Abstract
A new class of multi-target compounds was synthesized by linking a novel selective serotonin reuptake inhibitor (SSRI)a to a PDE4 inhibitor. The new dual PDE4 inhibitor/SSRI showed antidepressant-like activity in the forced swim test in mice The SSRIs 14, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine and 15, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-propyl]-tetrahydro-furan-2-yl}-ethylamine were both individually linked to the PDE4 inhibitor 19, (4-(3,4-dimethoxy-phenyl)-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one) via a five carbon chain. The dual PDE4 inhibitor/SSRI 21, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-pentyl]-4,5,8,8a-tetrahydro-2H-phthalazin-1-one showed potent and selective serotonin reuptake inhibition (IC50 value of 127 nM). The dual PDE4 inhibitor/SSRI 21 also inhibited PDE4D3 with a Ki value of 2.0 nM. The dual PDE4 inhibitor/SSRI was significantly more effective than the individual SSRI alone or fluoxetine in the forced swim test at standard doses. On a molar basis, the antidepressant-like effect of the dual PDE4 inhibitor/SSRI 21 showed a 129-fold increase in in vivo efficacy compared to fluoxetine.
Introduction
The neurodegeneration hypothesis of depression proposes that depression may be caused by maladaption of immune system and monoaminergic function1. Chronic antidepressant treatment up-regulates hippocampal neurogenesis, and could thereby block or reverse cellular atrophy. Antidepressants imipramine (C19H24N2) and amitriptyline (C20H23N) inhibit the uptake of serotonin (5-HT) and norepinephrine (NE) in vitro and in vivo. Based on clinical observations inhibition of the reuptake of either of these monoamines is responsible in part for the mood-elevating action of these compounds2. Overall, the literature suggest that 5-HT may be involved in depression and elevation of 5-HT by reuptake inhibition could be clinically useful. Reuptake agents such as fluoxetine (C17H18F3NO), sertraline (C17H17Cl2N), citalopram (C20H21FN2O),and fluvoxamine (C15H21F3N2O2) are relatively selective serotonin reuptake inhibitors (SSRIs) and selectively elevate 5-HT in vitro and in the brain of animals by inhibiting the human serotonin transporter (hSERT)3,4.
SSRIs do not have marked efficacy in animal models of depression such as the forced swim test (FST) and SSRI antidepressants may require 1 to 4 weeks of administration to observe behavioral improvement involving longer-term adaptive changes in receptor sensitivity5. Animals treated repeatedly with SSRIs, such as paroxetine (C19H20FNO3) down-regulate the SERT6. The extent of 5-HT reuptake inhibition after repeated treatment is greater than that observed after acute drug treatment4. This results in progressive increase in extracellular 5-HT and stimulation of postsynaptic receptors7. SSRIs are extensively metabolized and metabolites are found in the liver, lung and brain. Metabolism of antidepressants may play a significant role in overall efficacy because generally, the secondary amines (i.e., desmethyl metabolites) are more potent inhibitors of the uptake of NE than of 5-HT4. However, this is not the case with fluoxetine because the main metabolite (i.e.,nor-fluoxetine, C16H15F3NO) was equally selective a 5-HT reuptake inhibitor as its parent6. With acute treatment, SSRIs will increase 5-HT receptor-mediated cyclic adenosine monophosphate (cAMP) signaling8. Upon repeated treatment with SSRIs, this effect will be blunted (i.e., some tolerance development), since phosphodiesterases (PDEs) are up-regulated and cAMP hydrolysis is increased8.
Cellular cAMP concentrations are determined by synthesis (adenylyl cyclases) and hydrolysis (cAMP PDEs). PDEs comprise a diverse group of enzymes important as regulators of signal transduction9,10 coded by distinct genes, (e.g., PDE4A, PDE4B, PDE4C, and PDE4D, referred to as “subtypes”) and their selective inhibitors as “subtype-selective” inhibitors. PDE4 enzymes are particularly important in neuropsychopharmacology9 because PDE4 hydrolyzes cAMP formed by stimulation of beta adrenergic receptor-linked adenylyl cyclase in rat cerebral cortical slices11. Rolipram (C16H21NO3), a selective PDE4 inhibitor has antidepressant activity and produces memory-enhancing effects12. Increased cAMP increases the expression of a number of PDE4 variants in neurons. Thus, in the case of SSRIs, the adaptation of PDE4 that occurs in response to repeated treatment with SSRIs is homeostatic and in opposition to the acute effects of the drugs.
Selective inhibitors of PDE4, (i.e., rolipram), produce antidepressant-like effects in a number of preclinical tests sensitive to antidepressants. They antagonize the behavioral and physiological effects of reserpine (C33H40N2O9)12, decrease the time of immobility in the FST in rats and mice13 and decrease response rate and increase reinforcement rate of rats under a differential-reinforcement-of-low-rate (DRL) schedule14 and support the conclusion that PDE4 inhibitors have antidepressant activity9. If SSRIs produce their effects in part via activation of cAMP signaling, then loss of activity of the PDE4 subtype involved would enhance their ability to increase cAMP and alter subsequent pharmacology. Thus, one adaptation of PDE4 that occurs in response to repeated treatment with SSRIs increases 5-HT receptor-mediated cAMP signaling. With repeated treatment, this effect will be blunted as tolerance develops because PDE4 is up-regulated and cAMP hydrolysis is increased. When animals are repeatedly treated with SSRIs, PDE4 is up-regulated7. This may be a consequence of 5-HT receptor-mediated cAMP signaling. Increased cAMP augments the expression of a number of PDE4 variants in neurons8.
Compared to imipramine-treated rats, a combination of rolipram and imipramine showed that chronic co-administration of a PDE4 inhibitor with an antidepressant may be more effective for depression therapy and suggest that elevation of the cAMP signal transduction pathway is involved in the antidepressive effects15. Likewise, acute treatment with fluoxetine to elevate synaptic 5-HT but not dopamine (DA) significantly enhanced rolipram binding and accumulation16. Dual PDE4 inhibitor/SSRIs offer advantages beyond simple additive effects of individual agents. One hypothesis is that dual PDE4 inhibitors/SSRIs block the effect of the up-regulation of PDE4, so that the overall increase in 5-HT-mediated cAMP signaling is preserved with repeated treatment. Herein, we describe an SSRI that was chemically linked to a PDE4 inhibitor. The dual PDE4 inhibitor/SSRI was examined in vitro and in vivo for pharmacological activity.
Results
Chemistry
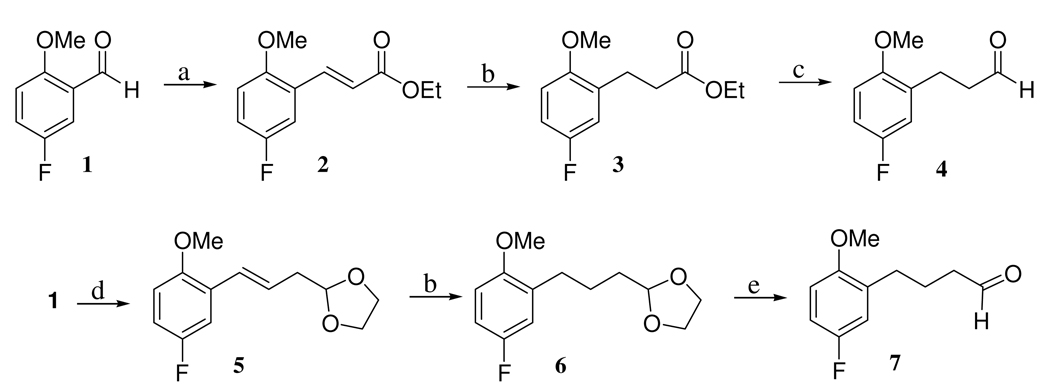
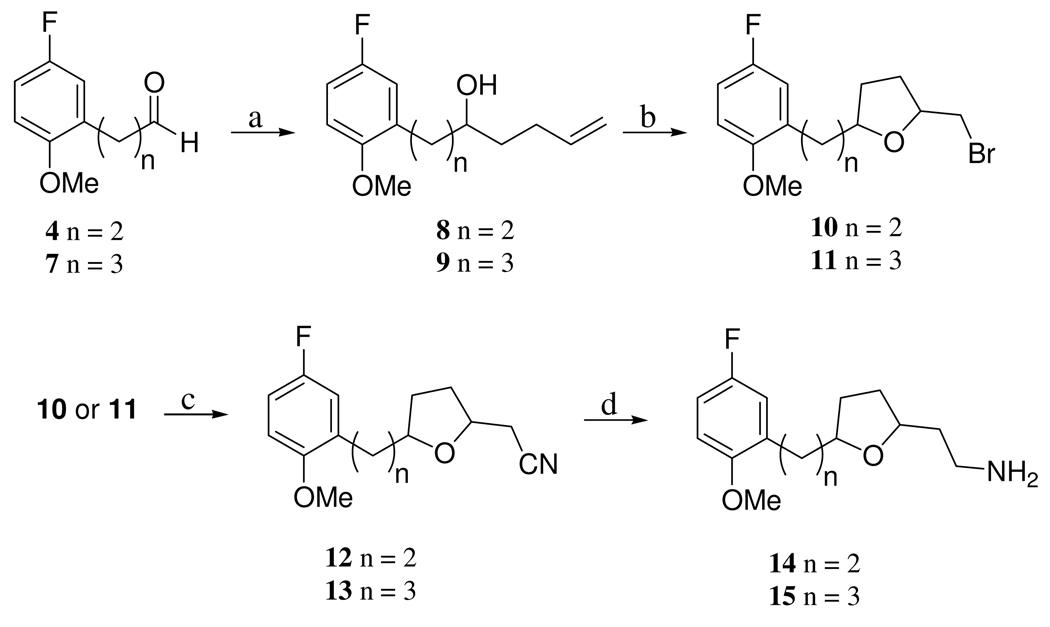
The chemical synthesis of the dual inhibitors consisted of coupling an SSRI (i.e., a 2,5-disubstituted tetrahydrofuran) with a known PDE4 inhibitor (i.e., a phthalazinone) via a five carbon linker to afford the target compound. The SSRI portion of the dual inhibitor was obtained as follows. The requisite aldehyde (i.e., 4, (3-(5’-fluoro-2’-methoxyphenyl)propanal) and 7, (4-(5’-fluoro-2’-methoxyphenyl)butyraldehyde), synthesized by standard chemistry (as described in Scheme 1) was combined with butenyl magnesium bromide to afford the alcohols 8, (7-(5’-fluoro-2’-methoxyphenyl)hept-1-en-5-ol) and 9, (8-(5’-fluoro-2’-methoxyphenyl)oct-1-en-5-ol), respectively (Scheme 2). Cyclization of 8 and 9 in the presence of NBS afforded the 2,5-disubstituted tetrahydrofurans 10, 2-(bromomethyl)-5-(2’-methoxy-5’-fluorophenethyl)tetrahydrafuran and 11, 2-(bromomethyl)-5-(3’-(2'’-methoxy-5'’-fluorophenyl)-1’-propyl)tetrahydrofuran, respectively (Scheme 2). Treatment of 10 or 11 with KCN resulted in the cyano compounds 12, 2-cyanomethyl-5-(5’-fluoro-2-methoxyphenethyl)tetrahydrofuran and 13, 2-cyanomethyl-5-(3’-(2”-methoxy-5”-fluorophenyl)-1’-propyl)tetrahydrafuran, respectively, that were hydrogenated with H2 in the presence of Raney Nickel and ammonia to afford 14, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine and 15, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-propyl]-tetrahydro-furan-2-yl}-ethylamine, respectively. The SSRIs 14 and 15 were used to couple with the PDE4 inhibitor portion of the molecule to afford the dual PDE4/SSRI.
