Abstract
Background and Aims
A comparative investigation was made of floral scent variation in the closely related, food-rewarding Anacamptis coriophora and the food-deceptive Anacamptis morio in order to identify patterns of variability of odour compounds in the two species and their role in pollinator attraction/avoidance learning.
Methods
Scent was collected from plants in natural populations and samples were analysed via quantitative gas chromatography and mass spectrometry. Combined gas chromatography and electroantennographic detection was used to identify compounds that are detected by the pollinators. Experimental reduction of scent variability was performed in the field with plots of A. morio plants supplemented with a uniform amount of anisaldehyde.
Key Results
Both orchid species emitted complex odour bouquets. In A. coriophora the two main benzenoid compounds, hydroquinone dimethyl ether (1,4-dimethoxybenzene) and anisaldehyde (methoxybenzaldehyde), triggered electrophysiological responses in olfactory neurons of honey-bee and bumble-bee workers. The scent of A. morio, however, was too weak to elicit any electrophysiological responses. The overall variation in scent was significantly lower in the rewarding A. coriophora than in the deceptive A. morio, suggesting pollinator avoidance-learning selecting for high variation in the deceptive species. A. morio flowers supplemented with non-variable scent in plot experiments, however, did not show significantly reduced pollination success.
Conclusions
Whereas in the rewarding A. coriophora stabilizing selection imposed by floral constancy of the pollinators may reduce scent variability, in the deceptive A. morio the emitted scent seems to be too weak to be detected by pollinators and thus its high variability may result from relaxed selection on this floral trait.
Key words: Anacamptis coriophora, Anacamptis morio, food deception, floral odour, GC-EAD, pollination, scentless flowers
INTRODUCTION
Insect-pollinated plants advertise their flowers with colour, shape and scent to attract their pollinators (Fægri and van der Pijl, 1979). Pollinators may be attracted to floral signals innately, as was shown for colour (Kugler, 1934; Gumbert, 2000) and scent (Henning et al., 1992; Andersson and Dobson, 2003), but pollinators also learn to associate floral rewards with flower traits, such as colour (Heinrich et al., 1977; Kunze and Gumbert, 2001), shape (Manning, 1956) and scent (Wells and Wells, 1985; Laloi et al., 1999). The association of floral signals with floral reward can lead to flower constancy, i.e. when insects restrict their visits to one flower type, even when other rewarding types are available (Heinrich et al., 1977; Waser, 1986). This behaviour of pollinators is favourable for plants, because it reduces inappropriate pollen transfer, and clogging of stigmas with non-compatible pollen (Jones, 2001). Flower constancy can be influenced by various factors, such as innate preferences of the pollinators, complexity of the flowers, amount of reward offered and the frequency of a particular flower type within a population or a plant community (Real, 1981; Smithson and Macnair, 1997a; Gegear and Laverty, 2001). Although it is doubtful that flower constancy plays an important role in plant speciation (Chittka, 1999; Goulson, 1999), it seems likely that this behaviour imposes stabilizing selection on flower signals, as it selects against phenotypes that fall out of the search image of the pollinators. Experiments have also shown that bees forage in a positive frequency-dependent manner, preferring the most common flower types over rare ones (Smithson and Macnair, 1996, 1997a). This positive frequency-dependent foraging behaviour of pollinators can also select for reduced variation in floral signals, because more common phenotypes have an advantage.
An opposite scenario is predicted when flowers are deceptive, i.e. offer no reward to their pollinators (Ackerman, 1986; Nilsson, 1992; Schiestl, 2005). Among orchids, this pollination syndrome is common, and can be classified into two different systems. Orchids that mimic a specific rewarding species are considered true Batesian mimics (Dafni and Ivri, 1981; Roy and Widmer, 1999; Johnson, 2000). By contrast, food-fraud orchids can be ‘non-model mimics’ or ‘general food mimics’ and attract inexperienced pollinators with general floral features, but do not mimic a specific plant species (Nilsson, 1984; Dafni, 1986). Non-model mimics often grow gregariously and produce conspicuous floral displays (Nilsson 1980, 1992). In non-model deceptive pollination systems, pollinators are expected to learn and avoid floral signals in subsequent visits, and because pollinators encounter common morphs more often than rare ones, they may proportionally over-visit rare morphs. Rare floral morphs of the non-model mimic are expected to experience a selective advantage in comparison with common morphs, and thus negative frequency-dependent selection (nFDS) may maintain high polymorphism of floral traits (Smithson and Macnair, 1997b; Ferdy et al., 1998). Indeed, in many systems, a high variability in floral characters has been documented and interpreted as an adaptive trait to delay avoidance learning of the pollinators (Heinrich, 1975; Nilsson, 1992; Moya and Ackerman, 1993). Only a few studies, however, have tested this hypothesis experimentally (Smithson and Macnair, 1997b; Gigord et al., 2001; Aragón and Ackerman, 2004), and only one of them has dealt with scent (Ackerman et al., 1997). Up to now, nFDS, maintaining high polymorphism of a floral trait, has been reported only for the two colour morphs of Dactylorhiza sambucina (Gigord et al., 2001).
In the present study, a phylogenetically balanced, comparative approach was used to investigate patterns of variation in floral scent in two different pollination systems and an experiment was used to test whether high variability is adaptive in a food deceptive system. With this aim, floral scent and its variability were analysed in a rewarding and in a non-model food-deceptive species of the orchid genus Anacamptis. Anacamptis coriophora produces nectar and is pollinated by various types of bees, including bumble-bees and honey-bees (Dafni and Ivri, 1979; Van der Cingel, 1995). Nectar production is probably a derived trait within the genus Anacamptis (Bateman et al., 2003). The closely related Anacamptis morio, by contrast, is food deceptive but also pollinated by bumble-bees (Nilsson, 1984; Cozzolino et al., 2005). Specifically, the following questions were addressed: (1) Which odour compounds are emitted by flowers of the two orchids? (2) Which compounds are detected by the pollinators? (3) What are patterns of variability of odour compounds in the two species? (4) Are high amounts of variability in the deceptive system adaptive by reducing avoidance learning by pollinators?
MATERIALS AND METHODS
Scent collection
Between 20 and 30 plants were chosen randomly from several populations in Italy and Switzerland for odour collection. Plants of Anacamptis morio (L.) R. M. Bateman, Pridgeon and M. W. Chase were sampled from three populations, one in southern Italy (Cilento National Park, Monte San Giacomo, Salerno) and two in Switzerland (Randen, Schaffhausen and Wollishofen, Zürich). Plants of Anacamptis coriophora (L.) R. M. Bateman, Pridgeon and M. W. Chase were sampled from two populations, one in northern Italy (Bassa del Bardello, Ravenna) and one in southern Italy (Varriconi, Caserta). Odour was collected by headspace sorption, a standard method that has been proven reliable in many systems (Schiestl and Marion-Poll, 2002; Tholl and Röse, 2006). The inflorescence was covered with a polyethylene terephtalate (PET) cooking bag (Nalophand, Kalle GmbH, Wiesbaden, Germany) and air was removed by using a battery-operated vacuum pump (SKC Inc., Eighty Four, PA, USA; Huber et al., 2005). Volatiles were trapped on 2·5 mg of Porapak Q sealed in a glass tube. Before use, the Porapak Q was cleaned with 100 mL dichloromethane and 100 mL of a hexane/acetone mixture (9 : 1). Ambient air was collected as control samples to identify and subtract background contamination. After a sampling period of 3–8 h (approx. 10 mL min−1), adsorbed volatiles were eluted from the Porapak Q with 50 mL of a mixture of hexane/acetone (9 : 1). Samples were sealed in glass vials and stored at −20 °C.
