 | .018 | 0.58 | < .001 |
Abbreviations: DFS, disease-free survival; OS, overall survival; ER+, estrogen receptor–positive patients; NA, not applicable; HR, hazard ratio; HER2, human epidermal growth factor receptor 2.
DISCUSSION
In this study, we examined the prognostic value of Tau in patients who received sequential AC-T or AC alone adjuvant chemotherapies in a large randomized clinical trial. The hypothesis behind this study was that patients with low Tau expression (ie, Tau negative) will preferentially benefit from the inclusion of paclitaxel in their adjuvant chemotherapy regimen. However, we observed no statistically significant interaction between low Tau-expression and benefit from adjuvant paclitaxel. Since the original report that linked Tau with increased sensitivity to chemotherapy, it also became apparent that Tau expression correlates closely with endocrine sensitivity in ER-positive cancers.14 Tau itself is an ER- and tamoxifen-induced gene. High Tau expression (ie, Tau-positive status) therefore may in fact predict for better survival when all breast cancer patients are considered because it identifies particularly endocrine sensitive, ER-positive patients for whom chemotherapy resistance is of limited relevance. Indeed, we observed this association in this study. Tau-positive patients had significantly better survival than patients with Tau-negative cancers, despite their presumed lesser chemotherapy sensitivity. Similar observations were also reported by others.15,16 We also examined the prognostic value of Tau separately in ER-positive patients only. Multivariate analysis indicated that Tau-positive status remained a significant predictor of survival even after adjusting for histologic grade, tumor size, nodal status, and age in ER-positive cancers (Appendix Table A3). These findings are consistent with a predictive value for endocrine sensitivity as it was observed previously.14
Tau illustrates several important caveats in predictive marker discovery. Despite clear recognition that ER-positive and ER-negative breast cancers are molecularly distinct diseases, many biomarker studies analyze these breast cancers together, mostly because this is how clinical studies were conducted in the past. Low Tau expression was identified as a predictor of pCR to neoadjuvant chemotherapy from such combined analysis. ER-negative, high grade, and HER2-positive cancers are known to have higher rates of pCR compared to other types of breast cancers.17 Low Tau expression is closely associated with these tumor characteristics and therefore with pCR. In this study, 85% of ER-negative cancers, 80% of HER2-positive tumors, and 70% of high-grade cancers were Tau negative. These associations explain to a large extent the association between low Tau expression and increased pCR rate. However, even after adjusting for ER status, low Tau expression was predictive of pCR in previous studies, while in this study only a very weak nonsignificant numerical trend was observed for greater paclitaxel sensitivity among ER-negative/Tau-negative patients. This highlights the importance of identifying chemotherapy sensitivity markers separately for ER-positive and ER-negative cancers to avoid discovery of clinical phenotype predictors. Such analysis will require large discovery sample sizes because response rates may be low in certain molecular subgroups (for example pCR in ER-positive cancers).
Another important consideration is the complex interaction between chemotherapy sensitivity markers and survival in adjuvant trials. Survival after stage I to III breast cancer is influenced by baseline prognosis (ie, probability of survival without any systemic therapy), sensitivity to chemotherapy, and sensitivity to endocrine therapy among hormone receptor-positive patients, and more recently sensitivity to trastuzumab among the HER2-positive patients. It is clear from several recent studies that the same marker can have multiple and often inverse interaction with more than one of these variables that influence survival. For example, high Oncotype Dx (Genomic Health, Redwood CA) recurrence score (RS) is associated with worse survival among ER-positive patients who receive adjuvant tamoxifen but at the same time it also predicts for greater benefit from adjuvant chemotherapy implying that chemotherapy sensitivity is more frequent among these tumors compared to tumors with low RS.18,19 However, patients with high RS, even with adjuvant chemotherapy, do worse than patients with low RS without