Abstract
Objective
To investigate whether repeated administration of modulating frequency transcutaneous electric nerve stimulation (TENS) prevents development of analgesic tolerance.
Design
Knee joint inflammation (3% carrageenan and kaolin) was induced in rats. Either mixed or alternating frequency was administered daily (20min) for 2 weeks to the inflamed knee under light halothane anesthesia (1%–2%).
Setting
Laboratory.
Animals
Adult male Sprague-Dawley rats (N=36).
Intervention
Mixed- (4Hz and 100Hz) or alternating- (4Hz on 1 day; 100Hz on the next day) frequency TENS at sensory intensity and 100μs pulse duration.
Main Outcome Measures
Paw and joint withdrawal thresholds to mechanical stimuli were assessed before induction of inflammation, and before and after daily application of TENS.
Results
The reduced paw and joint withdrawal thresholds that occur 24 hours after the induction of inflammation were significantly reversed by the first administration of TENS when compared with sham treatment or to the condition before TENS treatment, which was observed through day 9. By the tenth day, repeated daily administration of either mixed- or alternating-frequency TENS did not reverse the decreased paw and joint withdrawal thresholds.
Conclusions
These data suggest that repeated administration of modulating frequency TENS leads to a development of opioid tolerance. However, this tolerance effect is delayed by approximately 5 days compared with administration of low- or high-frequency TENS independently. Clinically, we can infer that a treatment schedule of repeated daily TENS administration will result in a tolerance effect. Moreover, modulating low and high frequency TENS seems to produce a better analgesic effect and tolerance is slower to develop.
Keywords: Hyperalgesia, Pain, Rehabilitation, Transcutaneous electric nerve stimulation
TRANSCUTANEOUS ELECTRIC nerve stimulation (TENS) is a nonpharmacologic treatment commonly used by health care practitioners as an adjunct treatment for pain relief. TENS is the application of surface electrodes applied to the skin for pain relief. Clinically, TENS has been applied at varying frequencies of stimulation, high (100Hz), low (4Hz), and modulated frequency (between high and low).1,2
Our group developed and refined an animal model of TENS that overcomes many of the shortcomings described in the clinical literature. This model minimizes the placebo effect, controls the extent and type of injury, and enables TENS to be applied in a highly reproducible manner. The size of electrodes (1.3cm [0.5in]) relative to the surface area of the rat is comparable with those used in humans. We showed that application of low- (4Hz) or high- (100Hz) frequency TENS to the knee joint at a sensory intensity (defined as just below motor threshold) completely reduces the secondary heat and mechanical hyperalgesia of the hind paw that develops after injection of 3% kaolin and carrageenan in the knee joint.3,4 This model is highly reproducible in the laboratory and is well suited to investigations of the neurobiologic mechanisms that mediate the antihyperalgesic effects of TENS in rats.
TENS reduces hyperalgesia through release of endogenous opioids in the central nervous system.5–7 Specifically, low- and high-frequency TENS activate μ- and δ-opioid receptors, respectively, in the spinal cord and rostral ventral medulla.6,7 Repeated administration of either μ- or δ-opioid agonists causes a gradual diminution of the analgesic effect.8,9 This phenomenom is termed opioid tolerance.10 Acute or chronic opioid tolerance develops following single or repeated administration of exogenous opioid agonists in animals8,11,12 or in clinical studies.13–15 Tolerance to morphine reduces the response to other morphine-like drugs as well, a phenomenon termed cross-tolerance.16 Repeated administration of either electroacupuncture or focal electric stimulation of the brain in rats leads to a gradual curtailment in efficacy and a cross-tolerance to morphine.17,18 Similarly, in morphine-tolerant rats, low-, but not high-frequency TENS is ineffective in reversing inflammation induced secondary hyperalgesia,19 suggesting cross-tolerance at the μ-opioid receptor. In human subjects who have received enough opioid analgesics to develop tolerance, a cross-tolerance to TENS also occurs.20
In rats, repeated administration of both low- and high-frequency TENS leads to a development of tolerance by the fourth day with a cross-tolerance to spinally administered μ- and δ-opioid agonists, respectively.4 A large number of biochemical and pharmacologic studies provide convincing evidence regarding the existence of modulatory interactions between μ-and δ-opioid receptors.4 Several studies indicate that δ-opioid receptor agonists as well as δ-opioid receptor antagonists can provide beneficial modulation to the pharmacologic effects of μ agonists. For example, δ-opioid agonists enhance the analgesic potency and efficacy of μ opioid agonists, and δ-opioid agonists and antagonists can prevent or diminish the development of tolerance and physical dependence by μ-opioid agonists.21
Modulating frequencies of TENS between 2 and 100Hz decreased morphine requirements postoperatively22 and produced a greater increase in heat pain thresholds,23 but had no increased analgesic effect on pain in people with osteoarthritis24 when compared with 2 or 100Hz alone. Further there was an increased release of enkephalin in the cerebral spinal fluid of orthopedic patients when the frequency of stimulation alternated between 2 and 15Hz compared with either 2 or 15Hz alone.25 Thus, the majority of the studies show that modulating frequency has a greater effect than a single frequency of stimulation.
Because repeated administration of either low or high frequency TENS results in cross tolerance at μ- and μ-opioid receptors, respectively, we modulated TENS frequency between low and high frequencies that would activate μ- and δ-opioid receptors. Thus, this study tested the hypothesis that modulating TENS frequency (either mixed or alternating frequency) in rats would prevent the development of analgesic tolerance to TENS.
METHODS
All experiments were approved by Animal Care and Use Committee at the University of Iowa and are in accordance with the guidelines of National Institute of Health and the International Association for the Study of Pain on use of laboratory animals. Adult male Sprague-Dawley rats (n=36; weight range, 225–250g) a were used for this study.
Induction of Inflammation
Immediately after baseline behavioral measurements, we anesthetized rats with 5% halothane, maintained with 1% to 2% halothane, and then induced knee joint inflammation by intra-articular injection of a mixture of 3% carrageenan and kaolin (0.1mL in sterile saline; pH, 7.4) into the left knee joint.26 The inflammation is considered acute for the first 24 hours, when there is neutrophil infiltration. By 1 week, the inflammation converts to chronic, as identified histologically by macrophage infiltration. This model is used to mimic arthritic conditions and shows good predictability for drug effects.27 After induction of knee inflammation, the rats were returned to their cages and allowed to recover for 24 hours. Within 24 hours, the animals exhibited signs of inflammation such as edematous and warm knee joints and also behavioral signs such as guarding and decreased weight bearing on the inflamed limb.28
Behavioral Testing
The paw and knee joint withdrawal thresholds were tested for all groups of rats. Both measurements were performed immediately before and after TENS on each day. The investigator was blinded for all stimulations.
