Summary
The conducting airways (bronchi and bronchioles) and peripheral gas exchange (alveolar) regions of the mammalian lung are generated by a process of branching morphogenesis. Evidence suggests that during embryonic development, the undifferentiated epithelial progenitors are located at the distal tips of the branching epithelium. To test this hypothesis, we used an Id2-CreERT2 knock-in mouse strain to lineage trace the distal epithelial tip cells during either the pseudoglandular or canalicular phases of development. During the pseudoglandular stage, the tip cells both self-renew and contribute descendents to all epithelial cell lineages, including neuroendocrine cells. In addition, individual Id2+ tip cells can self-renew and contribute descendents to both the bronchiolar and alveolar compartments. By contrast, during the later canalicular stage, the distal epithelial tip cells only contribute descendents to the alveoli. Taken together, this evidence supports a model in which the distal tip of the developing lung contains a multipotent epithelial population, the fate of which changes during development.
Keywords: Progenitor cells, Cre, Lineage tracing, Lung development, Epithelium
INTRODUCTION
Mouse lung development involves four histological stages (Cardoso and Lu, 2006). During the pseudoglandular stage [embryonic day (E) 10.5-16.5], the bronchiolar tree is laid down by branching morphogenesis (Metzger et al., 2008) and the epithelium begins to differentiate into secretory (Clara), neuroendocrine (NE), or ciliated cells. During the canalicular stage (E16.5-17.5), the undifferentiated distal epithelium continues to branch and gives rise to terminal sacs. These increase in number during the terminal sac stage [E17.5-postnatal day (P) 5], and type 1 and 2 alveolar epithelial cells begin to differentiate. During the alveolar stage (P5-30), the terminal sacs develop into mature alveoli.
Lineage tracing in the developing pancreas has shown that the distal tip epithelium comprises a population of multipotent progenitors (Zhou et al., 2007). There is evidence for a similar organization in the developing lung. For example, the distal tip cells have a higher proliferation index than more proximal epithelium (Okubo et al., 2005) and a distinct gene expression profile, including high levels of Etv5, Nmyc (Mycn - Mouse Genome Informatics), Id2, Sox9, Foxp1 and Foxp2 and high activities of the Wnt, Bmp and Fgf signaling pathways (reviewed by Rawlins, 2008). Loss- or gain-of-function phenotypes of a number of these genes are consistent with an effect on an epithelial progenitor population. For example, loss of Wnt7b, which is expressed in the distal epithelium, results in lungs that are small, but more or less normally patterned (Rajagopal et al., 2008). Results of lineage tracing in the developing lung, using a transgene driven by a regulatory region of the human SFTPC gene, are consistent with the presence of a multipotent epithelial population, but suggest that it does not include progenitors of NE cells (Perl et al., 2002).
Here, we use a different lineage-tracing strategy, based on the restricted expression of Id2 (inhibitor of differentiation 2; also known as inhibitor of DNA binding 2) in tip epithelial cells (Jen et al., 1996; Liu and Hogan, 2002). We have generated a new CreERT2 knock-in allele that allows us to follow the fate of these cells, including individual cells, at different times during lung development. This strategy, using the endogenous Id2 enhancer region, rather than a transgene, allowed us to lineage label a well-defined cell population. Our results show that the tip population is multipotent and, moreover, does gives rise to the NE lineage. However, we find no evidence that this Id2+ progenitor population is retained in adult lungs.
MATERIALS AND METHODS
Mouse strains
A 9.8 kb region of the 129/SvEv Id2 locus was used to generate a targeting vector with a CreERT2 or eGFP cassette inserted at the translation initiation codon replacing most of exon 1, except the final 25 nucleotides. Id2-CreERT2 [Id2tm1(CRE/ESR1)Blh], Id2-eGFP [Id2tm2(EGFP)Blh], Rosa26R-lacZ [Gt(Rosa)26Sortm1Sor] and Rosa26R-eYFP [Gt(Rosa)26Sortm1(eYFP)Cos] were used at the N3 C57Bl/6 backcross generation. For all experiments, at least three lungs from two litters were examined. Tamoxifen (tmx) (Sigma) was dissolved in Mazola corn oil. Pregnant females received a single intraperitoneal injection of 0.075 mg, 0.1 mg or 2 μg tmx per gram body weight. Adult mice (8-12 weeks old) received 0.25 mg per gram body weight every other day, four times.