chemotherapy suggesting that even though this former group includes more patients with chemotherapy sensitive tumors the absolute number of such cases is still low. The greater degree of chemotherapy benefit in this subset does not fully compensate for the lesser endocrine sensitivity and poor baseline prognosis. Similar, but less consistent, inverse association between survival, endocrine sensitivity, and chemotherapy sensitivity may exist for proliferation markers such as Ki67. High Ki67 expression at baseline is predictive of better response to preoperative chemotherapy in general,20 but it is also predictive of lesser endocrine sensitivity among ER-positive patients,21,22 poorer baseline prognosis, and therefore worse OS in general.23,24
An important question remains why analysis of this large randomized study could not confirm the preclinical observation that low Tau expression confers selective sensitivity to paclitaxel as it was seen in breast cancer cell lines. Observations in breast cancer cell lines may only capture the behavior of only relatively small subsets of human breast cancers.25 For example, a large body of in vitro evidence supports the role of drug efflux proteins in drug resistance but only very few patients benefit from inhibition of drug efflux proteins in the clinic.26,27 Similarly, inhibition of epidermal growth factor receptor inhibits the growth of breast cancer cells in vitro but the clinical activity of epidermal growth factor receptor inhibitors in breast cancer patients is limited.28 Many other examples in the literature suggest that molecular pathways that mediate drug resistance or represent effective targets for drug development in experimental cancer models play a similarly critical biologic role in only a small subset of patients. In this study, among the ER-negative patients where the predictive value of low Tau expression for higher paclitaxel response and consequently better survival is expected to be seen the most clearly (because of no interaction with endocrine therapy), there was indeed a small numerical advantage from inclusion of paclitaxel. The event rates were 48% (n = 143 of 295) among the ER- and Tau-negative cases treated with AC-T compared to 51% (n = 134 of 262) in those treated with AC alone (Fig 4). No similar numerical advantage from inclusion of paclitaxel was seen among the ER-negative and Tau-positive patients. This shows that if low Tau levels confer paclitaxel sensitivity, this is clinically relevant in only a very small (< 5%) patient population. Considering the numerous molecular events that can cause resistance to chemotherapy in general and paclitaxel in particular it may have been naïve to assume that a single molecular mechanism that increases vulnerability of microtubules to paclitaxel will have clinical value to define a drug sensitive population. Finally, in this trial, patients received tamoxifen concomitant with chemotherapy and tamoxifen is known to induce Tau expression in vitro.13 Tau levels during the treatment could have been higher than at baseline and this effect could also weaken correlation between pretreatment Tau expression and paclitaxel sensitivity.
In summary, these data show that Tau expression is not clinically useful to select or exclude patients for adjuvant paclitaxel chemotherapy. The previously reported association between low Tau and pCR is largely due to its phenotypic association with ER-negative, high-grade, and HER2-positive tumors. However, Tau-positive status is predictive of good overall prognosis that is partly independent of these phenotypic associations and may be due to greater endocrine sensitivity of Tau-positive and ER-positive tumors.
Appendix
Fig A1.
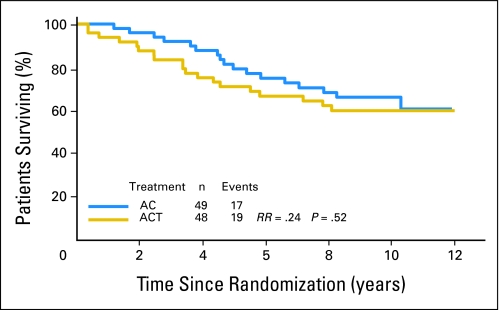
Overall survival of estrogen receptor–positive, Tau-negative patients by treatment arm. Kaplan-Meier survival curves are shown. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Fig A2.
Overall survival of estrogen receptor–positive, Tau-positive patients by treatment arm. Kaplan-Meier survival curves are shown. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Fig A3.
Overall survival of estrogen receptor–negative, Tau-negative patients by treatment arm. Kaplan-Meier survival curves are shown. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Fig A4.
Overall survival of estrogen receptor–negative, Tau-positive patients by treatment arm. Kaplan-Meier survival curves are shown. AC, doxorubicin and cyclophosphamide; ACT, AC and paclitaxel; RR, risk ratio.
Table A1.
