Abstract
We showed recently that IL-4 causes mitochondrial dysfunction in allergic asthma. IL-4 is also known to induce 12/15-lipoxygenase (12/15-LOX), a potent candidate molecule in asthma. Because vitamin E (Vit-E) reduces IL-4 and inhibits 12/15-LOX in vitro, here we tested the hypothesis that Vit-E may be effective in restoring key mitochondrial dysfunctions, thus alleviating asthma features in an experimental allergic murine model. Ovalbumin (OVA)-sensitized and challenged male BALB/c mice showed the characteristic features of asthma such as airway hyperresponsiveness (AHR), airway inflammation, and airway remodeling. In addition, these mice showed increase in the expression and metabolites of 12/15-LOX, reduction in the activity and expression of the third subunit of mitochondrial cytochrome-c oxidase, and increased cytochrome c in lung cytosol, which indicate that OVA sensitization and challenge causes mitochondrial dysfunction. Vit-E was administered orally to these mice, and 12/15-LOX expression, key mitochondrial functions, ultrastructural changes of mitochondria in bronchial epithelia, and asthmatic parameters were determined. Vit-E treatment reduced AHR, Th2 response including IL-4, IL-5, IL-13, and OVA-specific IgE, eotaxin, transforming growth factor-β1, airway inflammation, expression and metabolites of 12/15-LOX in lung cytosol, lipid peroxidation, and nitric oxide metabolites in the lung, restored the activity and expression of the third subunit of cytochrome-c oxidase in lung mitochondria and bronchial epithelia, respectively, reduced the appearance of cytochrome c in lung cytosol, and also restored mitochondrial ultrastructural changes of bronchial epithelia. In summary, these findings show that Vit-E reduces key mitochondrial dysfunctions and alleviates asthmatic features.
Keywords: 12/15-lipoxygenase, mitochondria
the deficiency of endogenous antioxidant defenses has been reported in asthma (37). In fact, childhood asthma is described as the state of fat-soluble vitamin deficiency (36). Furthermore, maternal diet intake, especially vitamin E (Vit-E), during pregnancy is consistently associated with the decreased frequency of childhood asthma (7, 33). Among eight known natural Vit-E analogs, only the α-tocopherol form has biological significance (40). Vit-E intake has been shown to be inversely associated with both allergen skin sensitization and total serum IgE levels. Although Vit-E has been shown to reduce airway inflammation and airway hyperresponsiveness (AHR) in animal models including mice (14, 24), it was not successful in clinical trials (22). This could be due to species difference in the metabolism of chylomicron and apolipoproteins, which are essential for Vit-E absorption (8). Evidently, Vit-E levels are different in mice and humans (5). In any event, the exact reason for this discrepancy is not yet clear. Therefore, exploring the mechanism of the antiasthma activity of Vit-E could be helpful for its efficient usage in humans.
Vit-E and its derivatives combat diseases associated with mitochondrion-derived oxidative stress (38) and increase the life span of mice by decreasing the degeneration of mitochondria (23). Interestingly, orally administered α-tocopherol predominantly localizes in mitochondrial fractions (2), and α-tocopherol is crucial in the maintenance of mitochondrial integrity (15). In addition, it was suggested previously that the state of 12/15-lipoxygenase (12/15-LOX) depletion and Vit-E administration share common pathways (39), and, importantly, Vit-E complexes with 12/15-LOX to inhibit its activity in in vitro studies (9). Increased Th2 cytokines, especially IL-4 and IL-13, upregulate murine 12/15-LOX (12), which is involved in asthma pathogenesis (10). Importantly, 15-LOX-1, a human counterpart of murine 12/15-LOX, induces airway epithelial apoptosis, which further activates epithelial mesenchymal trophic unit to initiate airway remodeling (13). 12/15-LOX is predominantly expressed in bronchial epithelial cells and degrades mitochondria (32). Recent evidence suggests that mitochondrial events are essential during antigen presentation, a crucial initiating step in asthma pathogenesis (6, 27). In addition, we recently showed (18) that mitochondrial dysfunction could be crucial in asthma. With this view, we hypothesized that Vit-E could have a role in the reduction of mitochondrial dysfunction in asthma. Hence, we selected α-tocopherol and evaluated its effect on mitochondrial dysfunction, using a mouse model of asthma.
MATERIALS AND METHODS
This study was conducted in conformity with the American Physiological Society's “Guiding Principles in the Care and Use of Animals.” Experimental protocols and study design were approved by the designated institutional review boards at the Institute of Genomics and Integrative Biology.
Animals.
Male BALB/c mice (8–10 wk old, weighing 18–20 g) were obtained from the National Institute of Nutrition (Hyderabad, India) and acclimatized for 1 wk under standard laboratory conditions (25 ± 2°C, 55% humidity) before the experiments were started.
Development of mouse model of asthma.
After acclimatization, each mouse was sensitized by intraperitoneal injections of 200 μl of PBS suspension containing 50 μg of chicken egg ovalbumin (OVA; grade V, Sigma) and 4 mg of alum (aluminum hydroxide, Sigma) on days 0, 7, and 14 as described previously (1, 18). From day 21 to day 32, mice were exposed to 3% OVA aerosols for 30 min/day in a Plexiglas chamber (20 × 20 × 10 cm3). The aerosol was generated by a nebulizer (Omron CX3) with an airflow rate of 9 l/min. Each control (Sham) mouse received intraperitoneal injections of 200 μl of PBS containing 4 mg of alum and was challenged with PBS alone.
Vit-E treatment of OVA-sensitized and challenged mice.
There were two experimental sets: 1) dose-titration experiments with different concentrations of Vit-E and 2) verification experiments. For dose-titration experiments, mice were divided into six groups after acclimatization; each group (n = 5) was named according to sensitization/challenge/treatment: Sham/PBS/Veh [PBS sensitized, PBS challenged, and vehicle (Veh) treated], OVA/OVA/Veh (OVA sensitized, OVA challenged, and vehicle treated), and OVA/OVA/Vit-E 5, 10, 15, and 20 IU/kg (vitamin E in the form of α-tocopherol, Sigma). In the verification experiments, there were three groups, Sham/PBS/Veh, OVA/OVA/Veh, and OVA/OVA/Vit-E (15 IU/kg Vit-E), and each group consisted of six mice. Vehicle or Vit-E was administered orally twice daily from day 19 to day 32 in a volume of 10 μl per dose. Ethanol (50%) was used as the vehicle in which Vit-E was dissolved.
