Abstract
OBJECTIVE
Insulin and contraction each increase muscle microvascular blood volume (MBV) and glucose uptake. Inhibiting nitric oxide synthase blocks insulin's but not contraction's effects. We examined whether contraction could augment the MBV increase seen with physiologic hyperinsulinemia and whether free fatty acid (FFA)-induced insulin resistance differentially affects contraction- versus insulin-mediated increases in MBV.
RESEARCH DESIGN AND METHODS
Rats were fasted overnight. Plasma FFAs were increased by intralipid/heparin infusion (3 h), insulin was increased with a euglycemic clamp (3 mU · min−1 · kg−1), and hindlimb muscle contraction was electrically stimulated. Muscle MBV was measured using contrast-enhanced ultrasound. Insulin transport into muscle was measured using 125I-insulin. BQ-123 (0.4 mg/h) was used to block the endothelin-1 (ET-1) receptor A.
RESULTS
Superimposing contraction on physiologic hyperinsulinemia increased MBV within 10 min by 37 and 67% for 0.1 or 1 Hz, respectively (P < 0.01). FFA elevation alone did not affect MBV, whereas 0.1 Hz stimulation doubled MBV (P < 0.05) and increased muscle insulin uptake (P < 0.05) despite high FFA. Physiologic hyperinsulinemia during FFA elevation paradoxically decreased MBV (P < 0.05). This MBV decrease was reversed by either 0.1 Hz contraction or ET-1 receptor A antagonism, and the combination raised MBV above basal.
CONCLUSIONS
Contraction recruits microvasculature beyond that seen with physiologic hyperinsulinemia by a distinct mechanism that is not blocked by FFA-induced vascular insulin resistance. The paradoxical MBV decline seen with insulin plus FFA may result from differential inhibition of insulin-stimulated nitric oxide–dependent vasodilation relative to ET-1 vasoconstriction. Our results implicate ET-1 as a potential mediator of FFA-induced vascular insulin resistance.
Insulin delivery to muscle interstitium is reported to be rate limiting for overall muscle insulin action (1,2). Insulin promotes its own access to muscle interstitium by increasing blood flow (3), by recruiting microvasculature (4,5) to expand the endothelial transporting surface available, and perhaps by also stimulating its own endothelial transport (6). Insulin's entry to muscle interstitium is delayed in insulin-resistant states (7). This implicates insulin's vascular actions as a significant regulator of overall insulin action in muscle.
Elevated plasma concentrations of free fatty acids (FFAs), as occur with obesity and type 2 diabetes, increase cellular lipid concentrations and are associated with insulin resistance in skeletal muscle, liver, and fat (8,9). Experimentally, increased dietary fat (10–12) or acute infusion of a lipid emulsion induces insulin resistance (13–16). Increased intramyocellular lipid content in the context of obesity and type 2 diabetes could be one factor that contributes to muscle insulin resistance. Postprandially or in response to a euglycemic-insulin clamp, plasma (FFA) falls in insulin-sensitive individuals (17–19). This response is impaired in states of insulin resistance (8,17,19,20).
Insulin also increases muscle blood flow and recruits microvasculature in both humans (21–24) and animals (4,25–27); both processes are inhibited by nitric oxide synthases (NOS) blockade (27). Raising plasma FFAs initiates hemodynamic effects that include decreased compliance, increased blood pressure and heart rate, and increased vascular resistance (28–31). Raising plasma (FFA) blunts insulin's NOS-dependent effects to mediate increases in both muscle microvascular blood volume (MBV) and glucose uptake (14,32,33). Thus, FFAs exert acute vascular as well as metabolic actions.
Insulin (34) and muscle contraction can each increase MBV and total flow in skeletal muscle (35–37). In addition, Wheatley et al. (38) observed that in the Zucker rat, insulin-mediated increases in MBV are blunted, but contraction-induced increases in MBV persisted. This suggests that exercise might recruit microvasculature via a mechanism that is distinct from that of insulin. Supporting this, we have recently shown that like insulin, brief low-frequency isometric contraction of the rat hindlimb (0.1 Hz, 10 min) robustly increases MBV without any observed increase in total femoral blood flow (FBF) and, unlike insulin's effect, this process is nitric oxide (NO)-independent (39).
In this study, we addressed whether 1) low-frequency contraction enhances muscle MBV and 3H-2-deoxyglucose (3H-2-DG) uptake beyond the effect of physiologic hyperinsulinemia; 2) lipid infusion differentially affects contraction- versus insulin-mediated increases in MBV; and 3) lipid infusion blunts combined insulin- and contraction-mediated effects on MBV.
RESEARCH DESIGN AND METHODS
Male Sprague-Dawley rats (225–300 g) were obtained from Charles River Laboratories (Wilmington, MA), housed at 22 ± 2°C with a 12:12-h light:dark cycle, allowed free access to water and standard rat diet, but fasted overnight before the study. The experimental protocols were approved by the Animal Care and Use Committee of the University of Virginia.
Surgical procedures.
Animals were anesthetized with an intraperitoneal injection of sodium pentobarbital (50–55 mg/kg body wt) and placed on a surgical platform. Body temperature was maintained at 38°C with a heating lamp and pad. Both jugular veins and the carotid artery were cannulated with polyethylene tubing (PE-50) and used for intravenous infusions, arterial blood sampling, and monitoring of mean arterial blood pressure. A tracheostomy was performed to facilitate respiration. Animals were maintained under anesthesia for the duration of the experiment by intravenous infusion of aqueous sodium pentobarbital solution (0.6 mg · min−1 · kg−1) via the carotid artery. For experiments in which total FBF was measured, the femoral vessels in the right hindlimb were exposed as described previously (23,38) and an ultrasound flow probe (VB series 0.5 mm; Transonic Systems) was positioned over the femoral artery. The flow probe was interfaced through a flow meter to a personal computer. FBF and arterial blood pressure were continuously acquired using Windaq software (Dataq Instruments, Akron, OH). The animals were allowed 30–45 min to stabilize after surgical procedures before beginning experimental protocols.