Characteristics of Patients Included in This Biomarker Study
| Characteristic | Treatment |
|||||
|---|---|---|---|---|---|---|
| AC |
AC-T |
Total |
||||
| No. | % | No. | % | No. | % | |
| Total cases on TMA | 950 | 100 | 992 | 100 | 1942 | 100 |
| Age, years | ||||||
| ≤ 49 | 473 | 50 | 507 | 51 | 980 | 50 |
| 50-59 | 297 | 31 | 299 | 30 | 596 | 31 |
| > 60 | 180 | 19 | 186 | 19 | 366 | 19 |
| Size, cm* | ||||||
| ≤ 2.0 | 545 | 57 | 553 | 56 | 1098 | 56 |
| 2.1-4.0 | 330 | 35 | 349 | 35.2 | 679 | 35 |
| > 4.0 | 74 | 8 | 89 | 9.0 | 163 | 8 |
| Lymph nodes, No. | ||||||
| 1-3 | 663 | 70 | 686 | 69 | 1349 | 69 |
| 4-9 | 250 | 26 | 263 | 26 | 513 | 26 |
| > 10 | 37 | 4 | 43 | 4 | 80 | 4 |
| Breast conserving surgery | 420 | 44 | 449 | 45 | 869 | 45 |
| Modified radical mastectomy | 530 | 56 | 543 | 55 | 1073 | 55 |
| Estrogen receptor | ||||||
| Negative | 311 | 33 | 344 | 35 | 655 | 34 |
| Positive | 639 | 67 | 648 | 65 | 1287 | 66 |
| Progesterone receptor | ||||||
| Negative | 364 | 38 | 397 | 40 | 761 | 39 |
| Positive | 586 | 62 | 595 | 60 | 1181 | 61 |
| HER2 | ||||||
| Negative | 651 | 68 | 686 | 69 | 1337 | 69 |
| Positive | 131 | 14 | 148 | 15 | 279 | 14 |
| Unknown | 168 | 18 | 158 | 16 | 326 | 17 |
| Grade | ||||||
| Low | 83 | 9 | 99 | 10 | 182 | 9 |
| Intermediate | 366 | 38 | 372 | 37 | 738 | 38 |
| High | 457 | 48 | 480 | 48 | 937 | 48 |
| Unknown | 44 | 5 | 41 | 4 | 85 | 4 |
| Tau | ||||||
| Negative | 525 | 55 | 583 | 59 | 1108 | 57 |
| Positive | 425 | 45 | 408 | 41 | 833 | 43 |
| Tamoxifen | ||||||
| Yes | 809 | 85 | 844 | 85 | 1653 | 85 |
| No | 141 | 14 | 148 | 15 | 289 | 15 |
Abbreviations: AC, doxorubicin and cyclophosphamide; AC-T, doxorubicin, cyclophosphamide, and paclitaxel; TMA, tissue microarray; HER2, human epidermal growth factor receptor 2.
The size of two cancers was unknown.
Table A2.
Results of Univariate Proportional Hazards Models for DFS and OS for All Patients
| Parameter | DFS |
OS |
||
|---|---|---|---|---|
| HR | P | HR | P | |
| Age, years | ||||
| ≤ 49 | Reference | Reference | ||
| 50-59 | 0.94 | .44 | 0.91 | .36 |
| > 60 | 1.22 | .21 | 1.46 | .0005 |
| Tumor size, cm | ||||
| < 2.0 | Reference | Reference | ||
| 2.1-4.0 | 1.45 | < .0001 | 1.62 | < .0001 |
| > 4.0 | 1.92 | < .0001 | 1.94 | < .0001 |
| Histologic grade | ||||
| Low | Reference | Reference | ||
| Intermediate | 1.61 | .0023 | 2.36 | .00028 |
| High | 2.14 | < .0001 | 3.57 | < .0001 |
| Lymph nodes, No. | ||||
| 0-3 | Reference | Reference | ||
| 3-9 | 1.81 | < .0001 | 1.97 | < .0001 |
| ≥ 10 | 2.95 | < .0001 | 3.61 | < .0001 |
| Estrogen receptor | ||||
| Negative | Reference | Reference | ||
| Positive | 0.58 | < .0001 | 0.48 | < .0001 |
| HER2 | ||||
| Negative | Reference | Reference | ||
| Positive | 1.51 | < .0001 | 1.59 | < .0001 |
| Tau | ||||
| Negative | Reference | Reference | ||
| Positive | 0.69 | < .0001 | 0.52 | < .0001 |
Abbreviations: DFS, disease free-survival; OS, overall survival; HR, hazard ratio; HER2, human epidermal growth factor receptor 2.
