Toxicity related to the infusion of dimethylsulfoxide-cryopreserved peripheral blood stem cells (DMSO-PBSC) manifests mostly as cardiovascular side effects. Neurotoxicity1 including transient global amnesia,2,3 seizures,4,5 and stroke6,7 has been reported as a rare complication primarily in adults. In children, data8 are sparse. In light of a recent report implicating morphine co-medication as a major contributing factor,8 we evaluated retrospectively our own data base, including all infusions of DMSO-PBSC applied in our pediatric center between January 1st 2002 and December 31st 2008. We report on 2 incidences of transient loss of consciousness following 131 infusions of DMSO-PBSC.
Case 1
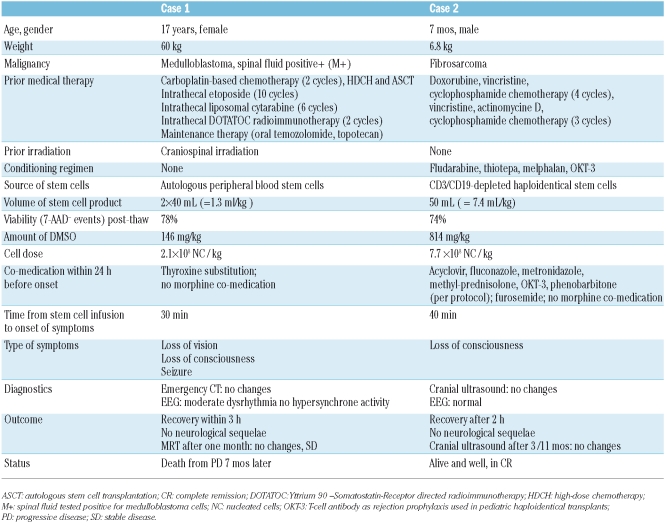
A 17-year old female suffering from recurrent medulloblastoma was admitted for prolonged pancytopenia due to topotecan maintenance therapy. Throughout her medical history, this patient had received intensive therapy, including high-dose chemotherapy and autologous peripheral blood stem cell transplantation, craniospinal irradiation, as well as CNS-directed therapy, namely intrathecal chemotherapy and radioimmunotherapy (Table 1). Thirty minutes after receiving a cryopreserved autologous stem cell boost, the patient initially complained of bilateral loss of vision. Fifteen minutes later, she became unconscious (Glasgow coma scale 3/15). The patient’s cardiorespiratory condition was stable. During transport to emergency CT, she developed one short episode of clonic seizure restricted to both arms, which responded promptly to dexamethasone. Her CT scan revealed no signs of ischemia or bleeding, her EEG displayed moderate dysrhythmia. Cerebrospinal fluid analysis revealed normal lactate, protein and glucose, and no leukocytes or malignant cells. Likewise, serum electrolytes, glucose, LDH were within normal ranges. The patient remained unconscious for approximately two hours. When she woke up, she was disoriented for an additional hour. Three hours after the incident, she had completely recovered, but showed retrograde amnesia for the complete episode. She was discharged the following day without any neurological symptoms. Following the stem cell boost, she recovered from neutropenia and thrombocytopenia after 14 days. Follow-up MRT one month later showed no specific findings.
Table 1.
Patients’ characteristics, medical history, clinical presentation and outcome.
Case 2
A 7-month old male infant diagnosed with fibrosarcoma of his lower back who had responded well to preceding chemotherapy, but still had non-operable residual disease extending into his spinal canal was considered eligible for haploidentical stem cell transplant as consolidation therapy after extensive consultation and written parental consent. Following the standard conditioning regimen of fludarabin, thiotepa and melphalan, he was transplanted with a cryopreserved CD3/CD19-depleted haploidentical stem cell graft from his father. Forty minutes following infusion, he suddenly lost consciousness. His cardiorespiratory condition was stable. Serum electrolytes and blood gases were within normal ranges. Emergency cranial ultrasound revealed no pathology, EEG was normal. In addition, he showed no clinical signs of seizure. One hour after the onset of symptoms, the infant gradually regained consciousness. Two hours after the onset, he had completely recovered.
DMSO is currently the only sufficiently validated reagent for cryopreservation of hematopoietic stem cells; standard practice in most centers is cryoconservation with 10% DMSO.9 However, stem cell washing10 before infusion or cryopreservation with DMSO mixed with hydroxyethyl starch11 may represent alternative approaches. In children, few data on neurotoxicity following infusion of DMSO-PBSC are available.8 The incidence in our series of consecutive stem cell infusions was found to be 2/131 (1.5 %). The only other report in children8 thus far had implicated morphine co-medication as contributing factor which can be ruled out in our 2 patients. Furthermore, post-thaw viability of both products was within the expected range (78% and 74%, respectively) excluding the assumption that an inadequately high number of dead or apoptotic cells might have contributed to neurotoxicity. Other triggers such as disturbances of electrolytes, glucose or blood gases, intravascular activation of coagulation could be excluded in both patients. The time to onset was within the first 60 min after infusion, in accordance with the majority of reports in adults.1,2,3,5
Toxicology analysis of DMSO in rodents indicates a dose-dependent increase in neurotoxicity.12 The first patient who had received only a relatively small amount of DMSO (146 mg/kg), may have been more sensitive to the neurotoxic effects due to her brain tumor and to prior intensive CNS-directed therapy. The second patient, an infant without prior CNS history, developed symptoms at a dose level of 814 mg/kg, which is at the higher end of the applicable dose range.2 Certainly, children with a body weight of 10kg and below are at risk for receiving higher dosage of DMSO. As a consequence of the observed toxicities, our institutional policy has changed to keep the maximum volume applied at 4 mL/kg cryopreserved stem cells containing 10% DMSO and to recommend splitting doses on subsequent infusion days if necessary.