Electrical stimulation.
Two metal electrodes were placed in the adductor tendons of the animal's right hindlimb and the limb was secured in place. The left hindlimb served as the sham control. Muscles were isometrically contracted for 10 min (2 V, 0.5 ms) at a frequency given in each protocol (Grass S88 Pulse Generator; Astro-Med, West Warwick, RI). We previously reported that 0.1 Hz stimulation can increase in basal hindlimb MBV approximately twofold (39) without an increased femoral artery flow.
Contrast-enhanced ultrasound.
MBV was measured at the time points indicated in the protocols described in Fig. 1. Where indicated, animals received an infusion of phosphatidylcholine/polyethylene glycol stearate– coated decafluorobutane-filled microbubbles manufactured at the University of Virginia as reported previously (40). Microbubbles were diluted 1:3 in deoxygenated saline and infused at a rate of 10–15 μl/min for 11 min. Pulse-inversion ultrasound (HDI-5000, Philips Ultrasound) images of the proximal adductor muscle group (adductor magnus and semimembranosus) were obtained as previously described (5,27) during the last 6 min at the time points specified in Fig. 1. Data were analyzed using a commercial software package (Q-lab, Philips Ultrasound).
FIG. 1.
Lipid and insulin infusion protocols. Time points for microbubble infusion and data acquisition are denoted by (*) for contrast- enhanced ultrasound. Blood samples (20 μl) were taken at t = 0, 30, 60, 120, and 180 min.
Experimental protocols.
Rats were studied in each of six experimental protocols (Fig. 1). 1) A 90-min euglycemic-insulin clamp was given as a primed (6 mU · kg−1 · min−1 × 8 min) continuous (3 mU · kg−1 · min−1 × 82 min) insulin infusion with a 30% glucose solution infused to maintain fasting euglycemia. During the last 30 min of the insulin clamp, 3 × 10 min periods of electrical stimulation (0.1, 1.0, and 2.0 Hz) were imposed. 2) Animals received a 70-min euglycemic-insulin clamp (6 mU · kg−1 · min−1 × 8 min, then 3 mU · kg−1 · min−1 × 62 min) with a 50-μCi bolus of 3H-2DG (specific activity 48.0 Ci/mmol) given at 60 min followed by 0.1 Hz electrical stimulation of the right leg for the last 10 min. 3) Animals were given a 3-h infusion of 10 ul/min of intralipid/heparin (3.3% and 30 units/ml, respectively) together with a saline infusion over the last 2 h (25 μl/min). 4) The same intralipid/heparin infusion as in protocol 3 with a euglycemic-insulin clamp (see protocol 1) infusion over the last 2 h was given. 5) The same infusion protocol as in protocol 4, with the addition of a continuous infusion of the ET-1 receptor A (ETA), was given. ETA receptor antagonist, BQ-123 (0.4 mg/h × 3 h) was given as reported previously (41). 6) Intralipid/heparin + saline was given as in protocol 3 with 125I-insulin given half-way through the electrical stimulation period. Total FBF was measured in separate groups of rats (n = 3–5) receiving infusion protocols 3 through 5.
3H-2-DG uptake assay.
Frozen muscles harvested from the stimulated and contralateral leg at the end of protocol 2 (Fig. 1) were ground under liquid nitrogen, and ∼100 mg of tissue was homogenized in 2 ml of deionized water for 20 s on ice and spun for 10 min at 0°C (13,000 rpm). Free and phosphorylated 3H-2-DG in ∼1.5 ml supernatant were separated using anion exchange chromatography (AG1-X8 resin) as previously described (14). 3H-2-DG-P was counted in a liquid scintillation system, and from the total counts in the supernatant and plasma-specific activity, muscle uptake was calculated (micrograms of glucose per gram of weight tissue per minute).
Muscle 125I-insulin content.
Low-frequency stimulation (0.1 Hz, 2 V, 0.5 ms) began 10 min before the end of the experiment (protocol 6). Five min after starting the electrical stimulation, the rats were given a 1.5 μCi bolus of 125I-insulin. This mono-iodinated species binds to the insulin receptor similarly to native insulin, and the tracer amount infused does not decrease systemic glucose concentrations. Gastrocnemius, soleus, and gracilis muscles were dissected from the stimulated and the control contralateral hindlimb. Protein- bound 125iodine in blood and muscle samples was precipitated using 10% trichloroacetic acid, and radioactivity was measured in a gamma counter and expressed as disintegrations per minute per gram of dry tissue. This was further normalized to plasma-specific activity.
Plasma insulin and ET-1 concentration.
Blood samples were taken at the time points indicated in each protocol (Fig. 1), quickly spun down, and plasma was kept frozen (−80°C). The insulin concentration in plasma samples was run in duplicate using a radioimmunoassay (LINCO Research, St. Charles, MI). ET-1 concentration in samples was measured in duplicate using an enzyme-linked immunoassay (ALPCO Diagnostics, Winham, NH).
Western detection of AMP-activated protein kinase phosphorylation.