Scheme 1.
a) EtOC(O)CH-PPH3, DCM; b) H2, Pd/C; c) DiBAL, toluene; d) [2-(1,3-dioxolan-2-yl)ehtyl]triphenylphosphonium bromide; e) acetone, H2SO4.
Scheme 2.
a) 1-Butenylmagnesium bromide; b) NBS, DCM; c) KCN, NaI, DMSO; d) Raney Ni, H2, 1 M NH3in MeOH.
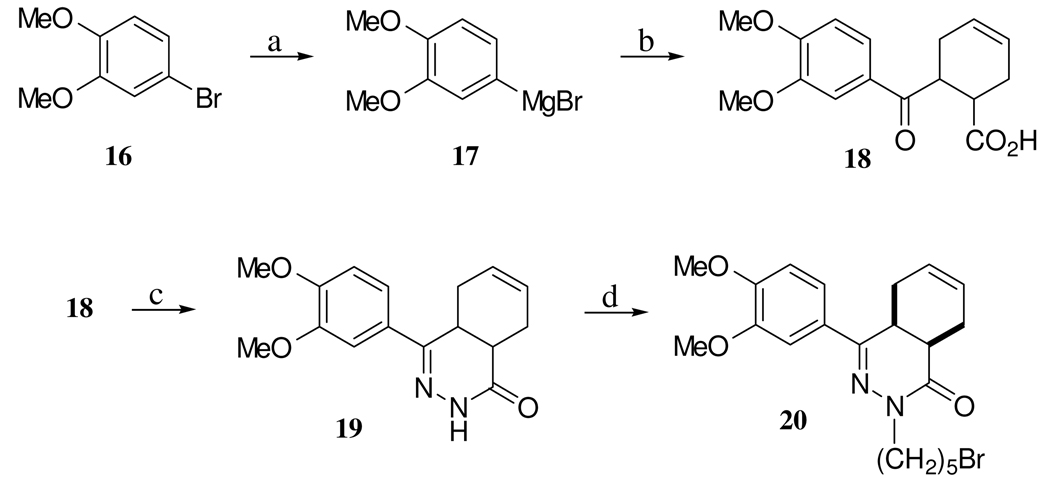
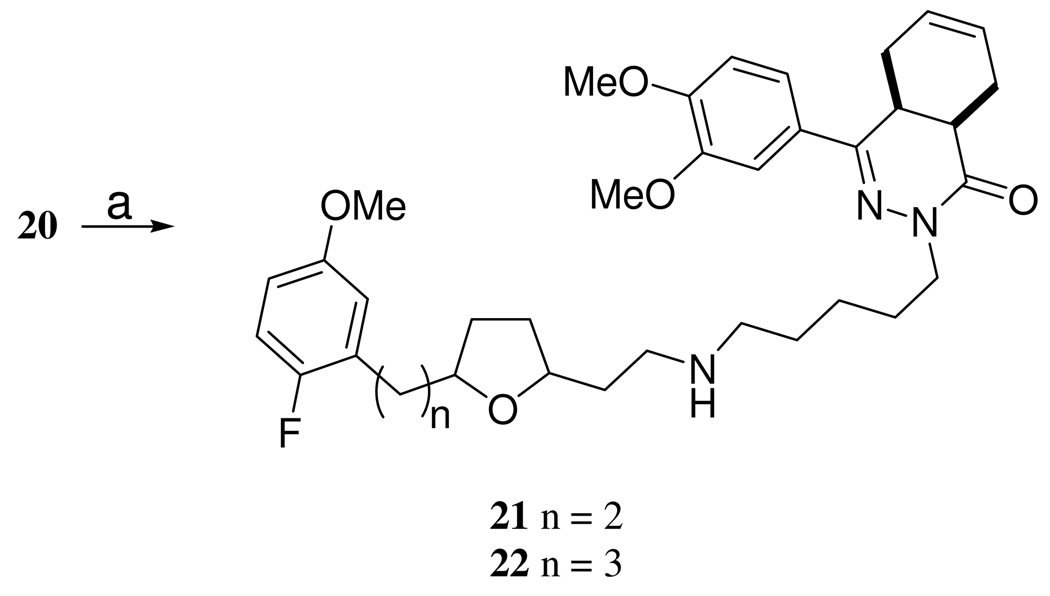
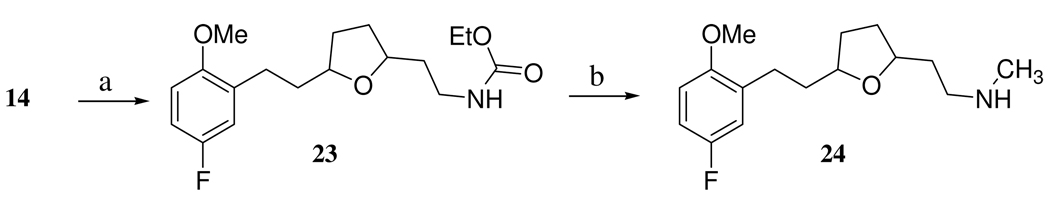
The PDE4 inhibitor portion of the dual inhibitor was synthesized by a previously described route17,18 (Scheme 3). Because it was known that the PDE4 inhibitor 19, 4-(3,4-dimethoxy-phenyl)-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one possessed increased inhibitory potency upon N-alkylation, we hypothesized that a more potent PDE4 inhibitor could arise from linking the SSRI and PDE4 compounds via N-alkylation. The final target compounds 21, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-pentyl]-4,5,8,8a-tetrahydro-2H-phthalazin-1-one and 22, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-propyl]-tetrahydro-furan-2-yl}-ethylamine)-pentyl-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one were prepared in a two step sequence involving: 1) attachment of a five carbon linker by treating the phthalazinone 19 with NaH and 1,5-dibromopentane to afford 20, 2-(5-bromo-pentyl)-4-(3,4-dimethoxy-phenyl-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one and 2) linking 20 to 14 and 15 with the aid of CsOH (to diminish formation of the tertiary amine) to afford 21 and 22, respectively (Scheme 4). The N-methyl derivative of 14 was synthesized in a two step sequence starting with protection with ethylchloroformate in the presence of base to from 23 and then reduction of 23 to 24 with lithium aluminum hydride in the presence of THF (Scheme 5).
Scheme 3.
a) Mg turlings, THF, reflux; b) cis-1,2,3,6-tetrahydrophthalic anhydride, THF; c) H2NNH2, EtOH, reflux; d) 1,5-dibromopentane, NaH, DMF.
Scheme 4.
a) CsOH.H2O, 4 Å sieves, 14 or 15, DMF.
Scheme 5.
a) Ethylchloroformate, K2CO3, THF; b) LAH, THF.
Biology
Inhibition of PDE4 enzymes was conducted with the synthetic agents (Table 1). In addition, binding affinity of each compound (i.e., Ki value) was calculated from the IC50 value and measured for each compound by assessing the potency of inhibition of binding radiolabeled 25 (RTI-55)19 to the hDAT, hSERT and hNET20,21 (Table 2). In vitro functional potency was measured by determining the reuptake inhibition (i.e., IC50 value) of [3H]-DA, [3H]-5-HT or [3H]-NE at the recombinant hDAT, hSERT or hNET in the presence of HEK-293 cells transfected with the respective transporter cDNA.
Table 1.
Inhibition of Recombinant PDE4 Isoforms with PDE4 inhibitors and Dual PDE4 Inhibitor/SSRIs
| Enzyme | |||
|---|---|---|---|
| Compound | Human PDE4D3 |
Human PDE4B3 |
Human PDE4A1 |
| Ki (nM) | Ki (nM) | Ki (nM) | |
| 19 | 6.3 | 250 | >1000 |
| 20 | 2.3 | NDa | ND |
| 21 | 2.0 | 199 | > 1000 |
| 22 | 1.2 | 500 | > 1000 |
| Rolipram | 58.9 | ND | ND |
ND, Not Done
Table 2.
Inhibition of radioligand binding and [3H] neurotransmitter uptake in HEK-hDAT, HEK-hSERT and HEK-hNET cellsa.
| Binding (Ki, nM) | Reuptake (IC50, nM) | ||||||
|---|---|---|---|---|---|---|---|
| Compound | Description | hDAT | hSERT | hNET | hDAT | hSERT | hNET |
| Cocaine | 371 ± 81 | 276 ± 87 | 1115 ± 198 | 303 ± 74 | 416 ± 135 | 835 ± 229 | |
| Fluoxetine b | 6670 ± 850 | 1.1 ± 0.5 | 1560 ± 300 | 19,500 ± 7600 | 7.3 ± 2.9 | 1020 ± 180 | |
| 14 | SSRI | 6500 ± 828 | 2.1 ± 0.2 | 2440 ± 215 | 27850 ± 5129 | 2.5 ± 0.6 | 481 ± 158 |
| 15 | SSRI | 7307 ± 1331 | 1.1 ± 1.0 | 10600 ± 1880 | 16987 ± 1352 | 2.3 ± 0.6 | 1930.0 ± 463.0 |
| 19 | PDE4 inhibitor | >100,000 | >100,000 | >100,000 | >100,000 | >100,000 | 1930.0 ± 463.0 |
| 20 | N-modified PDE4 inhibitor |
>100,000 | >100,000 | 39,119 ± 4915 | >100,000 | >100,000 | >100,000 |
| 21 | Dual inhibitor | 2478 ± 451 | 156 ± 30 | 1748 ± 257 | 3556 ± 377 | 127 ± 15 | 957 ± 146 |
| 22 | Dual inhibitor | 5340 ± 1083 | 194 ± 87 | 7441 ± 1269 | 5340 ± 1083 | 194 ± 87 | 7441 ± 1269 |
| 24 | SSRI | 66988 ± 14450 | 21.2 ± 4.3 | 19332 ± 3577 | > 100,000 | 18.4 ± 6.1 | 20370 ± 3751 |
Values are the mean ± S.E.M. of three to four independent experiments conducted with duplicate (binding) or with triplicate (uptake) determinations unless the mean of three experiments exceeded 100 µM. Drug inhibition of [125I]-25 binding in HEK-hDAT, HEK-hSERT, or HEK-hNET cell membranes. Inhibition of [3H]-DA, [3H]-5-HT, [3H]-NE in the presence of HEK-hDAT, HEK-hSERT, or HEK-hNET cells, respectively;
Data taken from reference 20.
PDE4 inhibitors
Compound 19 and rolipram are known PDE4 inhibitors and we confirmed this observation (Table 1). It was known that N-substitution was beneficial for PDE4 inhibition potency17 and in the phthalazinone series the cis-(±)-4a,5,8,8a-tetra- and cis-(±)-4a,5,6,7,8,8a-hexahydro-2H-phthalazin-1-ones showed potent inhibition of PDE4 by occupying a region of space different from that of other fused ring systems to afford selective inhibition17,18. N-Substituted derivatives distal to the fused ring system were synthesized to examine whether additional pharmacological properties could be introduced into 19 while still preserving PDE4 inhibitory activity. In fact, the N-substituted phthalazinone 20 was a potent inhibitor of PDE4D3 than was 19 (Table 1). Thus, a template with potent PDE4 inhibitory activity was chemically combined with a molecule possessing potent SSRI function to make a compound with a dual PDE4 inhibition/SSRI reuptake inhibition profile.
