Abstract
Human exposure to air pollutants, including ambient particulate matter, has been proposed as a mechanism for the rise in allergic disorders. Diesel exhaust particles, a major component of ambient particulate matter, induce sensitization to neoallergens, but the mechanisms by which sensitization occur remain unclear. We show that diesel exhaust particles upregulate thymic stromal lymphopoietin in human bronchial epithelial cells in an oxidant-dependent manner. Thymic stromal lymphopoietin induced by diesel exhaust particles was associated with maturation of myeloid dendritic cells, which was blocked by anti-thymic stromal lymphopoietin antibodies or silencing epithelial cell-derived thymic stromal lymphopoietin. Dendritic cells exposed to diesel exhaust particle-treated human bronchial epithelial cells induced Th2 polarization in a thymic stromal lymphopoietin-dependent manner. These findings provide new insight into the mechanisms by which diesel exhaust particles modify human lung mucosal immunity.
Keywords: Dendritic cells, bronchial epithelial cells, diesel exhaust particles, thymic stromal lymphopoietin, lung
Introduction
The prevalence of atopy and asthma has increased worldwide [1]. Allergic disorders result from T cell-derived immune responses that are the consequence of a complex interaction between environmental exposures and genetic susceptibility. Although industrialization and urbanization of populations with an increase in exposures to air pollutants such as ambient particulate matter (PM) has been proposed as a mechanism to explain the rise in allergic disorders, the immunologic mechanisms by which ambient PM enhance allergic sensitization and asthma remain unclear [2]. Diesel exhaust particles (DEP), major components of vehicular-derived ambient PM, induce sensitization to neoallergens [3]. Dendritic cells (DC) are pivotal in the induction and persistence of allergen-induced Th2 responses in the murine lung [4, 5]. Dendritic cells reside in close proximity to bronchial epithelial cells [6], which have potential to regulate recruitment, maturation, and activation of these DC.
DC induce a polarized Th1, Th2, Th3, or Treg response [7]. The ability of DC to induce each of these responses is determined by signals received when DC are immature [8–10]. Although DC maturation and polarization has been well studied to be a response to pathogens, cell–cell interactions may also instruct DC responses. We and others have recently demonstrated that primary culture human bronchial epithelial cells (HBEC) synthesize and release chemokines and cytokines with potential to induce DC migration and functional maturation [11–14]. We previously examined whether DEP instruct DC and showed that low concentrations of DEP did not directly induce DC maturation but induced phenotypic and functional maturation of DC in part, via HBEC-derived soluble signals. Moreover, we demonstrated that granulocyte-macrophage colony stimulating factor (GM-CSF) was one component of the HBEC-derived signal involved in DEP-induced DC maturation [15]. We now expand on this observation and suggest additional mechanisms by which DEP induce DC maturation and polarization that support Th2 responses.
In humans, thymic stromal lymphopoietin (TSLP), a member of the interleukin 7 (IL-7) family of cytokines [16], induces maturation of myeloid DC supporting Th2 cell polarization [16] and maintenance of the Th2 memory response [17]. TSLP upregulates expression of MCH class II, CD80, and CD83 but not IL-12 family members or type I interferons [18]. In contrast, TSLP upregulates myeloid DC messenger RNA (mRNA) for chemokines that attract neutrophils and eosinophils, as well as thymus and activation-regulated chemokine (TARC/CCL17), which attracts Th2 cells [19]. TSLP-treated myeloid DC induce an inflammatory Th2 response that is associated with IL-5, IL-4, IL-13, and tumor necrosis factor alpha (TNF-α) but low IL-10 [16, 19, 20]. TLSP induces expression of the TNF superfamily protein OX40 ligand (OX40L), engagement of which signals the generation of these inflammatory Th2 cells [18]. TSLP-activated DC also enhance functions of Th2 memory cells in the presence of IL-25 [21].
TSLP is expressed by human epithelial cells [16, 22] and is increased in asthmatic airways [23]. We therefore examined whether DEP provide signals for DC-induced T cell activation and polarization via epithelial-cell-derived TSLP and propose this as a potential mechanism for DEP-induced airway immune responses.
Methods
Reagents
Dulbecco’s modified Eagle’s medium (DMEM), MEM, penicillin-streptomycin, fetal bovine serum (FBS), trypsin, ethylene-diamine-tetraacetic acid (EDTA) solution, and phosphate-buffered saline (PBS) were purchased from Gibco Life Technologies (Grand Island, NY, USA). Bronchial epithelial cell growth medium (BEGM) and bronchial epithelial cell basal medium (BEBM) were purchased from Cambrex BioScience (Walkersville, MD, USA). Ficoll was obtained from Amersham Bioscience (Piscataway, NJ, USA). Recombinant GM-CSF and inter-leukin 4 (IL-4) were obtained from PeproTech (Princeton, NJ, USA). Anti-TSLP Abs and TSLP enzyme-linked immunosorbent assay (ELISA) were obtained from R&D systems (Minneapolis, MN, USA). Lipopolysaccharide (LPS) and phorbol 12-myristate 13-acetate (PMA) were obtained from Sigma (St. Louis, MO, USA) and N-acetyl cysteine (NAC) was obtained from Calbiochem (San Diego, CA, USA). Fluorescent reagents for fluorescence-activated cell sorting (FACS) analyses were obtained from Becton Dickinson Immunocytometry Systems (San Jose, CA, USA), Pharmingen (San Diego, CA, USA), or Coulter/Immunotech (Brea, CA, USA). BrdU-ELISA was obtained from Roche (Indianapolis, IN, USA). Carbon particles (Cabot Elftex 12) were from Cabot Corporation (Billerica, MA, USA).
DEP were derived from a 1.6 l Volkswagen Diesel Engine (40 kW) running under standard city driving cycle conditions according to US test protocol FTP 72 (EPA 1992) and were a kind gift of DL Costa (US EPA). DEP were diluted in cell culture medium, vortexed (five times, 10 s), sonicated (1 min), and added to cells in the defined concentrations. Because DEP sediment to the bottom of cell culture dishes, DEP concentrations were based on the available surface area (μg/cm2). Endotoxin content of DEP was measured using the PyroGene™ Recombinant Factor C Assay (Cambrex BioScience) according to the manufacturer’s recommendations. Endotoxin activity, in a concentration of DEP ten times higher than that used in most experiments, was below the lower limit of detection (0.01 EU/ml).