Quantitative gas chromatography (GC) analyses and GC-mass spectrometry
For quantitative analysis, 100 ng of n-octadecane was added to all samples as an internal standard. One millilitre of each odour sample was injected splitless at 40 °C (1 min) into a gas chromatograph (GC; Agilent 6890 N) followed by opening the split valve and programming to 300 °C at a rate of 10 °C min−1. The GC was equipped with an HP-5 column (30 m, diameter = 0·32 mm, film thickness = 0·25 mm, Agilent Technologies). Hydrogen was used as carrier gas and nitrogen as make-up gas. For compound identifications, selected samples were analysed by gas chromatography with mass selective detection (GC-MS; Hewlett Packard G1800 A) using the same GC parameters as described above. Compounds were identified by comparison of MS spectra and GC retention times with those of reference compounds. Reference compounds and mass spectra of the compounds for identification were kindly provided by Roman Kaiser (Givaudan Schweiz AG, Dübendorf, Switzerland).
Absolute amounts of odour compounds were calculated using the internal standard method (Schomburg, 1990) according to the formula: (area peak/area internal standard) × 100. Sampling times and sampling volumes were used to calculate the absolute amount per litre sampled air, per hour and per plant. Relative amounts were calculated by dividing individual amounts by the sum of all compounds. Only those compounds were used in the analyses that did not occur, or occurred in statistically significantly lower amounts in the control samples.
Identification of biologically active compounds
To identify compounds which are detected by the pollinators (‘biologically active compounds’), ten samples each of A. morio and A. coriophora were pooled to provide a representative sample comprising most compounds of both species. These samples were then analysed via combined gas chromatography and electroantennographic detection (GC-EAD; Schiestl and Marion-Poll, 2002). For each analysis, one antenna of a Bombus terrestris worker (Andermatt Biocontrol AG, Grossdietwil, Switzerland) or Apis mellifera worker (Botanic Garden Zürich, Switzerland) was cut off at its base and mounted between two silver electrodes. Contact was made with the help of an electroconductive gel (Parker Laboratories). The preparation was shielded with a Faraday cage to reduce electrical interference. A GC effluent splitter (SGE Australia; split ratio 1 : 1) was used and the outlet was placed in a purified and humidified air stream. This air was directed over the antennae from which summed olfactory neuron responses (EAD) were recorded via Syntech software (Kirchzarten, Germany). EAD signals and flame ionization detector (FID) responses were simultaneously recorded. For each bee species, at least five individuals were tested with each odour-type. A compound was judged EAD active when a clear EAD response was consistently present in at least three GC-EAD runs with three different antennae.
Scent variability and pollination success in A. morio
The aim of the experiment was to test the adaptive value of floral scent variation by making flowers of A. morio more similar to A. coriophora in terms of active compound emission and its variation. This was achieved by adding a uniform amount of anisaldehyde, an active compound naturally produced by A. coriophora, to the flowers of A. morio. A rubber septum (Supelco, Bellefonte, PA, USA) beaded on a thread was soaked for 1 h in a solution of anisaldehyde/dichloromethane (1 : 10). A scented septa was coiled around each A. morio inflorescence and was replaced every second day by a freshly scented one. In a pre-experimental study, the odour emission of septa treated in that way was found to correspond to the amount of natural anisaldehyde emission of A. coriophora (data not shown).
The experiment was conducted from 26 April to 2 May, 2005 in Portici (Naples), southern Italy. Forty-six artificial plots in three patches of an agricultural grassland at the foot of Mt. Vesuvius were established. A plot consisted of a polystyrene rack of 0·58 × 0·32 × 0·06 m, in which nine vials were placed evenly, each filled with water and containing a cut inflorescence of A. morio. The plants originated from populations in the area and were randomly assigned to the plots. Flowers with pollen on the stigma or with pollinia removed were removed before the onset of the experiment. The distance between plots was 3 m. All plants of a plot were treated identically: plots either contained plants that received scent treatment (scented rubber septum) or plants that received a control treatment (scentless septum). The application of the same scent to all plants in treatment plots enhanced odour emission and reduced odour variability among individuals – individuals in control plots, however, retained the high odour variability of natural populations. After the experiment, male and female reproductive success (removed pollinia or massulae on stigma, respectively) of each plant were recorded. As male and female pollination success did not differ, the two measurements were combined to give: pollination success = (sum of flowers pollinated or pollinia removed)/(total number of flowers).
Statistical analyses