The pattern of effects of PDE4 inhibitors on behavior is distinct from that of selective inhibitors of other PDE families9,14. Generalized increases in cAMP do not adequately explain the behavioral effects of PDE4 inhibitors. Thus, changes in cAMP signaling in particular pathways (e.g., beta adrenergic or NMDA receptor-mediated signaling) result in the pattern of behavioral changes observed11. There are four PDE4 genes (i.e., PDE4A, PDE4B, PDE4C and PDE4D). The enzyme PDE4C is mainly in the periphery and was not studied herein9,10 but PDE4A, PDE4B and PDE4D are present in the brain and their distribution suggest distinct roles in neuropsychopharmacology. Repeated treatment of animals with the antidepressant desipramine (C18H21N2) which indirectly stimulates beta receptors alters PDE4A and PDE4B expression8. The PDE4 inhibitor rolipram does not produce antidepressant-like effects in PDE4D knockout mice and PDE4D may play a role in pharmacotherapy of depression9,10. Consequently, the inhibition of these PDE4 forms was studied (Table 1).
Selective Serotonin Reuptake Inhibitors
Compounds 14 and 15 were derived from molecular dissection of aryltropanes21 and were potent inhibitors of serotonin reuptake (Table 2). Compounds 14 and 15 were potent inhibitors of radiolabeled 25 binding to the hSERT (i.e., Ki values of 2.1 and 1.1 nM, respectively) and reuptake (i.e., Ki values of 2.5 and 2.3 nM, respectively) (Table 2). Compounds 14 and 15 were equal to or more potent and selective than fluoxetine in reuptake inhibition of the hSERT. For 14 and 15, the selectivity of either binding potency or reuptake efficacy for hSERT was very great. Thus, the binding selectivity ratios Ki(hDAT)/Ki(hSERT) and Ki(hNET)/Ki(hSERT) for 14 and 15 were 3095, 1162 and 6642, 9636, respectively. The reuptake inhibition selectivity ratios IC50(hDAT)/IC50(hSERT) and IC50(hNET)/IC50(hSERT) for 14 and 15 were 11,140, 192 and 7386, 839, respectively. Compounds related to the PDE4 inhibitor (i.e., 19 and 20) were ineffective at inhibiting 25 binding to the hDAT, hSERT or hNET (i.e., Ki values 39-100 µM).
Dual PDE4 inhibitor/SSRIs
Compounds 19, 21, and 22 were tested for inhibition of cAMP hydrolysis by recombinant forms of human PDE4D3, human PDE4B3, and human PDE4A1, in vitro. The Ki value for inhibition of human PDE4D3 for rolipram, 19, 21 and 22 was 58.9, 6.3 nM, 2.0 nM and 1.2 nM, respectively (Table 1). The increase in potency for 21 and 22 compared with 19 suggested that N-substitution increased inhibition of human PDE4D17. For human PDE4B3, the Ki values for inhibition of cAMP hydrolysis of 19, 21 and 22 were 250 nM, 199 nM and 500 nM, respectively. In the case of human PDE4A1, compounds 19, 21 and 22 all showed Ki values for inhibition of cAMP hydrolysis with values greater than 1 µM. Thus, 19, 21 and 22 possessed considerable inhibitory selectivity for PDE4D.
The dual PDE4 inhibitor/SSRIs (i.e., compounds 21 and 22) were moderately potent inhibitors of binding of radiolabeled 25 to the hSERT having Ki values of 156 and 194 nM, respectively. For compounds 21 and 22, inhibition of neurotransmitter reuptake was highly selective for the hSERT (i.e., IC50 values of 127 and 194 nM, respectively) (Table 2). For 21 and 22, the selectivity of either binding potency or reuptake efficacy was greatest for hSERT. Finally, the data showed that dual PDE4 inhibitor/SSRIs 21 and 22 were relatively selective for the hSERT and did not potently interact with hDAT or hNET (Table 2).
In Vivo Studies
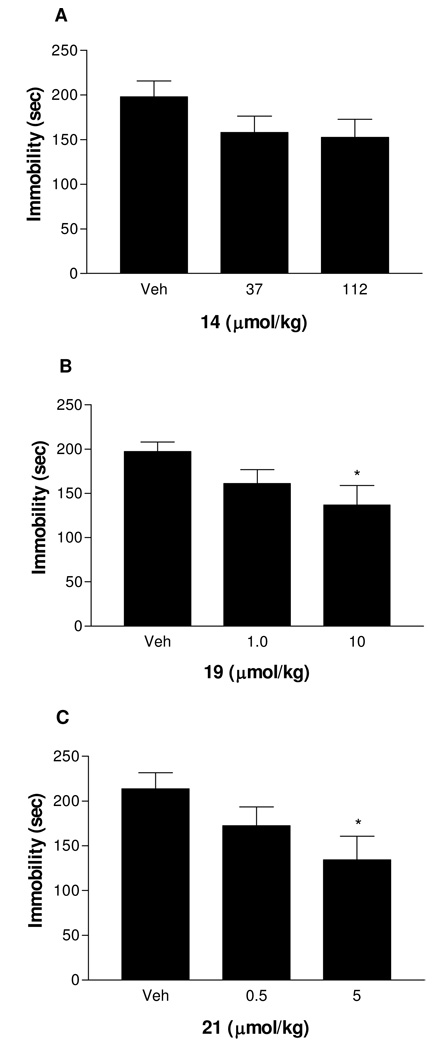
The PDE4 inhibitor (i.e., 19), the SSRI (i.e., 14) and the dual PDE4 inhibitor/SSRI (i.e., 21) were separately examined for acute and subchronic antidepressant-like effects in vivo. The two compounds were not administered in combination. Acute treatment of Inbred Charles River (ICR) mice with either 19 or 21, at doses of 0.3 and 3 mg/kg, decreased duration of immobility in a dose-dependent manner in the forced-swim test (FST)22,23 (Figure 1). Compared to vehicle control, the higher dose of 19 or 21 (3 mg/kg) significantly decreased immobility in the FST (P < 0.05). In contrast, acute administration of fluoxetine (40 mg/kg) or repeated treatment with rolipram (0.5 mg/kg, i.p., once a day for 8 days, (data not shown) was required to produce similar antidepressant-like effects in the FST. On a molar basis, compound 21 (0.5 µmol/kg) was 129-fold more efficacious than fluoxetine (65 µmol/kg) in the FST to give the same effect. Acute treatment with rolipram (0.5 mg/kg, data not shown) did not alter FST behavior in agreement with the literature8. The SSRI, compound 14, at the doses used (i.e., 10 and 30 mg/kg) tended to decrease immobility, but this effect was not statistically significant (Figure 1).
Figure 1.
Effect of acute treatment of test compounds on immobility in the forced-swim test in ICR mice: (A) compound 14 (10 mg/kg, 37 µmol/kg and 30 mg/kg, 112 µmol/kg); (B) compound 19 (0.3 mg/kg, 1.0 µmol/kg and 3 mg/kg, 10 µµmol/kg) and (C) compound 21 (0.3 mg/kg, 0.5 µmol/kg and 3 mg/kg, 5 µmol/kg). Vehicle (Veh) or test agent was injected i.p. 30 min prior to the test. n = 9–11; * P < 0.05 vs. Veh.
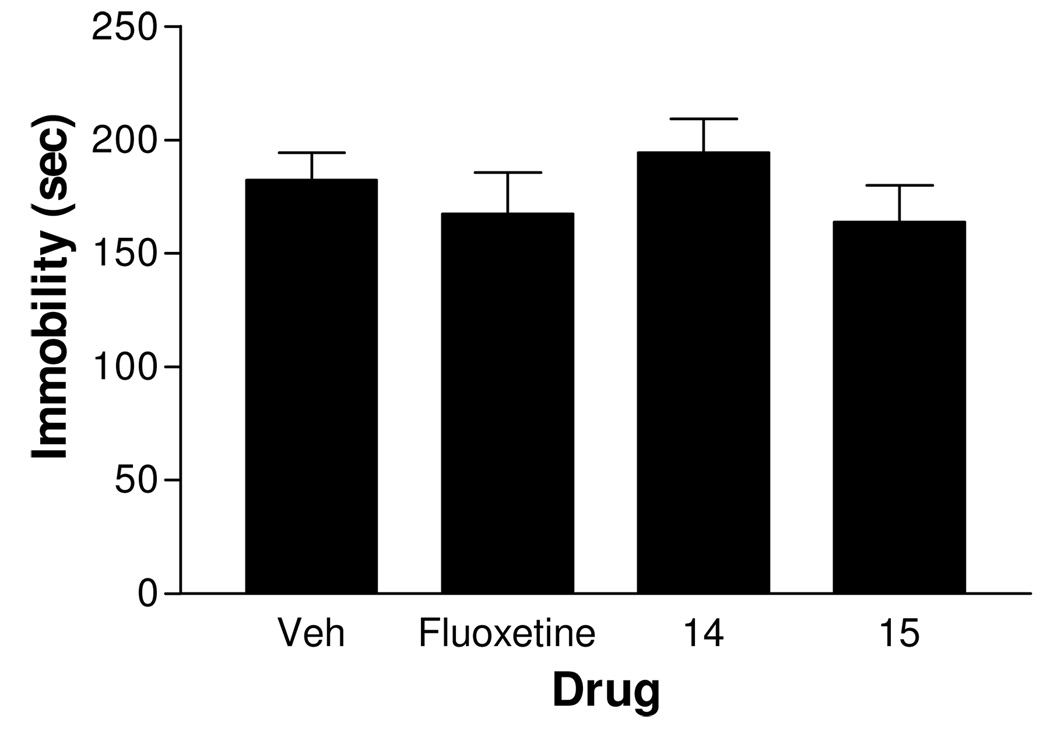
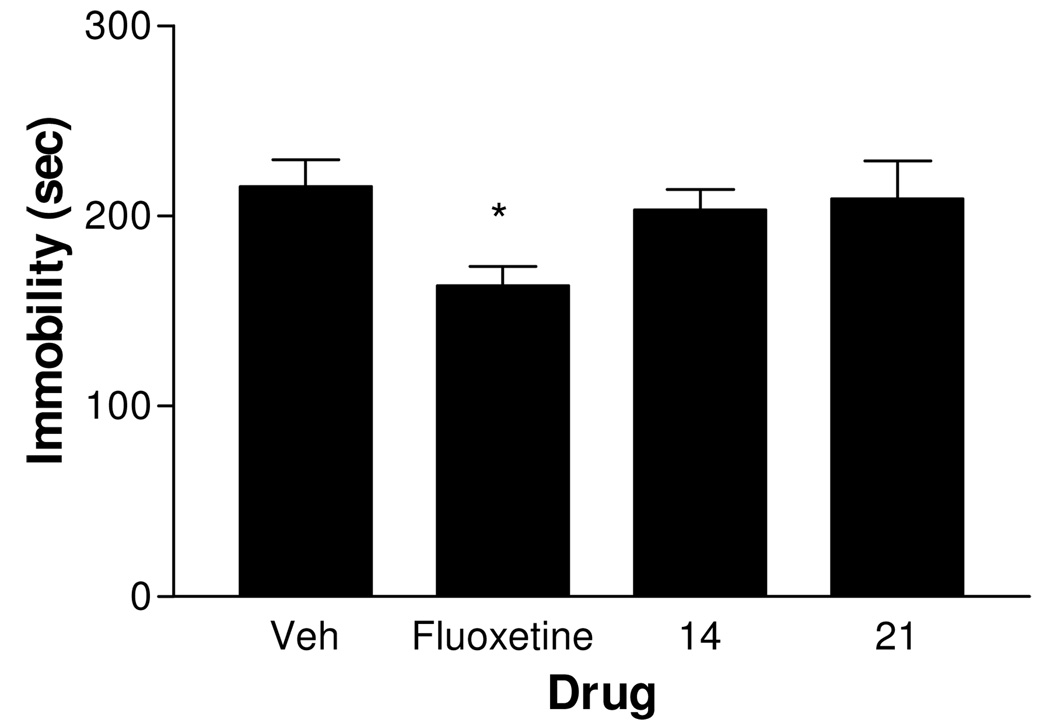
Because Balb/c mice have been reported to exhibit a relatively high immobility baseline and are sensitive to fluoxetine challenge in the FST23, the in vivo studies described above were repeated. Acute administration of fluoxetine (20 mg/kg), 14 (10 mg/kg), or 15 (10 mg/kg) did not markedly alter the duration of immobility in the FST in Balb/c mice (Figure 2). In contrast, subchronic administration of a lower dose of fluoxetine (10 mg/kg) significantly decreased immobility (P < 0.05). However, subchronic treatment with 14 (10 mg/kg) or 21 (1 mg/kg) did not change immobility compared to vehicle control (Figure 3). It is possible that the lack of in vivo activity for 14 or 15 was a consequence of metabolic instability. To test this possibility, the in vitro metabolic stability of 14, 15, 19, 21, 22 and 24 was examined.
