Abstract
We investigated the capability of biodegradable silica xerogel as a novel carrier of antibiotic and the efficacy of treatment compared to that with the same dose of free drug against murine salmonellosis. The drug molecules (31%) entrapped in the sol-gel matrix remained in biologically active form, and the bactericidal effect was retained upon drug release. The in vitro drug release profiles of the gentamicin from the xerogel and that from the xerogel-polyethylene glycol (PEG) were distinctly different at pH 7.4. A delayed release of gentamicin was observed from the silica xerogel network (57% in 33 h), and with the addition of 2% PEG, the release rate reached 90% in 33 h. Administration of two doses of the silica xerogel significantly reduced the Salmonella enterica serovar Typhimurium load in the spleens and livers of infected AJ 646 mice. The silica xerogel and xerogel-PEG achieved a 0.45-log and a 0.41-log reduction in the spleens, respectively, while for the free drug there was no reduction. On the other hand, silica xerogel and xerogel-PEG achieved statistically significant 1.13-log and 1.15-log reductions in the livers, respectively, while for the free drug the reduction was a nonsignificant value of 0.07 log. This new approach, which utilizes a room-temperature synthetic route for incorporating therapeutic drugs into the silica matrix, should improve the capability for targeting intracellular pathogens.
Targeting intracellular pathogens like Salmonella, Brucella, and Mycobacterium species remains a medical challenge. These intracellular pathogens can cause persistent infection due to their ability to maintain infections even in the presence of inflammation, specific antimicrobial mechanisms, and a robust adaptive immune response of the host (12). Some individuals who are infected with Salmonella enterica serovar Typhi become lifelong carriers, periodically shedding large numbers of bacteria (11). In host-adapted salmonelloses such as typhoid fever, the Salmonella-infected phagocytes gain access to the lymphatics and bloodstream, allowing the bacteria to spread to the liver and the spleen (20). Treatment and eradication are difficult since infections are localized within phagocytic cells, and it is difficult to achieve the optimum relatively high concentration of therapeutics within the infected cells (15). Thus, an optimum strategy to treat these infections should address targeting of active drugs to the intracellular compartment where bacteria replicate and should prolong release of the antibiotics so that the number of doses and associated drug toxicity can be reduced. The associated problems with delivering free antibiotics to the intracellular space have led to the investigations of improved drug carriers for treating intracellular pathogens, including antibiotics loaded into liposomes, microspheres, polymeric carriers, and nanoplexes (15).
The success of any approach for drug delivery to intracellular pathogens depends on the ability to construct a biocompatible carrier with a high loading capacity of therapeutic drugs with no or minimum premature release of the cargo before reaching the replicative niche of the bacteria. The recently developed sol-gel technique has shown great promise for biomedical applications by offering new possibilities for embedding antibiotics within silica and for controlling their release from the host matrix into the surrounding medium (7, 16, 19, 22, 23). The composition of silica xerogels that can be tuned and the fabrication method at low temperatures enable them to carry biologically active agents and to be useful as a drug delivery system (8). Sol-gel silica materials have been shown to be biocompatible in vivo as they are readily degradable inside the body (6), which eliminates the problem of accumulation that has remained a major drawback for many other nanoparticle delivery systems. The various glass compositions cause no adverse tissue reactions and are easily eliminated from the body through the kidneys as they degrade into Si(OH)4 (1, 18, 21).
Also, the size, zeta potential, pore structure, and the surface characteristics of silica xerogel make it a suitable carrier for therapeutics to target the replicative niche of intracellular pathogens.
Although gentamicin exhibits several characteristics that make it a useful antimicrobial agent in vitro, it does not kill intracellular Salmonella due to the polar nature of the drug and the associated low level of intracellular penetration. Therefore, the usefulness of gentamicin seems to be limited to eradication of the extracellular pathogens. In the present study, we used a mouse model of human typhoid fever to investigate the capability of sol-gel processed silica as a carrier for gentamicin and the efficacy of treatment compared to that with the free form of the same drug against Salmonella enterica serovar Typhimurium, a leading cause of human gastroenteritis (10).