Variability of odour compounds was assessed for both species by calculating the coefficient of variance (CV = standard deviation/mean). Mean CV was compared among populations within species and between species as well as between active and non-active odour compounds in A. coriophora (Mann–Whitney U-test with subsequent Bonferroni correction, where appropriate). The absolute amount of scent was also compared between species by using the Mann–Whitney U-test.
Data from the A. morio experiment for scent variability were analysed with an ANOVA with mean pollination success per plot as the dependent variable, treatment and plot as fixed factors and number of flowers as covariate. For all statistical analyses, the program SPSS 12·0·1 for Windows (SPSS Inc., Chicago, IL, USA) was used.
RESULTS
Scent analyses
In both orchid species, numerous floral volatiles were found; 34 compounds were identified in A. morio and 54 compounds in A. coriophora (Tables 1 and 2). The floral bouquet of A. coriophora was mainly characterized by high amounts of benzenoids (>80 %). Two benzenoids, anisaldehyde and hydroquinone dimethyl ether, dominated the scent profile, making up 83 % (‘Varriconi’ population) and 60 % (‘Ravenna’ population) of the total odour bouquet. In A. morio, monoterpenes were present in the scent profile of the Italian population, comprising more than 50 % of the total, but made up only a minor proportion in the two Swiss populations (16 and 22 %, respectively). The absolute amount of scent emission was significantly lower in A. morio than in A. coriophora (Mann–Whitney U-test, U = 341, P < 0·001; Fig. 1).
Table 1.
Mean relative values (± s.e.) of odour compounds identified in headspace samples of Anacamptis morio
| Italy (Cilento), n = 20 | Switzerland (Schaffhausen), n = 21 | Switzerland (Zurich), n = 18 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Mean ± s.e. | Min. | Max. | CV | % | Mean ± s.e. | Min. | Max. | CV | % | Mean ± s.e. | Min. | Max. | CV | % |
| Fatty acid derivatives | 0·18 ± 0·13 | 0·00 | 2·56 | 3·18 | 1·44 ± 0·35 | 0·16 | 7·51 | 1·13 | 3·13 ± 0·39 | 0·60 | 6·30 | 0·53 | |||
| Aldehydes | 0·18 ± 0·13 | 0·00 | 2·56 | 3·18 | 1·44 ± 0·35 | 0·16 | 7·51 | 1·13 | 3·13 ± 0·39 | 0·60 | 6·30 | 0·53 | |||
| Nonanal | 0·18 ± 0·13 | 0·00 | 2·56 | 3·18 | 30 | 1·44 ± 0·35 | 0·16 | 7·51 | 1·13 | 100 | 3·13 ± 0·39 | 0·60 | 6·30 | 0·53 | 100 |
| Benzenoids | 1·87 ± 0·74 | 0·08 | 12·56 | 1·77 | 2·30 ± 0·32 | 0·10 | 6·09 | 0·64 | 4·15 ± 0·60 | 0·00 | 8·25 | 0·61 | |||
| Aldehydes | 1·87 ± 0·74 | 0·08 | 12·56 | 1·77 | 2·30 ± 0·32 | 0·10 | 6·09 | 0·64 | 4·15 ± 0·60 | 0·00 | 8·25 | 0·61 | |||
| Benzaldehyde | 1·87 ± 0·74 | 0·08 | 12·56 | 1·77 | 100 | 2·30 ± 0·32 | 0·10 | 6·09 | 0·64 | 100 | 4·15 ± 0·60 | 0·00 | 8·25 | 0·61 | 89 |
| Isoprenoids | 53·15 ± 7·27 | 0·00 | 93·28 | 0·61 | 15·75 ± 4·59 | 3·02 | 73·59 | 1·33 | 21·68 ± 4·68 | 5·6 | 73·7 | 0·92 | |||
| Irregular terpenes | 0·30 ± 0·12 | 0·00 | 1·53 | 1·78 | 0·51 ± 0·14 | 0·00 | 2·94 | 1·27 | 0·75 ± 0·17 | 0·00 | 2·30 | 0·97 | |||
| 6-Methyl-5-hepten-2one | 0·30 ± 0·12 | 0·00 | 1·53 | 1·78 | 40 | 0·51 ± 0·14 | 0·00 | 2·94 | 1·27 | 91 | 0·75 ± 0·17 | 0·00 | 2·30 | 0·97 | 72 |
| Monoterpenes | 52·85 ± 7·31 | 0·00 | 93·28 | 0·62 | 15·24 ± 4·61 | 2·50 | 73·35 | 1·39 | 20·93 ± 4·65 | 5·6 | 73·5 | 0·94 | |||
| α-Pinene | 11·34 ± 2·63 | 0·00 | 37·10 | 1·04 | 85 | 2·50 ± 1·60 | 0·00 | 30·78 | 2·93 | 71 | 3·25 ± 2·11 | 0·00 | 31·38 | 2·75 | 39 |
| Camphene | 0·31 ± 0·22 | 0·00 | 4·54 | 3·20 | 50 | 0·03 ± 0·03 | 0·00 | 0·50 | 3·50 | 10 | 0·02 ± 0·02 | 0·00 | 0·24 | 2·93 | 11 |
| Sabinene | 8·65 ± 1·71 | 0·00 | 20·50 | 0·88 | 85 | 1·95 ± 0·66 | 0·13 | 11·85 | 1·55 | 100 | 2·07 ± 0·97 | 0·00 | 13·54 | 1·98 | 50 |
| β-Pinene | 1·84 ± 0·35 | 0·00 | 4·98 | 0·86 | 75 | 0·92 ± 0·26 | 0·15 | 5·07 | 1·27 | 100 | 1·93 ± 0·34 | 0·00 | 4·58 | 0·74 | 83 |
| Myrcene | 5·60 ± 1·18 | 0·00 | 21·43 | 0·94 | 95 | 2·26 ± 0·50 | 0·24 | 9·21 | 1·02 | 100 | 2·95 ± 0·53 | 0·00 | 6·62 | 0·77 | 83 |
| β-Phellandrene | 0·26 ± 0·06 | 0·00 | 0·76 | 0·97 | 70 | 0·91 ± 0·12 | 0·25 | 2·16 | 0·61 | 100 | 3·29 ± 1·19 | 0·00 | 20·60 | 1·53 | 83 |
| Limonene/eucalyptol | 13·08 ± 2·36 | 0·00 | 36·32 | 0·81 | 80 | 1·48 ± 0·62 | 0·09 | 11·33 | 1·90 | 100 | 2·45 ± 0·87 | 0·00 | 12·23 | 1·50 | 67 |
| γ-Terpinene | 1·00 ± 0·44 | 0·00 | 8·19 | 1·97 | 45 | 0·16 ± 0·11 | 0·00 | 1·74 | 3·17 | 10 | 0·15 ± 0·08 | 0·00 | 0·94 | 2·31 | 17 |
| Linalool | 7·68 ± 4·03 | 0·00 | 77·65 | 2·35 | 85 | 4·16 ± 3·06 | 0·00 | 65·23 | 3·37 | 95 | 2·54 ± 0·57 | 0·00 | 9·18 | 0·95 | 89 |
| α-Pinene-epoxide | 1·10 ± 0·27 | 0·00 | 3·81 | 1·11 | 75 | 0·09 ± 0·06 | 0·00 | 0·92 | 3·16 | 10 | 0·29 ± 0·14 | 0·00 | 1·75 | 2·03 | 22 |
| Terpinene-4-ol | 1·37 ± 0·54 | 0·00 | 9·73 | 1·77 | 65 | 0·67 ± 0·22 | 0·00 | 3·31 | 1·46 | 86 | 1·93 ± 0·28 | 0·39 | 4·04 | 0·61 | 100 |
| α-Terpineol | 0·62 ± 0·29 | 0·00 | 6·01 | 2·10 | 80 | 0·10 ± 0·05 | 0·00 | 0·72 | 2·03 | 38 | 0·05 ± 0·04 | 0·00 | 0·52 | 2·94 | 11 |
| Miscellaneous | 4·72 ± 2·18 | 0·00 | 43·88 | 2·06 | 3·90 ± 0·89 | 0·28 | 19·01 | 1·05 | 6·19 ± 1·50 | 0·00 | 23·76 | 1·02 | |||
| 2,3-Benzofuran | 4·72 ± 2·18 | 0·00 | 43·88 | 2·06 | 95 | 3·90 ± 0·89 | 0·28 | 19·01 | 1·05 | 100 | 6·19 ± 1·50 | 0·00 | 23·76 | 1·02 | 94 |
| Unidentified compounds | 40·07 ± 6·47 | 5·95 | 94·76 | 0·72 | 76·61 ± 4·57 | 25·87 | 93·79 | 0·27 | 64·85 ± 4·59 | 23·73 | 86·99 | 0·30 | |||