Measurement of airway responsiveness to methacholine.
These measurements were estimated for all mice on day 33 in unrestrained and restrained conscious mice by single-chamber (SCP) and double-chamber (DCP) body plethysmography, respectively (models PLY 3211 and PLY 3351, Buxco Electronics), as described previously with minor modification, especially in methacholine (MCh) volume (18). Enhanced pause (Penh) was estimated by SCP and specific airway conductance (sGaw) and specific airway resistance (sRaw) were estimated by DCP. Final results for SCP were expressed as either partial concentration of methacholine required to increase baseline Penh to 200% (MCh PC200 Penh) or as percent baseline Penh with each concentration of MCh, with baseline Penh (with PBS aerosol) considered as 100%. Final results for DCP were expressed as percent baseline sGaw and sRaw.
Transmission electron microscopy.
Whole body perfusion fixation was performed as described previously (18, 19). First-generation bronchi were located with a dissection microscope (SZX-12, Olympus) and processed to view under the transmission electron microscope. Approximately 50 bronchial epithelial cells were randomly viewed, and representative images are shown.
Euthanasia and histopathology of lung.
After AHR was determined, mice were euthanized; blood was collected and processed to separate sera (18, 28, 29). For lung histology, a portion of the harvested lung was fixed in 10% buffered formalin (28, 29). The fixed, paraffin-embedded tissues were sectioned into 4-μm sections and stained with hematoxylin and eosin, periodic acid-Schiff (PAS), and Masson trichrome (MT) to assess airway inflammation, goblet cell metaplasia, and subepithelial fibrosis, respectively. To determine airway mucin content and collagen deposition, quantitative morphometry was performed in PAS- and MT-stained lung sections as described previously (18, 19).
Isolation of whole lung mitochondria and cytosolic separation.
After euthanasia, the lung portion below the trachea was removed and washed three times to isolate mitochondrial and cytosolic fractions (Cyto) as described previously (18) with a mitochondria isolation kit (MITO-ISO1, Sigma). Protein estimation was done for both mitochondrial fractions and Cyto by bicinchoninic acid (Sigma) assay.
Measurement of OVA-specific immunoglobulins.
These measurements were performed as described previously (18) with minor modifications. Briefly, each well of the microplate was coated with 100 μl of OVA (2 μg), sera were added, bound IgE or IgG were detected with biotinylated anti-mouse IgE or anti-mouse IgG2a (BD Pharmingen), streptavidin-horseradish peroxidase (HRP) conjugates (BD Pharmingen) were added, bound enzymes were detected by addition of a tetramethylbenzidine substrate system (BD Pharmingen), and finally absorbances were read at 450 nm. Absorbances were converted to arbitrary units.
Measurements of IL-4, IL-5, IL-13, eotaxin, and transforming growth factor-β1 in lung homogenates.
Lung tissue homogenates (prepared by homogenization of 50 mg of tissue with 500 μl of PBS and centrifugation at 10,000 g for 30 min) in duplicate were used for sandwich ELISA [BD Biosciences for IL-4, IL-5, and transforming growth factor-β1 (TGF-β1) and R&D Systems for IL-13 and eotaxin]. Measurements were performed as per manufacturers' instructions. Results are expressed in picograms and normalized by protein concentrations.
12/15-LOX Western blot.
Cyto proteins were separated on 8% SDS-PAGE and transferred onto polyvinylidene difluoride (PVDF) membrane (Millipore), and the membrane was blocked with blocking buffer (3% skim milk), incubated with antibody that cross-reacts with 12- and 15-LOX forms (Santa Cruz Biotechnology), and detected with HRP-conjugated anti-rabbit secondary antibody and diaminobenzidine (DAB)-H2O2. α-Tubulin was used as a loading control.
13-(S)-hydroxyoctadecaenoic acid and 12-(S)-hydroxyeicosatetraenoic acid measurement in cytosolic fractions.
Fifty micrograms of Cyto per well in duplicate was used for ELISA of 13-(S)-hydroxyoctadecaenoic acid (HODE) and 12-(S)-hydroxyeicosatetraenoic acid (HETE) (Assay Designs). Measurements were performed as per manufacturer's instructions without further extraction. Results are expressed in nanograms per 50 μg of protein.
Measurements of lipid peroxidation, nitric oxide, and peroxynitrite levels in lung.
8-Isoprostane, nitric oxide (NO) metabolites (nitrates/nitrites), and nitrotyrosine (Cayman) were measured in lung homogenates by competitive ELISA except for nitrates/nitrites (calorimetric assay). The results are expressed in picograms, micromolar, and nanomoles, respectively, and normalized by protein concentrations.
Total cytochrome-c oxidase activity measurement.
These measurements were performed as described previously (18). The activity of cytochrome-c oxidase of electron transport chain (COXETC) is based on the decrease in absorbance of ferrocytochrome c due to its oxidation to ferricytochrome c by COXETC present in 10 μg of mitochondrial fraction in duplicate lysed with n-dodecyl-β-d-maltoside or recombinant enzyme. Net citrate synthase activities were estimated with a 96-well microplate reader (Bio-Rad) as per manufacturer's instructions (Sigma Chemical). The ratio of total COXETC to net citrate synthase activities is reported.
Immunohistochemistry.
This was performed as described previously (18) with commercial goat polyclonal antibodies for cytochrome-c oxidase III (Santa Cruz Biotechnology) as primary antibody and HRP-conjugated anti-goat IgG as secondary antibody (18). Negative control experiments were performed either by using γ-globulin as isotype control (Jackson Immunoresearch Laboratories) or by omission of primary antibody.
