Abstract
The last decade has witnessed dramatic growth in the number of reactions catalyzed by electrophilic gold complexes. While proposed mechanisms often invoke the intermediacy of gold-stabilized cationic species, the nature of bonding in these intermediates remains unclear. Herein, we propose that the carbon-gold bond in these intermediates is comprised of varying degrees of both σ and π-bonding; however, the overall bond order is generally less than or equal to unity. The bonding in a given gold-stabilized intermediate, and the position of this intermediate on a continuum ranging from gold-stabilized singlet carbene to gold-coordinated carbocation, is dictated by the carbene substituents and the ancillary ligand. Experiments show that the correlation between bonding and reactivity is reflected in the yield of gold-catalyzed cyclopropanation reactions.
The unique reactivity of organogold intermediates1 has recently enabled the development of a wide variety of new carbon–carbon bond forming reactions2. Based on the reactivity patterns that have emerged3, several mechanistic pathways4 and bonding models5 for key intermediates have been proposed, including intermediates ranging from gold carbenes6 to gold-stabilized carbocations7. In the last year, theoretical investigations8 and experimental observations9 have further polarized the discussion surrounding the carbenoid or cationic character of organogold species, mostly in support of their carbocationic character10. However, gold catalysis has been applied successfully to perform reactions that are traditionally carried out with carbenic systems11–16. Given this apparent lack of a consistent and clear understanding of the Au–CR2+ bond, we have performed a broad theoretical analysis on key intermediates relevant to gold(I) catalysis. Experimental results in support of our analysis are also presented.
The study of carbon–metal multiple bonds has been an area of intense discussion since their conception. For example, after the discovery of rhodium carbenoids, debate ensued as to the nature of the rhodium–carbon bond17. While certain calculations and experiments showed that the rhodium–carbon bond order was close to unity, eventually it was accepted, on the basis of reactivity, that a metal–carbon double bond18 was a more useful and convenient descriptor19. A similar discussion has recently emerged concerning the nature of gold–carbon bond. While partially semantic in nature, the correct description is useful for both predicting and explaining reactivity.
Results and Discussion
Barrier to bond rotation energy
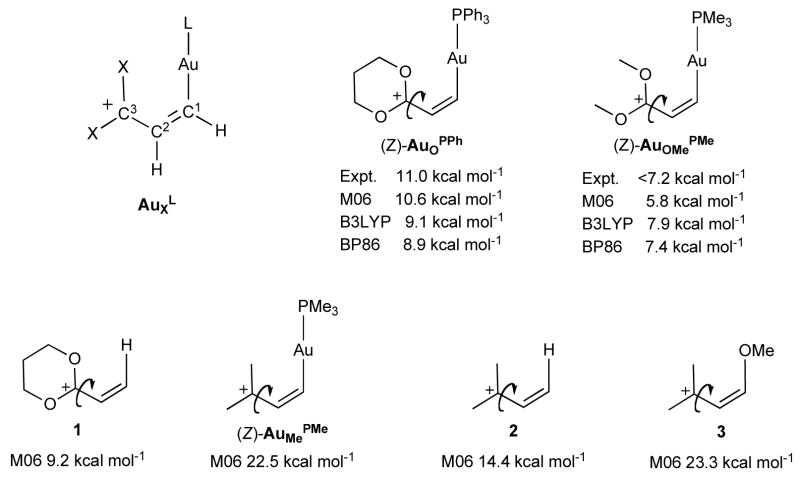
The magnitude of rotational barriers is a practical way of estimating the strength of π-bonds. Fürstner and co-workers devised a clever experiment to evaluate the carbenic/carbocationic character of (Z)-AuOPPh and (Z)-AuOMePMe by measuring the rotational barrier of bonds adjacent to gold10. They concluded that the contribution of the “carbene” resonance was marginal. We used Fürstner’s experiments to validate our theoretical methodology based on the M06 flavor20 of density functional theory (DFT).