| Compounds 1–18 | 40·07 ± 6·47 | 5·95 | 94·76 | 0·72 | 100 | 76·61 ± 4·57 | 25·87 | 93·79 | 0·27 | 100 | 64·85 ± 4·59 | 23·73 | 86·99 | 0·30 | 100 |
Within a chemical class, compounds are ordered according to retention time. Min.: minimal relative value; Max.: maximal relative value; CV: coefficient of variance (= standard deviation/mean); %: proportion of individuals emitting a particular compound.
Table 2.
Mean relative values (±s.e.) of odour compounds identified in headspace samples of Anacamptis coriophora
| Italy (Riserva Varriconi), n = 17 | Italy (Ravenna), n = 26 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean ± s.e. | Min. | Max. | CV | % | Mean ± s.e. | Min. | Max. | CV | % | |
| Fatty acid derivatives | 2·58 ± 0·42 | 0·42 | 6·19 | 0·68 | 7·77 ± 1·02 | 2·35 | 21·38 | 0·67 | ||
| Aldehydes | 0·17 ± 0·04 | 0·05 | 0·62 | 0·89 | 0·21 ± 0·04 | 0·06 | 0·52 | 0·57 | ||
| Nonanal | 0·17 ± 0·04 | 0·05 | 0·62 | 0·89 | 100 | 0·21 ± 0·02 | 0·06 | 0·52 | 0·57 | 100 |
| Miscellaneous | 2·41 ± 0·40 | 0·36 | 5·57 | 0·68 | 7·56 ± 1·01 | 2·20 | 21·12 | 0·68 | ||
| 4-Methyl-4-pentanolide | 0·19 ± 0·03 | 0·00 | 0·50 | 0·69 | 94 | 0·23 ± 0·04 | 0·05 | 1·07 | 0·86 | 100 |
| δ-Caprolactone | 1·16 ± 0·20 | 0·12 | 2·65 | 0·70 | 100 | 1·31 ± 0·35 | 0·00 | 8·46 | 1·35 | 96 |
| trans-4-Methyl-5-hexanolide | 1·07 ± 0·23 | 0·07 | 3·17 | 0·90 | 100 | 6·01 ± 0·86 | 2·08 | 19·32 | 0·73 | 100 |
| Benzenoids | 90·47 ± 1·52 | 73·88 | 96·79 | 0·07 | 81·84 ± 1·74 | 60·63 | 91·63 | 0·11 | ||
| Aldehydes | 28·37 ± 1·66 | 17·66 | 40·84 | 0·24 | 27·38 ± 2·54 | 3·83 | 47·85 | 0·47 | ||
| Anisaldehyde | 28·14 ± 1·63 | 17·62 | 40·53 | 0·24 | 100 | 27·28 ± 2·52 | 3·83 | 47·62 | 0·47 | 100 |
| p-Methoxy-cinnamic aldehyde | 0·23 ± 0·05 | 0·00 | 0·69 | 0·90 | 94 | 0·10 ± 0·03 | 0·00 | 0·45 | 1·38 | 50 |
| Alcohols | 1·45 ± 0·26 | 0·06 | 2·97 | 0·73 | 2·20 ± 0·31 | 0·09 | 5·90 | 0·71 | ||
| Anisyl alcohol | 0·50 ± 0·12 | 0·00 | 1·68 | 0·99 | 100 | 0·41 ± 0·08 | 0·06 | 1·71 | 0·94 | 100 |
| p-Methoxy phenylethyl alcohol | 0·95 ± 0·20 | 0·00 | 2·88 | 0·87 | 94 | 1·79 ± 0·26 | 0·00 | 4·19 | 0·73 | 96 |
| Esters | 5·42 ± 0·63 | 1·65 | 11·09 | 0·48 | 18·32 ± 2·65 | 2·28 | 43·68 | 0·74 | ||
| Methyl benzoate | 0·01 ± 0·01 | 0·00 | 0·14 | 4·12 | 6 | 0·18 ± 0·05 | 0·00 | 0·68 | 1·33 | 46 |
| Methyl anisate | 3·38 ± 0·36 | 1·17 | 5·88 | 0·44 | 100 | 14·97 ± 2·47 | 1·66 | 39·57 | 0·84 | 100 |
| Methyl-(E)-cinnamate | 0·65 ± 0·31 | 0·09 | 5·47 | 1·95 | 100 | 1·06 ± 0·21 | 0·00 | 3·15 | 1·00 | 96 |
| Methyl-(Z)-p-methoxycinnamate | 0·66 ± 0·20 | 0·00 | 3·26 | 1·27 | 100 | 1·13 ± 0·14 | 0·14 | 3·44 | 0·64 | 100 |
| Methyl-(E)-p-methoxycinnamate | 0·72 ± 0·14 | 0·13 | 2·06 | 0·79 | 100 | 0·98 ± 0·12 | 0·09 | 2·96 | 0·63 | 100 |
| Ethers | 55·23 ± 2·70 | 38·05 | 74·04 | 0·20 | 33·93 ± 2·26 | 17·53 | 59·29 | 0·34 | ||
| p-Methyl anisole | 0·36 ± 0·08 | 0·08 | 1·23 | 0·92 | 100 | 1·64 ± 0·24 | 0·16 | 4·51 | 0·75 | 100 |
| Hydroquinone dimethyl ether | 54·87 ± 2·69 | 37·77 | 73·86 | 0·20 | 100 | 32·29 ± 2·27 | 16·01 | 56·29 | 0·36 | 100 |
| Unidentified compounds | 6·95 ± 1·18 | 2·47 | 19·93 | 0·70 | 10·39 ± 0·91 | 4·87 | 22·95 | 0·45 | ||
| Compounds 1–39 | 6·95 ± 1·18 | 2·47 | 19·93 | 0·70 | 100 | 10·39 ± 0·91 | 4·87 | 22·95 | 0·45 | 100 |
Within a chemical class, compounds are ordered according to retention time. Min.: minimal relative value; Max.: maximal relative value; CV: coefficient of variance (= standard deviation/mean); %: proportion of individuals emitting a particular compound.
Fig. 1.
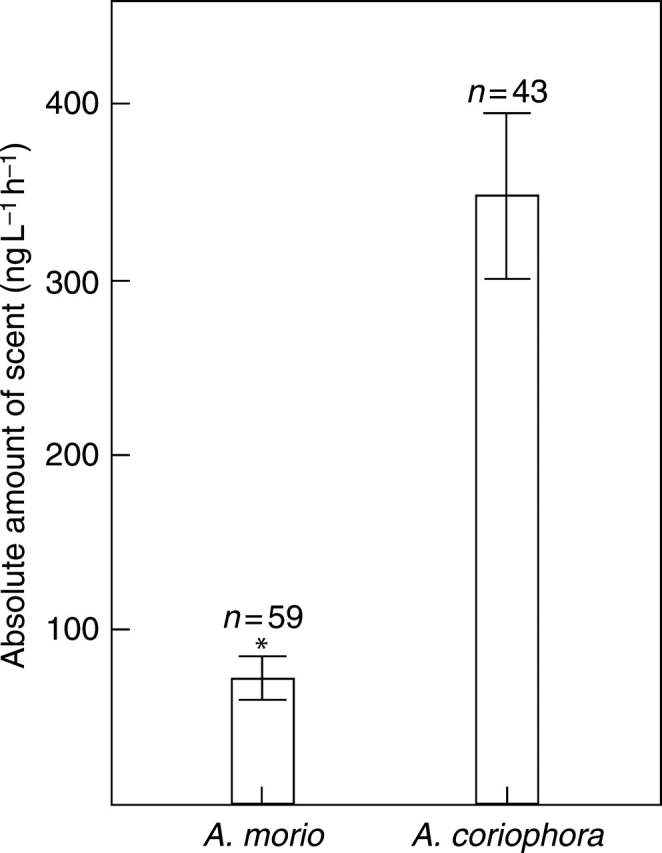
Absolute amount of scent emitted by inflorescences of Anacamptis morio and A. coriophora Mann–Whitney U-test, *P < 0·001. Bars show mean ± s.e.
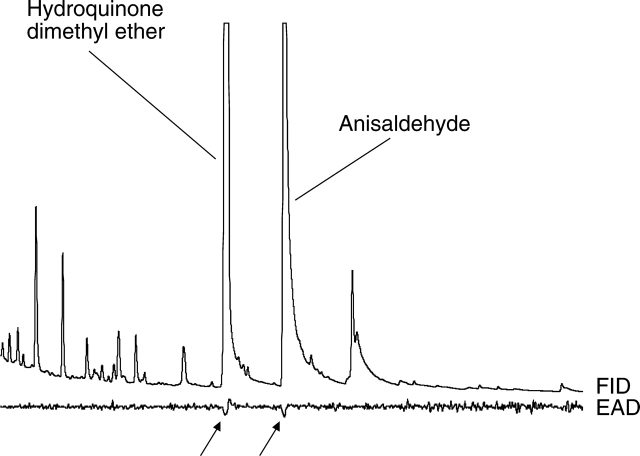
GC-EAD analyses showed that the two main scent compounds in A. coriophora, hydroquinone dimethyl ether (1,4-dimethoxybenzene) and anisaldehyde (methoxybenzaldehyde), elicited a physiological response in the antennae of the pollinator species Bombus terrestris and Apis mellifera (Fig. 2; data only shown for B. terrestris). In the A. morio samples, however, no compound elicited a physiological response in the pollinators' antennae.
Fig. 2.
Gas chromatographic analysis and electroantennographic detection (GC-EAD) of ten pooled headspace samples of Anacamptis coriophora using an antenna of a Bombus terrestris queen. Two compounds, hydroquinone dimethyl ether and anisaldehyde, showed a consistent physiological response (arrows) in antennae of Bombus terrestris and Apis mellifera. FID: flame ionization detector trace from the gas chromatograph; EAD: electroantennographic detection trace from the antenna.
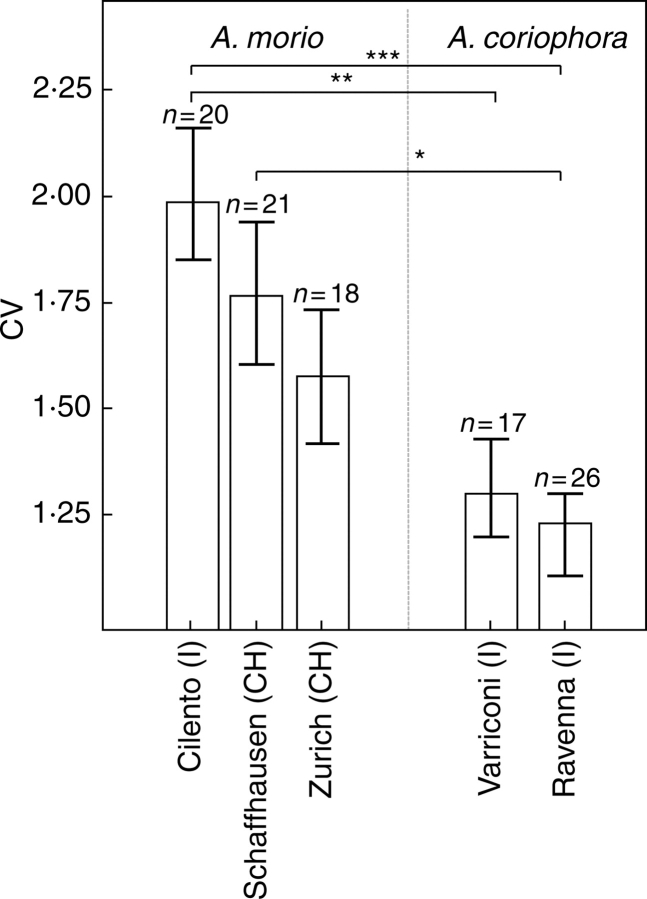
Comparing the mean CV of the odour compounds between all populations sampled, no significant differences were found between populations within a species (Fig. 3). However, populations of the food-deceptive A. morio were overall more variable than in the food-rewarding A. coriophora. Most pronounced were the differences between the Italian populations of A. morio ‘Cilento’ – A. coriophora ‘Varriconi’ and A. morio ‘Cilento’ – A. coriophora ‘Ravenna’ (Mann–Whitney U-test, U = 474, P = 0·001 and U = 374 and P < 0·001, respectively). Within A. coriophora the mean CV of the physiologically active odour compounds was significantly lower than the CV of the non-active compounds (Mann–Whitney U-test, U = 2, P < 0·001; Fig. 4).