Muscle samples were rapidly frozen and subsequently prepared for PAGE and Western blotting as previously described (5). Proteins were transferred to nitrocellulose membranes and detected using antibodies against AMP-activated protein kinase (AMPK)-α and p-AMPKα (Thr172) according to manufacturers' protocols. Membranes were incubated with IgG-peroxidase–linked secondary antibody for 60 min. Bands were detected via chemiluminescence using enhanced chemiluminescence reagents and quantified by ImageQuant 3.3 software.
Chemicals and reagents.
Chemicals and reagents were as follows: intralipid 20% (Baxter Healthcare, Deerfield, IL); ETA antagonist BQ-123 (Alexis Biochemicals, San Diego, CA); 3H-2-DG (Amersham Biosciences, U.K.); 125I-insulin (Perkin Elmer, Waltham, MA); AG-1X8 anion exchange resin (Bio-Rad Laboratories, Hercules, CA); AMPK antibodies (Cell Signaling Technology, Beverly, MA); ECL reagents (Amersham Biosciences, U.K.).
Statistical analysis.
Statistical calculations were performed using SigmaStat software. Individual tests are described in the figure legends. Data are presented as means ± SE.
RESULTS
Contraction further increases MBV after insulin-induced microvascular recruitment.
In agreement with previous studies (4,5), 60 min of insulin increased MBV by ∼65% compared with baseline (Fig. 2). We previously reported that doses of insulin above the 3 mU · min−1 · kg−1 dose used here further enhanced MBV only minimally (34). Isometric contraction for 10 min at 0.1 Hz significantly (P < 0.05) increased MBV by an additional ∼37% over 60 min of insulin alone. Stimulation at 1 or 2 Hz further increased MBV. The increases seen at 1 and 2 Hz cannot simply be attributed to the longer duration of stimulation, as in separate experiments, we observed that 45 min of continuous stimulation at 0.1 Hz does not increase MBV beyond that seen during the first 10 min of stimulation (data not shown). We previously reported (39) that contraction alone (in the absence infused insulin) increased MBV by 131, 200, and 200% above basal at frequencies of 0.1, 1.0, and 2.0 Hz, respectively. These incremental increases above basal were similar to those seen in the current study (100, 176, and 176% above basal for these same frequencies in the setting of hyperinsulinemia, Fig. 2). These findings demonstrate that contraction increases MBV beyond that observed at insulin concentrations that are physiological and near-maximally effective in recruiting the microvasculature (34).
FIG. 2.
Combined effect of insulin and contraction on MBV. Protocol 1 results (n = 7): Effect of insulin (3 mU · kg−1 · min−1 × 90 min) and isometric hindlimb contraction (0.1, 1.0, 2.0 Hz; 10 min/bout) on MBV. *P < 0.05 vs. baseline (white bar); **P < 0.05 vs. baseline, insulin 60 min (light gray); #P < 0.05 vs. baseline, insulin 60 min and 0.1 Hz (dark gray); one-way repeated-measures ANOVA.
Low-frequency contraction further enhances insulin-stimulated 3H-2DG uptake in muscle.
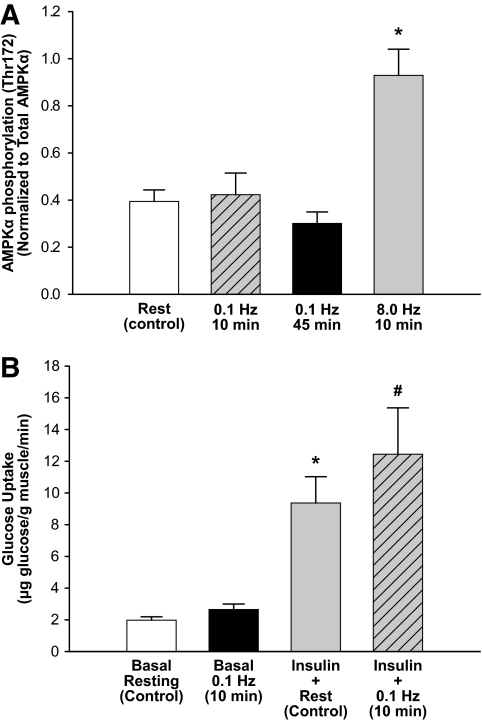
Study protocol 2 addressed whether very low-frequency (0.1 Hz) electrical stimulation of brief (0.5 msec) duration, which would be expected to minimally effect leg energy expenditure (but effectively enhances MBV), affects muscle glucose disposal during a euglycemic clamp. We first observed that in the absence of a systemic insulin infusion, this brief contractile stimulus did not affect basal muscle 2-deoxyglucose uptake (2.7 ± 0.3 vs. 2.0 ± 0.2 μg glucose · g wt tissue−1 · min−1 for 0.1 Hz and control legs, respectively) (Fig. 3B). At the end of the euglycemic-clamp muscle 2-deoxyglucose uptake was significantly stimulated compared with saline controls, and glucose uptake by the electrically stimulated leg exceeded that in the control contralateral limb by ∼1.3-fold (Fig. 3B; P < 0.05).
FIG. 3.
Effect of contraction on AMPKα phosphorylation and glucose uptake. A: Effect of isometric contraction on fasting basal muscle AMPKα phosphorylation (Thr172). *P < 0.05 vs. 0.1 Hz (10 min), 0.1 Hz (45 min); one-way ANOVA (n = 5). B: Protocol 2 results: Combined effect of insulin and brief, low-frequency contraction on muscle glucose utilization (n = 6). A 3H-2DG bolus (50 μCi) was given right before the onset of contraction and circulated a total of 10 min. *P < 0.05 vs. basal conditions, #P < 0.05 vs. insulin + rest, paired t test.