Measurement of cytochrome c in cytosol.
The levels of cytochrome c were measured in cytosolic fractions by ELISA as per manufacturer's instructions (R&D Systems). Briefly, 5 μg of Cyto protein per well in duplicate was used to measure cytochrome c with a rat/mouse cytochrome c enzyme immunoassay (EIA) kit.
Statistics.
Data are expressed as means ± SE. Since we conducted hypothesis-driven comparisons between selected groups, we used unpaired Student's t-test and statistical significance was set at P ≤ 0.05.
RESULTS
Vitamin E reduces airway hyperresponsiveness to methacholine.
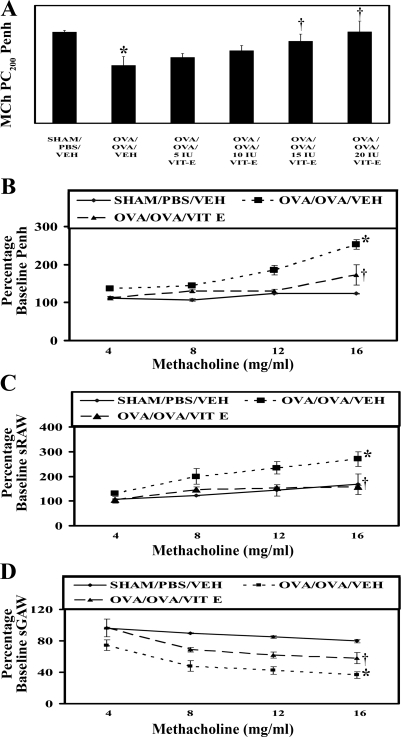
In dose-titration experiments, Vit-E reduced AHR to MCh as shown in Fig. 1A. The optimal effective dose was found to be 15 IU/kg, so further experiments were performed with this dose unless otherwise specified. In verification experiments, OVA/OVA/Veh mice showed a dose-dependent increase in Penh and sRaw compared with Sham/PBS/Veh mice with MCh (Fig. 1, B and C). On the other hand, OVA/OVA/Veh mice showed a dose-dependent decrease in sGaw compared with Sham/PBS/Veh mice with MCh (Fig. 1D). These results indicate that the OVA/OVA/Veh mice had developed significant AHR. However, Vit-E treatment attenuated the increase of Penh and sRaw and decrease of sGaw (Fig. 1, B–D).
Fig. 1.
Vitamin E (Vit-E) treatment reduced airway hyperresponsiveness in asthmatic mice. A: in dose-titration experiments, partial concentration of methacholine required to increase baseline enhanced pause (Penh) to 200% (MCh PC200 Penh) was determined in 6 groups of mice: Sham/PBS/Veh [PBS sensitized, PBS challenged, and orally administered 50% ethanol (vehicle, Veh)], OVA/OVA/Veh [ovalbumin (OVA) sensitized, OVA challenged, and orally administered vehicle], and OVA/OVA/Vit-E 5 IU, 10 IU, 15 IU, and 20 IU (OVA sensitized, OVA challenged, and orally administered 5, 10, 15 and 20 IU/kg of Vit-E). In verification experiments, % baseline Penh (B), specific airway resistance (sRaw; C), and specific airway conductance (sGaw; D), indicators of bronchoconstriction, were determined in 3 groups of mice: Sham/PBS/Veh, OVA/OVA/Veh, and OVA/OVA/Vit-E (15 IU/kg Vit-E). Each mouse was challenged with increasing concentrations of MCh (0–16 mg/ml), and final results for single-chamber plethysmography are expressed either in MCh PC200 Penh or in % baseline Penh; double-chamber plethysmography results are expressed in % baseline sGaw and sRaw, respectively, while the PBS aerosol values are considered as baseline. Values are means ± SE of 3 independent experiments. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh.
Vitamin E reduces airway inflammation.
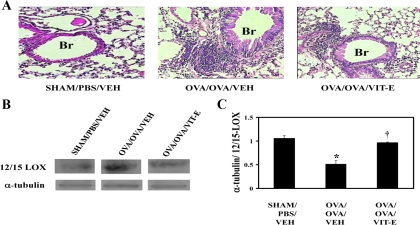
To assess the effects of Vit-E on airway inflammation, hematoxylin and eosin staining was performed on lung sections. The lungs of Sham control mice showed normal lung structure (Fig. 2A). In contrast, the lungs of OVA/OVA/Veh mice showed a dense infiltration of inflammatory cells in perivascular and peribronchial regions. However, Vit-E treatment showed a significant reduction of inflammation both in perivascular and peribronchial regions (Fig. 2A). Inflammation scoring also confirmed this (data not shown).
Fig. 2.
Vit-E treatment reduced airway inflammation and expression of 12/15 lipoxygenase (12/15-LOX). A: to determine the effect of Vit-E on airway inflammation, hematoxylin and eosin staining was performed in lung tissue sections. Representative photographs are shown; all photographs are at ×10 magnification. Br, bronchus. B: Western blot for 12/15-LOX was performed in lung cytosols and compared with α-tubulin. C: signals were estimated with spot densitometry. Results shown are representative of at least 3 independent experiments. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh.
Vitamin E reduces Th2 cytokines and eotaxin in lung and OVA-specific IgE in sera.
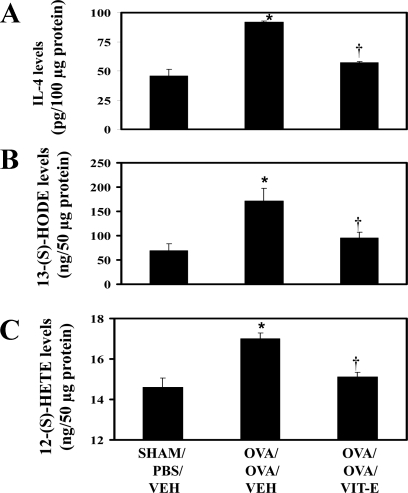
To examine the effect of Vit-E on IL-4, IL-5, IL-13, eotaxin, and OVA-specific IgE and IgG2a, they were measured in lung tissue homogenates and sera as described in materials and methods. As shown in Fig. 3A and Table 1, the levels of IL-4, IL-5, IL-13, eotaxin, and OVA-specific IgE were found to be markedly increased in OVA/OVA/Veh compared with Sham/PBS/Veh mice. However, treatment of asthmatic mice with Vit-E (OVA/OVA/Vit-E) significantly reduced these levels (Fig. 3A and Table 1). On the other hand, there was no difference in the levels of IgG2a between normal control and asthmatic control mice. However, it was observed that Vit-E treatment significantly increased IgG2a levels compared with either normal or asthmatic control mice (Table 1).