Fig. 3.
Comparison of the coefficient of variance (CV = standard deviation/mean) between populations of Anacamptis morio (‘Cilento’, ‘Schaffhausen’, ‘Zurich’) and A. coriophora (‘Varriconi’, ‘Ravenna’). Mann–Whitney U-test and subsequent Bonferroni correction, ***P < 0·001, **P < 0·01, *P < 0·05. Bars show mean ± s.e.
Fig. 4.
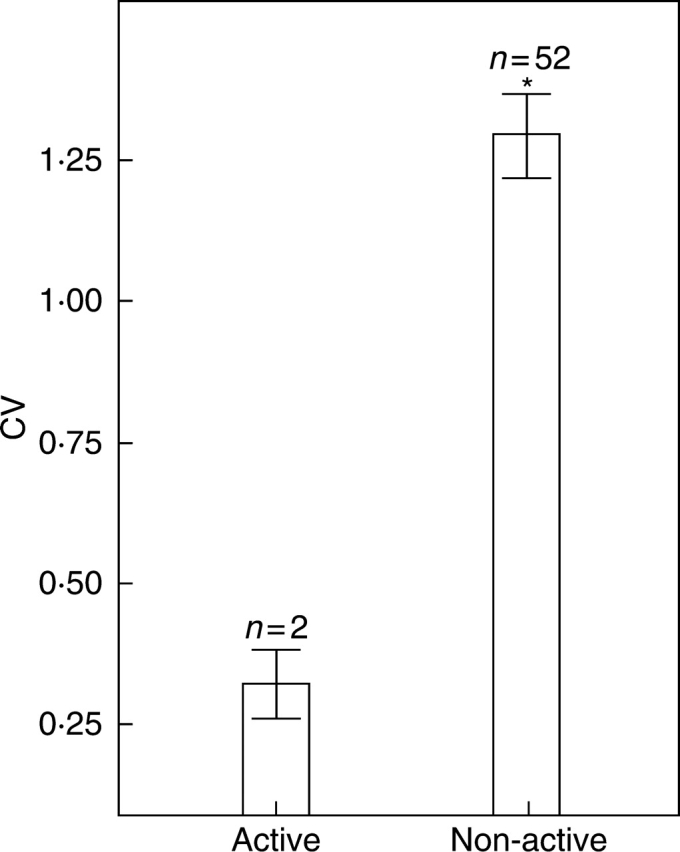
Comparison of the coefficient of variance (CV = standard deviation/mean) between the physiologically active and non-active compounds in Anacamptis coriophora. Mann–Whitney U-test, *P < 0·001). Bars show mean ± s.e.
Scent variability and pollination success in A. morio
Analyses of variance did not reveal a significant effect of odour treatment on pollination success of A. morio, nor did the plot or number of flowers (Table 3). When the data were pooled over the whole experiment, only a trend that odour-treated plants were less pollinated than untreated plants was found (Fig. 5; ANOVA, F1,44 = 1·307, P = 0·259).
Table 3.
Factors that explain variation in pollination success in Anacamptis morio. Data were analysed using an ANOVA with treatment and plot as fixed factors and number of flowers as covariate.
| Source | d.f. | SS* | MS | F | P |
|---|---|---|---|---|---|
| Dependent variable: percentage pollination success | |||||
| Corrected model | 6 | 0·064† | 0·011 | 1·137 | 0·360 |
| Treatment | 1 | 0·018 | 0·018 | 1·876 | 0·179 |
| Plot | 2 | 0·031 | 0·016 | 1·666 | 0·202 |
| Plot × treatment | 2 | 0·007 | 0·003 | 0·349 | 0·707 |
| Number of flowers | 1 | 0·009 | 0·009 | 0·911 | 0·346 |
* Type III SS.
† Adjusted R2 = 0·018.
Fig. 5.
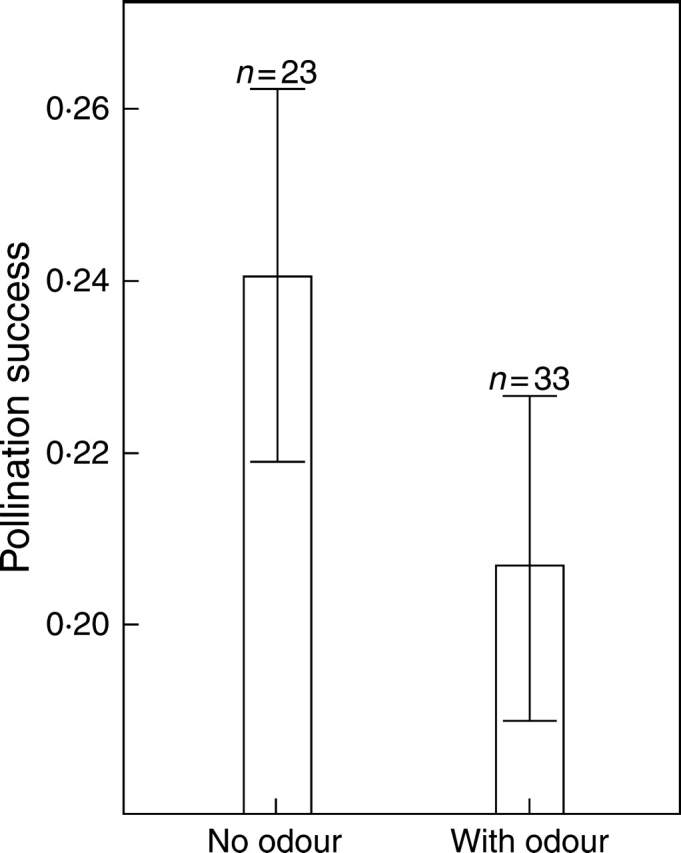
Difference in pollination success (= sum of flowers pollinated or pollinia removed/total amount of flowers) between untreated and odour-treated plants across all plots (ANOVA, F1,44 = 1·307, P = 0·259). Bars show mean ± s.e.
DISCUSSION
Variation in floral traits is the basis for evolutionary change in floral characters. Consequently, a large number of studies have investigated and documented such variation, but little is still known about the selective forces, or random genetic and environmental factors, that shape patterns of variation. In the present comparative study it was found that the rewarding A. coriophora varies significantly less in floral odour over two populations than the deceptive A. morio. This pattern is consistent with the hypothesis of stabilizing selection imposed by pollinator learning and floral constancy.
