Abstract
OBJECTIVE
To examine sex-specific black/white differences in lipoprotein profile and the role of visceral adiposity and to assess the relationship between insulin sensitivity and lipoprotein profiles in each group.
RESEARCH DESIGN AND METHODS
Fasting lipoprotein particle size and concentration and visceral adipose tissue (VAT) were determined in 226 children (117 black, 101 male) aged 8 to <18 years. The relationship between lipoproteins and insulin sensitivity was evaluated in a subset of 194 children (100 black, 88 male) who underwent a hyperinsulinemic-euglycemic clamp.
RESULTS
Black male children had smaller VLDL and black female children had larger HDL size than their white counterparts. Overall, blacks had larger LDL size with no sex-specific race differences. After adjusting for VAT and sex, only VLDL size and concentrations remained significantly favorable in blacks. Analysis of lipoprotein particle size and concentration across insulin sensitivity quartiles revealed that in both racial groups, the most insulin-resistant children had higher concentrations of small dense LDL, small HDL, and large VLDL and smaller LDL and HDL sizes than their more insulin-sensitive counterparts.
CONCLUSIONS
The previously reported favorable lipoprotein profiles in black versus white children is partly due to race differences in VAT. In both groups, however, the most insulin-resistant youths have a high-risk atherogenic profile of small dense LDL, small HDL, and large VLDL, akin to the atherogenic lipoprotein pattern in adults with coronary artery disease.
Type 2 diabetes and insulin resistance in children are associated with dyslipidemia (1,2), characterized by elevated triglycerides and LDL cholesterol and low concentrations of HDL cholesterol (1–3). In addition to traditional lipid profiles, evidence suggests that insulin resistance and type 2 diabetes are associated with changes in lipoprotein particle size and subclass concentration (2,4). These are important to assess, as traditional lipid measurements only partially predict disease risk (5). Recently, the SEARCH for Diabetes in Youth study (2) reported that 36% of youth with type 2 diabetes and 62% of those with poorly controlled diabetes had small dense LDL. Similarly, low proportions of large and high proportions of small HDL particles are found in children with type 2 diabetes and overweight, insulin-resistant children (4). However, whereas some investigators reported associations between LDL (6,7), HDL (8), and VLDL (6) particle size and fasting insulin, others did not (9). High triglyceride and low HDL cholesterol concentrations together with small, dense LDL in children with type 2 diabetes and insulin resistance are similar to the atherogenic lipoprotein phenotype in adults with coronary artery disease (10,11).
Black children, despite being insulin resistant and hyperinsulinemic (12,13) compared with their white counterparts, have favorable lipid concentrations including lower LDL and triglyceride and higher HDL concentrations (3,14,15), larger HDL and LDL and smaller VLDL particles, and favorable lipoprotein subclass concentrations (6,8). Why black children have favorable lipoprotein profiles despite insulin resistance is not clear. One explanation could be lower visceral adiposity in black than in white children despite similar overall adiposity (15). In black adults insulin resistance is not a good marker of triglyceride or HDL cholesterol concentrations or lipoprotein particle size (16). Thus, the relationship between in vivo insulin sensitivity and lipoprotein profiles in black and white children needs to be examined if at-risk children are to be identified for early treatments to improve lipoprotein profiles and if those treatments are to be pertinent in children of different ethnicity.
In the present study, therefore, we determined lipoprotein particle size and subclass concentrations in black and white children and measured in vivo insulin sensitivity to test the following hypotheses: 1) the favorable lipoprotein phenotype in black children is probably due to lower visceral adipose tissue (VAT) than in whites and 2) the relationship between insulin sensitivity and lipoprotein profile is similar between black and white children.
RESEARCH DESIGN AND METHODS
Participants, aged 8 to <18 years old, consisted of 117 black and 109 white normal-weight and overweight otherwise healthy children, except for 16 girls (10 white and 6 black; all with BMI ≥98th percentile) with untreated polycystic ovary syndrome (PCOS). Some participants were reported before as part of an ongoing National Institutes of Health (NIH)-funded R01 grant investigating race-related differences in childhood insulin sensitivity and secretion (3,12). Studies took place at the Children's Hospital of Pittsburgh NIH-funded Pediatric Clinical and Translational Research Center after institutional review board approval. Participants and their parents gave written informed consent. Of the 226 youth, 194 had a hyperinsulinemic-euglycemic clamp. Exclusion criteria included diagnosed diabetes and use of medications that influence glucose, lipid metabolism, or blood pressure. Participants' health was assessed by medical history, physical examination, and hematological and biochemical tests. Pubertal development was assessed using Tanner criteria.
