Abstract
Priming is an unconscious (nondeclarative) form of memory whereby identification or production of an item is improved by an earlier encounter. It has been proposed that declarative memory and priming might be related—for example, that conceptual priming results in more fluent processing, thereby providing a basis for familiarity judgments. In two experiments, we assessed conceptual priming and recognition memory across a 5-min interval in 5 memory-impaired patients. All patients exhibited fully intact priming in tests of both free association (study tent; at test, provide an association to canvas) and category verification (study lemon; at test, decide: Is lemon a type of fruit?). Yet the 2 most severely amnesic patients performed at chance on matched tests of recognition memory. These findings count against the notion that conceptual priming provides feelings of familiarity that can support accurate recognition judgments. We suggest that priming is inaccessible to conscious awareness and does not influence declarative memory.
Declarative memory supports the capacity for conscious recollection of facts and events and can be contrasted with a collection of non-declarative memory abilities, including habits and skills, simple forms of conditioning, and other ways by which experience can change how people interact with the world (Milner, Squire, & Kandel, 1998). One well-studied example of nondeclarative memory is the phenomenon of priming, whereby an encounter with an item improves the ability to identify or produce the same item (perceptual priming) or a related item (conceptual or semantic priming; Schacter & Buckner, 1998; Tulving & Schacter, 1990).
There has been extended exploration of the possibility that priming and declarative memory might be related in some way. For example, it has been proposed that recognition memory may be supported in part by the fluency with which items are processed. That is, priming may result in improved, more fluent processing that provides a basis for judging items as familiar (Jacoby & Dallas, 1981; Verfaellie & Keane, 2002; Yonelinas, Regehr, & Jacoby, 1995). This idea has been addressed most thoroughly in the case of perceptual (or repetition) priming, which is demonstrated in tasks such as word identification and word-stem completion. In perceptual priming, a specific perceptual operation is enhanced, and the priming effect does not depend on carrying out semantic or elaborative encoding at the time of study.
Three studies are particularly relevant to this issue. First, in undergraduate volunteers, familiarity judgments and repetition priming (word identification or word-stem completion) were affected differently by manipulations of perceptual and conceptual processing. Thus, recognition and repetition priming were sharply dissociable (Wagner, Gabrieli, & Verfaellie, 1997). Second, the severely amnesic patient E.P. was fully intact at perceptual priming, despite performing at chance on matched tests of recognition memory (Hamann & Squire, 1997). Third, in five additional experiments, E.P. performed at chance on recognition memory tasks, despite instructions that encouraged him to use familiarity and even when he produced the studied items in a priming task that was given just prior to the recognition test (Stark & Squire, 2000). These results count against the idea that perceptual priming makes a noticeable contribution to declarative memory. Thus, perceptual priming does not appear to be a source for feelings of familiarity that can accurately guide recognition memory judgments.
An alternative, perhaps more likely, possibility is that recognition memory is supported not by perceptual fluency but by conceptual priming, which results in improved processing of the meaning of items (Verfaellie & Keane, 2002; Wagner et al., 1997). Indeed, conceptual priming shares more characteristics with declarative memory than does perceptual priming. Accordingly, conceptual priming and recognition could rely on common mechanisms (Yonelinas, 2002).
Studies of amnesic patients have demonstrated intact conceptual priming in tasks of free association (e.g., study cry; at test, provide an associate for baby; Carlesimo, 1994; Shimamura & Squire, 1984; Vaidya, Gabrieli, Keane, & Monti, 1995) and production of category exemplars (e.g., study strawberry; at test, generate examples of fruits; Graf, Squire, & Mandler, 1984; Keane et al., 1997). Electrophysiological studies of event-related potentials (ERPs) suggest that conceptual priming may be indexed by the N400, which is reduced during word repetition in memory-impaired patients just as it is in control participants (Olichney et al., 2000). However, findings of intact conceptual priming in amnesia are not strong evidence against the idea that conceptual priming and recognition memory are related, because virtually all the memory-impaired patients tested to date have some residual declarative memory. When patients have intact conceptual priming and impaired, albeit above-chance recognition memory scores, conceptual priming could support some or all of the residual capacity for recognition.
It is also true that not all studies have found intact conceptual priming in amnesia (Cermak, Hill, & Wong, 1998; Cermak & Wong, 1998; Keane et al., 1997). However, in these cases, it is difficult to know whether the control participants may have outperformed the patients because they engaged in conscious (declarative) memory strategies (Schacter & Buckner, 1998; Vaidya, Gabrieli, Demb, Keane, & Wetzel, 1996).