We tested rats for paw withdrawal threshold with von Frey filaments applied to the paw. Initially, the animals were maintained in their cages in the behavior testing room for acclimation for 30 minutes. Then, the animals were placed in transparent Lucite cubicles over a wire mesh and acclimated for another 30 minutes before testing. A series of filaments with increasing bending forces (range, 9.4–495.8mN) were applied on the plantar surface of the hind paw until the rat withdrew from the stimulus.28 Each filament was applied twice. The lowest force at which the rat withdrew its paw from 1 of 2 applications was recorded as the paw withdrawal threshold for mechanical hyperalgesia and interpreted as cutaneous hyperalgesia. This testing method has shown significant test-retest reliability.6
Rats were also tested for knee joint withdrawal threshold with a pair of forceps applied to the knee joint as previously described for the gastrocnemius muscle.29 The forceps were equipped with 2 strain gauges to measure force. To measure knee joint withdrawal threshold, animals were placed in the restrainer (glove), and the experimenter compressed the knee joint with the tip of the forceps while the hind limb was extended. Compression was continued until the animal withdrew the leg. The maximum force applied at withdrawal was recorded as the knee joint withdrawal threshold. Three trials, 5 minutes apart, at each time period were performed and averaged to obtain 1 reading a time period. A decrease in withdrawal threshold of the knee joint was interpreted as hyperalgesia.
Application of TENS
We anesthetized the rats with halothane, initially with 5% and maintained with 1% to 2% for 20 minutes of TENS.30 We previously showed that (1) application of halothane without TENS has no effect on the paw withdrawal latency to heat induced by joint inflammation, and (2) application of TENS to a noninflamed knee joint has no effect on the paw withdrawal latency.3 EMPI Select TENS unitsb with an asymmetrical biphasic square wave and circular electrodes were used. Under halothane anesthesia, the knee joint was shaved and 1.3cm (0.5in) pre-gelled surface electrodes were applied to the medial and lateral aspects of the inflamed knee joint in the groups receiving TENS or sham TENS. We observed the animals continuously during TENS to ensure adequate anesthesia and to ensure that the electrodes remained in contact with the skin.
We administered mixed (low- and high-frequency TENS applied in the same session, cycled in each 3 seconds [adjustable parameters in the TENS unit: frequency, 2–150Hz in 5-Hz intervals; cycle time, 1–20s]) or alternating (low- and high-frequency TENS 4Hz and 100Hz, respectively, applied separately in alternating days) frequency TENS keeping other parameters constant for example, sensory intensity, and 20 minutes for stimulation. This strategy allowed a comparison of frequency differences without confounding differences in amplitude. Sensory intensity was determined by increasing the intensity until a muscle contraction was visibly observed and then reducing the intensity to just below this level. This intensity was between 1.5 and 4mA for all animals. The parameters were selected to model those used clinically and which produced full inhibition of secondary heat hyperalgesia.3 The sham TENS group was anesthetized with 1% to 2% halothane and electrodes were placed on their shaved knee joint, but they did not receive TENS treatment. Importantly, 3 rats always were anesthetized with the same vaporizer; at least 1 rat receiving the sham TENS treatment and 1 rat receiving the active TENS treatment were anesthetized at the same time. This procedure ensured that there always were animals in the sham TENS treatment group that received the same dose of anesthesia.
Experimental Design
We measured baseline paw and joint withdrawal thresholds bilaterally before the induction of the knee joint inflammation. Twenty-four hours after the knee joint inflammation, paw and joint withdrawal thresholds were reassessed and then the animals were lightly anesthetized with 1% to 2% halothane for placement of the electrodes. TENS was then applied for 20 minutes, and withdrawal thresholds were assessed immediately after TENS and recovery from the anesthesia (10–15min after removal of TENS). The same procedure (assessment of withdrawal thresholds, TENS application, and reassessment of withdrawal thresholds) was applied on each day for the next 13 days (total, 14d). The rats were randomly divided in 3 groups: (1) sham TENS (n=6); (2) mixed-frequency TENS (n=6), and (3) alternating-frequency TENS (n=6).
It is possible that TENS was ineffective 10 days after knee joint inflammation. Thus, in a separate group of animals mixed frequency (n=6), alternating frequency (n=6), and sham treatment (n=6) were administered only on the tenth day following the induction of inflammation with no TENS treatment in the previous 9 days.
Statistical Analysis
Mechanical withdrawal threshold of the paw was tested for differences between groups of treatment (mixed, alternating, sham) with the nonparametric Kruskal-Wallis test, and joint withdrawal threshold with analysis of variance for dependent samples. Tukey test, paired t test, signed-rank test, or Wilcoxon-matched pairs test were used as appropriate. P value less than .05 was considered significant.
