Abstract
Genetic factors are presumed to contribute to individual differences in alcohol expectancies, but few studies have examined this possibility directly. Genes with functional implications for alcohol metabolism and sensitivity are good candidates for studies of genetic effects on expectancy processes. The present study evaluated influences of ALDH2 and ADH1B genotypes on alcohol expectancies and drinking behavior in a sample of Asian-American young adults. In addition to assessing global alcohol expectancies, a measure of physiological expectancies was developed to evaluate an expectancy phenotype specific to the mechanism by which ALDH2 and ADH1B variations presumably influence drinking behavior. Compared to individuals with the ALDH2*1/*1 genotype, those with the ALDH2*2 allele reported greater negative alcohol expectancies, greater expectancies for physiological effects of alcohol and lower rates of alcohol use. ADH1B was not associated with alcohol expectancies or drinking behavior. Hierarchical models showed that demographic factors, ALDH2 genotype and expectancy variables explained unique variance in drinking outcomes. Mediation tests showed significant indirect effects of ALDH2 on drinking frequency and peak lifetime consumption via expectancies. These results provide support for influences of genetic factors and alcohol sensitivity on alcohol-related learning and suggest the importance of developing biopsychosocial models of drinking behavior in Asian Americans.
Keywords: ALDH2, ADH1B, genetic, alcohol expectancies, Asian Americans, college students
Psychiatric epidemiology studies have established that genetic and environmental factors each account for substantial variability in the risk for alcohol use and dependence (reviewed in Heath, 1995; McGue, 1994; 1999). A consequent goal is to enhance understanding of how genetic and psychosocial variables jointly influence drinking behavior (Dick et al., 2006; Heath & Nelson, 2002). Although biological and behavioral research efforts have tended to progress independently (Young, Lawford, Feeney, Ritchie, & Noble, 2004), there is increasing theoretical and empirical justification for integrating biological and behavioral approaches (e.g., Beauchaine, Neuhaus, Brenner, & Gatzke-Kopp, 2008; Bryan, Hutchison, Seals, & Allen, 2007; Hutchison et al., 2007; Plomin & Crabbe, 2000). This goal will be facilitated by the use of theoretical frameworks capable of accommodating hypotheses from both perspectives. Alcohol expectancy theory (Goldman, Darkes, & Del Boca, 1999; Goldman, Darkes, Reich, & Brandon, 2006) implicates cognitive and learning mechanisms as intermediary processes that may partly account for genetic influences on alcohol use behavior. In offering a bio-behavioral account of drinking behavior, expectancy theory serves as a promising theoretical framework for translational studies of alcohol use.
The notion of biological influences on alcohol-related learning is supported by preliminary evidence that genetic factors account for variability in alcohol expectancies. Relevant twin studies suggest that additive genetic factors explain a modest but significant proportion (up to 20-40%) of individual variability in self-reported expectancies, although heritable effects appear to differ by drinking status and expectancy subtype (Agrawal et al., 2007; Gabrielli & Plomin, 1985; Slutske et al., 2002; Vernon et al., 1996).1 Genetic influences on expectancies can also be inferred from their association with biologically based risk markers for alcohol dependence. For instance, expectancies are shown to partly mediate effects of dispositional risk factors (e.g., behavioral undercontrol, disinhibition and sensation seeking; Anderson, Smith, & Fischer, 2003; Finn, Sharkansky, Brandt, & Turcotte; 2000; Henderson et al., 1994; McCarthy, Kroll, & Smith, 2001; Schuckit & Smith, 2000) and familial risk for alcohol dependence (Sher, Walitzer, Wood, & Brent, 1991; Sher et al., 1996) on drinking outcomes. Among the most widely studied risk markers for alcohol dependence is level of response (LR) to alcohol (Schuckit, Smith, & Kalmijn, 2004), a heritable phenotype that predicts the risk for alcohol dependence prospectively (Heath et al., 1999; Schuckit & Smith, 1996) and may predict drinking in part via expectancies (Schuckit et al., 2005).
Expectancy research has begun to incorporate molecular genetic approaches. Not surprisingly, early studies have focused on genes with direct implications for alcohol metabolism, in particular ALDH2 and ADH1B. ALDH2 encodes the mitochondrial aldehyde dehydrogenase (ALDH) enzyme, the primary catalyst for the removal of acetaldehyde, a toxic byproduct of ethanol metabolism. The ALDH2*2 allele, common in 30-50% of individuals of northeast Asian descent, encodes a functionally inactive ALDH enzyme subunit that leads to impaired acetaldehyde removal. ALDH2*2 is associated with increased sensitivity to alcohol, as indexed by symptoms like flushing, tachycardia and nausea (Thomasson & Li, 1993). In turn, ALDH2*2 is associated with a significantly reduced likelihood of heavy drinking and alcohol dependence (Luczak et al., 2006; Wall & Ehlers, 1995). ADH1B is one of several genes that encode the alcohol dehydrogenase (ADH) enzymes, which regulate oxidation of ethanol to acetaldehyde (Edenberg, 2007). The ADH1B*2 and ADH1B*3 variants, prevalent among people of Asian and African descent, respectively, encode ADH enzyme subunits with a substantially greater turnover rate for converting ethanol to acetaldehyde relative to the common ADH1B*1 variant. ADH1B*2 and ADH1B*3 have been associated with increases in some measures of acute sensitivity to alcohol (Cook et al., 2005; Duranceaux et al., 2006; McCarthy, Pedersen, Lobos, Todd, & Wall, under review) as well as a reduced risk of alcohol dependence (Ehlers et al., 2007; Hasin et al., 2002; Luczak et al., 2006; Whitfield, 1997) compared to ADH1B*1. Presumably, the protection afforded by ALDH2*2, ADH1B*2 and ADH1B*3 occurs via a pathway involving increases in transient acetaldehyde during alcohol metabolism and greater sensitivity to alcohol, although empirical support for acetaldehyde as a mediating mechanism exists only for ALDH2*2 (Wall, 2005).
Differences in alcohol sensitivity can theoretically influence how drinking events are experienced, interpreted and stored in memory (Brown, Tate, Vik, Haas & Aarons, 1999; Goldman et al., 2006). It follows that functional variations in alcohol metabolizing genes are reasonable candidates for studies of genetic influences on expectancy processes. In an initial study of ALDH2 and alcohol expectancies, ALDH2*2 was associated with lower endorsement of positive expectancies, which partially accounted for ALDH2 effects on drinking quantity in women (McCarthy, Wall, Brown and Carr, 2000). In a follow-up study using an alcohol challenge paradigm, ALDH2*2 predicted greater expectancies for cognitive and behavioral impairment among men, an association that was fully explained by response to alcohol in the laboratory (McCarthy, Brown, Carr & Wall, 2001). Associations of ALDH2 with expectancies have also been reported in a study of alcohol-dependent patients in Taiwan (Hahn et al., 2006). These initial studies did not examine the additional influence of ADH1B. Others, however, have found that ADH1B*2 was associated with retrospectively reported physiological symptoms (Carr et al., 2002; Wall et al., 2005). Also, ADH1B*3 was associated with alcohol expectancies in a study of African Americans (Ehlers et al., 2003). 2 Theory and research therefore suggest that genetic differences in alcohol metabolism and sensitivity could influence expectancy development.
An imperative in genetic association studies is maximal precision in the definition and measurement of phenotypes (e.g., Hines, Ray, Hutchison, & Tabakoff, 2005; Hutchison, Stallings, McGeary, & Bryan, 2004). Similarly, expectancy theorists have stressed the importance of optimal measurement specificity (Goldman et al., 1999). Notably, previous attempts to study genetic influences on expectancies have used established expectancy instruments (e.g., Brown et al., 1980; Fromme et al., 1993) with item content spanning several social and behavioral domains. Depending on the genetic variable in question and its proposed mechanism of influence, traditional expectancy measures may lack sensitivity for detecting genetic influences. For example, although ALDH2 and ADH1B variations are proposed to influence drinking via differences in physiological responses to alcohol, the most popular expectancy instruments lack substantive item content in this domain. Tailoring expectancy measures to the proposed pathway of genetic influence would theoretically increase the likelihood of detecting genetic associations.