Figure 2.
Effect of acute treatment with fluoxetine (20 mg/kg, 65 µmol/kg), compound 14 (10 mg/kg, 37.4 µmol/kg), and compound 15 (10 mg/kg, 35.5 µmol/kg) on immobility in the forced-swim test in Balb/c mice. Vehicle or test agent was injected i.p. 30 min prior to the test. n = 10–11.
Figure 3.
Effect of subchronic administration of fluoxetine (10 mg/kg, 32.3 µmol/kg), compound 14 (10 mg/kg, 37.4 µmol/kg) and compound 21 (1.0 mg/kg, 1.6 µmol/kg) on immobility in the forced-swim test in Balb/c mice. Vehicle or the test agent was injected i.p. 23, 5, and 1 hr prior to the test. n = 10–11; * P < 0.05 vs. vehicle.
In Vitro Metabolic Studies
The in vitro metabolism of compounds 14, and 24, 2-{5-[3-(5-fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-methyl-amine were examined in the presence of mouse and human liver microsomes supplemented with NADPH to examine the metabolic stability of the compounds and because metabolism plays a large role in the pharmacological activity of SSRIs. We hypothesized that by blocking a prominent route of metabolism of SSRIs (i.e., aliphatic amine metabolism) a more efficacious agent could be obtained. Compounds 19, 21 and 22 were examined in the presence of mouse and human liver microsomes supplemented with NADPH. An efficient HPLC method was established to afford separation of the starting material from any putative metabolites (e.g., aldehyde, hydroxylamine, nitrone). Aerobic incubations of 14 and 24 in the presence of mouse and rat liver microsomes supplemented with NADPH showed a time-dependent loss of substrate. For 14 and 24, the calculated half life was 56 or 106 and 58 or 129 mins, respectively, in the presence of mouse and rat liver microsomes (Table 3). In the presence of mouse liver microsomes, compounds 19 and 21 showed considerable metabolic stability having calculated half lives of 211 and 154 mins, respectively. Compound 21 was not detectably metabolized in the presence of human liver microsomes. No detectable metabolic instability of compound 22 was observed in the presence of either mouse or human liver microsomes up to 60 min (Table 3) and we judged these compounds were metabolically stable enough to undertake in vivo studies. Lack of efficacy of 14 and 15 was likely not due to metabolism.
Table 3.
Effect of Hepatic Microsomes on the Metabolic Stability of SSRIs, PDE4 Inhibitors and Dual PDE4 inhibitor/SSRIs.
| Comp | RLMa t1/2 (min) |
MLMb t1/2 (min) |
HLMc t1/2 (min) |
|---|---|---|---|
| 14 | 106.3 | 56.0 | NAd |
| 19 | NA | 211.3 | NDe |
| 21 | NA | 153.9 | ND |
| 22 | NA | ND | ND |
| 24 | 129.0 | 58.5 | NA |
RLM, rat liver microsomes
MLM, mouse liver microsomes
HLM, human liver microsomes
NA, Not available.
ND, No detectable degradation observed in the presence of animal liver microsomes supplemented with NADPH for 60 mins.
Discussion
A dual PDE4 inhibitor/SSRI was synthesized by linking an SSRI with a PDE4 inhibitor via a five carbon linker. Previously, it was shown that the potency of PDE4 inhibition of 19 was not diminished by addition of lipophilic bulk to the phthalazin-1-one nitrogen atom and 20 was a more potent PDE4D3 inhibitor than 19. Thus, elaboration of 21 and 22 afforded PDE4 inhibitors linked to an SSRI that retained considerable pharmacological activity. Compared to 19, compounds 21 and 22 possessed more potency at inhibiting PDE4. The dual PDE4 inhibitor/SSRIs 21 and 22 possessed considerable potency for inhibition of binding of 25 and inhibition of 5-HT reuptake in the presence of the hSERT. Compared to 14 and 15, however, some functional activity was lost. Because studies have shown that the trans diastereomer of 14 or 15 possess about 7-fold more hSERT re-uptake inhibition activity than the cis diastereomer (unpublished data) preparation of a dual inhibitor with only the trans diastereomer should increase potency. While some loss of in vitro binding potency and reuptake inhibition of the hSERT was observed in chemically linking SSRIs 14 and 15 to compound 20, nevertheless, significant in vivo activity was retained.
A dual PDE4 inhibitor/SSRI offers advantages beyond simple additive effects of co-administration of the individual agents including providing greater symptomatic efficacy and better utility. The ‘message-address’ concept of a dual agent could afford proximal inhibition of PDE4 thus keeping ample cAMP concentrations present near the activated transporter for greater functional selectivity. Decrease in the catabolism of cAMP might be expected to increase the sensitivity to and effectiveness of SSRI antidepressants. Evidence points to the PDE4D subtype as an important component of the signaling pathway involved in mediation of antidepressant effects on behavior. Dual PDE4 inhibitor/SSRI can affect local control of 5-HT and cAMP in a stimulus-selective manner because of the compartmentalization of the PDE enzymes. In fact, acute treatment of animals with fluoxetine that elevated synaptic 5-HT and NE but not DA levels, significantly enhanced rolipram binding and retention16. By chemically combining a selective PDE4 inhibitor with an SSRI, synergistic antidepressant effects might be observed.
The greater PDE4 inhibitor potency of the dual agent will allow lower doses to be used and decrease side effects. Because CNS disorders are recognized as poly-etiological in nature, drugs that modulate multiple targets will contribute to the multi-factorial processes in disease treatment. PDE4D inhibition results in a pattern of behavior that is indicative of an antidepressant-like effects13, 14. This suggests that PDE4D is critical in mediating the antidepressant-like effects of PDE4 inhibitors. The PDE4 inhibitor 19 and the dual PDE4 inhibitor/SSRI 21 at doses of 0.3 and 3 mg/kg, respectively, decreased duration of immobility in the FST in a dose-dependent manner in ICR mice. Compared to vehicle control, 3 mg/kg significantly decreased immobility in the FST (P < 0.05). Acute administration of 14 decreased immobility in the FST but this did not reach statistical significance. In contrast, 40 mg/kg of fluoxetine or repeated treatment with rolipram (0.5 mg/kg, i.p., once a day for 8 days) was required to produce similar antidepressant-like effects in the FST; acute treatment with 0.5 mg/kg rolipram did not alter FST behavior.
Acute administration of fluoxetine (20 mg/kg) or 14 (10 mg/kg) did not alter the duration of immobility in the FST in Balb/c mice but SSRIs are known to not significantly affect changes in FST after acute administration. In contrast, subchronic administration of a lower dose of fluoxetine (10 mg/kg) significantly decreased immobility (P < 0.05). However, subchronic treatment with 14 or 15 (10 or 1 mg/kg, respectively) did not change immobility compared to the vehicle control.
In summary, we hypothesize that dual PDE4 inhibitor/SSRIs offer an advantage over simple additive effects of co-administered agents. Dual PDE4 inhibitor/SSRIs offer the advantage of blocking the effect of the up-regulation of the PDE4 in a compartmentalized manner. While PDE4 expression will still increase, its hydrolytic activity will be blocked. Thus, the overall increase in serotonin receptor-mediated cAMP signaling will be preserved with repeated treatment. Moreover, we suggest that compounds such as 21 show the utility of addressing multiple targets to alleviate complex disease such as depression.
Experimental Section
General
Chemicals used in this study were of the highest purity available. Commercially available reagents including 2-methoxy-5-fluoro benzaldehyde were purchased from Aldrich Chemical Company (Milwaukee, WI) or VWR (San Diego, CA) and were used as received. All moisture sensitive reactions were carried out in flame-dried glassware under an argon atmosphere. Tetrahydrofuran (THF) and toluene were freshly distilled from calcium hydride under an argon atmosphere. Methanol (CH3OH) was passed through a column of neutral alumina and stored over 3Å molecular sieves prior to use. Rolipram, fluoxetine, diethyleneaminetetracetic acid (DETAPAC) and all the compounds of the NADPH-generating system were obtained from Sigma Chemical Company (St. Louis, MO). Human liver, pooled female liver and pooled male rat liver microsomes were purchased from BD Gentest Corp. (Woburn, MA). The human liver microsomes had the following functional activities (nmol/min/mg of protein): phenacetin O-deethylase (0.18), coumarin 7-hydroxylase (2.0), (S)-mephenytoin N-demethylase (0.05), diclofenac 4’-hydroxylase (1.9), (S)-mephenytoin 4’-hydroxylase (0.03), bufuralol 1’-hydroxylase (0.12), chlorzoxazone 6-hydroxylase (1.5), testosterone 6β-hydroxylase (4.6), and methyl p-tolyl sulfide oxidase (4.3). The pooled male rat liver microsomes had the following functional activities (nmol/min/mg of protein): testosterone 6β-hydroxylase (5.8) and nicotine oxidase (2.2). The pooled female mouse liver microsomes had the following functional activities (nmol/min/mg of protein): 7-ethoxyresorufin O-deethylase (0.39), p-nitrophenol hydroxylase (1.9), lauric acid-hydroxylase (0.86), and testosterone 6β-hydroxylase (5.0). Cocaine was provided by the National Institute on Drug Abuse, NIH (Bethesda, MD). Compound 25) was a kind gift of Dr. Ivy Carroll (RTI, Research Triangle Park, NC). [3H]-DA, [3H]-5-HT, [3H]-NE and [125I]-25 were purchased from Perkin Elmer Life Sciences (Boston, MA). The preparation of the hDAT used was described previously20. The hSERT cDNA and HEK cells transfected with hNET cDNA was generously supplied by Dr. Randy Blakely (Vanderbilt University, Nashville, TN). Analytical thin-layer chromatography (TLC) was done on K6F silica gel 60 Å glass-backed plates from Whatman (Clifton, NJ). Compounds were detected using UV absorption at 254 nm and/or stained with I2 (iodine). Flash chromatography was done on (60 Å) pore silica gel from E. Merck (Darmstadt, Germany).