Primary culture HBEC were derived after endobronchial brushing during bronchoscopy of normal human volunteers following a protocol approved by the Institutional Review Board of the New York University School of Medicine. HBEC were cultured as described previously [15] and plated on tissue culture plates coated with human collagen (Vitrogen-100, Cohesion Technologies, Palo Alto, CA, USA). Studies were performed with cells in passage 2. Confluent cultures were deprived of hydrocortisone, epinephrine, and retinoic acid for 24 h before exposure studies. 16HBE14o− cells, a simian virus 40 large T antigen-transformed HBEC line was generously provided by Dr. D. Gruenert (Univ. of Vermont, Burlington, VT, USA) [24]. 16HBE14o− cells were cultured as previously described [15].
Monocyte-derived dendritic cells (MDDC) were generated from human peripheral blood monocytes after isolation of peripheral blood mononuclear cells (PBMC) by Ficoll density gradient centrifugation of heparinized blood from healthy volunteers. CD14+-PBMC were isolated by positive selection using magnetically labeled CD14 antibodies and a magnetic cell separator (Miltenyi Biotech, Auburn, CA, USA) according to the manufacturer’s instructions. CD14+-PBMC were cultured in DMEM supplemented with FBS (10%, v/v), β-mercaptoethanol (5 mM), and supplemented with GM-CSF (50 ng/ml) and IL-4 (10 ng/ml). Immature MDDC (iMDDC) were harvested on day 6, and MDDC were subsequently characterized by flow cytometry as lineage negative (CD3, 14, 16, 19, 20, 56; lin), CD11c+, and HLA-DR bright cells. Immature DC were identified as CD83− CD86− within the lin− CD11c+ HLA-DR+ population. For some experiments, immature myeloid DC were directly isolated from PBMC by positive selection using magnetically labeled BDCA-1 antibodies and a magnetic cell separator (Miltenyi Biotech) according to the manufacturer’s instructions.
For co-culture experiments, immature DC (1×105 cells/well) were added to confluent epithelial cells (HBEC or 16HBE14o− as defined) in 12-well plates (48 h) and cells subsequently trypsinized (0.5 mg/ml, 5 min). DC were discriminated from epithelial cells by FACS according to their expression of CD11c and cytokeratin (CK) 18/19 [15].
Immunoflourescence Labeling and Flow Cytometry
Cells were labeled (30 min, 0°C) using fluorescein isothio-cyanate-, phycoerythrin-, or allophycocyanin (APC)-conjugated murine mAb against lineage markers (lin), CD11c, cytokeratins (CK) 18/19, CD80, CD83, CD86, and HLA-DR, or mouse IgG isotype control, washed with cold Dulbecco’s phosphate-buffered saline (DPBS), and fixed in DPBS containing 1% formaldehyde. Cytometric analyses were performed on a FACSCalibur (BD Biosciences, San Jose, CA, USA). Forward and 90° angle scattered laser light intensities were used to exclude cellular debris. Fluorescence spillover detected by inappropriate channels, including autofluorescence above the first log, was corrected by electronic compensation. Irrelevant mAb conjugated to the same fluorophores were used to determine nonspecific cell surface binding. In some experiments, viable cells recovered from DC/HBEC co-cultures were labeled and separated by flow-sorting on a FACSVantage SE (BD Biosciences) equipped with an aerosol containment unit for handling isolates of human origin.
Analysis of Intracellular ROS Levels by Flow Cytometry
Intracellular reactive oxygen species (ROS) levels were assessed with carboxy-H2DCF-DA, an oxidation-sensitive fluorescent probe. Inside the cell, this probe is deacetylated by intracellular esterases and, in the presence of a variety of intracellular peroxides, is oxidized to fluorescent 2,7-dichlorofluorescein (DCF). Before treatment with DEP, cells were loaded with carboxy-H2DCF-DA (20 min, 37°C, 5% CO2, 20 μM), washed once, and DEP (3 μg/cm2) added (3 h). After treatment, cells were washed, detached with trypsin, and relative fluorescence intensity (RFI) determined by flow cytometry.
RNA Quantification
RNA was isolated from HBEC using the Micro-to-Midi total RNA purification System (Invitrogen, Carlsbad, CA, USA). Quantification by real-time PCR was performed using the One-Step Quantitech SyberGreen Real Time PCR kit (Qiagen, Valencia, CA) following the manufacturer’s instructions. In vitro transcription was performed using T7/SP6 RNA polymerase (Invitrogen) followed by complementary DNA (cDNA) digestion (DNAse I, Amplification Grade, Invitrogen) and cDNA purification. Levels of respective transcripts were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) transcript level as an internal control [ΔCt(target)=Ct(target)−Ct(GAP-DH)]. Change in respective transcripts after treatment were assessed by ΔΔCt(target)=ΔCt(target, stimulated)−ΔCt (target, resting).
Quantification of TSLP
TSLP was measured in culture supernatant of HBEC by a commercial ELISA. HBEC were cultured as previously described, supernatant removed, and TSLP measured according to the manufacturer’s instructions. The lower limit of detection of the ELISA is 3.5 pg/ml.
siRNA Gene Silencing
For small interfering RNA (siRNA) studies, 16HBE14o− were seeded (70% confluence), and after 24 h, cells were transfected with a siRNA duplex-pool targeted against human TSLP (Dharmacon siGENOME™ SMARTpool M-011166, Dharmacon, Lafayette, CO, USA) using lipo-fectamine (Invitrogen) according to the manufacturer’s instructions. Additional cells were transfected with a non-targeting duplex-pool (siCONTROL, Dharmacon) or siCONTROL RISC-free. After 48 h, cells were again transfected with the same siRNA duplex-pools (48 h). Transfected 16HBE14o− were subsequently exposed to iMDDC as described previously.
MLR Assay
Mixed leukocyte reaction (MLR) assays were performed with naive T cells derived from allogeneic PBMC. Allogeneic naive CD4+ CD45RA+ CD4RO− T cells were isolated from PBMC using the CD4 multisort kit in combination with a depletion of CD45RO+ cells using CD45RO-magnetic beads (Miltenyi Biotech) according to the manufacturer’s instructions. T cells (2.5×105) were cultured (5 days) in 96-well round-bottom microplates in the presence of mitomycin C-treated DC (5×103). Cells were incubated with BrdU (10 μM, 24 h) and BrdU incorporation by proliferating responder cells was quantified using a BrdU-ELISA according to the manufacturer’s instructions.
