Abstract
Sirtuins are a conserved family of proteins found in all domains of life. The first known sirtuin, Sir2 (silent information regulator 2) of Saccharomyces cerevisiae, from which the family derives its name, regulates ribosomal DNA recombination, gene silencing, DNA repair, chromosomal stability and longevity. Sir2 homologues also modulate lifespan in worms and flies, and may underlie the beneficial effects of caloric restriction, the only regimen that slows aging and extends lifespan of most classes of organism, including mammals. Sirtuins have gained considerable attention for their impact on mammalian physiology, since they may provide novel targets for treating diseases associated with aging and perhaps extend human lifespan. In this review we describe our current understanding of the biological function of the seven mammalian sirtuins, SIRT1–7, and we will also discuss their potential as mediators of caloric restriction and as pharmacological targets to delay and treat human age-related diseases.
Keywords: ADP-ribosyl transferase activity, caloric restriction, deacetylase activity, resveratrol, SIR2 (silent information regulator 2), sirtuin (SIRT1–7)
DISCOVERY OF THE SIRTUINS
It is a little known fact that the first sirtuin gene, SIR2 from Saccharomyces cerevisiae, was originally known as MAR1 (for mating-type regulator 1). Klar and collegues [1] discovered MAR1 by virtue of a spontaneous mutation that caused sterility by relieving silencing at the mating-type loci HMR and HML. A variety of additional mutations with a sterile phenotype were co-discovered by Jasper Rine, who named the set of four genes responsible SIR (silent information regulator)1–4 [2-4], thereby replacing the MAR nomenclature.
Gottlieb and Esposito [5] demonstrated, 10 years after this initial finding, that SIR2 is the only SIR gene required to suppress recombination between the 100–200 copies of the ribosomal RNA genes repeated in tandem on chromosome XII. By 1991, thanks primarily to work by Gottschling and colleagues [6], SIR2 was also known to be part of the mechanism that silences genes near telomeres. About 2 years later, Braunstein et al. showed that silent regions at telomeres and mating-type loci are associated with histones that are relatively hypoacetylated at the ε-amino group of N-terminal lysine residues [7]. SIR2 overexpression caused substantial histone deacetylation, an additional characteristic that distinguished SIR2 from the other SIR genes.
In 1995, Brachman et al. [8] and Derbyshire et al. [9] discovered four additional S. cerevisiae genes with high homology to SIR2, HST (homologues of SIR2)1–4. None of the four genes were essential, but they all were involved in silencing at the mating-type loci and telomeres, as well as cell-cycle progression and genomic integrity. The finding of SIR2 homologues in yeast and shortly thereafter in organisms ranging from bacteria to plants and mammals, demonstrated SIR2 is a member of a large and ancient family of genes we now refer to as ‘sirtuins’.
SIRTUINS ARE NAD+-DEPENDENT DEACETYLASES AND MONO-ADP-RIBOSYL TRANSFERASES
Over the next 2 years a variety of interesting features about Sir2 were discovered: it localizes to the nucleolus [10], it is a component of the Ku-associated apparatus that repairs double-stranded DNA breaks [11], it localizes to DNA breaks in a check-point-dependent manner [12-14] and it silences marker genes and alters chromatin structure at the rDNA (ribosomal DNA) locus [15,16]. The first insight into Sir2 enzymatic activity came from the characterization of CobB, a Salmonella typhimurium protein with nicotinate mononucleotide:5,6-dimethylbenzimidazole phosphoribosyl transferase activity. CobB contains a conserved diagnostic feature of the sirtuins: the amino acid sequences GAGISAESGIRTFR and YTQNID (conserved residues are underlined) [17]. By 1999, Roy Frye had identified five of the human SIR2 homologues, SIRT1–5, given the name ‘sirtuins’, and found that SIRT2 could act as an ADP-ribosyl transferase using, as a donor, one of the major nicotinamide nucleotides, NAD+. The transfer of an ADP-ribose from radioactively labelled NAD+ to BSA, and the loss of catalysis when a highly conserved amino acid in the Sir2 homology domain (H171Y) was mutated, were both good indicators that this family of proteins might act as ADP-ribosyl transferases [18]. Only a few months later, Moazed and colleagues [19] found that the yeast Sir2 had the ability to covalently modify a mixture of histones and itself using NAD+ as a donor, via the transfer of ADP-ribose to acceptor amino acids. Accordingly, the analogous mutation in yeast, H364Y, abolished silencing at the mating-type locus, telomeric DNA and rDNA locus. In light of these findings, researchers suspected that ADP-ribosylation of histones by Sir2 interfered with histone acetylation, which explained the higher acetylation and loss of silencing in sir2 mutants.
A few months later, however, two groups showed that Sir2 has histone deacetylase activity that is absolutely dependent on NAD+ [20,21]. Acetylated lysine residues at positions 9 and 14 of histone H3 and specifically Lys16 of histone H4 were found to be in vivo targets, and the H364Y mutation abolished SIR2 deacetylation. Furthermore, a G270A mutant that was defective in ADP-ribosyl tranferase activity, but retained 80 % of its deacetylase activity, proficiently silenced and suppressed rDNA recombination. Thus, Imai et al. [21] concluded that deacetylation rather than mono-ADP-ribosylation was the means by which Sir2 regulated processes in vivo. NAD+-dependent deacetylase activity in vitro has also been described for numerous other sirtuins, including bacterial CobB, archeabacterial SIR2-AF (Archaeoglobus fulgidus) and human SIRT1–3 and 5 [22].
By the end of 2000, Landry et al. [23] had described the catalytic mechanism of deacetylation: they found it to be tightly coupled to the generation of a unique acetyl-ADP-ribose metabolite, OAADPr (2′-O-acetyl-ADP-ribose). Hydrolysis of one NAD+ molecule produced one molecule of nicotinamide, and one of OAADPr (Figure 1) [24]. The subsequent X-ray crystallographic structures of various sirtuins [including archaeal Sir2Af1 and Sir2Af2, bacterial cobB and Sir2Tm (Thermotoga maritime), human SIRT2 and yeast Hst2; as well as sirtuin core domains in complex with NAD+, ADP-ribose, OAADPr or p53 acetyl lysine peptide] have helped us to understand the sirtuin deacetylation mechanism in greater detail (reviewed in [25,26]).
Figure 1. Sirtuin enzymatic activities.
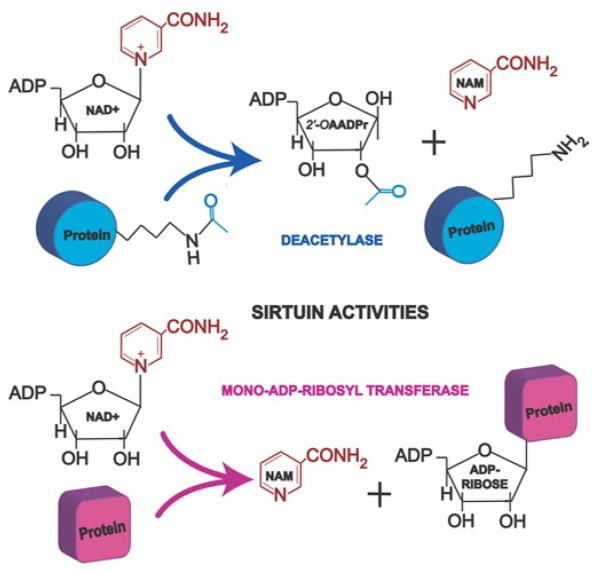
Sirtuins are NAD+-dependent deacetylases and mono-ADP-ribosyl transferases that regulate a wide array of proteins involved in metabolism and cell survival. The ε-acetyl lysine residues of the target protein serve as substrates for sirtuin deacetylation, which generate 2′-OAADPr as a by-product. NAM, nicotinamide.
The ADP-ribosyl transferase activity of sirtuins was considered a low efficiency side-reaction due to the partial uncoupling of intrinsic deacetylation and acetate transfer to ADP-ribose [24]. Recently, however, mono-ADP-ribosyl transferase activity has been found to be the main enzymatic activity of at least two of the mammalian sirtuins, SIRT4 and SIRT6 (Figure 1), as discussed below.
In the last 6 years, close to 500 publications have described different aspects of sirtuin biology, ranging from small molecule agonists to finding molecular targets. Functions for almost all of the mammalian sirtuins have now been ascribed, although many more probably await discovery. The requirement for NAD+ as a co-substrate suggested that the sirtuins might have evolved as sensors of cellular energy and redox states coupled to the metabolic status of the cell. The generation of OAADPr, a unique by-product of the NAD+ dependent deacetylation reaction, raised this molecule as a candidate for sirtuin-triggered signalling pathways, another prediction that has been proven experimentally.
SIRTUINS AND AGING IN NON-MAMMALIAN SPECIES
In 1997, Sinclair and Guarente [27] showed that one of the major causes of aging in S. cerevisiae is the exponential accumulation of ERCs (extrachromosomal rDNA circles). ERCs result from homologous recombination between rDNA repeats, and the introduction of a single ERC into a young cell is sufficient to cause premature aging. In addition, mutation of the SGS1 gene, a homologue of the premature aging disease gene, WRN (Werner syndrome), led to increased rDNA instability and decreased longevity [28]. Consequently, Kaeberlein et al. [29] tested whether extra copies of SIR2 could suppress rDNA recombination and hence extend lifespan. In 1999, they showed that the integration of an extra copy of the SIR2 gene increased the replicative lifespan up to 30 % and that deletion of SIR2, which accelerates ERC formation, shortens lifespan. Interestingly, the replication fork gene (FOB1), which reduces rDNA recombination and ERC formation, largely suppressed the short lifespan of a sir2 mutant. Today, some laboratories study sir2 mutants in the context of a fob1 deletion so that ERCs do not overwhelm the cells before they can be analysed for SIR2-independent longevity pathways.
Metazoans also live longer with extra copies or expression of SIR2, but the mechanism through which SIR2 homologues retard aging remains unknown. Caenorhabditis elegans carrying a chromosome duplication containing sir-2.1, the worm sirtuin most similar to yeast SIR2, exhibits up to a 50 % lifespan increase, and this is strictly dependent on the forkhead transcriptional factor DAF-16, the downstream target of the insulin/IGF (insulin-like growth factor)-1 signalling pathway [30]. Recently, SIR-2.1 was shown to associate with 14-3-3 proteins, which direct its interaction with DAF-16 in an insulin/IGF-1-independent manner under stress conditions [31]. Whether or not this interaction occurs in mammals is unknown, but, given the conservation of the sirtuin, 14-3-3 and Daf16 (abnormal dauer formation 16)/FOXO gene families, it seems very likely. In Drosophila melanogaster, an extra copy of the Sir2 gene (dSir2) expressed ubiquitously causes females and males to live longer by 29 % and 18 % respectively. Likewise, overexpressing dSir2 in the nervous system produced a considerable lifespan extension in both fly genders [32]. Expressing Sir2 specifically in motor neurons did not extend lifespan, however.
THE MAMMALIAN SIRTUINS: SIRT1–7
Although we do not yet know whether SIRT1 or the other mammalian sirtuins regulate health and longevity, knowing the variety of functions of these proteins will certainly give important insights into which biological functions and systems they may influence, and point towards which diseases they may be useful in treating. The mammalian sirtuin family consists of seven members, SIRT1–7. Each sirtuin is characterized by a conserved 275 amino acid catalytic core domain and also by unique additional N-terminal and/or C-terminal sequences of variable length (Figure 2).
Figure 2. Mammalian sirtuins.
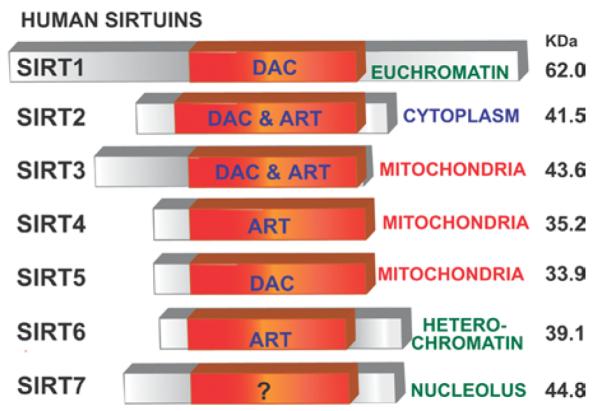
Mammals have seven sirtuins, SIRT1–7. All have an NAD+-dependent catalytic core domain that may act preferentially as a mono-ADP-ribosyl transferase (ART) and/or NAD+-dependent deacetylase (DAC). Additional N-terminal and/or C-terminal sequences of variable length may flank this core domain. The seven sirtuins show different cellular localization. Reprinted from Biochemical and Biophysical Research Communications, 273, Frye, R.A., Phylogenetic classification of prokaryotic and eukaryotic Sir2-like proteins, 793–798, © 2000, with permission from Elsevier.