Instrument Analysis
NMR spectra were recorded with a Bruker spectrometer operating at 500 MHz (NuMega Resonance Labs, Inc., San Diego, CA) or at 300 MHz by an in-house Varian spectrometer (Palo Alto, CA) using the solvent specified. Chemical shifts were reported in parts per million (ppm, δ) using residual solvent signals as internal standards. Low resolution mass spectroscopy (LRMS) was done with an HP 1100 mass spectrometer (HT Laboratories, San Diego, CA) using electrospray ionization (ESI) or with an inhouse Hitachi M-8000 3DQMS (ion trap) mass spectrometer using ESI. High resolution mass spectroscopy (HRMS) was done with a Micromass LCT time of flight mass spectrometer at the University of Montana Mass Spectral Facility (Missoula, MT) using ESI.
The 2,5-disubstituted tetrahydrofurans and the dual PDE4 inhibitor/SSRIs were characterized by 1H NMR, LRMS, HRMS and their purities (> 95%) were quantified by HPLC in two distinct solvent systems. Analytical HPLC measurements were run on a Hitachi L-6200 system equipped with a Hitachi L-7400 UV detector. Separations were done with a 5 µm, 50 mm, Axxi-chrom silica column (Richard Scientific, Novato, CA) or with a 5 µm, 4.6 mm × 250 mm Supelco HS F5 pentafluorophenyl column (Supelco Inc., Bellefonte, PA). HPLC analysis is described in the Supporting Information. 1H NMR and mass spectra were consistent with the assigned structures.
Transporter Binding Assays
HEK-hDAT, -hSERT, or -hNET cells were grown until confluent as described previously20, 24. Cells were scraped from plates and centrifuged for 20 min at 30,000 × g and the pellet was resuspended in 0.32 M sucrose with a Polytron at a setting of 1 for 5 sec. Assays contained 50 µL of membrane preparation, 25 µL of the test compound and 25 µL of [125I]-25 (40–80 pM final concentration) in a final volume of 250 µL Krebs HEPES buffer (25 mM HEPES, 122 mM NaCl, 5 mM KCl, 1.2 mM MgSO4, 2.5 mM CaCl2, 1 µM pargyline, 100 µM tropolone, 2 mg glucose/mL and 0.2 mg ascorbic acid/mL at pH 7.4.). Membranes were pre-incubated with test compounds for 10 min before addition of [125I]-25. Specific binding was determined as the difference in binding observed in the presence and absence of 5 µM mazindol (HEK-hDAT and hNET) or 5 µM imipramine (HEK-hSERT). The incubations were done in the dark and terminated by filtration onto a Whatman GF/C filters using a 96-well Tomtech cell harvester (Tomtech, Orange, CT). Scintillation fluid was added to each filter spot and radioactivity remaining on the filter was determined using a Wallace β-plate reader (Wallace Labs, Cranbury, NJ.). Specific binding was defined as the difference in binding in the presence and absence of 5 µM mazindol (hDAT and hNET) or 5 µM imipramine (hSERT).
Inhibition of Substrate Uptake
HEK-hDAT, -hSERT or -hNET cells were grown as described above. Cells were scraped from the plates and suspended cells were added to a 96-well plate containing test compounds and Krebs-HEPES buffer in a final assay volume of 0.5 mL. After a 10 min preincubation in a 25 °C water bath, [3H]-labeled neurotransmitter (50 µL, 20 nM final concentration) was added and the assay was initiated. After 10 min the incubation was terminated by filtration onto GF/C filters presoaked with 0.05% polyethylenimine using a Tomtech call harvester and radioactivity remaining on the filters was determined as described above. Specific uptake was defined as the difference in uptake in the presence and absence of 5 µM mazindol (hDAT and hNET) or 5 µM imipramine (hSERT).
PDE4 Assay
PDE4 enzyme assays were carried out as described previously25. Studies with recombinant PDE4 enzymes were carried out in the laboratory of Professor Marco Conti (Stanford University, Palo Alto, CA).
Data Analysis
GraphPad Prism (GraphPad Software, San Diego, CA) was used to determine the saturation and binding kinetic data. IC50 values were converted to Ki values using the Cheng-Prusoff equation.
Mouse and Rat Liver Microsome Stability Assay
Microsomal incubations (final volume 0.25 mL) with the SSRIs, dual PDE4 inhibitor/SSRIs or PDE4 inhibitor was done with an HPLC assay as described above. Diluted stocks of either mouse, human or rat liver microsomes (0.5 mg of protein), 100 µM potassium phosphate buffer (pH 7.4), 0.5 mM NADP+, 0.5 mM glucose-6-phosphate, 5 IU/mL glucose-6-phosphate dehydrogenase, 0.6 mM DETAPAC, and 3 mM MgCl2 were combined and placed on ice. For a metabolic stability assay, either vehicle or 40 µM test compound was added and the incubation was initiated at 37 °C with constant shaking. After 0, 10, 25, 40 and 60 min, the incubations were stopped by the addition of 1 mL CH2Cl2/2-propanol (3:1, v:v). After thorough mixing, the organic layer was separated from the aqueous portion by centrifugation at 12,000 × g. The organic material was evaporated with a stream of argon. The residue was dissolved in methanol (200 µL) and the sample was analyzed by HPLC.
In Vivo Evaluation of Selected Compounds
Separate non-habituated male mice were used in the conduct of this work. Fully approved animal protocols were used and the studies were done in keeping with the NIH standards for use of experimental animals.
Experiment 1
Forty male ICR mice, weighing 26.2 ± 0.3 g, were housed in a temperature-controlled room (22–23°C) and maintained on a 12-hour on 12-hour off light cycle (lights on at 6:00 AM). Water and food were freely available in the home cages. The mice were randomly divided into four groups: (A) vehicle; (B) compound 14; (C) compound 19; and (D) compound 21 and administered compounds were dissolved in 0.9% saline containing 5% dimethyl sulfoxide (DMSO). Saline containing the same percentage of DMSO was used as vehicle. The lower doses (i.e., 0.3 mg/kg for 19 and 21 or 10 mg/kg for 14) were tested followed by the larger doses (3 mg/kg for 19 and 21 or 30 mg/kg for 14) 1 week later, when the mice were again randomly divided into four groups as described above (Figure 1). The forced-swim test (FST) was carried out as described previously23. Mice were given a swimming pretest session, once a day for two successive days. Twenty-four hours after the last session, mice were injected with vehicle or the lower dose of each test compound by the i.p. route 30 min prior to the FST. One week later, administration of the larger dose of each of the test compounds was repeated. During the pretest and test sessions, each mouse was placed for 6 min in a plastic cylinder (45 cm high × 20 cm diameter), that was filled to a depth of 28 cm with water (23 ± 1°C). The duration of immobility, that was defined as floating in an upright position without additional activity other than that necessary for the animal to keep its head above water, was recorded22,23. In a control experiment, acute administration of fluoxetine (40 mg/kg) or repeated treatment with rolipram (0.5 mg/kg, i.p., once a day for 8 days, was required to produce similar antidepressant-like effects in the FST (data not shown).
Experiment 2
Forty Balb/c male mice weighing 22.4 ± 0.1 g were used in this experiment, which was carried out in two sessions. In each session, the mice were divided into four groups: (A) Vehicle (saline containing 5% DMSO), (B) fluoxetine, 20 mg/kg, (C) compound 21, 10 mg/kg, and (D) compound 14, 10 mg/kg for the first session; (A) Vehicle, (B) Fluoxetine, 10 mg/kg, (C) compound 14, 10 mg/kg, and (D) compound 21, 10 mg/kg for the second session, which was carried out 4 weeks later. Mice in the first session (Figure 2) were treated with vehicle or the test agents acutely, whereas mice in the second sessions (Figure 3) were treated subchronically, i.e., vehicle or drugs were given i.p. 23, 5, and 1 hr before the FST. The experiment was done similar to Experiment 1 except for the pretest training, which was not carried out in the second session.
In Vivo Statistics
All in vivo data were analyzed by one-way analyses of variance (ANOVA) followed by Dunnett’s tests for post hoc comparisons of individual means.
Synthesis
Ethyl-3-(2’- Methoxy-5’-fluorophenyl)-2-propenoate (2)
To a solution of 2-methoxy-5-fluoro benzaldehyde (4.3 g, 28.0 mmol, 1 equiv.) in dry CH2Cl2 (30 mL) at 0 °C was added carbethoxymethylenetriphenylphosphorane (10.7 g, 30.1 mmol, 1.1 equiv.) portion-wise and the reaction was warmed to room temperature and stirred for 12 h. The solvent was then removed in vacuo and the residue was purified by flash column chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.2) to yield the product (6.0 g, 95%) as a 4:1 mixture of trans:cis diastereomers; 1 H NMR (CDCl3, 500 MHz): δ 7.93 (d, J = 16.2 Hz, 0.8 H), 7.33 (dd, J = 3.1, 9.2 Hz, 0.2 H), 7.21 (dd, J = 3.1, 9.2, Hz, 0.8 H), 7.08 (d, J = 12.5 Hz, 0.2 H), 7.03 (dt, J = 3.0, 8.8 Hz, 0.8 H), 6.99 (dt, J = 3.0, 8.8 Hz, 0.2 H), 6.84 (dd, J = 3.1, 8.6 Hz, 0.8 H), 6.79 (dd, J = 3.3, 9.1 Hz, 0.2 H), 6.47 (d, J = 16.2 Hz, 0.8 H), 5.99 (d, J = 12.5 hz, 0.2 H), 4.25 (q, J = 7.1 Hz, 1.6 H), 4.15 (q, J = 7.0 Hz, 0.4 H), 3.86 (s, 2.4 H), 3.81 (s, 0.6 H), 1.34 (t, J = 7.1 Hz, 2.4 H), 1.22 (t, J = 7.0 Hz, 0.6 H).
Ethyl-4-(5’-fluoro-2’-methoxyphenyl)propanoate (3)
To a solution of 2 (6.0 g, 27.0 mmol, 1 equiv.) in ethanol (200 proof, 40 mL) under Ar was added Pd/C (10%, 250 mg, 0.1 equiv.) and the flask containing the mixture was evacuated and purged with H2 three times. H2 was attached to the flask and the reaction was allowed to stir for 15 h at room temperature. The reaction mixture was filtered through a pad of silica and eluted with EtOAc/hexane, 50:50, v:v. The crude product obtained by removal of the solvent in vacuo was purified by a flash column chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.27) to give the product (5.8 g, 95%); 1 H NMR (CDCl3, 500 MHz): δ 6.89-6.84 (m, 2 H), 6.74 (dd, J = 4.7, 9.8 Hz, 1 H), 4.12 (q, J = 7.1 Hz, 2 H), 3.79 (s, 3 H), 2.91 (t, J = 7.7 Hz, 2 H), 2.59 (t, J = 7.7 Hz, 2 H), 1.24 (t, J = 7.2 Hz, 3 H).
3-(5’-Fluoro-2’-methoxyphenyl)propanal (4)
To a solution of 3 (5.8 g, 25.7 mmol, 1 equiv.) in dry toluene (40 mL) under an atmosphere of Ar was added a DIBAL solution (1 M in toluene, 30 mL, 30 mmol, 1.2 equiv.) at −78°C. The reaction was stirred at −78°C for 2 h. Methanol (2 mL) was then added to the mixture and the reaction was allowed to warm to 0 °C. The reaction mixture was then poured into a separatory funnel containing HCl solution (1 N, 100 mL). The organic fraction was extracted with EtOAc (3 × 60 mL) and the combined organic layers were washed with brine (80 mL) and dried over sodium sulfate. The crude product obtained by removal of the solvent in vacuo was purified by flash column chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.1) to give the product (3.1 g, 66%); 1 H NMR (CDCl3, 500 MHz): δ 9.80 (s, 1 H), 6.88-6.86 (m, 2 H), 6.75 (dd, J = 4.6, 8.6 Hz, 1 H), 3.79 (s, 3 H), 2.91 (t, J = 7.4 Hz, 2 H), 2.72 (t, J = 7.4 Hz, 2 H).