Recently, the M06 flavor of DFT has been shown to accurately describe21,22 transition metal catalyzed organic transformations. To confirm this, we calculated rotational barriers for (Z)-AuOPPh and (Z)-AuOMePMe (Figure 1) and obtained ΔG‡=10.6 kcal mol−1 for (Z)-AuOPPh, in excellent agreement with experiment (ΔG‡=11.0) and ΔG‡=5.8 for (Z)-AuOMePMe, also consistent with experiment (<7.2 kcal mol−1). Previous DFT studies8 used either B3LYP or BP86 functionals. Although these methods have proven valuable for many organometallic studies, we find that they are insufficient to resolve the issues of interest here. For example, B3LYP predicts rotational barriers of ΔG‡=9.1 and 7.9 kcal mol−1, while BP86 predicts ΔG‡=8.9 and 7.4 kcal mol−1 for (Z)-AuOPPh and (Z)-AuOMePMe, respectively. This suggests that both B3LYP and BP86 cannot resolve the effects of the more electron donating PMe3 from the less electron donating PPh3.
Figure 1.
Calculated and experimental activation energies to bond rotation (indicated with arrows). C3-C2 bond rotation barriers are decreased when C3 is substituted with carbocation-stabilizing oxygen atoms. Throughout the text gold complexes will be referred to according to the AuXL notation, where L indicates the auxiliary ligand on gold and X indicates the C3 substituents (Me = methyl, O = 1,3-dioxanyl, OMe = methoxy, E = methyl ester).
Using our validated computational method, we calculated the barriers to bond rotation for metal-free allyl cations 1, 2, and 3, as well as the gold species (Z)-AuMePMe. The results confirm the conclusion of Fürstner and coworkers10 that the gold-moiety has little effect on the barrier to rotation in (Z)-AuOPPh ((Z)-AuOPPh versus 1 ~ 1.4 kcal mol−1); however, this conclusion is only valid in the presence of the highly carbocation-stabilizing oxygen atoms. When these heteroatoms are absent, the effect of the gold moiety is quite large ((Z)-AuMePMe versus 2 ~ 8.1 kcal mol−1). For comparison, the highly stabilizing methoxy group in 3 increases the barrier to rotation by 8.9 kcal mol−1 versus 2.
These results suggest that the reactivity of a given gold-stabilized carbene is highly dependent upon the carbene substituents. To gain further insight into the nature of the gold-carbon bond, we examined the structures of these types of intermediates, while varying both carbene and auxiliary ligand substituents.
Impact of the carbene
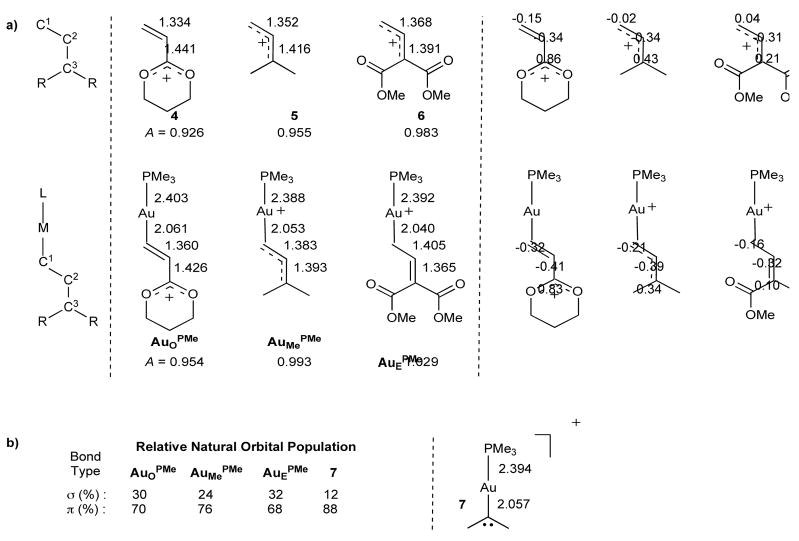
As a basis for comparison, we began by calculating the structures and natural atomic charges23 of metal-free allyl cations 4, 5, and 6 (Figure 2a). We also defined parameter A as the ratio of the bond lengths: A ≡ (C1-C2)/(C2-C3), so that A indicates roughly whether the partial positive charge on the substrate is more stabilized by its C1 or C3 substituents. The low value of A in 4 (0.926) is indicative of the stabilizing nature of the oxygen lone pairs. The magnitude of A increases with less-donating C3-methyl substituents (A = 0.955) and even further for ester-substituted substrate 6 (A = 0.983). The corresponding gold-coordinated structures AuOPMe, AuMePMe and AuEPMe, all show increased A values as a result of the ability