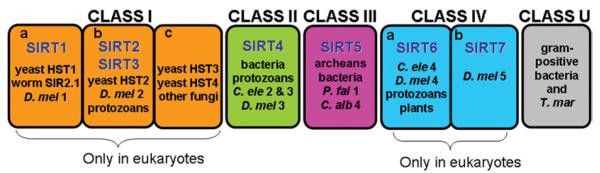
Phylogenetic analysis of 60 core domains from different eukaryotes and prokaryotes places the mammalian sirtuins into four different classes (I—IV). SIRT1, SIRT2 and SIRT3 are known as Class I sirtuins, which groups all yeast sirtuins and also at least one of the Sir2-related proteins in most eukaryotes. Class I is divided in three sub-classes: a, b and c. SIRT1 shares the same Class Ia with Sir2 and Hst1 from S. cerevisiae, C. elegans SIR-2.1 and D. melanogaster D.mel1. SIRT2 and SIRT3 reside in Class Ib, together with yeast Hst2, fly D.mel2 and other fungi and protozoa sirtuins. SIRT4 is part of Class II, which also includes sirtuins from bacteria, insects, nematodes, mould fungus and protozoans. SIRT5 is the mammalian member of Class III sirtuins, distributed widely in all prokaryotes either bacteria or archaea. Finally, Class IV contains SIRT6 and SIRT7 in two different sub-classes IVa and IVb respectively; and unlike Class III, sirtuins of this class are not present in prokaryotes, but are broadly distributed in metazoans, plants and vertebrates. Thus sirtuins from Class II and Class III together with Class U (which groups bacterial Sir2 homologues with undifferentiated motifs that are intermediate between classes II and III, and classes I and IV) seem to be the ones that appeared earliest in evolution (Figure 3) [33].
Figure 3. Classification of mammalian sirtuins.
The seven mammalian sirtuins are classified into four classes according to molecular phylogenetic analysis [33]. Sirtuins from Class II, Class III and Class U (undetermined) seem to have evolved earlier than the other classes. Thus, SIRT4 and SIRT5 may be the most ancient mammalian sirtuins. Class I and Class IV sirtuins, which group SIRT2 and SIRT3 and SIRT6 and SIRT7 respectively, are only present in eukaryotes. C. alb, Candida albicans; C. ele, Caenorhabditis elegans, D.mel, Drosophila melanogaster; P. fal, Plasmodium falciparum; T. mar, Termatoga maritime.
Mammalian sirtuins also differ in their sub-cellular localization. SIRT1, SIRT6 and SIRT7 are predominately in the nucleus (although SIRT1 does have some important cytoplasmic functions as well). In the nucleus a large fraction of SIRT1 is associated with euchromatin, whereas SIRT6 associates with heterochromatin and SIRT7 is found in the nucleolus [34]. The sirtuin that resides most prominently in the cytoplasm is SIRT2 [18,35]. SIRT3, SIRT4 and SIRT5 have been described as mitochondrial sirtuins. In terms of activity, SIRT1 and SIRT5 exhibit robust and weak deacetylase activity respectively [35,36]. SIRT4 and SIRT6 are mono-ADP-ribosyl tranferases [37,38], and both deacetylase and mono-ADP-ribosyl tranferase activities have been detected for SIRT2 and SIRT3 [18,35,39]. No robust activity has been found for SIRT7 as yet (Figure 2).
SIRTUINS MAY UNDERLIE THE BENEFICIAL EFFECTS OF CALORIC RESTRICTION
CR (calorie restriction) is a dietary regimen in which an organism is provided with at least 20 % fewer calories than it would naturally consume ad libitum, while maintaining adequate nutrition [40]. Although it is not correct to say that CR extends the lifespan of every organism tested, it comes close. CR-mediated lifespan extension has been demonstrated in organisms from very different taxa, including yeast (S. cerevisiae) [41], fruit flies (D. melanogaster) [42], nematodes (C. elegans) [43], crustaceans (Daphnia longispina) [44], spiders (Frontinella pyramitela) [45] and rodents [46]. This diversity argues that the mechanisms underlying CR are ancient, relatively simple and well conserved.
CR is the only non-genetic method that consistently increases maximal lifespan in mammals. It does so not by keeping animals in a unhealthy state for a longer time, but by actually retarding age-related deterioration, such as by decreased collagen elasticity, development of insulin resistance, a decline in immune function, neuro-behavioural impairments and by delaying the onset (and incidence) of age-related diseases such as cancer and auto-immune disorders [47]. Although it is unknown whether CR also extends lifespan in primates, ongoing studies in rhesus monkeys (Macaca mulatta) show many benefits from CR, including reduced body weight, lower body fat, decreased blood glucose and lower incidence of diabetes and heart disease. They also show altered hormonal profiles that do not compromise circadian patterns or reproductive timing [48].
A role for sirtuins in mediating CR was first demonstrated in S. cerevisiae. On medium with 0.5 % glucose versus standard 2 % (w/v) glucose, which has been defined as CR for yeast cells [49,50], the replicative lifespan was approx. 30 % longer. Strikingly, this effect is not seen if cells lack the SIR2 gene [32,49,51]. When cells become starved, sirtuin-independent pathways, such as TOR (target of rapamycin), are activated and can also extend lifespan [52]. At least two of the other four yeast sirtuins, Hst1 and Hst2 also participate in the CR response [51], indicating that the entire sirtuin family may control lifespan, perhaps having evolved from a primordial sirtuin that responded to stress and calorie availability. In Drosophila, lifespan extension by dietary restriction also requires the Sir2 gene and overexpression of Sir2 extends lifespan, paralleling the yeast data [32]. These findings have led credence to the hypothesis that sirtuins underlie the beneficial effects of caloric restriction in diverse species, including mammals. Further support for this hypothesis has come from studying the mammalian sirtuins [53], which as we will discuss below, have varied functions that are consistent with the physiological changes observed in rodents and primates on a CR diet.
FUNTIONS OF THE MAMMALIAN SIRTUINS
Chromatin structure and transcription
Similar to yeast Sir2, mammalian SIRT1 facilitates the formation of heterochromatin, the more tightly packed form of chromatin associated with histone hypoacetylation and gene repression. SIRT1 targets for deacetylation include lysine residues at positions 9 and 26 of histone H1, 14 of H3, and 16 of H4 (Figure 4) [21,54]. Multiple non-histone targets have also been described (Figure 4). TAFI68 [TBP (TATA-box binding protein)-associated factor I 68], the second largest subunit of the TBP-containing complex, TIF (transcription initiation factor)-IB/SL, which regulates transcription of RNA Pol I (RNA polymerase I), was the first non-histone target described for the mouse SIRT1 protein.
Figure 4. Cellular functions of mammalian sirtuins.
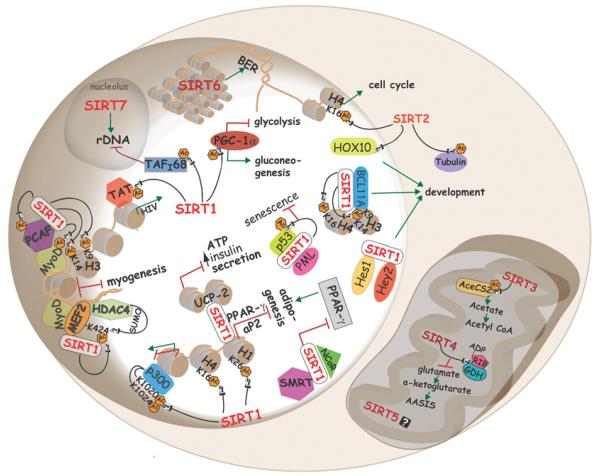
Sirtuins regulate a variety of process in mammalian cells. For example, in the nucleus, SIRT1 modulates chromatin structure by deacetylating specific lysine residues in histones H1, H3 and H4. Also, SIRT1 alters gene expression by targeting Lys1020 and Lys1024 of p300. Deacetylation of Tat by SIRT1 promotes HIV-1 replication. SIRT1 complexes with PML and mediates p53 deacetylation, thus protecting cells from senescence. SIRT1 represses muscle differentiation by two different mechanisms that involve MyoD: (i) in co-operation with PCAF and (ii) by deacetylating Lys424 of MEF2, which promotes its sumoylation by HDAC4, leading to suppression of myogenic genes. SIRT1 interacts with PPAR-γ and aP2 promoters, as well as with the PPAR-γ cofactors NcoR and SMRT to inhibit adipogenesis. Interactions of SIRT1 with BCL11A or Hes1/Hey2, and SIRT2 with the homeobox transcription factor, HOXA10, may regulate development. Glucose metabolism and energy modulated through deacetylation of PCG-1α and transcriptional repression of UCP2 respectively. SIRT6 mainly localizes to heterochromatin to regulate base excision repair, and SIRT2 shuffles from the cytoplasm to the nucleus during mitosis, where it deacetylates Lys16 of histone H4. In the nucleolus, SIRT7 activates RNA Pol I transcription, whereas SIRT1 represses it by deacetylating TAFI68. Cytoplasmic SIRT1 and mitochondrial SIRT3 deacetylate and activate AceCS1 and AceCS2 respectively. SIRT4 mono-ADP-ribosylates GDH, which inhibits amino-acid-stimulated insulin secretion. AC, acetylation.
Deacetylation of TAFI68 decreases its DNA binding activity and represses RNA Pol I-mediated transcription in vitro [55]. Interestingly, the nucleolar sirtuin, SIRT7, activates RNA Pol I-mediated transcription and expression of ribosomal RNA genes. SIRT7 has been found to be part of the RNA Pol I machinery, interacting with both the promoter and with transcribed regions of the rDNA locus, as well as with histones, predominantly H2A and H2B [56].
Bouras et al. [57] showed that, in HEK (human embryonic kidney)-293 cells, SIRT1 negatively regulates the activity of the histone acetytransferase p300 by deacetylating the lysine residues at positions 1020 and 1024, both of which reside in a key regulatory domain. Since p300 is a limiting transcriptional cofactor, its inhibition by SIRT1 may play an important role in the orchestration of metabolism and cellular differentiation.
The acetyltransferases PCAF [p300/cAMP-response-element-binding protein-associated factor] and GCN5 interact with SIRT1 and mediate the formation of a complex with the muscle-specific transcription factor MyoD (myogenic differentiation). Once the complex is recruited to chromatin, SIRT1 deacetylates the proteins of this complex and also Lys9 and Lys14 of H3 in the myogenin and myosin heavy chain promoters; all these events collectively inhibit muscle gene expression, which produces retardation of muscle differentiation [58]. SIRT1 also has been described to regulate MEF2 (MADS box transcription enhancer factor 2), a family of transcriptional factors important for muscle differentiation. In skeletal muscle cells, MEF2 members co-operate with MyoD to activate critical genes involved in muscle differentiation. Zhao et al. [59] showed that Lys424 on MEF2D, which when acetylated activates MEF2D and when sumoylated inhibits it, is also a SIRT1 target. In addition, the histone deacetylase HDAC4, which has been shown to have SUMO E3 ligase activity, also forms a complex with SIRT1, suggesting that both of these enzymes together enhance MEF2 sumoylation, thereby negatively regulating myogenesis (Figure 4).
An additional study also shows that SIRT1 activators promote osteoblast differentiation but markedly decrease adipocyte formation [60].
Apoptosis and cell survival: implication in cancer
p53-dependent apoptosis
The second target described for mouse and human SIRT1 was the tumour suppressor p53. SIRT1 and p53 are intimately linked: they not only physically associate, but they also regulate each other’s activity (Figure 5). SIRT1 has been shown to deacetylate multiple lysine residues of p53 including Lys317,Lys370 and Lys379 in mouse p53, which correspond to Lys320,Lys373 and Lys382 of human p53 [61,62]. SIRT1-dependent deacetylation of p53 inhibits its transactivation activity and suppresses apoptosis in response to oxidative stress and DNA damage [36,61].
Figure 5. SIRT1 in regulating apoptosis and cell survival.