Although floral odour is generally acknowledged as highly relevant for plant reproduction, the chemical composition and ecological functions of floral odour are unknown in many plant species, which is largely due to the long-term bias of pollination ecology towards floral colour (Fægri and van der Pijl, 1979; Dobson, 2006). One of the reasons for this is its often high chemical complexity that serves multiple functions, ranging from pollinator attraction to chemical defence, and may also comprise ‘biochemical noise’ introduced by the often low specificity of enzymes producing floral odour compounds (Raguso, 2003). The present investigations have confirmed this overall complexity in two Anacamptis species, although most of the identified compounds herein are commonly found in other bee-pollinated plants (Knudsen et al., 1993; Hansted et al., 1994; Olesen and Knudsen, 1994; Gaskett et al., 2005; Wright et al., 2005). Nevertheless, the two orchid species were clearly separated by their scent profiles. A monoterpene blend dominated the scent of the Italian A. morio population, confirming earlier studies by Nilsson (1984) in Swedish A. morio plants. The scent of A. coriophora, by contrast, consisted mainly of benzenoids, which are structurally less diverse and hence occur in fewer numbers among bee-pollinated plants (Dobson, 2006). One means by which to break down the complexity of floral odour, and to identify compounds involved in pollinator attraction, is the application of EAD. This powerful method analyses all compounds in the bouquet for their potential to elicit responses in olfactory neurons of pollinators and has been highly successful for identifying pollinator-attracting compounds in rewarding (Plepys et al., 2002; Huber et al., 2005; Dötterl et al., 2006) and deceptive pollination systems (Schiestl et al., 1999, 2003). By using this method, the two main benzenoid compounds emitted by A. coriophora, hydroquinone dimethyl ether and anisaldehyde, were demonstrated to be EAD-active in honey-bees and bumble-bees. Hydroquinone dimethyl ether was found in the flowers of two Salix species, and was shown to be EAD-active as well as attractive in field bioassays for the pollinator bee (Dötterl et al., 2005). This compound may thus represent a general bee attractant, although the attractiveness of hydroquinone dimethyl ether and anisaldehyde was not specifically investigated in the present study.
Floral scent can influence pollinator behaviour in multiple ways. Beside the mere attraction of pollinators, it has been shown that scent increases the attention of pollinators towards visual stimuli and stimulates their landing or feeding behaviour (Nilsson, 1983; Lunau, 1992; Dobson et al., 1999; Andersson and Dobson, 2003; Raguso and Willis, 2005). Floral scent can also mediate preferential visitation and flower constancy. Bumble-bees preferentially visited sweet smelling morphs over skunky smelling morphs in Polemonium, irrespective of their flower colour (Galen and Kevan, 1983), and in an experiment with artificial flowers, honey-bees became flower constant to either scent or colour when given the choice (Wells and Wells, 1985). When floral scent is paired with other floral traits, it may increase flower constancy, as bumble-bees become more constant when flowers differ in more than one sensory signal, e.g. colour and odour (Gegear and Laverty, 2001) or size and odour (Gegear, 2005), than when they differ in one of the traits alone. A strong odour emission, as in A. coriophora, may thus be selectively advantageous in a rewarding plant, as it increases the attraction of pollinators and enables the association of odour and reward. Flower constancy may cause stabilizing selection in the odour bouquet of A. coriophora by promoting the mating of similar-smelling individuals with each other, expressed by a consistent occurrence and lower variability of the two EAD-active compounds compared with the non-active compounds in all individuals. Different variability in active and non-active compounds suggests stabilizing selection on active compounds, probably imposed by pollinators (Mant et al., 2005). Reduced variability in active compounds was also found in the food-rewarding Gymnadenia (Huber et al., 2005) and in sexually deceptive Ophrys species (Ayasse et al., 2000; Mant et al., 2005). In the interspecific comparison, A. coriophora also showed less overall scent variability than the deceptive A. morio, further supporting the stabilizing selection scenario.
As much as floral scent is advantageous for nectar-producing plants to advertise their rewards and thus increase their reproductive fitness, different rules apply for food-deceptive species, where scent may be used by pollinators to learn and avoid the deceptive flowers, and scent may be learned more quickly than colour (Kunze and Gumbert, 2001; Galizia et al., 2005). The current study suggests that the amounts of compounds emitted by A. morio may be too weak to be consistently detected by the pollinators. No compound in the fragrance of A. morio elicited a response in bumble-bee or honey-bee antennae. This is probably due to the low concentration of the odour compounds emitted from the flowers, as some of them, such as myrcene, limonene and linalool, are known to be principally detected by honey-bees and bumble-bees (Henning et al., 1992; Henning and Teuber, 1992; Laloi et al., 1999). Flowers with scent emission below the detection level of the olfactory neurons in the antennae – de facto scentless for pollinators – may be adaptive in this pollination system. An experiment performed by Kunze and Gumbert (2001) with artificial flowers of a rewarding model and a differently coloured non-rewarding mimic showed that bumble-bees discriminated better between scented than between non-scented model and mimic. Thus, the mere presence of scent enhanced the discrimination ability of the bumble-bees, suggesting that scentless non-model deceptive flowers should have a selective advantage. The present results support this hypothesis by the low total scent emission in the food-deceptive A. morio, with a lack of detectable EAD responses to any floral odour compound. In future experiments, more sensitive methods, such as electroantennography with single sensillum recording (Schiestl and Marion-Poll, 2002), or behavioural experiments could be used to confirm the lack of detection of A. morio compounds. Alternatively, the low amounts of odour may also be explained with resource-saving by avoidance of the costly odour production.
Scent variability and reproductive success
Although high degrees of variation are common in deceptive orchids (summarized in Schiestl, 2005), this is the first comparative study showing that variation in floral odour is considerably higher in a deceptive orchid as compared with the closely related rewarding species. High degrees of variation in floral signals of deceptive orchids have been explained by evolutionary processes including nFDS (Ackerman et al., 1997; Smithson and Mcnair, 1997b; Gigord et al., 2001) and genetic drift (Ackerman and Zimmerman, 1994; Aragón and Ackerman, 2004). Variation in floral traits in deceptive orchids may be adaptive for the plants, as it may delay the avoidance learning of the pollinators (Heinrich, 1975). The aim of our field experiment was to show whether individuals of the deceptive A. morio in treatment plots, having a strong odour and reduced variation similar to that of A. coriophora, have lower pollination success compared with control plots with natural high odour variability. This hypothesis could not be confirmed, as low variability among plants in the plots had no significant effect on pollination success. Thus, the data suggest that odour variability is non-adaptive in A. morio. This is in accordance with earlier studies by Ackerman et al. (1997), who did not find any evidence for nFDS on the odour of Tolumnia variegata. Similarly, in floral colour, although nFDS was predicted in a study on Dactylorhiza sambucina using experimental plots (Gigord et al., 2001), no evidence for it was found in natural populations of that species (Pellegrino et al., 2005; Jersakova et al., 2006). The present study adds to these findings by suggesting that high degrees of variation are not necessarily adaptive in a food-deceptive system. Alternatively, high variation in floral scent of deceptive orchids may be the outcome of relaxed selection (Ackerman et al., 1997). If the low amounts of scent emitted by A. morio are indeed not detected by the pollinators, this trait would not influence pollinator behaviour and ‘neutral’ variation may accumulate.
Conclusions
The results support the hypothesis that stabilizing selection shapes floral scent variability in the food-rewarding A. coriophora. Evidence for this is the low variability of odour compounds, especially among the main compounds that are also biologically active in the pollinators. Odour compounds in the food-deceptive A. morio, by contrast, were too weak to elicit any EAD responses in the pollinator antennae but were significantly more variable perhaps as a consequence of relaxed selection on this trait.
ACKNOWLEDGEMENTS
We thank Roman Kaiser (Givaudan Schweiz AG, Dübendorf, Switzerland) for identifying floral odour compounds and providing anisaldehyde for experiments and Stefania Impagliazzo, Antonio Croce, Olga De Castro, Giovanni Scopece, Stefano Giorgio and Enzo Avolio (Università degli studi di Napoli ‘Federico II’, Naples, Italy) for helping with fieldwork. We also thank Vesuvio National Park and the Cilento and Vallo di Diano National Park for issuing collection licences. We further thank Adrian Leuchtmann (ETH Zürich) for helpful comments on the manuscript. This study was financially supported by the Federal Institute of Technology Zürich (ETH Research Grant 28/01-4 to F.P.S.) and by the PRIN programme.