of the gold moiety to stabilize positive charge at C1. In AuMePMe, A is close to 1 (0.993), suggesting that a secondary gold-stabilized carbocation is as stabilized as a tertiary carbocation. For the diester-substituted allyl carbene AuEPMe, the π system is now polarized towards the electron deficient C3 leading to A=1.029. Importantly, the magnitude of stabilization from the gold-moiety increases with increasing electrophilicity of the allyl-cation. This conclusion can also be reached by considering the natural atomic charge on C3. This charge is essentially unaffected in 4 (0.86) versus AuOPMe (0.83), while it is significantly reduced in 5 (0.43) versus AuMePMe (0.34) and 6 (0.21) versus AuEPMe (0.10).
Figure 2.
Structural and electronic comparison of cationic metal-free and [AuPMe3]+ substituted substrates. (a) On the left, calculated bond distances (Å), and on the right, natural charges for C1, C2, and C3. Parameter A is defined as the ratio of bond distances (C1–C2)/(C2–C3) and correlates to the polarization of the π-electrons along the delocalized C1–C2–C3 system. (b) A comparison of with Au-alkylidene 7 showing the relative natural orbital populations of σ and π contributions to the Au-C bond. On the right calculated bond distances (Å) for 7.
In order to examine the bonding and reactivity of non-vinylic gold(I) intermediates, we also examined gold alkylidene 7 and compared it to the gold vinylic carbene species (Figure 2b). Our results show that the gold–carbon bond distance (2.057 Å) varies little from the vinyl carbenes. However, in the absence of a delocalized neighboring vinyl group, the Au–C bond possesses a higher π-character, with decreased σ-donation from alkylidene to gold24. This demonstrates the ability of the gold moiety to stabilize carbenes of varying electrophilicity by modulating the nature of the gold carbon bond.
A direct consequence of the differences between AuOPMe and AuMePMe is the increased barrier to C2–C3 bond rotation in AuMePMe (Figure 1). In addition, a difference in reactivity could be expected: AuOPMe may react as a gold-stabilized carbocation, while AuMePMe may react more as a gold-stabilized carbene. We tested and confirmed this hypothesis experimentally. We were unable to observe productive cyclopropanation of intermediates resulting from the gold-catalyzed reaction of cyclopropene 8 (as a precursor to AuOPMe, Figure 3a). On the other hand, gold-catalyzed reaction of cyclopropene 9 (which should decompose to an intermediate similar to AuMePMe) with cis-stilbene provided the product of stereospecific olefin cyclopropanation (Figure 3b). As the yield of this reaction was highly dependent on the auxiliary ligand, we next examined the effect of this ligand on nature of the Au–C1 bond.
Figure 3.
Experimental and theoretical comparison for the carbene reactivity of the substrate with different ancillary ligands. (a) Attempted and (b) observed carbene-like reactivity, demonstrating the impact of the auxiliary ligand on the yield of cyclopropanation product. (c) Bond distances in AuMeL complexes. (d) A stylized depiction of the most important bonding interactions in L-Au(I)-CR2+ species.