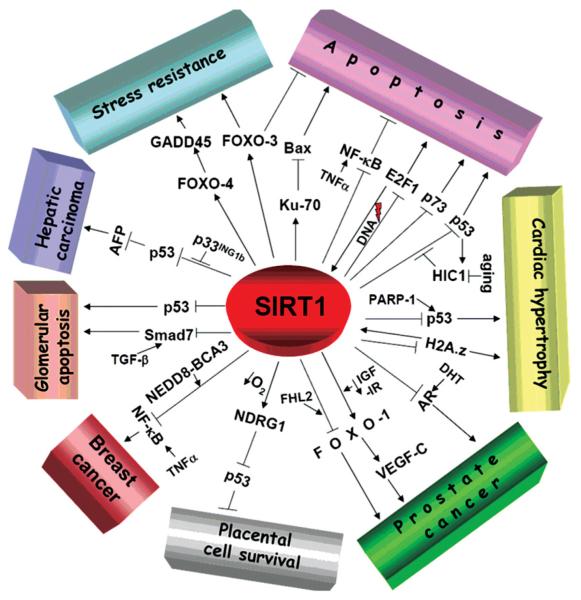
Different apoptotic and cell survival pathways regulated by SIRT1. Refer to the text for details. Whether or not SIRT1 is a tumour suppressor or oncogene in vivo is, as yet, unclear.
Although the SIRT1/p53 pathway may promote a beneficial effect in different diseases, Chen et al. [63] described a novel regulatory mechanism using mouse models. In co-operation with the HIC1 (hypermethylated in cancer 1) transcriptional repressor, SIRT1 mediates the bypass of apoptosis, potentially by promoting cell survival and tumorigenesis via p53 (Figure 5). HIC1 forms a complex with SIRT1 via its N-terminal POZ domain, which binds directly to the SIRT1 promoter and represses its expression, and attenuates p53-dependent apoptotic responses. Since p53 is able to transactivate HIC1 transcription, it has been proposed that SIRT1—HIC1—p53 act in a complex feedback loop. Under normal physiological conditions, HIC1 represses SIRT1, promoting p53 activity and apoptosis under stress. However, in cells set to recover from DNA damage, p53 down-regulates HIC1, which induces SIRT1 transcription and promotes cell survival. The authors of the study speculate that HIC1 hypermethylation experienced during aging may lead to increased SIRT1 expression, which may deacetylate p53 and increase cellular risk for neoplasic transformation and tumorigenesis (Figure 5).
Thymocytes from SIRT1-deficient mice show p53 hyperacetylation after DNA damage, and increased sensitivity to ionizing radiation [62]. However, recent reports surmise that, despite p53 deacetylation by SIRT1, this pathway does not impair thymocyte and/or splenocyte cell survival following radiation, nor cell viability or growth after DNA damage of primary human mammary epithelial cells. Accordingly, the elimination of p53 in SIRT1 knockout mice does not relieve their phenotype, suggesting that p53 hyperacetylation is not the main cause of those effects. [64,65]. These discrepancies suggest that regulation of p53 by SIRT1 may involve the input of other complex mechanisms, in addition to p53 deacetylation.
Bax-, E2F1- and p73-mediated apoptosis
Other mechanisms by which SIRT1 regulates cell survival have been described (Figure 5), for example by deacetylating Ku70, a DNA-repair factor and inhibitor of Bax-mediated apoptosis. Deacetylated Ku70 complexes with the proapoptotic factor Bax, sequestering it away from mitochondria and preventing it from triggering apoptosis in HEK-293 cells in response to stress [66].
E2F1, a regulator of cell proliferation and the cell cycle, together with SIRT1, form a negative regulatory feedback loop. E2F1 binds directly to the SIRT1 promoter and induces its transcription, whereby SIRT1 interaction becomes an efficient inhibitor of E2F1 apoptotic function. This mutual regulation of SIRT1 and E2F1 protects against DNA damage in U20S and H1299 cancer cell lines [67].
p73, a p53-related tumour suppressor, is one of the most recent targets described for SIRT1. Similar to p53, deacetylation of p73 suppresses its transcriptional activity and inhibits apoptosis in HEK-293 cells (Figure 5) [68].
Stress resistance and cell survival mediated by FOXO (Forkhead box class O) transcription factors
Another alternative pathway by which SIRT1 increases cell survival is through the regulation of FOXO transcription factors (Figure 5). There are four FOXOs and so far SIRT1 has been shown to deacetylate three of them: Foxo1, Foxo3a and Foxo4. The LXXLL motif of Foxo1 has been shown to be indispensable for its transcriptional regulation and for SIRT1 binding. Implications of Foxo1 in prostate cancer will be discussed below [69,70]. SIRT1 also affects Foxo3a function in mammalian cells, including neurons and fibroblasts, reducing apoptosis in response to stress stimuli, but increasing in expression of DNA repair and cell-cycle checkpoint genes [71,72].
Hydrogen peroxide treatment induces acetylation of FOXO4 and inhibits its transactivation potential. Interestingly, this suppression is reverted by the interaction with SIRT1, which enhances mammalian cellular defences against oxidative stress, such as the expression of the growth arrest and DNA repair protein, GADD45 (growth arrest and DNA-damage-inducible α) [73,74]. In addition, it has been shown that SIRT1 acting via FOXO4 suppresses the pro-apoptotic proteases caspase-3 and caspase-7 in transformed, but not in untransformed epithelial cells [75]. In what appears to be a feedback loop, caspase-9 and Bcl-xL regulate SIRT1 cleavage during apoptosis, shifting its localization from the nucleus to the cytoplasm [76].
Breast, liver and prostate cancer
Despite its numerous anti-apoptotic activities, SIRT1 may also promote apoptosis under certain conditions. For example, SIRT1 deacetylates RelA/p65, the most prevalent form of NF-κB (nuclear factor κB). Deacetylation inhibits the transactivation potential of RelA/p65, which sensitizes human cells to apoptosis in response to TNFα (tumour necrosis factor α) [77]. A study shows that the breast cancer associated protein, BCA3, when neddylated (modified by NEDD8) interacts with SIRT1 and suppress NF-kB-dependent transcription, also sensitizing human breast and prostate cancer cells (such as MCF7 and DU145 cells respectively) to TNF-α-induced apoptosis (Figure 5) [78].
In addition, it has been shown recently that SIRT7 levels of expression increase significantly in breast cancer, and that SIRT7 and SIRT3 both are highly transcribed in lymph-node positive breast biopsies, a stage in which the tumour size is at least 2 mm and the cancer has already spread to the lymph nodes [79]. SIRT7 is also overexpressed in human thyroid carcinoma cell lines and tissues [80,81].
The aberrant expression of human AFP (α-fetoprotein) is characteristic of hepatocellular carcinoma cases and serves as a diagnostic tumour-specific marker. Interestingly, it was shown that, in hepatocarcinoma cells (HepG2 cells), p33ING1b, a regulator of the cell cycle and apoptosis, interacts with human SIRT1, thereby inhibiting its ability to deacetylate p53, which negatively regulates transcription of AFP (Figure 5) [82].
Abnormal function of the androgen receptor has been associated with both the pathogenesis and the progression of human prostate cancer. SIRT1 interacts and preferentially deacetylates Lys630 in the androgen receptor lysine motif, which represses its oncogenic signalling and inhibits prostate cancer cells from growing in response to the androgen dihydrotestosterone [83]. Conversely, the four and a half LIM domain protein, FHL2, enhances the interaction and deacetylation of Foxo1 by SIRT1 in prostate cancer cells, and this effect has been proposed to promote tumorigenesis in response to increased stress during aging [84].
Brain cancer
The mammalian sirtuin, SIRT2, seems to suppress certain brain tumours known as gliomas. SIRT2 resides in a genomic region frequently deleted in human gliomas and ectopic expression of SIRT2 in glioma-derived cell lines markedly reduces their capacity to form colonies in vitro [85]. These findings have led to the hypothesis that inactivation of SIRT2 may be a cause of gliomas and that its activation may protect against these diseases or even potentially treat them.
Consistent with this hypothesis, SIRT2 expression is intimately linked to the cell cycle and may orchestrate its entry following DNA damage. The expression of this sirtuin increases dramatically during mitosis and is hyperphosphorylated during the G2/M phase transition. The phosphatase CDC (cell division cycle) 14B regulates SIRT2 phosphorylation, leading to its subsequent ubiquitination and degradation by the 26S proteosome, a process that may promote exit from mitosis. SIRT2 also interacts with and deacetylates Lys16 on histone H4, leading to the formation of condensed chromatin during the G2/M transition [86]. Exogenously expressed SIRT2 blocks chromosomal condensation and hyperploidy in glioma cell lines, accompanied by the presence of cyclin B/cdc2 activity in response to mitotic stress. Thus, SIRT2 may be a novel metaphase check-point protein that promotes genomic integrity and inhibits the uncontrolled proliferation of transformed cells [87]. There is still considerable work to be done, particularly with mouse models, to determine the role of SIRT2 in the initiation and progression of brain tumours or other types of cancer in vivo.
Placental cell survival
SIRT1 is up-regulated in primary trophoblasts from human placenta exposed to hypoxia and enhances the expression of the NDRG1 (N-Myc down-regulated gene 1), which inhibits p53 transcription. Therefore by direct inactivation of p53 or by reducing its expression via NDRG1, SIRT1 promotes cell resistance to oxygen deficiency in the placenta (Figure 5) [88].
Kidney diseases
Glomerular apoptosis is experienced in different kidney diseases. Interestingly, SIRT1 diminishes mesangial cell apoptosis induced by oxidative stress via p53 [89]. SIRT1 also attenuates TGF-β (transforming growth factor-β) apoptotic signalling that is mediated by the effector molecule Smad7. SIRT1-dependent deacetylation of Smad7 at Lys60 and Lys70 enhances its ubiquitin-dependent proteasomal degradation via Smurf1 (Smad ubiquitination regulatory factor 1), thus protecting glomerular mesangial cells from TGF-β-dependent apoptosis (Figure 5) [90].
Cardiac hypertrophy
Heart failure is a pathological state in which the heart is unable to pump blood efficiently. It begins with cardiac hypertrophy, followed by increased apoptosis, which culminates with organ failure. Thus, decreasing hypertrophy or apoptosis in cardiac myocytes can ameliorate the disease, and there is reason to suspect that SIRT1 activation may be useful in this regard. SIRT1 protects primary cultured myocytes from programmed cell death induced by serum starvation or by the activation of PARP-1 [poly(ADP-ribose)polymerase-1] in a p53-dependent manner [91,92]. SIRT1 also promotes PARP-1-mediated cell survival in response to DNA damage via AIF (apoptosis inducing factor) [93]. SIRT1 also deacetylates Lys115 and Lys121 of the histone variant H2A.Z, a factor known to promote cardiac hypertrophy. In doing so, SIRT1 promotes the ubiquitination and proteosome-dependent degradation of H2A.Z, which may help to protect against heart failure [94].
Cellular senescence
Cellular senescence is a state of permanent cell cycle arrest, accompanied by defined morphological changes that may be induced by several stimuli. Cellular senescence has been proposed as a major contributor to mammalian aging and also as a tumour suppressor mechanism. It is not currently clear whether SIRT1 inhibits or contributes to cellular senescence. Under certain conditions, SIRT1 localizes to discrete nuclear substructures with PML (promyelocytic leukaemia) protein to form nuclear bodies. PML may co-activate or co-repress various transcription factors localized in the nuclear bodies, thus mediating different apoptotic signals. Up-regulation of a PML isoform, PML-IV, recruits SIRT1 into the nuclear bodies together with p53. Thus, SIRT1 rescues primary mouse embryonic fibroblast from PML-mediated premature cellular senescence by inhibiting the pro-apoptotic action of p53 (Figure 4) [95]. Conversely, SIRT1 has been found to promote cellular senescence. Mouse embryonic fibroblasts lacking SIRT1 had an extended replicative potential and showed greater proliferative capacity following chronic but sublethal stress, a phenotype shown to require the tumour suppressor p19ARF and its downstream target p53 [96]. A recent study showed that SIRT1 levels are lower in cells that have been serially passaged or in dividing tissues of aged mice, such as thymus and testis, but not in immortalized cells or post-mitotic organs [97].
The regulatory role of SIRT1 in senescence or apoptosis, whether it is positive or negative, could have a major bearing on its influence on tumorigenesis in the elderly. Given that cell culture studies can be relatively poor predictors of how tumorigenic cells behave in vivo, mouse transgenic studies are now critical for answering questions about the roles of sirtuins in cancer and cell survival.