Immunohistochemistry, X-Gal staining and in situ hybridization
Lungs were inflated with up to 1 ml 4% paraformaldehyde (PFA), fixed 4 hours (for mice 3-weeks old or older), or 1-2 hours (P1-20) at 4°C, washed, embedded in OCT Compound (Tissue-Tek) and sectioned at 12 μm. Primary antibodies were rat anti-GFP (1:500, Nacalai Tesque), goat anti-Scgb1a1 (1:10,000, kindly provided by Barry Stripp, Duke University, Durham, NC, USA), rabbit anti-SftpC (1:200, Chemicon), mouse anti-β-tubulin (1:1000, BioGenex), mouse anti-acetylated-tubulin (1:3000, Sigma-Aldrich), rabbit anti-CGRP (1:500, Penninsular Labs), hamster anti-T1α (1:1000, Developmental Studies Hybridoma Bank) and rat anti-Pecam (1:200, BD Pharmingen). Alexa-Fluor-coupled secondary antibodies (Invitrogen) were used at 1:500 dilution. Biotinylated secondary antibodies (Jackson ImmunoResearch) were amplified using the Vectastain Elite ABC Kit and detected with DAB (Vector Laboratories). For X-Gal staining, postnatal lungs were inflated with 4% PFA and fixed 1-4 hours; E14.5-18.5 lungs were fixed for 1 hour in 4% PFA; E12.5-13.5 lungs were fixed for 30 minutes in 0.2% glutaraldehyde all at 4°C. X-Gal staining was as described previously (Eblaghie et al., 2006). Lungs were paraffin embedded, or cleared in methyl salicylate (Sigma) for wholemount imaging. In situ hybridization was as described previously (Liu and Hogan, 2002).
Microscopy
Images used to assess antibody co-localization consisted of a z-stack of optical sections captured on a Zeiss LSM 510 Meta laser scanning confocal microscope.
RESULTS AND DISCUSSION
Id2-CreERT2 for lineage tracing
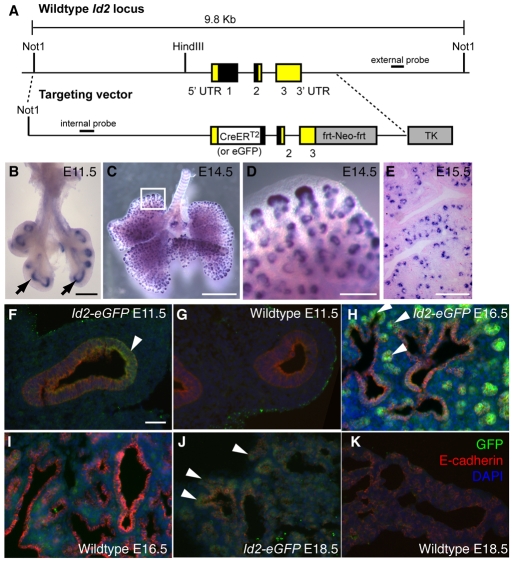
To lineage trace distal tip epithelial cells we used an Id2-CreERT2 knock-in allele (Fig. 1A). Evidence suggests that this recapitulates the epithelial expression of the endogenous wild-type allele. In situ hybridization shows that Id2 is transcribed in the epithelial tip cells of the developing lung throughout branching morphogenesis up to E16.5 (Fig. 1B-E). Immunohistochemistry of sectioned lungs also clearly shows eGFP expression in these cells from the Id2-eGFP allele (Fig. 1F-I). Similar results were obtained using antibodies to Id2 and Cre protein with lungs from wild-type and Id2-CreERT2 animals (see Fig. S1 in the supplementary material). By the terminal sac stage, levels of Id2 were much reduced, making analysis by immunohistochemistry difficult. However, at E18.5, Id2-eGFP+ epithelial cells could still be detected above background, particularly in more distal regions (Fig. 1J,K). At all stages, the expression of Id2 in mesodermal cells was very low. However, some Id2+ and eGFP+ cells were detected in the distal mesenchyme during the pseudoglandular stage (see Fig. S1 in the supplementary material) and some eGFP+ endothelial cells at E18.5 (not shown). For this study, we focus only on the epithelial population.
