Abstract
Background and purpose:
The chicken anterior mesenteric artery contains an outer longitudinal smooth muscle layer, whose neural regulation remains to be elucidated. ATP evokes a depolarization in the smooth muscle through P2Y purinoceptors. However, there may be an additional inhibitory regulation because blockade of P2Y purinoceptors converts the depolarization to hyperpolarization. The objective of the present study was to examine the mechanism underlying this hyperpolarization.
Experimental approach:
Membrane potentials of longitudinal smooth muscle of the chicken mesenteric artery were recorded with a microelectrode technique. Perivascular nerves were stimulated by applying electrical field stimulation (EFS).
Key results:
EFS induced a hyperpolarization in preparations obtained from 5-week-old chickens, whereas it evoked a depolarization in those from 12-week-old chickens. The EFS-evoked hyperpolarization in 5-week-old chickens was blocked by a non-specific purinoceptor antagonist, suramin, and by a specific P2X purinoceptor antagonist, pyridoxal phosphate-6-azophenyl-2′,4′-disulphonic acid. Desensitization of the P2X purinoceptor with its agonist α,β-MeATP significantly suppressed EFS-evoked hyperpolarization. Blockade of the P2Y purinoceptor did not affect EFS-evoked hyperpolarization. The application of the NOS inhibitor Nω-nitro-L-arginine methyl ester or the removal of the endothelium inhibited the hyperpolarization. The application of the nitric oxide (NO) donor sodium nitroprusside mimicked the hyperpolarization. Reverse transcriptase-PCR showed that P2X purinoceptors are expressed in the endothelium of the anterior mesenteric artery.
Conclusions and implications:
Hyperpolarization in the longitudinal smooth muscle of the chicken anterior mesenteric artery was induced by ATP. ATP released from perivascular nerves may act on P2X purinoceptors in the endothelium and thereby stimulate NO production.
Keywords: ATP, chicken, endothelium, mesenteric artery, NANC, nitric oxide, perivasucular nerve, purinergic neuron, purinoceptor, smooth muscle
Introduction
The mammalian arterial smooth muscle is oriented circularly and helically (Furchgott, 1955). However, the anterior mesenteric artery of the chicken differs structurally from the basic pattern observed in mammalian arteries. The chicken anterior mesenteric artery contains two well-developed muscular components, an inner circular and an outer longitudinal layer (Ball et al., 1963), analogous to the arrangement of the muscularis externa of the venous wall. In addition to the specialized wall structure, the anterior mesenteric artery of the chicken has unique innervation. In general, arterial muscles are innervated by excitatory adrenergic nerves (Keatinge, 1966). In accordance with this, adrenergic nerve stimulation produces contraction of the circular muscle, which is mimicked and blocked by α-adrenoceptor agonists and antagonists, respectively (Bell, 1969; Gooden, 1980). In contrast, the longitudinal muscle is relaxed by adrenergic nerve stimulation, acting through β-adrenoceptors (Bolton, 1968; 1969; Bell, 1969). Furthermore, the longitudinal muscle of this vessel receives cholinergic innervation (Bell, 1969), and stimulation of cholinergic nerve fibres causes the muscles to contract (Bolton, 1968; 1969; Bell, 1969).
Accumulating evidence indicates that ATP is a non-adrenergic, non-cholinergic (NANC) transmitter, colocalized with noradrenaline in the sympathetic nerves and being released with noradrenaline (Burnstock, 1986). ATP was shown to have regulatory roles in rabbit saphenous (Burnstock and Warland, 1987a) and mesenteric (Burnstock and Warland, 1987b), rat mesenteric (Rummery et al., 2007), guinea pig mesenteric (Smyth et al., 2000) and hamster mesenteric (Thapaliya et al., 1999) arteries. Also, ATP mediates relaxation of the longitudinal muscle in the rabbit portal vein (Kennedy and Burnstock, 1985) and contraction of the longitudinal muscle in the guinea pig (Orre et al., 1996). Receptors mediating responses to ATP have been characterized as P2 purinoceptors (nomenclature follows Alexander et al., 2008) and are subdivided into two major classes, P2X and P2Y purinoceptors, which are ligand-gated ion channels and G-protein-coupled receptors, respectively (Burnstock, 2007). When ATP acts on smooth muscle P2X purinoceptors, it evokes a transient depolarization termed excitatory junction potential (McLaren et al., 1995). The transmitter also acts on endothelial P2Y purinoceptors, resulting in release of hyperpolarizing substances and thereby eliciting a hyperpolarization of smooth muscle (Pearson and Carter, 1990). Nitric oxide (NO) and prostanoids are known to act as hyperpolarizing substances that are released from endothelial cells in some blood vessels (Triggle and Ding, 2002).
In agreement with the regulatory roles of ATP, we have demonstrated that ATP evoked a P2Y-mediated, endothelium-dependent hyperpolarization in the circular smooth muscle of the chicken anterior mesenteric artery (Draid et al., 2005). However, the neurotransmitter evoked a depolarization in the longitudinal smooth muscle through P2Y but not through P2X purinoceptors, suggesting the presence of specific neural regulation of the longitudinal smooth muscle (Khalifa et al., 2005). Interestingly, blockade of P2Y purinoceptors by a specific receptor antagonist or receptor desensitization converted the depolarization to hyperpolarization (Khalifa et al., 2005). This suggests that there is an additional inhibitory regulation of membrane response in the longitudinal smooth muscle. In a preliminary experiment using younger chickens than those used in our previous studies (Khalifa et al., 2005), stimulation of perivascular nerves under NANC conditions evoked a biphasic response consisting of depolarization followed by hyperpolarization. These results suggest that the inhibitory and stimulatory purinergic components in the longitudinal smooth muscle are changed during growth of the chicken. Hence, the aim of the present study was to examine the purinergic neuronal regulation in the longitudinal smooth muscle of the anterior mesenteric artery in young chickens. We hypothesized that the inhibitory purinergic component is dominantly operated before establishing the stimulatory purinergic component, and the nature of the inhibitory component can be clarified by using artery preparations obtained from young chickens.