Impact of the ligand1,25
The computed structures of AuMeL, for various ligands (L) are shown in Figure 3c. The L-Au–C1 bonding network can be partitioned into three components (Figure 3d)26,27. Since there is only one vacant valence orbital on Au (6s), the Pauli exclusion principle tells us that a 3c/4e σ-hyperbond28 (where hyperbond refers to bonding beyond the reduced 12e valence space) must be formed for the P–Au–C triad as [P:Au–C↔P–Au:C] (or C–Au–C triad for NHC ligand). As a result, the Au–C1 bond order decreases with increasing trans ligand σ-donation (trans influence). In the absence of a trans ligand, the gold–carbon bond in AuMe is notably shorter (1.985 Å); whereas, in AuMePMe, the electron donating PMe3 (strong trans influence) results in a correspondingly long Au–C1 bond length of 2.053 Å.
In addition, the metal center is able to form two π-bonds by donation from perpendicular filled d-orbitals into empty π-acceptors on the ligand and C1. While these two bonds are not mutually exclusive, they compete for electron density from gold. As a result, strongly π-acidic ligands decrease back donation to the substrate, resulting in even longer Au–C1 bonds (2.057 Å for AuMePOMe). In contrast, the π-donating chloride ligand in AuMeCl increases back-donation to C1 resulting in a very short gold–carbon bond (1.969 Å). In general, the strength of the back-donation to C1 is dependent on both ligand and, (as demonstrated in the previous section) the electrophilicity of the π-acceptor on C1.
The impact of these changes on reactivity is directly apparent in the yield of cyclopropanation product (Figure 3b). π-Acidic ligands are expected to increase carbocation-like reactivity by decreasing gold to C1 π-donation. Accordingly, we found that strongly π-acidic phosphite ligands provide only traces of the desired product and significant polymerization. Conversely, ligands which increase gold to C1 π-donation are expected to reduce carbocation-like reactivity, while those ligands that decrease C1 to gold σ-donation should increase carbene-like reactivity. The N-heterocyclic ligand IPr (1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene) should affect both of these changes (it is strongly σ-donating and only weakly π-acidic). Gratifyingly, we found that IPrAu+ provides the product of stereospecific cyclopropanation in excellent yield and with high diastereoselectivity (80% yield, 11:1 cis:trans). Phosphine ligands fell in between these two extremes. Finally, AuCl was unreactive under these conditions.
Charge distribution
In order to provide a better and more general view of the bonding in gold vinyl carbene species, we calculated natural charge distributions for the ligand (L), Au, and substrate using NBO analyses. Table 1 shows that the charge is relatively equally distributed between the substrate, gold, and the ligand. Across AuMeL and AuOL series the charge on ligand and Au is well correlated: a change in ligand that results in an increase in ligand charge is correlated with a similar decrease in charge on gold, but these changes have little effect on the charge on the substrate. On the other hand, changing the substrate from AuMeL to AuOL results in an increase in substrate charge. This correlates with a decrease in the charges on both Au and ligand. These results again demonstrate that both the ligand and the substrate play important roles in determining the overall electronic structure.
Table 1.
Calculated natural populations (charge) for the ancillary ligand, gold atom, and substrate.