Fig. 1.
Id2 expression and gene targeting. (A) Targeting vectors. The majority of the mouse Id2 coding region was replaced with a CreERT2 or eGFP cassette. The PGK-Neo cassette (frt-Neo-frt) was removed by FLP-mediated excision after germline transmission (Farley et al., 2000). TK, PGK-thymidine kinase. (B-E) In situ hybridization for Id2 mRNA. (B) E11.5; (C) E14.5; (D) Higher magnification of box in C. (E) E15.5. Id2 is highly expressed in tip epithelium (arrows). (F-K) Id2-eGFP (F,H,J) and wild-type (G,I,K) lungs. (F,G) E11.5; (H,I) E16.5; (J,K) E18.5. Green, anti-GFP; red, anti-E-cadherin (epithelium); blue, DAPI (nuclei). Arrowheads, eGFP+ epithelial tips. Scale bars: 0.25 cm in B; 0.5 cm in C; 200 μm in D,F-K.
In lineage-tracing studies using the CreERT2/loxP system, recombination of the reporter is dependent on the dose of Tamoxifen (tmx) and on the level of Cre expression, and occurs stochastically in a fraction of the Cre-expressing cells (Hayashi and McMahon, 2002). Consequently, high doses of tmx will cause the labeling of many of the cells that express a high level of CreER and only rarely of cells that express low levels of CreER. In each experiment, we initially defined the cell population(s) labeled by tmx and then followed their descendents.
The lungs of Id2eGFP/+ and Id2CreERT2/+ heterozygous mice were indistinguishable from their Id2+/+ siblings (not shown).
Lineage tracing Id2+ distal epithelial cells during early lung development
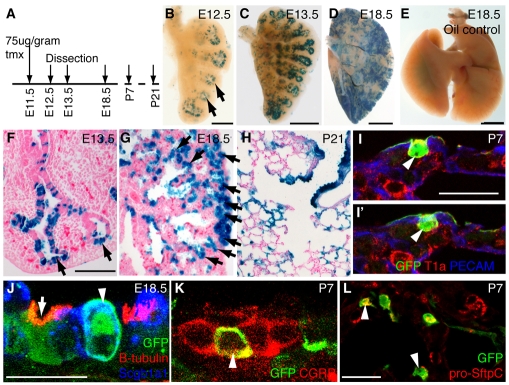
At E11.5, 75 μg tmx per gram mother's body weight resulted in the lineage labeling of a large proportion of the epithelial cells in the distal tips (Fig. 2A,B). There was no reporter activation in lungs exposed to vehicle only (Fig. 2E). Following the descendents of the E11.5 tip population showed that cells contributed to both the bronchioles and alveoli (Fig. 2C,D,F-H). Some lacZ-expressing cells were retained in the tip at E18.5, suggesting that the tip population self-renews during embryogenesis (Fig. 2F,G). We used a Rosa26R-eYFP lineage reporter to demonstrate that all of the major epithelial cell types (Clara, ciliated, NE, type 1, type 2, and SftpC/Scgb1a1 dual-positive cells whose function, if any, is unclear) can be derived from Id2+ tip epithelium labeled at E11.5 (Fig. 2I-L). Note that T1α (Pdpn - Mouse Genome Informatics), a commonly accepted type 1 cell marker, is not specific to type 1 cells in the developing lung (see Fig. S2 in the supplementary material). Similar results were obtained if embryos were exposed to tmx at E10.5 or E12.5 (not shown).