DNA repair
Novel insights into the role of mammalian sirtuins in DNA repair processes were first described by Motoslavsky et al. [98], who showed that SIRT6 knockout mice displayed signs of premature aging, such as decreased size, loss of subcutaneous fat, lymphocyte apoptosis, colitis, abnormal spine curvature and metabolic defects leading to premature death after only 4 weeks of age. A primary defect in the animals seems to be impairment in base excision repair. Mechanistically, however, it is unclear how SIRT6 acts in this process, and the authors speculate that this protein may regulate accessibility of the base excision repair machinery to the targeted DNA damage sites.
No role for SIRT1 in DNA repair has yet been described, although it seems very probable that one will be found, given its structural and functional homology to yeast Sir2.
HIV-1 replication
Tat (transactivator of transcription) is a protein of the HIV-1 virus essential for the transcriptional activation of the integrated provirus. Without Tat, HIV-1 viral replication is compromised due to the elevated level of aborted transcription resulting from inefficient elongation. SIRT1 deacetylates Lys50 of Tat, leading to its activation, which increases viral transcription (Figure 4) [99]. The relevance of this finding to the proliferation of HIV-1 in infected individuals remains to be determined.
Ion channel regulation
The metabolite OAADPr is a product of sirtuin-mediated deacetylation. OAADPr binds to and activates the cytoplasmic domain of the TRPM2 (transient receptor potential melastatin-related channel 2), a non-selective cation channel, whose prolonged activation leads to cell death. In experiments using puromycin, a selective drug that activates the TRPM2 channel, cell death is reduced by incubating cells with sirtuin inhibitors or by decreasing the expression of SIRT2 or SIRT3 [100]. Thus, by generating OAADPr, sirtuins may regulate channel gating and possibly other biological functions that remain to be elucidated.
Development
High levels of SIRT1 mRNA are detectable in heart, brain, spinal cord and dorsal root ganglia of embryos, indicating that it may play a critical role in development [101]. Consistent with this, SIRT1 knockout mice show developmental defects. About half of the numbers of pups expected are typically born, and of these approx. 20 % survive to adulthood. SIRT1 knockout mice are also markedly smaller than their littermates, develop more slowly and show dramatic perturbations in eye morphogenesis and cardiac septation. Both males and females that survive to adulthood are sterile, males have low sperm counts and females fail to ovulate, which is probably due to a hormonal inefficiency [62,102].
Explanations for the phenotype of the knockouts have come from biochemical studies. The Hairy and Hey subfamilies of the bHLH (basic helix—loop—helix) proteins primarily function as transcriptional repressors that direct metazoan development. The mammalian homologue of Drosophila Hairy is Hes1 and of Hey is Hey2. SIRT1 interacts with Hes1 and Hey2, both in vivo and in vitro, and modulates their ability to repress target promoters. Even though it has not yet been addressed how SIRT1 represses transcription in this system, one possibility is that Hes1 and Hey2 recruit this sirtuin to chromatin, where it may deacetylate histones and inhibit the expression of target promoters [103]. SIRT1 also interacts with BCL11A and CTIP2, a mammalian and chicken protein respectively, which are involved in haematopoietic cell development and malignances. Through this interaction, SIRT1 is recruited to target promoters, whereby it deacetylates histones H3/H4 and stimulates transcriptional repression (Figure 4) [104].
There are hints that other sirtuins may also be involved in development. For example, SIRT2 interacts with HOXA10, an evolutionarily conserved homeobox transcription factor important for cell-type determination during embryogenesis [105], and microinjection of starfish oocytes with AADPR delays or blocks embryonic cell division [106].
Inflammation
One of the hallmarks of rodents on a CR diet is reduced inflammation, and a key regulator of inflammation is NF-κB. As mentioned above, SIRT1 deacetylates the RelA/p65 subunit of NF-κB, which inhibits its transactivation potential [77]. A recent report showed that the exposure of either monocyte/macrophage cells or rat lung inflammatory cells to cigarette smoke extracts decreases the interaction between SIRT1 and RelA/p65, thus resulting in increased acetylation and activation of pro-inflammatory responses mediated by NF-κB [107]. Conversely, higher levels of SIRT1 in CR rodents may be one of the reasons for their decreased inflammatory responses.
Regulation of energy metabolism
At present, three mammalian sirtuins have been shown to play key roles in regulating metabolism in response to dietary changes: namely SIRT1, SIRT3 and SIRT4. SIRT1 promotes fat mobilization in white adipose tissue by binding to and repressing genes involved in adipogenesis, such as PPAR-γ (peroxisome proliferator-activated receptor γ ) and aP2 (fatty acid binding protein), and also by quenching the PPAR-γ cofactors NCoR (nuclear receptor co-repressor) and SMRT (silencing mediator of retinoid and thyroid hormone receptors) [108]. Interestingly, SIRT1 over-expression or pharmacological activation by resveratrol results in PPAR-γ-mediated transcriptional repression, inhibition of adipogenesis, enhanced lipolysis and the release of free fatty acids [108].
Adiponectin, an adipocyte-derived hormone whose plasma concentrations mostly correlates inversely with adiposity, is also regulated by SIRT1. FOXO1 forms a transcriptional complex at the mouse adiponectin promoter with C/EBPα (CCAAT/enhancer-binding protein α). SIRT1 deacetylates FOXO1 and enhances its interaction with C/EBPα, thereby increasing adiponectin concentrations [109]. Adiponectin regulates energy homoeostasis and glucose and lipid metabolism [110]. Administration of adiponectin to high-fat-fed obese mice and to a mouse model of lipoatrophic diabetes improves insulin sensitivity while lowering blood glucose, which suggests that adiponectin may confer protection against the development of insulin resistance and Type 2 diabetes [111]. Strikingly, it has been shown that CR increases the levels of circulating adiponectin in rats [112], an effect that may be mediated by SIRT1.
By deacetylating PGC-1α (PPAR-γ co-activator 1α), SIRT1 represses glycolysis and increases hepatic glucose output [113]. Also, through PGC-1α regulation, SIRT1 modulates mitochondrial function, metabolic homoeostasis, increases consumption of oxygen in muscle fibres and induces oxidative phosphorylation genes and mitochondrial biogenesis [114].
When SIRT1 is overexpressed in pancreatic β-cells, mice exhibit enhanced glucose-stimulated insulin secretion and ATP production [115,116]. The mechanism appears to be through SIRT1-mediated transcriptional repression of the UCP2 (uncoupling protein 2) gene, which encodes a mitochondrial inner membrane protein that uncouples O2 consumption from ATP generation and generates heat. High UCP2 expression is observed in SIRT1 knockout mice, along with blunted insulin secretion and lower ATP levels following glucose stimulation of β-cells (Figure 4) [116].
Also, by deacetylating FOXO1, SIRT1 promotes activation and transcription of two genes, NeuroD (neurogenic differentiation) and MafA, which may preserve insulin secretion and promote cell survival in vivo [117].
Roles for the mitochondrial sirtuins in regulating key metabolic pathways were recently uncovered. Tong and colleagues [39] have shown previously that SIRT3 decreases mitochondrial membrane potential and the production of reactive oxygen species, while increasing cellular respiration. In addition, during cold exposure, SIRT3 is induced in brown adipose tissue, which promotes the expression of mitochondrial genes, including those encoding UCP1, PGC-1α, cytochrome c oxidase subunits II and IV (COX II and COX IV) and ATP synthetase [39]. Interestingly the brown adipose tissue of obese mice has less SIRT3 protein and decreased expression of mitochondrial proteins.
CR induces numerous metabolic changes, including a shift away from glycolysis and fatty acid biosynthesis in the liver, and induction of gluconeogenesis and improved insulin sensitivity. How this occurs is not well understood, but some sirtuins may underlie many of these changes. Acetyl-CoA is a small molecule that is central to the synthesis of fatty acids, amino acids, ketone bodies and the tricarboxylic acid cycle. In mammals, acetate from the diet or from endogenous reactions is converted into acetyl-CoA in the cytoplasm by AceCS1 (acetyl-CoA synthetase 1) or in the mitochondria by AceCS2. New results show that SIRT1 and SIRT3 deacetylate and thereby activate AceCS1 [118] and AceCS2 [119] respectively. Given that SIRT1 and SIRT3 are both induced by CR, it seems plausible that during periods of food limitation, these sirtuins alter the rate of fatty acid synthesis and regulate how much carbon is shuttled into the tricarboxylic acid cycle for ATP production.
The first comprehensive study of SIRT4 was published recently by Haigis et al. [37], who showed that this mitochondrial sirtuin regulates amino-acid-stimulated insulin secretion in pancreatic β-cells. The enzyme GDH (glutamate dehydrogenase) regulates glutamate and glutamine metabolism, promotes ATP synthesis and enhances insulin secretion. GDH can be mono-ADP-ribosylated by SIRT4, which inhibits GDH activity, and slows the conversion of glutamate into α-ketoglutarate (Figure 4). Accordingly, pancreatic islets of SIRT4 knockout mice display an increase in GDH activity, higher insulin levels, up-regulation of amino-acid-stimulated insulin secretion and secrete insulin in response to glutamine. Similar effects are also observed in pancreatic islets of mice under a CR diet, suggesting that SIRT4 is down-regulated by CR, thus inducing the expression of GDH and allowing glutamine to serve as an insulin secretagogue.
Sirtuins and the nervous system
Although most of the sirtuins are highly expressed in brain [34,38,120], at present very little is known about their roles in the nervous system. In the brain, only SIRT1 and SIRT2 have been studied to any significant extent, and it is already clear that these two proteins may have different functions. SIRT1 is present mainly in neuronal bodies, whereas SIRT2 is present in oligodendrocytes and Schwann cells that form the myelin sheaths covering the axons (Figure 6) and in olfactory sensory neurons [121]. Abnormalities in the central nervous system have been described in SIRT1 knockout mice whose retinal cell layers are significantly thinner, and the inner and outer segments of photoreceptor cells are difficult to discern. In addition, embryos also show abnormal multiple retinal involutions [62].
Figure 6. SIRT1 and SIRT2 localize differentially in the brain.
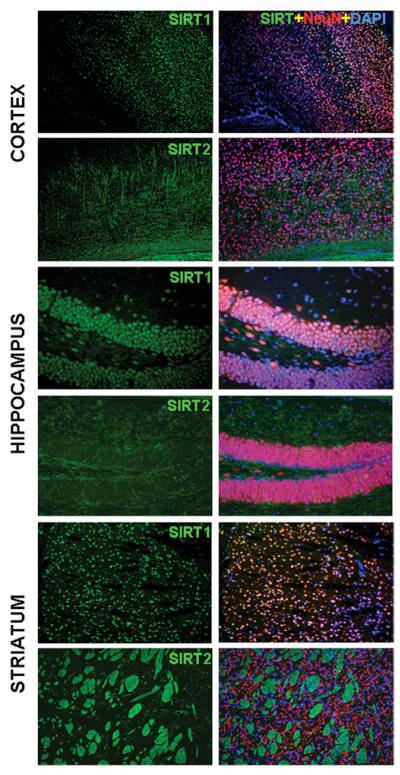
Immunofluorescence microscopy using specific SIRT1 and SIRT2 antibodies in cortex, hippocampus and striatum. SIRT1 (green) co-localizes with the neuronal marker NeuN (red) and DAPI (4′,6-diamidino-2-phenylindole; blue) in cortical neurons, hippocampal granular neurons and striatal neurons, while SIRT2 does not co-label with NeuN (neuron-specific nuclear-proteins) or DAPI; it mainly co-localizes with the fibres of myelinated axons in white matter and oligodendrocytes. Samples were processed as described previously [141].
Most studies of SIRT1 in the brain have focused on its role in neuroprotection, stemming from a discovery by Milbrandt and colleagues [122] that an NAD+ biosynthetic enzyme, Nmnat (nicotinate mononucleotide adenylyltransferase), is primarily responsible for protection from axonal or Wallerian degeneration. Using neuronal explants from primary dorsal root ganglia, Milbrandt et al. showed that protection by Nmnat from chemical or mechanical injury is due primarily to the actions of SIRT1. SIRT1 has since been shown to also protect neurons against β-amyloid-induced toxicity by inhibiting NF-κB signalling in microglia [123] or by down-regulating the serine/threonine ROCK1 (Rho kinase) expression in neurons, resulting in induction of α-secretase [124]. Interestingly, CR prevents genetically engineered mice prone to Alzheimer’s disease from developing β-amyloid neuropathology, and also increases SIRT1 and NAD+/nicotinamide ratios in the brain, strongly suggesting that the mechanism by which CR protect neurons against β-amyloid deposition is through the modulation of SIRT1 [124].