Fig. 3.
Vit-E treatment reduced IL-4 and metabolites of 12/15-LOX. IL- 4 (in lung homogenates; A), 13-(S)-hydroxyoctadecaenoic acid (HODE) (B), and 12-(S)-hydroxyeicosatetraenoic acid (HETE) (C) (lung cytosols) were estimated. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh.
Table 1.
Effects of vitamin E on IL-5, IL-13, OVA-specific IgE, IgG2a, and TGF-β1 levels
| IL-5, pg/100 μg protein | IL-13, pg/100 μg protein | OVA-Specific IgE, AU | OVA-Specific IgG2a, AU | TGF-β1, pg/50 μg protein | Eotaxin, pg/25 μg protein | |
|---|---|---|---|---|---|---|
| Sham/PBS/Veh | 9.0±7.9 | 17.9±1.1 | 0.7±0.08 | 1.4±0.2 | 362.9±40.6 | 15.9±1.4 |
| OVA/OVA/Veh | 66.2±7.7* | 41.5±6.3* | 2.2±0.18* | 1.4±0.1* | 511.4±8.8* | 44.0±0.7* |
| OVA/OVA/Vit-E | 22.9±1.1† | 21.7±4.8† | 1.3±0.18† | 4.1±1.5† | 432.8±19.1† | 31.0±0.6† |
Data are means ± SE of 3 independent experiments. Vit-E, vitamin E; Sham, sham-treated (control); Veh, vehicle; OVA, ovalbumin; TGF-β1, transforming growth factor-β1; AU, arbitrary unit.
P < 0.05 vs. Sham/PBS/Veh group;
P < 0.05 vs. OVA/OVA/Veh group.
Vitamin E reduces expression of 12/15-LOX and levels of 13-(S)-HODE and 12-(S)-HETE.
To determine the effect of Vit-E on the expression of 12/15-LOX and its metabolites, Western blot analysis and ELISA were performed in Cyto. 12/15-LOX expression (Fig. 2, B and C) and its metabolites (Fig. 3, B and C) were increased in OVA/OVA/Veh compared with Sham/PBS/Veh mice. However, treatment of asthmatic mice with Vit-E showed a significant reduction of 12/15-LOX expression and its metabolites (Fig. 2, B and C, and Fig. 3, B and C).
Vitamin E reduces oxidative stress in lung.
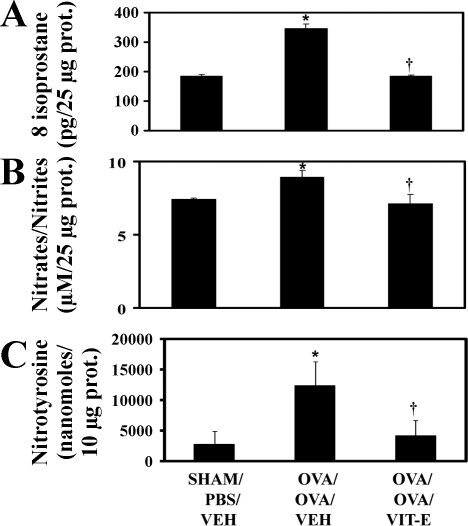
Since 12/15-LOX leads to lipid peroxidation, the levels of 8-isoprostane, a reliable marker of lipid peroxidation (37), were estimated in lung homogenates. As shown in Fig. 4A, the levels of 8-isoprostane were found to be significantly increased in OVA/OVA/Veh mice compared with Sham/PBS/Veh mice. However, Vit-E treatment significantly reduced the levels of 8-isoprostane.
Fig. 4.
Vit-E treatment reduced nitrooxidative stress in lung. After euthanasia of mice, 8-isoprostane (A), nitrates/nitrites (B), and nitrotyrosine (C) levels were measured in lung homogenates. Data are means ± SE of 3 independent experiments. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh.
Vitamin E reduces nitrative stress in lung.
Since NO is crucial in the development of mitochondrial dysfunction by inhibiting cytochrome-c oxidase activity (18), final metabolites of NO such as nitrates/nitrites were measured. As shown in Fig. 4B, the levels of nitrates/nitrites were found to be significantly increased in OVA/OVA/Veh mice compared with Sham/PBS/Veh mice. However, Vit-E treatment significantly reduced these levels. In addition, Vit-E at higher doses was found to reduce the levels of nitrates/nitrites in dose-titration experiments (Table 2).
Table 2.
Effects of vitamin E on nitrates/nitrites, cytochrome-c oxidase activity and cytosolic cytochrome c in dose-titration experiments
| Nitrates/nitrites, μM/25 μg protein | COXETC/Citrate Synthase | Cytochrome c in cytosol, ng/5 μg protein | |
|---|---|---|---|
| Sham/PBS/Veh | 7.4±0.1 | 11.3±0.8 | 0.9±0.08 |
| OVA/OVA/Veh | 9.2±0.6* | 3.8±0.8* | 1.8±0.07* |
| OVA/OVA/Vit-E 5 IU | 9.4±0.1 | 6.8±1.3 | 1.9±0.16 |
| OVA/OVA/Vit-E 10 IU | 6.7±0.1 | 7.5±1.6 | 2.2±0.38 |
| OVA/OVA/Vit-E 15 IU | 6.8±0.2† | 11.2±0.9† | 1.0±0.26† |
| OVA/OVA/Vit-E 20 IU | 6.8±0.6† | 12.5±1.3† | 1.3±0.18† |
Data are means ± SE. COXETC, cytochrome-c oxidase of electron transport chain.