The present study tested the relationship between conceptual priming and recognition memory using two tests of conceptual priming (free-association priming in Experiment 1 and category-verification priming in Experiment 2) and matched tests of recognition memory. We tested 2 severely amnesic patients (E.P. and G.P.) who have little or no capacity for declarative memory, as well as 3 other patients with less severe amnesia. The question of interest was whether conceptual priming can be fully intact even when recognition memory performance is at chance.
EXPERIMENT 1
Method
Participants
We tested 2 patients (E.P. and G.P., both male) who have severe amnesia and large medial temporal lobe (MTL) lesions as the result of herpes simplex encephalitis (E.P. in 1992 and G.P. in 1987). Three additional patients (2 males, 1 female) with less severe amnesia were also tested; their damage is thought to be limited to the hippocampal region (dentate gyrus, cell fields of the hippocampus proper, and subicular complex; Table 1). A.B. became amnesic in 1976 following an anoxic episode associated with cardiac arrest. L.J. became amnesic in 1988 during a 6-month period with no known etiology. Her memory impairment has remained stable since that time. G.W. became amnesic in 2001 following a drug overdose and associated respiratory failure. For all 5 patients, immediate and delayed (12-min) recall of a short prose passage (Gilbert, Levee, & Catalano, 1968) averaged 4.2 and 0 segments, respectively. Six healthy male volunteers served as a control group (mean age = 69.3 years, range = 55–82; mean education = 12.7 years, range = 12–16). Their immediate and delayed prose recall averaged 7.2 and 6.2, respectively.
TABLE 1.
Characteristics of the Amnesic Patients
| WMS–R |
|||||||||
|---|---|---|---|---|---|---|---|---|---|
| Patient | Group | Year of birth | Education (years) | WAIS–III IQ | Attention | Verbal | Visual | General | Delay |
| E.P. | MTL | 1922 | 12 | 98 | 94 | 59 | 82 | 68 | 56 |
| G.P. | MTL | 1946 | 16 | 98 | 102 | 79 | 62 | 66 | <50 |
| A.B. | H | 1937 | 20 | 107 | 87 | 62 | 72 | 54 | <50 |
| L.J. | H | 1937 | 12 | 101 | 105 | 83 | 60 | 69 | <50 |
| G.W. | H | 1959 | 12 | 108 | 105 | 67 | 86 | 70 | <50 |
Note. The Wechsler Adult Intelligence Scale–III (WAIS–III) and the Wechsler Memory Scale–Revised (WMS–R) yield mean scores of 100 in the normal population, with a standard deviation of 15. MTL=medial temporal lobe lesion; H=damage thought to be limited to the hippocampal region.
For E.P. and G.P., estimates of MTL damage were based on quantitative analysis of magnetic resonance images (MRIs) from the patients and 4 control subjects for each patient, following published procedures for segmenting the medial temporal lobe (Amaral & Insausti, 1990; Insausti, Insausti, Sobreviela, Salinas, & Martinez-Penuala, 1998; Insausti, Juottonen, et al., 1998; also see Gold & Squire, in press). These measurements are based on improved techniques and on more control subjects than available previously. Accordingly, the estimates presented here are similar but not identical to what has been reported previously (Levy, Bayley, & Squire, 2004; Schmolck, Kensinger, Corkin, & Squire, 2002; Stefanacci, Buffalo, Schmolck, & Squire, 2000). Briefly, E.P.'s lesion extends 7 cm caudally from the temporal pole bilaterally and includes all of the amygdala and all of the hippocampal region except for a small tag of abnormally appearing vestigial tissue that comprises approximately 3% of hippocampal volume. In addition, the damage includes all the entorhinal cortex, all the perirhinal cortex, and much of the parahippocampal cortex (about 70% on the left and 71% on the right). The lesion also extends laterally to include the rostral fusiform gyrus (about 46% on the left and 71% on the right). The lateral temporal lobe (inferior, middle, and superior temporal gyri) is of normal size (within 7% of mean control volume). Finally, the insula is reduced in size (about 38% on the left and 36% on the right).
G.P.'s lesion is also primarily medial temporal, but extends more laterally than E.P.'s lesion. The damage extends through the anterior 7 cm of the left temporal lobe and the anterior 6 cm of the right temporal lobe. The damage includes bilaterally all of the amygdala, all of the hippocampal region (except for 7% on the right), all the entorhinal cortex, all the perirhinal cortex, and much of the parahippocampal cortex (about 87% on the left and 55% on the right). The damage also extends into the rostral fusiform gyrus (about 29% on the left and 48% on the right). The lateral temporal lobe is also reduced in volume by about 14% on the left and 12% on the right, and the insula is reduced in volume (about 76% on the left and 39% on the right).