MATERIALS AND METHODS
All chemicals were purchased from Sigma-Aldrich Chemicals. Tetraethylorthosilica (TEOS), gentamicin sulfate, polyethylene glycol (PEG; molecular weight, 8,000), ammonium hydroxide (NH4OH), and hydrochloric acid (HCl) were used as received.
Preparation of antibiotic-loaded xerogel and xerogel-PEG.
The antibiotic-loaded mesoporous silica nanoparticles were prepared at room temperature by hydrolysis and polycondensation of tetraethoxysilane (TEOS) with water and catalyst contents (2). Silica sol was prepared by two-phase catalysis with HCl and NH3. Immobilization of antibiotic in the silica gels was carried out by mixing antibiotic solution with prehydrolyzed TEOS sol. Eleven milliliters of TEOS was added to 9 ml of deionized water at room temperature to form a homogenous solution. The pH of the sol (pH 6.5) was adjusted by addition of nitric acid as catalyst followed by addition of ammonium hydroxide base. To control the hydrolysis process of the TEOS, the temperature was kept under 20°C. After 1 h of hydrolysis, 750 mg of gentamicin was dissolved in 3 ml of water and added into the reaction mixture. The resulting solution was stirred for 3 h, covered with Parafilm, and left for gelation for 72 h. After gelation, holes were made in the Parafilm and the gels were allowed to continue aging and drying under ambient conditions. During the drying process, evaporation of solvents and reaction by-products occurred from the interconnected pore network to reach a constant weight and the so-called “xerogels” were obtained. The xerogel-PEG was fabricated like the xerogel described above except with the addition of 2% PEG to control the pore structure of the materials. The drug-loaded xerogels and xerogel-PEG were rinsed with deionized water to remove unreacted reagents. The final hybrid sol-gels were crushed into powder by ball milling, and powders were sieved to less than ∼5 μm in size for further characterization.
Characterization and instrumentation.
The particle size of the silica xerogels was analyzed in a Malvern Zetasizer Nano Series HT. For the analysis, the particles were suspended in phosphate-buffered saline (PBS) at a concentration of 0.50 mg/ml and sonicated in a glass vial with no heating (Branson 1510 sonifier) for 30 min. The suspension was transferred to a polystyrene cuvette, and the particle size distribution was measured. The results were processed with DTS software version 4.20 (Malvern Zetasizer). The microstructure pore size parameters, specific surface area, and pore volume were determined from N2 adsorption isotherms at 77K with an Autosorb-1 MP (Quantachrome Co.). Prior to the measurements, the samples were degassed at 25°C and 1 Pa overnight.
In vitro release of gentamicin.
The profiles of gentamicin release from silica xerogel and xerogel-PEG matrix were studied using suspensions (50 μg/ml) of the antibiotic-loaded xerogel and xerogel-PEG in PBS. The suspensions were kept at room temperature, and the release was determined by taking aliquots at different time points. At the end of each time point, 1 ml of the solution was removed for sampling and replaced immediately with an identical volume of PBS. The aliquots were centrifuged, and the absorbance value of the gentamicin sulfate antibiotic in the supernatant was measured (23). The quantitative measurement in each elution sample was performed using a UV/VIS spectrophotometer (Perkin-Elmer). The analytical wavelength λ = 246 nm, corresponding to the absorbance maximum, was selected for the determination of the drug. The drug-PBS standard solutions were used for calibration. The calibration curve of gentamicin was determined by taking absorbance versus drug concentration between 0 and 100 mg/ml.
Bacterial strain.
Salmonella enterica serovar Typhimurium strain LT2 (10) was routinely grown at 37°C in tryptic soy broth or on tryptic soy agar (Difco).
Animal experiment (in vivo infection assay).