References
- 1.Mueller LP, Theurich S, Christopeit M, Grothe W, Muetherig A, Weber T, et al. Neurotoxicity upon infusion of dimethylsulfoxide-cryopreserved peripheral blood stem cells in patients with and without pre-existing cerebral disease. Eur J Haematol. 2007;78:527–31. doi: 10.1111/j.1600-0609.2007.00851.x. [DOI] [PubMed] [Google Scholar]
- 2.Junior AM, Arrais CA, Saboya R, Velasques RD, Junqueira PL, Dulley FL. Neurotoxicity associated with dimethylsul-foxide-preserved hematopoietic progenitor cell infusion. Bone Marrow Transplant. 2008;41:95–6. doi: 10.1038/sj.bmt.1705883. [DOI] [PubMed] [Google Scholar]
- 3.Otrock ZK, Beydoun A, Barada WM, Masroujeh R, Hourani R, Bazarbachi A. Transient global amnesia associated with the infusion of DMSO-cryopreserved autologous blood stem cells. Haematologica. 2008;93:e36–7. doi: 10.3324/haematol.12249. [DOI] [PubMed] [Google Scholar]
- 4.Bauwens D, Hantson P, Laterre PF, Michaux L, Latinne D, De Tourtchninoff M, et al. Recurrent seizure and sustained encephalopathy associated with dimethylsulfoxide-preserved stem cell infusion. Leuk Lymphoma. 2005;46:1671–4. doi: 10.1080/10428190500235611. [DOI] [PubMed] [Google Scholar]
- 5.Hequet O, Dumontet C, El Jaafari-Corbin A, Salles G, Espinouse D, Arnaud P, et al. Epileptic seizures after autologous peripheral blood progenitor infusion in a patient treated with high-dose chemotherapy for myeloma. Bone Marrow Transplant. 2002;29:544. doi: 10.1038/sj.bmt.1703383. [DOI] [PubMed] [Google Scholar]
- 6.Windrum P, Morris TCM. Severe neurotoxicity because of dimethyl sulphoxide following peripheral blood stem cell transplantation. Bone Marrow Transplant. 2003;31:315. doi: 10.1038/sj.bmt.1703848. [DOI] [PubMed] [Google Scholar]
- 7.Hoyt R, Szer J, Grigg A. Neurological events associated with the infusion of cryopreserved bone marrow and/or peripheral blood progenitor cells. Bone Marrow Transplant. 2000;25:1285–7. doi: 10.1038/sj.bmt.1702443. [DOI] [PubMed] [Google Scholar]
- 8.Caselli D, Tintori V, Messeri A, Frenos S, Bambi F, Arico M. Respiratory depression and somnolence in children receiving dimethylsulfoxide and morphine during hematopoietic stem cell transplantation. Haematologica. 2009;94:152–3. doi: 10.3324/haematol.13828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Windrum P, Morris TC, Drake MB, Niederwieser D, Ruutu T EBMT Chronic Leukaemia Working Party Complications Subcommittee. Variation in dimethyl sulfoxide use in stem cell transplantation: a survey of EBMT centres. Bone Marrow Transplant. 2005;36:601–3. doi: 10.1038/sj.bmt.1705100. [DOI] [PubMed] [Google Scholar]
- 10.Syme R, Bewick M, Stewart D, Porter K, Chadderton T, Glück S. The role of depletion of dimethyl sulfoxide before autografting: on hematologic recovery, side effects, and toxicity. Biol Blood Marrow Transplant. 2004;10:135–41. doi: 10.1016/j.bbmt.2003.09.016. [DOI] [PubMed] [Google Scholar]
- 11.Rowley SD, Feng Z, Chen L, Holmberg L, Heimfeld S, MacLeod B, Bensinger WI. A randomized phase III clinical trial of autologous blood stem cell transplantation comparing cryopreservation using dimethylsulfoxide vs dimethyl-sulfoxide with hydroyethylstarch. Bone Marrow Transplant. 2003;31:1043–51. doi: 10.1038/sj.bmt.1704030. [DOI] [PubMed] [Google Scholar]
- 12.Cavaletti G, Oggioni N, Sala F, Pezzoni G, Cavalletti E, Marmiroli P, et al. Effect on the peripheral nervous system of systemically administered dimethysulfoxide in the rat: a neurophysiological and pathological study. Toxicology Lett. 2000;118:103–7. doi: 10.1016/s0378-4274(00)00269-1. [DOI] [PubMed] [Google Scholar]