1-(5’-Fluoro-2’-methoxyphenyl)-4-ethylenedioxy-1-butene (5)
To a suspension of 2-(1,3-dioxolan-2-yl)ethyltriphenylphosphonium bromide (5.0 g, 11.3 mmol, 1 equiv.) in THF (30 mL) under an atmosphere of Ar was added NaH (60% in mineral oil, 0.48 g, 11.3 mmol, 1 equiv.) and the mixture obtained was heated to reflux for 1 h. The orange suspension obtained was cooled to 0 °C and 5-fluoro-2-methoxybenzaldehyde (1.54 g, 10 mmol, 0.9 equiv.) was added and the reaction was warmed to room temperature and continued with stirring for 12 h. The reaction mixture was poured into a separatory funnel containing ammonium chloride aqueous solution (sat. NH4Cl/H2O 50:50, v:v, 50 mL). The organic material was extracted with EtOAc (3 × 80 mL) and the combined organic layers were washed with brine (60 mL) and dried over sodium sulfate. The solvent was then removed in vacuo and the crude product obtained was purified by flash column chromatography (EtOAc/hexane, 7:93, v:v, Rf = 0.15) to give the product (2.63 g, 66%) as a 4:1 mixture of cis and trans diastereomers: 1 H NMR (major isomer): (CDCl3, 500 MHz): δ 7.08 (dd, J = 3.1, 8.7 Hz, 1 H), 6.93 (dt, J = 3.1, 8.7 Hz, 1 H), 6.78 (dd, J = 4.3, 8.7 Hz, 1 H), 6.63 (d, J = 11.9 Hz, 1 H), 5.83 (td, J = 7.3 Hz, 11.9 Hz, 1 H), 4.99 (t, J = 4.6 Hz, 1 H), 4.02-3.99 (m, 2 H), 3.9-3.87 (m, 2 H), 3.8 (s, 3 H), 2.64-2.62 (m, 2 H).
4-(5’-Fluoro-2’-methoxyphenyl)-1-ethylenedioxybutane (6)
To a solution of 5 (1.75 g, 7.3 mmol, 1 equiv) in ethanol (200 proof, 80 mL) under an atmosphere of Ar was added Pd/C (10%, 300 mg, 0.1 equiv.) and the flask containing the mixture was evacuated and purged with H2 three times. H2 gas was attached to the flask and the reaction was allowed to proceed for 72 h at room temperature. The reaction mixture was then filtered through a pad of Celite and eluted with EtOH. The crude product was obtained by removal of the solvent in vacuo. 1 H NMR (CDCl3, 500 MHz): δ 6.87-6.72 (m, 3 H), 4.87 (t, J = 3.8 Hz, 1 H), 3.97-3.95 (m, 2 H), 3.86-3.83 (m, 2 H), 3.79 (s, 3 H), 2.64-2.62 (m, 2 H), 2.44 (td, J = 1.7, 7.2 Hz, 2 H), 1.92 (quintet, J = 7.5 Hz, 2 H).
4-(5’-Fluoro-2’-methoxyphenyl)butyraldehyde (7)
To a solution of 6 (1.7g, 7.0 mmol, 1 equiv.) in THF (60 mL) was added HCl (1 N, 3 mL) and the reaction was stirred at room temperature for 48 h. The reaction mixture was then poured into a separatory funnel containing water (60 mL). The organic fraction was extracted with EtOAc (3 × 60 mL) and the combined organic layers were washed with brine (80 mL) and dried over sodium sulfate. The crude product obtained by removal of the solvent in vacuo was purified by flash column chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.1) to give the product (1.03 g, 53% from 3a); 1 H NMR (CDCl3, 500 MHz): δ 9.76 (s, 1 H), 6.88-6.83 (m, 2 H), 6.75 (dd, J = 4.6, 8.6 Hz, 1 H), 3.79 (s, 3 H), 2.63 (t, J = 7.4 Hz, 2 H), 2.44 (t, J = 7.4 Hz, 2 H), 1.92 (quintet, J = 7.4 Hz, 2 H).
7-(5’-Fluoro-2’-methoxyphenyl)hept-1-en-5-ol (8)
To a solution of 4 (3.0 g, 19.5 mmol, 1 equiv.) in THF (20 mL) at 0 °C was added a solution 1-butenylmagnesium bromide (0.5 M in THF, 45 mL, 22.5 mmol, 1.1 equiv.) dropwise for 15 min. The reaction mixture was poured into a solution of saturated ammonium chloride (80 mL) in a separatory funnel. The organic fraction was extracted with EtOAc (3 × 60 mL) and the combined organic layers were washed with brine (50 mL) and dried over sodium sulfate. The product was purified by flash column chromatography (EtOAc/hexane, 15:85, v:v, Rf = 0.2) to give the product as a colorless oil (3.7 g, 94%); 1 H NMR (CDCl3, 500 MHz): δ 6.86-6.82 (m, 2 H), 6.76 (dd, J = 4.6, 8.6 Hz, 1 H), 5.82 (m, 1 H), 5.04 (d, J = 17.3 Hz, 1 H), 4.95 (d, J = 10.2 Hz, 1 H), 3.81 (s, 3 H), 3.55 (m, 1 H),2.75-2.69 (m, 2 H), 2.2 (m, 1 H), 2.1 (m, 1 H), 1.96 (bs, 1 H), 1.73-1.68 (m, 2 H), 1.6-1.53 (m, 3 H).
The following compound was prepared in a similar manner.
8-(5’-Fluoro-2’-methoxyphenyl)oct-1-en-5-ol (9)
The product was purified by flash column chromatography (EtOAc/hexane, 12.5:87.5, v:v, Rf = 0.15) as a colorless oil (1.06 g, 61%); 1 H NMR (CDCl3, 500 MHz): δ 6.86-6.82 (m, 2 H), 6.74 (dd, J = 4.6, 8.6 Hz, 1 H), 5.85 (m, 1 H), 5.04 (d, J = 17.3 Hz, 1 H), 4.97 (d, J = 10.2 Hz, 1 H), 3.79 (s, 3 H), 3.66 (m, 1 H), 2.6 (m, 2 H), 2.22-2.1 (m, 2 H), 1.74-1.47 (m, 7 H).
2-(Bromomethyl)-5-(2’-methoxy-5’-fluorophenethyl)tetrahydrafuran (10)
To a solution of 8 (3.24 g, 18.0 mmol, 1 equiv.) in dry CH2Cl2 (50 mL) at 0 °C was added N-bromosuccinimide (3.56 g, 20 mmol, 1.1 equiv.) portion-wise and the reaction was warmed to room temperature and stirred for 12 h. Solvent was then removed in vacuo and the product was purified by flash column chromatography (EtOAc/hexane, 5:95, v:v, Rf = 0.1) and resulted in a colorless oil as a 2:1 mixture of trans:cis diastereomers; 1 H NMR (CDCl3, 500 MHz): δ 6.86-6.81 (m, 2 H), 6.72 (dd, J = 4.6, 8.6 Hz, 1 H), 4.22 (m, 1 H), 4.03 (m, 1 H), 3.78 (s, 3 H), 3.44 (dd, J = 4.3, 9.7 Hz, 1 H), 3.34 (dd, J = 6.4, 9.7 Hz, 1 H), 2.6 (t, J = 7.3 Hz, 2 H), 2.13 (m, 1 H), 2.05 (m, 1 H), 1.75 (m, 1 H), 1.67-1.46 (m, 5 H).
The following compound was prepared in a similar manner.
2-(Bromomethyl)-5-(3’-(2'’-methoxy-5'’-fluorophenyl)-1’-propyl)tetrahydrofuran (11)
the product was purified by flash column chromatography (EtOAc/hexane, 5:95, v:v, Rf = 0.1) to provide a product (530 mg, 40%) as a colorless oil; 1 H NMR (CDCl3, 500 MHz): δ 6.86-6.81 (m, 2 H), 6.72 (dd, J = 4.6, 8.6 Hz, 1 H), 4.22 (m, 1 H), 4.03 (m, 1 H), 3.78 (s, 3 H), 3.44 (dd, J = 4.3, 9.7 Hz, 1 H), 3.34 (dd, J = 6.4, 9.7 Hz, 1 H), 2.6 (t, J = 7.3 Hz, 2 H), 2.13 (m, 1 H), 2.05 (m, 1 H), 1.75 (m, 1 H), 1.67-1.46 (m, 5 H).
2-Cyanomethyl-5-(5’-fluoro-2-methoxyphenethyl)tetrahydrofuran (12)
To a vial under an atmosphere of Ar(g) was added 10 (1.23 g, 4.3 mmol, 1 equiv.), NaI (100 mg, 1.3 mmol, 0.3 equiv.), potassium cyanide (0.7 g, 10.6 mmol, 2.4 equiv.), and dry DMSO (15 mL). The mixture obtained was heated to 70 °C under an atmosphere of Ar for 12 h. After cooling to room temperature, the reaction mixture was poured into a separatory funnel containing sodium bicarcarbonate aqueous solution (sat. NaHCO3/H2O, 50:50, v:v, 80 mL). The organic fraction was extracted with EtOAc (3 × 60 mL) and the combined organic layers were washed with brine (50 mL) and dried over sodium sulfate. The solvent was then removed in vacuo and the crude product obtained (0.45 g, 86%) was purified by flash column chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.11); as a 2:1 mixture of trans/cis diastereomers. 1 H NMR (500 MHz, CDCl3) δ 6.88-6.82 (m, 2 H), 6.74 (dd, J = 4.3, 8.5 Hz, 1 H), 4.26 (m, 1 H), 4.09 (m, 0.65 H), 3.90 (m, 0.35 H)3.79 (s, 3 H), 2.72-2.53 (m, 4 H), 2.22-2.04 (m, 2 H), 1.84-1.6 (m, 4 H).
The following compound was prepared in a similar manner.
2-Cyanomethyl-5-(3’-(2”-methoxy-5”-fluorophenyl)-1’-propyl)tetrahydrafuran (13)
the product (0.2 g, 80%) was purified by chromatography (EtOAc/hexane, 10:90, v:v, Rf = 0.11) to give a 2:1 mixture of trans/cis diastereomers; 1 H NMR (CDCl3, 500 MHz): δ 6.86-6.81 (m, 2 H), 6.73 (dd, J = 4.2, 8.5 Hz, 1 H), 4.22 (p, J = 6.8 Hz, 0.65 H), 4.11 (m, 0.35 H) 4.09 (m, 0.65 H), 3.9 (m, 0.35 H), 3.79 (s, 3 H), 2.62-2.52 (m, 4 H), 2.18 (m, 1 H), 2.12 (m, 1 H), 1.78 (m, 1 H), 1.69-1.46 (m, 3 H).
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine (14)
To a round bottom flask under an atmosphere of Ar was added Raney Ni (5 mg, 0.03 mmol, 0.1 equiv.) that was washed with methanol. Compound 12 (80 mg, 0.3 mmol, 1 equiv.), dissolved in 2 M NH3 in methanol was added to the flask. The flask was evacuated and purged with H2 once. H2 gas was then attached to the flask and the reaction was allowed to stirr for 4 h at room temperature. The reaction mixture was filtered through a plug of Celite and the crude product obtained after the removal of the solvent was purified by preparative TLC and eluted with (MeOH/CH2Cl2, 10:90, v:v, Rf = 0.1) to give the product (55 mg, 70%) as a 2:1 mixture of trans/cis diastereomers; 1 H NMR: (CDCl3, 500 MHz): δ 6.87 (m, 1 H), 6.82 (dd, J = 3.0, 8.5 Hz, 1 H), 6.72 (m, 1 H), 4.03 (m, 0.65 H), 3.96 (m, 0.65 H), 3.89 (m, 0.35 H), 3.82 (m, 0.35 H), 3.78 (s, 3 H), 2.68 (m, 1 H), 2.58 (m, 1 H), 2.03 (m, 2 H), 1.83 (bs, 2 H), 1.86-1.6 (m, 4 H), 1.54 (m, 2 H); HRMS (ESI) [M+H]+ calcd for C15H23FNO2 268.1713, found 268.1701.