Body weight and height were measured using standardized equipment. Waist circumference was obtained at the midpoint between the lowest rib and the iliac crest (17). Body composition and abdominal adiposity were assessed by dual-energy X-ray absorptiometry and computed tomography, respectively, as described previously (12). Fasting blood samples were collected from all 226 children for analysis of lipoprotein particle size and concentration.
In vivo insulin sensitivity
A subset of children (100 black and 94 white, including 16 girls with PCOS) underwent a 3-h hyperinsulinemic-euglycemic clamp after 10–12 h of overnight fasting. Briefly, intravenous crystalline insulin (Humulin; Lilly, Indianapolis, IN) was infused at a constant rate of 40 mU · m−2 · min−1 in normal-weight subjects and 80 mU · m−2 · min−1 in obese subjects to suppress hepatic glucose production, as described previously (12). Plasma glucose was clamped at 5.6 mmol/l with a variable rate infusion of 20% dextrose based on arterialized plasma glucose determined every 5 min.
Biochemical measurements
Plasma glucose was measured using a glucose analyzer (YSI, Yellow Springs, OH), and insulin concentrations were measured by radioimmunoassay (12). Plasma lipid concentrations were determined using the standards of the Centers for Disease Control and Prevention as described previously (18), and lipoprotein particle size and subclass concentration were determined using nuclear magnetic resonance spectroscopy (LipoScience, Raleigh, NC) (19).
Calculations
Insulin-stimulated glucose disposal was calculated using the average exogenous glucose infusion rate during the final 30 min of the clamp (12). Insulin sensitivity was calculated by dividing the insulin-stimulated glucose disposal rate by steady-state plasma insulin concentrations during the last 30 min of the clamp, as described previously (3,12).
Statistical analysis
Independent t tests or χ2 tests for categorical variables were used to examine race-related differences in subject characteristics and lipoprotein particle size and concentration in the group as a whole and stratified by sex. ANCOVA was used to determine the influence of sex and visceral adiposity on race-related differences in lipoprotein particle size and concentration. Black and white subjects were divided into insulin sensitivity quartiles by sex. One-way ANOVA or the nonparametric Kruskal-Wallis test, based on the nonviolation of statistical assumptions, was used to compare differences in lipoprotein particle size and concentration among quartiles. Tukey's post hoc comparison was used to identify differences among quartiles. Because insulin sensitivity changes with puberty, we analyzed differences among quartiles with an ANCOVA including Tanner stage as a covariate. Data were also analyzed with exclusion of the 16 girls with PCOS to determine whether this condition may have affected our results. Stepwise multiple regression was used to assess the contribution of race, sex, age, insulin sensitivity, and VAT to lipoprotein particle size. Data are presented as means ± SEM with significance at P < 0.05.
RESULTS
Sex-specific, race-related differences
Participant characteristics are summarized in Table 1. Black and white youth had similar body weight, BMI, body composition, and subcutaneous abdominal adipose tissue. VAT was lower in black than in white girls, with a similar tendency in black versus white boys (P = 0.055).
Table 1.