| AuMeNHC | AuMePMe | AuMePOMe | AuONHC | AuOPMe | AuOPOMe | |
|---|---|---|---|---|---|---|
| Ligand | 0.32 | 0.40 | 0.45 | 0.31 | 0.37 | 0.42 |
| Metal | 0.39 | 0.30 | 0.25 | 0.36 | 0.27 | 0.23 |
| Substrate | 0.29 | 0.31 | 0.31 | 0.33 | 0.36 | 0.35 |
Bonding and reactivity
The model in Figure 3d for bonding in gold-stabilized carbenes proposes that these intermediates possess highly electron-deficient α-carbons that are stabilized, to varying degrees, by back-donation from the metal to the vacant pπ-orbital of the singlet carbene. This electronic deficiency reduces donation from the filled sp2 σ-orbital of the carbene to the metal, therefore minimizing gold–carbon σ-bonding. Thus, our model suggests that the conversion of a vinylgold intermediate into a gold-stabilized carbene, which is commonly proposed in gold-catalyzed reactions29–34, occurs with an increase in gold–carbon π-bonding and a decrease in the σ-bonding (Figure 4). The bonding situation in these carbene intermediates has often been depicted by two extreme resonance structures: a carbocation with a gold-carbon single bond or a carbene with a gold-carbon double bond. Much like the double “half-bond” model proposed for rhodium-carbenoid intermediates35,36, the depiction of a gold-stabilized carbene with a gold-carbon double bond should not be taken as an indication of a bond order of 2; but rather a means to convey that both σ and π components to the bond are present. To illustrate this, we calculated gold-carbon natural bond orders for AuOPMe (0.53), AuMePMe (0.91), and 7 (1.14). Nucleophilic attack on the now highly electrophilic pπ-orbital of the carbon adjacent to gold restores the gold-carbon sigma bond37–39. In this scenario, divergence towards carbocation-like or carbene-like reactivity may also be influenced by the potential of the nucleophile to intercept the developing positive charge. Alternatively, gold-stabilized carbene intermediates may react with concerted carbene-like reactivity (i.e. cyclopropanation), especially when the gold is coordinated to electron donating ligands.
Figure 4.
Arrow pushing in the formation of gold-stabilized carbenes is a useful mnemonic for keeping track of electrons, but it can lead to misconceptions about bonding. While the Au-C bonsd in the intermediate carbene has both σ and π components, the overall bond order is generally less than or equal to unity.
Conclusions
We suggest the reactivity in Au(I)-coordinated carbenes is best accounted for by a continuum ranging from metal stabilized singlet carbene to metal-coordinated carbocation. The position of a given gold species on this continuum is largely determined by the carbene substituents and the ancillary ligand. Consideration of the bonding description described herein provides insight into previously reported gold-catalyzed transformations and a basis for the ab initio prediction of reactivity, optimization of ligand effects, and design of new gold-catalyzed reactions.
Methods
Calculations were performed using density functional theory (DFT) with the M06 functional, as implemented in Jaguar 7.640. All calculations used the Hay and Wadt small core-valence relativistic effective-core-potential41 (ECP) to describe the 1s-3d core electrons of the gold atom, leaving the outer 16 electrons (4s, 4p, 4d, 5s, etc.) to be treated explicitly. The LACVP** basis set was used for all geometry optimizations and LACV3P++**(2f) for energies. LACV3P++**(2f) utilizes the LACV3P++** basis set as implemented in Jaguar plus a double-zeta f-shell with exponents from Martin and Sundermann42. All electrons were described for all other atoms using the 6-31G** or 6-311++G** basis sets43,44. For each optimized structure, the M06 analytic Hessian was calculated to obtain the vibrational frequencies, which in turn were used to obtain the zero point energies and free energy corrections (without translational or rotational components). Solvent corrections were based on single point self-consistent Poisson-Boltzmann continuum solvation calculations for CH2Cl2 (ε = 8.93 and R0= 2.33 Å using the PBF45 module in Jaguar.
Supplementary Material
Acknowledgments
FDT gratefully acknowledges NIHGMS (RO1 GM073932), Bristol-Myers Squibb, and Novartis for funding, and Johnson Matthey for the generous donation of AuCl3. The MSC computational facilities were funded by grants from ARO-DURIP and ONR-DURIP. DB and ET thank Dr. Robert “Smith” Nielsen for useful suggestions.
Footnotes
Author contributions
DB, NDS and FDT originated the idea and wrote the manuscript; NDS and YW performed the experiments; DB and ET performed the calculations; all authors contributed to discussions and edited the manuscript; DB and NDS contributed equally to this work.
Additional information
Supplementary Information accompanies this paper at www.nature.com/naturechemistry. Reprints and permission information is available online at http://npg.nature.com/reprintsandpermissions/.