Methods
Animals and tissue preparation
All animal care and experimental procedures were approved by the Gifu University Animal Care and Use Committee, and all efforts were made to minimize animal suffering and to reduce the number of animals used. Male white Leghorn chickens aged 5 and 12 weeks were killed by neck dislocation. The anterior mesenteric artery (3–5 cm in length, 200–250 µm in diameter) was severed at its origin from the aorta and at the region of the sub-branches supplying the intestine and was placed in a physiological salt solution (PSS; see later) at room temperature. The middle portions of the anterior mesenteric artery were cleaned from connective tissue, and the vessels were cannulated at the proximal end with a glass micropipette (200 µm tip diameter) attached to a gravity-driven perfusion apparatus to perfuse the vessels with warmed (29°C) PSS in order to remove blood from the vessels.
Electrophysiological recordings
Arteries were placed in a partition chamber in which large extracellular silver–silver chloride plates were used to elicit nerve stimulation, as described previously (Khalifa et al., 2005). The chamber was perfused at a constant flow rate (3 mL·min−1) with pre-warmed (29°C) PSS. This temperature was used in this experiment because spontaneous activity, which makes recording from the cells difficult, was frequently produced at 35°C (Khalifa et al., 2005). To establish an NANC condition, a muscarinic acetylcholine receptor antagonist atropine (0.5 µM), an α1-adrenoceptor antagonist prazosin (5 µM) and a β-adrenoceptor antagonist propranolol (1 µM) were added to PSS. Tissue preparations were allowed to equilibrate for approximately 1 h before the experiments were undertaken. Membrane potentials were recorded with a conventional microelectrode technique, using glass capillary microelectrodes filled with 3 M KCl with tip resistances ranging from 50 to 80 MΩ (Takewaki and Ohashi, 1977). Microelectrode insertions were made in the longitudinal muscle cells through the adventitial side, within 2 mm of the stimulation electrode. Electrical activity was monitored on an oscilloscope (CS 4026, Kenwood, Tokyo, Japan) and recorded using a PowerLab system (ADInstruments, Bella Vista NSW, Australia). The perivascular nerves were stimulated by applying electrical field stimulation (EFS) (stimulus intensity: 15 V; pulse duration: 1 ms; frequency: 20 Hz) for 0.5-s periods.
PSS
The PSS used in this study had the following composition in mM: NaCl 118, KCl 4.6, CaCl2 2.7, MgCl2 1.2, KH2PO4 1.2, NaHCO3 25, glucose 11. The solution in the supply reservoir was gassed continuously with 95% O2: 5% CO2 gas mixture creating a pH of 7.2 and was warmed to 29°C.
Removal of the endothelium
Endothelium removal was achieved by injection of warmed (29°C) PSS containing collagenase (1 mg·mL−1) into the lumen of the anterior mesenteric artery for 15 min. Complete removal of the endothelium was confirmed by the absence of the ability of ACh to induce relaxation of the mesenteric artery. The removal of the endothelium had no effect on resting membrane potential. After the removal of the endothelium, EFS, in the absence of atropine, prazosin and propranolol, elicited depolarization of the longitudinal smooth muscle, proving that collagenase treatment does not damage nerves and smooth muscles.
RNA isolation and reverse transcriptase-PCR (RT-PCR)
The expression of P2X receptor and P2Y receptor gene mRNA was assessed by RT-PCR. Total cellular RNA was extracted from tissue homogenates using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). First-strand cDNA was synthesized from 2 µg of total RNA by using Superscript III RNase H- reverse transcriptase (Invitrogen) and Random primers (Invitrogen). The absence of PCR-amplified DNA fragments in the samples without reverse transcription indicated the isolation of RNA free from genomic DNA contamination.
The PCR were performed with platinum Taq DNA polymerase (Invitrogen). The primer sets were as follows: P2X1 receptor sense 5′-ACT GGT GCG GAA CAA GAA GG-3′ and anti-sense 5′- TCA CAG TTC CAG TCG ATG GT -3′ (predicted size = 720 bp); P2X4 receptor sense 5′- ACA GAT TCC GTG GTC AGT TC -3′ and anti-sense 5′- CAA ATG TTG TTC TTT ACC AG -3′ (predicted size = 413 bp); P2X5 receptor sense 5′- GTG TCC TGG AAG GGT TTA T-3′ and anti-sense 5′- GAA GTG GTT TCT TGG GCT TG -3′ (predicted size = 535 bp); P2Y1 receptor sense 5′- CAT CTC TGC TGC TCT GAA CG -3′ and anti-sense 5′- TGG TTT TGT TCC TCC TCA CC -3′ (predicted size = 551 bp); P2Y5 receptor sense 5′- TAA GCT CTA ACT GCT CCA CT -3′ and anti-sense 5′- TCA GGG TAA TGT TAT AAG GC -3′ (predicted size = 735 bp); P2Y6 receptor sense 5′- TGG TCT ACT CAG TGG TGT TC -3′ and anti-sense 5′- AGT GAT GGT CAA CGT GAT GC -3′ (predicted size = 506 bp); β-actin sense 5′- TGA CCC TGA AGT ACC CCA TTG -3′ and anti-sense 5′- TCA GGA TCT TCA TGA GGT AG -3′ (predicted size = 387 bp); myosin alkali light chain (MLC) sense 5′- AGC CAT GTG CGA TTT CTC GG -3′ and anti-sense 5′- TCA GCC GCT CAG CAC CAT CC -3′ (predicted size = 460 bp); and platelet/endothelial cell adhesion molecule 1 (PECAM1) sense 5′- TAG TAC CGA TCA GGA ACA CC -3′ and anti-sense 5′- TTC TGT ACA GGA GCT CTA TG -3′ (predicted size = 631 bp). All primers were purchased from Invitrogen. The amplifications were performed by 30 cycles. The reaction products were electrophoresed on 1% agarose gels and stained with ethidium bromide (0.2 µg·mL−1). The gels were exposed to UV light with a UV transiluminator (UVP Laboratory Products, Upland, CA, USA), and the images were quantified using Scion Images NIH software.