Physical characteristics of the participants
| Male |
Female |
P
|
||||
|---|---|---|---|---|---|---|
| Blacks | Whites | Blacks | Whites | Black vs. white male | Black vs. white female | |
| n | 49 | 52 | 68 | 57 | ||
| Age (years) | 12.7 ± 0.3 | 13.5 ± 0.3 | 12.8 ± 0.3 | 12.6 ± 0.3 | 0.033 | NS |
| Tanner stage | ||||||
| I | 9 | 6 | 10 | 8 | NS | NS |
| II–III | 22 | 26 | 15 | 15 | NS | NS |
| IV–V | 18 | 20 | 43 | 34 | NS | NS |
| Height (cm) | 158.9 ± 2.0 | 164.5 ± 1.8 | 154.8 ± 1.3 | 154.8 ± 1.6 | 0.037 | NS |
| Weight (kg) | 70.3 ± 3.9 | 76.1 ± 4.6 | 70.6 ± 3.4 | 69.5 ± 3.7 | NS | NS |
| BMI (kg/m2) | 27.0 ± 1.1 | 27.1 ± 1.2 | 28.4 ± 1.1 | 28.2 ± 1.2 | NS | NS |
| BMI percentile | 83.0 ± 3.1 | 79.3 ± 4.0 | 82.7 ± 3.0 | 84.2 ± 2.9 | NS | NS |
| Fat mass (kg) | 20.8 ± 2.2 | 21.5 ± 2.3 | 26.8 ± 2.0 | 25.7 ± 2.1 | NS | NS |
| Fat-free mass (kg) | 43.9 ± 1.8 | 44.7 ± 1.9 | 40.0 ± 1.4 | 37.1 ± 1.3 | NS | NS |
| Body fat (%) | 27.9 ± 2.1 | 27.7 ± 1.8 | 35.3 ± 1.4 | 36.2 ± 1.5 | NS | NS |
| Waist circumference (cm) | 85.6 ± 3.0 | 90.8 ± 3.3 | 83.0 ± 2.9 | 80.1 ± 2.9 | NS | NS |
| VAT (cm2) | 39.4 ± 5.4 | 56.1 ± 6.7 | 37.3 ± 3.6 | 52.9 ± 5.4 | 0.055 | 0.018 |
| SAT (cm2) | 245.8 ± 32.3 | 303.9 ± 37.3 | 327.3 ± 29.0 | 339.2 ± 31.6 | NS | NS |
Data are means ± SEM. Tanner stages compared using χ2. All other variables were compared using independent t tests. SAT, subcutaneous adipose tissue.
As a group, black children had lower VLDL, total LDL, and small dense LDL and higher large HDL concentrations and larger HDL and LDL and smaller VLDL particle sizes than whites (Supplementary Table A1, available in an online appendix at http://care.diabetesjournals.org/cgi/content/full/dc09-0380). After correcting for sex and VAT, race differences remained in VLDL particle size and total, large, and medium VLDL and total HDL concentrations (Table A1).
Table 2 depicts race data grouped by sex. Black male children had smaller VLDL particle size and black female children had larger HDL size than their white counterparts. After adjustment for VAT, differences in VLDL particle size remained in male children (P = 0.028) and for HDL particle size persisted in female children (P = 0.084). Black male and female children had lower concentrations of total, large, and medium VLDL, and black male children had lower concentrations of total, small, and very small LDL than their white counterparts. After adjustment for VAT, differences in VLDL remained in both sexes but differences in LDL disappeared in male children.
Table 2.
Lipoprotein subclass concentration, particle size, and plasma lipids in black versus white children
| Male |
Female |
Nominal P |
||||
|---|---|---|---|---|---|---|
| Blacks | Whites | Blacks | Whites | Black vs. white male | Black vs. white female | |