We also examined whether 0.1 Hz stimulation enhanced muscle AMPKα phosphorylation as this is often correlated with exercise-stimulated glucose uptake and metabolism in muscle. In rats receiving only a saline infusion, neither 10 min nor an extended 45 min of 0.1 Hz contraction significantly increased AMPKα phosphorylation as compared with control (Fig. 3A). In contrast, as a positive control, 10 min of high-frequency electrical stimulation at 8.0 Hz significantly increased AMPKα phosphorylation.
FFAs do not affect basal or contraction-mediated increases in MBV nor prevent contraction-mediated 125I-insulin uptake.
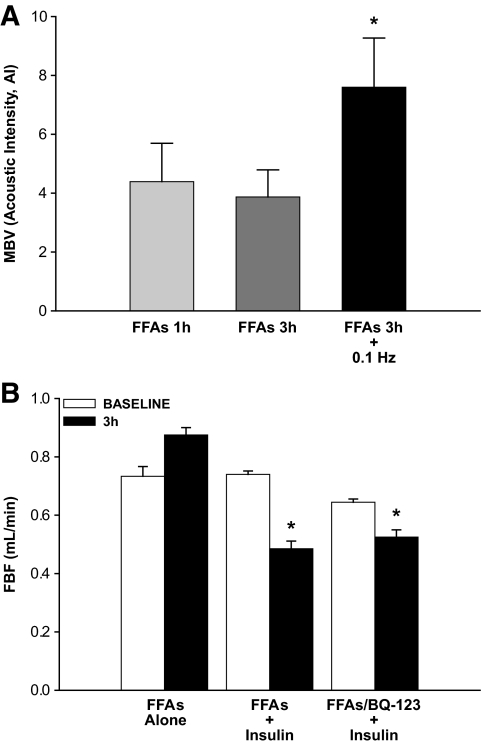
We previously reported that total FBF remains unchanged in response to 10 min of 0.1 Hz contraction, whereas skeletal muscle insulin uptake is modestly increased by this brief 0.1 Hz contractile stimulus (39). In the present study, plasma FFA levels increased ∼1.5-fold over baseline after 1 and 3 h, of the infusion protocol (Table 1). With lipid infusion, total FBF trended toward a slight increase after 3 h, but this was not significant (Fig. 4B). When insulin was added to the FFA infusion, FBF declined significantly. Presumably, this was secondary to increased tone of resistance vessels in the leg. Others had reported a paradoxic vasoconstriction induced by insulin when FFAs were infused (42). Infusing BQ 123 for 1 h before and throughout the insulin infusion did not prevent this decline (Fig. 4B). In response to intralipid/heparin alone, MBV remained unchanged after either 1 or 3 h of infusion (Fig. 4A). However, MBV increased ∼1.8-fold within 10 min of initiating 0.1 Hz contraction (vs. 1 h and 3 h; P < 0.05). Here again, in the context of FFA-provoked insulin resistance, 0.1 Hz contraction still significantly promoted the delivery of 125I-insulin to muscle as compared with the resting control limb (Fig. 5). We had previously observed a similar effect when saline was infused instead of FFA (39).
TABLE 1.
Physiological parameters
| AVG | |
|---|---|
| Weight | 266 ± 6 |
| Fasting blood glucose (mmol/l) | 5.3 ± 0.1 |
| GIR (mg · kg−1 · min−1) | |
| Insulin only | 9.5 ± 0.8 |
| Insulin + FFAs | 3.5 ± 0.7* |
| Insulin + FFAs + BQ-123 | 3.3 ± 0.9* |
| Insulin (pmol) | |
| Baseline | 104 ± 16 |
| FFAs 1 h | 294 ± 63 |
| FFAs 3 h | 203 ± 25 |
| FFAs 3 h + 0.1 Hz | 223 ± 49 |
| FFAs 1.5 h + insulin 30 min | 543 ± 188† |
| FFAs 3 h + insulin 2 h | 1,029 ± 205† |
| FFA (mM) | |
| Baseline | 0.62 ± 0.06 |
| FFAs 3 h | 0.98 ± 0.07‡ |
| FFAs 3 h + insulin 2 h | 1.13 ± 0.11‡ |
| ET-1 (fmol/ml) | |
| Baseline | 0.93 ± 0.19 |
| FFAs 3 h | 0.76 ± 0.12 |
Data are means ± SE (n = 6, 9 per group).
*P < 0.05 vs. insulin;
†P < 0.05 vs. baseline;
‡P < 0.05 vs. baseline (one-way ANOVA). GIR, glucose infusion rate.
FIG. 4.
Effect of FFAs and contraction on MBV and FBF. A: Protocol 3 results: The effect of FFA-provoked insulin resistance on MBV after 1 h, 3 h, and 3 h + 10 min 0.1 Hz isometric hindlimb muscle contraction (n = 6). *P < 0.05 vs. FFAs 1 h, FFAs 3 h; one-way repeated-measures ANOVA. B: Effects of treatment on FBF; protocols 3–5 (n = 4 per group). *P < 0.05; one-way repeated-measures ANOVA (vs. baseline).
FIG. 5.
Effect of FFAs on contraction-mediated muscle 125I-insulin uptake. An intravenous 1.5 μCi bolus of 125I-insulin was given 5 min after starting low-frequency (0.1 Hz) isometric contraction (protocol 2). Intact insulin in wet tissue was determined using trichloroaetic acid precipitation and then normalized to dry tissue weight (n = 7). *P < 0.05.