Inhibition of apoptosis is probably not the only mechanism by which SIRT1 protects neurons. It is becoming clear that the maintenance of mitochondrial number and function is critical for normal neuronal function, and that the loss of mitochondria is an underlying cause of many neurodegenerative diseases [125], as evidenced by the fact that PGC-1α, one of the downstream effectors of SIRT1, is a master regulator of mitochondrial number and function and is highly neuroprotective. PGC-1α null-mice are, for example, considerably more sensitive to neuronal loss by the reactive oxygen species generating compounds and neurotoxic agents MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydro-pyridine) and kainic acid, which disrupt the substantia nigra and hippocampus respectively. Increasing PGC-1α levels substantially protects neurons from oxidative-stress-induced death [126]. Therefore it seems likely that the ability of SIRT1 to provide neuroprotection stems, in part, from its regulation of PGC-1α activity. Therefore analysis of mice overexpressing SIRT1 in the brain may provide valuable clues as to how SIRT1 acts as a neuroprotectant in vivo and which downstream pathways are important for SIRT1-mediated protection.
Apart from the aforementioned link between SIRT2 and glioma, very little is known about the function of SIRT2 in the brain. Its expression pattern, however, may provide some clues. The high level of SIRT2 expression in olfactory neurons indicates that it may play a role in sensory perception [121]. SIRT2 is also developmentally regulated and has a pattern of expression that resembles the Nf155 gene, which encodes the 155 kDa isoform of neurofascin, a protein required for the adequate assembly of paranodal axoglial junctions in myelinated axons, suggesting that this sirtuin could play a role in axonal myelinization [127].
Currently, the field has no knowledge about the role of the mitochondrial sirtuins SIRT3–5 in the nervous system, but there are some important clues. The mitochondrial uncoupling proteins, UCP2, UCP4 and UCP5/BMCP1, are highly expressed in the central nervous system, but the brains of Alzheimer’s disease patients show significantly reduced expression of these genes [128]. Also, UCP2 has been shown to be neuroprotective in experimental stroke and brain trauma models [129] as well as in Parkinson’s disease [130]. UCPs are induced by free radicals and non-esterified fatty acids, and their activity regulates mitochondrial biogenesis, calcium flux, free radical production and local temperature [131]. Thus, by modulating UCP proteins, sirtuins may influence these key neuronal functions.
With regard to SIRT4, this enzyme controls glutamate metabolism in response to amino acid availability, by modifying and inhibiting GDH in the pancreas. Given that glutamate is a neuro-transmitter at most excitatory synapses in the brain, it seems plausible that SIRT4 could also regulate neuronal impulses or even the ability of the brain to respond to the toxic quantities of glutamate that are released following a stroke.
STACS (SIRTUIN ACTIVATING COMPOUNDS)
The fact that additional SIR2 extends the lifespan of diverse organisms prompted a search for STACs (sirtuin activating compounds) that might extend lifespan in the absence of CR or genetic manipulation. A series of STACs, including resveratrol, a small plant molecule synthesized in response to stress, were identified as activators of mammalian SIRT1 in vitro [132]. Consequently, this polyphenolic compound found naturally in the skin of grapes, and a variety of other plants, has been shown to extend lifespan in organisms of different phyla, such as yeast, worms, flies and fish [133-136]. Resveratrol only extends lifespan when the SIR2 gene is present, i.e. resveratrol has no apparent effect when the SIR2 gene is deleted. Experiments in different mammalian cell lines show that resveratrol modulates a variety of cellular process in a SIRT1-dependent manner, including neuroprotection, tumour suppression, differentiation and inflammation (reviewed in [137-139]).
Two recent studies have addressed the effects of resveratrol in mammals in vivo. Baur et al. [139] showed that, when this polyphenol is administrated in a daily dose of 22.4 mg/kg to middle-aged mice on a high-calorie diet, the animals displayed a shift in their physiology towards that of mice on standard diet, with increased mitochondria, lower blood glucose and insulin, and a hepatic gene expression profile matching that of lean mice, concomitant with increased SIRT1 and PGC1-α activity. Similarly, Lagouge et al. [114] showed that mice fed with a diet that induced obesity supplemented with resveratrol at a dose 20 times higher than the former study (400 mg/kg) were actually leaner than the controls that did not receive resveratrol. They also had increased oxidative-type muscle fibres, enhanced resistance to muscle fatigue, lower blood glucose and insulin, and a greater tolerance to cold. The activities of SIRT1 and PGC-1α were also increased by resveratrol and, even more interestingly, three single nucleotide polymorphisms in the SIRT1 gene of Finnish human subjects were significantly associated with systemic energy expenditure, now implicating SIRT1 as a regulator of energy and metabolic homoeostasis in humans.
PERSPECTIVES
When SIR2 was first discovered in yeast 27 years ago, it could hardly have been anticipated how much interest would be taken in this family of proteins. Several studies over the last 7 years have illuminated our understanding of the role of sirtuins in different organisms. Currently, biological functions ranging from DNA repair to metabolism have been attributed to six of the seven sirtuins found in mammals, with SIRT1 being the most extensively studied.
Arguably, some of the most noteworthy and intriguing discoveries in recent years have been the polymorphisms in human sirtuin genes, which suggest a role for sirtuins in human health and longevity. Allele variants of SIRT1 are associated with energy expenditure and a variant of the SIRT3 gene, specifically in an enhancer within intron 5, correlates with male lifespan beyond 90 years [140].
Many questions remain to be answered, such as whether sirtuins regulate aging in mammals, or if resveratrol effects are indeed SIRT1-dependent. As researchers mine through existing human genome databases and patient records, together with additional studies in conditional knockout and transgenic mice, as well as disease models, these questions may be answered. Finally, all this will give us a far clearer perspective as to whether the sirtuins may provide novel therapies to alleviate age-associated changes, such as diabetes, cancer and cardiovascular disease, and possibly extend healthy human lifespan.
Acknowledgments
We thank Juan Carmona for a presubmission critique of this review.
Abbreviations used
- AceCS1
acetyl-CoA synthetase 1
- AFP
α-fetoprotein
- CDC
cell division cycle
- C/EBPα
CCAAT/enhancer-binding protein α
- CR
calorie restriction
- ERC
extrachromosomal circle
- FOXO
Forkhead box class O
- GDH
glutamate dehydrogenase
- HEK
human embryonic kidney
- HIC1
hypermethylated in cancer 1
- HST
homologues of SIR2
- IGF
insulin-like growth factor
- MAR1
mating-type regulator 1
- MEF2
MADS box transcription enhancer factor 2
- MyoD
myogenic differentiation
- NDRG1
N-Myc down-regulated gene 1
- NF-κB
nuclear factor κB
- Nmnat
nicotinate mononucleotide adenylyltransferase
- OAADPr
2′-O-acetyl-ADP-ribose
- PARP-1
poly(ADP-ribose) polymerase-1
- PGC-1α
PPAR-γ co-activator 1α
- PML
promyelocytic leukaemia
- PPAR-γ
peroxisome proliferator-activated receptor γ
- rDNA
ribosomal DNA
- RNA Pol I
RNA polymerase I
- SIR
silent information regulator
- SIRT
sirtuin
- STAC
sirtuin activating compounds
- TAFI68
TBP-associated factor I 68
- Tat
transactivator of transcription
- TBP
TATA-box binding protein
- TGF-β
transforming growth factor-β
- TNFα
tumour necrosis factor α
- TRPM2
transient receptor potential melastatin-related channel 2
- UCP2
uncoupling protein 2.
REFERENCES
- 1.Klar AJ, Seymour F, Macleod K. MAR1-A regulator of the HMa and HMα locus in Saccharomyces cerevisiae. Genetics. 1979;93:37–50. doi: 10.1093/genetics/93.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shore D, Squire M, Nasmyth KA. Characterization of two genes required for the position-effect control of yeast mating-type genes. EMBO J. 1984;3:2817–2823. doi: 10.1002/j.1460-2075.1984.tb02214.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ivy JM, Hicks JB, Klar AJ. Map positions of yeast genes SIR1, SIR3 and SIR4. Genetics. 1985;111:735–744. doi: 10.1093/genetics/111.4.735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rine J, Herskowitz I. Four genes responsible for a position effect on expression from HML and HMR in Saccharomyces cerevisiae. Genetics. 1987;116:9–22. doi: 10.1093/genetics/116.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gottlieb S, Esposito RE. A new role for a yeast transcriptional silencer gene, SIR2, in regulation of recombination in ribosomal DNA. Cell. 1989;56:771–776. doi: 10.1016/0092-8674(89)90681-8. [DOI] [PubMed] [Google Scholar]
- 6.Aparicio OM, Billington BL, Gottschling DE. Modifiers of position effect are shared between telomeric and silent mating-type loci in S. cerevisiae. Cell. 1991;66:1279–1287. doi: 10.1016/0092-8674(91)90049-5. [DOI] [PubMed] [Google Scholar]
- 7.Braunstein M, Rose AB, Holmes SG, Allis CD, Broach JR. Transcriptional silencing in yeast is associated with reduced nucleosome acetylation. Genes Dev. 1993;7:592–604. doi: 10.1101/gad.7.4.592. [DOI] [PubMed] [Google Scholar]
- 8.Brachmann CB, Sherman JM, Devine SE, Cameron EE, Pillus L, Boeke JD. The SIR2 gene family, conserved from bacteria to humans, functions in silencing, cell cycle progression, and chromosome stability. Genes Dev. 1995;9:2888–2902. doi: 10.1101/gad.9.23.2888. [DOI] [PubMed] [Google Scholar]
- 9.Derbyshire MK, Weinstock KG, Strathern JN. HST1, a new member of the SIR2 family of genes. Yeast. 1996;12:631–640. doi: 10.1002/(SICI)1097-0061(19960615)12:7%3C631::AID-YEA960%3E3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 10.Gotta M, Strahl-Bolsinger S, Renauld H, Laroche T, Kennedy BK, Grunstein M, Gasser SM. Localization of Sir2p: the nucleolus as a compartment for silent information regulators. EMBO J. 1997;16:3243–3255. doi: 10.1093/emboj/16.11.3243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsukamoto Y, Kato J, Ikeda H. Silencing factors participate in DNA repair and recombination in Saccharomyces cerevisiae. Nature. 1997;388:900–903. doi: 10.1038/42288. [DOI] [PubMed] [Google Scholar]
- 12.Mills KD, Sinclair DA, Guarente L. MEC1-dependent redistribution of the Sir3 silencing protein from telomeres to DNA double-strand breaks. Cell. 1999;97:609–620. doi: 10.1016/s0092-8674(00)80772-2. [DOI] [PubMed] [Google Scholar]