Cell Polarization
DC were exposed to DEP, in the absence or presence of epithelial cells (48 h). Cells were trypsinized and DC were isolated (FACS) from co-culture. Mitomycin C-treated DC were used as stimulator cells to allogeneic naive T cells at a stimulator–responder ratio of 1:10 in round bottom 96-well plates (Costar, Cambridge, MA, USA). After 5 days, T cells were stimulated with rhIL-2 (10 μ/ml) and expanded for another 5 days. On day 10, T cells were washed, counted, and viable cells were re-stimulated with PMA (10 ng/ml) and ionomycin (1 μg/ml) for 24 h. Cell-free supernatants were analyzed for IL-5 and interferon gamma (IFN-) using commercially available ELISAs (R&D). The lower detection limits were 3 and 8 pg/ml for each cytokine, respectively. To generate defined polarized DC for comparison, DC were exposed to maturation factors that have been well-described to induce mature DC that support Th1 polarization (MF1). These factors include IL-1β (10 ng/ml), TNF-α (10 ng/ml), IL-6 (100 ng/ml), IFN-γ (50 ng/ml) [25]. Alternatively, DC were exposed to maturation factors that have been well-described to induce mature DC that support Th2 polarization (MF2). These factors include IL-1β (10 ng/ml), TNF-α (10 ng/ml), IL-6 (100 ng/ml), PGE2 (10 μM) [26].
Data Analysis
Data are presented as mean±standard error (SE). Experiments were performed from HBEC derived from at least three individual donors. Significance was determined by Student’s t test, with a p<0.05 considered significant.
Results
DEP Induced TSLP Expression by HBEC
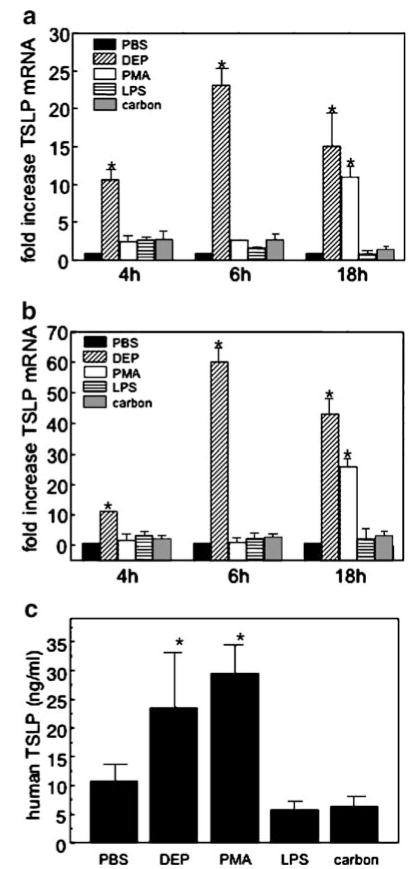
Although there is increasing information about the regulation of TSLP, the stimuli that induce TSLP in airway cells remain incompletely described [27, 28]. We first investigated whether DEP and proinflammatory stimuli induced TSLP transcripts in primary culture HBEC. HBEC were treated with defined agents (4–18 h) and RNA isolated. DEP was used at a physiologically relevant concentration (3 μg/cm2), previously shown to induce cell activation without cell toxicity [15]. Exposure of HBEC to DEP induced a rapid (4 h) and persistent (18 h) increase in TSLP mRNA compared to resting HBEC (10.7±1.5- and 15.1± 4.3-fold induction respectively, n=3, mean ± SE, p<0.05; Fig. 1a). Exposure of HBEC to PMA (10 nM) resulted in a delayed increase in TSLP (11.1±1.3-fold increase, 18 h). In contrast, neither LPS (1 μg/ml) nor carbon particles (3 μg/cm2) induced TSLP mRNA in HBEC at either time point (Fig. 1a). To confirm that 16HBE14o− cells, a transformed HBEC line, serve as model epithelial cells for these studies, TSLP transcripts were also monitored in 16HBE14o− cells. DEP and PMA induced TSLP in a time dependent manner in 16HBE14o− cells, and neither LPS nor carbon induced an increase in TLSP transcript (Fig. 1b). Thus, DEP induced TSLP transcripts in both primary and transformed HBEC.
Fig. 1.
DEP upregulate TLSP in HBEC and 16HBE14o− cells. TSLP mRNA (4–18 h) was measured in response to DEP (3 μg/cm2), PMA (10 nM), LPS (1 μg/ml), or carbon (3 μg/cm2) in a HBEC or b 16HBE14o− cells. RNA was isolated and quantified by real-time PCR, normalized to GAPDH and expressed (ΔΔCt) as fold increase compared to unstimulated. c TSLP protein was measured in HBEC supernatants by commercial ELISA. Data are derived from three different donors and are expressed as mean±SE (n=3, *p<0.05).
To confirm that TSLP transcripts were associated with TSLP expression, HBEC were treated with DEP, PMA, carbon, or LPS in the previously defined concentrations, and TSLP release in supernatants was determined by a commercial ELISA. TSLP expression was upregulated in HBEC exposed to DEP and PMA but was not increased compared to PBS in supernatants derived from HBEC treated with carbon or LPS (Fig. 1c).
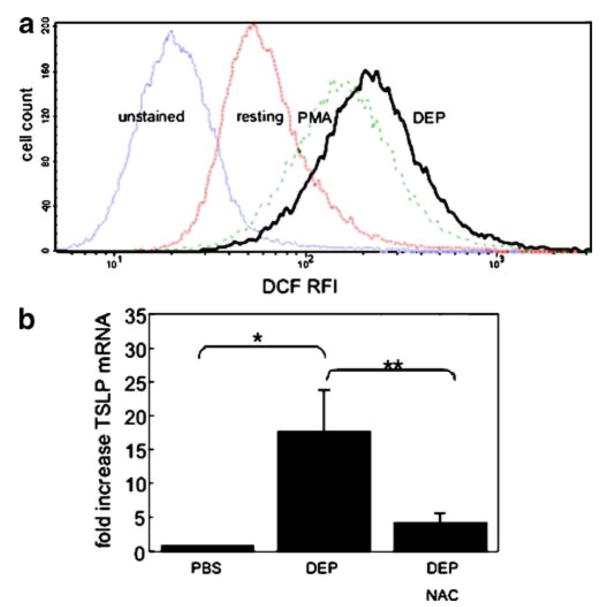
To investigate whether upregulation of TSLP mRNA in HBEC was mediated by the induction of ROS, we first confirmed the ability of DEP (3 μg/cm2) to induce ROS using carboxy-H2DCF-DA, an oxidation-sensitive fluorescent probe. RFI was increased in DEP-treated compared to resting 16HBE14o− cells (641±126.1 vs 269±65.3, respectively, mean±SE, n=4, p<0.05), and a representative experiment is shown in Fig. 2a. Carbon particles failed to induce ROS (data not shown).