Statistics
Data are expressed as means ± SEM. Only one preparation of mesenteric artery was isolated per chicken, and thus n indicates the number of animals used. Statistical analysis was performed with Student's unpaired t-test, and P-values <0.05 were considered to be statistically significant.
Materials
The following drugs were used in the present study: ACh, atropine sulphate monohydrate, collagenase, guanethidine sulphate, suramin sodium, cibacron blue F3GA 1-amino-4-[[4-[[4-chloro-6-[[3(or 4)-sulphophenyl]-amino-1,3,5-triazin-2-yl]amino]-3-sulphophenyl]-amino-9,10-dihydro-9,10-dioxo-2-anthracene sulphonic acid (CBF3GA), prazosin hydrochloride, pyridoxalphosphate-6-azophenyl-2', 4'-disulphonic acid (PPADS), α,β-methylene ATP (α,β-MeATP) lithium salt, tetrodotoxin (TTX), dl-propranolol hydrochloride, and Nω-nitro-L-arginine methyl ester (L-NAME), 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one (ODQ), and sodium nitroprusside dihydrate (SNP) (Sigma, St Louis, MO, USA). Prazosin and ODQ were dissolved in methanol and dimethyl sulphoxide, respectively. All other drugs were dissolved in distilled water. Drugs were serially diluted in the PSS just before the experiments and were applied at required concentrations by addition to superfusing PSS.
Results
Resting membrane potentials and EFS-evoked responses
The mean resting membrane potentials from the longitudinal smooth muscle cells of the mesenteric artery in 5- and 12-week-old chickens were −35.2 ± 2.3 mV (n= 30) and −34.7 ± 3.2 mV (n= 20), respectively. In preparations obtained from 12-week-old chickens, EFS induced a prominent depolarization response (Figure 1B), confirming our previous report (Khalifa et al., 2005). In contrast, in preparations obtained from 5-week-old chickens, EFS evoked hyperpolarization (Figure 1A). The inhibitory or excitatory membrane responses at 5 and 12 weeks of age, respectively, were totally blocked by TTX (0.3 µM, n= 4) or guanethidine (5 µM, n= 4) (Figure 1C,D), indicating that they are neurogenic and mediated by the activation of sympathetic nerve axons.
Figure 1.
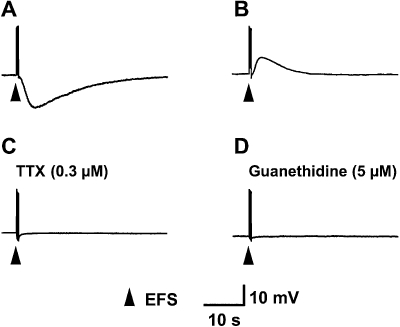
Effects of EFS on membrane responses of longitudinal smooth muscle of the chicken mesenteric artery. EFS-evoked hyperpolarization and depolarization in 5-week-old (A) and in 12-week-old (B) chickens, respectively. Resting membrane potentials for A and B were −38 and −36 mV, respectively. Similar results were obtained in six independent experiments. The EFS-evoked hyperpolarization at 5 weeks of age was blocked by TTX (0.3 µM, n= 4) (C) or guanethidine (5 µM, n= 4) (D). EFS, electrical field stimulation; TTX, tetrodotoxin.
Effects of suramin, PPADS and CBF3GA on EFS-evoked responses
As the NANC components that regulate longitudinal smooth muscles of the anterior mesenteric artery in mature chickens (10–14 weeks old) have already been clarified (Khalifa et al., 2005), we focused on those in 5-week-old chickens. To investigate whether the NANC hyperpolarizing response is because of the effect of ATP, the non-specific purinoceptor antagonist suramin was employed. Suramin (500 µM) completely abolished the EFS-evoked response (Figure 2). The drug did not cause any changes in the resting membrane potential (−35.2 ± 1.6 mV, n= 5).
Figure 2.
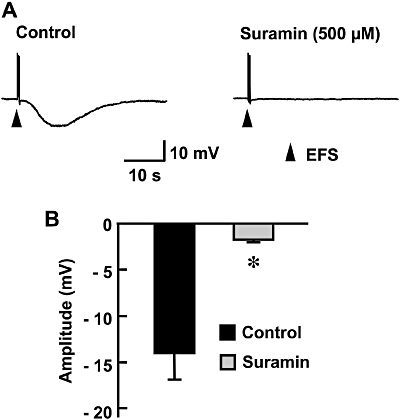
Effect of suramin on EFS-evoked hyperpolarization in the longitudinal smooth muscle of the chicken mesenteric artery. (A) Typical recordings of EFS-evoked responses in the absence (control) or presence of suramin (500 µM) are shown. (B) Peak amplitudes of EFS-evoked hyperpolarization are summarized as means ± SEM (n= 5). *Significantly different from control values (P < 0.05). EFS, electrical field stimulation.
The application of the P2X purinoceptor blocker PPADS (50 µM) significantly inhibited the EFS-evoked hyperpolarization (Figure 3A,B) without affecting the resting membrane potential (−32.1 ± 1.3 mV, n= 10). On the other hand, the application of a P2Y purinoceptor blocker, CBF3GA (100 µM), did not affect the EFS-evoked hyperpolarization (n= 5). This concentration of CBF3GA has been shown to be specific for P2Y purinoceptors (Burnstock and Warland, 1987b) and was sufficient to significantly inhibit the depolarization in preparations obtained from 12-week-old chickens, being consistent with results of our previous study (Khalifa et al., 2005). This concentration CBF3GA did not change the resting membrane potential.