To study the efficacy of the silica-loaded antibiotic in vivo, female AJ 646 mice (Charles River Laboratories, Wilmington, MA) 6 weeks old were used. Groups of five mice each were infected intraperitoneally with Salmonella serovar Typhimurium (4 × 103 CFU/mouse). The animals were treated intraperitoneally with two doses of (i) silica control, (ii) free gentamicin (150 μg), (iii) silica xerogel gentamicin (150 μg), (iv) silica xerogel-PEG-gentamicin (150 μg), and (v) PBS at days 3 and 5 postinfection. Forty-eight hours after administration of the last dose, the animals were euthanized. The spleens and livers were collected, and bacterial CFU per individual organ were determined by plating a series of 10-fold serial dilutions of the organ homogenates on tryptic soy agar plates. The number of colonies was determined after incubation for 24 h at 37°C. The experimental procedures on mice and the facilities used to hold the experimental animals were in compliance with the rules of the Virginia Tech Institutional Animal Care and Use Committee.
Statistical analysis.
All statistical analyses for bacterial clearance from organs were performed using the Student two-tailed t test using Microsoft Excel. P values of ≤0.05 were considered significant.
RESULTS
Characterization of silica xerogels.
The synthesized acid-base-catalyzed sol-gel silica and silica modified by the addition of low-molecular-weight PEG matrices both were transparent, glassy, and crack-free monoliths. The addition of gentamicin to the xerogel did not change the physical characteristics of the particles. This indicates that the gentamicin was homogeneously distributed throughout the xerogel without any macroscopic phase separation. To avoid any damage or loss of activity of the antibiotic, room temperature and low-acidity parameters were used. The particle sizes of the porous silica xerogel and porous silica-PEG were 190 to 395 and 220 to 342 nm, respectively, and the zeta potentials (surface charges) were −1.78 and −6.24, respectively.
N2 adsorption-desorption isotherm analysis.
Nitrogen adsorption-desorption isotherms and pore parameters of porous silica xerogel and porous silica xerogel-PEG matrix are shown in Table 1. The data were recorded over the relative pressure (P/Po) from 0 to 1 at −196°C. Based on the IUPAC classification scheme, the isotherm curve observed is of type IV, which has a characteristic loop at around 0.5 and 0.8 of P/Po. Accordingly, these isotherms show low adsorption at lower relative pressures and a sudden increase in adsorption at high relative pressure due to capillary condensation in the mesoporous structure. These isotherms describe solids with a well-defined distribution of the mesoporous structure.
TABLE 1.
The structural parameters of the silica xerogel and silica xerogel-PEGa
| Sample | SBET (m2/g) | Vpore (m3/g) | Dpeak (nm) |
|---|---|---|---|
| Silica xerogel | 412.2 | 1.459 | 9.60 |
| Silica xerogel-gentamicin | 21.14 | 0.068 | 3.60 |
| Silica xerogel-PEG | 230.5 | 1.096 | 1.430 |
| Silica xerogel-PEG-gentamicin | 38.89 | 0.054 | 1.717 |
SBET, BET surface area; Vpore, pore volume; Dpeak, pore diameter.
In vitro release studies of entrapped gentamicin from xerogel and xerogel-PEG matrix.
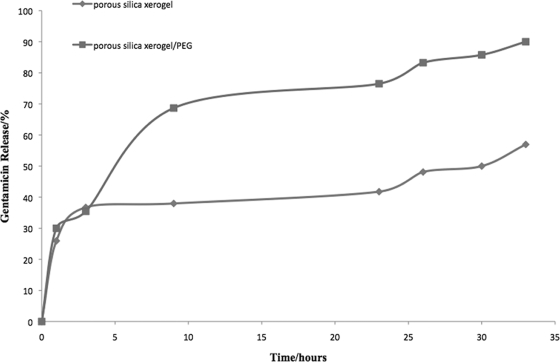
In vitro release profiles of gentamicin immobilized in the silica xerogel and xerogel-PEG matrix are presented in Fig. 1. All samples showed a higher release rate followed by a slower, steady release. Silica xerogel-PEG was used to investigate how the polymer (PEG) concentration influences the release of gentamicin. Incorporation of 2% PEG in the fabrication process increased the release of gentamicin from the xerogel (Fig. 1). There was a sustained release of gentamicin (57% in 33 h) from the silica xerogel network, while with the addition of 2% PEG the gentamicin release rate reached 90% in 33 h.