For 2 of the 3 other amnesic patients, new estimates of MTL damage were obtained using recent MRI scans and a larger control group than was available previously. Relative to the age- and gender-matched control group (12 females and 19 males), L.J. and G.W. have an average bilateral reduction in hippocampal size of 46% and 48%, respectively. The parahippocampal gyrus is relatively unaffected (−8% and +112%). The 3rd patient (A.B.) is ineligible for MRI but is thought to have hippocampal damage on the basis of etiology (anoxia) and a neurological examination indicating circumscribed amnesia. In addition, high-resolution computed tomography (CT) images obtained in 2001 were consistent with restricted damage to the hippocampal region (Schmolck et al., 2002).
Materials
Twelve lists of 12 paired associates were constructed, such that each pair consisted of a cue word followed by its associate (e.g., strap-belt, noon-lunch, knit-yarn, quiet-noisy, canvas-tent). According to data from a large normative sample (Nelson, McEvoy, & Schreiber, 1998), each associate was produced as the first associate to its cue word by about 20% of participants. Eight of the lists provided studied and nonstudied words for the four free-association tests, and four of the lists were used for the four cued-recall tests. The assignment of lists to these three conditions (studied and nonstudied words for free association and studied words for cued recall) was balanced across participants, and the order of the words in each list was randomized each time the list was used.
For the four recognition memory tests, 8 different lists of 12 words each were constructed. The words were matched to the words in the 12 paired-associate lists according to frequency (Kucera & Francis, 1967), proportion of concrete and abstract words, and part of speech. Four of these lists provided study words, and four lists provided foils.
Procedure
All 12 tests (4 free association, 4 cued recall, and 4 recognition) began with a study phase of 16 words (12 target words preceded and followed by 2 filler words to reduce primacy and recency effects). Participants were instructed to read each word aloud from a computer screen and to indicate by button press (on a scale from 1 to 5) how much they liked each word. Presentation of the words was self-paced. The procedure was then repeated so that the study list was presented a total of two times. After a 5-min, conversation-filled interval, the test phase was presented on a computer screen.
Free Association
In each free-association test phase, 24 words were presented. Twelve of these were cue words for previously studied associates (e.g., the word belt had been studied and now the word strap was presented), and the other 12 words were cue words for associates that had not been studied. Participants were instructed to say the first word that came to mind in response to each cue word. Four such tests were given in two sessions on 2 different days.
Cued Recall
The cued-recall tests were given in two sessions on separate days, alternating with the days on which the free-association tests were given. In the test phase for cued recall, 12 cue words for previously studied associates were presented. Participants were told that the words they would see were hints to help them recollect the words they had seen a few minutes earlier. They were asked to try to recall the studied words and to guess if necessary.
Recognition Memory
The four recognition tests were given on a separate day after the other tests were completed. In each test phase, 24 words were presented; 12 of these words had been studied and 12 had not. Participants indicated by button press (yes/no) whether each word had been presented previously.
Results
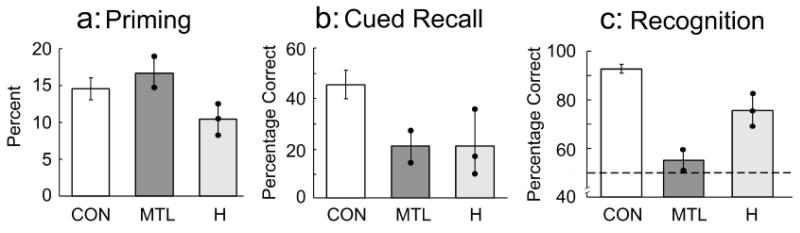
When asked to free-associate to a cue word (Fig. 1a), participants responded with words they had seen earlier substantially more frequently than would be expected if the words had not been presented (MTL patients: 21.9% for studied items vs. 5.2% for baseline items; hippocampal patients: 22.2% for studied items vs. 11.8% for baseline items; control group: 25.3% for studied items vs. 10.8% for baseline items). The scores for both baseline items and studied items were similar across groups (Fs < 1.0), as was the magnitude of priming (hippocampal: 10.4%, MTL: 16.7%, control: 14.6%). E.P.'s priming score was 18.7%, and G.P.'s score was 14.6%. An analysis of variance (Studied vs. Baseline Items × Group) yielded an effect of priming, F(1, 8)=160.1, p < .001, ηp2=.95, but no effect of group (p=.7) and no interaction (p=.14).