References
- 1.Gorin DJ, Sherry BD, Toste FD. Ligand effects in homogeneous Au catalysis. Chem Rev. 2008;108:3351–3378. doi: 10.1021/cr068430g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hashmi ASK. Gold-catalyzed organic reactions. Chem Rev. 2007;107:3180–3211. doi: 10.1021/cr000436x. [DOI] [PubMed] [Google Scholar]
- 3.Fürstner A, Davies PW. Catalytic carbophilic activation: catalysis by platinum and gold π acids. Angew Chem, Int Ed. 2008;46:3410–3449. doi: 10.1002/anie.200604335. [DOI] [PubMed] [Google Scholar]
- 4.Jiménez-Núñez E, Echavarren AM. Gold-catalyzed cycloisomerizations of enynes: a mechanistic perspective. Chem Rev. 2008;108:3326–3350. doi: 10.1021/cr0684319. [DOI] [PubMed] [Google Scholar]
- 5.Gorin DJ, Toste FD. Relativistic effects in homogeneous gold catalysis. Nature. 2007;446:395–403. doi: 10.1038/nature05592. [DOI] [PubMed] [Google Scholar]
- 6.Fedorov A, Moret ME, Chen P. Gas-phase synthesis and reactivity of a gold carbene complex. J Am Chem Soc. 2008;130:8880–8881. doi: 10.1021/ja802060t. [DOI] [PubMed] [Google Scholar]
- 7.Hashmi ASK. “High noon” in gold catalysis: Carbene versus carbocation intermediates. Angew Chem, Int Ed. 2008;47:6754–6756. doi: 10.1002/anie.200802517. [DOI] [PubMed] [Google Scholar]
- 8.Correa A, Marion N, Fensterbank L, Malacria M, Nolan SP, Cavallo L. Golden carousel in catalysis: the cationic gold/propargylic ester cycle. Angew Chem, Int Ed. 2008;47:718–721. doi: 10.1002/anie.200703769. [DOI] [PubMed] [Google Scholar]
- 9.Fürstner A, Morency L. On the nature of the reactive intermediates in gold-catalyzed cycloisomerization reactions. Angew Chem, Int Ed. 2008;47:5030–5033. doi: 10.1002/anie.200800934. [DOI] [PubMed] [Google Scholar]
- 10.Seidel G, Mynott R, Fürstner A. Elementary steps of gold catalysis: NMR spectroscopy reveals the highly cationic character of a “gold carbenoid. Angew Chem, Int Ed. 2009;48:2510–2513. doi: 10.1002/anie.200806059. [DOI] [PubMed] [Google Scholar]
- 11.Johansson MJ, Gorin DJ, Staben ST, Toste FD. Gold(I)-catalyzed stereoselective olefin cyclopropanation. J Am Chem Soc. 2005;127:18002–18003. doi: 10.1021/ja0552500. [DOI] [PubMed] [Google Scholar]
- 12.Horino Y, Yamamoto T, Ueda K, Kuroda S, Toste FD. Au(I)-catalyzed cycloisomerizations terminated by sp3 C–H bond insertion. J Am Chem Soc. 2009;131:2809–2811. doi: 10.1021/ja808780r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lemière G, et al. Generation and trapping of cyclopentenylidene gold species: four pathways to polycyclic compounds. J Am Chem Soc. 2009;131:2993–3006. doi: 10.1021/ja808872u. [DOI] [PubMed] [Google Scholar]
- 14.Fructos MR, Belderrain TR, Frémont P, Scott NM, Nolan SP, Díaz-Requejo MM, Pérez PJ. A gold catalyst for carbene-transfer reactions from ethyl diazoacetate. Angew Chem, Int Ed. 2005;44:5284–5288. doi: 10.1002/anie.200501056. [DOI] [PubMed] [Google Scholar]
- 15.López S, Herrero-Gómez E, Pérez-Galán P, Nieto-Oberhuber C, Echavarren AM. Gold(I)-catalyzed intermolecular cyclopropanation of enynes with alkenes: trapping of two different gold carbenes. Angew Chem, Int Ed. 2005;45:6029–6032. doi: 10.1002/anie.200602448. [DOI] [PubMed] [Google Scholar]