FIG. 1.
Cumulative release of gentamicin from silica xerogel and silica xerogel-PEG complex. In vitro release profiles of gentamicin immobilized in the silica xerogel and that in the xerogel-PEG matrix showed a higher release rate followed by a slower, steady release. There was a sustained release of gentamicin (57% in 33 h) from the silica xerogel network, while with the addition of 2% PEG the gentamicin release rate reached 90% in 33 h.
In vivo infection assay.
The efficacy of the antibiotic-loaded silica versus the free form of the same concentration of gentamicin was tested in AJ 646 mice. The results are summarized in Table 2. The xerogel-gentamicin and xerogel-PEG-gentamicin therapies induced a significant (P < 0.05) reduction in the log CFU of Salmonella serovar Typhimurium in the spleens and livers of infected mice. The empty xerogel and the free gentamicin did not produce a significant reduction of Salmonella serovar Typhimurium CFU.
TABLE 2.
Efficacy of two doses of silica-antibiotic hybrid against infection with Salmonella serovar Typhimuriuma
| Treatment | Log CFU/spleen | Reduction (log) | Log CFU/liver | Reduction (log) |
|---|---|---|---|---|
| No treatment | 3.16 ± 0.06 | 0.00 | 2.80 ± 0.09 | 0.00 |
| Silica control | 3.34 ± 0.14 | +0.18 | 2.65 ± 0.03 | 0.15 |
| Free gentamicin | 3.38 ± 0.15 | +0.22 | 2.73 ± 0.11 | 0.07 |
| Silica-gentamicin | 2.71 ± 0.07 | 0.45* | 1.67 ± 0.22 | 1.13* |
| Silica-PEG-gentamicin | 2.75 ± 0.11 | 0.41* | 1.65 ± 0.43 | 1.15* |
Groups of five mice each were infected intraperitoneally with Salmonella serovar Typhimurium (4 × 103 CFU/mouse). After 72 h, the animals received two doses of free or silica-loaded antibiotics (150 μg/mouse). At 48 h after administration of the last dose, the animals were euthanized; liver and spleen were assayed for CFU. Values represent the mean ± standard error numbers of CFU. Statistical significance levels were defined as P < 0.05 (shown by asterisks).
DISCUSSION
The development of a new biodegradable drug carrier capable of intracellular delivery of sustained released therapeutics to where the intracellular pathogen resides will allow intracellular accumulation of the antibiotic after particle degradation and will improve drug targeting. Moreover, sustained drug delivery could reduce long-term treatment, reduce dosing frequency, and eliminate some of the toxic side effects associated with the free drug. In the present study, we investigated the capability of silica xerogel as a carrier of gentamicin and the efficacy of treatment compared to that with free drug against murine salmonellosis. The drug molecules entrapped in the sol-gel matrix remained in biologically active form, and the bactericidal effect was retained upon its release. The fabrication method using a room-temperature synthetic route and low acid was essential to preserve the antibiotic activity.
The pore size of the mesoporous xerogel and xerogel-PEG matrix is large enough to allow gentamicin molecules to diffuse through the pore channels, which leads to different levels of decrease in nitrogen adsorption, reflecting the different storage amounts of the drug in the porous network. The incorporation of gentamicin molecules into the silica leads to a decrease in pore diameter, surface area, and pore volume (Table 1).