Fig. 1.
Free-association priming, cued recall, and recognition memory in Experiment 1. Results are shown separately for the control group (CON, n=6), amnesic patients with large medial temporal lobe lesions (MTL, n=2), and amnesic patients with lesions thought to be limited to the hippocampal region (H, n=3). Participants were given each of the three tests four different times, using similar materials. The measure of priming (a) is the difference between the percentage of studied and unstudied words produced at test in response to an associate of each word. The measure of cued recall (b) is the percentage of studied words recalled in response to an associate of each studied word. The measure of recognition memory (c) is the percentage of correct responses (hits plus correct rejections) for yes/no recognition. The data points for the MTL and H groups show the scores of individual patients averaged across the four separate tests. The dashed line in (c) indicates chance performance. Brackets show standard errors of the mean.
On the cued recall test (Fig. 1b), the control group performed markedly better than the patients (44.8% correct vs. 20.8% correct), t(9)=3.24, p < .01. Indeed, the cued-recall score for the 5 MTL and hippocampal patients was not significantly better than their priming score (20.8% vs. 12.9%), t(4)=1.54, p > .20. In the cued-recall task, guessing was encouraged (as in the free-association task), and primed words not available to declarative memory could be produced (also see Graf et al., 1984). The 5 patients were also markedly impaired at recognition memory (Fig. 1c). Notably, patient E.P. performed at chance (51.0% correct, 60.4% hit rate, d′=0.05). G.P. performed better, but also not above chance for either percentage correct or d′ (59.4% correct, 93.8% hit rate, d′=0.68), ts(3) < 1.7, ps > .18. The hippocampal patients performed above chance levels (75.7% correct, 80.5% hit rate, d′=1.89), ts(2) > 4.12, ps < .06, but were nevertheless impaired relative to the control group (92.8% correct, 88.4% hit rate, d′=3.25), ts(7) > 2.40, ps < .05.
These results demonstrate a striking dissociation between intact conceptual priming and severely impaired cued recall and recognition memory. Thus, patients E.P. and G.P. could not recognize as familiar the words they had read 5 min earlier, but they tended to produce these words when they were given cue words and asked to produce associates.
EXPERIMENT 2
Experiment 2 compared recognition memory performance with performance on a second test of conceptual priming (category-verification priming). Whereas, the free-association test in Experiment 1 assessed priming by measuring word production, the category-verification test assessed priming by measuring response time.
Method
Participants
The 5 patients and the 6 control subjects from Experiment 1 also participated in Experiment 2, along with 6 additional control subjects (4 males, 2 females). Together, the 12 control subjects averaged 68.8 years of age and 13.7 years of education. Their immediate and delayed (12-min) recall of a prose passage averaged 7.2 and 6.2 segments, respectively.
Materials
Four low-frequency exemplars for each of 24 categories were selected, on the basis of material collected by Yoon et al. (2002). Participants contributing to that database were asked to list 5 exemplars from these categories. The items used in the current experiment were ones rarely included in those lists (<1% probability; e.g., fruit: apricot, lemon, fig, blueberry; bird: falcon, peacock, raven, vulture). The 24 categories and their 96 exemplars provided study words and nonstudied words for four tests of category-verification priming. Thus, each test consisted of 12 words from 6 categories for the study phase and 24 words from the same 6 categories for the test phase. The order of the words in each list was randomized each time the list was used. Four recognition memory tests were also constructed, as in Experiment 1, using words matched to the category exemplars.
Procedure
As in Experiment 1, all eight tests (four category verification and four recognition) began with a study phase in which 16 words were presented (12 target words preceded and followed by 2 filler words). Participants read each word aloud and made a liking judgment (scale from 1 to 5). Each list was presented twice, and the test phase followed after 5 min.
For the test phase of the category-verification tests, reaction time and accuracy data were collected for 24 yes/no questions. Half of the questions concerned words that had been presented, and half concerned nonstudied words (e.g., Is apricot a type of fruit? Is vulture a type of ship?). The correct answer was “yes” for half the questions and “no” for half the questions. (The category names for the “no” questions came from the same database—Yoon et al., 2002—as the other category names but were not used elsewhere in our tests.) Participants were instructed to answer each question as quickly as possible with a button push (“yes” or “no”). The four tests were given in two sessions on 2 different days.
Finally, the four recognition tests were given on a separate day after the other tests were completed. Testing followed exactly the procedure of Experiment 1.
