Abstract
The sulfur K-edge x-ray absorption spectra for the amino acids cysteine and methionine and their corresponding oxidized forms cystine and methionine sulfoxide are presented. Distinct differences in the shape of the edge and the inflection point energy for cysteine and cystine are observed. For methionine sulfoxide the inflection point energy is 2.8 eV higher compared with methionine. Glutathione, the most abundant thiol in animal cells, also has been investigated. The x-ray absorption near-edge structure spectrum of reduced glutathione resembles that of cysteine, whereas the spectrum of oxidized glutathione resembles that of cystine. The characteristic differences between the thiol and disulfide spectra enable one to determine the redox status (thiol to disulfide ratio) in intact biological systems, such as unbroken cells, where glutathione and cyst(e)ine are the two major sulfur-containing components. The sulfur K-edge spectra for whole human blood, plasma, and erythrocytes are shown. The erythrocyte sulfur K-edge spectrum is similar to that of fully reduced glutathione. Simulation of the plasma spectrum indicated 32% thiol and 68% disulfide sulfur. The whole blood spectrum can be simulated by a combination of 46% disulfide and 54% thiol sulfur.
This paper describes an application of sulfur K-edge x-ray absorption spectroscopy (XAS) to determine the ratio of thiol to disulfide in biological systems. Sulfur is an important element in chemistry and biology, and often it is desirable to assay thiols and disulfide and their ratio in biological samples. There are no spectroscopic techniques available that can distinguish between thiol and disulfide sulfur or sulfur in higher formal oxidation states. This is not surprising, because there are no significant optical markers for sulfur. Also, only one sulfur isotope of <1% natural abundance has a nuclear magnetic moment; but this isotope possesses an unfavorable nuclear spin with a small magnetic moment and a large quadrupole moment, making NMR unattractive for sulfur analysis.
Sulfur K-edge XAS has been shown to exhibit a strong correlation between oxidation state and inflection point energy (IPE) and to have a rich x-ray absorption near-edge structure (XANES) that is related to chemical structure (1–4) and can be used to distinguish between thiol and disulfide sulfur.
The two sulfur-containing amino acids found in all cells are cysteine and methionine. Cysteine can be oxidized to the disulfide form cystine. Additionally there are “mixed” disulfides (cysteine bound to proteins by formation of a disulfide linkage). Methionine can be reversibly oxidized by HO⋅, 1O2, or H2O2 to the sulfoxide or even further to the sulfone (5).
The number and importance of enzymes and proteins whose function has been found to be significantly modified by the oxidation of their thiol-containing amino acid is growing rapidly. The reversible oxidation–reduction of thiol groups is intimately linked to oxidative stress in a number of respects. For cysteine and glutathione, the formation of disulfide bonds as such cannot be considered as damage, because it is a reversible process, but disulfide bridges in peptides and proteins may drastically alter biological functions. Alteration in the thiol/disulfide status, for example, has been found to lead to biological consequences, including changes in enzyme properties (Km or Vmax effects) (6). Thus the thiol redox status seems to serve as a metabolic signal. There are many enzymes and proteins whose activities are reported to be protein modulated by mixed disulfide formation or changes in the ratio of the reduced form of glutathione (GSH) to the oxidized form of glutathione (GSSG), such as liver phosphorylase phosphatase (7), pineal indoleamine N-acetyltransferase (8), glycogen synthase (9, 10), thiol proteinase inhibitor (11), adenylate cyclase (12, 13), guanylate cyclase (14), 3-hydroxy-3-methylglutaryl-CoA reductase (15), carbonic anhydrase I (16), and phosphofructokinase (17). Oxidation of methionine to methionine sulfoxide can be associated with the loss of function; a particularly striking example is that of antitrypsin (18).
The most abundant molecule containing cysteinyl thiol in animal cells is glutathione. Glutathione (GSH) is a tripeptide, γ-l-glutamyl-l-cysteinylglycine, with cellular concentrations in the range 0.1–10 mM. Glutathione is a component of a pathway that uses reduced NADPH to maintain the cellular redox state. It has an essential role in maintaining various components of cells in their reduced state. Furthermore, it is involved in the reduction of ribonucleotides to deoxyribonucleotides (precursors of DNA) and is implicated in protecting the cell against oxidative damage and free radical damage and in detoxification via a conjugation mechanism (19). In its “free” form, it is present intracellularly, mainly in the thiol-reduced form (GSH). The ratio between the oxidized (GSSG) and the reduced (GSH) forms is kept at a low value, approximately 0.1, in normal cells (20). Glutathione may exist in many forms as GSH, GSSG, glutathione mixed disulfides with low molecular weight compounds (e.g., cysteine) and with high molecular weight substances (e.g., peptides and proteins). The level of oxidized to reduced glutathione can change significantly upon oxidative stress and provides useful information about the redox and detoxification status of cells and tissues (21–24).