RESULTS
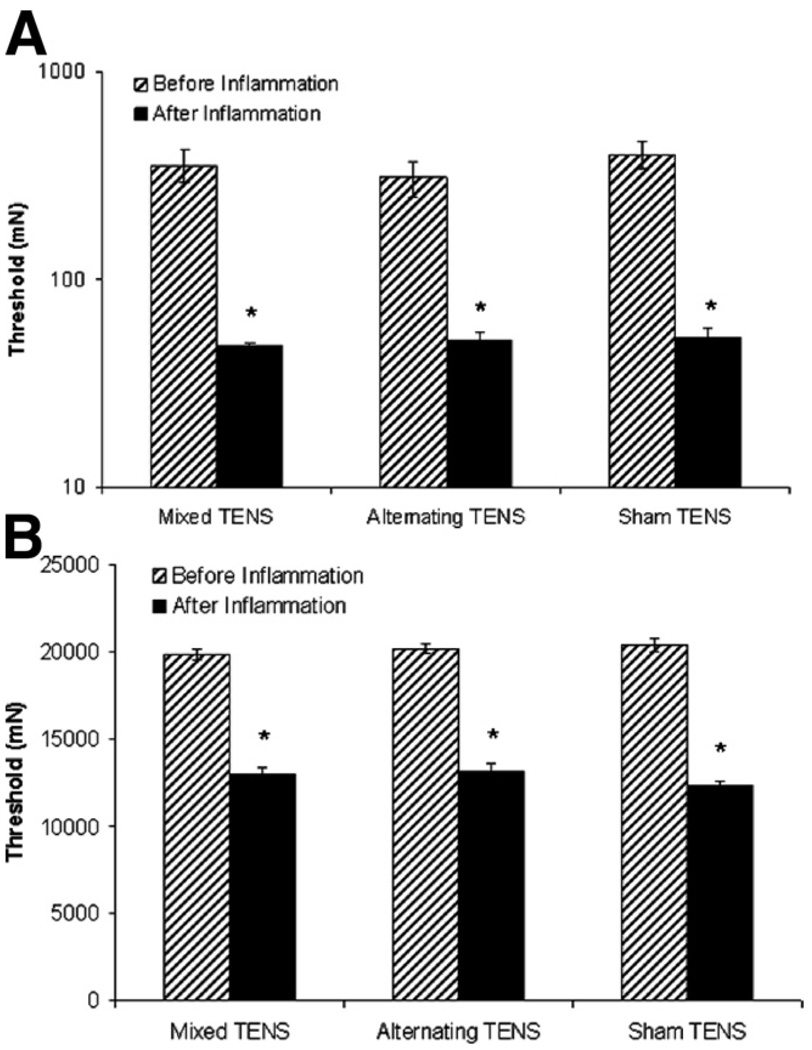
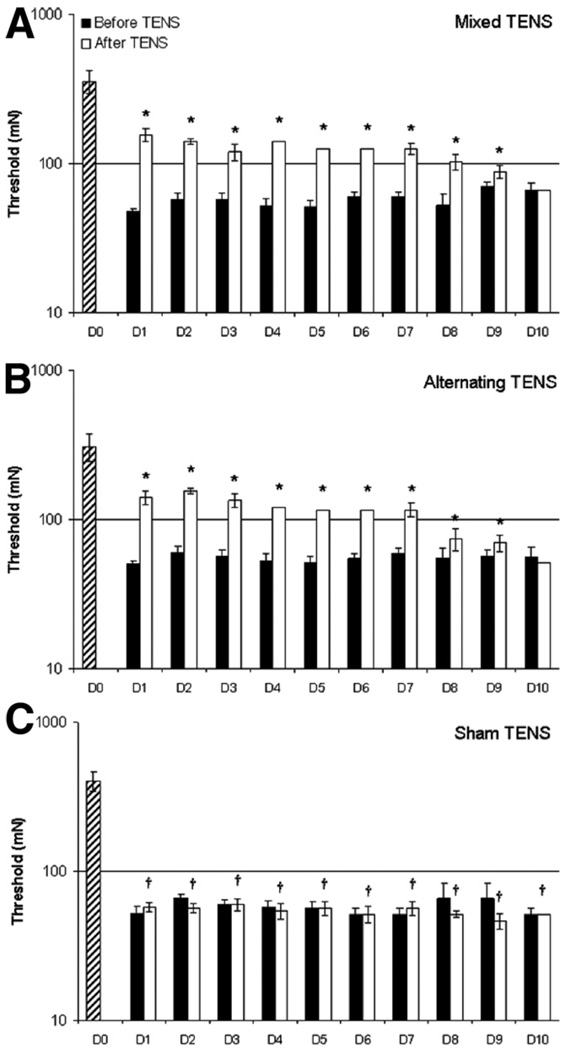
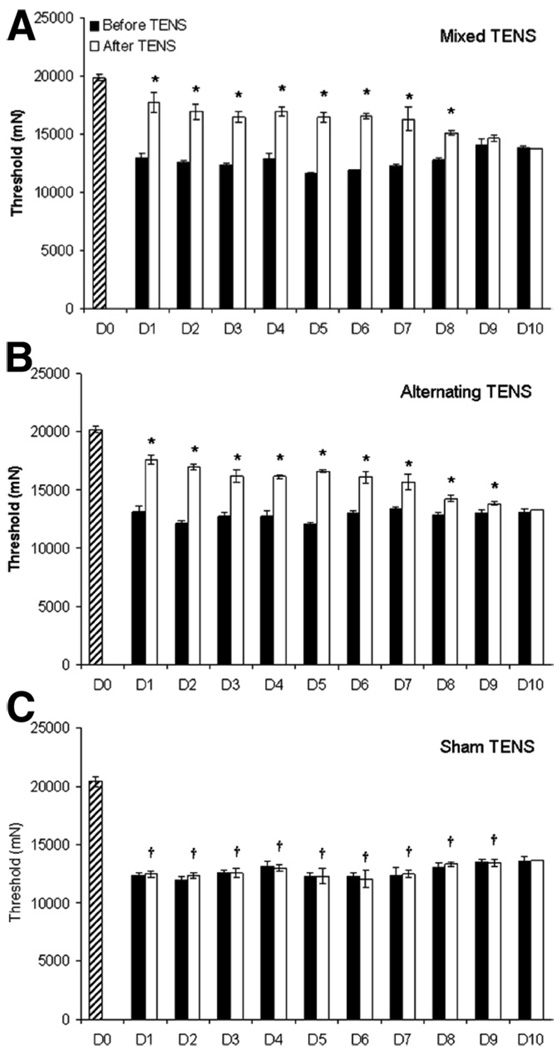
Twenty-four hours after carrageenan and kaolin injection-induced knee joint inflammation, the paw (P =.001) and joint (P = .001) withdrawal thresholds were reduced on the ipsilateral paw (figs 1A, B, respectively) and the animals showed overt behavioral signs, for example, limping and guarding of the limb. Following the administration of either mixed or alternating-frequency TENS, there was a significant reversal of the reduced paw and knee joint withdrawal thresholds. When TENS was repeatedly administered for 14 days, its effectiveness in reversing the decreased withdrawal thresholds of the paw and knee (fig 2A, 2B, fig 3A, 3B) was lost by the tenth day in both the mixed and alternating frequency TENS groups.
Fig. 1.
Bar graphs representing mechanical thresholds of the ipsilateral (A) paw and (B) knee joint from animals before and after knee joint inflammation in the groups mixed, alternating, and sham TENS. Plot represents mean values, error bars represent the standard error of the mean (SEM). *Significantly different from before knee joint inflammation (P<.05).
Fig. 2.