PEG in high concentration has been widely used as a successful strategy to incorporate hydrophilic chains or to change the surface charges of the therapeutic nanoparticles, resulting in increased blood circulation time and decreased phagocytosis possibly due to decreased nonspecific interaction of the complex with serum components (14). In this study, PEG was used at a low concentration to control the pore structure and enhance the release of the cargo. By incorporation of 2% PEG during hydrolysis, the channels were better connected with increased pore volume (Table 1). Thus, it is obvious that the pore structure of silica xerogel can be modified by addition of PEG and that the silica xerogel-PEG with various pore structures can be prepared by changing the PEG ratio.
The larger pore volume and hydrophilic factors facilitate more penetration of the immersion solution inside the silica lattice and accelerate the diffusion of the gentamicin molecules in a given period of time. The in vitro drug release profiles of the gentamicin from the xerogel and that from the xerogel-PEG were distinctly different at pH 7.4.
Experimental results suggest that the release of gentamicin from the silica xerogel matrices is a combination of diffusion and swelling of the matrix. For silica alone, due to limited interconnection of the channels, the immersion solution penetrates slowly through the silica matrix to the interior, and therefore, the release of gentamicin from the silica matrix was delayed. For the PEG-silica xerogel complex, during the immersion time a partial physical erosion of the material surface could have taken place followed by penetration of solution inside the PEG-silica complex. Incorporation of PEG in the silica matrix allows formation of a more interconnected porous network. This allows the immersion solution to freely penetrate the pores of the matrices, causing swelling. This allows the gentamicin to be more easily leached out through the interconnected channels.
Both release rates were sufficiently low to signify that the xerogel silica remained intact under physiological conditions at pH 7.4, and this could enhance phagocytosis by the reticuloendothelial system (20) and reduce ototoxic and nephrotoxic effects associated with free gentamicin (4, 15). Although the faster release of the drug after addition of 2% PEG did not alter the efficacy of treatment (Table 2), it should be useful for other applications that require faster release of therapeutics without interfering with the phagocytosis process.
Determination of the efficiency of the silica xerogel and xerogel-PEG for elimination of Salmonella from the spleens and livers of infected mice was assessed after treatment with two doses. The results (shown in Table 2) indicated that both silica xerogel and xerogel-PEG were more effective in clearing the infection in the spleen and liver than was the same dose of free drug. The silica xerogel and xerogel-PEG achieved a 0.45-log and a 0.41-log reduction in the spleens, respectively, while for the free drug there was no reduction. Both of these values were statistically significant (P < 0.05) in comparison with the results achieved for the negative control. On the other hand, silica xerogel and xerogel-PEG achieved statistically significant 1.13-log and 1.15-log reductions in the livers (P < 0.05), respectively, while for the free drug the reduction was a nonsignificant value of 0.07 log from the results achieved for the control.
To achieve intracellular delivery of aminoglycosides, several drug delivery strategies have been utilized using liposomes and polymeric carriers (3, 9). Although these drug delivery systems have improved the intracellular delivery of aminoglycosides, they still suffer from low drug incorporation. Silica xerogel obtained by the techniques used in this study contained 31% of its weight in antibiotic, 31-fold higher than the values reported earlier using liposomes (5). The higher loading capacity of the silica xerogel is attributed to the significant clearance of the infection from the mice. Our previous experiment used only 17% loading of gentamicin, and in these studies the clearance was not statistically significant (13). This clearly indicates that we could achieve a higher rate of clearance of infection by changing the porosity and increasing the loading capacity of the silica xerogel. It is possible that, by proper engineering of the sol-gel process, even higher drug loading capacities could be achieved, but the data show that at the current drug loading levels used in this study, a sufficient amount of drug is delivered. One variation of the process, which could achieve much higher drug loading amounts, would be using aerogel particles as drug delivery carriers. Aerogels have an open mesoporic structure with a porosity up to 98% by volume and a high surface area of 400 to 1,000 m2/g, which would be suited for high loading of the drugs (17).