Contraction, but not insulin, increases MBV in the presence of FFAs.
Infusion of intralipid/heparin (protocol 3) decreased the glucose infusion rate required to maintain euglycemia during the insulin clamp indicating insulin resistance (Table 1). Interestingly, MBV did not increase but declined slightly during the first 30 min after the onset of insulin infusion and was significantly below baseline after 2 h (Fig. 6; P < 0.05). This was paralleled by a gradual and significant decrease in total FBF over the last 60 min studied in a separate group of five animals (Fig. 4B; P < 0.05). At the end of the 3 h of intralipid/heparin and 2 h of insulin, 0.1 Hz muscle contraction increased MBV by more than threefold within 10 min, reaching levels comparable to but not significantly above baseline.
FIG. 6.
Effect of FFA microvascular responses to insulin and contraction. Intralipid/heparin (FFAs) and insulin were infused as described in experimental protocol 4. In the presence of elevated FFAs, insulin had a paradoxical effect to decrease MBV vs. baseline. Brief low-frequency contraction (0.1 Hz, 10 min) restored MBV to baseline (n = 9). *P < 0.05 vs. all other treatment groups; one-way repeated-measures ANOVA.
ETA receptor antagonism prevents the effect of combined lipid and insulin to decrease MBV.
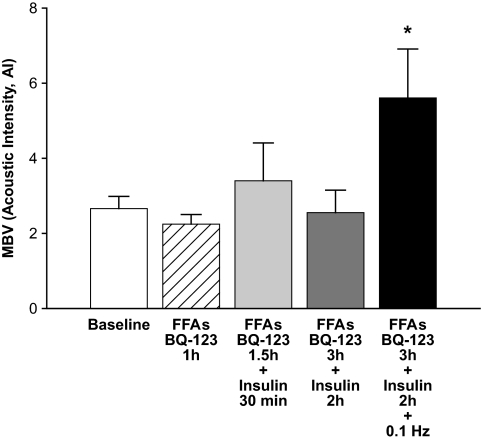
In addition to stimulating NO production, insulin can enhance production of the vasoconstrictor ET-1 via the ERK1/2 protein kinase cascade. To address whether the unexpected MBV decrease produced by combined intralipid/heparin and insulin infusion might be caused by increased ET-1 production, we measured plasma ET-1 concentrations at baseline and at the end of protocol 4. The circulating ET-1 concentrations did not change during the infusion protocol (Table 1). As ET-1 is thought to act principally in a paracrine fashion, we investigated this further by infusing the ETA receptor antagonist BQ-123 (0.4 mg/h) (41) concomitantly with intralipid/heparin throughout the duration of the experiment. In this protocol, MBV did not change significantly during combined BQ-123, intralipid/heparin, and insulin infusion (Fig. 7) (mean 14% rise, P = not significant), unlike the 32% decline observed in MBV in protocol 4 without BQ-123 present (Fig. 6). When the change in MBV was compared between animals studied using protocols 4 and 5, the MBV decline in protocol 4 was of borderline (P = 0.07) significance. This pattern of MBV response contrasts with the lack of effect of BQ 123 on the insulin-induced decline in FBF (Fig. 4B) noted previously. However, low frequency contraction at 0.1 Hz (10 min) significantly increased MBV in the BQ-123, intralipid/heparin, and insulin group.
FIG. 7.
Combined ETA antagonism and FFA elevation on the microvascular response to insulin and contraction. The ETA receptor anagonist, BQ-123 (0.4 mg/h), was infused with intralipid/heparin (FFAs) as described in protocol 5. The physiologic euglycemic-hyperinsulinemic clamp (3 mU · kg−1 · min−1 × 90 min) started 1 h after FFA/BQ infusion. Contraction (0.1 Hz) was superimposed over the last 10 min. MBV was measured at the indicated time points (n = 6). *P < 0.05 vs. all other groups; one-way repeated-measures ANOVA.
DISCUSSION
In the present study, we confirmed that insulin alone increased the skeletal muscle microvascular volume accessible to microbubbles significantly over basal values. However, when compared with basal (preinsulin or contraction) the combination of insulin plus exercise had no greater effect on MBV versus contraction alone at either 0.1, 1.0, or 2.0 Hz. Muscle contraction did increase MBV above that seen with insulin alone. As the insulin infusion rate used here exerts near-maximal effects on MBV, the significant augmentation after contraction suggests that contraction is a more potent stimulus to microvascular recruitment. We had previously found that insulin recruits microvasculature by a NO-dependent (27) and exercise by a NO-independent mechanism (39). The additional finding that FFA elevation blocked insulin's but not contraction's ability to recruit microvasculature further differentiates these several mechanisms that regulate capillary recruitment.
Low-frequency muscle contraction (0.1 Hz) also significantly increased muscle 2-deoxyglucose uptake during hyperinsulinemia but not when insulin concentrations remained at postabsorptive levels. In addition, it did not affect AMPKα phosphorylation. Lack of any increase of 2-deoxyglucose uptake under conditions of basal insulin underscores the very light workload from this contraction stimulus protocol. It appears likely that the increased muscle 2-deoxyglucose uptake during insulin infusion (euglycemic clamp) plus muscle contraction could be explained by the observed changes in MBV. Such changes would increase endothelial surface available for insulin delivery to muscle interstitium, which in turn may have enhanced insulin's action to stimulate muscle glucose uptake. Indeed, we had previously reported that a 0.1 Hz contraction significantly increased the muscle uptake of iodinated insulin (39). In the current study, we observed that FFA-provoked insulin resistance did not block the effect of 0.1 Hz contraction to increase 125I-insulin delivery to muscle. These findings raise the possibility that muscle contraction–induced increases in MBV and enhanced insulin delivery to muscle interstitium may be one of the mechanisms by which exercise ameliorates insulin resistance.