Bar graphs representing mechanical thresholds of the ipsilateral paw from animals that received (A) mixed-frequency TENS, (B) alternating-frequency TENS, or (C) sham TENS for the time before (black bars) and after (white bars) application of TENS. Striped bar represents time before knee joint inflammation. Plot represents mean values, error bars represent the SEM. *Significantly different from before application of TENS; †significantly different from before inflammation (P<.05).
Fig. 3.
Bar graphs representing mechanical thresholds of the ipsilateral knee joint from animals that received (A) mixed-frequency TENS, (B) alternating-frequency TENS, or (C) sham TENS for the time before (black bars) and after (white bars) application of TENS. Striped bar represents time before knee joint inflammation. Plot represents mean values, error bars represent the SEM. *Significantly different from before application of TENS; †significantly different from before inflammation (P<.05).
Treatment with both mixed- and alternating-frequency TENS significantly increased paw withdrawal thresholds compared with the results obtained before TENS treatment (P≤.02, for each day of treatment) or with sham TENS treatment (P≤.01, for each day of treatment) for 1 to 9 days. Similarly, joint withdrawal threshold was also significantly increased when compared with the results obtained before TENS treatment (P≤.006, for each day of treatment) or with sham TENS treatment (P≤.003, for each day of treatment) for 1 to 9 days. Thus, following 10 days of repeated application of both mixed-and alternating-frequency TENS during 20 minutes daily to the inflamed knee joint in separate groups of rats, a tolerance-like effect was developed after 9 days of repeated stimulation. This TENS tolerance effect was exemplified as an ineffectiveness of TENS in reversing both the decreased withdrawal thresholds of the paw and the joint. No effect was observed in control animals (sham TENS group), which were similarly anesthetized with halothane for 20 minutes daily for 10 days with the electrodes placed on the inflamed knee; for example, the paw and joint withdrawal thresholds remained decreased.
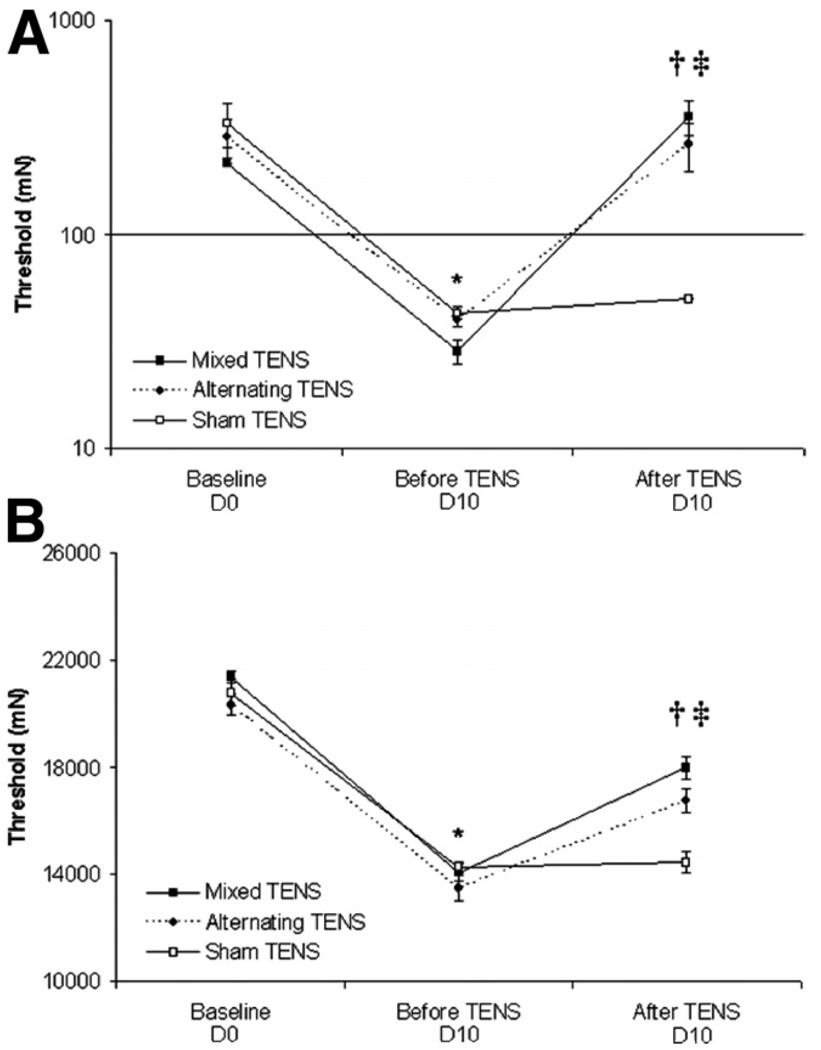
If TENS was given only on day 10 (after induction of joint inflammation), both mixed- and alternating-frequency TENS significantly increased the paw and knee joint withdrawal thresholds when compared with sham TENS (figs 4A, 4B). This finding refutes the possibility of a time-effect for the action of TENS, implying that recurrent TENS administration leads to a reduction in the antinociceptive effect.
Fig. 4.
Line graphs representing mechanical thresholds of the (A) ipsilateral paw and (B) knee joint from animals that received mixed- and alternating-frequency TENS, and sham TENS. Baseline corresponds to time before the induction of knee inflammation. Error bars represent the SEM. *Significantly different from baseline; †mixed- and alternating-frequency TENS significantly different from before TENS treatment; ‡mixed-and alternating-frequency TENS significantly different from sham TENS (P<.05).