LITERATURE CITED
- Ackerman JD. Mechanisms and evolution of food-deceptive pollination systems in orchids. Lindleyana. 1986;1:108–113. [Google Scholar]
- Ackerman JD, Zimmerman JK. Bottlenecks in the life histories of orchids: resources, pollination, population structure, and seedling establishment. In: Pridgeon A, editor. Proceedings of the 14th world orchid conference. London: Her Majesty's Stationary Office; 1994. pp. 125–129. [Google Scholar]
- Ackerman JD, Méléndez-Ackerman EJ, Salguero-Faria J. Variation in pollinator abundance and selection on fragrance phenotypes in an epiphytic orchid. American Journal of Botany. 1997;84:1383–1390. [PubMed] [Google Scholar]
- Andersson S, Dobson HE. Behavioral foraging responses by the butterfly Heliconius melpomene to Lantana camara floral scent. Journal of Chemical Ecology. 2003;29:2303–2318. doi: 10.1023/a:1026226514968. [DOI] [PubMed] [Google Scholar]
- Aragón S, Ackerman JD. Does flower color variation matter in deception pollinated Psychilis monensis (Orchidaceae)? Oecologia. 2004;138:405–413. doi: 10.1007/s00442-003-1443-9. [DOI] [PubMed] [Google Scholar]
- Ayasse M, Schiestl FP, Paulus HF, Löfstedt C, Hansson BS, Ibarra F, Francke W. Evolution of reproductive strategies in the sexually deceptive orchid Ophrys sphegodes: how does flower-specific variation of odor signals influence reproductive success? Evolution. 2000;54:1995–2006. doi: 10.1111/j.0014-3820.2000.tb01243.x. [DOI] [PubMed] [Google Scholar]
- Bateman RM, Hollingsworth PM, Preston J, Yi-Bo L, Pridgeon AM, Chase MW. Molecular phylogenetics and evolution of Orchidinae and selected Habenariinae (Orchidaceae) Botanical Journal of the Linnaean Society. 2003;142:1–40. [Google Scholar]
- Chittka L. Flower constancy, insect psychology, and plant evolution. Naturwissenschaften. 1999;86:361–377. [Google Scholar]
- Cozzolino S, Schiestl FP, Müller A, De Castro O, Nardella AM, Widmer A. Evidence for pollinator sharing in Mediterranean nectar-mimic orchids: absence of premating barriers? Proceedings of the Royal Society B-Biological Sciences. 2005;272:1271–1278. doi: 10.1098/rspb.2005.3069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dafni A. Floral mimicry – mutualism and unidirectional exploitation of insects by plants. In: Juniper B, Sothwood R, editors. Insects and the plant surface. London: Edward Arnold; 1986. pp. 81–90. [Google Scholar]
- Dafni A, Ivri Y. Pollination ecology of, and hybridization between, Orchis coriophora L. and O. collina Sol. ex Russ. (Orchidaceae) in Israel. New Phytologist. 1979;83:181–187. [Google Scholar]
- Dafni A, Ivri Y. The flower biology of Cephalanthera longifolia (Orchidaceae) – pollen imitation and facultative floral mimicry. Plant Systematics and Evolution. 1981;137:229–240. [Google Scholar]
- Dobson HEM. Relationship between floral fragrance composition and type of pollinator. In: Dudareva N, Pichersky E, editors. Biology of floral scent. Boca Raton, FL: Taylor and Francis; 2006. pp. 147–198. [Google Scholar]
- Dobson HEM, Danielson EM, Van Wesep ID. Pollen odor chemicals as modulators of bumble bee foraging on Rosa rugosa Thunb. (Rosaceae) Plant Species Biology. 1999;14:153–166. [Google Scholar]
- Dötterl S, Füssel U, Jürgens A, Aas G. 1,4-Dimethoxybenzene, a floral scent compound in willows that attracts an oligolectic bee. Journal of Chemical Ecology. 2005;31:2993–2998. doi: 10.1007/s10886-005-9152-y. [DOI] [PubMed] [Google Scholar]
- Dötterl S, Jürgens A, Seifert K, Laube T, Weißbecker B, Schütz S. Nursery pollination by a moth in Silene latifolia: the role of odours in eliciting antennal and behavioural responses. New Phytologist. 2006;169:707–718. doi: 10.1111/j.1469-8137.2005.01509.x. [DOI] [PubMed] [Google Scholar]
- Fægri K, van der Pijl L. The principles of pollination ecology. Oxford: Pergamon Press; 1979. [Google Scholar]
- Ferdy JB, Gouyon PH, Moret J, Godelle B. Pollinator behavior and deceptive pollination: learning process and floral evolution. American Naturalist. 1998;152:696–705. doi: 10.1086/286200. [DOI] [PubMed] [Google Scholar]
- Galen C, Kevan PG. Bumblebee foraging and floral scent dimorphism: Bombus kirbyellus Curtis (Hymenoptera: Apidae) and Polemonium viscosum Nutt. (Polemoniaceae) Canadian Journal of Zoology. 1983;61:1207–1213. [Google Scholar]
- Galizia GC, Kunze J, Gumbert A, Borg-Karlson A-K, Sachse S, Markl C, Menzel R. Relationship of visual and olfactory signal parameters in a food-deceptive flower mimicry system. Behavioral Ecology. 2005;16:159–168. [Google Scholar]
- Gaskett A, Conti E, Schiestl FP. Floral odor variation in two heterostylous species of. Primula. Journal of Chemical Ecology. 2005;31:1223–1228. doi: 10.1007/s10886-005-5351-9. [DOI] [PubMed] [Google Scholar]
- Gegear RJ. Multicomponent floral signals elicit selective foraging in bumblebees. Naturwissenschaften. 2005;92:269–271. doi: 10.1007/s00114-005-0621-5. [DOI] [PubMed] [Google Scholar]
- Gegear RJ, Laverty TM. The effect of variation among floral traits on the flower constancy of pollinators. In: Chittka L, Thomson JD, editors. Cognitive ecology of pollination. Animal behavior and floral evolution. Cambridge: Cambridge University Press; 2001. pp. 1–20. [Google Scholar]
- Gigord LDB, Macnair MR, Smithson A. Negative frequency-dependent selection maintains a dramatic flower color polymorphism in the rewardless orchid Dactylorhiza sambucina (L.) Soò. Proceedings of the National Academy of Science USA. 2001;98:6253–6255. doi: 10.1073/pnas.111162598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goulson D. Foraging strategies of insects for gathering nectar and pollen, and implications for plant ecology and evolution. Perspectives in Plant Ecology Evolution and Systematics. 1999;2:185–209. [Google Scholar]
- Gumbert A. Color choices by bumble bees (Bombus terrestris): innate preferences and generalization after learning. Behavioral Ecology and Sociobiology. 2000;48:36–43. [Google Scholar]
- Hansted L, Jakobsen HB, Olsen CE. Influence of temperature on the rhythmic emission of volatiles from Ribes nigrum flowers in situ. Plant Cell and Environment. 1994;17:1069–1072. [Google Scholar]
- Heinrich B. Bee flowers: a hypothesis on flower variety and blooming times. Evolution. 1975;29:325–334. doi: 10.1111/j.1558-5646.1975.tb00212.x. [DOI] [PubMed] [Google Scholar]
- Heinrich B, Mudge PR, Deringis PG. Laboratory analysis of flower constancy in foraging bumblebees. Bombus ternarius and B. terricola. Behaviour Ecology Sociobiology. 1977;2:247–265. [Google Scholar]
- Henning JA, Teuber LR. Combined gas chromatography-electroantennogram characterization of alfalfa floral volatiles recognized by honey bees (Hymenoptera, Apidae) Journal of Economic Entomology. 1992;85:226–232. [Google Scholar]
- Henning JA, Peng YS, Montague MA, Teuber LR. Honey bee (Hymenoptera: Apidae) behavioral response to primary alfalfa (Rosales, Fabaceae) floral volatiles. Journal of Economic Entomology. 1992;85:233–239. [Google Scholar]