Results
On the category-verification tests (Fig. 2a), priming was evident as an increase in the speed with which participants answered questions about category exemplars that they had studied, relative to the speed with which they answered questions about nonstudied items (MTL patients: 2,142 ms vs. 2,300 ms; hippocampal patients: 1,819 ms vs. 1,975 ms; control group: 1,781 ms vs. 1,945 ms). The response times for both nonstudied and studied items were similar across groups (F < 1.0), as was the magnitude of priming (hippocampal: 156 ms, MTL: 158 ms, control: 164 ms). E.P.'s priming score was 155 ms, and G.P.'s priming score was 160 ms. An analysis of variance (Studied vs. Nonstudied Items × Group) yielded an effect of priming, F(1, 14)=14.6, p < .01, ηp2=.51, but no effect of group (p=.9) and no interaction (p=.8).
Fig. 2.
Category-verification priming and recognition memory in Experiment 2. Results are shown separately for the control group (CON, n=12), amnesic patients with large medial temporal lobe lesions (MTL, n=2), and amnesic patients with lesions thought to be limited to the hippocampal region (H, n=3). Participants were given each of the two tests four different times, using similar materials. The measure of priming in (a) is response time for studied words minus response time for unstudied words. Panel (b) shows the priming score from (a) divided by the response time for unstudied words (baseline response time). The measure of recognition memory (c) is the percentage correct responses (hits plus correct rejections) for yes/no recognition. The data points for the MTL and H groups show the scores of individual patients averaged across the four separate tests. The dashed line in (c) indicates chance performance. Brackets show standard errors of the mean.
Because the magnitude of priming in the control group was strongly correlated with the response time to nonstudied items (r=.924), we also calculated the magnitude of priming as a percentage of baseline (unprimed, nonstudied) response time (Fig. 2b). Again, the three groups exhibited a similar magnitude of priming (MTL patients: 7.0%; hippocampal patients: 8.0%; control group: 7.6%), F < 1.0. E.P. and G.P. scored 6.0% and 7.9%, respectively.
Priming was also similar among groups when the data were analyzed separately for test items that should be answered “yes” and test items that should be answered “no.” Across groups, it took longer to answer “no” than to answer “yes” (1,987 ms vs. 1,835 ms), t(16)=4.18, p < .01. Nevertheless, the magnitude of priming was similar across groups for both kinds of test item (p > .14 for all between-groups comparisons).
Occasionally, participants made errors in answering the yes/no questions, perhaps because some of the low-frequency items (e.g., pewter, mutton) were unfamiliar. The control groups averaged 2.9 errors across all four tests, the hippocampal group averaged 1.3 errors, and the MTL group averaged 8.5 errors. The larger number of errors in the MTL group may reflect the mild impairment in semantic knowledge that has been described for these 2 patients (Schmolck et al., 2002) and the fact that both patients have some damage to antero-lateral temporal cortex. In any case, priming was also similar across the three groups when the trials on which errors occurred were eliminated from the data analysis (MTL group: 139 ms; hippocampal group: 156 ms; control group: 140 ms; p > .9 for all between-groups comparisons).
To ensure that the observed priming effect did not depend on the particular combination of studied and nonstudied words that were used, we gave the same four priming tests to 6 new control participants (mean age=68.0 years; mean education=15.8 years) but with the studied and nonstudied words reversed. Robust priming was observed (1,700 ms for studied words and 1,913 ms for nonstudied words), t(5)=4.3, p < .01, and the magnitude of priming (213 ms) was similar to what was observed for the 12 control participants in Figure 2a, t(16)=0.8, p=.5.
Although both patient groups exhibited intact priming, they were impaired at recognition memory (Fig. 2c). Indeed, the recognition memory results closely matched the findings from Experiment 1. Accuracy was 96.0% for the control group (96.4% hit rate, d′=3.43), and the patients in the MTL group performed near chance (E.P.: 46.9% correct, 62.5% hit rate, d′=−0.17; G.P.: 59.4% correct, 77.1% hit rate, d′=0.53). G.P.'s recognition scores were marginally above chance, for both percentage correct and d′, ts(3) > 2.8, ps < .07. Finally, the hippocampal group performed well above chance (82.3% correct, 84.7% hit rate, d′=2.25), ts(2) > 8.2, ps < .02, but less well than the control group, ts(13) > 3.0, ps < .05.