| n | 49 | 52 | 68 | 57 | ||
| VLDL and chylomicrons (nmol/l) | ||||||
| Total VLDL and chylomicrons | 41.7 ± 2.9 | 54.8 ± 2.9 | 40.3 ± 2.2 | 53.5 ± 2.5 | 0.002 | <0.001 |
| Large VLDL and chylomicrons | 1.7 ± 0.4 | 4.1 ± 0.7 | 1.3 ± 0.3 | 2.7 ± 0.4 | 0.005 | 0.007 |
| Medium VLDL | 11.4 ± 1.5 | 19.4 ± 1.9 | 10.8 ± 1.1 | 18.3 ± 1.3 | 0.001 | <0.001 |
| Small VLDL | 28.6 ± 1.7 | 31.4 ± 2.1 | 28.2 ± 1.6 | 32.4 ± 1.6 | NS | NS |
| LDL (nmol/l) | ||||||
| Total LDL | 833.0 ± 40.4 | 993.8 ± 53.3 | 828.6 ± 35.6 | 887.7 ± 43.9 | 0.019 | NS |
| Large LDL | 316.1 ± 20.0 | 302.6 ± 22.2 | 321.3 ± 15.3 | 296.4 ± 16.4 | NS | NS |
| Small LDL | 481.6 ± 41.3 | 649.8 ± 55.3 | 474.5 ± 36.1 | 553.7 ± 48.3 | 0.017 | NS |
| Medium small LDL | 104.9 ± 8.4 | 133.8 ± 12.4 | 100.2 ± 7.1 | 116.4 ± 9.4 | NS | NS |
| Very small LDL | 376.8 ± 33.2 | 515.9 ± 43.2 | 374.3 ± 29.3 | 437.3 ± 39.1 | 0.012 | NS |
| HDL (μmol/l) | ||||||
| Total HDL | 27.1 ± 0.7 | 25.8 ± 0.7 | 25.1 ± 0.5 | 24.7 ± 0.5 | NS | NS |
| Large HDL | 6.2 ± 0.5 | 5.5 ± 0.5 | 6.2 ± 0.3 | 5.4 ± 0.4 | NS | NS |
| Medium HDL | 4.8 ± 0.5 | 4.5 ± 0.7 | 3.5 ± 0.4 | 3.7 ± 0.3 | NS | NS |
| Small HDL | 16.1 ± 0.6 | 15.8 ± 0.7 | 15.4 ± 0.6 | 15.6 ± 0.5 | NS | NS |
| IDL (nmol/l) | 35.3 ± 4.7 | 41.4 ± 7.4 | 32.7 ± 4.1 | 37.4 ± 4.8 | NS | NS |
| Lipoprotein particle size (nm) | ||||||
| VLDL | 50.8 ± 1.2 | 56.4 ± 1.5 | 50.7 ± 1.3 | 52.8 ± 1.0 | 0.004 | NS |
| LDL | 21.2 ± 0.1 | 20.9 ± 0.1 | 21.2 ± 0.1 | 21.1 ± 0.1 | NS | NS |
| HDL | 9.1 ± 0.1 | 9.0 ± 0.1 | 9.1 ± 0.1 | 8.9 ± 0.1 | NS | 0.007 |
| Plasma lipids (mmol/l) | ||||||
| Total cholesterol | 4.09 ± 0.12 | 4.49 ± 0.13 | 3.93 ± 0.09 | 4.12 ± 0.09 | 0.031 | NS |
| LDL cholesterol | 2.44 ± 0.10 | 2.66 ± 0.12 | 2.36 ± 0.09 | 2.45 ± 0.08 | NS | NS |
| HDL cholesterol | 1.20 ± 0.05 | 1.16 ± 0.04 | 1.18 ± 0.03 | 1.11 ± 0.03 | NS | NS |
| Total triglycerides | 1.00 ± 0.08 | 1.50 ± 0.14 | 0.94 ± 0.07 | 1.26 ± 0.08 | 0.003 | 0.003 |
| VLDL triglycerides | 0.20 ± 0.02 | 0.30 ± 0.03 | 0.19 ± 0.01 | 0.25 ± 0.02 | 0.003 | 0.002 |
Data are means ± SEM. All comparisons were made using independent t tests. Nominal P values indicate that significance values are unadjusted for multiple comparisons. IDL, intermediate-density lipoprotein.
Insulin sensitivity, lipoprotein particle size, and concentrations
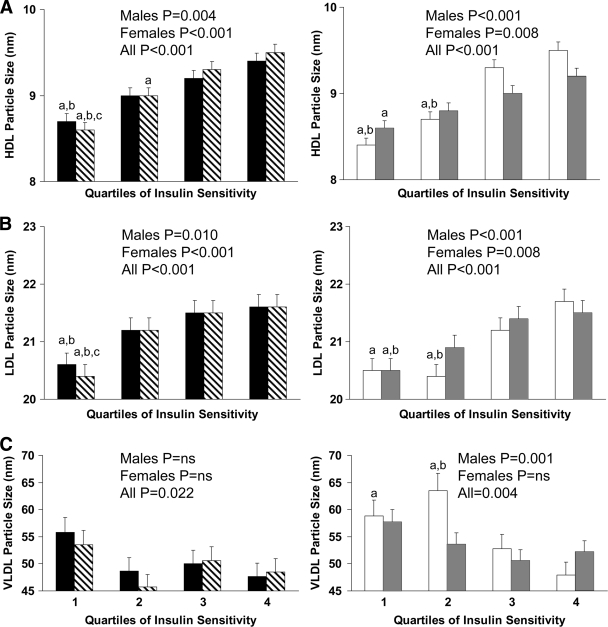
Figure 1 depicts lipoprotein particle size by in vivo insulin sensitivity quartiles. Irrespective of sex, black and white children in the lowest quartile of insulin sensitivity had smaller HDL (Fig. 1A) and LDL (Fig. 1B) size than children in the upper quartiles. White male children in the lowest two quartiles of insulin sensitivity had larger VLDL particle size (Fig. 1C) than their counterparts in the upper quartiles.