The following compound was prepared in a similar manner.
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-propyl]-tetrahydro-furan-2-yl}-ethylamine (15)
the product (24 mg, 47%), was isolated as 3:1 mixture of trans/cis diastereomers (MeOH/CH2Cl2, 10:90, v:v, Rf = 0.1): 1 H NMR: (CDCl3, 500 MHz): δ 6.86-6.8 (m, 2 H), 6.73 (m, 1 H), 4.02 (m, 0.65 H), 3.96 (m, 0.65 H), 3.92 (m, 0.35 H), 3.82 (m, 0.35 H), 3.78 (s, 3 H), 3.09 (m, 1 H), 2.99 (m, 1 H), 2.6 (bs, 2 H), 1.96 (m, 2 H), 1.83-1.77 (m, 2 H), 1.65-1.47 (m, 8 H); HRMS (ESI) [M+H]+ calcd for C16H25FNO2 282.1875, found 282.1869.
3,4-Dimethoxyphenylmagnesium bromide (17)
Into a flame dried round bottom flask with a magnetic stir bar was placed anhydrous THF (150 mL) and Mg(s) turnings (1.43 g, 58.8 mmol, 1.0 equiv.). The flask was then fitted with a pressure equalizing addition funnel containing 4-bromo-1,2-dimethoxybenzene (8.2 g, 58.8 mmol, 1 equiv.) (16) solution that was added slowly over a period of 45 min while stirring at rt. Once the addition was complete I2(s) (500 mg) was added and the reaction was brought to reflux and stirred for 12 hrs. The Grignard reagent was cooled and used immediately in the following reaction.
6-(3,4-Dimethoxy-benzoyl)-cyclohex-3-enecarboxylic acid (18)
A solution of 17 (9.6 g, 59 mmol, 0.29 M, 1 equiv.) in THF was added dropwise to an ice-cooled solution of cis-1,2,3,6-tetrahydrophthalic anhydride (8.9 g, 59 mmol, 1 equiv.) in THF (120 mL) over a 1 hr period. After the addition was complete, the resulting mixture was stirred for another 30 min at 0 °C. The reaction mixture was allowed to warm to rt and stirred overnight. The reaction was then stopped with sat. NH4Cl and the pH adjusted to 2 with concentrated HCl(aq) and extracted with diethyl ether. The organic layer was washed with water and subsequently extracted with 1 M NaOH. The combined aqueous extract was acidified with concentrated HCl and extracted with EtOAc (3× 100 mL). The organic layers were combined and dried over MgSO4, filtered, and concentrated under reduced pressure to afford oil. The oil was dissolved in CH2Cl2 and filtered through silica gel to remove the dicarboxylic acid formed during workup. The product was recrystallized from diethyl ether to afford pure product (1.62 g, 10%) as a white solid; mp = 109 °C; LRMS ESI [M-H]− calcd for C16H18O5 289, found m/z 289; 1H NMR (CDCl3, 500 MHz): δ 7.60-7.53 (m, 2 H), 6.92 (d, J = 8.4 Hz, 1 H), 5.84-5.82 (m, 1 H), 5.71-5.68 (m, 1 H), 4.03-4.00 (m, 1 H), 3.98 (s, 3 H), 3.95 (s, 1 H), 3.10-3.07 (m, 1 H), 2.90-2.84 (m, 1 H), 2.54-2.44 (m, 3 H).
4-(3,4-Dimethoxy-phenyl)-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one (19)
A mixture of 18 (610 mg, 2.1 mmol, 1.0 equiv.) and hydrazine hydrate (168 mg, 5.25 mmol, 2.5 equiv.) in EtOH (10 mL) was refluxed for 4 hrs. The reaction was then cooled to rt and concentrated under reduced pressure to afford a white precipitate. The precipitate was dissolved in EtOAc and washed with Na2SO4(aq), 1 N HCl(aq), and water. The organic layer was then dried over MgSO4, filtered, and concentrated to give a white precipitate. The precipitate was recrystalized in EtOH to afford the product as white crystals (376 mg, 63%); HRMS ESI [M+H]+ calcd for C16H18N2O3 287.3385, found m/z 287.3379; 1H NMR (CDCl3, 500 MHz): δ 8.53 (bs, 1 H), 7.46 (d, J = 2.0 Hz, 1 H), 7.23 (dd, J = 2.0, 8.4 Hz, 1 H), 6.87 (d, J = 8.4 Hz, 1 H), 5.8-5.77 (m, 1 H), 5.72-5.7 (m, 1 H), 3.94 (s, 3 H), 3.93 (s, 3 H), 3.4 (dt, J = 5.5, 8.7 Hz, 1 H), 3.01-2.97 (m, 1 H), 2.85 (t, J = 6.0 Hz, 1 H), 2.26-2.19 (m, 3 H); HRMS (ESI) [M+H]+ calcd for C16H19N2O3 287.3385, found 287.3376; HPLC > 98% pure (tR = 4.93, 70(A):30(E); tR = 5.33, 50(A):50(E)).
2-(5-Bromo-pentyl)-4-(3,4-dimethoxy-phenyl_-4a,5,8,8a-tetrahydro-2H-phthalazin-1-one (20)
Sodium hydride (60% dispersion in oil, 44 mg, 1.1 mmol, 1.1 equiv.) was added to a solution 19 (287 mg, 1.0 mmol, 1.0 equiv.). The mixture was stirred at rt for 30 min whereupon it took on a slight yellow color. 1,5-Dibromopentane (600 mg, 2.6 mmol, 2.6 eq) was added via syringe and the reaction was stirred for 30 min at rt while the yellow color dissipated to an almost clear solution. The reaction was stopped with the addition of water and then transferred to a separatory funnel and extracted with Et2O (3 × 30 mL). The organic fractions were combined and dried over MgSO4, filtered, and concentrated to afford an oil. The product was purified on silica and eluted with CH2Cl2 to afford a clear oil; 1H NMR (CDCl3, 500 MHz): δ 7.47 (d, J = 2.0 Hz, 1 H), 7.25 (dd, J = 2.0, 8.5 Hz, 1 H), 6.87 (d, J = 8.5 Hz, 1 H), 5.8-5.77 (m, 1 H), 5.7-5.67 (m, 1 H), 4.01-3.97 (m, 1 H), 3.95 (s, 3 H), 3.92 (s, 3 H), 3.79-3.74 (m, 1 H), 2.22-2.18 (m, 2 H), 2.1-2.04 (m, 1 H), 1.94-1.86 (m, 4 H), 1.74-1.69 (m, 4 H), 1.63-1.56 (m, 1 H), 1.52-1.46 (m, 2 H); LRMS ESI [M+H]+ calcd for C21H28BrN2O3 436, found m/z 435 (Br79), 437 (Br81). HRMS (ESI) [M+H]+ calcd for C21H28BrN2O3 436.8007, found 436.8025
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-pentyl]-4,5,8,8a-tetrahydro-2H-phthalazin-1-one (21)
Into a flame dried roundbottom flask under an atmosphere of Ar(g), cesium hydroxide monohydrate (23 mg, 0.14 mmol, 1.0 equiv.) and DMF (0.5 mL) was stirred for 30 min at rt. Compound 14 (37 mg, 0.14 mmol, 1.0 equiv.) in DMF (0.3 mL) was then added via syringe and the resulting mixture was stirred at rt. After stirring an additional 30 min, compound 20 (72 mg, 0.16 mmol, 1.2 eq) dissolved in DMF (0.3 mL) was added and stirred for an additional 18 hrs at rt. The mixture was washed with EtOAc (30 mL) and then filtered and the filtrate was washed with water in a separatory funnel. The organic layer was dried with Na2SO4, filtered, and concentrated to oil. The product was purified by preparative TLC (developed with MeOH/CH2Cl2, 5:95, v:v, Rf = 0.2) to afford an oil (27 mg, 0.04 mmol, 31%); 1H NMR (CDCl3, 500 MHz) δ 7.47 (d, J = 2.0 Hz, 1 H), 7.25 (dd, J = 2.0, 8.5 Hz, 1 H), 6.87 (d, J = 8.5 Hz, 1 H), 6.86-6.81 (m, 2 H), 6.76-6.72 (m, 1 H), 5.81-5.79 (m, 1 H), 5.71 (m, 1 H), 4.01-3.97 (m, 1 H), 3.94 (s, 3 H), 3.91 (s, 3 H), 3.8 (s, 3 H), 3.79-3.74 (m, 1 H), 3.36-3.31 (m, 1 H), 3.03-3.0 (m, 1 H), 2.81-2.78 (m, 2 H), 2.73-2.37 (m, 5 H), 2.30-2.19 (m, 3 H), 2.09-1.97 (m, 3 H), 1.87-1.26 (m, 14 H); HRMS (ESI) [M+H]+ calcd for C36H49FN3O5 622.8007, found 622.8025.
The following compound was prepared in a similar manner.
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-propyl]-tetrahydro-furan-2-yl}-ethylamine)-pentyl-4a,5,8,8atetrahydro-2H-phthalazin-1-one (22)
the product was isolated by preparative TLC and (developed with MeOH/CH2Cl2, 5:95, v:v, Rf = 0.2); 1H NMR (CDCl3, 500 MHz) δ 7.47 (d, J = 2.0 Hz, 1 H), 7.25 (dd, J = 2.0, 8.5 Hz, 1 H), 6.87 (d, J = 8.5 Hz, 1 H), 6.86-6.81 (m, 2 H), 6.76-6.72 (m, 1 H), 5.8-5.77 (m, 1 H), 5.69-5.66 (m, 1 H), 4.01-3.97 (m, 1 H), 3.94 (s, 3 H), 3.91 (s, 3 H), 3.77 (s, 3 H), 3.79-3.74 (m, 1 H), 3.33-3.3 (m, 1 H), 3.0-2.96 (m, 1 H), 2.81-2.57 (m, 7 H), 2.22-2.17 (m, 3 H), 2.04-1.34 (m, 19 H); HRMS (ESI) [M+H]+ calcd for C37H51FN3O5 636.8275, found 636.826.
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-carbamic ethyl ester (23)
Into a flame dried 20 mL vial was placed K2CO3 (239 mg, 1.73 mmol, 6.0 equiv) and anhydrous THF (5.0 mL). The vial was purged with Ar(g) and then cooled in an ice bath. Ethyl chloroformate (156 mg, 1.44 mmol, 5.0 equiv) was then added via syringe followed by slow addition of 14 (77 mg, 0.29 mmol, 1.0 equiv) dissolved in THF (1.5 mL). The reaction was stirred at 0° C for 0.5 hrs and then warmed to rt and allowed to stir an additional 3 hrs. The reaction was then stopped with sat. NaHCO3(aq) and extracted with EtOAc (3 × 20 mL). The organic layer was washed with brine and dried over Na2SO4, filtered, and concentrated under reduced pressure to yield crude product that was used in the next step of the synthesis. TLC: MeOH/CH2Cl2 (2:98, v:v), Rf 0.4; 1H NMR (CDCl3, 500 MHz) 6.87-6.82 (m, 2 H), 6.74-6.71 (m, 1 H), 4.12 (q, J = 7.1 Hz, 2 H), 4.05-3.77 (m, 2 H), 3.78 (s, 3 H), 2.71-2.56 (m, 2 H), 2.08-1.51 (m, 10 H) 1.3 (t, J = 7.2 Hz, 3 H). LRMS (ESI) [M+H]+ calcd for C18H27FNO4 340 found 340.