DISCUSSION
Five amnesic patients were given tests of conceptual priming and cued recall, along with matched tests of recognition memory. Across eight different priming tests (four free-association tests and four category-verification tests), the patients exhibited fully intact priming across a 5-min study-test interval. Indeed, each of the 5 patients obtained above-baseline priming scores on seven of the eight priming tests that were given (p=.035). The most telling results involved patients E.P. and G.P., who have large MTL lesions and little or no capacity for recognition memory. Both E.P. and G.P. exhibited robust priming performance on the two kinds of priming tests, and their priming scores closely matched the priming scores obtained by the control group (Figs. 1 and 2). Yet, despite fully intact priming, neither E.P. nor G.P. performed measurably above chance (50%) on the recognition tests (E.P.: 49.0%; G.P.: 59.4%). In short, conceptual priming was intact, but no declarative memory could be detected.
These results count against the idea that conceptual priming is a source of familiarity that can be used in making accurate recognition memory judgments. Instead, the evidence favors the idea that priming and recognition memory are independent phenomena. If either repetition priming or conceptual priming contributes to recognition memory judgments on typical recognition memory tasks, and if both forms of priming are intact in amnesia, then recognition performance should be above chance even in severe cases of amnesia. Yet patient E.P. has fully intact repetition priming (Hamann & Squire, 1997; Stark & Squire, 2000) and fully intact conceptual priming (this study), but in the many traditional tests of recognition memory he has been given (this study and Stefanacci et al., 2000), he has performed no better than chance.
In the cued-recall test (Experiment 1), the patients were markedly impaired relative to the control group, and their cued-recall score was similar to their priming score (Figs. 1a and 1b). This result is reminiscent of the finding that amnesic patients obtained similar scores on tests of cued recall and word-stem completion priming (Graf et al., 1984). The cued-recall performance of the patients was impaired relative to that of the control group, but their word-stem completion was intact. In both studies, it seems reasonable to suppose that cued-recall scores matched the priming scores because participants were instructed to guess when they could not recall words from the study list. These instructions enabled the patients to produce primed words as responses. Thus, the scores obtained by patients on these cued-recall tests do not reflect successful cued-recall performance, but are better viewed as the minimum scores to be expected given that the instructions encouraged the production of primed words.
The conclusion that priming and accuracy of recognition memory performance are independent is not inconsistent with the finding that the fluency with which an item is processed (e.g., the time needed to identify an item) can influence whether healthy control participants (Johnston, Dark, & Jacoby, 1985; Rajaram & Geraci, 2000; Whittlesea, 1993) or amnesic patients (Verfaellie & Cermak, 1999) will endorse an item as familiar. This effect is similar for studied and nonstudied items and appears to reflect a change in the criterion by which recognition decisions are made (i.e., a shift in bias), not a change in recognition accuracy. We are unaware of evidence that either perceptual or conceptual priming contributes to the accuracy of recognition memory.
Neuroimaging studies of perceptual and conceptual priming suggest that priming is typically associated with a reduction in activation for primed items relative to unprimed items (Schacter & Buckner, 1998). In the case of perceptual priming of visual stimuli, reduced activation is most evident in extrastriate visual cortex. In the case of conceptual priming, prominent reductions in activation are evident in left inferior prefrontal cortex and left inferior temporal cortex, regions that are often active during semantic encoding or semantic processing (Buckner, Koutstaal, Schacter, & Rose, 2000; Schacter & Buckner, 1998; Thompson-Schill, D'Esposito, & Kan, 1999; Wagner, Koutstaal, Maril, Schacter, & Buckner, 2000). A dissociation between priming and recognition memory was demonstrated by the finding of a network of brain regions (including lateral and medial parietal cortex, left dorsal middle frontal gyrus, and left anterior prefrontal cortex) that exhibited increased activity associated with successful recognition performance and a different network of brain regions (including left ventral and dorsal inferior frontal gyrus and left temporal cortex) that exhibited reduced activity associated with conceptual priming (Donaldson, Petersen, & Buckner, 2001). Our results extend these by showing that recognition memory can fail altogether, even while conceptual priming is fully intact. We suggest that changes in the cortical regions related to priming are inaccessible to conscious awareness and do not influence the operation of declarative memory.
Acknowledgments
This work was supported by the Medical Research Service of the Department of Veterans Affairs, National Institute of Mental Health Grant MH24600, and the Metropolitan Life Foundation. We thank Peter Bayley, Jeffrey Gold, Jennifer Frascino, and Leah Swalley for assistance.