Figure 1.
HDL (A), LDL (B), and VLDL (C) particle size by quartiles of insulin sensitivity in black male (n = 43; ■) and female (n = 57; ▧) and white male (n = 45; □) and female (n = 49;  ) children. Differences within each group were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; c, significant difference versus 2; P < 0.05. Range of insulin sensitivity (in micromoles per kilogram per minute per picomole per liter) for black children: quartile 1, 0.45–1.95; quartile 2, 2.16–4.37; quartile 3, 4.40–9.20; quartile 4, 9.21–18.39; for white children: quartile 1, 0.68–1.75; quartile 2, 1.78–3.76; quartile 3, 3.80–9.21; quartile 4, 9.28–25.32.
) children. Differences within each group were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; c, significant difference versus 2; P < 0.05. Range of insulin sensitivity (in micromoles per kilogram per minute per picomole per liter) for black children: quartile 1, 0.45–1.95; quartile 2, 2.16–4.37; quartile 3, 4.40–9.20; quartile 4, 9.21–18.39; for white children: quartile 1, 0.68–1.75; quartile 2, 1.78–3.76; quartile 3, 3.80–9.21; quartile 4, 9.28–25.32.
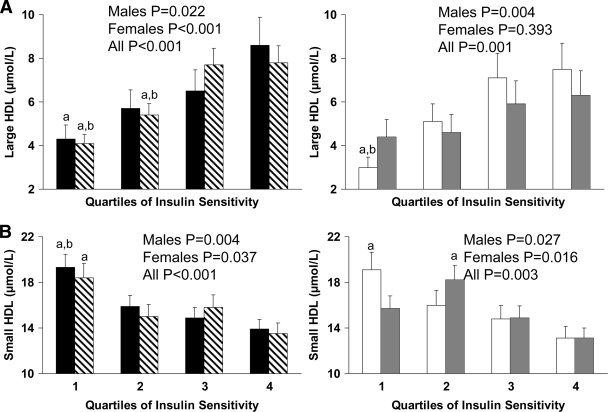
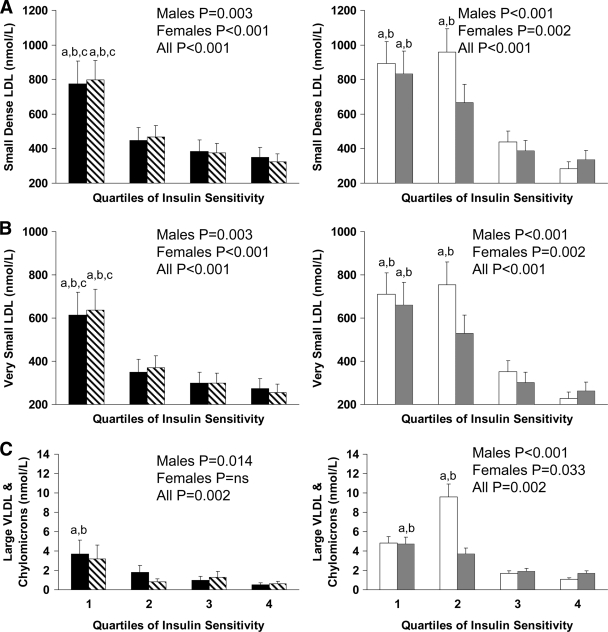
Figures 2 and 3 depict lipoprotein particle concentrations by in vivo insulin sensitivity quartiles. In white male children and in both sexes for black children, those in the lowest quartile of insulin sensitivity had lower concentrations of large and higher concentrations of small HDL than children in the top quartile (Fig. 2A and B). Similarly, small dense LDL and very small LDL concentrations were higher in the lowest than in the uppermost quartile of insulin sensitivity in both races (Fig. 3A and B), irrespective of sex. Large VLDL and chylomicron concentrations were significantly higher in the most insulin-resistant quartile of black male children (P < 0.05) and in the bottom two quartiles of white children (both P < 0.05) compared with the most insulin-sensitive children in each group (Fig. 3C).