Several procedures for the determination of GSH and GSSG from different biological sources have been reported in the literature, but all require extraction procedures for the determination of glutathione in biological materials. An in situ determination is not possible with chemical (25) and enzymatic (26–28) methods, HPLC (29–38), flow cytometry (39–41) and, more recently, capillary electrophoresis (42, 43). GSH and GSSG can be detected by using spectrophotometric (44, 45), spectrofluorimetric (46–52), and electrochemical methods (30, 34, 37, 53–58) with levels of sensitivity ranging from nanomolar (33, 35) to picomolar (31, 32). For spectrophotometric detection, glutathione extracts are treated with iodoacetate to block free thiol groups and then with Sanger’s reagent (as one example) to produce a N-dinitrophenyl derivative that can be analyzed. Neither GSH nor GSSG, as such, have significant intrinsic fluorescence (59). Some complexes and some covalent or other derivatives of GSH and GSSG, however, have fluorescent properties, and the fluorescence and fluorescence quenching of these derivatives can be used for determination of GSH and GSSG levels. However, a difficult problem in determining glutathione levels is the sample and standard preparation. Sample preparation, protein precipitating agents, and sample treatment must be carefully considered, because the oxidation of glutathione to glutathione disulfide, including protein mixed disulfides, must be minimized; otherwise erroneously low values of glutathione and high values of glutathione disulfide and mixed disulfides will be obtained (60–62). The difficulties associated with existing procedures, especially for use in vivo, led us to investigate a method to measure thiol and disulfide levels in cells without any previous chemical treatment. An optimal sample preparation procedure for measuring the RSH and RSSR levels in erythrocytes, plasma, and blood would be fast freezing of freshly taken samples. We show that sulfur K-edge XAS, on such samples, allows a determination of thiol, disulfide (GSSG and GSSR), sulfoxide, and sulfone sulfur in vivo.
In whole blood, plasma, and red blood cells, the two major thiol and disulfide components are glutathione and cyst(e)ine. The major sulfur-containing constituents of whole blood are glutathione (∼34 mg/100 ml), cysteine (∼0.9 mg/100 ml), and methionine (∼0.5 mg/100 ml), which are there in free form as well as part of proteins. Additionally, there is sulfate (∼1.0 mg/100 ml) (63). Whole blood is mostly a mixture of red blood cells (erythrocytes) and blood plasma. In red blood cells, the level of cyst(e)ine is at least one order of magnitude lower than the glutathione concentration (64, 65). This cyst(e)ine/glutathione ratio is reversed in blood plasma (66, 67).
Sulfur K-edge XAS spectra have been collected and the redox state of sulfur in whole blood, as well as separated plasma and red blood cells, has been determined. The sulfur K-edge spectra of reduced/oxidized forms of the amino acids cysteine/cystine and methionine/methionine sulfoxide and reduced/oxidized glutathione also are reported.
MATERIALS AND METHODS
Cysteine, cystine, methionine, methionine sulfoxide, reduced glutathione, and oxidized glutathione were of reagent grade, purchased from Sigma and used as received. A small amount (micrograms) of each compound was ground into a fine powder and dispersed as thinly as possible (over about 1 cm2) on Mylar tape. The compounds were used at successively lower concentrations until a reproducible spectrum was obtained, to avoid spectral distortion and loss of resolution caused by self-absorption of the sample.
Whole human blood was collected in heparin-coated syringes and centrifuged for 5 min at 2,500 × g. Whole blood, erythrocytes, or blood plasma samples were placed in sample holders (Lucite frames with inner dimensions 18 × 2.5 × 0.8 mm, backed with Mylar tape) and frozen in liquid nitrogen immediately after collection.
Sulfur K-edge spectra were recorded at the Stanford Synchrotron Radiation Laboratory by using the 54-pole wiggler beamline 6–2, with a Pt-coated focusing mirror and a Si(111) double crystal monochromator. Details of the setup have been described (see refs. 1–4, 68, and 69 and references therein). All sulfur K-edge spectra were run with sample between 130 and 140 K in a home-built liquid He flow cryostat. The data were collected as fluorescence excitation spectra with a N2 gas-filled Lytle detector (70, 71). Scans were run from 2,420 to 2,740 eV, with a step size of 0.08 eV from 2,465 to 2,495 eV to obtain high resolution (∼0.5 eV) (2) in the preedge and edge region and a step size of 1.0 eV in the other regions.
For each compound, two to four scans were collected. The energy was calibrated from the sulfur K-edge spectra of Na2S2O3⋅5H2O (Sigma), run after every two sample scans. The maximum of the first preedge feature in this spectrum occurs at 2,472.02 eV (68). Reproducibility of the sulfur K-edge energies with the above conditions have been found to be ±0.1 eV (68).