DISCUSSION
The data from this study show that mixed- or alternating-frequency TENS inhibits the primary (knee) and secondary (paw) mechanical hyperalgesia associated with inflammation of the knee joint. Further, repeated applications of either mixed- or alternating-frequency TENS in rats produces analgesic tolerance to TENS by the tenth day of treatment. This tolerance is delayed when compared to previous studies showing that tolerance to either low- or high-frequency TENS develops by the fourth day.4 We propose that the development of tolerance is a result of tolerance at μ- and δ-opioid receptors in the spinal cord as observed in previous studies with TENS4 and in pharmacologic studies that show repeated administration of μ- and δ-opioid agonists spinally or supraspinally leading to a development of tolerance.8,9,30
Both spinally and supraspinally located μ- and δ-opioid receptors mediate the analgesic effects of low- and high-frequency TENS, respectively.6,7 Evidence points to a lack of cross-tolerance between spinally administered μ- (morphine) and δ-opioid (D-Ala2-D-Leu5) enkephalin agonists suggesting that the tolerance produced at μ- and δ-opioid receptors occur independent of each other.30,31 Similarly, in the study by Sluka et al,19 there is no cross-tolerance between low-frequency TENS, μ-opioid mediated analgesia, and the δ-opioid agonist, SNC-80. Conversely, there is no cross-tolerance between high-frequency TENS, δ-opioid mediated analgesia, and the μ-opioid agonist, morphine. In animals tolerant to morphine, low-frequency electroacupuncture does not reduce heat hyperalgesia in models of inflammation when secondary heat hyperalgesia was tested by measuring the paw withdrawal latency to radiant heat.17 However, high-frequency TENS is still effective in morphine tolerant rats.19 High-frequency electroacupuncture, on the other hand, is effective in animals tolerant to δ-opioid agonists, but ineffective in rats made tolerant to dynorphin, a k-opioid receptor agonist.32,33 These data show that low- and high-frequency TENS produce their effects through activation of different opioid receptors, and repeated administration produces tolerance at different opioid receptors. Differences between TENS and electroacupuncture for high-frequency TENS could relate to different parameters of stimulation (electroacupuncture used longer pulse durations than TENS), different fibers activated (deep vs cutaneous), or different intensities of stimulation (motor vs sensory).
Endomorphin-1, an endogenous ligand for the μ-opioid receptor mediates the analgesic effect of low-frequency (2Hz), but not high-frequency (100Hz) electroacupuncture,34 whereas, cross-tolerance studies show that the analgesic effect of low-frequency electroacupuncture is mediated by δ-opioid receptors in the spinal cord. On the other hand, high-frequency electroacupuncture is mediated by k-opioid receptor and the dense-disperse mode (2–15Hz) for example, low frequency alternating with high frequency at 10 to 12 cycles/min, is mediated by a combined action at all 3 opioid receptors in the spinal cord.33 Wang et al35 demonstrated a similar analgesic activity for electroacupuncture and TENS in rats. Using 3 different frequencies (2, 15, 100Hz), an intense cross-tolerance effect occurs between the corresponding frequencies of electroacupuncture and TENS. Also, low doses of subcutaneously injected naloxone (2mg/kg) have been shown to cause a complete, partial, and no blockade of the analgesic effects of 2, 15, and 100Hz frequencies, respectively, for both electroacupuncture and TENS. This leads to a conclusion that there is a common neurochemical basis for the analgesia obtained for these 2 analgesic modalities.
Our current data extend the results of a previous study of our laboratory that examined the effects of TENS at high and low frequencies on the hyperalgesia induced by knee inflammation.4 In the current study, the reversal of the primary and secondary mechanical hyperalgesia at 9 to 10 days was surprising. Different from results obtained in the present study, our previous study showed that both high- and low-frequency TENS lost its effectiveness in reversing the secondary hyperalgesia by the fourth day of repeated daily TENS. Importantly, in the current study, the pulse duration and intensity and time for stimulation were similar to those in a study by Chandran and Sluka.4
Evidence points to the existence of physical and functional interactions between the opioid receptors, particularly between the μ- and δ-opioid receptors. Several biochemical and pharmacologic studies using μ- and δ-opioid receptor ligands gave an early indication of such interactions.36–38 The μ- and δ-opioid receptors exist on overlapping populations of neurons in pain modulating regions of the central nervous system, and the presence of both μ- and δ- opioid receptors within the same neuron has been shown.39 The physiologic and pharmacologic significance of δ–μ-opioid receptor interactions have been substantiated by recent studies using opioid receptor gene knockout animals.40
Three studies describe δ-receptor–mediated analgesia in μ-opioid receptor–deficient mice.41–43 Spinal analgesia (measured by the tail-withdrawal response) elicited by 2 highly δ-receptor–selective agonists,41–43 D-Pen2,D-Pen5-enkephalin and deltorphin II,41 was reported to be unchanged,43 slightly lower,41 or dramatically lower43 in mutant mice compared with wild type animals. The 2 latter studies suggest that the presence of μ-receptors is required to obtain full δ-receptor-mediated analgesia in the tail-withdrawal response. Importantly, reversal by a selective δ-receptor antagonist, naltrindole, indicated that the higher δ-receptor mediated analgesia in wild-type mice was not a result of a nonselective activation of μ-receptors.41 The supraspinal antinociceptive response (jumping from the hotplate) measured in a parallel experiment, was found to be diminished43 or preserved41,42 in μ-opioid-receptor– deficient mice. Although alterations of δ-receptor–mediated analgesia are not concordant in the 3 studies, the data support the existence of synergistic interactions between μ- and δ-receptors, which are revealed under certain experimental conditions. Whether receptor interactions take place between receptors located on distinct neurons, which interact functionally within neural circuits, or from receptor cross-talk in co-expressing cells, is not clear.
Co-administration of μ- and δ-opioid receptor showed that both the potency and efficacy of μ-opioid agonists can be increased by δ-opioid agonists. Vaught and Takemori44 observed that Leu 5-enkephalin given at subantinociceptive doses potentiates the analgesic actions of the μ-opioid ligand morphine. Several other studies extended these observations to synergistic antinociceptive effects between other μ-opioid agonists and δ-opioid agonists.45,46 The activation of δ-opioid receptors has synergistic effects on μ-opioid functional activities in cells transfected with μ- and δ-opioid receptors.38,47,48 Treatment of rats or mice for several days with μ-opioid agonists leads to translocation of δ-opioid receptors to neuronal plasma membranes and enhances δ-receptor mediated antinociception.49 In this present study, the modulation of high and low frequencies of TENS presented a greater antinociceptive effect because the development of tolerance to TENS was significantly delayed. Thus, we hypothesize that modulating frequency of TENS, at both mixed and alternating frequencies, results in synergistic antinociceptive interaction between μ- and δ-opioid receptors because effects of low- or high-frequency TENS are mediated by activation of μ- and δ-opioid receptors, respectively.6,7
Of particular interest among peptide ligands possessing dual agonist actions at μ- and δ-opioid receptors are biphalin and biphalin analogs. Biphalin (Tyr-D-Ala-Gly-Phe-NH)2, binds to both μ- and δ-opioid receptors with high affinity. This peptide also produces antinociceptive effects comparable with morphine after systemic injection and has been shown to produce less dependence than morphine on chronic use.50,51 Similarly to biphalin, it seems that modulating frequency exerts a dual agonist action at μ- and δ-opioid receptors which has been shown to cause a delay in the development of tolerance to TENS treatment.