P < 0.05 vs. Sham/PBS/Veh group;
P < 0.05 vs. OVA/OVA/Veh group.
Since peroxynitrite is also known to cause mitochondrial dysfunction by degrading heme centers of cytochrome-c oxidase (18), we determined it in lung homogenates. As shown in Fig. 4C, the levels of nitrotyrosine, a marker of peroxynitrite, were found to be significantly increased in OVA/OVA/Veh mice compared with Sham/PBS/Veh mice. However, Vit-E treatment significantly reduced these levels.
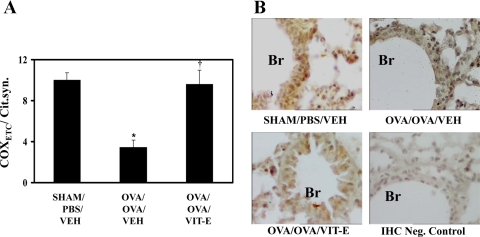
Vitamin E increases activity of COXETC.
Since lipid peroxidation and NO lead to reduction in the activity of cytochrome-c oxidase (18), we measured COXETC in lung mitochondria and normalized by the respective citrate synthase activity. The results indicated that COXETC/citrate synthase was decreased in OVA/OVA/Veh mice compared with Sham/PBS/Veh (Fig. 5A). Interestingly, Vit-E treatment significantly restored COXETC activity. It is also important to note that there was a dose-dependent restoration of COXETC activity in dose-titration experiments (Table 2).
Fig. 5.
Vit-E treatment restored activity of cytochrome-c oxidase of electron transport chain (COXETC) and expression of subunit III of COXETC. A: after euthanasia, mitochondria were isolated from fresh lung tissue; COXETC activity was estimated and normalized by respective citrate synthase activity. Data are means ± SE of 3 independent experiments. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh. B: immunohistochemistry (IHC) was performed for subunit III of COXETC in lung tissue sections. Brown color indicates positive expression of respective subunit. Br, bronchus. Primary antibody against subunit III of COXETC was omitted in IHC negative control. Representative photomicrographs from 3 independent experiments are shown at ×40 magnification.
Vitamin E restores reduction in expression of COXETC subunit III.
Since subunit III is critical in the catalytic activity of COXETC (18), its expression was determined by immunohistochemistry. COXETC subunit III was found to be predominantly expressed in bronchi of Sham/PBS/Veh mice compared with other pulmonary structures such as alveoli and vascular endothelia, and it was found to be significantly decreased in OVA/OVA/Veh mice (Fig. 5B). Interestingly, Vit-E significantly restored its expression (Fig. 5B).
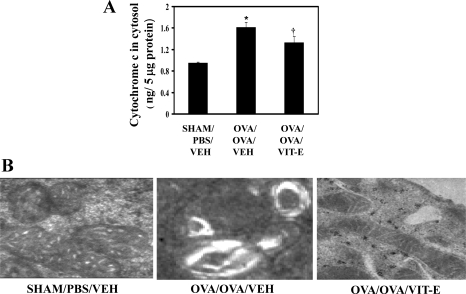
Vitamin E reduces cytochrome c levels in cytosol.
Because there was a reduction in COXETC activity in asthmatic mice lung, we further analyzed the cytochrome c levels by ELISA. As shown in Fig. 6, OVA/OVA/Veh mice showed increased levels of cytochrome c in the cytosol compared with Sham/PBS/Veh mice. However, OVA/OVA/Vit-E mice showed a significant reduction of cytochrome c in the cytosol. In addition, a similar reduction was found with higher doses of Vit-E in dose-titration experiments (Table 2).
Fig. 6.
Vit-E treatment reduced levels of cytochrome c in lung cytosol and restored mitochondrial ultrastructural changes in bronchial epithelia. A: cytochrome c levels were measured in cytosolic fractions by ELISA. Data are means ± SE of 3 independent experiments. *P < 0.05 vs. Sham/PBS/Veh; †P < 0.05 vs. OVA/OVA/Veh. B: transmission electron microscopy was performed with first-generation bronchi to determine the ultrastructural changes of mitochondria with Vit-E treatment. Representative images are shown from each group.
Vitamin E restores ultrastructural changes of mitochondria.
To determine the mitochondrial morphological changes in bronchial epithelia, transmission electron microscopy was performed. As shown in Fig. 6B, OVA/OVA/Veh mice showed features of mitochondrial damage such as loss or disruption of cristae and swelling. However, these ultrastructural changes were restored by Vit-E treatment.
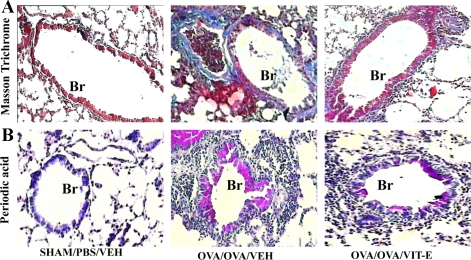
Vitamin E reduces subepithelial fibrosis and TGF-β1 levels.
Because mitochondrial dysfunction is associated with fibrosis (20, 35) and subepithelial fibrosis is one of the critical components of airway remodeling, MT staining was performed. As shown in Fig. 7A, there was no collagen in lung sections of normal control mice. In contrast, dense accumulation of collagen was found in subepithelial regions of bronchi and also around vascular regions. However, Vit-E-treated asthmatic mice showed a significant reduction in collagen deposition in the bronchovascular regions compared with control asthmatic mice (Fig. 7A). Quantitative morphometry also confirmed this [subepithelial collagen content in nl/mm2 (means ± SE): 3.9 ± 0.2, 12.4 ± 1.0, and 5.9 ± 1.1 for Sham control, OVA control, and Vit-E treated, respectively; P = 0.0002 for Sham control vs. OVA control and P = 0.005 for OVA control vs. Vit-E treated]. Since TGF-β1 is crucial in the development of subepithelial fibrosis, we measured it in lung homogenates. As shown in Table 1, Vit-E treatment reduced TGF-β1 levels compared with OVA/OVA/Veh mice.