A preedge background was removed from all spectra by fitting a straight line to the preedge region and subtracting this straight line from the entire spectrum. Normalization of the spectra was achieved by extrapolating into the edge region a quadratic fit to the data in the far post-edge region. Each spectrum was then multiplied by a factor so that the value of the extrapolated line was unity at the energy of the first absorption peak. The position of the main edge was defined by the first IPE of the steeply rising absorption edge, determined from the zero crossing of the second derivative of the spectrum. Analytical differentiation of a third-order polynomial fit to the data over an interval of 2.0 eV on each side of a data point produced the derivative spectra.
For erythrocytes, plasma, and whole blood, simulations were compared with the original data in the region from 2,471 to 2,475 eV by following a least-squares method. A limited range of energy around the IPE was used in the simulation to reduce the effects of scattering from more distant atoms and multiple scattering effects that contribute at higher energies. Percentages of the two constituent spectra (GSH and GSSG) were combined to form a simulated spectrum. The sum of the squares of the residuals between the original data and the simulation was designated the error and referred to as the residuals in the figures. The method of combination of edges in a least squares method to determine respective amounts of a component has been used (72). The linear combination, in percentages of GSH and GSSG, found to give the minimum error (least squares) was judged to be the best fit of the data. The uncertainty is estimated to be ±5%. The plot of residuals is shown as insets in the figures.
RESULTS
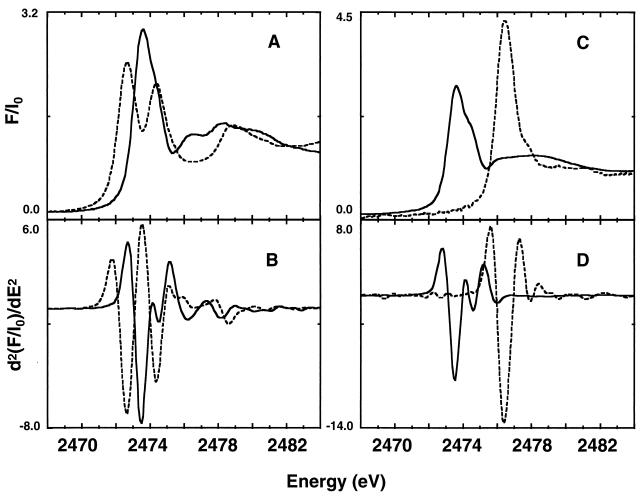
The sulfur K-edge x-ray absorption near-edge spectra of the thiol cysteine and thioether methionine are shown in Fig. 1. The IPE for cysteine and methionine, 2,473.1 eV, are the same (Table 1). The oxidized form of cysteine is cystine, a disulfide. The IPE changes to 2,472.1 eV for cystine. The S XANES spectra of cysteine and cystine are clearly different (Fig. 1A). For cystine the intense absorption maximum is split into two features with almost equal intensity. Additionally the region 5–10 eV beyond the maximum shows less structure compared with cysteine. Methionine sulfoxide (Fig. 1C, dashed line), the oxidized form of methionine (IPE = 2,473.1 eV; see Fig. 1C, solid line), shows a shift in IPE of more than 2 eV (IPE = 2,475.9 eV), whereas the shape of the edge stays about the same. The second derivatives of the spectra shown in Fig. 1 B and D accentuate the changes in spectral position and shape. The significant changes seen in the second derivatives demonstrate the sensitivity of the XAS method for distinguishing between the different kinds of S-containing molecules.
Figure 1.
(A) Normalized S K-edge x-ray absorption spectra of cysteine (solid line) and cystine (dashed line). (B) Second derivatives of cysteine (solid line) and cystine (dashed line). (C) Normalized S K-edge x-ray absorption spectra of methionine (solid line) and methionine sulfoxide (dashed line). (D) Second derivatives of methionine (solid line) and methionine sulfoxide (dashed line). All sample materials were in the form of solids.
Table 1.
S K-edge IPEs
| Compound | Edge IPE, eV |
|---|---|
| Cysteine (Cys-SH) | 2,473.1 |
| Cystine (Cys-SS-Cys) | 2,472.1 |
| Methionine | 2,473.1 |
| Methionine sulfoxide | 2,475.9 |
| GSH | 2,473.0 |
| GSSG | 2,472.2 |
| Blood | 2,472.4 |
| Erythrocytes | 2,473.0 |
| Plasma | 2,472.3 |
Precision of the IPE is < ±0.1 eV; the inflection point reported is determined from the zero crossing of the second derivative of the spectrum.