The current study examined genetic influences on alcohol-related learning by evaluating associations of ALDH2 and ADH1B with alcohol expectancies and drinking behavior among individuals of northeast Asian descent. In addition to using a traditional alcohol expectancy instrument, we developed a measure of physiological alcohol expectancies (Study 1) in order to evaluate a relatively narrower expectancy phenotype with theoretical relevance for the mechanism by which ALDH2 and ADH1B variations are presumed to influence drinking behavior. Study 2 aimed to examine joint influences of genetic, expectancy and demographic variables on drinking outcomes and to evaluate mediating effects of genetic variables on drinking outcomes via expectancies. We predicted that ALDH2*2 and ADH1B*2 would each be associated with lower positive expectancies and higher negative expectancies as measured with an established instrument (Fromme et al., 1993). It was also anticipated that ALDH2*2 and ADH1B*2 would predict higher expectancies for physiological drinking outcomes and lower rates of drinking. We expected that genetic, expectancy and demographic factors would each explain unique variance in drinking outcomes and that expectancies would partly mediate the effects of ALDH2 and ADH1B on alcohol use.
Study 1
Method and Results
The aims of Study 1 were to generate information about expected physiological drinking outcomes, to develop a quantitative measure to assess these expectancies, and to evaluate scale factor structure. Two independent samples were recruited, each of which included participants from introductory psychology courses who reported 100% northeast Asian heritage and lifetime alcohol consumption. In the item generation phase, a sample of 91 participants (mean age = 19.75, SD = 1.45, 50% female) volunteered in exchange for course extra credit. Participation took place in small groups and included completing an open-ended questionnaire designed to elicit data about physiological alcohol expectancies. Specifically, participants were asked to report the physiological effects (e.g., changes in physical feelings or bodily sensations) that they expected following alcohol consumption. Participants were instructed to list as many relevant outcomes as possible and to be as specific as possible in their responses. Overall, a total of 693 expectancies were generated. All responses were transcribed into an electronic database and examined to derive common themes/categories based on participant responses. Expectancies that were judged not to reflect physiological outcomes were categorized separately and omitted from consideration during the scale construction phase. Based on participants' responses a preliminary version of the Physiological Effects of Alcohol Questionnaire (PEAQ) was developed, which included 69 items reflective of the content that emerged during the item generation stage. The questionnaire was designed to parallel the structure of the Comprehensive Effects of Alcohol Questionnaire (Fromme et al., 1993) such that expectancy items were preceded by the stem “If I were under the influence from drinking alcohol…”, and followed by scales assessing both the likelihood (1 = disagree, 4 = agree) and the subjective evaluation (1 = bad, 5 = good) of each drinking outcome.
The preliminary questionnaire was then administered to the second sample of participants (n = 131; 59% female). Participants' responses to the 69 PEAQ expectancy items were subjected to exploratory factor analysis (EFA) using principal components extraction and promax rotation. Consistent with previous research, expectancy evaluation items were not included in the initial EFA (Fromme, Stroot, & Kaplan, 1993). Examination of eigenvalues and visual inspection of scree plots identified four primary factors that cumulatively accounted for 50% of the common variance, each with an eigenvalue > 2.5. The initial step in item reduction consisted of retaining items that demonstrated high (> .40) loadings on one, but not more than one of these factors. Thirty-seven items met these criteria and were retained. These items were further reduced to five items per subscale, using high factor loadings (range: .41 – 1.0) as the primary selection criterion and non-redundancy of item content as a secondary criterion.
Internal consistency analyses conducted with the resulting 5-item subscales indicated no items whose deletion improved subscale reliability. Mean inter-item correlations across the subscales ranged from .46 - .57, approximating the range recommended for a measure assessing a relatively circumscribed construct (Clark & Watson, 1995). Inter-scale correlations showed that all subscales were moderately and significantly correlated (range = .34 - .66). Based on item content the initial four factors were labeled flushing (α = .91), sensorimotor impairment (α = .87), dizziness/nausea (α = .85), and physical sensations (α = .78). Internal consistencies (α) for the evaluation subscales corresponding with these respective expectancy subscales were .63, .92, .87 and .88. We added three additional items to form a subscale for tachycardia, labeled heart rate. These items were added on theoretical grounds because elevated heart rate following alcohol consumption has been observed among individuals with the ALDH2*2 allele (e.g., Peng et al., 1999).
Study 2
Method
Participants
Study 2 participants included 193 of 200 undergraduates enrolled in a study of genetic and environmental influences on alcohol use. Individuals were recruited after they completed a web-based survey of alcohol use norms and behavior that was sent to 4000 randomly selected undergraduates as part of a different study. People who completed this survey, indicated 100% Chinese, Korean or Japanese heritage and consented to follow-up contact received email and phone invitations to participate in the current study. A total of 292 students qualified and were targeted for recruitment via email and/or phone. Recruitment continued until a sample of 200 was reached; 48 students declined participation. Of the 193 participants included in the present analyses, 53.4% were female and the mean age was 20.17 years (SD = 1.55). The ethnic breakdown was 58.5% Chinese, 32.6% Korean and 8.8% Japanese.
Procedure
All participants who enrolled in the study attended a laboratory visit to provide informed consent and a fingertip-puncture blood sample for DNA analysis. Shortly following the laboratory visit participants received an email with a link to complete a web-based survey assessing alcohol use, expectancies and related constructs. Participants logged onto a secure server using a randomly generated identification number to complete the survey. Of the 200 participants enrolled in the study, 193 (96.5%) completed this survey and were included in the present analyses. Previous research has demonstrated the equivalence of web-based versus traditional administration of alcohol questionnaires in terms of reliability, validity and participant responses (Miller et al., 2002). Participants received $15 for providing a blood sample at the laboratory visit and another $15 upon completing the online survey.
Measures
Alcohol expectancies
Alcohol expectancies were measured with the 38-item Comprehensive Effects of Alcohol questionnaire (CEOA; Fromme et al., 1993). The CEOA has demonstrated reliability and validity in college students and adolescents (Fromme et al., 1993; 2000) and has been used in previous research examining relationships between ALDH2 status and expectancies (McCarthy et al., 2000; 2001). The CEOA has been found to have two higher-order factors, positive and negative expectancies. The positive expectancy domain comprises four subfactors (sociability, tension reduction, liquid courage, sexuality) and the negative expectancy domain comprises three subfactors (cognitive/behavioral impairment, risk/aggression, self perception) (Fromme et al., 1993). Other researchers have replicated this factor structure (Valdivia & Stewart, 2005). Each expectancy item is rated on two components: the perceived likelihood of the drinking outcome (1 = disagree, 4 = agree) and the subjective evaluation of the outcome (1 = bad, 5 = good). The subjective evaluation component reflects the perspective that behaviors are a function of both anticipated likelihood and perceived desirability of an expected outcome (Fishbein & Ajzen, 1975). Expectancy evaluations have been shown to predict variance in drinking above and beyond that explained by expectancies alone (Burden & Maisto, 2000; Leigh, 1987).
Participants' anticipated physiological drinking outcomes were measured with the 23-item Physiological Effects of Alcohol Questionnaire (PEAQ), developed in Study 1. A confirmatory factor analysis (CFA) of PEAQ expectancy items was conducted using the factor structure observed in Study 1. Full information maximum likelihood estimation was used to estimate the model. The independence test that all variables were uncorrelated was not tenable [χ2 (276) = 3419.95, p < .001]. The five-factor model was significantly better than the independence model [χ2 diff (56) = 2726.88, p < .001] and provided reasonable fit with no items appearing problematic [χ2 (220) = 693.07, p = .01, CFI = .96, RMSEA = 0.105, 95% CI = .096 - .114]. Scale reliability coefficients (α) in this sample were: .91 for flushing, .87 for sensorimotor impairment, .89 for dizziness/nausea, .85 for physical sensations and .86 for heart rate (scale items are listed in the Appendix). Participants completed scales assessing both the likelihood (1 = disagree, 4 = agree) and subjective evaluation (1 = bad, 5 = good) of each physiological drinking outcome. Internal consistencies (α) for the evaluation components of the aforementioned expectancy subscales were .93, .92, .89, .91 and .84 respectively. Pearson correlations evaluated the bivariate association of each expectancy subscale with its corresponding evaluation subscale. Expectancies and evaluations for the heart rate subscale were significantly correlated (r = .79, p < .001); there were no significant correlations for any other subscales. Consistent with procedures in Study 1, a separate CFA was not conducted on the evaluation items because the expectancy items are considered of primary importance in the context of scale development.