- 13.Martin SG, Laroche T, Suka N, Grunstein M, Gasser SM. Relocalization of telomeric Ku and SIR proteins in response to DNA strand breaks in yeast. Cell. 1999;97:621–633. doi: 10.1016/s0092-8674(00)80773-4. [DOI] [PubMed] [Google Scholar]
- 14.McAinsh AD, Scott-Drew S, Murray JA, Jackson SP. DNA damage triggers disruption of telomeric silencing and Mec1p-dependent relocation of Sir3p. Curr. Biol. 1999;9:963–966. doi: 10.1016/s0960-9822(99)80424-2. [DOI] [PubMed] [Google Scholar]
- 15.Smith JS, Boeke JD. An unusual form of transcriptional silencing in yeast ribosomal DNA. Genes Dev. 1997;11:241–254. doi: 10.1101/gad.11.2.241. [DOI] [PubMed] [Google Scholar]
- 16.Fritze C. a. E., R. Direct evidence for Sir2 modulation of chromatin structure in yeast rDNA. EMBO J. 1997;16:6495–6509. doi: 10.1093/emboj/16.21.6495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tsang AW, Escalante-Semerena JC. CobB, a new member of the SIR2 family of eukaryotic regulatory proteins, is required to compensate for the lack of nicotinate mononucleotide:5,6-dimethylbenzimidazole phosphoribosyltransferase activity in cobT mutants during cobalamin biosynthesis in Salmonella typhimurium LT2. J. Biol. Chem. 1998;273:31788–31794. doi: 10.1074/jbc.273.48.31788. [DOI] [PubMed] [Google Scholar]
- 18.Frye RA. Characterization of five human cDNAs with homology to the yeast SIR2 gene: Sir2-like proteins (sirtuins) metabolize NAD and may have protein ADP-ribosyltransferase activity. Biochem. Biophys. Res. Commun. 1999;260:273–279. doi: 10.1006/bbrc.1999.0897. [DOI] [PubMed] [Google Scholar]
- 19.Tanny JC, Dowd GJ, Huang J, Hilz H, Moazed D. An enzymatic activity in the yeast Sir2 protein that is essential for gene silencing. Cell. 1999;99:735–745. doi: 10.1016/s0092-8674(00)81671-2. [DOI] [PubMed] [Google Scholar]
- 20.Landry J, Sutton A, Tafrov ST, Heller RC, Stebbins J, Pillus L, Sternglanz R. The silencing protein SIR2 and its homologs are NAD-dependent protein deacetylases. Proc. Natl. Acad. Sci. U.S.A. 2000;97:5807–5811. doi: 10.1073/pnas.110148297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Imai S, Armstrong CM, Kaeberlein M, Guarente L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 2000;403:795–800. doi: 10.1038/35001622. [DOI] [PubMed] [Google Scholar]
- 22.Smith JS, Brachmann CB, Celic I, Kenna MA, Muhammad S, Starai VJ, Avalos JL, Escalante-Semerena JC, Grubmeyer C, Wolberger C, Boeke JD. A phylogenetically conserved NAD+-dependent protein deacetylase activity in the Sir2 protein family. Proc. Natl. Acad. Sci. U.S.A. 2000;97:6658–6663. doi: 10.1073/pnas.97.12.6658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Landry J, Slama JT, Sternglanz R. Role of NAD(+) in the deacetylase activity of the SIR2-like proteins. Biochem. Biophys. Res. Commun. 2000;278:685–690. doi: 10.1006/bbrc.2000.3854. [DOI] [PubMed] [Google Scholar]
- 24.Tanner KG, Landry J, Sternglanz R, Denu JM. Silent information regulator 2 family of NAD-dependent histone/protein deacetylases generates a unique product, 1-O-acetyl-ADP-ribose. Proc. Natl. Acad. Sci. U.S.A. 2000;97:14178–14182. doi: 10.1073/pnas.250422697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Denu JM. The Sir2 family of protein deacetylases. Curr. Opin. Chem. Biol. 2005;9:431–440. doi: 10.1016/j.cbpa.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 26.Sauve AA, Wolberger C, Schramm VL, Boeke JD. The biochemistry of sirtuins. Annu. Rev. Biochem. 2006;75:435–465. doi: 10.1146/annurev.biochem.74.082803.133500. [DOI] [PubMed] [Google Scholar]
- 27.Sinclair DA, Guarente L. Extrachromosomal rDNA circles — a cause of aging in yeast. Cell. 1997;91:1033–1042. doi: 10.1016/s0092-8674(00)80493-6. [DOI] [PubMed] [Google Scholar]
- 28.Sinclair DA, Mills K, Guarente L. Accelerated aging and nucleolar fragmentation in yeast sgs1 mutants. Science. 1997;277:1313–1316. doi: 10.1126/science.277.5330.1313. [DOI] [PubMed] [Google Scholar]
- 29.Kaeberlein M, McVey M, Guarente L. The SIR2/3/4 complex and SIR2 alone promote longevity in Saccharomyces cerevisiae by two different mechanisms. Genes Dev. 1999;13:2570–2580. doi: 10.1101/gad.13.19.2570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tissenbaum HA, Guarente L. Increased dosage of a sir-2 gene extends lifespan in Caenorhabditis elegans. Nature. 2001;410:227–230. doi: 10.1038/35065638. [DOI] [PubMed] [Google Scholar]
- 31.Wang Y, Tissenbaum HA. Overlapping and distinct functions for a Caenorhabditis elegans SIR2 and DAF-16/FOXO. Mech. Ageing Dev. 2006;127:48–56. doi: 10.1016/j.mad.2005.09.005. [DOI] [PubMed] [Google Scholar]
- 32.Rogina B, Helfand SL. Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc. Natl. Acad. Sci. U.S.A. 2004;101:15998–16003. doi: 10.1073/pnas.0404184101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Frye RA. Phylogenetic classification of prokaryotic and eukaryotic Sir2-like proteins. Biochem. Biophys. Res. Commun. 2000;273:793–798. doi: 10.1006/bbrc.2000.3000. [DOI] [PubMed] [Google Scholar]
- 34.Michishita E, Park JY, Burneskis JM, Barrett JC, Horikawa I. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Mol. Biol. Cell. 2005;16:4623–4635. doi: 10.1091/mbc.E05-01-0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.North BJ, Marshall BL, Borra MT, Denu JM, Verdin E. The human Sir2 ortholog, SIRT2, is an NAD+-dependent tubulin deacetylase. Mol. Cell. 2003;11:437–444. doi: 10.1016/s1097-2765(03)00038-8. [DOI] [PubMed] [Google Scholar]
- 36.Vaziri H, Dessain SK, Eaton E. Ng, Imai SI, Frye RA, Pandita TK, Guarente L, Weinberg RA. hSIR2 (SIRT1) functions as an NAD-dependent p53 deacetylase. Cell. 2001;107:149–159. doi: 10.1016/s0092-8674(01)00527-x. [DOI] [PubMed] [Google Scholar]
- 37.Haigis MC, Mostoslavsky R, Haigis KM, Fahie K, Christodoulou DC, Murphy AJ, Valenzuela DM, Yancopoulos GD, Karow M, Blander G, et al. SIRT4 inhibits glutamate dehydrogenase and opposes the effects of calorie restriction in pancreatic β cells. Cell. 2006;126:941–954. doi: 10.1016/j.cell.2006.06.057. [DOI] [PubMed] [Google Scholar]
- 38.Liszt G, Ford E, Kurtev M, Guarente L. Mouse Sir2 homolog SIRT6 is a nuclear ADP-ribosyltransferase. J. Biol. Chem. 2005;280:21313–21320. doi: 10.1074/jbc.M413296200. [DOI] [PubMed] [Google Scholar]
- 39.Shi T, Wang F, Stieren E, Tong Q. SIRT3, a mitochondrial sirtuin deacetylase, regulates mitochondrial function and thermogenesis in brown adipocytes. J. Biol. Chem. 2005;280:13560–13567. doi: 10.1074/jbc.M414670200. [DOI] [PubMed] [Google Scholar]
- 40.McCay CM. The effect of retarded growth upon the length of life and upon ultimate size. J. Nutr. 1935;10:63–79. [Google Scholar]
- 41.Jiang JC, Jaruga E, Repnevskaya MV, Jazwinski SM. An intervention resembling caloric restriction prolongs life span and retards aging in yeast. FASEB J. 2000;14:2135–2137. doi: 10.1096/fj.00-0242fje. [DOI] [PubMed] [Google Scholar]
- 42.Mair W, Goymer P, Pletcher SD, Partridge L. Demography of dietary restriction and death in Drosophila. Science. 2003;301:1731–1733. doi: 10.1126/science.1086016. [DOI] [PubMed] [Google Scholar]
- 43.Lakowski B, Hekimi S. The genetics of caloric restriction in Caenorhabditis elegans. Proc. Natl. Acad. Sci. U.S.A. 1998;95:13091–13096. doi: 10.1073/pnas.95.22.13091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ingle L, Wood T, Banta AM. A study of longevity, growth, reproduction and heart rate in Daphnia longispina as influenced by limitations in quantity of food. J. Exp. Zool. 1937;76:325–352. [Google Scholar]
- 45.Austad SN. Life extension by dietary restriction in the bowl and doily spider, Frontinella pyramitela. Exp. Gerontol. 1989;24:83–92. doi: 10.1016/0531-5565(89)90037-5. [DOI] [PubMed] [Google Scholar]
- 46.Weindruch R, Walford RL. Dietary restriction in mice beginning at 1 year of age: effect on life-span and spontaneous cancer incidence. Science. 1982;215:1415–1418. doi: 10.1126/science.7063854. [DOI] [PubMed] [Google Scholar]
- 47.Barger JL, Walford RL, Weindruch R. The retardation of aging by caloric restriction: its significance in the transgenic era. Exp. Gerontol. 2003;38:1343–1351. doi: 10.1016/j.exger.2003.10.017. [DOI] [PubMed] [Google Scholar]
- 48.Mattison JA, Roth GS, Lane MA, Ingram DK. Dietary restriction in aging nonhuman primates. Interdiscip. Top. Gerontol. 2007;35:137–158. doi: 10.1159/000096560. [DOI] [PubMed] [Google Scholar]
- 49.Lin SJ, Defossez PA, Guarente L. Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science. 2000;289:2126–2128. doi: 10.1126/science.289.5487.2126. [DOI] [PubMed] [Google Scholar]
- 50.Anderson RM, Bitterman KJ, Wood JG, Medvedik O, Sinclair DA. Nicotinamide and PNC1 govern lifespan extension by calorie restriction in Saccharomyces cerevisiae. Nature. 2003;423:181–185. doi: 10.1038/nature01578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lamming DW, Latorre-Esteves M, Medvedik O, Wong SN, Tsang FA, Wang C, Lin SJ, Sinclair DA. HST2 mediates SIR2-independent life-span extension by calorie restriction. Science. 2005;309:1861–1864. doi: 10.1126/science.1113611. [DOI] [PubMed] [Google Scholar]
- 52.Kaeberlein M, Powers RW, 3rd, Steffen KK, Westman EA, Hu D, Dang N, Kerr EO, Kirkland KT, Fields S, Kennedy BK. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science. 2005;310:1193–1196. doi: 10.1126/science.1115535. [DOI] [PubMed] [Google Scholar]
- 53.Guarente L. Calorie restriction and SIR2 genes: towards a mechanism. Mech. Ageing. Dev. 2005;126:923–928. doi: 10.1016/j.mad.2005.03.013. [DOI] [PubMed] [Google Scholar]
- 54.Vaquero A, Scher M, Lee D, Erdjument-Bromage H, Tempst P, Reinberg D. Human SirT1 interacts with histone H1 and promotes formation of facultative heterochromatin. Mol. Cell. 2004;16:93–105. doi: 10.1016/j.molcel.2004.08.031. [DOI] [PubMed] [Google Scholar]
- 55.Muth V, Nadaud S, Grummt I, Voit R. Acetylation of TAF(I)68, a subunit of TIF-IB/SL1, activates RNA polymerase I transcription. EMBO J. 2001;20:1353–1362. doi: 10.1093/emboj/20.6.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ford E, Voit R, Liszt G, Magin C, Grummt I, Guarente L. Mammalian Sir2 homolog SIRT7 is an activator of RNA polymerase I transcription. Genes Dev. 2006;20:1075–1080. doi: 10.1101/gad.1399706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bouras T, Fu M, Sauve AA, Wang F, Quong AA, Perkins ND, Hay RT, Gu W, Pestell RG. SIRT1 deacetylation and repression of p300 involves lysine residues 1020/1024 within the cell cycle regulatory domain 1. J. Biol. Chem. 2005;280:10264–10276. doi: 10.1074/jbc.M408748200. [DOI] [PubMed] [Google Scholar]
- 58.Fulco M, Schiltz RL, Iezzi S, King MT, Zhao P, Kashiwaya Y, Hoffman E, Veech RL, Sartorelli V. Sir2 regulates skeletal muscle differentiation as a potential sensor of the redox state. Mol. Cell. 2003;12:51–62. doi: 10.1016/s1097-2765(03)00226-0. [DOI] [PubMed] [Google Scholar]