Table A3.
Results of Univariate Proportional Hazards Models for DFS and OS for ER-Positive Patients Only
| Parameter | DFS |
OS |
||
|---|---|---|---|---|
| HR | P | HR | P | |
| Age, years | ||||
| ≤ 49 | Reference | Reference | ||
| 50-59 | 0.98 | .85 | 0.99 | .95 |
| > 60 | 1.06 | .64 | 1.47 | .01 |
| Tumor size, cm | ||||
| < 2.0 | Reference | Reference | ||
| 2.1-4.0 | 1.59 | < .0001 | 1.76 | < .0001 |
| > 4.0 | 2.60 | < .0001 | 2.60 | < .0001 |
| Histologic grade | ||||
| Low | Reference | Reference | ||
| Intermediate | 1.57 | .0093 | 2.27 | .0028 |
| High | 2.26 | < .0001 | 3.81 | < .0001 |
| Lymph nodes, No. | ||||
| 0-3 | Reference | Reference | ||
| 3-9 | 2.00 | < .0001 | 2.19 | < .0001 |
| ≥ 10 | 2.55 | < .0001 | 2.92 | < .0001 |
| HER2 | ||||
| Negative | Reference | Reference | ||
| Positive | 1.71 | < .0001 | 1.96 | < .0001 |
| Tau | ||||
| Negative | Reference | Reference | ||
| Positive | 0.73 | .0007 | 0.55 | < .0001 |
Abbreviations: DFS, disease free-survival; OS, overall survival; ER, estrogen receptor; HR, hazard ratio; HER2, human epidermal growth factor receptor 2.
Footnotes
Supported by a grant from the Breast Cancer Research Foundation (L.P.) and by the Nellie B. Connally Breast Cancer Research Fund. NSABP Trial B-28 was supported by Public Health Service Grants No. U10-CA-12027, U10-CA-69651, U10-CA-37377, U10-CA-69974, and U24-CA-114732 from the National Cancer Institute, Department of Health and Human Services.
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Clinical Trials repository link available on JCO.org.
Clinical trial information can be found for the following: NSABP B28.
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
The author(s) indicated no potential conflicts of interest.
AUTHOR CONTRIBUTIONS
Conception and design: Lajos Pusztai, Soonmyung Paik, Roman Rouzier, Fabrice Andre, Gabriel N. Hortobagyi, W. Fraser Symmans
Financial support: Lajos Pusztai, Gabriel N. Hortobagyi, Norman Wolmark, W. Fraser Symmans
Administrative support: Lajos Pusztai, Soonmyung Paik, Gabriel N. Hortobagyi, Norman Wolmark, W. Fraser Symmans
Provision of study materials or patients: Yun Gong, Jeffrey S. Ross, Chungyeul Kim, Soonmyung Paik, W. Fraser Symmans
Collection and assembly of data: Lajos Pusztai, Yun Gong, Jeffrey S. Ross, Chungyeul Kim, W. Fraser Symmans
Data analysis and interpretation: Lajos Pusztai, Jong-Hyeon Jeong, Yun Gong, Jeffrey S. Ross, Soonmyung Paik, Roman Rouzier, Fabrice Andre, W. Fraser Symmans
Manuscript writing: Lajos Pusztai, Jong-Hyeon Jeong, Soonmyung Paik
Final approval of manuscript: Lajos Pusztai, Jong-Hyeon Jeong, Yun Gong, Jeffrey S. Ross, Chungyeul Kim, Soonmyung Paik, Roman Rouzier, Fabrice Andre, Gabriel N. Hortobagyi, Norman Wolmark, W. Fraser Symmans
REFERENCES
- 1.Hess KR, Anderson K, Symmans WF, et al. Pharmacogenomic predictor of sensitivity to preoperative chemotherapy with paclitaxel and fluorouracil, doxorubicin, and cyclophosphamide in breast cancer. J Clin Oncol. 2006;24:4236–4244. doi: 10.1200/JCO.2006.05.6861. [DOI] [PubMed] [Google Scholar]
- 2.Peintinger F, Anderson K, Mazouni C, et al. Thirty-gene pharmacogenomic test correlates with residual cancer burden after preoperative chemotherapy breast cancer. Clin Cancer Res. 2007;13:4078–4082. doi: 10.1158/1078-0432.CCR-06-2600. [DOI] [PubMed] [Google Scholar]