We also wanted to ascertain the effect of acute infusion of intralipid/heparin on contraction- and insulin-mediated increases on muscle MBV. Contraction appeared to increase MBV normally during intralipid/heparin infusion (Fig. 4). In contrast, infusing insulin with intralipid/heparin paradoxically decreased MBV below baseline. Adding low-frequency muscle contraction restored MBV to baseline values but did not produce increases over baseline as was seen when either insulin or FFA alone were followed by electrical stimulation (Fig. 6).
Insulin stimulates the release of both ET-1 and NO by endothelial cells. Inhibiting PI-3-kinase reduces NO-mediated arteriolar dilation but enhances constriction by ET-1 (42). Insulin-resistant individuals have higher fasting ET-1 levels than healthy control subjects. Acute increases in both plasma insulin and FFAs in normal subjects raises plasma ET-1 to concentrations seen with insulin resistance (43). Interestingly, it was recently shown that ETA receptor antagonism in obese insulin-resistant individuals increased skeletal muscle hemodynamic and metabolic responses to insulin (44). We hypothesized that the balance between insulin's effect to increase NO (vasodilation) and ET-1 (vasoconstriction) might be affected in the context of elevated plasma FFAs.
We were not able to detect an effect of elevated plasma FFA concentrations on plasma ET-1 concentration. However, antagonism of ETA receptors with BQ-123 prevented the decline in MBV observed in response to combined FFAs and insulin. This suggests that FFAs might enhance insulin-mediated ET-1 release. Although BQ-123 prevented MBV from decreasing in response to combined FFA elevation and insulin, MBV did not rise above baseline values. Adding a contraction stimulus further increased MBV. Therefore, antagonism of ETA alone might not be expected to fully restore the vascular response to insulin in the presence of FFAs. Another possible mechanism that might explain these results is that FFAs might facilitate conversion of NO to reactive oxygen species as has been previously suggested (45,46), and in the context of elevated plasma FFAs, insulin-mediated NO production might lead to biologically unfavorable effects.
We conclude that in the absence of elevated plasma FFAs, insulin and low-frequency muscle contraction can each increase MBV. Muscle contraction stimulates muscle insulin uptake, and this contributes to the enhancing effect of contraction on glucose metabolism. Raising plasma FFA concentration does not block the effect of contraction to recruit microvasculature. However, raising FFA alters endothelial function to provoke a paradoxical insulin-mediated de- or recruitment of the microvasculature. The effect of BQ123 to block MBV decreases, triggered by combining hyperinsulinemia with elevated plasma FFA, suggests a shift in a balance of insulin action on the microvascular endothelium toward increased ET-1 production and at least partly underlies this paradoxic vasoconstrictor response. However, this does not exclude the possibility that in the context of elevated plasma FFAs, insulin signaling in the vascular endothelium might activate other hemodynamically unfavorable actions. The fact that BQ123 did not also prevent the decline in leg blood flow (Fig. 4B) may also suggest either other pathways by which insulin may cause vasoconstriction or that resistance vessels are not as sensitive to BQ123 and the dosages used here. These findings extend our understanding of the distinct mechanisms used by insulin and exercise to act on the vasculature within skeletal muscle.
Acknowledgments
No potential conflicts of interest relevant to this article were reported.
Parts of this study were presented in abstract form at the 68th Scientific Sessions of the American Diabetes Association, San Francisco, California, 6–10 June 2008.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
REFERENCES
- 1.Yang YJ, Hope ID, Ader M, Bergman RN: Insulin transport across capillaries is rate limiting for insulin action in dogs. J Clin Invest 1989;84:1620–1628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Miles PD, Levisetti M, Reichart D, Khoursheed M, Moossa AR, Olefsky JM: Kinetics of insulin action in vivo. Identification of rate-limiting steps. Diabetes 1995;44:947–953 [DOI] [PubMed] [Google Scholar]
- 3.Baron AD: Hemodynamic actions of insulin. Am J Physiol 1994;267:E187–E202 [DOI] [PubMed] [Google Scholar]
- 4.Rattigan S, Clark MG, Barrett EJ: Hemodynamic actions of insulin in rat skeletal muscle: Evidence for capillary recruitment. Diabetes 1997;46:1381–1388 [DOI] [PubMed] [Google Scholar]