- Huber FK, Kaiser R, Sauter W, Schiestl FP. Floral scent emission and pollinator attraction in two species of Gymnadenia (Orchidaceae) Oecologia. 2005;142:564–575. doi: 10.1007/s00442-004-1750-9. [DOI] [PubMed] [Google Scholar]
- Jersakova J, Kindlmann P, Renner SS. Is the colour dimorphism in Dactylorhiza sambucina maintained by differential seed viability instead of frequency-dependent selection? Folia Geobotanica. 2006;41:61–76. [Google Scholar]
- Johnson SD. Batesian mimicry in the non-rewarding orchid Disa pulchra and its consequences for pollinator behavior. Biological Journal of the Linnaean Society. 2000;71:119–132. [Google Scholar]
- Jones KN. Pollinator-mediated assortative mating: causes and consequences. In: Chittka L, Thomson JD, editors. Cognitive ecology of pollination. animal behavior and floral evolution. Cambridge: Cambridge University Press; 2001. pp. 259–273. [Google Scholar]
- Knudsen JT, Tollsten L, Bergström GL. Floral scents – a checklist of volatile compounds isolated by head-space techniques. Phytochemistry. 1993;33:253–280. [Google Scholar]
- Kugler H. Blütenökologische Untersuchungen mit Hummeln VII. Die Anlockung von “Neulingen” durch Blüten. Planta. 1934;23:692–714. [Google Scholar]
- Kunze J, Gumbert A. The combined effect of color and odor on flower choice behavior of bumble bees in flower mimicry systems. Behaviour Ecology. 2001;12:447–456. [Google Scholar]
- Laloi D, Sondoz JC, Picard-Nizou AL, Marchesi A, Pouvreau A, Taséi JN, et al. Olfactory conditioning of the proboscis extension in bumble bees. Entomologia Experimentalis et Applicata. 1999;90:123–129. [Google Scholar]
- Lunau K. Innate recognition of flowers by bumble bees: orientation of antennae to visual stamen signals. Canadian Journal of Zoology. 1992;70:2139–2144. [Google Scholar]
- Manning A. Some aspects of the foraging behaviour of bumble-bees. Behaviour. 1956;9:164–201. [Google Scholar]
- Mant J, Peakall R, Schiestl FP. Does selection on floral odor promote differentiation among populations and species of the sexually deceptive orchid genus Ophrys? Evolution. 2005;59:1449–1463. [PubMed] [Google Scholar]
- Moya S, Ackerman JD. Variation in the floral fragrance of Epidendrum ciliare (Orchidaceae) Nordic Journal of Botany. 1993;13:41–47. [Google Scholar]
- Nilsson LA. The pollination ecology of Dactylorhiza sambucina (Orchidaceae) Botaniska Notizer. 1980;133:367–385. [Google Scholar]
- Nilsson LA. Processes of isolation and introgressive interplay between Platanthera bifolia (L.) Rich and Platanthera chlorantha (Custer) Reichb. (Orchidaceae) Botanical Journal of the Linnaean Society. 1983;87:325–350. [Google Scholar]
- Nilsson LA. Anthecology of Orchis morio (Orchidaceae) at its outpost in the north. Nova Acta Regiae Societatis Scientarum Upsaliensis. 1984;V:166–179. [Google Scholar]
- Nilsson LA. Orchid pollination biology. Trends in Ecology and Evolution. 1992;7:255–259. doi: 10.1016/0169-5347(92)90170-G. [DOI] [PubMed] [Google Scholar]
- Olesen JM, Knudsen JT. Scent profiles of flower color morphs of Corydalis cava (Fumariaceae) in relation to foraging behavior of bumblebee queens (Bombus terrestris) Biochemical Systematics and Ecology. 1994;22:231–237. [Google Scholar]
- Pellegrino G, Caimi D, Noce ME, Musacchio A. Effects of local density and flower colour polymorphism on pollination and reproduction in the rewardless orchid Dactylorhiza sambucina (L.) Soò. Plant Systematics and Evolution. 2005;251:119–129. [Google Scholar]
- Plepys D, Ibarra F, Francke W, Lofstedt C. Odour-mediated nectar foraging in the Silver Y Moth, Autographa gamma (Lepidoptera: Noctuidae): behavioural and electrophysiological responses to floral volatiles. Oikos. 2002;99:75–82. [Google Scholar]
- Raguso RA. Olfactory landscapes and deceptive pollination: signal, noise and convergent evolution in floral scent. In: Blomquist GJ, editor. Insect pheromone biochemistry and molecular biology: the biosynthesis and detection of pheromones and plant volatiles. Amsterdam: Elsevier Academic Press; 2003. pp. 631–650. [Google Scholar]
- Raguso RA, Willis MA. Synergy between visual and olfactory cues in nectar feeding by wild hawkmoths, Manduca sexta. Animal Behaviour. 2005;69:407–418. [Google Scholar]
- Real LA. Uncertainty and pollinator–plant interactions: the foraging behavior of bees and wasps on artificial flowers. Ecology. 1981;62:20–26. [Google Scholar]
- Roy BA, Widmer A. Floral mimicry: a fascinating yet poorly understood phenomenon. Trends in Ecology and Evolution. 1999;4:325–330. doi: 10.1016/s1360-1385(99)01445-4. [DOI] [PubMed] [Google Scholar]
- Schiestl FP. On the success of a swindle: pollination by deception in orchids. Naturwissenschaften. 2005;92:255–264. doi: 10.1007/s00114-005-0636-y. [DOI] [PubMed] [Google Scholar]
- Schiestl FP, Marion-Poll F. Detection of physiologically active flower volatiles using gas chromatography coupled with electroantennography. In: Jackson JF, Linskens HF, editors. Analyses of taste and aroma. Berlin: Springer; 2002. pp. 173–198. [Google Scholar]
- Schiestl FP, Ayasse M, Paulus HF, Löfstedt C, Hansson BS, Ibarra F, Francke W. Orchid pollination by sexual swindle. Nature. 1999;399:421–422. [Google Scholar]
- Schiestl FP, Peakall R, Mant J, Ibarra F, Schulz C, Francke S, Francke W. The chemistry of sexual deception in an orchid-wasp pollination system. Science. 2003;302:437–438. doi: 10.1126/science.1087835. [DOI] [PubMed] [Google Scholar]
- Schomburg G. Gas chromatography. A practical course. Weinheim: VCH; 1990. [Google Scholar]
- Smithson A, Macnair MR. Frequency-dependent selection by pollinators: mechanisms and consequences with regard to behaviour of bumblebees Bombus terrestris (L.) (Hymenoptera: Apidae) Journal of Evolutionary Biology. 1996;9:571–588. [Google Scholar]
- Smithson A, Macnair M. Density-dependent and frequency-dependent selection by bumblebees Bombus terrestris (L.) (Hymenoptera: Apidae) Biological Journal of the Linnaean Society. 1997;60:401–417. [Google Scholar]
- Smithson A, Macnair M. Negative frequency-dependent selection by pollinators on artificial flowers without rewards. Evolution. 1997;51:715–723. doi: 10.1111/j.1558-5646.1997.tb03655.x. [DOI] [PubMed] [Google Scholar]
- Tholl D, Röse USR. Detection and identification of floral scent compounds. In: Dudareva N, Pichersky E, editors. Biology of floral scent. Boca Raton, FL: Taylor and Francis; 2006. pp. 3–26. [Google Scholar]
- Van der Cingel NA. An atlas of orchid pollination: European orchids. Rotterdam: Balkema; 1995. [Google Scholar]
- Waser NM. Flower constancy: definition, cause, and measurement. American Naturalist. 1986;127:593–603. [Google Scholar]
- Wells PH, Wells H. Ethological isolation of plants 2. Odour selection by honeybees. Journal of Apicultural Research. 1985;24:86–92. [Google Scholar]
- Wright GA, Lutmerding A, Dudareva N, Smith BH. Intensity and the ratios of compounds in the scent of snapdragon flowers affect scent discrimination by honeybees (Apis mellifera) Journal of Comparative Physiology A – Sensory Neural and Behavioral Physiology. 2005;191:105–114. doi: 10.1007/s00359-004-0576-6. [DOI] [PubMed] [Google Scholar]