Drinking behavior
Drinking behavior was assessed using the Alcohol Use Disorder Identification Test (Babor, Higgins-Biddle, Saunders, & Monteiro, 2001) and the National Institute on Alcohol Abuse and Alcoholism question set (NIAAA, 2003). Three outcomes were selected for the current analyses: drinking frequency, heavy episodic drinking and peak lifetime consumption. Drinking frequency was assessed with the AUDIT item: How often do you have a drink containing alcohol? Response options range from 0 (never) to 4 (4 or more times per week). Frequency of heavy drinking episodic drinking was assessed with the question: During the last 3 months how often did you have [4 or more (females) / 5 or more (males)] drinks containing any kind of alcohol within a two-hour period? Response options ranged from 0 (never) to 9 (every day). Peak lifetime consumption was assessed with the item: During your lifetime, what is the maximum number of drinks containing alcohol that you drank within a 24-hour period? (NIAAA, 2003).
Genotyping
Blood samples were sent to the Alcohol Research Center at Indiana University for genotyping. DNA was isolated using the “HotSHOT” method (Truett et al., 2000), which is a fast and cost-effective way to isolate PCR-quality genomic DNA. Further details on genotyping procedures for this study are reported elsewhere (Hendershot et al., 2009). ALDH2 and ADH1B status were each coded 0, 1 or 2 according the number of variant (*2) alleles.
Analytic plan
Analyses were structured to address three aims: 1) to assess the relation of ALDH2 and ADH1B genotypes to expectancies and drinking behavior, 2) to evaluate the joint influence of genetic factors, expectancies and demographic factors on drinking behavior, and 3) to evaluate whether expectancies mediated associations between genetic factors and drinking. Aims 1 and 3 were addressed using correlation and regression analyses and included only those participants who reported lifetime alcohol consumption (n = 171). Lifetime abstainers were omitted because ALDH2 and ADH1B would theoretically be unrelated to alcohol expectancies in the absence of any drinking experience. Aim 2, which involved evaluation of a multivariate model of alcohol use that included demographic and expectancy factors as predictors, was addressed using the full sample because demographic and expectancy factors can influence the decision to abstain from alcohol use.
Results
Genotype results for the 200 participants who enrolled in the study showed that 105 (52.5%) had the ALDH2*1/*1 genotype, 73 (36.5%) had the ALDH2*1/*2 genotype, and 22 (11%) had the ALDH2*2/*2 genotype. For ADH1B, 19 (9.5%) were ADH1B*1/*1, 73 (36.7%) were AHD1B*1/*2, and 107 (53.8%) were ADH1B*2/*2. The distributions of ALDH2 and ADH1B genotypes conformed to Hardy-Weinberg Equilibrium. One person had missing data for ADH1B. Among participants reporting lifetime alcohol consumption (n=171), mean response to the drinking frequency item was 1.42 (SD = .89), mean response to the heavy episodic drinking item was 0.82 (SD = 1.34), and the mean value for peak alcohol consumption was 7.16 (SD = 6.36).
Associations of CEOA and PEAQ subscales
Preliminary analyses evaluated correlations between CEOA and PEAQ expectancies to provide an initial test of convergent/discriminant validity of the PEAQ. All PEAQ subscales were significantly and positively correlated with CEOA negative expectancy subscales (Table 1). CEOA positive expectancies did not relate significantly to PEAQ subscales with the exception of one CEOA positive expectancy subscale, sexuality, which was positively correlated with the sensorimotor impairment subscale (r = .18), physical sensations subscale (r = .15) and total score (r = .15) on the PEAQ (p's < .05).
Table 1.
Correlations between CEOA negative expectancy subscales and PEAQ subscales
| CEOA | PEAQ | |||||
|---|---|---|---|---|---|---|
| Flushing | Sensorimotor impairment | Dizziness/ Nausea | Physical Sensations | Heart rate | PEAQ total | |
| Negative | .40** | .69** | .68** | .56** | .46** | .68** |
| CBI | .49** | .73** | .71** | .57** | .53** | .74** |
| Risk/aggression | .07 | .26** | .27** | .26** | .15* | .25** |
| Self perception | .15* | .42** | .38** | .34** | .20** | .37** |
Note. CBI = CEOA Cognitive-Behavioral Impairment subscale.
p <.05,
p <.01.
Bivariate associations of ALDH2 and ADH1B with alcohol expectancies
Bivariate associations of ALDH2 and ADH1B with alcohol expectancies were examined among lifetime drinkers. Genotypes were entered as 0, 1 or 2, corresponding to the number of *2 alleles, and examined in relation to all CEOA and PEAQ expectancy and evaluation subscales. Individuals with the ALDH2*2 allele reported significantly higher scores for CEOA global negative and cognitive-behavioral impairment (CBI) expectancies. ALDH2*2 allele also predicted significantly lower expectancy evaluations for the CBI subscale; thus, participants with ALDH2*2 reported both an increased likelihood and more negative appraisals of drinking outcomes related to CBI. ALDH2 status was not significantly associated with any positive expectancy subscales of the CEOA. Examination of PEAQ scores showed that the ALDH2*2 allele predicted significantly higher physiological expectancies (measured by the PEAQ total score) and higher expectancies on 3 PEAQ subscales (flushing, dizziness/nausea and heart rate). Overall, significant correlations between ALDH2 status and expectancies appeared relatively higher in magnitude for physiological than for CEOA expectancies. ADH1B was not associated with any expectancy scales (Table 2).
Table 2.
Bivariate correlations of ALDH2 and ADH1B with alcohol expectancies and evaluations
| Expectancies | Evaluations | |||
|---|---|---|---|---|
| CEOA | ALDH2 | ADH1B | ALDH2 | ADH1B |
| Negative | .15* | -.04 | -.11 | -.03 |
| CBI | .16* | -.05 | -.17* | -.05 |
| Risk/aggression | .03 | .08 | -.06 | .01 |
| Self-perception | .11 | -.08 | .00 | -.05 |
| PEAQ | ||||
| Flushing | .49** | -.02 | .11 | -.03 |
| Sensorimotor Impariment | .02 | -.10 | -.02 | -.05 |
| Dizziness/nausea | .24** | -.05 | -.01 | .01 |
| Physical sensations | .09 | -.07 | .12 | .00 |
| Heart rate | .42** | .02 | .08 | .01 |
| Total score | .31** | -.06 | .07 | -.01 |
Note. ALDH2 and ADH1B coded 0, 1, 2 corresponding with number of variant (*2) alleles. CEOA = Comprehensive Effects of Alcohol Questionnaire. CBI = CEOA Cognitive-Behavioral Impairment subscale. PEAQ = Physiological Effects of Alcohol Questionnaire.
p < .05
p < .01.
Additive influences of demographic, expectancy and genetic factors on drinking outcomes
Using hierarchical regression we proceeded to evaluate the joint effects of genetic and expectancy variables on drinking outcomes while controlling for demographic factors. Models were run separately for each drinking outcome and included six steps. Step 1 included gender and ethnicity. Ethnicity was entered as a dummy-coded variable contrasting the Chinese group (coded 0) with the Korean and Japanese groups (both coded 1). This scheme was used because Chinese had the lowest drinking rates and the Japanese group was too small to evaluate separately. Step 2 included ALDH2 and ADH1B. Steps 3 and 4 included PEAQ expectancies (total score) and evaluations, respectively. Step 5 included CEOA positive and negative expectancies and Step 6 included the evaluation components of these scales. Models included the global CEOA and PEAQ scores (rather than including the expectancy and evaluation scores for all subscales) in the interest of parsimony. PEAQ expectancies were entered in the model prior to CEOA expectancies because the latter measure is more comprehensive and is likely to partly subsume physiological expectancies, as was suggested by the significant positive correlations between PEAQ scores and all CEOA negative subscales. All expectancy variables were mean centered prior to analyses (Aiken & West, 1991).
Results from hierarchical analyses are presented in Table 4. Step 1 of the model was significant for one outcome (maximum drinks lifetime), with female gender and Chinese ethnicity predicting lower drinking. Step 2 was significant for heavy episodic drinking and peak lifetime consumption, with ALDH2*2 showing a significant protective effect on these outcomes. ADH1B was not predictive of any drinking outcomes. Step 3 (PEAQ total score) explained unique variance for drinking frequency and peak lifetime consumption, such that higher physiological expectancies predicted lower rates of drinking. Step 4 (PEAQ evaluations) was significant for all drinking outcomes, such that relatively more negative appraisals of physiological outcomes predicting decreased drinking. The addition of CEOA positive and negative expectancies at Step 5 also predicted unique variance for all outcomes, with both positive and negative expectancies generally predicting significant variance in drinking. Finally, the addition of positive and negative CEOA evaluations at Step 6 explained unique variance for one drinking outcome (frequency). The overall model explained 26% of the variance in drinking frequency, 19% of the variance in heavy episodic drinking and 23% of the variance in peak lifetime consumption.