Fig. 7.
Vit-E treatment reduced subepithelial fibrosis and goblet cell metaplasia. To determine the effect of Vit-E on subepithelial fibrosis and goblet cell metaplasia, Masson trichrome (A) and periodic acid-Schiff (B) staining were performed in lung tissue sections. All photographs are at ×10 magnification. Br, bronchus.
Vitamin E reduces goblet cell metaplasia.
Since oxidative stress is involved in goblet cell metaplasia, PAS staining was performed to determine the effect of Vit-E on this feature. As shown in Fig. 7B, control asthmatic mice showed marked goblet cell metaplasia. Interestingly, Vit-E treatment of asthmatic mice significantly reduced the goblet cell metaplasia (Fig. 7B). This was also confirmed by quantitative morphometry [airway mucin content in nl/mm2 (means ± SE): 1.6 ± 0.2, 14.3 ± 1.6, and 7.3 ± 0.6 for Sham control, OVA control, and Vit-E treated, respectively; P = 0.0003 for Sham control vs. OVA control and P = 0.007 for OVA control vs. Vit-E treated].
DISCUSSION
Although Vit-E is shown to have antiasthmatic properties, there is no evidence to suggest its role in the restoration of mitochondrial dysfunction in an in vivo model of asthma. In this study, we have demonstrated for the first time, using a mouse model of allergic asthma, that Vit-E treatment restores key mitochondrial functions, reduces 12/15-LOX expression, and reduces the features of asthma such as AHR, airway inflammation, and airway remodeling changes. Thus the restoration of mitochondrial dysfunction could be one of the ways for Vit-E to mediate its antiasthmatic activity.
12/15-LOX, inducible by IL-4, plays a critical role in various chronic inflammatory diseases such as atherosclerosis and diabetes (3, 11). It preferentially metabolizes linoleic acid to produce HODE and also metabolizes arachidonic acid to produce HETE by stereospecific incorporation of molecular oxygen into respective fatty acids. It was recently demonstrated that the deficiency of 12/15-LOX alleviates the features of asthma (10). In this study, Vit-E not only reduced IL-4 but also reduced the expression and the metabolites of 12/15-LOX. Interestingly, Vit-E is known to inhibit IL-4 transcription by suppressing the binding of NF-κB and Sp-1 with IL-4 promoter binding sites (17). In addition, Vit-E also reduced OVA-specific IgE. The increase of OVA-specific IgG2a by Vit-E indicates the possible reduction of IFN-γ since IFN-γ induces the production of IgG2a.
Vit-E treatment also reduced other components of Th2 response such as IL-5, IL-13, and eotaxin, which are crucial in the recruitment of inflammatory cells including eosinophils. Recruited inflammatory cells release oxygen and nitrogen free radicals to produce a local oxidative/nitrosative milieu in the airway. At early stages of the disease, antioxidant systems neutralize the oxidative insults to avoid the development of oxidative stress. However, repeated allergen exposures lead to reduction of both enzymatic and nonenzymatic components of the antioxidant defense system including α-tocopherol (31) to cause oxidative stress in the airway. 12/15-LOX, a prooxidant enzyme, has a unique property since it oxidizes biomembranes including mitochondrial membranes, unlike other mammalian lipoxygenases, and the resultant free radicals further initiate other deleterious oxidative processes (3). Also, under hypoxic conditions this enzyme shows hydroperoxidase activity that produces hydro-peroxy phospholipids to cause deleterious effects on mitochondria (32).
Lipid peroxidation and NO are known to inhibit the activity of cytochrome-c oxidase (21, 25). In addition, peroxynitrite, a combined product of superoxide and nitrative free radicals, irreversibly inhibits COXETC by degrading its heme centers (18). The reduction of COXETC activity has been observed in many inflammatory oxidative diseases such as Alzheimer disease and the initial stages of diabetes mellitus (18) and also in lymphocytes isolated from tobacco smokers (18). It is also important to note that oxidative insults specifically inhibit the third subunit of cytochrome-c oxidase (18). In this study, Vit-E administration decreased lipid peroxidation and NO metabolites along with the restoration of cytochrome-c oxidase activity. It is also important to note that Vit-E restores cytochrome-c oxidase activity in a dose-dependent manner. This restoration was almost parallel with the reduction of nitrates/nitrites levels by Vit-E, at least with 15 IU/kg and 20 IU/kg doses. Furthermore, Vit-E restored the expression of subunit III of COXETC in bronchial epithelial cells and reduced the levels of cytochrome c in lung cytosol. It should be noted that the expression of subunit III of COXETC with Vit-E treatment, although not completely restored, might be optimal for its enzymatic activity. Similar reduction in cytochrome c release has been shown in myocytes of myocardial infarction after combined treatment with vitamins C and E (26). In addition, Vit-E restores ultrastructural changes of mitochondria in bronchial epithelia. However, whether Vit-E has any effect on bronchial smooth muscle cells remains to be investigated.
Bronchial epithelial injury activates epithelial mesenchymal trophic unit to initiate airway remodeling changes. Interestingly, oxygen free radicals alone are able to activate epidermal growth factor receptor to develop goblet cell metaplasia (4). In this study, we have demonstrated that Vit-E reduced both goblet cell metaplasia and subepithelial fibrosis and also reduced the levels of TGF-β1. Since Vit-E is not a specific inhibitor of IL-4 or 12/15-LOX, it is not clear whether the observed effects of Vit-E on the asthmatic lung are primary or secondary to any systemic steps in asthma pathogenesis. However, we recently showed (19) that esculetin, a potent inhibitor of 12/15-LOX, restores mitochondrial dysfunction and ultrastructural changes along with the reduction of airway inflammation and AHR. Similarly, Vit-E restores mitochondrial function and alleviates airway inflammation in this study. Thus it is likely that mechanistically Vit-E may act through the 12/15-LOX and mitochondrial pathway. On the other hand, Vit-E is also known to inhibit 5-LOX, LTC-4 synthase, and PLA2 enzymes, which play important roles in asthma pathogenesis (16, 30, 34). Therefore, the contribution of inhibition of these enzymatic pathways by Vit-E toward its observed antiasthmatic activity remains to be investigated.