Fig. 2.
DEP increase ROS and TSLP mRNA is inhibited by NAC. a Representative experiment (n=4) of ROS induction by DEP in epithelial cells (16HBE14o−). ROS was determined by monitoring RFI of carboxy-H2DCF-DA for unstained, resting, PMA-treated, or DEP-treated 16HBE14o− cells. b DEP-induced TSLP mRNA is inhibited by treatment of 16HBE14o− cells with NAC (*PBS vs DEP, **DEP vs DEP+NAC, p<0.05, n=3).
To examine whether DEP-induction of ROS was associated with TSLP transcription, we used NAC, an anti-oxidant that is a precursor of glutathione and increases free radical scavenging. Bronchial epithelial cells (16HBE14o−) were exposed to DEP (3 μg/cm2) in the absence or presence of the NAC (25 mM), and TSLP mRNA measured. DEP-induced TSLP mRNA was significantly decreased in the presence of NAC (17.8±10.4- vs 4.3±2.1-fold induction above resting, respectively; n=3; p<0.05; Fig. 2b). These data suggested that oxidant-induced pathways were associated with the DEP-induced TSLP mRNA in bronchial epithelial cells.
TSLP Derived from DEP-treated HBEC and Functional DC Maturation
We have previously demonstrated that exposure of iMDDC to DEP-treated HBEC, but not to DEP alone, results in phenotypic and functional maturation of MDDC [15]. As TSLP has been proposed as an epithelial-cell-derived cytokine that regulates T cell differentiation via its effect on DC maturation [16], we examined whether DEP-induced upregulation of TSLP mRNA in HBEC was associated with functional MDDC maturation. HBEC were cultured, treated with DEP (3 μg/cm2), and iMDDC added (48 h). Cells were subsequently disassociated and MDDC flow-sorted from the co-cultures. MDDC were used as stimulator cells in an MLR (24 h, 1:50; DC/T cell), and proliferation of T cells was measured by BrdU incorporation [15]. MDDC isolated after exposure to DEP-treated HBEC supported an increased T cell proliferation compared to MDDC exposed to resting HBEC (2.6±0.4-fold increase, n=3; p<0.05; Fig. 3). Results were similar with 16HBE14o− cells, and the effect was not seen with carbon particles at a similar dose (data not shown). Anti-TSLP Ab included during exposure of MDDC to DEP-treated epithelial cells significantly but incompletely reduced the ability of those MDDC to support T cell proliferation above that of resting (1.6±0.2-fold above resting, n=3). These data suggested that TSLP participated in functional maturation of MDDC exposed to DEP-treated HBEC.
Fig. 3.
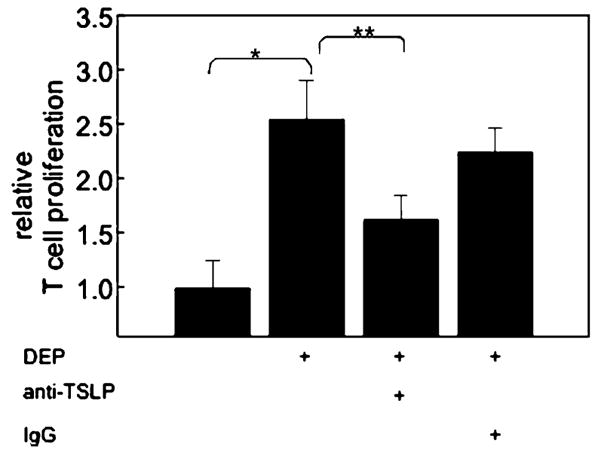
TSLP derived from DEP-treated epithelial cells support DC maturation. Immature MDDC were exposed to resting or DEP (3 μg/cm2)-treated HBEC (48 h) in the absence or presence of anti-TSLP Ab. DC were FACS sorted, and their ability to support proliferation of allogeneic naive T cells monitored by BrdU incorporation (n=3, *DC derived from DEP-treated HBEC vs resting, **DC derived from DEP-treated HBEC compared to anti-TSLP, p<0.05).
To confirm that DEP-treated HBEC induced MDDC maturation via epithelial-cell-derived TSLP, we silenced mRNA encoding for TSLP in 16HBE14o− cells and tested whether these cells retained the ability to induce MDDC maturation in response to DEP (Fig. 4). 16HBE14o− cells were transfected with siTSLP duplex pools, a control non-targeting siRNA duplex-pool (siCONTROL), or a non-targeting siRNA duplex pool that does not activate the RISC pathway. Cells were subsequently treated with DEP, and iMDDC were added to the culture (48 h). MDDC were harvested and functional maturation of MDDC was monitored by MLR. MDDC that had been exposed to DEP-treated 16HBE14o− cells and vehicle control induced a significant increase in T cell proliferation compared to MDDC exposed to resting 16HBE14o− cells (2.1±0.3-fold increase, n=3, p<0.05; Fig. 4). MDDC exposed to DEP-treated 16HBE14o− cells that had been transfected with control non-targeting siRNA duplex-pools retained the ability to induce T cell proliferation to a level similar to that of untransfected cells. In contrast, MDDC exposed to DEP-treated 16HBE14o− cells that had been transfected with siTSLP duplex pools failed to induce T cell proliferation above that of resting cells. These studies confirmed the ability of DEP-induced epithelial-cell-derived TSLP to stimulate functional MDDC maturation.
Fig. 4.