- 59.Zhao X, Sternsdorf T, Bolger TA, Evans RM, Yao TP. Regulation of MEF2 by histone deacetylase 4- and SIRT1 deacetylase-mediated lysine modifications. Mol. Cell Biol. 2005;25:8456–8464. doi: 10.1128/MCB.25.19.8456-8464.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Backesjo CM, Li Y, Lindgren U, Haldosen LA. Activation of Sirt1 decreases adipocyte formation during osteoblast differentiation of mesenchymal stem cells. J. Bone Miner. Res. 2006;21:993–1002. doi: 10.1359/jbmr.060415. [DOI] [PubMed] [Google Scholar]
- 61.Luo J, Nikolaev AY, Imai S, Chen D, Su F, Shiloh A, Guarente L, Gu W. Negative control of p53 by Sir2α promotes cell survival under stress. Cell. 2001;107:137–148. doi: 10.1016/s0092-8674(01)00524-4. [DOI] [PubMed] [Google Scholar]
- 62.Cheng HL, Mostoslavsky R, Saito S, Manis JP, Gu Y, Patel P, Bronson R, Appella E, Alt FW, Chua KF. Developmental defects and p53 hyperacetylation in Sir2 homolog (SIRT1)-deficient mice. Proc. Natl. Acad. Sci. U.S.A. 2003;100:10794–10799. doi: 10.1073/pnas.1934713100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chen WY, Wang DH, Yen RC, Luo J, Gu W, Baylin SB. Tumor suppressor HIC1 directly regulates SIRT1 to modulate p53-dependent DNA-damage responses. Cell. 2005;123:437–448. doi: 10.1016/j.cell.2005.08.011. [DOI] [PubMed] [Google Scholar]
- 64.Kamel C, Abrol M, Jardine K, He X, McBurney MW. SirT1 fails to affect p53-mediated biological functions. Aging Cell. 2006;5:81–88. doi: 10.1111/j.1474-9726.2006.00191.x. [DOI] [PubMed] [Google Scholar]
- 65.Solomon JM, Pasupuleti R, Xu L, McDonagh T, Curtis R, DiStefano PS, Huber LJ. Inhibition of SIRT1 catalytic activity increases p53 acetylation but does not alter cell survival following DNA damage. Mol. Cell. Biol. 2006;26:28–38. doi: 10.1128/MCB.26.1.28-38.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B, Howitz KT, Gorospe M, de Cabo R, Sinclair DA. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science. 2004;305:390–392. doi: 10.1126/science.1099196. [DOI] [PubMed] [Google Scholar]
- 67.Wang C, Chen L, Hou X, Li Z, Kabra N, Ma Y, Nemoto S, Finkel T, Gu W, Cress WD, Chen J. Interactions between E2F1 and SirT1 regulate apoptotic response to DNA damage. Nat. Cell Biol. 2006;8:1025–1031. doi: 10.1038/ncb1468. [DOI] [PubMed] [Google Scholar]
- 68.Dai JM, Wang ZY, Sun DC, Lin RX, Wang SQ. SIRT1 interacts with p73 and suppresses p73-dependent transcriptional activity. J. Cell. Physiol. 2007;210:161–166. doi: 10.1002/jcp.20831. [DOI] [PubMed] [Google Scholar]
- 69.Nakae J, Cao Y, Daitoku H, Fukamizu A, Ogawa W, Yano Y, Hayashi Y. The LXXLL motif of murine forkhead transcription factor FoxO1 mediates Sirt1-dependent transcriptional activity. J. Clin. Invest. 2006;116:2473–2483. doi: 10.1172/JCI25518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li J, Wang E, Rinaldo F, Datta K. Upregulation of VEGF-C by androgen depletion: the involvement of IGF-IR—FOXO pathway. Oncogene. 2005;24:5510–5520. doi: 10.1038/sj.onc.1208693. [DOI] [PubMed] [Google Scholar]
- 71.Motta MC, Divecha N, Lemieux M, Kamel C, Chen D, Gu W, Bultsma Y, McBurney M, Guarente L. Mammalian SIRT1 represses forkhead transcription factors. Cell. 2004;116:551–563. doi: 10.1016/s0092-8674(04)00126-6. [DOI] [PubMed] [Google Scholar]
- 72.Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y, Tran H, Ross SE, Mostoslavsky R, Cohen HY, et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science. 2004;303:2011–2015. doi: 10.1126/science.1094637. [DOI] [PubMed] [Google Scholar]
- 73.van der Horst A, Tertoolen LG, de Vries-Smits LM, Frye RA, Medema RH, Burgering BM. FOXO4 is acetylated upon peroxide stress and deacetylated by the longevity protein hSir2 (SIRT1) J. Biol. Chem. 2004;279:28873–28879. doi: 10.1074/jbc.M401138200. [DOI] [PubMed] [Google Scholar]
- 74.Kobayashi Y, Furukawa-Hibi Y, Chen C, Horio Y, Isobe K, Ikeda K, Motoyama N. SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int. J. Mol. Med. 2005;16:237–243. [PubMed] [Google Scholar]
- 75.Ford J, Jiang M, Milner J. Cancer-specific functions of SIRT1 enable human epithelial cancer cell growth and survival. Cancer Res. 2005;65:10457–10463. doi: 10.1158/0008-5472.CAN-05-1923. [DOI] [PubMed] [Google Scholar]
- 76.Ohsawa S, Miura M. Caspase-mediated changes in Sir2α during apoptosis. FEBS Lett. 2006;580:5875–5879. doi: 10.1016/j.febslet.2006.09.051. [DOI] [PubMed] [Google Scholar]
- 77.Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW. Modulation of NFκB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004;23:2369–2380. doi: 10.1038/sj.emboj.7600244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gao F, Cheng J, Shi T, Yeh ET. Neddylation of a breast cancer-associated protein recruits a class III histone deacetylase that represses NFκB-dependent transcription. Nat. Cell Biol. 2006;8:1171–1177. doi: 10.1038/ncb1483. [DOI] [PubMed] [Google Scholar]
- 79.Ashraf N, Zino S, Macintyre A, Kingsmore D, Payne AP, George WD, Shiels PG. Altered sirtuin expression is associated with node-positive breast cancer. Br. J. Cancer. 2006;95:1056–1061. doi: 10.1038/sj.bjc.6603384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.de Nigris F, Cerutti J, Morelli C, Califano D, Chiariotti L, Viglietto G, Santelli G, Fusco A. Isolation of a SIR-like gene, SIR-T8, that is overexpressed in thyroid carcinoma cell lines and tissues. Br. J. Cancer. 2002;86:917–923. doi: 10.1038/sj.bjc.6600156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Frye R. “SIRT8” expressed in thyroid cancer is actually SIRT7. Br. J. Cancer. 2002;87:1479. doi: 10.1038/sj.bjc.6600635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kataoka H, Bonnefin P, Vieyra D, Feng X, Hara Y, Miura Y, Joh T, Nakabayashi H, Vaziri H, Harris CC, Riabowol K. ING1 represses transcription by direct DNA binding and through effects on p53. Cancer Res. 2003;63:5785–5792. [PubMed] [Google Scholar]
- 83.Fu M, Liu M, Sauve AA, Jiao X, Zhang X, Wu X, Powell MJ, Yang T, Gu W, Avantaggiati ML, et al. Hormonal control of androgen receptor function through SIRT1. Mol. Cell. Biol. 2006;26:8122–8135. doi: 10.1128/MCB.00289-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yang Y, Hou H, Haller EM, Nicosia SV, Bai W. Suppression of FOXO1 activity by FHL2 through SIRT1-mediated deacetylation. EMBO J. 2005;24:1021–1032. doi: 10.1038/sj.emboj.7600570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hiratsuka M, Inoue T, Toda T, Kimura N, Shirayoshi Y, Kamitani H, Watanabe T, Ohama E, Tahimic CG, Kurimasa A, Oshimura M. Proteomics-based identification of differentially expressed genes in human gliomas: down-regulation of SIRT2 gene. Biochem. Biophys. Res. Commun. 2003;309:558–566. doi: 10.1016/j.bbrc.2003.08.029. [DOI] [PubMed] [Google Scholar]
- 86.Dryden SC, Nahhas FA, Nowak JE, Goustin AS, Tainsky MA. Role for human SIRT2 NAD-dependent deacetylase activity in control of mitotic exit in the cell cycle. Mol. Cell. Biol. 2003;23:3173–3185. doi: 10.1128/MCB.23.9.3173-3185.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Inoue T, Hiratsuka M, Osaki M, Yamada H, Kishimoto I, Yamaguchi S, Nakano S, Katoh M, Ito H, Oshimura M. SIRT2, a tubulin deacetylase, acts to block the entry to chromosome condensation in response to mitotic stress. Oncogene. 2007;26:945–957. doi: 10.1038/sj.onc.1209857. [DOI] [PubMed] [Google Scholar]
- 88.Chen B, Nelson DM, Sadovsky Y. N-myc down-regulated gene 1 modulates the response of term human trophoblasts to hypoxic injury. J. Biol. Chem. 2006;281:2764–2772. doi: 10.1074/jbc.M507330200. [DOI] [PubMed] [Google Scholar]
- 89.Kume S, Haneda M, Kanasaki K, Sugimoto T, Araki S, Isono M, Isshiki K, Uzu T, Kashiwagi A, Koya D. Silent information regulator 2 (SIRT1) attenuates oxidative stress-induced mesangial cell apoptosis via p53 deacetylation. Free Radical Biol. Med. 2006;40:2175–2182. doi: 10.1016/j.freeradbiomed.2006.02.014. [DOI] [PubMed] [Google Scholar]
- 90.Kume S, Haneda M, Kanasaki K, Sugimoto T, Araki SI, Isshiki K, Isono M, Uzu T, Guarente L, Kashiwagi A, Koya D. SIRT1 inhibits TGFβ-induced apoptosis in glomerular mesangial cells via Smad7 deacetylation. J. Biol. Chem. 2006;282:151–158. doi: 10.1074/jbc.M605904200. [DOI] [PubMed] [Google Scholar]
- 91.Alcendor RR, Kirshenbaum LA, Imai S, Vatner SF, Sadoshima J. Silent information regulator 2α, a longevity factor and class III histone deacetylase, is an essential endogenous apoptosis inhibitor in cardiac myocytes. Circ. Res. 2004;95:971–980. doi: 10.1161/01.RES.0000147557.75257.ff. [DOI] [PubMed] [Google Scholar]
- 92.Pillai JB, Isbatan A, Imai S, Gupta MP. Poly(ADP-ribose) polymerase-1-dependent cardiac myocyte cell death during heart failure is mediated by NAD+ depletion and reduced Sir2α deacetylase activity. J. Biol. Chem. 2005;280:43121–43130. doi: 10.1074/jbc.M506162200. [DOI] [PubMed] [Google Scholar]
- 93.Kolthur-Seetharam U, Dantzer F, McBurney MW, de Murcia G, Sassone-Corsi P. Control of AIF-mediated cell death by the functional interplay of SIRT1 and PARP-1 in response to DNA damage. Cell Cycle. 2006;5:873–877. doi: 10.4161/cc.5.8.2690. [DOI] [PubMed] [Google Scholar]
- 94.Chen IY, Lypowy J, Pain J, Sayed D, Grinberg S, Alcendor RR, Sadoshima J, Abdellatif M. Histone H2A.z is essential for cardiac myocyte hypertrophy but opposed by silent information regulator 2α. J. Biol. Chem. 2006;281:19369–19377. doi: 10.1074/jbc.M601443200. [DOI] [PubMed] [Google Scholar]
- 95.Langley E, Pearson M, Faretta M, Bauer UM, Frye RA, Minucci S, Pelicci PG, Kouzarides T. Human SIR2 deacetylates p53 and antagonizes PML/p53-induced cellular senescence. EMBO J. 2002;21:2383–2396. doi: 10.1093/emboj/21.10.2383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Chua KF, Mostoslavsky R, Lombard DB, Pang WW, Saito S, Franco S, Kaushal D, Cheng HL, Fischer MR, Stokes N, et al. Mammalian SIRT1 limits replicative life span in response to chronic genotoxic stress. Cell. Metab. 2005;2:67–76. doi: 10.1016/j.cmet.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 97.Sasaki T, Maier B, Bartke A, Scrable H. Progressive loss of SIRT1 with cell cycle withdrawal. Aging Cell. 2006;5:413–422. doi: 10.1111/j.1474-9726.2006.00235.x. [DOI] [PubMed] [Google Scholar]
- 98.Mostoslavsky R, Chua KF, Lombard DB, Pang WW, Fischer MR, Gellon L, Liu P, Mostoslavsky G, Franco S, Murphy MM, et al. Genomic instability and aging-like phenotype in the absence of mammalian SIRT6. Cell. 2006;124:315–329. doi: 10.1016/j.cell.2005.11.044. [DOI] [PubMed] [Google Scholar]
- 99.Pagans S, Pedal A, North BJ, Kaehlcke K, Marshall BL, Dorr A, Hetzer-Egger C, Henklein P, Frye R, McBurney MW, et al. SIRT1 regulates HIV transcription via Tat deacetylation. PLoS Biol. 2005;3:e41. doi: 10.1371/journal.pbio.0030041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Grubisha O, Rafty LA, Takanishi CL, Xu X, Tong L, Perraud AL, Scharenberg AM, Denu JM. Metabolite of SIR2 reaction modulates TRPM2 ion channel. J. Biol. Chem. 2006;281:14057–14065. doi: 10.1074/jbc.M513741200. [DOI] [PubMed] [Google Scholar]