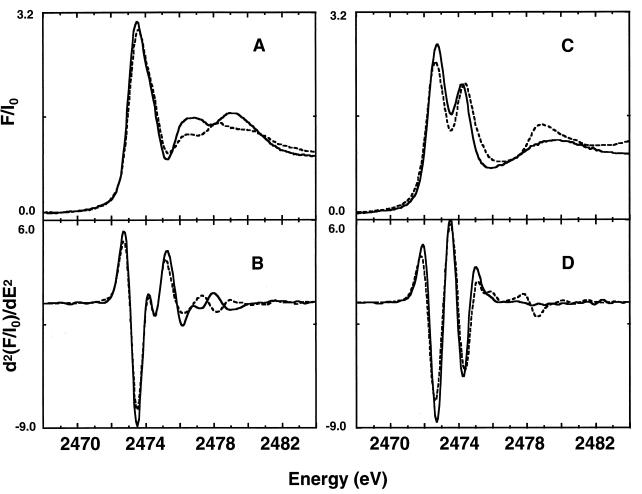
The S XANES spectra of GSH (Fig. 2A) and GSSG (Fig. 2C) are shown. Plotting these spectra over the corresponding cysteine and cystine spectra reveals major similarities, as expected. This also is reflected in similar inflection point energies for GSH and cysteine and for GSSG and cystine and in the similarity of the second derivatives of the spectra (Fig. 2 B and D) at energies near the IPE. Differences become obvious more than 10 eV beyond the IPE, where more distant atoms and multiple scattering effects begin to contribute to the spectral features.
Figure 2.
(A) Normalized S K-edge x-ray absorption spectra of reduced glutathione (solid line) and cysteine (dashed line). (B) Second derivatives of reduced glutathione (solid line) and cysteine (dashed line). (C) Normalized S K-edge x-ray absorption spectra of oxidized glutathione (solid line) and cystine (dashed line). (D) Second derivatives of oxidized glutathione (solid line) and cystine (dashed line). All sample materials were in the form of solids.
The sulfur K-edge spectra of whole blood, plasma, and erythrocytes were collected to determine the ratio of thiol to disulfide sulfur in these biological samples.
Erythrocytes.
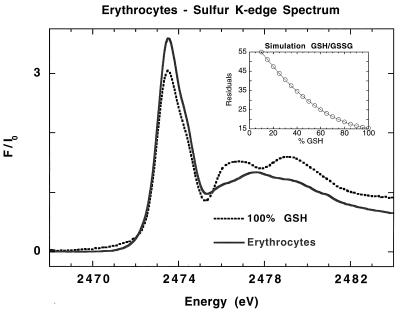
Fig. 3 shows the sulfur K-edge XANES spectrum from erythrocytes (solid line). Comparison with the reduced glutathione spectrum (dashed line) shows near identity in the rising edge region and indicates that glutathione is present predominantly in its reduced form. The postedge region shows some difference, again because the scattering of more distant atoms and multiple scattering effects begin to contribute. It is appropriate to simulate the erythrocyte spectrum only with glutathione and without any non-glutathione-derived cysteine contributions because it is known that the level of cysteine and cystine is at least one order of magnitude lower than the glutathione concentration in red blood cells (64, 65). The similarities of the sulfur XAS spectra of reduced glutathione and cysteine do not allow a differentiation between these two moieties. This is not required because the focus of this study is the determination of the ratio of thiol to disulfide sulfur and not the absolute amounts of all individual thiol and/or disulfide components. There is good agreement between the thiol (GSH) concentrations obtained herein by sulfur K-edge spectroscopy and those determined spectrophotometrically and by high-performance capillary electrophoresis analysis (73). Spectrophotometric investigations of human red blood cells have shown that the level of GSH to GSSG is about 245:1. By high-performance capillary electrophoresis analysis, no GSSG was detectable. Another study that analyzed GSH and GSSG levels of normal human red blood cells by HPLC and electrochemical detection reported a ratio of 297:1 (30). The uncertainty of the least squares fitting methodology is ∼5% and hence the detectability of GSSG in excess GSH by using XAS is 5:100.
Figure 3.
Normalized S K-edge x-ray absorption spectra of erythrocytes (solid line) and 100% reduced glutathione (dashed line). (Inset) Plot of residuals for the GSH/GSSG simulation.
Plasma.
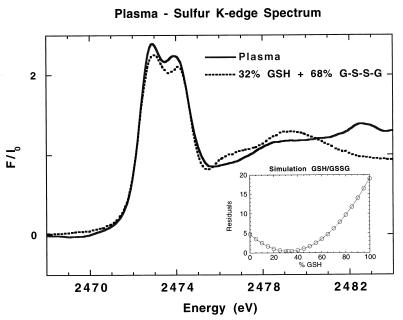
The concentration of GSH and GSSG in extracellular media such as plasma is orders of magnitude lower than the intracellular content. In plasma the total cyst(e)ine (cysteine and cystine) concentration is ∼20 times greater than the total glutathione concentration (66, 67). It has been reported by Malloy et al. (74) that in blood plasma up to 50% of cysteine is found to be “mixed” cysteine. The blood plasma spectrum (Fig. 4, solid line) can be simulated with GSH (or cysteine) and GSSG (or cystine) (Fig. 4, dashed line). Best results were obtained by using 68% GSSG and 32% GSH, indicating that 32% are in the thiol form and 68% are disulfides and mixed disulfides. The thiol/disulfide values reported in the literature differ because of the use of different processing methods (75). A small peak is seen at 2,482.5 eV, which corresponds to the K-edge inflection point of S in SO42− that is known to be present in plasma.