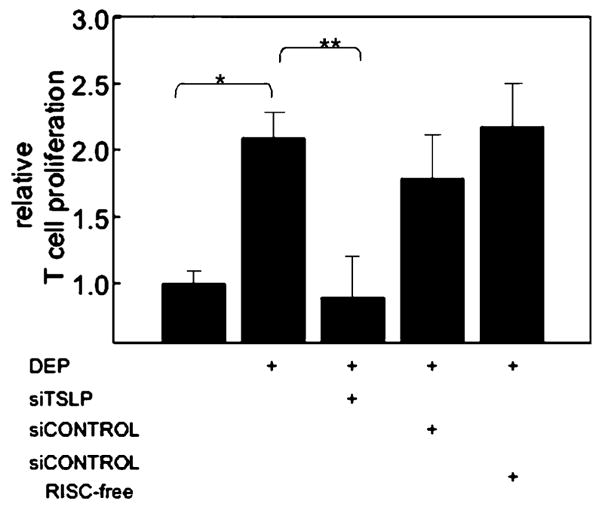
DEP-induced epithelial cell-derived TSLP supports DC maturation. Immature MDDC were exposed to resting, DEP-treated, or DEP-treated 16HBE14o− cells in the presence of siTSLP or siCONTROLs. DC were FACS sorted, and their ability to support proliferation of allogeneic naive T-cells monitored by BrdU incorporation (n=3, *p<0.05 DC derived from DEP-treated 16HBE14o− cells vs resting, **p<0.05 DC derived from DEP-treated 16HBE14o− cells compared to siTSLP).
DEP and DC Polarization
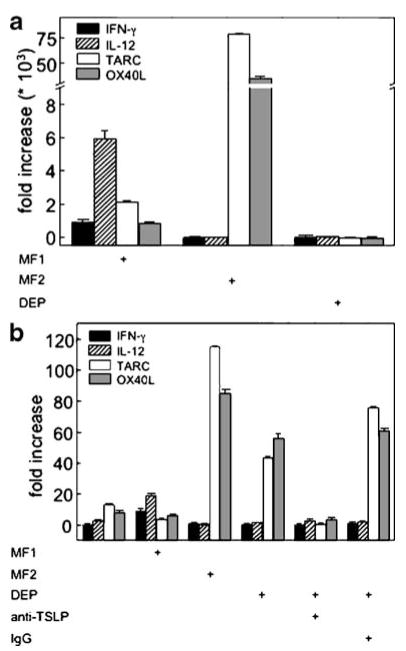
The ability of DC to support T cell polarization depends on their ability to express T helper polarizing cytokines [29]. Dendritic cells activated by TSLP express OX40L and Th2-attracting chemokines such as TARC/CCL17 but not Th1 cell polarizing cytokines such as IL-12 or interferon IFN-γ [16, 19]. We therefore examined whether human myeloid DC derived from whole blood and exposed to DEP or DEP-treated HBEC displayed a pattern of markers consistent with Th2 priming DC. Myeloid DC were exposed to maturation factors that have previously been described to induce mature DC that support Th1 polarization (MF1) [25]. Alternatively, myeloid DC were exposed to maturation factors that induce mature DC, which support Th2 polarization (MF2) [26]. Exposure of myeloid DC to MF1 resulted in small but significant increases in mRNA for IFN-γ, IL-12p35, and less TARC, and OX40L (Fig. 5a). Myeloid DC exposed to MF2 failed to upregulate IFN-γ or IL-12 but induced a high level of TARC and OX40L (Fig. 5a). These cytokine patterns are consistent with the generation of DC that would support Th1 and Th2 T cell polarization, respectively [19]. In contrast to myeloid DC exposed to MF1 or MF2, myeloid DC exposed to DEP did not express elevated levels of any of these markers, consistent with the absence of Th2 polarization by direct exposure of DC to DEP (Fig. 5a).
Fig. 5.
TSLP derived from DEP-treated epithelial cells supports DC expression of Th2 priming cytokines. a Immature myeloid DC were treated with defined cytokines that support Th1 (MF1) or Th2 (MF2) priming DC or with DEP (3 μg/cm2). RNA was isolated from DC and quantified for IFN-γ, IL-12p35, TARC, or OX40L by real-time PCR and expressed as fold increase compared to unstimulated (mean±SE, n=3). b Immature myeloid DC were exposed to epithelial cells (16HBE14o−) in the presence of MF1, MF2, DEP, or anti-TSLP Ab. DC were sorted by FACS, and RNA was isolated from DC and quantified for IFN-γ, IL-12p35, TARC, and OX40L by real-time PCR (mean±SE, n=3).
To examine whether HBEC regulated the DC response to DEP, myeloid DC were exposed to 16HBE14o− cells treated with MF1, MF2, or DEP. Dendritic cells were subsequently disassociated and myeloid DC flow-sorted (CD11c+) from the DC-HBEC co-cultures by FACS. DC mRNA was measured for IFN-γ, IL-12p35, TARC, and OX40L (Fig. 5b). Myeloid DC exposed to resting 16HBE14o− cells expressed a Th2 bias, with an increase in TARC and OX40L compared to IFNγ and IL-12p35. As expected, levels of TARC and OX40L remained low in the presence of MF1 but were elevated when cells were exposed to MF2. Myeloid DC exposed to DEP-treated 16HBE14o− cells expressed elevated TARC and OX40L, consistent with a Th2-priming pattern. These data suggested that DEP-induced epithelial-cell-derived signals were associated with polarization of DC.
To determine whether the polarized response of myeloid DC in the presence of DEP-treated epithelial cells was dependent on TSLP, we performed experiments in the presence of anti-TSLP Ab (Fig. 5b). The increased TARC and OX40L mRNA induced by DEP was significantly reduced in the presence of anti-TSLP Ab. These studies suggested that DEP-treated bronchial epithelial cells supported the generation of DC with the potential to elicit a Th2 response via TSLP.
DC exposed to DEP-treated Epithelial Cells Induce T Cell Polarization
To confirm that DEP-treated bronchial epithelial cells elicited DC with a functional ability to induce Th2 polarized T cells, T cells were stimulated with isolated myeloid DC that were untreated or that had been exposed to bronchial epithelial cells (16HBE14o− cells). T cell supernatants were isolated and IL-5 and IFN-γ measured by ELISA and expressed as cytokine concentrations (Table I) or as the IL-5/IFN-γ ratio (Fig. 6). As expected, T cells exposed to myeloid DC treated with MF1 expressed low levels of IL-5 and high levels of IFN-γ (Table I). T cells exposed to myeloid DC treated with MF2 expressed high levels of IL-5 and a lower level of IFN-γ compared to those exposed to MF1. We previously showed that DEP-treated myeloid DC fail to stimulate T cell proliferation [15], and in these current studies, exposure of T cells to DEP-treated myeloid DC failed to induce IL-5 consistent with absence of Th2 polarization.