Fig. 2.
Id2+ tip epithelial cells labeled at the pseudoglandular stage contribute descendents to all lineages. (A) Id2-CreERT2; Rosa26R-lacZ or Rosa26R-eYFP mouse embryos were exposed to tmx at E11.5 and sacrificed at intervals. (B-E) X-Gal stained (blue) lungs. (B) E12.5. Epithelial staining is restricted to tips (arrows). Rare mesenchymal cells are also labeled, consistent with a low level of Id2 expression in this population. (C) E13.5. (D) E18.5. Note the staining throughout the epithelium. (E) E18.5 vehicle-only control showing no tmx-independent recombination. (F-H) X-Gal-stained (blue) lung sections. (F) E13.5. (G) E18.5. Staining is prominent in the tip epithelium (arrows). (H) P21. Staining is seen throughout the bronchioles and alveoli. (I,I′) P7. Two optical sections from a confocal stack. Green, anti-GFP, lineage label; red, anti-T1α, type 1 and 2 cells at P7 (see Fig. S2 in the supplementary material); blue, anti-Pecam, endothelium. Arrowhead, labeled type 1 cell, based on morphology and position adjacent to endothelial cell. (J) E18.5 confocal maximum projection. Green, anti-GFP, lineage label; red, anti-β-tubulin, ciliated cells; blue, anti-Scgb1a1, Clara cells. Arrowhead, lineage-labeled Clara cell. Arrow, lineage-labeled ciliated cell. (K) P7 confocal maximum projection. Green, anti-GFP, lineage label; red, anti-CGRP (Calca), neuroendocrine (NE) cells. Arrowhead, lineage-labeled NE cell (n=24 neuroendocrine bodies with one or more GFP+ cells, five lungs scored). (L) P7 confocal maximum projection. Green, anti-GFP, lineage label; red, anti-pro-SftpC, type 2 cells. Arrowheads, lineage-labeled type 2 cells. Scale bars: 1.5 mm in B; 3 mm in C; 6 mm in D; 7.5 mm in E; 500 μm in F-H; 20 μm in I-L.
Distal epithelial progenitors labeled at later stages of lung development give rise to alveolar cells
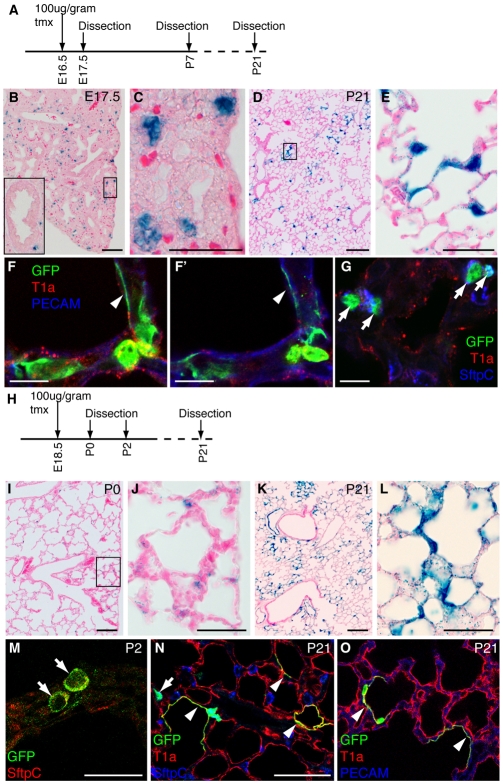
We next asked about the fate of epithelial cells present in the distal epithelium during the early canalicular stage. We exposed pregnant Id2-CreERT2; Rosa26-lacZ females to tmx at E16.5 and dissected the progeny at E17.5. This confirmed that only distal epithelial tip, not bronchiolar, cells were labeled (Fig. 3A-C). When the progeny were dissected at P21, lineage labeling was observed throughout the alveoli (Fig. 3D,E) in both type 2 and 1 alveolar cells (Fig. 3F,G). We did not observe any lineage-labeled bronchiolar cells, including the putative bronchioalveolar stem cell (BASC) population, at P21. In conclusion, tip cells present at the canalicular stage give rise to alveolar cells only, not to bronchiolar cells.