Table 4.
Results of mediation tests evaluating indirect effects of ALDH2 on alcohol use via expectancies.
| Mediator | α*β | SE α*β | 95% CI (lower) | 95% CI (upper) |
|---|---|---|---|---|
| Drinking Frequency | ||||
| CEOA Negative | -0.060* | 0.034 | -0.133 | -0.002 |
| CEOA CBI | -0.060* | 0.032 | -0.133 | -0.004 |
| CEOA CBI Evaluations | -0.029 | 0.021 | -0.079 | 0.003 |
| PEAQ Total score | -0.069* | 0.036 | -0.147 | -0.007 |
| PEAQ Dizziness/Nausea | -0.065* | 0.032 | -0.136 | -0.013 |
| Maximum Drinks | ||||
| CEAO Negative | -0.204 | 0.148 | -0.555 | 0.009 |
| CEOA CBI | -0.266* | 0.169 | -0.656 | -0.010 |
| PEAQ Total score | -0.445* | 0.245 | -0.980 | -0.022 |
| PEAQ Flushing | -0.749 | 0.401 | -1.575 | 0.005 |
| PEAQ Dizziness/Nausea | -0.321* | 0.196 | -0.763 | -0.003 |
| PEAQ Heart Rate | -0.062 | 0.322 | -0.706 | 0.573 |
Note. CEOA = Comprehensive Effects of Alcohol Questionnaire. Negative = global negative expectancies. CBI = Cognitive behavioral impairment. PEAQ = Physiological Effects of Alcohol Questionnaire. α*β = product of coefficients (estimate of the indirect effect). CI = asymmetric confidence interval.
p <.05.
Evaluating mediating relationships among genotype, expectancies and drinking
We next conducted mediation analyses to evaluate indirect effects of ALDH2 on drinking outcomes via expectancies. Our consideration of multiple CEOA and PEAQ expectancy and evaluation subscales, as well as three drinking outcomes, yielded a sizable number of potential mediation models. To limit the number of models tested to a reasonable number, mediation tests were only conducted for expectancy or expectancy evaluation subscales that initially showed significant associations with ALDH2 and a given drinking outcome. Of the expectancy subscales that were significantly related to ALDH2, two CEOA subscales (negative expectancies and cognitive behavioral impairment expectancies) and four PEAQ subscales (flushing, dizziness/nausea, heart rate and total score) had significant, negative associations with at least one drinking outcome (Pearsons' r ranged from -.19 to -.32 for the CEOA subscales and from -.16 to -.29 for the PEAQ subscales; all ps < .05). In addition, the one expectancy evaluation subscale showing a significant association with ALDH2 (CEOA cognitive behavioral impairment) showed a significant correlation with drinking frequency (r = .16, p < .05). Therefore, 11 potential mediation models were tested: 5 for drinking frequency (with CEOA negative expectancies, CEOA CBI expectancies, CEOA CBI evaluations, PEAQ total score and PEAQ dizziness/nausea expectancies examined as mediators) and 6 for peak lifetime consumption (with CEOA negative expectancies, CEOA CBI expectancies, PEAQ total score, PEAQ flushing expectancies, PEAQ dizziness/nausea expectancies and PEAQ heart rate expectancies examined as mediators).
Each mediation model consisted of a two-part linear regression analysis in which a) expectancy was regressed on ALDH2, and b) drinking was regressed on expectancy with ALDH2 entered simultaneously in the model. Finally, to test for mediation, we utilized the product of coefficients method (α*β), which has been shown to be the most powerful test of mediation in simulation studies (MacKinnon, Lockwood, Hoffman, West & Sheets, 2004). Although the individual coefficients comprising the indirect effect are normally distributed, their product is not (Meeker, Cornwell, & Aroian, 1981). Thus, standard calculations for the standard error of the indirect effect that assume normality, such as Sobel's method (Sobel, 1982) can lead to an underestimation of the mediated effect (MacKinnon et al., 2004). For the current study, we tested for significance of the mediated effects by computing asymmetric confidence intervals with the program PRODCLIN using a 95% confidence criterion, described by MacKinnon, Fritz, Williams and Lockwood (2007). A significant mediated effect was supported if the confidence interval excluded zero. A significant indirect effect of ALDH2 via expectancies was supported in 7 of the 11 models tested. Results supported that the protective effect of ALDH2*2 on drinking outcomes included a significant indirect effects via higher negative expectancies and higher physiological expectancies. Although the model evaluating PEAQ dizziness/nausea expectancies as a mediator of peak lifetime consumption suggested a marginal effect of expectancies on drinking (p = .058) when controlling for ALDH2 the indirect effect was significant. Mediation test statistics are presented in Table 4 and models yielding significant mediating effects are presented in Figures 1-2.
Figure 1.
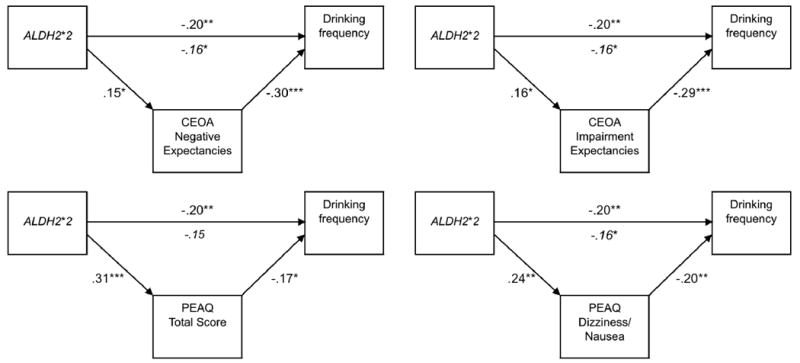
Mediation models supported for ALDH2, expectancies and drinking frequency. CEOA = Comprehensive Effects of Alcohol Questionnaire. PEAQ = Physiological Effects of Alcohol Questionnaire. Values reflect standardized regression coefficients (betas). Associations of expectancy variables and drinking variables were evaluated controlling for ALDH2. Italicized values reflect the association of ALDH2 and drinking with expectancies included in the model. *p <.05, ** p <.01, *** p <.001.
Figure 2.
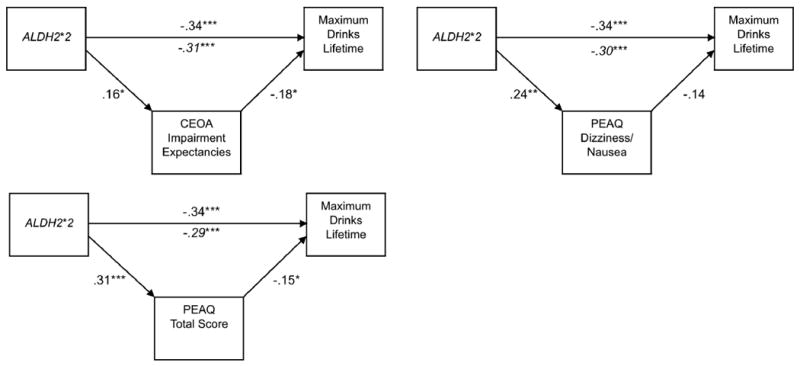
Mediation models supported for ALDH2, expectancies and peak lifetime consumption. CEOA = Comprehensive Effects of Alcohol Questionnaire. PEAQ = Physiological Effects of Alcohol Questionnaire. Values reflect standardized regression coefficients (betas). Associations of expectancy variables and drinking variables were evaluated controlling for ALDH2. Italicized values reflect the association of ALDH2 and drinking with expectancies included in the model. *p <.05, ** p <.01, *** p <.001.
Discussion
This study evaluated potential biological influences on alcohol-related learning by examining alcohol expectancies in relation to genetic variants with functional implications for alcohol metabolism and sensitivity. The current study extends previous research by examining both ALDH2 and ADH1B in relation to expectancies and by including an expectancy measure that was relatively specific to the theoretical mechanism of ALDH2 and ADH1B influences on drinking behavior. The ALDH2*2 allele was associated with higher self-reported expectancies in global negative, cognitive-behavioral impairment and physiological domains. Hierarchical models showed additive influences of ALDH2 and expectancy variables on drinking behavior and mediation analyses showed indirect effects of ALDH2 on drinking outcomes via expectancies.