Table I.
T Cell-Derived IL5 and IFN-γ in Response to Myeloid DC Treated with Defined Agents
| Treatment | IL-5 (ng/ml ± SE) | IFN-γ (ng/ml ± SE) |
|---|---|---|
| MF1 | 0.10±0.02 | 16.02±1.01 |
| MF2 | 1.25±0.20 | 5.60±0.43 |
| DEP | 0.14±0.06 | 3.59±0.08 |
| 16HBE+Medium | 0.55±0.03 | 5.600±0.39 |
| 16HBE+MF1 | 0.26±0.02 | 9.06±1.24* |
| 16HBE+MF2 | 0.92±0.07* | 3.98±0.24 |
| 16HBE+DEP | 0.86±0.07* | 5.02±0.89 |
| 16HBE+DEP+α TSLP | 0.33±0.04 | 7.57±0.38* |
| 16HBE+DEP+IgG | 0.92±0.10* | 4.40±0.24 |
DC were derived from the defined culture conditions (MF1, MF2, and DEP) in the absence or presence of 16HBE14o− cells (16HBE). When indicated, IgG or anti-TLSP was included during the DC-16HBE co-culture. Naive T cells were stimulated by DC (5 days), expanded (5 days) and treated with PMA and ionomycin (24 h) in a standard T cell polarization assay as described in “Methods”. Supernatants from T cells were analyzed for IL-5 and IFN-γ by commercial ELISA. Data are presented as mean±SE, n=3
p<0.05 for an increase vs 16HBE+medium.
Fig. 6.
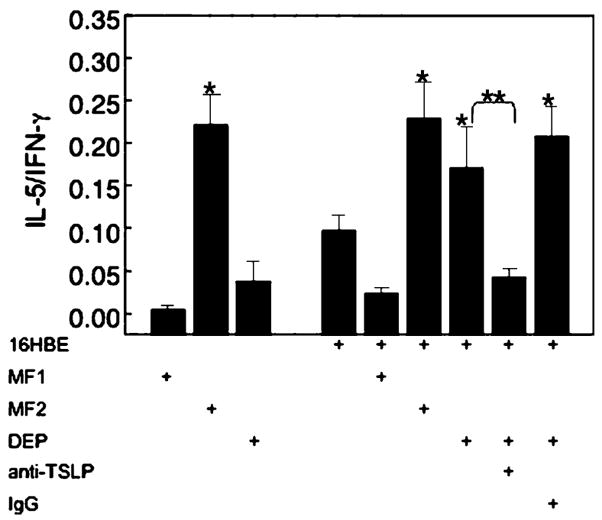
TSLP derived from DEP-treated epithelial cells supports Th2 polarization by DC. Immature myeloid DC were stimulated (48 h) with specific polarizing cytokines (MF1 and MF2) or DEP in the absence or presence of epithelial cells (16HBE14o−) and anti-TSLP Ab. DC were isolated and used as stimulator cells with allogeneic naive T cells (1:10, 5 days). T cells were subsequently expanded (IL-2, 10 U/ml, 5 days), stimulated (PMA, 10 ng/ml and ionomycin, 1 μg/ml, 24 h) and cell-free supernatants analyzed for IFN-γ and IL-5 by ELISA. Data are expressed as ratio of IL-5/IFN-γ (n=3, *p<0.05 vs resting epithelial cells, **p<0.05 DEP-treated epithelial cells vs anti-TSLP Ab).
T cells exposed to DC cultured with resting epithelial cells (16HBE14o−) showed an IL-5/IFN-γ ratio that was elevated compared to that seen in T cells exposed to DC cultured with MF1 (Fig. 6). T cells exposed to myeloid DC that had been cultured with DEP-treated epithelial cells released IL-5 and IFN-γ and had a high IL-5/IFN-γ ratio that was greater than that seen for cells exposed to DC cultured with resting epithelial cells and similar to the ratio seen in T cells exposed to DC that had been cultured with MF2 and epithelial cells (Fig. 6).
To test whether TSLP was associated with the elevated IL-5/IFN-γ ratio, T cells were exposed to myeloid DC that had been cultured with DEP-treated epithelial cells in the presence of anti-TSLPAb. The presence of anti-TSLP during the DC-16HBE14o− co-culture resulted in a significant decrease in DC-stimulated T cell-derived IL-5/IFN-γ ratio (Fig. 6). These studies suggested that DEP-treated epithelial cells induced functional polarization of DC as determined by the polarization of naive CD4+ T cells and that TSLP was necessary for this response.
Discussion
The role of pollutants in the development of allergic disorders has gained increased recognition, [2] and the importance of DC and allergic diseases is increasingly being recognized [4]. We have previously suggested that DEP modify airway immune responses via activation of epithelial cells resulting in maturation of DC [15]. We now expand our previous observation and demonstrate that DEP-treated airway epithelial cells upregulate TSLP in an oxidant-dependent manner and, furthermore, that airway epithelial-cell-derived TSLP results in functional maturation and Th2 polarization of DC.
TSLP promotes DC-mediated Th2 responses and augments responsiveness to T cell receptor engagement [30]. Thus, TSLP has the potential to modify airway immune responses in multiple ways. Upregulation of human TSLP by IL-1β, TNF-α, or selective toll-like receptors (TLR2, 8, and 9) has been shown to be mediated via NFκB in human airway epithelial cells [27]. More recently, Il-4, IL-13, and rhinovirus have been shown to upregulate TSLP mRNA in a corticosteroid-sensitive manner in human epithelial cells [28]. We now show that DEP upregulate TSLP in HBEC and that this upregulation was rapid and prolonged. The increase in TSLP mRNA induced by DEP was associated with an increase in TSLP protein release by HBEC. Moreover, we demonstrated that, as expected, DEP induced ROS in epithelial cells and that TSLP was upregulated in an oxidant-dependent manner. These data provide information about additional stimuli for epithelial cell-derived TSLP and suggest a mechanism by which ambient environmental stimuli have the potential to upregulate this immunoregu-latory cytokine.
DC maturation is functionally characterized by the ability to support T cell proliferation. We now show that DEP-treated epithelial cells induced DC that supported DC maturation as measured by a MLR. This effect was dependent on TSLP, as both a blocking antibody and silencing epithelial-cell-derived TSLP inhibited the ability of DEP-treated epithelial cells to induce myeloid DC that supported T cell proliferation.