Figure 4.
Normalized S K-edge x-ray absorption spectra of blood plasma (solid line) and a simulation of 32% GSH and 68% GSSG (dashed line). (Inset) Plot of residuals for the GSH/GSSG simulation.
Whole Blood.
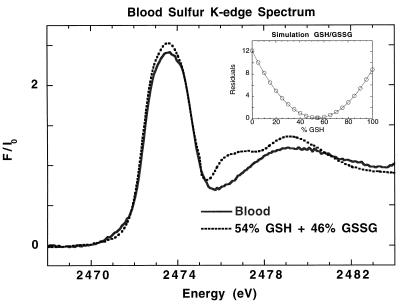
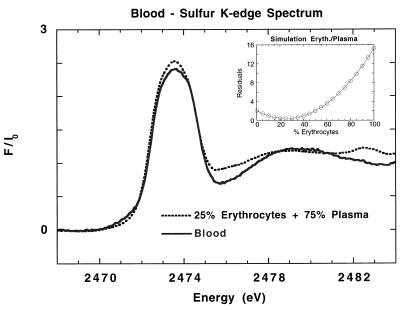
Sulfur K-edge spectrum of whole blood is shown in Fig. 5. An excellent simulation of the whole blood spectrum (Fig. 5) is obtained by a combination of 54% GSH and 46% GSSG. The inflection point of the whole blood spectrum is satisfactorily simulated, although there are some small differences in amplitudes between the simulation and the spectrum of blood. This might be because of small differences in self absorption of the blood samples, which were frozen glasses, compared with GSH and GSSG samples, which were solid powders. The S K-edge spectrum of whole blood is also satisfactorily simulated (Fig. 6) by the addition of the spectrum of erythrocytes (25%) with the spectrum from plasma (75%), indicating that the separation protocol does not alter the composition and the redox status of the erythrocytes and plasma.
Figure 5.
Normalized S K-edge x-ray absorption spectra of whole blood (solid line) and a simulation of 54% GSH and 46% GSSG (dashed line). (Inset) Plot of residuals for the GSH/GSSG simulation.
Figure 6.
Normalized S K-edge x-ray absorption spectra of whole blood (solid line) and a simulation with 25% erythrocytes and 75% plasma (dashed line). (Inset) Plot of residuals for the erythrocytes/plasma simulation.
DISCUSSION
Conflicting literature values for erythrocyte, plasma, and whole blood thiol and disulfide (cysteine or GSH and cystine or GSSG) values have resulted from the use of different analytical methods (75). Also, a remaining question is whether all thiol and disulfide forms in a blood sample have been quantitatively measured by using standard analytical methods. Moreover, the stability of GSH and its possible oxidation to GSSG during the period between collection and analytical determination have been questioned (76).
To resolve these matters, it is desirable to have an analytical tool that measures the thiol to disulfide ratio in biological samples without chemical treatment before determination. Sulfur K-edge spectroscopy provides such a tool. We have recorded spectra of whole biological cells, where the sulfur concentration is in the millimolar range. This sulfur concentration in the biological tissues was high enough that the spectra of the samples could be measured without any previous concentration and at the same time did not lead to problems of self absorption. A sulfur K-edge scan takes around 20 min and usually one scan has a signal-to-noise ratio high enough that the IPE can be determined and the shape of the edge can be analyzed. Sulfur K-edge spectroscopy detects changes of ∼5% in the thiol-to-disulfide ratio. Thus this tool does not have the absolute sensitivity of other analytical methods. Additionally sulfur K-edge spectroscopy does not distinguish between glutathione and cyst(e)ine or between GSSG and GSSR. However, we have shown that this method is an excellent tool to determine the thiol redox status in whole cells and to within an accuracy of 5%. The reported numbers vary over a much wider range.
Thus with this noninvasive spectroscopic determination of the oxidation states of intracellular thiol groups, it becomes possible to measure the changes induced by drugs and to explore the idea that radiation damage to cells may be determined by the state of sulfur.
Acknowledgments
We thank Joy C. Andrews, Gary T. Olsen, John H. Robblee, and Henk Visser for help with XAS data collection. We also thank Dr. Diane L. Tribble and Lillie Taat for help with preparation of the whole blood, erythrocytes, and plasma samples. This work was supported by the Director, Division of Energy Biosciences, Office of Basic Energy Sciences, and by the Office of Energy Research, Office of Health and Environmental Research and Health Effects Research, U. S. Department of Energy under Contract DE-AC03–76SF00098. The Stanford Synchrotron Radiation Laboratory is supported by the U.S. Department of Energy. The Biotechnology Laboratory at Stanford Synchrotron Radiation Laboratory is supported by the National Center for Research Resources of the National Institutes of Health. A.R. was supported by a Forschungsstipendium of the Deutsche Forschungsgemeinschaft, which is gratefully acknowledged.