A specific aim of this study was to evaluate an expectancy construct that was relevant to the mechanism by which ALDH2 and ADH1B are presumed to influence drinking. The physiological expectancy measure developed for this study showed significant associations with ALDH2 status and with CEOA negative expectancy subscales, providing some initial support for its construct and convergent validity. Physiological expectancies showed somewhat stronger direct associations with ALDH2 than did CEOA expectancies, although CEOA expectancies generally showed stronger associations with drinking. The latter finding may be not be surprising in that a comprehensive measure assessing several social, cognitive and behavioral domains would presumably explain more variance in comparison to a more focused measure. Both CEOA and PEAQ expectancies explained variance in drinking outcomes, and both accounted for significant indirect effects of ALDH2 on alcohol use. These findings suggest that negative expectancies, including those related to physiological outcomes, are important predictors of drinking in this population. The results also provide preliminary evidence that tailoring expectancy measures to the theoretical mechanism of genetic influence might be helpful in the context of future genetic association studies.
The current study also extended previous research by examining associations of genetic factors with expectancy evaluations (subjective appraisals of anticipated drinking outcomes). Compared to individuals with the ALDH2*1/*1 genotype, those with the ALDH2*2 allele reported significantly more negative evaluations of cognitive behavioral impairment expectancies. It is also noteworthy that subjective appraisals of physiological expectancies consistently predicted drinking behavior after controlling for other variables, generally accounting for more variance than physiological expectancies themselves. This result could relate partly to the fact that ALDH2*2 is associated with objective symptoms (i.e., skin flushing) as well as subjective physiological changes following alcohol consumption. The anticipation of overt physiological reactions associated with ALDH2*2 might deter drinking for social or interpersonal reasons, irrespective of anticipated subjective physiological experiences (e.g., Sue & Nakamura, 1984). If so, subjective appraisals of physiological expectancies could be important determinants of whether (or in what contexts) drinking takes place. Expectancy evaluations related to physiological outcomes are potentially important predictors of drinking behavior in this population.
ADH1B genotype was not related to expectancies or drinking in this sample. Some studies have linked ADH1B*2 to retrospective reports of physiological symptoms (e.g., Carr et al., 2002; W.-J. Chen et al., 1998; Takeshita et al., 2001; Wall et al., 2005) whereas others have found no influence of ADH1B on measures of alcohol sensitivity (Duranceaux et al., 2008; Shea et al., 2001). Although the protective effect of ADH1B*2 on the risk for alcohol dependence is established (Luczak et al., 2006), many studies have focused on adult samples comprised of alcohol-dependent participants and controls. Of studies examining ADH1B in college populations, some have found protective effects of ADH1B*2 (Wall et al., 2005) whereas others have not (Carr et al., 2002; Shea et al., 2001). There is some evidence that the effects of ADH1B may be more pronounced among heavier drinkers (e.g., Heath et al., 2001; Neumark et al., 1998), which may explain inconsistent findings in younger samples and the lack of significant effects in this study. Additionally, because in vivo increases in blood acetaldehyde have been associated with ALDH2*2 but not ADH1B*2 (reviewed in Peng & Yin, 2009), stronger associations of expectancies with ALDH2 compared with ADH1B would be predicted.
The examination of expectancies as a mediator of genetic influences might raise the question of whether expectancies can be considered a candidate endophenotype for alcohol dependence. Expectancies could be viewed as partly fulfilling some of the criteria for an endophenotype (Bearden & Freimer, 2006; Gottesman & Gould, 2003) in that they show heritability (Agrawal et al., 2007; Slutske et al., 2002), anticipate drinking initiation (Christiansen, Smith, Roehling, & Goldman, 1989), predict risk for alcohol dependence and family history of the disorder (Kilbey, Downey, & Breslau, 1998; Sher, Walitzer, Wood, & Brent, 1991) and may predict alcohol use disorder severity among individuals from affected families (Schuckit & Smith, 2001). However, it is important to note that useful endophenotypes are ideally state-independent, moderately or highly heritable and governed primarily by biological rather than environmental processes (Bearden & Freimer, 2006; Gottesman & Gould, 2003; Gould & Gottesman, 2006). Expectancies, as traditionally defined, are unlikely to satisfy these criteria because they show substantial environmental influence (Agrawal et al., 2007; Slutske et al., 2002) as well as changes across development (Sher, Wood, Wood and Raskin, 1996). Also, given that expectancies are theoretically shaped by ongoing drinking experiences, genetic influences would to a large extent necessitate interactions with environment (i.e., alcohol exposure) to emerge. Expectancies might therefore be conceptualized as an intermediate phenotype whose development reflects the product of gene-environment interactions and possibly epigenetic processes (c.f. Goldman et al., 2006).
In considering whether alcohol expectancies can index genetic risk, it should be noted that expectancy research has historically relied on self-report and language-based assessment methods. There is increasing interest in developing implicit (i.e., automatic) measures of substance use cognitions (e.g., Wiers, van Woerden, Smulders, & de Jong, 2002) and these approaches have promise for refining the conceptualization and measurement and expectancy processes. Similarly, incorporating neurocognitive and neuroimaging methods in expectancy research (Anderson, Schweinsburg, Paulus, Brown, & Tapert, 2005; Fishman, Goldman, & Donchin, 2008) has potential for characterizing expectancy processes that would go unobserved using conventional approaches. Advances in these areas might ultimately identify expectancy processes that are less susceptible to environmental influence, therefore serving as potentially better candidates for endophenotypes. Considerably more research is needed to evaluate this possibility.
There are several limitations to this study. Our focus on Asian participants allowed examination of theoretically relevant polymorphisms that are rare (ADH1B*2) or nonexistent (ALDH2*2) in other populations. Nonetheless, a focus on this sample restricts the ability to generalize these findings to other populations. We did not subject the PEAQ to extensive psychometric validation given our goal of providing a preliminary test of physiological expectancies as related to ALDH2 and ADH1B. In addition, the relevance of this measure for other populations is unknown. The lack of genetic data for participants in the measurement development phase is an additional limitation, although the prevalence rates of ADH1B*2 and ALDH2*2 in northeast Asian populations suggest that the majority of participants possessed either or both variants. An additional limitation is that this study focused on functional variants that have shown relatively strong associations with alcohol use and dependence. While a focus on ALDH2*2 and ADH1B*2 was theoretically justified, other polymorphisms (Joslyn et al., 2008) and chromosomal regions (Schuckit, Smith, & Kalmijn, 2004) have been implicated in the level of response to alcohol (LR) phenotype. As additional genetic variants relevant to LR are identified their examination in relation to expectancies will likely be warranted. A reported association of the GABRB3 G1 allele and expectancies further suggests that variants relevant for neurotransmitter function might be important to consider (Young, Lawford, Feeney, Richard, & Noble, 2004). Other limitations to the current study include the use of a cross-sectional design, a moderate sample size and a focus on undergraduate participants.
This study adds to the few existing efforts to examine associations of candidate genes with alcohol expectancies (e.g., McCarthy et al., 2000; 2001; Young et al., 2004), thereby considering biological factors in the context of an established learning framework for understanding drinking behavior. The current results provide general support for the notion of expectancy theory as a promising theoretical framework for translational studies of alcohol use (Goldman et al., 2006). These findings also suggest that genetic, expectancy and cultural or demographic factors contribute uniquely and additively to predict drinking among Asian-American young adults. Joint consideration of these domains appears important for characterizing drinking behavior in this population. The refinement of bio-behavioral approaches, as well as efforts to identify intervening mechanisms, will likely aid the development of comprehensive theoretical models of drinking behavior.
Table 3.