REFERENCES
- Amaral DG, Insausti R. Hippocampal formation. In: Paxinos G, editor. The human nervous system. Academic Press; San Diego, CA: 1990. pp. 711–755. [Google Scholar]
- Buckner RL, Koutstaal W, Schacter DL, Rose BR. Functional MRI evidence for a role of frontal and inferior temporal cortex in amodal components of priming. Brain. 2000;123:620–640. doi: 10.1093/brain/123.3.620. [DOI] [PubMed] [Google Scholar]
- Carlesimo GA. Perceptual and conceptual priming in amnesic and alcoholic patients. Neuropsychologia. 1994;32:903–921. doi: 10.1016/0028-3932(94)90042-6. [DOI] [PubMed] [Google Scholar]
- Cermak LS, Hill R, Wong B. The effects of spacing and repetition on amnesic patients' performance during perceptual identification, stem completion and category exemplar production. Neuropsychology. 1998;12:65–77. doi: 10.1037//0894-4105.12.1.65. [DOI] [PubMed] [Google Scholar]
- Cermak LS, Wong BM. Amnesic patients' impaired category exemplar production priming. Journal of the International Neuropsychological Society. 1998;4:576–583. doi: 10.1017/s1355617798466062. [DOI] [PubMed] [Google Scholar]
- Donaldson DI, Petersen SE, Buckner RL. Dissociating memory retrieval processes using fMRI: Evidence that priming does not support recognition memory. Neuron. 2001;31:1047–1059. doi: 10.1016/s0896-6273(01)00429-9. [DOI] [PubMed] [Google Scholar]
- Gilbert J, Levee R, Catalano K. A preliminary report on a new memory scale. Perceptual and Motor Skills. 1968;27:277–278. doi: 10.2466/pms.1968.27.1.277. [DOI] [PubMed] [Google Scholar]
- Gold JJ, Squire LR. Quantifying medial temporal lobe damage in memory-impaired patients. Hippocampus. doi: 10.1002/hipo.20032. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf P, Squire LR, Mandler G. The information that amnesic patients do not forget. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1984;10:164–178. doi: 10.1037//0278-7393.10.1.164. [DOI] [PubMed] [Google Scholar]
- Hamann SB, Squire LR. Intact perceptual memory in the absence of conscious memory. Behavioral Neuroscience. 1997;111:850–854. doi: 10.1037//0735-7044.111.4.850. [DOI] [PubMed] [Google Scholar]
- Insausti R, Insausti AM, Sobreviela M, Salinas A, Martinez-Penuela J. Human medial temporal lobe in aging: Anatomical basis of memory preservation. Microscopy Research and Technique. 1998;43:8–15. doi: 10.1002/(SICI)1097-0029(19981001)43:1<8::AID-JEMT2>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- Insausti R, Juottonen K, Soininen H, Insausti AM, Partanen K, Vainio P, Laakso MP, Pitkanen A. MR volumetric analysis of the human entorhinal, perirhinal, and temporopolar cortices. American Journal of Neuroradiology. 1998;19:659–671. [PMC free article] [PubMed] [Google Scholar]
- Jacoby LL, Dallas M. On the relationship between autobiographical memory and perceptual learning. Journal of Experimental Psychology: General. 1981;3:306–340. doi: 10.1037//0096-3445.110.3.306. [DOI] [PubMed] [Google Scholar]
- Johnston WA, Dark VJ, Jacoby LL. Perceptual fluency and recognition judgments. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1985;11:3–11. doi: 10.1037//0278-7393.17.2.210. [DOI] [PubMed] [Google Scholar]
- Keane MM, Gabrieli JDE, Monti LA, Fleischman DA, Cantor JM, Noland JS. Intact and impaired conceptual memory processes in amnesia. Neuropsychology. 1997;11:59–69. doi: 10.1037//0894-4105.11.1.59. [DOI] [PubMed] [Google Scholar]
- Kucera H, Francis WN. Computational analysis of present-day American English. Brown University Press; Providence, RI: 1967. [Google Scholar]
- Levy DA, Bayley PJ, Squire LR. The anatomy of semantic knowledge: Medial vs. lateral temporal lobe. Proceedings of the National Academy of Sciences, USA. 2004;101:6710–6715. doi: 10.1073/pnas.0401679101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner B, Squire LR, Kandel ER. Cognitive neuroscience and the study of memory. Neuron. 1998;20:445–468. doi: 10.1016/s0896-6273(00)80987-3. [DOI] [PubMed] [Google Scholar]
- Nelson DL, McEvoy CL, Schreiber TA. The University of South Florida word association, rhyme, and word fragment norms. 1998 doi: 10.3758/bf03195588. Retrieved September 2002 from http://www.usf.edu/FreeAssociation/ [DOI] [PubMed]