ABBREVIATIONS
- GSH
glutathione reduced form
- GSSG
glutathione oxidized form
- IPE
inflection point energy
- XANES
x-ray absorption near-edge structure
- XAS
x-ray absorption spectroscopy
References
- 1.Hedman B, Frank P, Penner-Hahn J E, Roe A L, Hodgson K O, Carlson R M K, Brown G, Cerino J, Hettel R, Troxel T, et al. Nucl Instrum Methods Phys Res A. 1986;246:797–800. [Google Scholar]
- 2.Frank P, Hedman B, Carlson R M K, Tyson T A, Roe A L, Hodgson K O. Biochemistry. 1987;26:4975–4979. doi: 10.1021/bi00390a014. [DOI] [PubMed] [Google Scholar]
- 3.Frank P, Kustin K, Robinson W E, Linebaugh L, Hodgson K O. Inorg Chem. 1995;34:5942–5949. [Google Scholar]
- 4.Frank P, Hedman B, Carlson R M K, Hodgson K O. Inorg Chem. 1994;33:3794–3803. [Google Scholar]
- 5.Brot N, Weissbach H. Arch Biochem Biophys. 1983;223:271–281. doi: 10.1016/0003-9861(83)90592-1. [DOI] [PubMed] [Google Scholar]
- 6.Kosower E M, Kosower N S. Int Rev Cytol. 1978;54:109–160. doi: 10.1016/s0074-7696(08)60166-7. [DOI] [PubMed] [Google Scholar]
- 7.Usami M, Matsushita H, Shimazu T. J Biol Chem. 1980;255:1928–1931. [PubMed] [Google Scholar]
- 8.Namboodiri M A A, Weller J L, Klein D C. J Biol Chem. 1980;255:6032–6035. [PubMed] [Google Scholar]
- 9.Ernest M J, Kim K-H. J Biol Chem. 1973;248:1550–1555. [PubMed] [Google Scholar]
- 10.Lau K-H W, Thomas J A. J Biol Chem. 1983;258:2321–2326. [PubMed] [Google Scholar]
- 11.Wakamatsu N, Kominami E, Takio K, Katunuma N. J Biol Chem. 1984;259:13832–13838. [PubMed] [Google Scholar]
- 12.Mukherjee S P, Lynn W S. Biochim Biophys Acta. 1979;568:224–233. doi: 10.1016/0005-2744(79)90289-4. [DOI] [PubMed] [Google Scholar]
- 13.Mukherjee S P, Mukherjee C. Biochim Biophys Acta. 1981;677:339–349. doi: 10.1016/0304-4165(81)90245-2. [DOI] [PubMed] [Google Scholar]
- 14.Tsai S-C, Adamik R, Manganiello V C, Vaughan M. Biochem Biophys Res Commun. 1981;100:637–643. doi: 10.1016/s0006-291x(81)80223-9. [DOI] [PubMed] [Google Scholar]
- 15.Dotan I, Shechter I. Biochim Biophys Acta. 1982;713:427–434. doi: 10.1016/0005-2760(82)90262-4. [DOI] [PubMed] [Google Scholar]
- 16.Kondo T, Taniguchi N, Hirano T, Kawakami Y. J Biol Chem. 1984;259:15517–15522. [PubMed] [Google Scholar]
- 17.Gilbert H F. J Biol Chem. 1982;257:12086–12091. [PubMed] [Google Scholar]
- 18.Beatty K, Bieth J, Travis J. J Biol Chem. 1980;255:3931–3934. [PubMed] [Google Scholar]
- 19.Meister A. Pharmacol Ther. 1991;51:1–194. doi: 10.1016/0163-7258(91)90076-x. [DOI] [PubMed] [Google Scholar]
- 20.Meister A. In: Glutathione: Chemical, Biochemical and Medical Aspects. Dolphin D, Avramovic O, Poulson R, editors. New York: Wiley; 1988. pp. 1–48. [Google Scholar]
- 21.Sies H. Angew Chem Int Ed Engl. 1986;25:1058–1071. [Google Scholar]
- 22.Reddy V N. Exp Eye Res. 1990;50:771–778. doi: 10.1016/0014-4835(90)90127-g. [DOI] [PubMed] [Google Scholar]
- 23.Ziegler D M. Annu Rev Biochem. 1985;54:305–329. doi: 10.1146/annurev.bi.54.070185.001513. [DOI] [PubMed] [Google Scholar]
- 24.Hughes H, Jaeschke H, Mitchell J R. Methods Enzymol. 1991;186:681–685. doi: 10.1016/0076-6879(90)86167-t. [DOI] [PubMed] [Google Scholar]
- 25.Hissin P J, Hilf R. Anal Biochem. 1976;74:214–226. doi: 10.1016/0003-2697(76)90326-2. [DOI] [PubMed] [Google Scholar]