Hierarchical regression analyses examining effects of demographic, genetic and expectancy variables.
| Drinking frequency | Heavy episodes | Maximum drinks lifetime | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B(SE) | β | R2Δ | B(SE) | β | R2Δ | B(SE) | β | R2Δ | |
| Step 1 | .01 | .02 | .06** | ||||||
| Gender | -0.02 (0.14) | -0.01 | -0.19 (0.22) | -0.06 | -2.16 (0.91) | -0.17* | |||
| Ethnicity | 0.16 (0.14) | 0.09 | 0.40 (0.22) | 0.13 | 2.40 (0.92) | 0.19* | |||
| Step 2 | .03 | .07** | .06** | ||||||
| ALDH2 | -0.27 (0.14) | -.014 | -0.80 (0.22) | -0.26*** | -3.11 (0.91) | -0.24** | |||
| ADH1B | 0.30 (0.24) | 0.09 | 0.44 (0.37) | 0.09 | 0.84 (1.53) | 0.04 | |||
| Step 3 | .04** | .00 | .04** | ||||||
| PEAQ | -0.30 (0.10) | -0.21** | -0.15 (0.16) | -0.07 | -1.93 (0.65) | -0.20** | |||
| Step 4 | .06*** | .04** | .02* | ||||||
| PEAQ (E) | 0.36 (0.10) | 0.25*** | 0.49 (0.16) | 0.21** | 1.34 (0.67) | 0.14* | |||
| Step 5 | .09*** | .03* | .05** | ||||||
| CEOA Pos | 0.71 (0.20) | 0.24** | 0.74 (0.33) | 0.16* | 4.33 (1.37) | 0.22** | |||
| CEOA Neg | -0.92 (0.23) | -0.39*** | -0.72 (0.37) | -0.19 | -3.13 (1.52) | -0.20* | |||
| Step 6 | .03* | .02 | .01 | ||||||
| CEOA Pos (E) | 0.21 (0.09) | 0.18* | 0.31 (0.15) | 0.16 | 0.20 (0.60) | 0.03 | |||
| CEOA Neg (E) | 0.12 (0.16) | 0.07 | 0.20 (0.26) | 0.07 | 1.52 (1.09) | 0.13 | |||
| Model statistics | R2 =.26, F(10, 181) = 6.28*** | R2 =.19, F(10, 181) = 4.33*** | R2 =.23, F(10, 180) = 5.36*** | ||||||
Note. Gender was coded 0 (Male) and 1 (Female). Ethnicity was coded 0 (Chinese) and 1 (Korean and Japanese). ALDH2 and ADH1B were coded 0, 1 or 2 corresponding with number of variant (*2) alleles. CEOA = Comprehensive Effects of Alcohol Questionnaire Pos = Global positive expectancies Neg = Global negative expectancies. PEAQ = Physiological Effects of Alcohol Questionnaire (total score). (E) = Expectancy evaluation component.
p < .05,
p <.01,
p< .001
Acknowledgments
This research was supported by the National Institute on Alcohol Abuse and Alcoholism grants F31AA016440 and K02AA00269, a small grant award from the University of Washington Alcohol and Drug Abuse Institute and a Dissertation Research Award from the American Psychological Association. Genotyping services were provided by the Genomics and Molecular Biology Core of the Alcohol Research Center at Indiana University, which is funded by NIAAA grant P60AA07611-20. The authors thank Kevin M. King for his helpful comments on earlier drafts of this paper.
Appendix: PEAQ Subscales/items
Flushing
My cheeks would turn red
My face would look flushed
I would feel flushed
My skin would turn red
I would feel blood rushing to my face
Sensorimotor Impairment
My hearing would be different
I would have vision problems
I wouldn't be able to speak as well
My hearing would be less clear
I wouldn't see things as clearly
Dizziness/Nausea
My head would start spinning
I would feel dizzy
My head would begin to hurt
I would feel lightheaded
I would feel queasy
Physical Sensations
I would feel tingling in my feet
My body would feel numb
I would get a tingling sensation in my fingers/toes
I would feel like I was floating
I would feel light
Heart Rate
My heart would start beating faster
My pulse would get stronger
My breathing would get faster
Footnotes
A few other studies have been cited as examples of behavioral genetic investigations of alcohol expectancies. However, we did not consider studies without twin data (e.g., Johnson et al., 1990), that were unpublished (e.g., cited in Prescott et al., 2004) or that did not include a face-valid measure of alcohol expectancies (e.g., Perry, 1973).
Some studies have examined ALDH2 or ADH1B in relation to retrospectively reported physiological symptoms (e.g., Carr et al., 2002; Chen et al., 1998; Takeshita, Yang, & Morimoto, 2001; Wall et al., 2005). Because the distinction between retrospective experiences vs. anticipated future outcomes (i.e., expectancies) is conceptually important, we discuss only those studies that examined expectancies as traditionally defined.
Publisher's Disclaimer: The following manuscript is the final accepted manuscript. It has not been subjected to the final copyediting, fact-checking, and proofreading required for formal publication. It is not the definitive, publisher-authenticated version. The American Psychological Association and its Council of Editors disclaim any responsibility or liabilities for errors or omissions of this manuscript version, any version derived from this manuscript by NIH, or other third parties. The published version is available at www.apa.org/journals/adb.
Contributor Information
Christian S. Hendershot, Department of Psychology, University of Washington
Clayton Neighbors, Department of Psychiatry and Behavioral Sciences, University of Washington.
William H. George, Department of Psychology, University of Washington
Denis M. McCarthy, Department of Psychological Sciences, University of Missouri-Columbia
Tamara L. Wall, Department of Psychiatry, University of California - San Diego, Psychology Service, Veterans Affairs San Diego Healthcare System and Veterans Medical Research Foundation, San Diego
Tiebing Liang, Indiana University School of Medicine.
Mary E. Larimer, Department of Psychiatry and Behavioral Sciences, University of Washington
References
- Agrawal A, Dick DM, Bucholz KK, Madden PAF, Cooper ML, Sher KJ, et al. Drinking expectancies and motives: a genetic study of young adult women. Addiction. 2008;103(2):194–204. doi: 10.1111/j.1360-0443.2007.02074.x. [DOI] [PubMed] [Google Scholar]
- Aiken LS, West SG. Multiple regression: Testing and interpreting interactions. Newbury Park, CA: Sage Publications, Inc.; 1991. [Google Scholar]
- Anderson KG, Schweinsburg A, Paulus MP, Brown SA, Tapert S. Examining personality and alcohol expectancies using functional magnetic resonance imaging (fMRI) with adolescents. Journal of Studies on Alcohol. 2005;66(3):323–331. doi: 10.15288/jsa.2005.66.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson KG, Smith GT, Fischer SF. Women and acquired preparedness: Personality and learning implications for alcohol use. Journal of Studies on Alcohol. 2003;64(3):384–392. doi: 10.15288/jsa.2003.64.384. [DOI] [PubMed] [Google Scholar]
- Babor TF, Biddle-Higgins JC, Saunders JB, Monteiro MG. AUDIT: The Alcohol Use Disorders Identification Test: Guidelines for Use in Primary Health Care. Geneva, Switzerland: World Health Organization; 2001. [Google Scholar]
- Bearden CE, Freimer NB. Endophenotypes for psychiatric disorders: Ready for primetime? Trends in Genetics. 2006;22:306–313. doi: 10.1016/j.tig.2006.04.004. [DOI] [PubMed] [Google Scholar]
- Beauchaine TP, Neuhaus E, Brenner SL, Gatzke-Kopp L. Ten good reasons to consider biological processes in prevention and intervention research. Development and Psychopathology. 2008;20:745–774. doi: 10.1017/S0954579408000369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown SA, Goldman MS, Inn A, Anderson LR. Expectations of reinforcement from alcohol: Their domain and relation to drinking patterns. Journal of Consulting and Clinical Psychology. 1980;48(4):419–426. doi: 10.1037//0022-006x.48.4.419. [DOI] [PubMed] [Google Scholar]
- Bryan A, Hutchison KE, Seals DR, Allen DL. A transdisciplinary model integrating genetic, physiological, and psychological correlates of voluntary exercise. Health Psychology. 2007;26(1):30–39. doi: 10.1037/0278-6133.26.1.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown SA, Tate SR, Vik PW, Haas AL, Aarons GA. Modeling of alcohol use mediates the effect of family history of alcoholism on adolescent alcohol expectancies. Experimental and Clinical Psychopharmacology. 1999;7(1):20–27. doi: 10.1037//1064-1297.7.1.20. [DOI] [PubMed] [Google Scholar]