- 16.Fedorov A, Chen P. Electronic effects in the reactions of olefin-coordinated gold carbene complexes. Organometallics. 2009;28:1278–1281. [Google Scholar]
- 17.Sheehan SM, Padwa A, Snyder JP. Dirhodium(II) tetracarboxylate carbenoids as catalytic intermediates. Tetrahedron Lett. 1998;39:949–952. [Google Scholar]
- 18.Doyle MP. Electrophilic metal carbenes as reaction intermediates in catalytic reactions. Acc Chem Res. 1986;19:348–356. [Google Scholar]
- 19.Nowlan DT, Gregg TM, Davies HML, Singleton DA. Isotope effects and the nature of selectivity in rhodium-catalyzed cyclopropanations. J Am Chem Soc. 2004;125:15902–15911. doi: 10.1021/ja036025q. [DOI] [PubMed] [Google Scholar]
- 20.Zhao Y, Truhlar DG. Density functionals with broad applicability in chemistry. Acc Chem Res. 2008;41:157–167. doi: 10.1021/ar700111a. [DOI] [PubMed] [Google Scholar]
- 21.Truhlar DG. Molecular modeling of complex chemical systems. J Am Chem Soc. 2008;130:16824–16827. doi: 10.1021/ja808927h. [DOI] [PubMed] [Google Scholar]
- 22.Zhao Y, Truhlar DG. Benchmark energetic data in a model system for Grubbs II metathesis catalysis and their use for the development, assessment, and validation of electronic structure methods. J Chem Theory Comput. 2009;5:324–333. doi: 10.1021/ct800386d. [DOI] [PubMed] [Google Scholar]
- 23.Reed AE, Curtiss LA, Weinhold F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Re. 1988;88:899–926. [Google Scholar]
- 24.Irikura KK, Goddard WA., III Energetics of third-row transition metal methylidene ions MCH2+ (M = La, Hf, Ta, W, Re, Os, Ir, Pt, Au) J Am Chem Soc. 1994;116:8733–8740. [Google Scholar]
- 25.Padwa A, Austin DJ. Ligand effects on the chemoselectivity of transition metal catalyzed reactions of α-diazo carbonyl compounds. Angew Chem Int Ed Engl. 1994;33:1797–1815. [Google Scholar]
- 26.Dewar M. Bull Soc Chim Fr. 1951;18:C71–C77. [Google Scholar]
- 27.Chatt J, Duncanson LA. Olefin co-ordination compounds. Part III. Infra-red spectra and structure: attempted preparation of acetylene complexes. J Chem Soc. 1953:2939–2947. [Google Scholar]
- 28.Landis CR, Weinhold F. Valence and extra-valence orbitals in main group and transition metal bonding. J Comput Chem. 2007;28:198–203. doi: 10.1002/jcc.20492. [DOI] [PubMed] [Google Scholar]
- 29.Mamane V, Gress T, Krause H, Fürstner A. Platinum- and gold-catalyzed cycloisomerization reactions of hydroxylated enynes. J Am Chem Soc. 2004;126:8654–8655. doi: 10.1021/ja048094q. [DOI] [PubMed] [Google Scholar]
- 30.Luzung MR, Markham JP, Toste FD. Catalytic isomerization of 1,5-enynes to bicyclo[3.1.0]hexenes. J Am Chem Soc. 2004;126:10858–10859. doi: 10.1021/ja046248w. [DOI] [PubMed] [Google Scholar]
- 31.Gorin DJ, Davis NR, Toste FD. Gold(I)-catalyzed intramolecular acetylenic Schmidt reaction. J Am Chem Soc. 2005;127:1126–1127. doi: 10.1021/ja053804t. [DOI] [PubMed] [Google Scholar]