In studies with rats, additive analgesic effects were observed when the μ-opioid agonist fentanyl was administered in combination with the δ-opioid agonist BW373U86, a nonpeptide agonist ligand. In other studies it has been found that prolonged co-administration of morphine and BW373U86 attenuated the development and expression of morphine-induced dependence and tolerance in rats.52 Moreover, co-administration of BW373U86 with alfentanil led to significant attenuation of alfentanil-induced respiratory depression.53 This suggests that compounds with mixed μ- and δ-receptor agonist activity may be useful in producing analgesic effects with fewer adverse effects. Conversely, no side effect has been shown when TENS is used in any frequency. On the basis of this knowledge and our results, it can be inferred that modulating-frequency TENS has potential clinical advantage given that it does not cause side effects and the delay of the tolerance effect means a better antinociceptive effect.
The application of current of modulated frequencies not only could delay tolerance but also likely will increase the analgesic effectiveness. Previous literature on electroacupuncture shows that alternating frequencies between 2 and 100Hz in the same session (2 and 100Hz were alternated in each 3 seconds) was 40% more effective than applying frequencies of 2 and 100Hz simultaneously (simultaneous stimulation with 2Hz applied to the left and 100Hz to the right hind leg). Further, alternating between 2 and 100Hz electroacupuncture stimulation released both dynorphin and endomorphin opioid peptides, whereas only dynorphin was released by combined application of 2 and 100Hz.54 In postoperative pain, modulating frequencies of TENS between 2 and 100Hz also decreased morphine requirements postoperatively22 when compared with those receiving either 2 or 100Hz alone, or placebo. Thus, modulating frequency may increase release of multiple opioid peptides to reduce pain and increase analgesia.
Although this present study involved no neurophysiologic or neurochemical techniques, it can be suggested that the development of analgesic tolerance to TENS occurs (1) after the tissue injury when the knee joint is inflamed with 3% of carrageenan and kaolin, (2) in response to mechanical stimuli on the site and outside the injury, and (3) probably related to the existence of intermodulatory effects between μ- and δ-opioid receptors.
Study Limitations
Our findings represent data from a small number of animals and therefore any generalization to acute or chronic human pain response is speculative, although the temptation to infer the findings to human clinical practice is strong.
CONCLUSIONS
The current study is unique in that the effect of modulating TENS on primary and secondary mechanical hyperalgesia was studied after TENS application, showing that TENS is effective in both mixed and alternating frequencies of stimulation. If this finding translates clinically, TENS can be applied in patients who have inflammatory conditions such as arthritis. Moreover, there is evidence that there could be clear therapeutic advantages in combining the actions of high- and low-frequency TENS, providing more benefit from the analgesic effects of TENS to produce a maximal antinociceptive effect. Based on the findings in this study, it can be also inferred that a treatment schedule of repeated TENS administration should be avoided to prevent the development of tolerance. Administration of TENS for fewer treatment sessions and its combined usage with other pharmaceutical agents could be used to improve analgesic efficacy and prevent tolerance to TENS.
Acknowledgments
Supported by a competitive grant from EMPI Inc. and the National Institutes of Health (grant no. NIH AR052316).
Footnotes
No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the authors or upon any organization with which the authors are associated.
- Harlan, 8520 Allison Pointe Blvd, Ste 400, Indianapolis, IN 46250.
- EMPI Inc, 599 Cardigan Rd, St. Paul, MN 55126-4099.
References
- 1.Rakel B, Frantz R. Effectiveness of transcutaneous electrical nerve stimulation on postoperative pain with movement. J Pain. 2003;4:455–464. doi: 10.1067/s1526-5900(03)00780-6. [DOI] [PubMed] [Google Scholar]
- 2.Olsen MF, Elden H, Janson ED, Lilja H, Stener-Victorin E. A comparison of high- versus low-intensity, high-frequency transcutaneous electric nerve stimulation for painful postpartum uterine contractions. Acta Obstet Gynecol Scand. 2007;86:310–314. doi: 10.1080/00016340601040928. [DOI] [PubMed] [Google Scholar]
- 3.Sluka KA, Bailey K, Bogush J, Olson R, Ricketts A. Treatment with either high or low frequency TENS reduces the secondary hyperalgesia observed after injection of kaolin and carrageenan into the knee joint. Pain. 1998;77:97–102. doi: 10.1016/S0304-3959(98)00090-6. [DOI] [PubMed] [Google Scholar]
- 4.Chandran P, Sluka KA. Development of opioid tolerance with repeated transcutaneous electrical nerve stimulation administration. Pain. 2003;102:195–201. doi: 10.1016/s0304-3959(02)00381-0. [DOI] [PubMed] [Google Scholar]
- 5.Han JS, Xie GX, Ding ZX, Fan SG. High and low frequency electroacupuncture analgesia are mediated by different opioid peptides [abstract] Pain. 1984;18 Suppl 1:S369. [Google Scholar]
- 6.Sluka KA, Deacon M, Stibal A, Strissel S, Terpstra A. Spinal blockade of opioid receptors prevents the analgesia produced by TENS in arthritic rats. J Pharmacol Exp Ther. 1999;289:840–846. [PubMed] [Google Scholar]
- 7.Kalra A, Urban MO, Sluka KA. Blockade of opioid receptors in rostral ventral medulla prevents antihyperalgesia produced by transcutaneous electrical nerve stimulation (TENS) J Pharmacol Exp Ther. 2001;298:257–263. [PubMed] [Google Scholar]