Fig. 3.
Id2+ epithelial tip cells labeled at later stages only contribute descendents to the alveoli. (A) Time course of experiment shown in B-G. Id2-CreERT2; Rosa26R-lacZ or Rosa26R-eYFP mouse embryos were exposed to tmx at E16.5 and sacrificed at intervals. (B-E) X-Gal-stained (blue) lung sections. (B) E17.5. Note staining in epithelial tips and not in the more proximal airways (inset). (C) Higher magnification of box in B showing labeled tip cells. (D) P21. Note labeling throughout the alveoli. (E) Higher magnification of box in D showing lineage-labeled type 1 and 2 cells. (F-G). P7 sections. (F,F′) Two confocal sections from same z-stack showing lineage-labeled type 1 cell (arrowhead) tightly apposed to, but separate from, the endothelium. Green, anti-GFP, lineage label; red, anti-T1α, type 1 and 2 cells (see Fig. S2 in the supplementary material); blue, anti-Pecam, endothelium. (G) Lineage-labeled type 2 cells (arrows). Green, anti-GFP, lineage label; red, anti-T1α, type 1 and 2 cells; blue, anti-pro-SftpC, type 2 cells. (H) Time course of experiment in I-O. Id2-CreERT2; Rosa26R-lacZ or Rosa26R-eYFP embryos were exposed to tmx at E18.5 and sacrificed at intervals. (I-L) X-Gal-stained (blue) lung sections. (I) P0. Labeling in alveoli. (J) Higher magnification of box in I showing labeling restricted to type 2 cells and a small number of endothelial cells. (K) P21. Labeling throughout the alveoli. (L) Higher magnification of K, lineage-labeled type 1 and 2 cells. (M) P2. Lineage-labeled type 2 cells (arrows). Green, anti-GFP, lineage label; red, anti-pro-SftpC, type 2 cells. (N) P21. Lineage-labeled type 1 (arrowheads) and type 2 (arrow) cells. Green, anti-GFP, lineage label; red, anti-T1α, type 1 cells; blue, anti-pro-SftpC, type 2 cells. (O) P21. Lineage-labeled type 1 cells (arrowheads). Green, anti-GFP, lineage label; red, anti-T1α, type 1 cells; blue, anti-Pecam, endothelium. Scale bars: 50 μm in B-E,N,O; 10 μm in F-G; 1 mm in I,K; 20 μm in J,L,M.
When Id2-CreERT2; Rosa26R-lacZ embryos were exposed to tmx at E18.5 and examined at P0, it was mostly cells in the alveolar sacs that were lineage labeled (Fig. 3H-J). At P2, these labeled cells expressed SftpC (Fig. 3M). They subsequently gave rise to mature type 2 and type 1 cells (Fig. 3K,L,N,O). This supports the previously published hypothesis that type 2 cells, or their progenitors, generate type 1 cells (Kauffman et al., 1974).