DC that induce Th2 responses are characterized by their low level expression of IFN-γ and IL-12 but high level expression of chemokines associated with Th2 recruitment, such as TARC. Moreover, expression of OX40L by DC has been shown to be TSLP-induced and to trigger inflammatory Th2 cells [16, 19]. Direct exposure of myeloid DC to ambient outdoor air from the city of Baltimore has recently been shown to directly induce a complex Th1/Th2-like pattern of T cell activation [31]. Our previous studies [15] and this current study failed to demonstrate functional maturation and phenotypic polarization of myeloid DC exposed directly to DEP alone. Although we were able to detect TARC and OX40L in DC exposed to MF2, direct exposure of DC to DEP failed to induce TARC or OX40L in our studies. Our data suggest the importance of additional cell-derived signals in the induction of DEP responses and the abundance of airway epithelial cells, and their presence as a first target for inhaled particles suggest their important role in microenvironmental regulation of DC. Indeed, our data now show that DEP-treated epithelial cells induce DC that express both TARC and OX40L and thus have the potential to trigger inflammatory Th2 cells. Furthermore, expression of TARC and OX40L is inhibited in the presence of anti-TSLP, suggesting a role for DEP-induced TSLP. Finally, and importantly, we demonstrated that DEP-treated epithelial cells stimulated myeloid DC that were capable of inducing T cells characterized by a Th2 polarized response with an increased IL-5/IFN-γ ratio.
These data suggest an important cell–cell interaction between DC and associated epithelial cells. We have previously shown that HBEC synthesize and release chemokines with potential to recruit immature DC in response to ambient PM [14]. We and others have shown that ambient PM or DEP induce airway epithelial-cell-derived pro-inflammatory and immunoregulatory cytokines [11–13, 32]. We previously demonstrated that DEP-induced epithelial cell-derived GM-CSF was required but not sufficient for DEP-induced maturation of DC [15]. Our current studies suggest that resting epithelial cells induce DC that bias toward a Th2 polarization as shown by an intermediate IL-5/IFN-γ ratio, although they do not support T cell proliferation. However, DEP-treated epithelial cells induce both functional DC maturation that supports T cell proliferation and a marked increase in the IL-5/IFN-γ ratio, consistent with a strong ability to induce Th2 polarization.
Conclusion
We extend our previous observations and show that TSLP derived from DEP-treated epithelial cells induce myeloid DC maturation that supports a polarized Th2 response. These data suggest that DEP have the ability to modify DC towards a Th2 type response through a complex interplay of epithelial cell-derived cytokines, a finding that has important implications for understanding the role of environmental pollutants in the development of mucosal immunity and airway diseases.
Acknowledgments
This work was supported by National Institutes of Health Grants ES010187 (JR), T32 ES007267 (JR), MO1RR00096, AI27742-129007 (DT) and AI44729 (DT). We thank Rashedul Ahsan for his technical support for ROS studies.
References
- 1.Beasley R, Crane J, Lai CK, Pearce N. Prevalence and etiology of asthma. J Allergy Clin Immunol. 2000;105:S466–72. doi: 10.1016/s0091-6749(00)90044-7. [DOI] [PubMed] [Google Scholar]
- 2.Saxon A, Diaz-Sanchez D. Air pollution and allergy: you are what you breathe. Nat Immunol. 2005;6:223–6. doi: 10.1038/ni0305-223. [DOI] [PubMed] [Google Scholar]
- 3.Diaz-Sanchez D, Garcia MP, Wang M, Jyrala M, Saxon A. Nasal challenge with diesel exhaust particles can induce sensitization to a neoallergen in the human mucosa. J Allergy Clin Immunol. 1999;104:1183–8. doi: 10.1016/s0091-6749(99)70011-4. [DOI] [PubMed] [Google Scholar]
- 4.Hammad H, Lambrecht BN. Recent progress in the biology of airway dendritic cells and implications for understanding the regulation of asthmatic inflammation. J Allergy Clin Immunol. 2006;118:331–6. doi: 10.1016/j.jaci.2006.03.041. [DOI] [PubMed] [Google Scholar]
- 5.Lambrecht BN, Salomon B, Klatzmann D, Pauwels RA. Dendritic cells are required for the development of chronic eosinophilic airway inflammation in response to inhaled antigen in sensitized mice. J Immunol. 1998;160:4090–7. [PubMed] [Google Scholar]
- 6.Holt PG, Schon-Hegrad MA, Oliver J, Holt BJ, McMenamin PG. A contiguous network of dendritic antigen-presenting cells within the respiratory epithelium. Int Arch Allergy Appl Immunol. 1990;91:155–9. doi: 10.1159/000235107. [DOI] [PubMed] [Google Scholar]
- 7.Hammad H, Kool M, Soullie T, Narumiya S, Trottein F, Hoogsteden HC, et al. Activation of the D prostanoid 1 receptor suppresses asthma by modulation of lung dendritic cell function and induction of regulatory T cells. J Exp Med. 2007;204:357–67. doi: 10.1084/jem.20061196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kalinski P, Hilkens CM, Wierenga EA, Kapsenberg ML. T-cell priming by type-1 and type-2 polarized dendritic cells: the concept of a third signal. Immunol Today. 1999;20:561–7. doi: 10.1016/s0167-5699(99)01547-9. [DOI] [PubMed] [Google Scholar]
- 9.Liu YJ. Dendritic cell subsets and lineages, and their functions in innate and adaptive immunity. Cell. 2001;106:259–62. doi: 10.1016/s0092-8674(01)00456-1. [DOI] [PubMed] [Google Scholar]
- 10.Pulendran B, Banchereau J, Maraskovsky E, Maliszewski C. Modulating the immune response with dendritic cells and their growth factors. Trends Immunol. 2001;22:41–7. doi: 10.1016/s1471-4906(00)01794-4. [DOI] [PubMed] [Google Scholar]