- 3.Rouzier R, Rajan R, Wagner P, et al. Microtubule-associated protein tau: A marker of paclitaxel sensitivity in breast cancer. Proc Natl Acad Sci U S A. 2005;102:8315–8320. doi: 10.1073/pnas.0408974102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wagner P, Wang B, Clark E, et al. Microtubule associated protein (MAP)-tau: A novel mediator of paclitaxel sensitivity in vitro and in vivo. Cell Cycle. 2005;4:1149–1152. doi: 10.4161/cc.4.9.2038. [DOI] [PubMed] [Google Scholar]
- 5.Jimeno A, Hallur G, Chan A, et al. Development of two novel benzoylphenylurea sulfur analogues and evidence that the microtubule-associated protein tau is predictive of their activity in pancreatic cancer. Mol Cancer Ther. 2007;6:1509. doi: 10.1158/1535-7163.MCT-06-0592. [DOI] [PubMed] [Google Scholar]
- 6.Duran GE. Impaired paclitaxel- and epothilon-induced polymerization in non-MDR1 taxane-resistant MCF7 breast cancer variant with reduced MAP4 and elevated MAP-Tau content. Proc Annu Meet Am Assoc Cancer Res. 2006;47:883. abstr 3755. [Google Scholar]
- 7.Mimori K, Sadanaga N, Yoshikawa Y, et al. Reduced tau expression in gastric cancer can identify candidates for successful paclitaxel treatment. Br J Cancer. 2006;94:1894–1897. doi: 10.1038/sj.bjc.6603182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rody A, Karn T, Gatje R, et al. Gene expression profiling of breast cancer patients treated with docetaxel, doxorubicin, and cyclophosphamide within the GEPARTRIO trial: HER-2, but not topoisomerase II alpha and microtubule-associated protein tau, is highly predictive of tumor response. Breast. 2007;16:86–93. doi: 10.1016/j.breast.2006.06.008. [DOI] [PubMed] [Google Scholar]
- 9.Galmarini CM, Treilleux I, Cardoso F, et al. Class III beta-tubulin isotype predicts response in advanced breast cancer patients randomly treated either with single-agent doxorubicin or docetaxel. Clin Cancer Res. 2008;14:4511–4516. doi: 10.1158/1078-0432.CCR-07-4741. [DOI] [PubMed] [Google Scholar]
- 10.Mamounas EP, Bryant J, Lembersky BC, et al. Paclitaxel after doxorubicin plus cyclophosphamide as adjuvant chemotherapy for node-positive breast cancer: Results from NSABP-B 28. J Clin Oncol. 2005;23:3686–3696. doi: 10.1200/JCO.2005.10.517. [DOI] [PubMed] [Google Scholar]
- 11.Ferreira A, Caceres A. Estrogen-enhanced neurite growth: Evidence for a selective induction of Tau and stable microtubules. J Neurosci. 1991;11:392–400. doi: 10.1523/JNEUROSCI.11-02-00392.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lew GM. Changes in microtubular tau protein after estrogen in a cultured human neuroblastoma cell line. Gen Pharmacol. 1993;24:1383–1386. doi: 10.1016/0306-3623(93)90423-u. [DOI] [PubMed] [Google Scholar]
- 13.Frasor J, Stossi F, Danes JM, et al. Selective estrogen receptor modulators: Discrimination of agonistic versus antagonistic activities by gene expression profiling in breast cancer cells. Cancer Res. 2004;64:1522–1533. doi: 10.1158/0008-5472.can-03-3326. [DOI] [PubMed] [Google Scholar]
- 14.Andre F, Hatzis C, Anderson K, et al. Microtubule-associated protein-tau is a bifunctional predictor of endocrine sensitivity and chemotherapy resistance in estrogen receptor-positive breast cancer. Clin Cancer Res. 2007;13:2061–2067. doi: 10.1158/1078-0432.CCR-06-2078. [DOI] [PubMed] [Google Scholar]