- 26.Racker E. J Biol Chem. 1951;190:685–696. [PubMed] [Google Scholar]
- 27.Brehe E, Burck H B. Anal Biochem. 1976;74:189–197. doi: 10.1016/0003-2697(76)90323-7. [DOI] [PubMed] [Google Scholar]
- 28.Akerboom T P, Sies H. Methods Enzymol. 1981;77:373–382. doi: 10.1016/s0076-6879(81)77050-2. [DOI] [PubMed] [Google Scholar]
- 29.Anderson M E. Methods Enzymol. 1985;113:548–555. doi: 10.1016/s0076-6879(85)13073-9. [DOI] [PubMed] [Google Scholar]
- 30.Kuninori T, Nishiyama J. Anal Biochem. 1991;197:19–24. doi: 10.1016/0003-2697(91)90349-x. [DOI] [PubMed] [Google Scholar]
- 31.Neuschwander-Tetri B A, Roll F J. Anal Biochem. 1989;179:236–241. doi: 10.1016/0003-2697(89)90121-8. [DOI] [PubMed] [Google Scholar]
- 32.Svardal A M, Mansoor M A, Ueland P M. Anal Biochem. 1990;184:338–346. doi: 10.1016/0003-2697(90)90691-2. [DOI] [PubMed] [Google Scholar]
- 33.Reed D J, Babson J R, Beatty P W, Brodie A E, Ellis W W, Potter D W. Anal Biochem. 1980;106:55–62. doi: 10.1016/0003-2697(80)90118-9. [DOI] [PubMed] [Google Scholar]
- 34.Morier-Teissier E, Mestdagh N, Bernier J-L, Henichart J-P. J Liq Chromatogr. 1993;16:573–596. [Google Scholar]
- 35.Nischiyama J, Kuninori T. Anal Biochem. 1984;138:95–98. doi: 10.1016/0003-2697(84)90774-7. [DOI] [PubMed] [Google Scholar]
- 36.Richie J P, Jr, Lang C A. Anal Biochem. 1987;163:9–15. doi: 10.1016/0003-2697(87)90085-6. [DOI] [PubMed] [Google Scholar]
- 37.Awasthi S, Ahmad F, Sharma P, Ahmad H. J Chromatogr Biomed Appl. 1992;584:167–173. doi: 10.1016/0378-4347(92)80572-8. [DOI] [PubMed] [Google Scholar]
- 38.Crawhall J C, Kalant D. Anal Biochem. 1988;172:479–483. doi: 10.1016/0003-2697(88)90470-8. [DOI] [PubMed] [Google Scholar]
- 39.Cook J A, Pass H I, Russo A, Iype S, Mitchell J B. Int J Radiat Oncol Biol Phys. 1989;16:1321–1324. doi: 10.1016/0360-3016(89)90307-6. [DOI] [PubMed] [Google Scholar]
- 40.Rice G C, Bump E A, Shrieve D C, Lee W, Kovacs M. Cancer Res. 1986;46:6105–6110. [PubMed] [Google Scholar]
- 41.Ublacker G A, Johnson J A, Siegel F L, Mulcahy R T. Cancer Res. 1991;51:1783–1788. [PubMed] [Google Scholar]
- 42.Cappiello M, Del Corso A, Camici M, Mura U. J Biochem Biophys Methods. 1993;26:335–341. doi: 10.1016/0165-022x(93)90034-l. [DOI] [PubMed] [Google Scholar]
- 43.Pascual P, Martinezlara E, Barcena J A, Lopez-Barea J, Toridio F. J Chromatogr. 1992;581:49–56. doi: 10.1016/0378-4347(92)80446-w. [DOI] [PubMed] [Google Scholar]
- 44.Beales D, Finch R, Mc Lean A E, Smith M, Wilson I D. J Chromatogr. 1981;226:498–503. doi: 10.1016/s0378-4347(00)86088-0. [DOI] [PubMed] [Google Scholar]
- 45.Alpert A J, Gilbert H F. Anal Biochem. 1985;144:553–562. doi: 10.1016/0003-2697(85)90153-8. [DOI] [PubMed] [Google Scholar]
- 46.Nakamura H, Tamura Z. Anal Chem. 1981;53:2190–2193. [Google Scholar]
- 47.Nakamura H, Tamura Z. Bunseki Kagaku. 1988;37:35–40. [Google Scholar]
- 48.Keller D A, Menzel D B. Anal Biochem. 1985;151:418–423. doi: 10.1016/0003-2697(85)90197-6. [DOI] [PubMed] [Google Scholar]
- 49.Morineau G, Azoulay M, Frappier F. J Chromatogr. 1989;467:209–216. doi: 10.1016/s0021-9673(01)93965-2. [DOI] [PubMed] [Google Scholar]
- 50.Minchinton A I. Int J Radiat Oncol Biol Phys. 1984;10:1503–1506. doi: 10.1016/0360-3016(84)90490-5. [DOI] [PubMed] [Google Scholar]