Lineage labeling of single epithelial tip cells
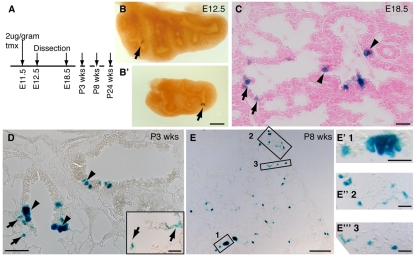
Is there a single self-renewing Id2+ progenitor cell type during the pseudoglandular stage? Or, are there separate bronchiolar and alveolar progenitors? To distinguish between these possibilities, we identified a dose of tmx that labeled either a single tip epithelial cell, or no cells, per lung. We exposed pregnant females to this dose at E11.5 and observed that 7/12 lungs at E12.5 had single labeled cells (Fig. 4A,B; note that pairs of labeled cells were often observed, consistent with the labeled cell dividing before lung harvest). No labeling was observed in the remaining lungs. The fate of the descendents of these single labeled cells was assessed by cutting serial sections through E18.5 lungs and scoring clones of labeled cells as bronchiolar only (3/26), alveolar only (2/26), or mixed (21/26) (Fig. 4C). These data demonstrate that more than 80% of individual Id2+ cells labeled at E11.5 were self-renewing progenitors for both the bronchioles and alveoli. Significantly, the few bronchiolar-only and alveolar-only clones that were observed consisted of single cells, suggesting they were derived from Id2+ cells that did not divide after labeling.
Fig. 4.
Single Id2+ tip epithelial cells can give rise to both bronchiolar and alveolar lineages. (A) Id2-CreERT2; Rosa26R-lacZ mouse embryos were exposed to a low dose of tmx at E11.5 and sacrificed at intervals. (B,B′) E12.5 wholemount lungs showing individual clones (arrows). Pairs of cells are frequently seen, suggesting that lineage-labeled cells divided prior to dissection. (C) E18.5 section. Note lineage-labeled bronchiolar (arrowheads) and alveolar (arrows) cells. (D) Lung section from a 3-week-old mouse. Note the larger groups of lineage-labeled cells. Inset shows labeled alveolar cells in the adjacent section. (E-E‴) Lung section from an 8-week-old mouse. Low-magnification image showing clone size. Box 1 shows a bronchiolar clone at higher magnification. Boxes 2 and 3 are high magnifications of alveolar regions showing labeled type 1 and 2 cells. Scale bars: 1 mm in B,E; 20 μm in C and insets in D,E′-E‴; 40 μm D.
When lungs were harvested at P21, clones were larger and contained a mixture of Clara, ciliated, type 2 and type 1 cells, based on morphology (Fig. 4D). This strongly suggests that single Id2+ cells at E11.5 not only contribute to both the bronchioles and alveoli, but are multipotent lung epithelial progenitors. Owing to technical difficulties, we did not specifically stain for NE cells and cannot exclude the possibility that NE cells are derived from a separate progenitor. As we were just as likely to see alveolar-only as bronchiolar-only single cell clones at E18.5, we favor the hypothesis that Id2+ cells are multipotent for the lung epithelium during the pseudoglandular stage.
The clones continued to increase in size over time (Fig. 4E), demonstrating that adult epithelial progenitors are derived from the embryonic tip population.
Our results also show that there is extensive cell mixing within the epithelium during embryonic lung development (for example, compare Fig. 4B with 4C). This is analogous to kidney development, in which cell mixing in the branching tip has been observed (Shakya et al., 2005). By contrast, patches of lineage-labeled cells were observed in all postnatal lungs examined (Fig. 4D,E), suggesting that little cell mixing occurs postnatally.