- 15.Gown AM, Goldstein LC, Porter PL, et al. Multivariate analysis of the microtubule-associated protein Tau predicts improved progression-free and overall survival in patients with metastatic, Her-2-negative breast cancer treated with docetaxel and vinorelbine plus filgrastim. J Clin Oncol. 2006;24(suppl):13s. abstr 543. [Google Scholar]
- 16.Dumontet C, Reed JC, Krajewska M, et al. BCIRG-001 molecular analysis: Identification of prognostic factors in patients receiving adjuvant therapy for node-positive (N+) breast cancer. J Clin Oncol. 2007;25(suppl):9s. doi: 10.1158/1078-0432.CCR-10-0079. abstr 525. [DOI] [PubMed] [Google Scholar]
- 17.Rouzier R, Pusztai L, Delaloge S, et al. Nomograms to predict pathologic complete response and metastasis-free survival after preoperative chemotherapy for breast cancer. J Clin Oncol. 2005;23:8331–8339. doi: 10.1200/JCO.2005.01.2898. [DOI] [PubMed] [Google Scholar]
- 18.Paik S, Shak S, Tang G, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351:2817–2826. doi: 10.1056/NEJMoa041588. [DOI] [PubMed] [Google Scholar]
- 19.Paik S, Tang G, Shak S, et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol. 2006;24:3726–3734. doi: 10.1200/JCO.2005.04.7985. [DOI] [PubMed] [Google Scholar]
- 20.Faneyte IF, Schrama JG, Peterse JL, et al. Breast cancer response to neoadjuvant chemotherapy: Predictive markers and relation with outcome. Br J Cancer. 2003;88:406–412. doi: 10.1038/sj.bjc.6600749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ponzone R, Montemurro F, Maggiorotto F, et al. Clinical outcome of adjuvant endocrine treatment according to PR and HER-2 status in early breast cancer. Ann Oncol. 2006;17:1631–1636. doi: 10.1093/annonc/mdl296. [DOI] [PubMed] [Google Scholar]
- 22.Jirstrom K, Ryden L, Anagnostaki L, et al. Pathology parameters and adjuvant tamoxifen response in a randomized premenopausal breast cancer trial. J Clin Pathol. 2005;58:1135–1142. doi: 10.1136/jcp.2005.027185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.de Azambuja E, Cardoso F, de Castro G, et al. Ki-67 as prognostic marker in early breast cancer: A meta-analysis of published studies involving 12,155 patients. Br J Cancer. 2007;96:1504–1513. doi: 10.1038/sj.bjc.6603756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Viale G, Regan MM, Mastropasqua MG, et al. Predictive value of tumor Ki-67 expression in two randomized trials of adjuvant chemo endocrine therapy for node-negative breast cancer. J Natl Cancer Inst. 2008;100:207–212. doi: 10.1093/jnci/djm289. [DOI] [PubMed] [Google Scholar]
- 25.Ethier SP. Human breast cancer cell lines as models of growth regulation and disease progression. J Mammary Gland Biol Neoplasia. 1996;1:111–121. doi: 10.1007/BF02096306. [DOI] [PubMed] [Google Scholar]
- 26.Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: Role of ATP-dependent transporters. Nat Rev Cancer. 2002;2:48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- 27.Pusztai L, Wagner P, Ibrahim N, et al. Phase II study of tariquidar, a selective P-glycoprotein inhibitor, in patients with chemotherapy-resistant, advanced breast carcinoma. Cancer. 2005;104:682–691. doi: 10.1002/cncr.21227. [DOI] [PubMed] [Google Scholar]
- 28.Arteaga CL, Truica CI. Challenges in the development of anti-epidermal growth factor receptor therapies in breast cancer. Semin Oncol. 2004;31(suppl 3):3–8. doi: 10.1053/j.seminoncol.2004.01.006. [DOI] [PubMed] [Google Scholar]