- 8.Kissin I, Brown PT, Robinson CA, Bradley EL., Jr Acute tolerance in morphine analgesia: continuous infusion and single injection in rats. Anesthesiology. 1991;74:166–171. doi: 10.1097/00000542-199101000-00025. [DOI] [PubMed] [Google Scholar]
- 9.Zhao GM, Bhargava HN. Effects of multiple intracerebroventricular injections of [D-Pen2,D-Pen5] enkephalin and [D-Ala2,Glu4] deltorphin II on tolerance to their analgesic action and on brain delta-opioid receptors. Brain Res. 1997;745:243–247. doi: 10.1016/s0006-8993(96)01156-0. [DOI] [PubMed] [Google Scholar]
- 10.Moulin DE, Ling GS, Pasternak GW. Unidirectional analgesic cross-tolerance between morphine and levorphanol in the rat. Pain. 1988;33:233–239. doi: 10.1016/0304-3959(88)90095-4. [DOI] [PubMed] [Google Scholar]
- 11.Mao J, Price DD, Mayer DJ. Thermal hyperalgesia in association with the development of morphine tolerance in rats: roles of excitatory amino acid receptors and protein kinase C. J Neurosci. 1994;14:2301–2312. doi: 10.1523/JNEUROSCI.14-04-02301.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fairbanks CA, Wilcox GL. Spinal plasticity of acute opioid tolerance. J Biomed Sci. 2000;7:200–212. doi: 10.1007/BF02255467. [DOI] [PubMed] [Google Scholar]
- 13.McQuay HJ, Bullingham RE, Moore RA. Acute opiate tolerance in man. Life Sci. 1981;28:2513–2517. doi: 10.1016/0024-3205(81)90593-2. [DOI] [PubMed] [Google Scholar]
- 14.Vinik HR, Kissin I. Rapid development of tolerance to analgesia during remifentanil infusion in humans. Anesth Analg. 1998;86:1307–1311. doi: 10.1097/00000539-199806000-00033. [DOI] [PubMed] [Google Scholar]
- 15.Chia YY, Liu K, Wang JJ, Kuo MC, Ho ST. Intraoperative high dose fentanyl induces postoperative fentanyl tolerance. Can J Anaesth. 1999;46:872–877. doi: 10.1007/BF03012978. [DOI] [PubMed] [Google Scholar]
- 16.Paronis CA, Holtzman SG. Development of tolerance to the analgesic activity of mu agonists after continuous infusion of morphine, meperidine or fentanyl in rats. J Pharmacol Exp Ther. 1992;262:1–9. [PubMed] [Google Scholar]
- 17.Han JS, Li SJ, Tang J. Tolerance to electroacupuncture and its cross tolerance to morphine. Neuropharmacology. 1981;20:593–596. doi: 10.1016/0028-3908(81)90213-6. [DOI] [PubMed] [Google Scholar]
- 18.Mayer DJ, Hayes RL. Stimulation-produced analgesia: development of tolerance and cross-tolerance to morphine. Science. 1975;188:941–943. doi: 10.1126/science.1094537. [DOI] [PubMed] [Google Scholar]
- 19.Sluka KA, Judge MA, McColley MM, Reveiz PM, Taylor BM. Low frequency TENS is less effective than high frequency TENS at reducing inflammation-induced hyperalgesia in morphinetolerant rats. Eur J Pain. 2000;4:185–193. doi: 10.1053/eujp.2000.0172. [DOI] [PubMed] [Google Scholar]
- 20.Duckworth WC, Solomon SS, Jallepalli P, Heckemeyer C, Finnern J, Powers A. Glucose intolerance due to insulin resistance in patients with spinal cord injuries. Diabetes. 1980;29:906–910. doi: 10.2337/diab.29.11.906. [DOI] [PubMed] [Google Scholar]
- 21.Ananthan S. Opioid ligands with mixed μ/δ opioid receptor interaction: an emerging approach to novel analgesics. AAPS J. 2006;8:E118–E124. doi: 10.1208/aapsj080114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hamza MA, White PF, Ahmed HE, Ghoname EA. Effect of the frequency of transcutaneous electrical nerve stimulation on the postoperative opioid analgesic requirement and recovery profile. Anesthesiology. 1999;91:1232–1238. doi: 10.1097/00000542-199911000-00012. [DOI] [PubMed] [Google Scholar]
- 23.Tong KC, Lo SK, Cheing GL. Alternating frequencies of transcutaneous electric nerve stimulation: does it produce greater analgesic effects on mechanical and thermal pain thresholds? Arch Phys Med Rehabil. 2007;88:1344–1349. doi: 10.1016/j.apmr.2007.07.017. [DOI] [PubMed] [Google Scholar]
- 24.Law PW, Cheing LY. Optimal stimulation frequency of transcutaneous electrical nerve stimulation on people with knee osteoarthritis. J Rehabil Med. 2004;36:220–225. doi: 10.1080/16501970410029834. [DOI] [PubMed] [Google Scholar]
- 25.Chen XH, Han JS, Yuan Y, Yan SC, Terenius L. 2/15 Hz electrical stimulation induced by an increase of both Met-enkephalin-Arg-Phe and dynorphin from spinal cord in humans: a preliminary report. J Beijing Med Univ. 1992:24–80. [Google Scholar]
- 26.Sluka KA, Westlund KN. Behavioral and immunohistochemical changes in an experimental arthritis model in rats. Pain. 1993;55:367–377. doi: 10.1016/0304-3959(93)90013-F. [DOI] [PubMed] [Google Scholar]
- 27.Radhakrishnan R, Moore SA, Sluka KA. Unilateral carrageenan injection into muscle or joint induces chronic bilateral hyperalgesia in rats. Pain. 2003;104:567–577. doi: 10.1016/s0304-3959(03)00114-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gopalkrishnan P, Sluka KA. Effect of varying frequency, intensity, and pulse duration of transcutaneous electrical nerve stimulation on primary hyperalgesia in inflamed rats. Arch Phys Med Rehabil. 2000;81:984–990. doi: 10.1053/apmr.2000.5576. [DOI] [PubMed] [Google Scholar]
- 29.Skyba DA, Radhakrishnan R, Sluka KA. Characterization of a method for measuring primary hyperalgesia of deep somatic tissue. J Pain. 2005;6:41–47. doi: 10.1016/j.jpain.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 30.Kalso EA, Sullivan AF, McQuay HJ, Dickenson AH, Roques BP. Cross-tolerance between mu opioid and alpha-2 adrenergic receptors, but not between mu and delta opioid receptors in the spinal cord of the rat. J Pharmacol Exp Ther. 1993;265:551–558. [PubMed] [Google Scholar]