- Burden JL, Maisto SA. Expectancies, evaluations and attitudes: Prediction of college student drinking behavior. Journal of Studies on Alcohol. 2000;61(2):323–331. doi: 10.15288/jsa.2000.61.323. [DOI] [PubMed] [Google Scholar]
- Carr LG, Foroud T, Stewart T, Castelluccio P, Edenberg HJ, Li TK. Influence of ADH1B polymorphism on alcohol use and its subjective effects in a Jewish population. American Journal of Medical Genetics. 2002;112(2):138–143. doi: 10.1002/ajmg.10674. [DOI] [PubMed] [Google Scholar]
- Chen WJ, Chen CC, Yu JM, Cheng ATA. Self-reported flushing and genotypes of ALDH2, ADH2, and ADH3 among Taiwanese Han. Alcoholism-Clinical and Experimental Research. 1998;22(5):1048–1052. [PubMed] [Google Scholar]
- Christiansen BA, Roehling PV, Smith GT, Goldman MS. Using Alcohol Expectancies To Predict Adolescent Drinking Behavior After One Year. Journal Of Consulting And Clinical Psychology. 1989;57(1):93–99. doi: 10.1037//0022-006x.57.1.93. [DOI] [PubMed] [Google Scholar]
- Clark L, Watson D. Constructing validity: Basic issues in objective scale development. Psychological Assessment. 1995;7:309–319. doi: 10.1037/pas0000626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook TAR, Luczak SE, Shea SH, Ehlers CL, Carr LG, Wall TL. Associations of ALDH2 and ADH1B genotypes with response to alcohol in Asian Americans. Journal of Studies on Alcohol. 2005;66(2):196–204. doi: 10.15288/jsa.2005.66.196. [DOI] [PubMed] [Google Scholar]
- Dick DM, Agrawal A, Schuckit MA, Bierut L, Hinrichs A, Fox L, et al. Marital status, alcohol dependence, and GABRA2: Evidence for gene-environment correlation and interaction. Journal of Studies on Alcohol. 2006;67(2):185–194. doi: 10.15288/jsa.2006.67.185. [DOI] [PubMed] [Google Scholar]
- Edenberg HJ. The genetics of alcohol metabolism - Role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Research & Health. 2007;30(1):5–13. [PMC free article] [PubMed] [Google Scholar]
- Ehlers CL, Carr L, Betancourt M, Montane-Jaime K. Association of the ADH2*3 allele with greater alcohol expectancies in African-American young adults. Journal of Studies on Alcohol. 2003;64:176–181. doi: 10.15288/jsa.2003.64.176. [DOI] [PubMed] [Google Scholar]
- Finn PR, Sharkansky EJ, Brandt KM, Turcotte N. The effects of familial risk, personality, and expectancies on alcohol use and abuse. Journal of Abnormal Psychology. 2000;109(1):122–133. doi: 10.1037//0021-843x.109.1.122. [DOI] [PubMed] [Google Scholar]
- Fishbein M, Ajzen I. Belief, attitude, intention and behavior: An introduction to theory and research. Reading, MA: Addison-Wesley; 1975. [Google Scholar]
- Fishman I, Goldman MS, Donchin E. The P300 as an electrophysiological probe of alcohol expectancy. Experimental and Clinical Psychopharmacology. 2008;16:341–356. doi: 10.1037/a0012873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fromme K, D'Amico EJ. Measuring adolescent alcohol outcome expectancies. Psychology of Addictive Behaviors. 2000;14(2):206–212. doi: 10.1037//0893-164x.14.2.206. [DOI] [PubMed] [Google Scholar]
- Fromme K, Stroot EA, Kaplan D. Comprehensive effects of alcohol: Development and psychometric assessment of a new expectancy questionnaire. Psychological Assessment. 1993;5:19–26. [Google Scholar]
- Gabrielli WF, Plomin R. Individual differences in anticipation of alcohol sensitivity. Journal of Nervous and Mental Disease. 1985;173(2):111–114. doi: 10.1097/00005053-198502000-00008. [DOI] [PubMed] [Google Scholar]
- Goldman MS, Darkes J, Del Boca FK. Expectancy mediation of biopsychosocial risk for alcohol use and alcoholism. In: Kirsch I, editor. How Expectancies Shape Experience. Washington, DC: American Psychological Association; 1999. [Google Scholar]
- Goldman MS, Darkes J, Reich RR, Brandon KO. From DNA to conscious thought: The influence of anticipatory processes on human alcohol consumption. In: Munafo M, Albery IP, editors. Cognition and addiction. New York: Oxford University Press; 2006. [Google Scholar]
- Gottesman II, Gould TD. The endophenotype concept in psychiatry: Etymology and strategic intentions. The American Journal of Psychiatry. 2003;160:636–645. doi: 10.1176/appi.ajp.160.4.636. [DOI] [PubMed] [Google Scholar]
- Gould TD, Gottesman II. Psychiatric endophenotypes and the development of valid animal models. Genes, Brain and Behavior. 2006;5:112–119. doi: 10.1111/j.1601-183X.2005.00186.x. [DOI] [PubMed] [Google Scholar]
- Heath AC. Genetic influence on drinking behavior in humans. In: Begleiter H, Kissin B, editors. The genetics of alcoholism. New York: Oxford University Press; 1995. [Google Scholar]
- Heath AC, Nelson EC. Effects of the interaction between genotype and environment. Research into the genetic epidemiology of alcohol dependence. Alcohol Research and Health. 2002;26:193–201. [PMC free article] [PubMed] [Google Scholar]
- Hasin D, Aharonovich E, Liu XH, Mamman Z, Matseoane K, Carr LG, et al. Alcohol dependence symptoms and alcohol dehydrogenase 2 polymorphism: Israeli Ashkenazis, Sephardics, and recent Russian immigrants. Alcoholism-Clinical and Experimental Research. 2002;26(9):1315–1321. doi: 10.1097/01.ALC.0000029597.07916.A9. [DOI] [PubMed] [Google Scholar]
- Henderson MJ, Goldman MS, Coovert MD, Carnevalla N. Covariance structure models of expectancy. Journal of Studies on Alcohol. 1994;55(3):315–326. doi: 10.15288/jsa.1994.55.315. [DOI] [PubMed] [Google Scholar]
- Hendershot CS, Collins SE, George WH, Wall TL, McCarthy DM, Liang T, Larimer ME. Associations of ALDH2 and ADH1B genotypes with alcohol-related phenotypes in Asian young adults. Alcoholism: Clinical and Experimental Research. 2009;33(5):839–847. doi: 10.1111/j.1530-0277.2009.00903.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutchison KE, Allen DL, Filbey FM, Jepson C, Lerman C, Benowitz NL, et al. CHRNA4 and tobacco dependence: From gene regulation to treatment outcome. Archives of General Psychiatry. 2007;64(9):1078–1086. doi: 10.1001/archpsyc.64.9.1078. [DOI] [PubMed] [Google Scholar]
- Hutchison KE, Stallings M, McGeary J, Bryan A. Population stratification in the candidate gene study: Fatal threat or red herring? Psychological Bulletin. 2004;130(1):66–79. doi: 10.1037/0033-2909.130.1.66. [DOI] [PubMed] [Google Scholar]
- Johnson RC, Nagoshi CT, Danko GP, Honbo KA, Chau LL. Familial transmission of alcohol use norms and expectancies and reported alcohol use. Alcoholism-Clinical and Experimental Research. 1990;14:216–20. doi: 10.1111/j.1530-0277.1990.tb00475.x. [DOI] [PubMed] [Google Scholar]
- Kilbey MM, Downey K, Breslau N. Predicting the emergence and persistence of alcohol dependence in young adults: The role of expectancy and other risk factors. Experimental and Clinical Psychopharmacology. 1998;6:149–156. doi: 10.1037//1064-1297.6.2.149. [DOI] [PubMed] [Google Scholar]
- Leigh BC. Evaluations of alcohol expectancies: Do they add to prediction of drinking patterns? Psychology of Addictive Behaviors. 1987;1:135–139. [Google Scholar]
- Luczak SE, Glatt SJ, Wall TL. Meta-analyses of ALDH2 and ADH1B with alcohol dependence in Asians. Psychological Bulletin. 2006;132(4):607–621. doi: 10.1037/0033-2909.132.4.607. [DOI] [PubMed] [Google Scholar]