Lineage labeling of adult Id2+ cells
We asked whether Id2+ multipotent progenitor cells persist in the adult. Although some Id2 transcripts can be detected in the adult lung by RT-PCR, we could not localize these to a population of highly expressing cells using in situ hybridization or the Id2-eGFP mice (not shown). No recombination was observed in Id2-CreERT2 adult mice treated with vehicle alone (n=8) or with two doses of tmx (n=4) (not shown). However, when adults were exposed to four successive doses of 0.25 mg tmx per gram body weight, some labeled cells were observed in the epithelium, including Clara, ciliated and type 2 cells, and endothelium (see Fig. S3A,B,E,F in the supplementary material). We asked whether these cells gave rise to clones of descendants over time, as would be expected if they were proliferating progenitors. When animals were sacrificed at intervals up to 10 months after the final tmx injection, we did not observe any clusters of lineage-labeled cells that would be expected if the labeled cells had proliferated (see Fig. S3C,D,G,H in the supplementary material). Rather, we noticed that although both Clara and ciliated cells were initially labeled in the bronchioles (74% lineage-labeled cells were ciliated, n=190 cells, two mice), by 10 months only labeled ciliated cells were found (99% lineage-labeled cells were ciliated, n=277 cells, three mice). This is consistent with labeled Clara cells differentiating into ciliated cells (Rawlins et al., 2009). Similar results were observed with the rare bronchiolar cells labeled by tmx exposure at E18.5 and analyzed at P21 (Fig. 3K). These data suggest that Id2 is not expressed in an important population of epithelial progenitors in the postnatal lung.
We tested the response of adult Id2+ bronchiolar cells to injury by exposing lineage-labeled Id2-CreERT2; Rosa26R-lacZ mice to naphthalene (see Fig. S3A in the supplementary material). Of the lineage-labeled cells, only ciliated cells survived naphthalene injury (see Fig. S3I,K in the supplementary material). Consistent with previous reports (Hong et al., 2001; Rawlins et al., 2007), these neither proliferated nor transdifferentiated to contribute to repair (see Fig. S3J,L in the supplementary material). This supports our conclusion that Id2 is not expressed in an important epithelial progenitor in the adult. We also tested the possibility that Id2 is upregulated during repair. We exposed Id2-eGFP adults to naphthalene and assessed eGFP levels 48-72 hours post-injury, when epithelial repair is beginning. In addition, we exposed Id2-CreERT2; Rosa26R-lacZ mice to naphthalene and then to tmx to lineage label Id2+ cells during repair. We did not detect any increase in the numbers of Id2+ epithelial cells in either of these experiments (not shown).
Conclusions
In conclusion, our results support a model in which the distal tip epithelium of the developing lung contains a self-renewing population of progenitors whose descendents exit the tip and give rise to all lung epithelial cell types, including NE cells (as illustrated in Fig. S4 in the supplementary material). Based on our single-cell lineage-tracing data, we favor the hypothesis that tip cells are multipotent progenitors. However, we cannot formally exclude the possibility that minor progenitor populations exist that differ in their ability to give rise to the various differentiated lineages (see Fig. S4 in the supplementary material). Resolving this question will require additional single-cell lineage tracing using a reporter allele more compatible with immunohistochemistry. The precise role of Id2 in the progenitor cells, and the regulation of their developmental potential, also remain to be determined.
Many additional important questions concerning the lung epithelial lineage remain to be resolved. Different strategies will be needed to ask whether late distal progenitors, which give rise to only alveolar lineages, still have the potential to generate proximal epithelial cell types if placed in a different environment. One approach to address this question is to recombine the cells with early embryonic mesenchyme and graft them to ectopic sites, such as the renal capsule. Finally, during development, the trachea and primary lung buds arise from a Nkx2.1+ domain of the foregut (Cardoso and Lu, 2006), so another important question is at what stage the SftpC+ epithelial cells in the primary lung buds become committed to lung fate.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/cgi/content/full/136/22/3741/DC1
Supplementary Material
We thank Yuan Zhuang for the Id2 genomic fragment. This work was funded by NIH grant HL071303 (B.L.M.H.) and a Parker B. Francis Fellowship (E.L.R.). Deposited in PMC for release after 12 months.