- 5.Vincent MA, Clerk LH, Lindner JR, Klibanov AL, Clark MG, Rattigan S, Barrett EJ: Microvascular recruitment is an early insulin effect that regulates skeletal muscle glucose uptake in vivo. Diabetes 2004;53:1418–1423 [DOI] [PubMed] [Google Scholar]
- 6.Wang H, Wang AX, Liu Z, Barrett EJ: Insulin signaling stimulates insulin transport by bovine aortic endothelial cells. Diabetes 2008;57:540–547 [DOI] [PubMed] [Google Scholar]
- 7.Sjostrand M, Gudbjornsdottir S, Holmang A, Lonn L, Strindberg L, Lonnroth P: Delayed transcapillary transport of insulin to muscle interstitial fluid in obese subjects. Diabetes 2002;51:2742–2748 [DOI] [PubMed] [Google Scholar]
- 8.Krebs M, Roden M: Molecular mechanisms of lipid-induced insulin resistance in muscle, liver and vasculature. Diabetes Obes Metab 2005;7:621–632 [DOI] [PubMed] [Google Scholar]
- 9.Han SH, Quon MJ, Koh KK: Reciprocal relationships between abnormal metabolic parameters and endothelial dysfunction. Curr Opin Lipidol 2007;18:58–65 [DOI] [PubMed] [Google Scholar]
- 10.Oakes ND, Cooney GJ, Camilleri S, Chisholm DJ, Kraegen EW: Mechanisms of liver and muscle insulin resistance induced by chronic high-fat feeding. Diabetes 1997;46:1768–1774 [DOI] [PubMed] [Google Scholar]
- 11.Halseth AE, Bracy DP, Wasserman DH: Limitations to basal and insulin-stimulated skeletal muscle glucose uptake in the high-fat-fed rat. Am J Physiol Endocrinol Metab 2000;279:E1064–E1071 [DOI] [PubMed] [Google Scholar]
- 12.Ellmerer M, Hamilton-Wessler M, Kim SP, Huecking K, Kirkman E, Chiu J, Richey J, Bergman RN: Reduced access to insulin-sensitive tissues in dogs with obesity secondary to increased fat intake. Diabetes 2006;55:1769–1775 [DOI] [PubMed] [Google Scholar]
- 13.Kruszynska YT, Worrall DS, Ofrecio J, Frias JP, Macaraeg G, Olefsky JM: Fatty acid-induced insulin resistance: decreased muscle PI3K activation but unchanged Akt phosphorylation. J Clin Endocrinol Metab 2002;87:226–234 [DOI] [PubMed] [Google Scholar]
- 14.Clerk LH, Rattigan S, Clark MG: Lipid infusion impairs physiologic insulin-mediated capillary recruitment and muscle glucose uptake in vivo. Diabetes 2002;51:1138–1145 [DOI] [PubMed] [Google Scholar]
- 15.Le Marchand-Brustel Y, Gual P, Gremeaux T, Gonzalez T, Barres R, Tanti JF: Fatty acid-induced insulin resistance: role of insulin receptor substrate 1 serine phosphorylation in the retroregulation of insulin signalling. Biochem Soc Trans 2003;31:1152–1156 [DOI] [PubMed] [Google Scholar]
- 16.Roden M, Price TB, Perseghin G, Petersen KF, Rothman DL, Cline GW, Shulman GI: Mechanism of free fatty acid-induced insulin resistance in humans. J Clin Invest 1996;97:2859–2865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shankar SS, Steinberg HO: FFAs: do they play a role in vascular disease in the insulin resistance syndrome? Curr Diab Rep 2005;5:30–35 [DOI] [PubMed] [Google Scholar]
- 18.Kelley DE, Simoneau JA: Impaired free fatty acid utilization by skeletal muscle in non-insulin- dependent diabetes mellitus. J Clin Invest 1994;94:2349–2356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bonadonna RC, Groop LC, Zych K, Shank M, DeFronzo RA: Dose-dependent effect of insulin on plasma free fatty acid turnover and oxidation in humans. Am J Physiol 1990;259:E736–E750 [DOI] [PubMed] [Google Scholar]
- 20.Kim JK, Wi JK, Youn JH: Plasma free fatty acids decrease insulin-stimulated skeletal muscle glucose uptake by suppressing glycolysis in conscious rats. Diabetes 1996;45:446–453 [DOI] [PubMed] [Google Scholar]
- 21.Bonadonna RC, Saccomani MP, Delprato S, Bonora E, Defronzo RA, Cobelli C: Role of Tissue-Specific Blood Flow and Tissue Recruitment in Insulin-Mediated Glucose Uptake of Human Skeletal Muscle. Circulation 1998;98:234–241 [DOI] [PubMed] [Google Scholar]
- 22.Baron AD, Tarshoby M, Hook G, Lazaridis EN, Cronin J, Johnson A, Steinberg HO: Interaction between insulin sensitivity and muscle perfusion on glucose uptake in human skeletal muscle: evidence for capillary recruitment. Diabetes 2000;49:768–774 [DOI] [PubMed] [Google Scholar]
- 23.Vincent MA, Dawson D, Clark AD, Lindner JR, Rattigan S, Clark MG, Barrett EJ: Skeletal muscle microvascular recruitment by physiological hyperinsulinemia precedes increases in total blood flow. Diabetes 2002;51:42–48 [DOI] [PubMed] [Google Scholar]
- 24.Coggins M, Lindner J, Rattigan S, Jahn L, Fasy E, Kaul S, Barrett E: Physiologic hyperinsulinemia enhances human skeletal muscle perfusion by capillary recruitment. Diabetes 2001;50:2682–2690 [DOI] [PubMed] [Google Scholar]
- 25.Gaudreault N, Santure M, Pitre M, Nadeau A, Marette A, Bachelard H: Effects of insulin on regional blood flow and glucose uptake in Wistar and Sprague-Dawley rats. Metabolism 2001;50:65–73 [DOI] [PubMed] [Google Scholar]