The present study demonstrates the feasibility and efficacy of targeting Salmonella by using gentamicin incorporated into bioactive porous silica by using a xerogel processing technique and a room-temperature synthesis route. The gentamicin-hybrid xerogel silica showed a significantly higher rate of bacterial clearance from organs than did the same dose of free drug. Although the mouse model did not provide direct evidence that the hybrid xerogel was taken up intracellularly, these results indicate that the porous xerogel silica could be used as a drug delivery system to control salmonellosis.
Footnotes
Published ahead of print on 10 August 2009.
REFERENCES
- 1.Ahola, M., P. Kortesuo, I. Kangasniemi, J. Kiesvaara, and A. Yli-Urpo. 2000. Silica xerogel carrier material for controlled release of toremifene citrate. Int. J. Pharm. 195:219-227. [DOI] [PubMed] [Google Scholar]
- 2.Conroy, J. F. T., M. E. Power, J. Martin, B. Earp, B. Hosticka, C. E. Daitch, and P. M. Norris. 2000. Cells in sol-gels I: a cytocompatible route for the production of macroporous silica gels. J. Sol Gel Sci. Technol. 18:269-283. [Google Scholar]
- 3.Fattal, E., J. Rojas, L. Roblot-Treupel, A. Andremont, and P. Couvreur. 1991. Ampicillin-loaded liposomes and nanoparticles: comparison of drug loading, drug release and in vitro antimicrobial activity. J. Microencapsul. 8:29-36. [DOI] [PubMed] [Google Scholar]
- 4.Forge, A., and J. Schacht. 2000. Aminoglycoside antibiotics. Audiol. Neurootol. 5:3-22. [DOI] [PubMed] [Google Scholar]
- 5.Khalil, R. M., F. E. Murad, S. A. Yehia, M. S. El-Ridy, and H. A. Salama. 1996. Free versus liposome-entrapped streptomycin sulfate in treatment of infections caused by Salmonella enteritidis. Pharmazie 51:182-184. [PubMed] [Google Scholar]
- 6.Kortesuo, P., M. Ahola, S. Karlsson, I. Kangasniemi, A. Yli-Urpo, and J. Kiesvaara. 2000. Silica xerogel as an implantable carrier for controlled drug delivery-evaluation of drug distribution and tissue effects after implantation. Biomaterials 21:193-198. [DOI] [PubMed] [Google Scholar]
- 7.Kortesuo, P., M. Ahola, S. Karlsson, I. Kangasniemi, J. Kiesvaara, and A. Yli-Urpo. 1999. Sol-gel-processed sintered silica xerogel as a carrier in controlled drug delivery. J. Biomed. Mater. Res. 44:162-167. [DOI] [PubMed] [Google Scholar]
- 8.Lee, E. J., D. S. Shin, H. E. Kim, H. W. Kim, Y. H. Koh, and J. H. Jang. 2009. Membrane of hybrid chitosan-silica xerogel for guided bone regeneration. Biomaterials 30:743-750. [DOI] [PubMed] [Google Scholar]
- 9.Lutwyche, P., C. Cordeiro, D. J. Wiseman, M. St.-Louis, M. Uh, M. J. Hope, M. S. Webb, and B. B. Finlay. 1998. Intracellular delivery and antibacterial activity of gentamicin encapsulated in pH-sensitive liposomes. Antimicrob. Agents Chemother. 42:2511-2520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McClelland, M., K. E. Sanderson, J. Spieth, S. W. Clifton, P. Latreille, L. Courtney, S. Porwollik, J. Ali, M. Dante, F. Du, S. Hou, D. Layman, S. Leonard, C. Nguyen, K. Scott, A. Holmes, N. Grewal, E. Mulvaney, E. Ryan, H. Sun, L. Florea, W. Miller, T. Stoneking, M. Nhan, R. Waterston, and R. K. Wilson. 2001. Complete genome sequence of Salmonella enterica serovar Typhimurium LT2. Nature 413:852-856. [DOI] [PubMed] [Google Scholar]