Figure 3.
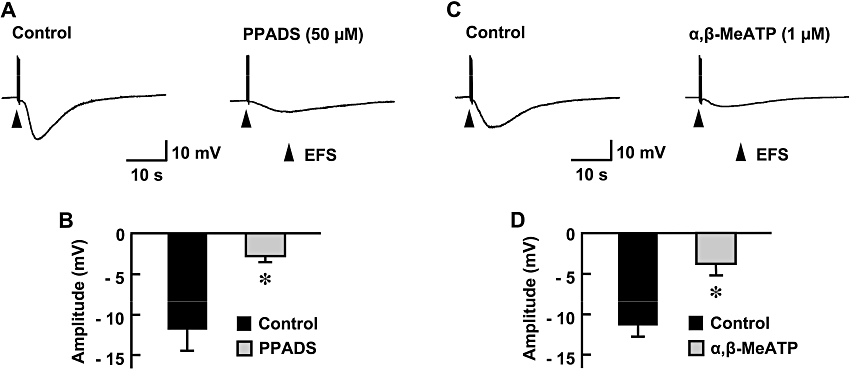
Effect of P2X purinoceptor blockade on EFS-evoked hyperpolarization in the longitudinal smooth muscle of the chicken mesenteric artery. The P2X purinoceptor was blocked by the specific blocker PPADS (50 µM) or by desensitization with the agonist α,β-MeATP (1 µM). Typical recordings of EFS-evoked responses in the absence (control) or presence of PPADS (A) and those in the absence (control) or presence of α,β-MeATP (C) are shown. Peak amplitudes of EFS-evoked hyperpolarization are summarized as means ± SEM (B, n= 10; D, n= 8). *Significantly different from corresponding control values (P < 0.05). EFS, electrical field stimulation; α,β-MeATP, α,β-methylene ATP; PPADS, pyridoxal phosphate-6-azophenyl-2′,4′-disulphonic acid.
Effects of desensitization of P2X purinoceptors on EFS-evoked responses
PPADS is generally recognized as a P2X purinoceptor antagonist, but in some mammalian cells, it also acts on P2Y purinoceptors (Ralevic and Burnstock, 1998). To confirm the contribution of the P2X purinoceptor to purinergic control of the longitudinal smooth muscle, desensitization of the P2X purinoceptor was carried out. Incubation of preparations obtained from 5-week-old chickens with a P2X purinoceptor agonist, α,β-MeATP (1 µM), for 30 min significantly suppressed the EFS-evoked hyperpolarization (Figure 3C,D) without affecting the resting membrane potential (−36.3 ± 1.3 mV, n= 8).
Effect of endothelium removal
Endothelium removal with collagenase did not affect the resting membrane potential. The EFS-evoked hyperpolarization disappeared after endothelium removal (n= 5, Figure 4), suggesting a dominant role of the endothelium in the EFS-evoked hyperpolarization.
Figure 4.
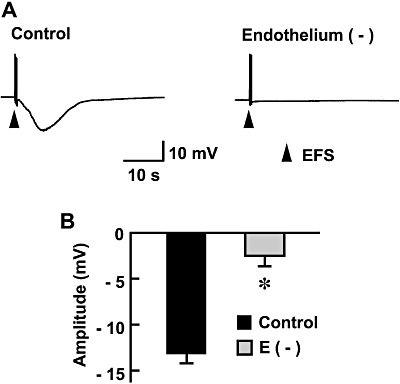
Effect of removal of the endothelium of the anterior mesenteric artery on EFS-evoked hyperpolarization in the longitudinal smooth muscle. The endothelium was removed by injecting collagenase (1 mg·mL−1) into the lumen of the artery for 15 min. (A) Typical recordings of EFS-evoked responses in the endothelium-intact (control) or endothelium-removed preparations are shown. (B) Peak amplitudes of EFS-evoked hyperpolarization are summarized as means ± SEM (n= 5). *Significantly different from control values (P < 0.05). EFS, electrical field stimulation.
Involvement of NO in EFS-evoked hyperpolarization
Endothelial cells are known to release inhibitory factors, including NO (Arnal et al., 1999). We therefore examined the involvement of NO in the EFS-evoked hyperpolarization. The application of L-NAME (100 µM), an NOS inhibitor, significantly suppressed the amplitude of EFS-evoked hyperpolarization (Figure 5) without affecting the resting membrane potential (−34 mV, n= 5). Hyperpolarization of the longitudinal smooth muscle was reproduced by exogenous application of an NO donor, SNP (10 µM; n= 5), not only in endothelium-intact but also in endothelium-removed preparations (Figure 6A,B).
Figure 5.
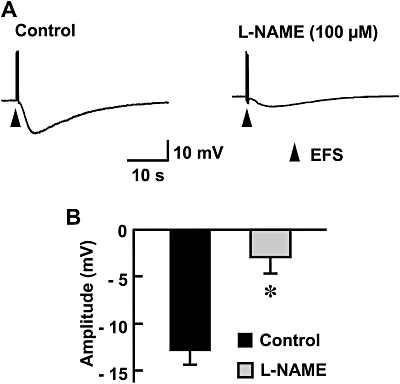
Effect of the NOS inhibitor L-NAME on EFS-evoked hyperpolarization in the longitudinal smooth muscle of the chicken mesenteric artery. (A) Typical recordings of EFS-evoked responses in the absence (control) or presence of L-NAME (100 µM) are shown. (B) Peak amplitudes of EFS-evoked hyperpolarization are summarized as means ± SEM (n= 5). *Significantly different from control values (P < 0.05). EFS, electrical field stimulation; L-NAME, Nω-nitro-L-arginine methyl ester; NOS, nitric oxide synthase.
Figure 6.