- MacKinnon DP, Lockwood CM, Hoffman JM, West SG, Sheets V. A comparison of methods to test the significance of the mediated effect. Psychological Methods. 2002;7:83–104. doi: 10.1037/1082-989x.7.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKinnon DP, Fairchild AJ, Fritz MS. Mediation analysis. Annual Review of Psychology. 2007;58:593–614. doi: 10.1146/annurev.psych.58.110405.085542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKinnon DP, Fritz MS, Williams J, Lockwood CM. Distribution of the product confidence limits for the indirect effect: Program PRODCLIN. Behavior Research Methods. 2007;39(3):384–389. doi: 10.3758/bf03193007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarthy DM, Brown SA, Carr LG, Wall TL. ALDH2 status, alcohol expectancies, and alcohol response: Preliminary evidence for a mediation model. Alcoholism-Clinical and Experimental Research. 2001;25(11):1558–1563. [PubMed] [Google Scholar]
- McCarthy DM, Wall TL, Brown SA, Carr LG. Integrating biological and behavioral factors in alcohol use risk: The role of ALDH2 status and alcohol expectancies in a sample of Asian Americans. Experimental and Clinical Psychopharmacology. 2000;8(2):168–175. doi: 10.1037//1064-1297.8.2.168. [DOI] [PubMed] [Google Scholar]
- McGue M. Genes, environment and the etiology of alcoholism. In: Zucker R, Boyd G, Howard J, editors. The Development of Alcohol Problems: Exploring the Biopsychosocial Matrix of Risk. US Department of Health and Human Services; 1994. [Google Scholar]
- McGue M. Behavioral genetic models of alcoholism and drinking. In: Leonard KE, Blane HT, editors. Psychological theories of drinking and alcoholism. 2nd. New York, NY: The Guilford Press; 1999. [Google Scholar]
- Meeker WQ, Cornwell LW, Aroian LA. The product of two normally distributed random variables. In: Kennedy WJ, Odeh RE, Davenport JM, editors. Selected tables in mathematical statistics. Vol. 7. Providence, RI: American Mathematical Society; 1981. pp. 1–256. [Google Scholar]
- Miller ET, Neal DJ, Roberts LJ, Baer JS, Cressler SO, Metrik J, Marlatt GA. Test-retest reliability of alcohol measures: Is there a difference between internet-based assessment and traditional measures? Psychology of Addictive Behaviors. 16(1):56–63. [PubMed] [Google Scholar]
- Perry A. The effect of heredity on attitudes toward alcohol, cigarettes and coffee. Journal of Applied Psychology. 1973;58(2):275–277. [Google Scholar]
- Peng GS, Yin SJ. Effect of the allelic variants of aldehyde dehydrogenase ALDH2*2 and alcohol dehydrogenase ADH1B*2 on blood acetaldehyde concentrations. Human Genomics. 2009;3(2):121–127. doi: 10.1186/1479-7364-3-2-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plomin R, Crabbe J. DNA. Psychological Bulletin. 2000;126(6):806–828. doi: 10.1037/0033-2909.126.6.806. [DOI] [PubMed] [Google Scholar]
- Prescott CA, Cross RJ, Kuhn JW, Horn JL, Kendler KS. Is risk for alcoholism mediated by individual differences in drinking motivations? Alcoholism: Clinical and Experimental Research. 2004;28:29–39. doi: 10.1097/01.ALC.0000106302.75766.F0. [DOI] [PubMed] [Google Scholar]
- Schuckit MA. Genetics of the risk for alcoholism. American Journal on Addictions. 2000;9(2):103–112. doi: 10.1080/10550490050173172. [DOI] [PubMed] [Google Scholar]
- Schuckit MA, Smith TL. An 8-year follow-up of 450 sons of alcoholic and control subjects. Archives of General Psychiatry. 1996;53(3):202–210. doi: 10.1001/archpsyc.1996.01830030020005. [DOI] [PubMed] [Google Scholar]
- Schuckit MA, Smith TL, Kalmijn J. The search for genes contributing to the low level of response to alcohol: patterns of findings across studies. Alcoholism: Clinical and Experimental Research. 2004;28:1449–1458. doi: 10.1097/01.alc.0000141637.01925.f6. [DOI] [PubMed] [Google Scholar]
- Schuckit MA, Smith TL. A comparison of correlates of DSM-IV alcohol abuse or dependence among more than 400 sons of alcoholics and controls. Alcoholism: Clinical and Experimental Research. 2001;25:1–8. [PubMed] [Google Scholar]
- Sher KJ, Walitzer KS, Wood PK, Brent EE. Characteristics of children of alcoholics: Putative risk factors, substance use and abuse, and psychopathology. Journal of Abnormal Psychology. 1991;100(4):427–448. doi: 10.1037//0021-843x.100.4.427. [DOI] [PubMed] [Google Scholar]
- Sher KJ, Wood MD, Wood PK, Raskin G. Alcohol outcome expectancies and alcohol use: A latent variable cross-lagged panel study. Journal of Abnormal Psychology. 1996;105(4):561–574. doi: 10.1037/0021-843X.105.4.561. [DOI] [PubMed] [Google Scholar]
- Slutske WS, Cronk NJ, Sher KJ, Madden PAF, Bucholz KK, Heath AC. Genes, environment, and individual differences in alcohol expectancies among female adolescents and young adults. Psychology of Addictive Behaviors. 2002;16(4):308–317. [PubMed] [Google Scholar]
- Sobel ME. Asymptotic confidence intervals for indirect effects in structural equation models. In: Leinhardt S, editor. Sociological Methodology 1982. Washington DC: American Sociological Association; 1982. pp. 290–312. [Google Scholar]
- Sue S, Nakamura CY. An integrative model of physiological and social/psychological factors in alcohol consumption among Chinese and Japanese Americans. Journal of Drug Issues. 1984;14:349–364. [Google Scholar]
- Takeshita T, Yang X, Morimoto K. Association of the ADH2 genotypes with skin responses after ethanol exposure in Japanese male university students. Alcoholism: Clinical and Experimental Research. 2001;25(9):1264–1269. [PubMed] [Google Scholar]
- Thomasson HR, Li TK. How alcohol and aldehyde dehydrogenase genes modify alcohol drinking, alcohol flushing, and the risk for alcoholism. Alcohol Health & Research World. 1993;17:167–172. [Google Scholar]
- Truett GE, Heeger P, Mynatt RL, Truett AA, Walker JA, Warman ML. Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT) BioTechniques. 2000;29:52–54. doi: 10.2144/00291bm09. [DOI] [PubMed] [Google Scholar]
- Valdivia I, Stewart SH. Further examination of the psychometric properties of the comprehensive effects of alcohol questionnaire. Cognitive Behaviour Therapy. 2005;24:22–33. doi: 10.1080/16506070410001009. [DOI] [PubMed] [Google Scholar]
- Vernon PA, Lee D, Harris JA, Jang KL. Genetic and environmental contributions to individual differences in alcohol expectancies. Personality And Individual Differences. 1996;21(2):183–187. [Google Scholar]
- Wall TL. Genetic associations of alcohol and aldehyde dehydrogenase with alcohol dependence and their mechanisms of action. Therapeutic Drug Monitoring. 2005;27(6):700–703. doi: 10.1097/01.ftd.0000179840.78762.33. [DOI] [PubMed] [Google Scholar]
- Wall TL, Ehlers CL. Genetic influences affecting alcohol use among Asians. Alcohol Health & Research Workd. 1995;19(3):184–189. [PMC free article] [PubMed] [Google Scholar]
- Wall TL, Shea SH, Luczak SE, Cook TAR, Carr LG. Genetic associations of alcohol dehydrogenase with alcohol use disorders and endophenotypes in White college students. Journal Of Abnormal Psychology. 2005;114(3):456–465. doi: 10.1037/0021-843X.114.3.456. [DOI] [PubMed] [Google Scholar]
- Whitfield JB. Meta-analysis of the effects of alcohol dehydrogenase genotype on alcohol dependence and alcoholic liver disease. Alcohol and Alcoholism. 1997;32(5):613–619. doi: 10.1093/oxfordjournals.alcalc.a008303. [DOI] [PubMed] [Google Scholar]
- Whitfield JB. Alcohol dehydrogenase and alcohol dependence: Variation in genotype-associated risk between populations. American Journal of Human Genetics. 2002;71:1247–1250. doi: 10.1086/344287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young RMD, Lawford BR, Feeney GF, Ritchie T, Noble EP. Alcohol-related expectancies are associated with the D2 dopamine receptor and GABAA receptor β3 subunit genes. Psychiatry Research. 2004;127(3):171–183. doi: 10.1016/j.psychres.2003.11.004. [DOI] [PubMed] [Google Scholar]
- Wiers RW, van Woerden N, Smulders FTY, de Jong PJ. Implicit and explicit alcohol-related cognitions in heavy and light drinkers. Journal of Abnormal Psychology. 2002;111(4):648–658. doi: 10.1037/0021-843X.111.4.648. [DOI] [PubMed] [Google Scholar]