- 11.Monack, D. M., D. M. Bouley, and S. Falkow. 2004. Salmonella typhimurium persists within macrophages in the mesenteric lymph nodes of chronically infected Nramp1+/+ mice and can be reactivated by IFNgamma neutralization. J. Exp. Med. 199:231-241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Monack, D. M., A. Mueller, and S. Falkow. 2004. Persistent bacterial infections: the interface of the pathogen and the host immune system. Nat. Rev. Microbiol. 2:747-765. [DOI] [PubMed] [Google Scholar]
- 13.Munusamy, P., M. N. Seleem, H. Alqublan, R. Tyler, Jr., N. Sriranganathan, and G. Pickrell. 2009. Targeted drug delivery using silica xerogel systems to treat diseases due to intracellular pathogens. Mater. Sci. Eng. C doi: 10.1016/j.msec.2009.05.020. [DOI]
- 14.Sato, A., S. W. Choi, M. Hirai, A. Yamayoshi, R. Moriyama, T. Yamano, M. Takagi, A. Kano, A. Shimamoto, and A. Maruyama. 2007. Polymer brush-stabilized polyplex for a siRNA carrier with long circulatory half-life. J. Control. Release 122:209-216. [DOI] [PubMed] [Google Scholar]
- 15.Seleem, M. N., N. Jain, N. Pothayee, A. Ranjan, J. S. Riffle, and N. Sriranganathan. 2009. Targeting Brucella melitensis with polymeric nanoparticles containing streptomycin and doxycycline. FEMS Microbiol. Lett. 294:24-31. [DOI] [PubMed] [Google Scholar]
- 16.Slowing, I. I., J. L. Vivero-Escoto, C. W. Wu, and V. S. Lin. 2008. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 60:1278-1288. [DOI] [PubMed] [Google Scholar]
- 17.Smirnova, I., J. Mamic, and W. Arlt. 2003. Adsorption of drugs on silica aerogels. Langmuir 19:8521-8525. [Google Scholar]
- 18.Trewyn, B. G., S. Giri, I. I. Slowing, and V. S.-Y. Lin. 2007. Mesoporous silica nanoparticle based controlled release, drug delivery, and biosensor systems. Chem. Commun. 31:3236-3245. [DOI] [PubMed] [Google Scholar]
- 19.Trewyn, B. G., I. I. Slowing, S. Giri, H.-T. Chen, and V. S.-Y. Lin. 2007. Synthesis and functionalization of a mesoporous silica nanoparticle based on the sol-gel process and applications in controlled release. Acc. Chem. Res. 40:846-853. [DOI] [PubMed] [Google Scholar]
- 20.Vazquez-Torres, A., J. Jones-Carson, A. J. Baumler, S. Falkow, R. Valdivia, W. Brown, M. Le, R. Berggren, W. T. Parks, and F. C. Fang. 1999. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 401:804-808. [DOI] [PubMed] [Google Scholar]
- 21.Wen, L. X., D. Hao-Min, J. X. Wang, and J. F. Chen. 2006. Porous hollow silica nanoparticles as carriers for controlled delivery of ibuprofen to small intestine. J. Nanosci. Nanotechnol. 6:3139-3144. [DOI] [PubMed] [Google Scholar]
- 22.Zhu, Y., and J. Shi. 2007. A mesoporous core-shell structure for pH-controlled storage and release of water-soluble drug. Microporous Mesoporous Mater. 103:243-249. [Google Scholar]
- 23.Zhu, Y., J. Shi, Y. Li, H. Chen, W. Shen, and X. Dong. 2005. Hollow mesoporous spheres with cubic pore network as a potential carrier for drug storage and its in vitro release kinetics. J. Mater. Res. 20:54-61. [Google Scholar]