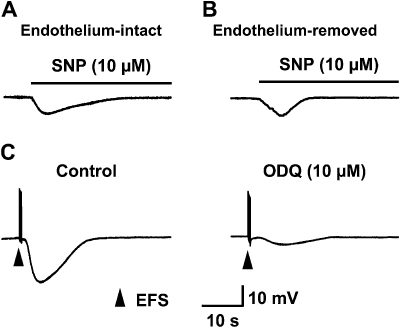
Action of the NO donor SNP and effect of the GC inhibitor ODQ on EFS-evoked hyperpolarization in the longitudinal smooth muscle of the chicken mesenteric artery. Typical recordings of membrane response after exogenous application of the NO donor SNP (10 µM) in endothelium-intact (A) and endothelium-removed (B) preparations are shown. (C) Typical recordings of EFS-evoked responses in the absence (control) or presence of ODQ (10 µM) are shown. Similar results were obtained in five independent experiments. EFS, electrical field stimulation; NO, nitric oxide; ODQ, 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one.
We then examined the involvement of soluble guanylate cyclase (sGC) in hyperpolarization of the longitudinal smooth muscle. As shown in Figure 6C, pretreatment with an sGC inhibitor, ODQ, (10 µM), significantly attenuated the EFS-induced hyperpolarization (n= 5).
Molecular identification of purinoceptors in the chicken anterior mesenteric artery by RT-PCR
We then examined the expression of purinoceptor isoforms in the chicken anterior mesenteric artery. As shown in Figure 7A, amplified products of P2X1, P2X4, P2X5, P2Y1, P2Y5 and P2Y6 were observed in appropriate sizes when homogenized chicken anterior mesenteric artery with intact endothelium was used for RT-PCR. On the other hand, P2X1, P2X4, P2X5 and P2Y1 isoforms were not detected in the tissue without endothelium, although P2Y5 and P2Y6 isoforms were amplified (Figure 7B). Efficacy of the endothelium removal was confirmed by amplifying the endothelium-specific protein PECAM1 (Newman, 1994) and smooth muscle-specific MLC (Nabeshima et al., 1987). An amplified product of PECAM1 was not observed in the endothelium-removed samples, whereas MLC was found both in endothelium-intact and endothelium-removed samples (Figure 7).
Figure 7.
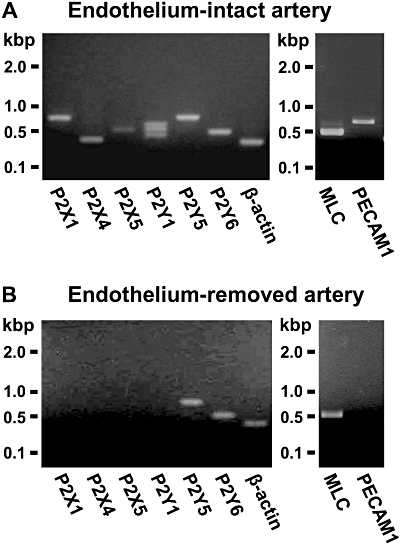
Identification of purinoceptor isoforms in the chicken anterior mesenteric artery by RT-PCR. Homogenized samples of the chicken anterior mesenteric artery with intact endothelium (A) or those without endothelium (B) were used for RT-PCR. Amplified products of P2X1, P2X4, P2X5, P2Y1, P2Y5 and P2Y6 purinoceptors were observed in appropriate sizes in the endothelium-intact artery (A). P2Y5 and P2Y6 isoforms, but not P2X1, P2X4, P2X5 and P2Y1 isoforms, were amplified in the tissue without endothelium (B). Efficacy of endothelium removal was confirmed by amplifying the endothelium-specific protein PECAM1 (platelet/endothelial cell adhesion molecule 1) and smooth muscle-specific MLC. An amplified product of PECAM1 was not observed in the endothelium-removed samples, whereas MLC was found both in endothelium-intact and endothelium-removed samples. Similar results were obtained in five independent experiments. MLC, myosin alkali light chain; RT-PCR, reverse transcriptase-PCR.
Discussion
It has been previously demonstrated that an activation of perivascular purinergic nerves causes an excitatory membrane response (i.e. depolarization) in longitudinal smooth muscles of the anterior mesenteric artery in mature chickens (10–14 weeks old) (Khalifa et al., 2005). In this study, we examined the purinergic neuronal regulation by using younger chickens. The major findings of this study are (i) the EFS of preparations obtained from young chickens (5 weeks old) induced hyperpolarization but not depolarization; (ii) the EFS-evoked hyperpolarization in 5-week-old chickens was blocked by the non-specific purinoceptor antagonist suramin and by the specific P2X purinoceptor antagonist PPADS; (iii) desensitization of the P2X purinoceptor with its agonist α,β-MeATP significantly suppressed EFS-evoked hyperpolarization; (iv) blockade of the P2Y purinoceptor did not affect EFS-evoked hyperpolarization; (v) the removal of the endothelium or the application of the NOS inhibitor L-NAME to endothelium-intact preparations inhibited the hyperpolarization; (vi) the EFS-evoked hyperpolarization was mimicked by an NO donor, SNP, and was inhibited by an sGC inhibitor, ODQ; and (vii) P2X purinoceptors are expressed in the endothelium in the anterior mesenteric artery. These findings, together with the results of our previous study (Khalifa et al., 2005), demonstrate that the purinergic neuronal component that regulates longitudinal smooth muscles of the anterior mesenteric artery varies depending on the age of the chicken. In young chickens, ATP released from perivascular nerves acts on P2X purinoceptors in endothelium and thereby stimulates NO production. NO-mediated inhibitory regulation precedes the establishment of excitatory neuromuscular transmission in mature chickens.