References
- Cardoso, W. V. and Lu, J. (2006). Regulation of early lung morphogenesis: questions, facts and controversies. Development 133, 1611-1624. [DOI] [PubMed] [Google Scholar]
- Eblaghie, M. C., Reedy, M., Oliver, T., Mishina, Y. and Hogan, B. L. (2006). Evidence that autocrine signaling through Bmpr1a regulates the proliferation, survival and morphogenetic behavior of distal lung epithelial cells. Dev. Biol. 291, 67-82. [DOI] [PubMed] [Google Scholar]
- Farley, F. W., Soriano, P., Steffen, L. S. and Dymecki, S. M. (2000). Widespread recombinase expression using FLPeR (flipper) mice. Genesis 28, 106-110. [PubMed] [Google Scholar]
- Hayashi, S. and McMahon, A. P. (2002). Efficient recombination in diverse tissues by a tamoxifen-inducible form of Cre: a tool for temporally regulated gene activation/inactivation in the mouse. Dev. Biol. 244, 305-318. [DOI] [PubMed] [Google Scholar]
- Hong, K. U., Reynolds, S. D., Giangreco, A., Hurley, C. M. and Stripp, B. R. (2001). Clara cell secretory protein-expressing cells of the airway neuroepithelial body microenvironment include a label-retaining subset and are critical for epithelial renewal after progenitor cell depletion. Am. J. Respir. Cell Mol. Biol. 24, 671-681. [DOI] [PubMed] [Google Scholar]
- Jen, Y., Manova, K. and Benezra, R. (1996). Expression patterns of Id1, Id2, and Id3 are highly related but distinct from that of Id4 during mouse embryogenesis. Dev. Dyn. 207, 235-252. [DOI] [PubMed] [Google Scholar]
- Kauffman, S. L., Burri, P. H. and Weibel, E. R. (1974). The postnatal growth of the rat lung. II. Autoradiography. Anat. Rec. 180, 63-76. [DOI] [PubMed] [Google Scholar]
- Liu, Y. and Hogan, B. L. (2002). Differential gene expression in the distal tip endoderm of the embryonic mouse lung. Gene Expr. Patterns 2, 229-233. [DOI] [PubMed] [Google Scholar]
- Metzger, R. J., Klein, O. D., Martin, G. R. and Krasnow, M. A. (2008). The branching programme of mouse lung development. Nature 453, 745-750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okubo, T., Knoepfler, P. S., Eisenman, R. N. and Hogan, B. L. (2005). Nmyc plays an essential role during lung development as a dosage-sensitive regulator of progenitor cell proliferation and differentiation. Development 132, 1363-1374. [DOI] [PubMed] [Google Scholar]
- Perl, A. K., Wert, S. E., Nagy, A., Lobe, C. G. and Whitsett, J. A. (2002). Early restriction of peripheral and proximal cell lineages during formation of the lung. Proc. Natl. Acad. Sci. USA 99, 10482-10487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajagopal, J., Carroll, T. J., Guseh, J. S., Bores, S. A., Blank, L. J., Anderson, W. J., Yu, J., Zhou, Q., McMahon, A. P. and Melton, D. A. (2008). Wnt7b stimulates embryonic lung growth by coordinately increasing the replication of epithelium and mesenchyme. Development 135, 1625-1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlins, E. L. (2008). Lung epithelial progenitor cells: lessons from development. Proc. Am. Thorac. Soc. 5, 675-681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlins, E. L., Ostrowski, L. E., Randell, S. H. and Hogan, B. L. (2007). Lung development and repair: Contribution of the ciliated lineage. Proc. Natl. Acad. Sci. USA 104, 410-417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlins, E. L., Okubo, T., Xue, Y., Brass, D. M., Auten, R. L., Hasegawa, H., Wang, F. and Hogan, B. L. (2009). The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 4, 525-534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shakya, R., Watanabe, T. and Costantini, F. (2005). The role of GDNF/Ret signaling in ureteric bud cell fate and branching morphogenesis. Dev. Cell 8, 65-74. [DOI] [PubMed] [Google Scholar]
- Zhou, Q., Law, A. C., Rajagopal, J., Anderson, W. J., Gray, P. A. and Melton, D. A. (2007). A multipotent progenitor domain guides pancreatic organogenesis. Dev. Cell 13, 103-114. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.