- 51.Burton N K, Aherne G W. J Chromatogr. 1986;382:253–257. doi: 10.1016/s0378-4347(00)83525-2. [DOI] [PubMed] [Google Scholar]
- 52.Velury S, Howell S B. J Chromatogr. 1988;424:141–146. doi: 10.1016/s0378-4347(00)81085-3. [DOI] [PubMed] [Google Scholar]
- 53.Iwamoto T, Yoshiura M, Iriyama K, Tomizawa N, Kurihara S, Lee T, Suzuki N. Jikeikai Med J. 1985;32:245–270. [Google Scholar]
- 54.Stein A F, Dills R L, Klaassen C D. J Chromatogr. 1986;381:259–270. doi: 10.1016/s0378-4347(00)83592-6. [DOI] [PubMed] [Google Scholar]
- 55.Dupuy D, Szabo S. J Liq Chromatogr. 1987;10:107–119. [Google Scholar]
- 56.Buchberger W, Winsauer K. Anal Chim Acta. 1987;196:251–254. [Google Scholar]
- 57.Iriyama K, Iwamoto T, Yoshiura M. J Liq Chromatogr. 1986;9:955–969. [Google Scholar]
- 58.Carro-Ciampi G, Hunt P G, Turner C J, Wells P G. J Pharmacol Methods. 1988;19:75–83. doi: 10.1016/0160-5402(88)90047-2. [DOI] [PubMed] [Google Scholar]
- 59.Megaw J M, Lee J, Lerman S. Lens Res. 1985;2:351–368. [Google Scholar]
- 60.Sacchetta P, DiCola D, Federici G. Anal Biochem. 1986;154:205–208. doi: 10.1016/0003-2697(86)90516-6. [DOI] [PubMed] [Google Scholar]
- 61.Eyer P, Podhradsky D. Anal Biochem. 1986;153:57–66. doi: 10.1016/0003-2697(86)90061-8. [DOI] [PubMed] [Google Scholar]
- 62.Beutler E, Srivastava S K, West C. Biochem Biophys Res Commun. 1970;38:341–347. doi: 10.1016/0006-291x(70)90718-7. [DOI] [PubMed] [Google Scholar]
- 63.Spector W S, editor. Handbook of Biological Data. Philadelphia: Saunders; 1956. [Google Scholar]
- 64.Johnson C A, Bergeim O. J Biol Chem. 1951;188:833–838. [PubMed] [Google Scholar]
- 65.Levy H L, Barkin E. J Lab Clin Med. 1971;78:517–523. [PubMed] [Google Scholar]
- 66.Liu A, Li T, Wang E. Anal Sci. 1995;11:597–603. [Google Scholar]
- 67.Jacobean S H, Molds P. Clin Nephrol. 1994;42:189–192. [Google Scholar]
- 68.Shadle S E, Hedman B, Hodgson K O, Solomon E I. J Am Chem Soc. 1995;117:2259–2272. doi: 10.1021/ja002183v. [DOI] [PubMed] [Google Scholar]
- 69.Waldo G S, Carlson R M K, Moldowan J M, Peters K E, Penner-Hahn J E. Geochim Acta. 1991;55:801–814. [Google Scholar]
- 70.Stern E, Heald S. Rev Sci Instr. 1979;50:1579–1582. doi: 10.1063/1.1135763. [DOI] [PubMed] [Google Scholar]
- 71.Lytle F W, Greegor R B, Sandstrom D R, Marques E C, Wong J, Spiro C L, Huffman G P, Huggins F E. Nucl Instr Methods. 1984;226:542. [Google Scholar]
- 72.Powers L, Blumberg W E, Chance B, Barlow C H, Leigh J S, Jr, Smith J, Yonetani T, Vik S, Peisach J. Biochim Biophys Acta. 1979;546:520–538. doi: 10.1016/0005-2728(79)90085-9. [DOI] [PubMed] [Google Scholar]
- 73.Piccoli G, Fiorani M, Biagiarelli B, Palma F, Potenya L, Amicucci A, Stocchi V. J Chromatogr A. 1994;676:239–246. doi: 10.1016/0021-9673(94)80465-6. [DOI] [PubMed] [Google Scholar]
- 74.Malloy M H, Rassin D K, Gaull G E. Anal Biochem. 1981;113:407–415. doi: 10.1016/0003-2697(81)90095-6. [DOI] [PubMed] [Google Scholar]
- 75.Mills B J, Richie J P, Jr, Lang C A. Anal Biochem. 1990;184:263–267. doi: 10.1016/0003-2697(90)90678-3. [DOI] [PubMed] [Google Scholar]
- 76.Fahey R C. In: Glutathione: Chemical, Biochemical and Medical Aspects. Dolphin D, Avramovic O, Poulson R, editors. New York: Wiley; 1989. pp. 304–337. [Google Scholar]