- 11.Ohtoshi T, Takizawa H, Okazaki H, Kawasaki S, Takeuchi N, Ohta K, et al. Diesel exhaust particles stimulate human airway epithelial cells to produce cytokines relevant to airway inflammation in vitro. J Allergy Clin Immunol. 1998;101:778–85. doi: 10.1016/S0091-6749(98)70307-0. [DOI] [PubMed] [Google Scholar]
- 12.Boland S, Bonvallot V, Fournier T, Baeza-Squiban A, Aubier M, Marano F. Mechanisms of GM-CSF increase by diesel exhaust particles in human airway epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2000;278:L25–32. doi: 10.1152/ajplung.2000.278.1.L25. [DOI] [PubMed] [Google Scholar]
- 13.Reibman J, Hsu Y, Chen LC, Kumar A, Su WC, Choy W, et al. Size fractions of ambient particulate matter induce granulocyte macrophage colony-stimulating factor in human bronchial epithelial cells by mitogen-activated protein kinase pathways. Am J Respir Cell Mol Biol. 2002;27:455–62. doi: 10.1165/rcmb.2001-0005OC. [DOI] [PubMed] [Google Scholar]
- 14.Reibman J, Hsu Y, Chen LC, Bleck B, Gordon T. Airway epithelial cells release MIP-3alpha/CCL20 in response to cytokines and ambient particulate matter. Am J Respir Cell Mol Biol. 2003;28:648–54. doi: 10.1165/rcmb.2002-0095OC. [DOI] [PubMed] [Google Scholar]
- 15.Bleck B, Tse DB, Jaspers I, Curotto de Lafaille MA, Reibman J. Diesel exhaust particle-exposed human bronchial epithelial cells induce dendritic cell maturation. J Immunol. 2006;176:1–7437. doi: 10.4049/jimmunol.176.12.7431. [DOI] [PubMed] [Google Scholar]
- 16.Liu YJ, Soumelis V, Watanabe N, Ito T, Wang YH, Malefyt Rde W, et al. TSLP: an epithelial cell cytokine that regulates T cell differentiation by conditioning dendritic cell maturation. Annu Rev Immunol. 2007;25:193–219. doi: 10.1146/annurev.immunol.25.022106.141718. [DOI] [PubMed] [Google Scholar]
- 17.Wang YH, Ito T, Wang YH, Homey B, Watanabe N, Martin R, et al. Maintenance and polarization of human TH2 central memory T cells by thymic stromal lymphopoietin-activated dendritic cells. Immunity. 2006;24:827–38. doi: 10.1016/j.immuni.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 18.Liu YJ. Thymic stromal lymphopoietin and OX40 ligand pathway in the initiation of dendritic cell-mediated allergic inflammation. J Allergy Clin Immunol. 2007;120:238–44. doi: 10.1016/j.jaci.2007.06.004. [DOI] [PubMed] [Google Scholar]
- 19.Ito T, Wang YH, Duramad O, Hori T, Delespesse GJ, Watanabe N, et al. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J Exp Med. 2005;202:1213–23. doi: 10.1084/jem.20051135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu YJ. Thymic stromal lymphopoietin: master switch for allergic inflammation. J Exp Med. 2006;203:269–73. doi: 10.1084/jem.20051745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Angkasekwinai P, Park H, Wang YH, Wang YH, Chang SH, Corry DB, et al. Interleukin 25 promotes the initiation of proallergic type 2 responses. J Exp Med. 2007;204:1509–17. doi: 10.1084/jem.20061675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat Immunol. 2002;3:673–80. doi: 10.1038/ni805. [DOI] [PubMed] [Google Scholar]
- 23.Ying S, O’Connor B, Ratoff J, Meng Q, Mallett K, Cousins D, et al. Thymic stromal lymphopoietin expression is increased in asthmatic airways and correlates with expression of Th2-attracting chemokines and disease severity. J Immunol. 2005;174:8183–90. doi: 10.4049/jimmunol.174.12.8183. [DOI] [PubMed] [Google Scholar]
- 24.Cozens AL, Yezzi MJ, Kunzelmann K, Ohrui T, Chin L, Eng K, et al. CFTR expression and chloride secretion in polarized immortal human bronchial epithelial cells. Am J Respir Cell Mol Biol. 1994;10:38–47. doi: 10.1165/ajrcmb.10.1.7507342. [DOI] [PubMed] [Google Scholar]
- 25.Bender A, Sapp M, Schuler G, Steinman RM, Bhardwaj N. Improved methods for the generation of dendritic cells from nonproliferating progenitors in human blood. J Immunol Methods. 1996;196:121–35. doi: 10.1016/0022-1759(96)00079-8. [DOI] [PubMed] [Google Scholar]
- 26.Vieira PL, de Jong EC, Wierenga EA, Kapsenberg ML, Kalinski P. Development of Th1-inducing capacity in myeloid dendritic cells requires environmental instruction. J Immunol. 2000;164:4507–12. doi: 10.4049/jimmunol.164.9.4507. [DOI] [PubMed] [Google Scholar]
- 27.õLee HC, Ziegler SF. Inducible expression of the proallergic cytokine thymic stromal lymphopoietin in airway epithelial cells is controlled by NFkappaB. Proc Natl Acad Sci U S A. 2007;104:914–9. doi: 10.1073/pnas.0607305104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kato A, Favoreto S, Jr, Avila PC, Schleimer RP. TLR3- and Th2 cytokine-dependent production of thymic stromal lymphopoietin in human airway epithelial cells. J Immunol. 2007;179:1080–7. doi: 10.4049/jimmunol.179.2.1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kapsenberg ML. Dendritic-cell control of pathogen-driven T-cell polarization. Nat Rev Immunol. 2003;3:984–93. doi: 10.1038/nri1246. [DOI] [PubMed] [Google Scholar]
- 30.Rochman I, Watanabe N, Arima K, Liu YJ, Leonard WJ. Cutting Edge: Direct action of thymic stromal lymphopoietin on activated human CD4+ T cells. J Immunol. 2007;178:6720–4. doi: 10.4049/jimmunol.178.11.6720. [DOI] [PubMed] [Google Scholar]
- 31.Williams MA, Porter M, Horton M, Guo J, Roman J, Williams D, et al. Ambient particulate matter directs nonclassic dendritic cell activation and a mixed TH1/TH2-like cytokine response by naive CD4+ T cells. J Allergy Clin Immunol. 2007;119:488–97. doi: 10.1016/j.jaci.2006.10.022. [DOI] [PubMed] [Google Scholar]
- 32.Bayram H, Devalia JL, Sapsford RJ, Ohtoshi T, Miyabara Y, Sagai M, et al. The effect of diesel exhaust particles on cell function and release of inflammatory mediators from human bronchial epithelial cells in vitro. Am J Respir Cell Mol Biol. 1998;18:441–8. doi: 10.1165/ajrcmb.18.3.2882. [DOI] [PubMed] [Google Scholar]