In conclusion, Vit-E reduces asthmatic features, 12/15-LOX, and nitrooxidative stress in lung and restores mitochondrial dysfunction, and these findings might help in developing newer therapeutic modalities for asthma.
GRANTS
The study was funded by the Net Work Project (NWP0033) of the Council of Scientific and Industrial Research, Government of India. U. Mabalirajan and G. D. Leishangthem acknowledge the Indian Council of Medical Research, New Delhi, and J. Aich acknowledges Council of Scientific and Industrial Research for their fellowships.
ACKNOWLEDGMENTS
We thank Dr. Arjun Ram and Dr. Anurag Agrawal for their suggestions and Tanveer Ahmad and Dr. Vijay Pal Singh for their help.
Present address: for U. Mabalirajan: BSD/Dept. of Pathology, The Univ. of Chicago, 5841 S. Maryland Ave., MC 3083, Chicago, IL 60637.
REFERENCES
- 1. Agrawal A, Rengarajan S, Adler KB, Ram A, Ghosh B, Fahim M, Dickey BF. Inhibition of mucin secretion with MARCKS-related peptide improves airway obstruction in a mouse model of asthma. J Appl Physiol 102: 399–405, 2007 [DOI] [PubMed] [Google Scholar]
- 2. Bjørneboe A, Bjørneboe GE, Drevon CA. Absorption, transport and distribution of vitamin E. J Nutr 120: 233–242, 1990 [DOI] [PubMed] [Google Scholar]
- 3. Chanez P, Bonnans C, Chavis C, Vachier I. 15-Lipoxygenase: a Janus enzyme? Am J Respir Cell Mol Biol 27: 655–658, 2002 [DOI] [PubMed] [Google Scholar]
- 4. Cohn L. Mucus in chronic airway diseases: sorting out the sticky details. J Clin Invest 116: 306–308, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Cunningham ML. A mouse is not a rat is not a human: species differences exist. Toxicol Sci 70: 157–158, 2002 [DOI] [PubMed] [Google Scholar]
- 6. Del Prete AP, Zaccagnino M, Di Paola M, Saltarella C, Oliveros Celis Nico B, Santoro G, Lorusso M. Role of mitochondria and reactive oxygen species in dendritic cell differentiation and functions. Free Radic Biol Med 44: 1443–1451, 2008 [DOI] [PubMed] [Google Scholar]
- 7. Devereux G, Turner SW, Craig LC, McNeill G, Martindale S, Harbour PJ, Helms PJ, Seaton A. Low maternal vitamin E intake during pregnancy is associated with asthma in 5-year-old children. Am J Respir Crit Care Med 174: 499–507, 2006 [DOI] [PubMed] [Google Scholar]
- 8. Goldstein JL, Kita T, Brown MS. Defective lipoprotein receptors and atherosclerosis. Lessons from an animal counterpart of familial hypercholesterolemia. N Engl J Med 309: 288–296, 1983 [DOI] [PubMed] [Google Scholar]
- 9. Grossman S, Waksman EG. New aspects of the inhibition of soybean lipoxygenase by alpha-tocopherol. Evidence for the existence of a specific complex. Int J Biochem 16: 281–289, 1984 [DOI] [PubMed] [Google Scholar]
- 10. Hajek AR, Lindley AR, Favoreto S, Jr, Carter R, Schleimer RP, Kuperman DA. 12/15-Lipoxygenase deficiency protects mice from allergic airways inflammation and increases secretory IgA levels. J Allergy Clin Immunol 122: 633–639, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hatley ME, Srinivasan S, Reilly KB, Bolick DT, Hedrick CC. Increased production of 12/15 lipoxygenase eicosanoids accelerates monocyte/endothelial interactions in diabetic db/db mice. J Biol Chem 278: 25369–25375, 2003 [DOI] [PubMed] [Google Scholar]
- 12. Heydeck D, Thomas L, Schnurr K, Trebus F, Thierfelder WE, Ihle JN, Kühn H. Interleukin-4 and -13 induce upregulation of the murine macrophage 12/15-lipoxygenase activity: evidence for the involvement of transcription factor STAT6. Blood 92: 2503–2510, 1998 [PubMed] [Google Scholar]
- 13. Holgate ST, Davies DE, Puddicombe S, Richter A, Lackie P, Lordan J, Howarth P. Mechanisms of airway epithelial damage: epithelial-mesenchymal interactions in the pathogenesis of asthma. Eur Respir J 44: 24s–29s, 2003 [DOI] [PubMed] [Google Scholar]
- 14. Kiliç FS, Erol K. The effects of vitamin E in ovalbumin-sensitized guinea pigs. Methods Find Exp Clin Pharmacol 25: 27–31, 2003 [DOI] [PubMed] [Google Scholar]
- 15. Li X, May JM. Location and recycling of mitochondrial alpha-tocopherol. Mitochondrion 3: 29–38, 2003 [DOI] [PubMed] [Google Scholar]
- 16. Liu W, Li G, Zhang X. Effects of selenium and vitamin E on arachidonic acid metabolism in experimental myocardial ischaemia. Zhonghua Yu Fang Yi Xue Za Zhi 29: 279–282, 1995 [PubMed] [Google Scholar]
- 17. Li-Weber M, Giaisi M, Treiber MK, Krammer PH. Vitamin E inhibits IL-4 gene expression in peripheral blood T cells. Eur J Immunol 32: 2401–2408, 2002 [DOI] [PubMed] [Google Scholar]
- 18. Mabalirajan U, Dinda AK, Kumar S, Roshan R, Gupta P, Sharma SK, Ghosh B. Mitochondrial structural changes and dysfunction are associated with experimental allergic asthma. J Immunol 181: 3540–3548, 2008 [DOI] [PubMed] [Google Scholar]