2-{5-[3-(5-Fluoro-2-methoxy-phenyl)-ethyl]-tetrahydro-furan-2-yl}-ethylamine)-methyl-amine (24)
Into a flame dried round bottom flask, purged with Ar(g), was placed 23 (112 mg, 0.33 mmol, 1.0 equiv.) and anhydrous THF (1.6 mL). The flask was cooled with an ice bath and stirred for 15 min. LAH (1.0 M, 1.32 mL, 1.32 mmol, 4.0 equiv.) was then slowly added via a syringe over a period of 5 min. The reaction was allowed to warm to rt and stirred an additional 4 hrs. The reaction was then stopped with ice cold MeOH and then stirred for an additional 15 min at rt. The resulting solution was acidified with 1N HCl(aq) and then transferred to a beaker and then made basic with 10 M NaOH(aq). The basic solution was extracted with diethyl ether (3 × 20 mL) and then the organic layer was washed with brine. The diethyl ether extract was dried over Mg2SO4 for 15 min and then filtered and concentrated to oil under reduced pressure to afford the crude product. The oil was purified with preparative TLC (developed with MeOH/CH2Cl2, 5:95, v:v, Rf 0.05); 1H NMR (CDCl3, 500 MHz) 6.87-6.82 (m, 2 H), 6.74-6.71 (m, 1 H), 4.05-3.77 (m, 2 H), 3.78 (s, 3 H), 2.71-2.56 (m, 5 H), 2.08-1.51 (m, 10 H); HRMS (ESI) [M+H]+ calcd for C16H25FNO2 282.3784, found 282.3762.
Supplementary Material
Acknowledgments
The authors are indebted to Professor Marco Conti (Stanford University) for providing the results shown in Table 1. We also thank Dr. Aaron Janowsky and Robert Johnson (Portland VA Hospital, Portland, OR) for doing the binding and reuptake inhibition assays shown in Table 2. The work was supported in part by research grants and an Independent Scientist Award from the National Institute of Mental Health (JOD).
Footnotes
Abbreviations: SSRI, selective serotonin reuptake inhibitor; FST, forced-swim test; PDE, phosphodiesterase; cAMP, cyclic adenosine monophosphate; ICR mice, Inbred Charles River mice; 5-HT, 5-hydroxytryptamine; 5-HIAA, 5-hydroxy indole acetic acid; DA, dopamine; NE, norepinephrine; hSERT, human serotonin transporter; hNET, human norepinephrine transporter; hDAT, human dopamine transporter; DRL, differential-reinforcement-of-low-rate; ANOVA, analysis of variance; Veh, vehicle.
Supporting Information Available
Analytical data of the target compounds is provided in the Supporting Information. This information is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Warner Schmidt JL, Duman RS. Hippocampal neurogenesis: opposing effects of stress and antidepressant treatment. Hippocampus. 2006;16:239–249. doi: 10.1002/hipo.20156. [DOI] [PubMed] [Google Scholar]
- 2.Preskorn SH, Stanga CY, Ross R. Selective Serotonin Reuptake Inhibitor. In: Preskorn SH, Stanga CY, Feighner JP, Ross R, editors. Antidepressants: Past, Present, and Future. New York: Springer Publishing Co; 2004. pp. 242–264. [Google Scholar]
- 3.Wong DT, Bymaster FP, Horng JS, Molloy BB. A new selective inhibitor for uptake of serotonin into synaptosomes or rat brain: 3-(p-trifluoromethylphenoxy)-N-methyl-3-phenylpropylamine (Lilly 110140) J. Pharmacol. Exp. Ther. 1975;193:804–811. [PubMed] [Google Scholar]
- 4.Wong DT, Bymaster FP, Engleman EA. Prozac (fluoxetine, Lilly 110140), the first selective serotonin uptake inhibitor and an antidepressant drug (in US): twenty years since its first publication. Life Sci. 1995;57:411–441. doi: 10.1016/0024-3205(95)00209-o. [DOI] [PubMed] [Google Scholar]
- 5.Hirano K, Seki T, Sakai N, Kato Y, Hashimoto H, Uchida S, Yamada S. Effects of continuous administration of paroxetine on ligand binding site and expression of serotonin transporter protein in mouse brain. Brain Res. 2005;1053:54–61. doi: 10.1016/j.brainres.2005.06.038. [DOI] [PubMed] [Google Scholar]
- 6.Wong DT, Perry KW, Bymaster FP. The discovery of fluoxetine hydrochloride (Prozac) Nature Reviews Drug Disc. 2005;4:764–774. doi: 10.1038/nrd1821. [DOI] [PubMed] [Google Scholar]
- 7.Kobayashi K, Ikeda Y, Haneda E, Suzuki H. Chronic fluoxetine bidirectionally modulates potentiating effects of serotonin on the hippocampal mossy fiber synaptic transmission. J. Neurosci. 2008;28:6272–6280. doi: 10.1523/JNEUROSCI.1656-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ye Y, Jackson K, O’Donnell JM. Effects of repeated antidepressant treatment of type 4A phosphodiesterase (PDE4A) in rat brain. J. Neurochem. 2000;74:1257–1262. doi: 10.1046/j.1471-4159.2000.741257.x. [DOI] [PubMed] [Google Scholar]
- 9.Halene TB, Siegel SJ. PDE inhibitors in psychiatry-future options for dementia, depression and schizophrenia? Drug Discov. Today. 2007;12:870–878. doi: 10.1016/j.drudis.2007.07.023. [DOI] [PubMed] [Google Scholar]
- 10.Houslay MD, Shafer P, Zhang KY. Keynote review: phosphodiesterase-4 as a therapeutic target. Drug Discov. Today. 2005;10:1503–1519. doi: 10.1016/S1359-6446(05)03622-6. [DOI] [PubMed] [Google Scholar]
- 11.Ye Y, O'Donnell JM. Diminished noradrenergic activity in the brain down-regulates Type 4 phosphodiesterase in rat cerebral cortex. J. Neurochem. 1996;66:1894–1902. doi: 10.1046/j.1471-4159.1996.66051894.x. [DOI] [PubMed] [Google Scholar]
- 12.Wachtel H, Schneider HH. Rolipram, a novel antidepressant drug, reverses the hypothermia and hypokinesia of monoamine-depleted mice by an action beyond postsynaptic monoamine receptors. Neuropharmacology. 1986;25:1119–1126. doi: 10.1016/0028-3908(86)90159-0. [DOI] [PubMed] [Google Scholar]
- 13.Zhang HT, Huang Y, Jin SLC, Frith SA, Suvarna N, Conti M, O'Donnell JM. Antidepressant-like profile and reduced sensitivity to rolipram in mice deficient in the PDE4D phosphodiesterase enzyme. Neuropsychopharmacology. 2002;27:587–595. doi: 10.1016/S0893-133X(02)00344-5. [DOI] [PubMed] [Google Scholar]
- 14.O'Donnell JM, Frith S. Behavioral effects of family-selective inhibitors of cyclic nucleotide phosphodiesterases. Pharmacol. Biochem. Behav. 1999;63:185–192. doi: 10.1016/s0091-3057(98)00267-6. [DOI] [PubMed] [Google Scholar]
- 15.Itoh T, Tokumura M, Abe K. Effects of rolipram, a phosphodiesterase 4 inhibitor, in combination with imipramine on depressive behavior, CRE-binding activity and BDNF level in learned helplessness rats. Eur. J. Pharmacol. 2004;498:135–142. doi: 10.1016/j.ejphar.2004.07.084. [DOI] [PubMed] [Google Scholar]
- 16.Lourenco CM, Kenk M, Beanlands RS, DaSilva JN. Increasing synaptic noradrenaline, serotonin and histamine enhances in vivo binding of phosphodiesterase-4 inhibitor (R)-[11C]rolipram in rat brain, lung and heart. Life Sci. 2006;79:356–364. doi: 10.1016/j.lfs.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 17.Van der Mey M, Hatzelmann A, Van Klink GP, Van der Laan IJ, Sterk GJ, Thibaut U, Ulrich WR, Timmerman H. Novel selective PDE4 inhibitors. 2. Synthesis and structure-activity relationships of 4-aryl-substituted cis-tetra- and cis-hexahydrophthalazinones. J Med Chem. 2001;44:2523–2535. doi: 10.1021/jm010838c. [DOI] [PubMed] [Google Scholar]
- 18.Van der Mey M, Boss H, Couwenberg D, Hatzelmann A, Sterk GJ, Goubitz K, Schenk H, Timmerman H. Novel selective phosphodiesterase (PDE4) inhibitors. 4. Resolution, absolute configuration and PDE4 inhibitory activity of cis-tetra- and cis-hexahydrophthalazinones. J. Med. Chem. 2002;45:2526–2533. doi: 10.1021/jm0110338. [DOI] [PubMed] [Google Scholar]
- 19.Carroll FI, Runyon SP, Abraham P, Navarro H, Kuhar MJ, Pollard GT, Howard JL. Monoamine transporter binding, locomotor activity, and drug discrimination properties of 3-(4-substituted-phenyl)tropane-2-carboxylic acid methyl ester isomers. J Med. Chem. 2004;47:6401–6409. doi: 10.1021/jm0401311. [DOI] [PubMed] [Google Scholar]
- 20.Eshleman AJ, Carmolli M, Cumbay M, Neve KA, Janowsky A. Characteristics of drug interactions with recombinant biogenic amine transporters expressed in the same cell type. J. Pharmacol. Exper. Ther. 1999;289:877–885. [PubMed] [Google Scholar]
- 21.Feng X, Fandrick K, Johnson R, Janowsky A, Cashman JR. Synthesis of 3-carbomethoxy-4-(aryl)piperidines and in vitro and in vivo pharmacological evaluation: Identification of inhibitors of the human dopamine transporter. Bioorg. Med. Chem. 2003;11:775–780. doi: 10.1016/s0968-0896(02)00528-x. [DOI] [PubMed] [Google Scholar]
- 22.Lucki I, Dalvi A, Mayorga AJ. Sensitivity to the effects of pharmacologically selective antidepressants in different strains of mice. Psychopharmacology. 2001;155:315–322. doi: 10.1007/s002130100694. [DOI] [PubMed] [Google Scholar]
- 23.Lucki I. The forced swim test as a model for core and component behavioral effects of antidepressant drugs. Behav. Pharmacol. 1997;8:523–532. doi: 10.1097/00008877-199711000-00010. [DOI] [PubMed] [Google Scholar]
- 24.Eshleman AJ, Neve RL, Janowsky A, Neve KA. Characterization of a recombinant human dopamine transporter in multiple cell lines. J. Pharmacol. Exper. Ther. 1995;274:276–283. [PubMed] [Google Scholar]
- 25.Hansen G, Jin S, Umetsu DT, Conti M. Absence of muscarinic cholinergic airway responses in mice deficient in the cyclic nucleotide phosphodiesterase PDE4D. Proc. Natl. Acad. Sci. U.S.A. 2000;97:6751–6756. doi: 10.1073/pnas.97.12.6751. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.