- 31.Stevens CW, Yaksh TL. Studies of morphine and D-ala2-D-leu5-enkephalin (DADLE) cross-tolerance after continuous intrathecal infusion in the rat. Anesthesiology. 1992;76:596–603. doi: 10.1097/00000542-199204000-00017. [DOI] [PubMed] [Google Scholar]
- 32.Chen XH, Han JS. Analgesia induced by electroacupuncture of different frequencies is mediated by different types of opioid receptors: another cross-tolerance study. Behav Brain Res. 1992;47:143–149. doi: 10.1016/s0166-4328(05)80120-2. [DOI] [PubMed] [Google Scholar]
- 33.Chen XH, Han JS. All three types of opioid receptors in the spinal cord are important for 2/15 Hz electroacupuncture analgesia. Eur J Pharmacol. 1992;211:203–210. doi: 10.1016/0014-2999(92)90530-h. [DOI] [PubMed] [Google Scholar]
- 34.Han Z, Jiang YH, Wan Y, Wang Y, Chang JK, Han JS. Endomorphin-1 mediates 2 Hz but not 100 Hz electroacupuncture analgesia in the rat. Neurosci Lett. 1999;274:75–78. doi: 10.1016/s0304-3940(99)00670-9. [DOI] [PubMed] [Google Scholar]
- 35.Wang JQ, Mao L, Han JS. Comparison of the antinociceptive effects induced by electroacupuncture and transcutaneous electrical nerve stimulation in the rat. Int J Neurosci. 1992;65:117–129. doi: 10.3109/00207459209003283. [DOI] [PubMed] [Google Scholar]
- 36.Rapaka RS, Porreca F. Development of delta opioid peptides as nonaddicting analgesics. Pharm Res. 1991;8:1–8. doi: 10.1023/a:1015809702296. [DOI] [PubMed] [Google Scholar]
- 37.Traynor JR, Elliott J. Delta-opioid receptor subtypes and crosstalk with mu-receptors. Trends Pharmacol Sci. 1993;14:84–86. doi: 10.1016/0165-6147(93)90068-u. [DOI] [PubMed] [Google Scholar]
- 38.Rothman RB, Bykov V, Xue BG, et al. Interaction of opioid peptides and other drugs with multiple kappa receptors in rat and human brain. Evidence for species differences. Peptides. 1992;13:977–987. doi: 10.1016/0196-9781(92)90059-c. [DOI] [PubMed] [Google Scholar]
- 39.Egan TM, North RA. Both mu and delta opiate receptors exist on the same neuron. Science. 1981;214:923–924. doi: 10.1126/science.6272393. [DOI] [PubMed] [Google Scholar]
- 40.Kieffer BL. Opioids: first lessons from knockout mice. Trends Pharmacol Sci. 1999;20:19–26. doi: 10.1016/s0165-6147(98)01279-6. [DOI] [PubMed] [Google Scholar]
- 41.Matthes HW, Smadja C, Valverde O, et al. Activity of the delta-opioid receptor is partially reduced, whereas activity of the kappa-receptor is maintained in mice lacking the mu-receptor. J Neurosci. 1998;18:7285–7295. doi: 10.1523/JNEUROSCI.18-18-07285.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Loh HH, Liu HC, Cavalli A, Yang W, Chen YF, Wei LN. Mu opioid receptor knockout in mice: effects on ligand-induced analgesia and morphine lethality. Mol Brain Res. 1998;54:321–326. doi: 10.1016/s0169-328x(97)00353-7. [DOI] [PubMed] [Google Scholar]
- 43.Sora I, Funada M, Uhl GR. The mu-opioid receptor is necessary for [D-Pen2,D-Pen5] enkephalin-induced analgesia. Eur J Pharmacol. 1997;324:R1–R2. doi: 10.1016/s0014-2999(97)10016-4. [DOI] [PubMed] [Google Scholar]
- 44.Vaught JL, Takemori AE. Differential effects of leucine and methionine enkephalin on morphine-induced analgesia, acute tolerance and dependence. J Pharmacol Exp Ther. 1979;208:86–90. [PubMed] [Google Scholar]
- 45.Horan P, Tallarida RJ, Haaseth RC, Matsunaga TO, Hruby VJ, Porreca F. Antinociceptive interactions of opioid delta receptor agonists with morphine in mice: supra- and sub-additivity. Life Sci. 1992;50:1535–1541. doi: 10.1016/0024-3205(92)90144-e. [DOI] [PubMed] [Google Scholar]
- 46.Porreca F, Takemori AE, Sultana M, Portoghese PS, Bowen WD, Mosberg HI. Modulation of mu-mediated antinociception in the mouse involves opioid delta-2 receptors. J Pharmacol Exp Ther. 1992;263:147–152. [PubMed] [Google Scholar]
- 47.Gomes I, Jordan BA, Gupta A, Trapaidze N, Nagy V, Devi LA. Heterodimerization of mu and delta opioid receptors: A role in opiate synergy. J Neurosci. 2000;20:RC110. doi: 10.1523/JNEUROSCI.20-22-j0007.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Martin NA, Prather PL. Interaction of co-expressed mu- and delta-opioid receptors in transfected rat pituitary GH(3) cells. Mol Pharmacol. 2001;59:774–783. doi: 10.1124/mol.59.4.774. [DOI] [PubMed] [Google Scholar]
- 49.Cahill CM, Morinville A, Lee MC, Vincent JP, Collier B, Beaudet A. Prolonged morphine treatment targets delta opioid receptors to neuronal plasma membranes and enhances delta-mediated antinociception. J Neurosci. 2001;21:7598–7607. doi: 10.1523/JNEUROSCI.21-19-07598.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Silbert BS, Lipkowski AW, Cepeda MS, Szyfelbein SK, Osgood PF, Carr DB. Analgesic activity of a novel bivalent opioid peptide compared to morphine via different routes of administration. Agents Actions. 1991;33:382–387. doi: 10.1007/BF01986590. [DOI] [PubMed] [Google Scholar]
- 51.Yamazaki M, Suzuki T, Narita M, Lipkowski AW. The opioid peptide analogue biphalin induces less physical dependence than morphine. Life Sci. 2001;69:1023–1028. doi: 10.1016/s0024-3205(01)01194-8. [DOI] [PubMed] [Google Scholar]
- 52.Lee PH, McNutt RW, Chang KJ. A. nonpeptidic delta opioid receptor agonist, BW373U86, attenuates the development and expression of morphine abstinence precipitated by naloxone in rat. J Pharmacol Exp Ther. 1993;267:883–887. [PubMed] [Google Scholar]
- 53.Su YF, McNutt RW, Chang KJ. Delta-opioid ligands reverse alfentanil-induced respiratory depressions but not antinociception. J Pharmacol Exp Ther. 1998;287:883–887. [PubMed] [Google Scholar]
- 54.Wang Y, Zhang Y, Wang W, Cao Y, Han JS. Effects of synchronous or asynchronous electroacupuncture stimulation with low versus high frequency on spinal opioid release and tail flick nociception. Exp Neurol. 2005;192:156–162. doi: 10.1016/j.expneurol.2004.11.003. [DOI] [PubMed] [Google Scholar]