- 26.Clark MG, Wallis MG, Barrett EJ, Vincent MA, Richards SM, Clerk LH, Rattigan S: Blood flow and muscle metabolism: a focus on insulin action. Am J Physiol Endocrinol Metab 2003;284:E241–E258 [DOI] [PubMed] [Google Scholar]
- 27.Vincent MA, Barrett EJ, Lindner JR, Clark MG, Rattigan S: Inhibiting NOS blocks microvascular recruitment and blunts muscle glucose uptake in response to insulin. Am J Physiol Endocrinol Metab 2003;285:E123–E129 [DOI] [PubMed] [Google Scholar]
- 28.Stojiljkovic MP, Zhang D, Lopes HF, Lee CG, Goodfriend TL, Egan BM: Hemodynamic effects of lipids in humans. Am J Physiol Regul Integr Comp Physiol 2001;280:R1674–R1679 [DOI] [PubMed] [Google Scholar]
- 29.Monahan KD, Dyckman DJ, Ray CA: Effect of acute hyperlipidemia on autonomic and cardiovascular control in humans. J Appl Physiol 2007;103:162–169 [DOI] [PubMed] [Google Scholar]
- 30.Steinberg HO, Baron AD: Vascular function, insulin resistance and fatty acids. Diabetologia 2002;45:623–634 [DOI] [PubMed] [Google Scholar]
- 31.Tripathy D, Mohanty P, Dhindsa S, Syed T, Ghanim H, Aljada A, Dandona P: Elevation of free fatty acids induces inflammation and impairs vascular reactivity in healthy subjects. Diabetes 2003;52:2882–2887 [DOI] [PubMed] [Google Scholar]
- 32.Steinberg HO, Paradisi G, Hook G, Crowder K, Cronin J, Baron AD: Free fatty acid elevation impairs insulin-mediated vasodilation and nitric oxide production. Diabetes 2000;49:1231–1238 [DOI] [PubMed] [Google Scholar]
- 33.Clerk LH, Vincent MA, Clark MG, Barrett EJ: Physiological elevation of free fatty acids (FFA) inhibits insulin-mediated capillary recruitment in rat skeletal muscle. Diabetes 2003;52:A48 [Google Scholar]
- 34.Zhang L, Vincent MA, Richards SM, Clerk LH, Rattigan S, Clark MG, Barrett EJ: Insulin sensitivity of muscle capillary recruitment in vivo. Diabetes 2004;53:447–453 [DOI] [PubMed] [Google Scholar]
- 35.Dawson D, Vincent MA, Barrett EJ, Kaul S, Clark A, Leong-Poi H, Lindner JR: Vascular recruitment in skeletal muscle during exercise and hyperinsulinemia assessed by contrast ultrasound. Am J Physiol Endocrinol Metab 2002;282:E714–E720 [DOI] [PubMed] [Google Scholar]
- 36.Honig CR, Odoroff CL, Frierson JL: Capillary recruitment in exercise: rate, extent, uniformity, and relation to blood flow. Am J Physiol 1980;238:H31–H42 [DOI] [PubMed] [Google Scholar]
- 37.Vincent MA, Lindner JR, Jahn L, Leong-Poi H, Barrett EJ: Modest exercise enhances human skeletal muscle perfusion by capillary recruitment in the absence of changes in total blood flow. Diabetes 2002;51:A59 [Google Scholar]
- 38.Wheatley CM, Rattigan S, Richards SM, Barrett EJ, Clark MG: Skeletal muscle contraction stimulates capillary recruitment and glucose uptake in insulin-resistant obese Zucker rats. Am J Physiol Endocrinol Metab 2004;287:E804–E809 [DOI] [PubMed] [Google Scholar]
- 39.Inyard AC, Clerk LH, Vincent MA, Barrett EJ: Contraction stimulates nitric oxide independent microvascular recruitment and increases muscle insulin uptake. Diabetes 2007;56:2194–2200 [DOI] [PubMed] [Google Scholar]
- 40.Fisher NG, Christiansen JP, Klibanov A, Taylor RP, Kaul S, Lindner JR: Influence of microbubble surface charge on capillary transit and myocardial contrast enhancement. J Am Coll Cardiol 2002;40:811–819 [DOI] [PubMed] [Google Scholar]
- 41.Oparil S, Chen SJ, Meng QC, Elton TS, Yano M, Chen YF: Endothelin-A receptor antagonist prevents acute hypoxia-induced pulmonary hypertension in the rat. Am J Physiol 1995;268:L95–L100 [DOI] [PubMed] [Google Scholar]
- 42.Eringa EC, Stehouwer CD, Merlijn T, Westerhof N, Sipkema P: Physiological concentrations of insulin induce endothelin-mediated vasoconstriction during inhibition of NOS or PI3-kinase in skeletal muscle arterioles. Cardiovasc Res 2002;56:464–471 [DOI] [PubMed] [Google Scholar]
- 43.Piatti PM, Monti LD, Conti M, Baruffaldi L, Galli L, Phan CV, Guazzini B, Pontiroli AE, Pozza G: Hypertriglyceridemia and hyperinsulinemia are potent inducers of endothelin-1 release in humans. Diabetes 1996;45:316–321 [DOI] [PubMed] [Google Scholar]
- 44.Lteif A, Vaishnava P, Baron AD, Mather KJ: Endothelin limits insulin action in obese/insulin-resistant humans. Diabetes 2007;56:728–734 [DOI] [PubMed] [Google Scholar]
- 45.Inoguchi T, Li P, Umeda F, Yu HY, Kakimoto M, Imamura M, Aoki T, Etoh T, Hashimoto T, Naruse M, Sano H, Utsumi H, Nawata H: High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C–dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes 2000;49:1939–1945 [DOI] [PubMed] [Google Scholar]
- 46.Pleiner J, Schaller G, Mittermayer F, Bayerle-Eder M, Roden M, Wolzt M: FFA-induced endothelial dysfunction can be corrected by vitamin C. J Clin Endocrinol Metab 2002;87:2913–2917 [DOI] [PubMed] [Google Scholar]