In the present experiment, EFS under NANC conditions caused an inhibitory membrane response, that is, hyperpolarization in the longitudinal smooth muscle of the anterior mesenteric artery in young chickens (5 weeks old). This was in contrast to EFS-induced responses in mature chickens (10–14 weeks old), in which depolarization was observed (Figure 1 and Khalifa et al., 2005). Both the hyperpolarization in young chickens and the depolarization in mature animals were completely abolished by suramin. Suramin is generally recognized as a non-specific purinoceptor antagonist, but it also inhibits γ-aminobutyric acid and glutamate receptor channels (Nakazawa et al., 1995; Peoples and Li, 1998). However, the contribution of these receptor channels, if any, seems to be minor because EFS-induced membrane responses of the smooth muscles were also blocked by specific purinoceptor antagonists or by desensitization with specific purinoceptor agonists (Draid et al., 2005; Khalifa et al., 2005). Taken together, our results suggest that both the responses observed in young and mature chickens are commonly mediated by neurally released ATP. Therefore, it is reasonable to assume that purinergic regulation in the longitudinal smooth muscle shifts from inhibitory to excitatory during the growth of chickens.
In preparations in which the endothelium was removed, EFS failed to induce hyperpolarization (Figure 4), indicating that the inhibitory membrane response is endothelium dependent. It is known that several inhibitory mediators released from the endothelium diffuse to and act on smooth muscle cells, thereby causing hyperpolarization (Furchgott and Vanhoutte, 1989). In the longitudinal smooth muscle of the anterior mesenteric artery in young chickens, NO is the most probable mediator responsible for the endothelium-dependent hyperpolarization. This is based on our finding that EFS-induced hyperpolarization was significantly suppressed by treatment of the preparations with L-NAME, a NOS inhibitor (Figure 5), and the fact that it was reproduced by exogenous application of NO (Figure 6A,B). The mechanism by which NO caused hyperpolarization is thought to be the activation of sGC because the sGC inhibitor ODQ inhibited EFS-induced hyperpolarization (Figure 6C). In our pervious study, in which membrane responses were recorded from the circular smooth muscle cells of this artery, endothelium-dependent hyperpolarization was not blocked by L-NAME (Draid et al., 2005). These results indicate that the target muscle layer of the endothelium-derived inhibitory mediator is clearly separated. This may be beneficial for controlling blood flow of the mesenteric artery. NO generated at the endothelium needs to pass across the circular muscle layer before it reaches and exerts its action on longitudinal smooth muscles. If the circular smooth muscle also responds to NO in a manner similar to that of the longitudinal one, the timing of smooth muscle dilation of these layers cannot be controlled separately. The presence of the longitudinal muscle layer is unique to this artery, and it may contribute to enhancement of the capability for regional control of blood flow (Bell, 1969). Therefore, operation of specific neural control on the longitudinal smooth muscle is reasonable.
It is generally accepted that P2Y purinoceptors are involved in endothelium-dependent membrane responses of smooth muscles in the artery (Pearson and Carter, 1990). In agreement with this, we previously demonstrated that the hyperpolarizing effect of ATP on circular muscle cells in the chicken anterior mesenteric artery is mediated by the P2Y purinoceptor (Draid et al., 2005). However, blocking of the P2Y purinoceptor with a specific antagonist did not inhibit the suramin-sensitive hyperpolarization in longitudinal smooth muscle cells. In contrast, blocking of P2X purinoceptors by using an antagonist or agonist-induced receptor desensitization significantly inhibited the hyperpolarization. These results indicated that P2X, but not P2Y, purinoceptors mediated the action of ATP. There are a few reports in which P2X purinoceptor-mediated, endothelium-dependent vasodilatation is described. In the mesenteric artery of rats and mice, the response is mediated by P2X1 purinoceptors (Harrington and Mitchell, 2004; Harrington et al., 2007). In contrast, Yamamoto et al. (2006) have shown that the P2X4 purinoceptor plays a key role in endothelial NO production. In our study, RT-PCR analysis showed that both P2X1 and P2X4 purinoceptors were expressed in the endothelium of the chicken anterior mesenteric artery. P2X1 purinoceptors possess sensitivity to α,β-MeATP, PPADS and suramin, while P2X4 purinoceptors are much less sensitive to these drugs (Ralevic and Burnstock, 1998). As pharmacological blockade using these drugs abolished the response, the P2X1 purinoceptor may be responsible for the endothelium-dependent hyperpolarization in the chicken mesenteric artery.
P2Y purinoceptors were detected in the endothelium-removed anterior mesenteric artery of 5-week-old chickens (Figure 7), suggesting that the receptors are expressed in the smooth muscles. As direct action of ATP on P2Y purinoceptors in the longitudinal smooth muscle cells of this artery, at least in 12-week-old chickens, was depolarization (Figure 1 and Khalifa et al., 2005), it is possible that a large hyperpolarization caused by endothelium-derived NO overlaps with P2Y-mediated depolarization, thereby hiding the depolarizing response. However, this is unlikely because we failed to observe depolarization even in the presence of blockers of P2X purinoceptors or NOS. A possible explanation for the lack of depolarizing response is that purinergic innervation to the longitudinal smooth muscle has not yet been established in chickens at this age. According to this assumption, the age-dependent shift of membrane responses to purinergic stimuli would be brought about by the overlapping of depolarization response, which becomes dominant in mature chickens, to hyperpolarization. It should be noted, however, that samples used for RT-PCR were not separated into individual muscle layers. Therefore, an alternative explanation is that P2Y purinoceptors were not expressed in the longitudinal smooth muscle cells but in the circular smooth muscle cells. In this case, the shift can be simply explained by the timing of expression of P2Y purinoceptors in the longitudinal smooth muscle cells. Further study is needed to clarify the precise mechanisms responsible for the age-dependent shift of purinergic regulation.