- 101.Sakamoto J, Miura T, Shimamoto K, Horio Y. Predominant expression of Sir2α, an NAD-dependent histone deacetylase, in the embryonic mouse heart and brain. FEBS Lett. 2004;556:281–286. doi: 10.1016/s0014-5793(03)01444-3. [DOI] [PubMed] [Google Scholar]
- 102.McBurney MW, Yang X, Jardine K, Hixon M, Boekelheide K, Webb JR, Lansdorp PM, Lemieux M. The mammalian SIR2α protein has a role in embryogenesis and gametogenesis. Mol. Cell. Biol. 2003;23:38–54. doi: 10.1128/MCB.23.1.38-54.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Takata T, Ishikawa F. Human Sir2-related protein SIRT1 associates with the bHLH repressors HES1 and HEY2 and is involved in HES1- and HEY2-mediated transcriptional repression. Biochem. Biophys. Res. Commun. 2003;301:250–257. doi: 10.1016/s0006-291x(02)03020-6. [DOI] [PubMed] [Google Scholar]
- 104.Senawong T, Peterson VJ, Leid M. BCL11A-dependent recruitment of SIRT1 to a promoter template in mammalian cells results in histone deacetylation and transcriptional repression. Arch. Biochem. Biophys. 2005;434:316–325. doi: 10.1016/j.abb.2004.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Bae NS, Swanson MJ, Vassilev A, Howard BH. Human histone deacetylase SIRT2 interacts with the homeobox transcription factor HOXA10. J. Biochem. (Tokyo) 2004;135:695–700. doi: 10.1093/jb/mvh084. [DOI] [PubMed] [Google Scholar]
- 106.Borra MT, O’Neill FJ, Jackson MD, Marshall B, Verdin E, Foltz KR, Denu JM. Conserved enzymatic production and biological effect of O-acetyl-ADP-ribose by silent information regulator 2-like NAD+-dependent deacetylases. J. Biol. Chem. 2002;277:12632–12641. doi: 10.1074/jbc.M111830200. [DOI] [PubMed] [Google Scholar]
- 107.Yang SR, Wright J, Bauter M, Seweryniak K, Kode A, Rahman I. Sirtuin regulates cigarette smoke induced pro-inflammatory mediators release via RelA/p65 NF-κB in macrophages in vitro and in rat lungs in vivo. Am. J. Physiol. Lung Cell Mol. Physiol. 2006;292:L567–L576. doi: 10.1152/ajplung.00308.2006. [DOI] [PubMed] [Google Scholar]
- 108.Picard F, Kurtev M, Chung N, Topark-Ngarm A, Senawong T, De Oliveira R. Machado, Leid M, McBurney MW, Guarente L. Sirt1 promotes fat mobilization in white adipocytes by repressing PPARγ. Nature. 2004;429:771–776. doi: 10.1038/nature02583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Qiao L, Shao J. SIRT1 regulates adiponectin gene expression through Foxo1—C/EBPα transcriptional complex. J. Biol. Chem. 2006;281:39915–39924. doi: 10.1074/jbc.M607215200. [DOI] [PubMed] [Google Scholar]
- 110.Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K, et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med. 2002;8:1288–1295. doi: 10.1038/nm788. [DOI] [PubMed] [Google Scholar]
- 111.Yamauchi T, Kamon J, Waki H, Terauchi Y, Kubota N, Hara K, Mori Y, Ide T, Murakami K, Tsuboyama-Kasaoka N, et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001;7:941–946. doi: 10.1038/90984. [DOI] [PubMed] [Google Scholar]
- 112.Zhu M, Miura J, Lu LX, Bernier M, DeCabo R, Lane MA, Roth GS, Ingram DK. Circulating adiponectin levels increase in rats on caloric restriction: the potential for insulin sensitization. Exp. Gerontol. 2004;39:1049–1059. doi: 10.1016/j.exger.2004.03.024. [DOI] [PubMed] [Google Scholar]
- 113.Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P. Nutrient control of glucose homeostasis through a complex of PGC-1α and SIRT1. Nature. 2005;434:113–118. doi: 10.1038/nature03354. [DOI] [PubMed] [Google Scholar]
- 114.Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P, et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell. 2006;127:1109–1122. doi: 10.1016/j.cell.2006.11.013. [DOI] [PubMed] [Google Scholar]
- 115.Moynihan KA, Grimm AA, Plueger MM, Bernal-Mizrachi E, Ford E, Cras-Meneur C, Permutt MA, Imai S. Increased dosage of mammalian Sir2 in pancreatic β cells enhances glucose-stimulated insulin secretion in mice. Cell Metab. 2005;2:105–117. doi: 10.1016/j.cmet.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 116.Bordone L, Motta MC, Picard F, Robinson A, Jhala US, Apfeld J, McDonagh T, Lemieux M, McBurney M, Szilvasi A, et al. Sirt1 regulates insulin secretion by repressing UCP2 in pancreatic β cells. PLoS Biol. 2006;4:e31. doi: 10.1371/journal.pbio.0040031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Kitamura YI, Kitamura T, Kruse JP, Raum JC, Stein R, Gu W, Accili D. FoxO1 protects against pancreatic β cell failure through NeuroD and MafA induction. Cell Metab. 2005;2:153–163. doi: 10.1016/j.cmet.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 118.Hallows WC, Lee S, Denu JM. Sirtuins deacetylate and activate mammalian acetyl-CoA synthetases. Proc. Natl. Acad. Sci. U.S.A. 2006;103:10230–10235. doi: 10.1073/pnas.0604392103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Schwer B, Bunkenborg J, Verdin RO, Andersen JS, Verdin E. Reversible lysine acetylation controls the activity of the mitochondrial enzyme acetyl-CoA synthetase 2. Proc. Natl. Acad. Sci. U.S.A. 2006;103:10224–10229. doi: 10.1073/pnas.0603968103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Afshar G, Murnane JP. Characterization of a human gene with sequence homology to Saccharomyces cerevisiae SIR2. Gene. 1999;234:161–168. doi: 10.1016/s0378-1119(99)00162-6. [DOI] [PubMed] [Google Scholar]
- 121.Yu TT, McIntyre JC, Bose SC, Hardin D, Owen MC, McClintock TS. Differentially expressed transcripts from phenotypically identified olfactory sensory neurons. J. Comp. Neurol. 2005;483:251–262. doi: 10.1002/cne.20429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Araki T, Sasaki Y, Milbrandt J. Increased nuclear NAD biosynthesis and SIRT1 activation prevent axonal degeneration. Science. 2004;305:1010–1013. doi: 10.1126/science.1098014. [DOI] [PubMed] [Google Scholar]
- 123.Chen J, Zhou Y, Mueller-Steiner S, Chen LF, Kwon H, Yi S, Mucke L, Gan L. SIRT1 protects against microglia-dependent amyloid-β toxicity through inhibiting NF-κB signaling. J. Biol. Chem. 2005;280:40364–40374. doi: 10.1074/jbc.M509329200. [DOI] [PubMed] [Google Scholar]
- 124.Qin W, Yang T, Ho L, Zhao Z, Wang J, Chen L, Zhao W, Thiyagarajan M, MacGrogan D, Rodgers JT, et al. Neuronal SIRT1 activation as a novel mechanism underlying the prevention of Alzheimer disease amyloid neuropathology by calorie restriction. J. Biol. Chem. 2006;281:21745–21754. doi: 10.1074/jbc.M602909200. [DOI] [PubMed] [Google Scholar]
- 125.Beal MF. Mitochondria take center stage in aging and neurodegeneration. Ann. Neurol. 2005;58:495–505. doi: 10.1002/ana.20624. [DOI] [PubMed] [Google Scholar]
- 126.St-Pierre J, Drori S, Uldry M, Silvaggi JM, Rhee J, Jager S, Handschin C, Zheng K, Lin J, Yang W, et al. Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell. 2006;127:397–408. doi: 10.1016/j.cell.2006.09.024. [DOI] [PubMed] [Google Scholar]
- 127.Southwood CM, Peppi M, Dryden S, Tainsky MA, Gow A. Microtubule deacetylases, SirT2 and HDAC6, in the nervous system. Neurochem. Res. 2006;32:187–195. doi: 10.1007/s11064-006-9127-6. [DOI] [PubMed] [Google Scholar]
- 128.de la Monte SM, Wands JR. Molecular indices of oxidative stress and mitochondrial dysfunction occur early and often progress with severity of Alzheimer’s disease. J. Alzheimer’s Dis. 2006;9:167–181. doi: 10.3233/jad-2006-9209. [DOI] [PubMed] [Google Scholar]
- 129.Mattiasson G, Shamloo M, Gido G, Mathi K, Tomasevic G, Yi S, Warden CH, Castilho RF, Melcher T, Gonzalez-Zulueta M, et al. Uncoupling protein-2 prevents neuronal death and diminishes brain dysfunction after stroke and brain trauma. Nat. Med. 2003;9:1062–1068. doi: 10.1038/nm903. [DOI] [PubMed] [Google Scholar]
- 130.Conti B, Sugama S, Lucero J, Winsky-Sommerer R, Wirz SA, Maher P, Andrews Z, Barr AM, Morale MC, Paneda C, et al. Uncoupling protein 2 protects dopaminergic neurons from acute 1,2,3,6-methyl-phenyl-tetrahydropyridine toxicity. J. Neurochem. 2005;93:493–501. doi: 10.1111/j.1471-4159.2005.03052.x. [DOI] [PubMed] [Google Scholar]
- 131.Andrews ZB, Diano S, Horvath TL. Mitochondrial uncoupling proteins in the CNS: in support of function and survival. Nat. Rev. Neurosci. 2005;6:829–840. doi: 10.1038/nrn1767. [DOI] [PubMed] [Google Scholar]
- 132.Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG, Zipkin RE, Chung P, Kisielewski A, Zhang LL, et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 2003;425:191–196. doi: 10.1038/nature01960. [DOI] [PubMed] [Google Scholar]
- 133.Jarolim S, Millen J, Heeren G, Laun P, Goldfarb DS, Breitenbach M. A novel assay for replicative lifespan in Saccharomyces cerevisiae. FEMS Yeast Res. 2004;5:169–177. doi: 10.1016/j.femsyr.2004.06.015. [DOI] [PubMed] [Google Scholar]
- 134.Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature. 2004;430:686–689. doi: 10.1038/nature02789. [DOI] [PubMed] [Google Scholar]
- 135.Viswanathan M, Kim SK, Berdichevsky A, Guarente L. A role for SIR-2.1 regulation of ER stress response genes in determining C. elegans life span. Dev. Cell. 2005;9:605–615. doi: 10.1016/j.devcel.2005.09.017. [DOI] [PubMed] [Google Scholar]
- 136.Valenzano DR, Terzibasi E, Genade T, Cattaneo A, Domenici L, Cellerino A. Resveratrol prolongs lifespan and retards the onset of age-related markers in a short-lived vertebrate. Curr. Biol. 2006;16:296–300. doi: 10.1016/j.cub.2005.12.038. [DOI] [PubMed] [Google Scholar]
- 137.Anekonda TS. Resveratrol—a boon for treating Alzheimer’s disease? Brain Res. Brain Res. Rev. 2006;52:316–326. doi: 10.1016/j.brainresrev.2006.04.004. [DOI] [PubMed] [Google Scholar]
- 138.Labinskyy N, Csiszar A, Veress G, Stef G, Pacher P, Oroszi G, Wu J, Ungvari Z. Vascular dysfunction in aging: potential effects of resveratrol, an anti-inflammatory phytoestrogen. Curr. Med. Chem. 2006;13:989–996. doi: 10.2174/092986706776360987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, Prabhu VV, Allard JS, Lopez-Lluch G, Lewis K, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444:337–342. doi: 10.1038/nature05354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Bellizzi D, Rose G, Cavalcante P, Covello G, Dato S, De Rango F, Greco V, Maggiolini M, Feraco E, Mari V, et al. A novel VNTR enhancer within the SIRT3 gene, a human homologue of SIR2, is associated with survival at oldest ages. Genomics. 2005;85:258–263. doi: 10.1016/j.ygeno.2004.11.003. [DOI] [PubMed] [Google Scholar]
- 141.Cruz JC, Tseng HC, Goldman JA, Shih H, Tsai LH. Aberrant Cdk5 activation by p25 triggers pathological events leading to neurodegeneration and neurofibrillary tangles. Neuron. 2003;40:471–483. doi: 10.1016/s0896-6273(03)00627-5. [DOI] [PubMed] [Google Scholar]