- 32.Nieto-Oberhuber C, Muñoz MP, Buñuel E, Nevado C, Cárdenas DJ, Echavarren AM. Cationic gold(I) complexes: highly alkynophilic catalysts for the exo- and endo-cyclization of enynes. Angew Chem, Int Ed. 2004;43:2402–2406. doi: 10.1002/anie.200353207. [DOI] [PubMed] [Google Scholar]
- 33.Shapiro ND, Toste FD. Rearrangement of alkynyl sulfoxides catalyzed by gold(I) complexes. J Am Chem Soc. 2007;129:4160–4161. doi: 10.1021/ja070789e. [DOI] [PubMed] [Google Scholar]
- 34.Zhang G, Zhang L. Au-containing all-carbon 1,3-dipoles: generation and [3+2] cycloaddition reactions. J Am Chem Soc. 2008;130:12598–12599. doi: 10.1021/ja804690u. [DOI] [PubMed] [Google Scholar]
- 35.Snyder JP, Padwa A, Stengel T, Arduengo AJ, III, Jockisch A, Kim H. A stable dirhodium tetracarboxylate carbenoid: crystal structure, bonding analysis, and catalysis. J Am Chem Soc. 2001;123:11318–11319. doi: 10.1021/ja016928o. [DOI] [PubMed] [Google Scholar]
- 36.Costantino G, Rovito R, Macchiarulo A, Pellicciari R. Structure of metal–carbenoid intermediates derived from the dirhodium(II)tetracarboxylate mediated decomposition of α-diazocarbonyl compounds: a DFT study. J Mol Struct Theochem. 2002;581:111. [Google Scholar]
- 37.Amijs CHM, López-Carrillo V, Echavarren AM. Gold-catalyzed addition of carbon nucleophiles to propargyl carboxylates. Org Lett. 2007;9:4021–4024. doi: 10.1021/ol701706d. [DOI] [PubMed] [Google Scholar]
- 38.Davies PW, Albrecht SJ-C. Alkynes as masked ylides: gold-catalysed intermolecular reactions of propargylic carboxylates with sulfides. Chem Commun. 2008:238–240. doi: 10.1039/b714813e. [DOI] [PubMed] [Google Scholar]
- 39.Nieto-Oberhuber C, Muñoz MP, López S, Jiménez-Núñez E, Nevado C, Herrero-Gómez E, Raducan M, Echavarren AM. Gold(I)-catalyzed cyclizations of 1,6-enynes: alkoxycyclizations and exo/endo skeletal rearrangements. Chem Eur J. 2006;12:1677–1693. doi: 10.1002/chem.200501088. [DOI] [PubMed] [Google Scholar]
- 40.Jaguar 7.6. Schrodinger, LLC; New York, NY: 2006. [Google Scholar]
- 41.Hay PJ, Wadt WR. Ab initio effective core potentials for molecular calculations - potentials for K to Au including the outermost core orbitals. J Chem Phys. 1985;82:299–310. [Google Scholar]
- 42.Martin JML, Sundermann A. Correlation consistent valence basis sets for use with the Stuttgart-Dresden-Bonn relativistic effective core potentials: The atoms Ga-Kr and In-Xe. J Chem Phys. 2001;114:3408–3420. [Google Scholar]
- 43.Krishnan R, Binkley JS, Seeger R, Pople JA. Self-consistent molecular-orbital methods. XX A basis set for correlated wave-functions. J Chem Phys. 1980;72:650–654. [Google Scholar]
- 44.Frisch MJ, Pople JA, Binkley JS. Self-consistent molecular-orbital methods 25. Supplementary functions for Gaussian-basis sets. J Chem Phys. 1984;80:3265–3269. [Google Scholar]
- 45.Tannor DJ, et al. Accurate first principles calculation of molecular charge-distributions and solvation energies from ab-initio quantum-mechanics and continuum dielectric theory. J Am Chem Soc. 1994;116:11875–11882. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.