Bell (1969) demonstrated that contraction of the longitudinal smooth muscle had little effect on perfusion pressure in the isolated chicken mesenteric artery. If the longitudinal smooth muscle affects vascular tone, the experimentally induced contraction would be accompanied by changes in perfusion pressure. Therefore, the longitudinal smooth muscle would play a minor role, if any, in the regulation of vascular tone. However, contraction of the longitudinal smooth muscle potentiates perfusion pressure responses to vasoconstrictor stimuli directly affecting circular muscle (Bell, 1969), suggesting that the longitudinal smooth muscle may serve to modulate the response of the artery to vasomotor stimuli. As significant changes occur in intestinal haemodynamics during growth (Nankervis et al., 2001), adjustment of blood flow may be essential. Rationally, it is expected that modulatory roles of the longitudinal muscle would also be changed. We hypothesize that the age-dependent shift of purinergic neuronal control from inhibitory to excitatory in the longitudinal smooth muscle is related to age-dependent changes in intestinal demand for blood supply. A similar age-dependent change in endothelium-mediated vasodilator response has been reported by Hasegawa et al. (1993), who demonstrated that angiotensin II-induced vasorelaxation is marked in young domestic fowls but diminishes in adult birds. Alternatively, it is also possible that the longitudinal smooth muscle confers resistance to tension applied to blood vessels. In fact, the longitudinal muscle layer of the posterior mesenteric artery of female chickens develops remarkably when they begin egg production (unpublished observation). If age-dependent gain in intestinal weight causes an increase in mechanical tension in the anterior mesenteric artery, age-dependent shift of purinergic neuronal control is reasonable.
In conclusion, the present results demonstrate that activation of a purinergic neuronal component in the anterior mesenteric artery of young chickens evokes hyperpolarization in the longitudinal smooth muscle of the artery. ATP released from perivascular nerves acts on P2X purinoceptors in the endothelium and thereby stimulates NO production. The NO-mediated inhibitory regulation precedes the establishment of excitatory purinergic regulation that is dominantly expressed in mature chickens. The results of the present study provide an insight into the role of ATP in the regulation of smooth muscle layers in the mesenteric artery in birds.
Acknowledgments
This work was supported by a Grant-in-Aid Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Glossary
Abbreviations:
- α,β-MeATP
α,β-methylene ATP
- CBF3GA
cibacron blue F3GA
- EFS
electrical field stimulation
- MLC
myosin alkali light chain
- ODQ
1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one
- PECAM1
platelet/endothelial cell adhesion molecule 1
- PPADS
pyridoxal phosphate-6-azophenyl-2′,4′-disulphonic acid
- PSS
physiological salt solution
- RT-PCR
reverse transcriptase-PCR
- SNP
sodium nitroprusside
- TTX
tetrodotoxin
Statement of conflict of interest
None.
References
- Alexander SPH, Mathie A, Peters JA. Guide to receptors and channels (GRAC), 3rd edn. Br J Pharmacol. 2008;153(Suppl. 2):S1–S209. doi: 10.1038/sj.bjp.0707746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnal JF, Dinh-Xuan AT, Pueyo M, Darblade B, Rami J. Endothelium-derived nitric oxide and vascular physiology and pathology. Cell Mol Life Sci. 1999;55:1078–1087. doi: 10.1007/s000180050358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ball RA, Sautter JH, Katter MS. Morphological characteristics of the anterior mesenteric artery of fowl. Anat Rec. 1963;146:251–255. doi: 10.1002/ar.1091460311. [DOI] [PubMed] [Google Scholar]
- Bell C. Indirect cholinergic vasomotor control of intestinal blood flow in the domestic chicken. J Physiol. 1969;205:317–327. doi: 10.1113/jphysiol.1969.sp008967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton TB. Studies on the longitudinal muscle of the anterior mesentric artery of the domestic fowl. J Physiol. 1968;196:273–281. doi: 10.1113/jphysiol.1968.sp008506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton TB. Spontaneous and evoked release of neurotransmitter substances in the longitudinal muscle of anterior mesenteric artery of the domestic fowl. Br J Pharmacol. 1969;35:112–120. doi: 10.1111/j.1476-5381.1969.tb07971.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnstock G. Purines as co-transmitters in adrenergic and cholinergic neurons. In: Hokfelt T, Fuxe K, Pernow B, editors. Coexistance of Neuronal Messengers: A New Principle in Chemical Transmission. Amsterdam: Elsevier; 1986. pp. 193–203. [Google Scholar]
- Burnstock G. Purine and pyrimidine receptors. Cell Mol Life Sci. 2007;64:1471–1483. doi: 10.1007/s00018-007-6497-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnstock G, Warland JJ. A pharmacological study of the rabbit saphenous artery in vitro: a vessel with a large purinergic contractile response to sympathetic nerve stimulation. Br J Pharmacol. 1987a;90:111–120. doi: 10.1111/j.1476-5381.1987.tb16830.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnstock G, Warland JJ. P2-purinoceptors of two subtypes in the rabbit mesenteric artery: reactive blue 2 selectively inhibits responses mediated via the P2y-but not the P2x-purinoceptor. Br J Pharmacol. 1987b;90:383–391. doi: 10.1111/j.1476-5381.1987.tb08968.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Draid M, Shiina T, El-Mahmoudy A, Boudaka A, Shimizu Y, Takewaki T. Neurally released ATP mediates endothelium-dependent hyperpolarization in the circular smooth muscle cells of chicken anterior mesenteric artery. Br J Pharmacol. 2005;146:983–989. doi: 10.1038/sj.bjp.0706413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furchgott RF. The pharmacology of vascular smooth muscle. Pharmacol Rev. 1955;7:183–265. [PubMed] [Google Scholar]