- 19. Mabalirajan U, Dinda AK, Sharma SK, Ghosh B. Esculetin restores mitochondrial dysfunction and reduces allergic asthma features in experimental murine model. J Immunol 183: 2059–2067, 2009 [DOI] [PubMed] [Google Scholar]
- 20. Masini A, Ceccarelli D, Giovannini F, Montosi G, Garuti C, Pietrangelo A. Iron-induced oxidant stress leads to irreversible mitochondrial dysfunctions and fibrosis in the liver of chronic iron-dosed gerbils. The effect of silybin. J Bioenerg Biomembr 32: 175–182, 2000 [DOI] [PubMed] [Google Scholar]
- 21. Mason MG, Nicholls P, Wilson MT, Cooper CE. Nitric oxide inhibition of respiration involves both competitive (heme) and noncompetitive (copper) binding to cytochrome c oxidase. Proc Natl Acad Sci USA 103: 708–713, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. McKeever TM, Britton J. Diet and asthma. Am J Respir Crit Care Med 170: 725–729, 2004 [DOI] [PubMed] [Google Scholar]
- 23. Navarro A, Boveris A. The mitochondrial energy transduction system and the aging process. Am J Physiol Cell Physiol 292: C670–C686, 2007 [DOI] [PubMed] [Google Scholar]
- 24. Okamoto N, Murata T, Tamai H, Tanaka H, Nagai H. Effects of alpha tocopherol and probucol supplements on allergen-induced airway inflammation and hyperresponsiveness in a mouse model of allergic asthma. Int Arch Allergy Immunol 141: 172–180, 2006 [DOI] [PubMed] [Google Scholar]
- 25. Paradies G, Ruggiero FM, Petrosillo G, Quagliariello E. Peroxidative damage to cardiac mitochondria: cytochrome oxidase and cardiolipin alterations. FEBS Lett 424: 155–158, 1998 [DOI] [PubMed] [Google Scholar]
- 26. Qin F, Yan C, Patel R, Liu W, Dong E. Vitamins C and E attenuate apoptosis, beta-adrenergic receptor desensitization, and sarcoplasmic reticular Ca2+ ATPase downregulation after myocardial infarction. Free Radic Biol Med 40: 1827–1842, 2006 [DOI] [PubMed] [Google Scholar]
- 27. Raby BA, Klanderman B, Murphy A, Mazza S, Camargo CA, Jr, Silverman EK, Weiss ST. A common mitochondrial haplogroup is associated with elevated total serum IgE levels. J Allergy Clin Immunol 20: 351–358, 2007 [DOI] [PubMed] [Google Scholar]
- 28. Ram A, Mabalirajan U, Das M, Bhattacharya I, Dinda AK, Gangal SV, Ghosh B. Glycyrrhizin alleviates experimental allergic asthma in mice. Int Immunopharmacol 6: 1468–1477, 2006 [DOI] [PubMed] [Google Scholar]
- 29. Ram A, Mabalirajan U, Singh SK, Singh VP, Ghosh B. Mepacrine alleviates airway hyperresponsiveness and airway inflammation in a mouse model of asthma. Int Immunopharmacol 8: 893–899, 2008 [DOI] [PubMed] [Google Scholar]
- 30. Rhee SJ, Jeong YC, Choi JH. Effects of vitamin E on phospholipase A2 activity and oxidative damage to the liver in streptozotocin-induced diabetic rats. Ann Nutr Metab 49: 392–396, 2005 [DOI] [PubMed] [Google Scholar]
- 31. Sackesen C, Ercan H, Dizdar E, Soyer O, Gumus P, Tosun BN, Büyüktuncer Z, Karabulut E, Besler T, Kalayci O. A comprehensive evaluation of the enzymatic and nonenzymatic antioxidant systems in childhood asthma. J Allergy Clin Immunol 122: 78–85, 2008 [DOI] [PubMed] [Google Scholar]
- 32. Schewe T. 15-Lipoxygenase-1: a prooxidant enzyme. Biol Chem 383: 365–374, 2002 [DOI] [PubMed] [Google Scholar]
- 33. Seaton A. From nurture to Nature—the story of the Aberdeen asthma dietary hypothesis. QJM 101: 237–239, 2008 [DOI] [PubMed] [Google Scholar]
- 34. Taccone-Gallucci M, Manca-di-Villahermosa S, Maccarrone M. Leukotrienes. N Engl J Med 358: 746, 2008 [DOI] [PubMed] [Google Scholar]
- 35. Vendemiale G, Grattagliano I, Altomare E, Serviddio G, Portincasa P, Prigigallo F, Palasciano G. Mitochondrial oxidative damage and myocardial fibrosis in rats chronically intoxicated with moderate doses of ethanol. Toxicol Lett 123: 209–216, 2001 [DOI] [PubMed] [Google Scholar]
- 36. Weiss ST, Litonjua AA. Childhood asthma is a fat-soluble vitamin deficiency disease. Clin Exp Allergy 38: 385–387, 2008 [DOI] [PubMed] [Google Scholar]
- 37. Wood LG, Gibson PG, Garg ML. Biomarkers of lipid peroxidation, airway inflammation and asthma. Eur Respir J 21: 177–186, 2003 [DOI] [PubMed] [Google Scholar]
- 38. Zhang JG, Nicholls-Grzemski FA, Tirmenstein MA, Fariss MW. Vitamin E succinate protects hepatocytes against the toxic effect of reactive oxygen species generated at mitochondrial complexes I and III by alkylating agents. Chem Biol Interact 138: 267–284, 2001 [DOI] [PubMed] [Google Scholar]
- 39. Zhao L, Praticò D, Rader DJ, Funk CD. 12/15-Lipoxygenase gene disruption and vitamin E administration diminish atherosclerosis and oxidative stress in apolipoprotein E deficient mice through a final common pathway. Prostaglandins Other Lipid Mediat 78: 185–193, 2005 [DOI] [PubMed] [Google Scholar]
- 40. Zingg JM. Vitamin E and mast cells. Vitam Horm 76: 393–418, 2007 [DOI] [PubMed] [Google Scholar]