- Olichney JM, Van Petten C, Paller KA, Salmon DP, Iragui VJ, Kutas M. Word repetition in amnesia: Electrophysiological measures of impaired and spared memory. Brain. 2000;123:1948–1963. doi: 10.1093/brain/123.9.1948. [DOI] [PubMed] [Google Scholar]
- Rajaram S, Geraci L. Conceptual fluency selectively influences knowing. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2000;26:1070–1074. doi: 10.1037//0278-7393.26.4.1070. [DOI] [PubMed] [Google Scholar]
- Schacter DL, Buckner RL. Priming and the brain. Neuron. 1998;20:185–195. doi: 10.1016/s0896-6273(00)80448-1. [DOI] [PubMed] [Google Scholar]
- Schmolck H, Kensinger E, Corkin S, Squire LR. Semantic knowledge in patient H.M. and other patients with bilateral medial and lateral temporal lobe lesions. Hippocampus. 2002;12:520–533. doi: 10.1002/hipo.10039. [DOI] [PubMed] [Google Scholar]
- Shimamura AP, Squire LR. Paired-associate learning and priming effects in amnesia: A neuropsychological analysis. Journal of Experimental Psychology: General. 1984;113:556–570. doi: 10.1037//0096-3445.113.4.556. [DOI] [PubMed] [Google Scholar]
- Stark CEL, Squire LR. Recognition memory and familiarity judgments in severe amnesia: No evidence for a contribution of repetition priming. Behavioral Neuroscience. 2000;114:459–467. doi: 10.1037//0735-7044.114.3.459. [DOI] [PubMed] [Google Scholar]
- Stefanacci L, Buffalo EA, Schmolck H, Squire LR. Profound amnesia following damage to the medial temporal lobe: A neuroanatomical and neuropsychological profile of patient E.P. Journal of Neuroscience. 2000;20:7024–7036. doi: 10.1523/JNEUROSCI.20-18-07024.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson-Shill SL, D'Esposito M, Kan IP. Effects of repetition and competition on activity in left prefrontal cortex during word generation. Neuron. 1999;23:513–522. doi: 10.1016/s0896-6273(00)80804-1. [DOI] [PubMed] [Google Scholar]
- Tulving E, Schacter DL. Priming and human memory systems. Science. 1990;247:301–306. doi: 10.1126/science.2296719. [DOI] [PubMed] [Google Scholar]
- Vaidya CJ, Gabrieli JDE, Demb JB, Keane MM, Wetzel LC. Impaired priming on the general knowledge task in amnesia. Neuropsychology. 1996;10:529–537. [Google Scholar]
- Vaidya CJ, Gabrieli JDE, Keane MM, Monti LA. Perceptual and conceptual memory processes in global amnesia. Neuropsychology. 1995;9:580–591. [Google Scholar]
- Verfaellie M, Cermak LS. Perceptual fluency as a cue for recognition judgments in amnesia. Neuropsychology. 1999;13:198–205. doi: 10.1037//0894-4105.13.2.198. [DOI] [PubMed] [Google Scholar]
- Verfaellie M, Keane MM. Impaired and preserved memory processes in amnesia. In: Squire LR, Schacter DL, editors. Neuropsychology of memory. 3rd ed. Guilford Press; New York: 2002. pp. 35–46. [Google Scholar]
- Wagner AD, Gabrieli JDE, Verfaellie M. Dissociations between familiarity processes in explicit recognition and implicit perceptual memory. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1997;23:305–323. doi: 10.1037//0278-7393.23.2.305. [DOI] [PubMed] [Google Scholar]
- Wagner AD, Koutstaal W, Maril A, Schacter DL, Buckner RL. Task-specific repetition priming in left inferior prefrontal cortex. Cerebral Cortex. 2000;10:1176–1184. doi: 10.1093/cercor/10.12.1176. [DOI] [PubMed] [Google Scholar]
- Whittlesea BWA. Illusions of familiarity. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1993;19:1235–1253. [Google Scholar]
- Yonelinas AP. The nature of recollection and familiarity: A review of 30 years of research. Journal of Memory and Language. 2002;46:441–517. [Google Scholar]
- Yonelinas AP, Regehr G, Jacoby L. Incorporating response bias in a dual-process theory of memory. Journal of Memory and Language. 1995;34:835. [Google Scholar]
- Yoon C, Feinberg F, Hu P, Gutchess AH, Hedden T, Chen HY, Jing Q, Yao C, Park DC. Category norms as a function of culture and age: Comparisons of item responses to 105 categories by American and Chinese adults. University of Michigan; Ann Arbor: 2002. Unpublished manuscript. Available at http://agingmind.beckman.uiuc.edu/Cat_Norms/Publications.html. [DOI] [PubMed] [Google Scholar]