- Furchgott RF, Vanhoutte PM. Endothelium-derived relaxing and contracting factors. FASEB J. 1989;3:2007–2018. [PubMed] [Google Scholar]
- Gooden BA. The effect of hypoxia on vasoconstrictor responses of isolated mesenteric arterial vasculature from chicken and duckling. Comp Biochem Physiol. 1980;67:219–222. doi: 10.1016/0306-4492(80)90022-2. [DOI] [PubMed] [Google Scholar]
- Harrington LS, Evans RJ, Wray J, Norling L, Swales KE, Vial C, et al. Purinergic 2X1 receptors mediate endothelial dependent vasodilation to ATP. Mol Pharmacol. 2007;72:1132–1136. doi: 10.1124/mol.107.037325. [DOI] [PubMed] [Google Scholar]
- Harrington LS, Mitchell JA. Novel role for P2X receptor activation in endothelium-dependent vasodilation. Br J Pharmacol. 2004;143:611–617. doi: 10.1038/sj.bjp.0706004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa K, Nishimura H, Khosla MC. Angiotensin II-induced endothelium-dependent relaxation of fowl aorta. Am J Physiol. 1993;264:R903–R911. doi: 10.1152/ajpregu.1993.264.5.R903. [DOI] [PubMed] [Google Scholar]
- Keatinge WR. Electrical and mechanical response of arteries to stimulation of sympathetic nerves. J Physiol. 1966;185:701–715. doi: 10.1113/jphysiol.1966.sp008011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy C, Burnstock G. Evidence for two types of P2-purinoceptor in longitudinal muscle of the rabbit portal vein. Eur J Pharmacol. 1985;111:49–56. doi: 10.1016/0014-2999(85)90112-8. [DOI] [PubMed] [Google Scholar]
- Khalifa M, El-mahmoudy A, Shiina T, Shimizu Y, Nikami H, El-Sayed M, et al. An electrophysiological study of excitatory purinergic neuromuscular transmission in longitudinal smooth muscle of chicken anterior mesenteric artery. Br J Pharmacol. 2005;144:830–839. doi: 10.1038/sj.bjp.0706076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaren GJ, Kennedy C, Sneddon P. The effects of suramin on purinergic and noradrenergic neurotransmission in the rat isolated tail artery. Eur J Pharmacol. 1995;277:57–61. doi: 10.1016/0014-2999(95)00065-s. [DOI] [PubMed] [Google Scholar]
- Nabeshima Y, Nabeshima Y, Nonomura Y, Fujii-Kuriyama Y. Nonmuscle and smooth muscle myosin light chain mRNAs are generated from a single gene by the tissue-specific alternative RNA splicing. J Biol Chem. 1987;262:10608–10612. [PubMed] [Google Scholar]
- Nakazawa K, Inoue K, Ito K, Koizumi S, Inoue K. Inhibition by suramin and reactive blue 2 of GABA and glutamate receptor channels in rat hippocampal neurons. Naunyn Schmiedebergs Arch Pharmacol. 1995;351:202–208. doi: 10.1007/BF00169334. [DOI] [PubMed] [Google Scholar]
- Nankervis CA, Reber KM, Nowicki PT. Age-dependent changes in the postnatal intestinal microcirculation. Microcirculation. 2001;8:377–387. doi: 10.1038/sj/mn/7800110. [DOI] [PubMed] [Google Scholar]
- Newman PJ. The role of PECAM-1 in vascular cell biology. Ann N Y Acad Sci. 1994;714:165–174. doi: 10.1111/j.1749-6632.1994.tb12041.x. [DOI] [PubMed] [Google Scholar]
- Orre M, Pennefather JN, Story ME, Haynes JM. The effects of P2 purinoceptor agonists on the isolated portal vein of the guinea pig. Eur J Pharmacol. 1996;316:229–236. doi: 10.1016/s0014-2999(96)00687-5. [DOI] [PubMed] [Google Scholar]
- Pearson JD, Carter TD. Effects of extracellular ATP on the release of vasoactive mediators from endothelium. Ann N Y Acad Sci. 1990;603:267–273. doi: 10.1111/j.1749-6632.1990.tb37678.x. [DOI] [PubMed] [Google Scholar]
- Peoples RW, Li C. Inhibition of NMDA-gated ion channels by the P2 purinoceptor antagonists suramin and reactive blue 2 in mouse hippocampal neurones. Br J Pharmacol. 1998;124:400–408. doi: 10.1038/sj.bjp.0701842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralevic V, Burnstock G. Receptors for purines and pyrimidines. Pharmacol Rev. 1998;50:413–492. [PubMed] [Google Scholar]
- Rummery NM, Brock JA, Pakdeechote P, Ralevic V, Dunn WR. ATP is the predominant sympathetic neurotransmitter in rat mesenteric arteries at high pressure. J Physiol. 2007;582:745–754. doi: 10.1113/jphysiol.2007.134825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smyth L, Bobalova J, Ward SM, Keef KD, Mutafova-Yambolieva VN. Cotransmission from sympathetic vasoconstrictor neurons: differences in guinea-pig mesenteric artery and vein. Auton Neurosci. 2000;86:18–29. doi: 10.1016/S1566-0702(00)00203-4. [DOI] [PubMed] [Google Scholar]
- Takewaki T, Ohashi O. Non-cholinergic excitatory transmission to intestinal smooth muscle cells. Nature. 1977;268:749–750. doi: 10.1038/268749a0. [DOI] [PubMed] [Google Scholar]
- Thapaliya S, Matsuyama H, Takewaki T. ATP released from perivascular nerves hyperpolarizes smooth muscle cells by releasing an endothelium-derived factor in hamster mesenteric arteries. J Physiol. 1999;521:191–199. doi: 10.1111/j.1469-7793.1999.00191.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Triggle CR, Ding H. Endothelium-derived hyperpolarizing factor: is there a novel chemical mediator? Clin Exp Pharmacol Physiol. 2002;29:153–160. doi: 10.1046/j.1440-1681.2002.03632.x. [DOI] [PubMed] [Google Scholar]
- Yamamoto K, Sokabe T, Matsumoto T, Yoshimura K, Shibata M, Ohura N, et al. Impaired flow-dependent control of vascular tone and remodeling in P2X4-deficient mice. Nat Med. 2006;12:133–137. doi: 10.1038/nm1338. [DOI] [PubMed] [Google Scholar]