Figure 2.
Concentrations of large (A) and small (B) HDL by quartiles of insulin sensitivity in black male (■) and female (▧) and white male (□) and female ( ) children. Differences were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; P < 0.05.
) children. Differences were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; P < 0.05.
Figure 3.
Concentrations of small (A) and very small (B) LDL, and large VLDL and chylomicrons (C), by quartiles of insulin sensitivity in black male (■) and female (▧) and white male (□) and female ( ) children. Differences were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; c, significant difference versus 2; P < 0.05.
) children. Differences were compared using one-way ANOVA with post hoc Tukey correction. a, significant difference versus 4; b, significant difference versus 3; c, significant difference versus 2; P < 0.05.
After correction for pubertal development across insulin sensitivity quartiles, the significance for large HDL concentrations in black male children changed from P = 0.022 to P = 0.111; in black female children, the significance for small HDL changed from P = 0.037 to P = 0.109 and, in white male children, it changed from P = 0.027 to P = 0.078. Excluding black or white girls with PCOS from their respective datasets did not change significance values across quartiles.
Contribution of insulin sensitivity and visceral adiposity to lipoprotein particle size
In multiple regression analyses with lipoprotein particle size as the dependent variable and race, sex, age, insulin sensitivity, and VAT as the independent variables, VAT and insulin sensitivity independently and together explained 26% of the variance (P < 0.001) in LDL size (VAT, partial r = −0.293, P < 0.001; insulin sensitivity, partial r = 0.194, P = 0.008) and 41% of the variance (P < 0.001) in HDL size (VAT, partial r = −0.368, P < 0.001; insulin sensitivity, partial r = 0.301, P < 0.001), whereas VAT and race explained 12% of the variance (P < 0.001) in VLDL size (VAT, partial r = −0.266, P < 0.001; race, partial r = 0.199, P = 0.007).
CONCLUSIONS
The present study advances previous observations of favorable lipoprotein profiles in black compared with white children (3,6,8,14,15) and demonstrates that this finding is partly explained by lower visceral adiposity in blacks. Moreover, we show that in both racial groups, the most insulin-resistant youths have an atherogenic lipoprotein profile of small dense LDL, small HDL, and large VLDL, akin to the atherogenic pattern in adults with coronary artery disease (10,11). In consideration of the fact that atherosclerosis starts in childhood (20), such a lipoprotein pattern may have serious health consequences.
Our findings from the whole group (Table A1, available in an online appendix) are consistent with those of the Bogalusa Heart Study (6,8), in which black children had HDL and LDL particles on average 0.3 and 0.2 nm larger (6,8) and VLDL particles 3.6 nm smaller than those of their white peers (6). These values are similar to the mean differences we observed: 0.2 nm larger, 0.2 nm larger, and 3.8 nm smaller for HDL, LDL, and VLDL in blacks, respectively. Several studies demonstrated favorable lipid profiles in black compared with white children (6,8,14,15) despite insulin resistance (12,13) with similar observations in adults (21). Importantly, for similar overall adiposity, blacks have lower visceral adiposity than whites (3,15,22), an observation repeated in the current study. Controlling for VAT abolished black-white differences in LDL and HDL particle size and concentration in the present study. However, visceral adiposity did not account for the race-related differences in VLDL particle size and concentration, which remained significant after adjustment for VAT. Two potential explanations for this are 1) the lower concentrations of triglycerides in black children because lipoprotein size is related to concentration (6) and 2) increased lipoprotein triglyceride clearance (21), as postheparin lipoprotein lipase activity is reported to be higher and hepatic lipase activity lower in black adults (21,23).
The present study suggests that, besides race, sex-specific analyses are important. Black male children had smaller VLDL particles and black female children had larger HDL particles than their white counterparts. For particle concentration, race differences existed in the larger VLDL in children of both sexes and for male children only in the small dense LDL. One note of caution is that we report nominal significance values for race comparisons on lipid variables in both the group as a whole (Table A1) and for sex-specific analyses (Table 2). The use of multiple t tests may have increased the chance of finding a difference in our data. Nevertheless, significant race differences in some findings remain even if adjusted for multiple comparisons, particularly for VLDL concentrations in both sexes and LDL concentrations in male children.
Our study is the first to examine the relationship between in vivo insulin sensitivity and lipoprotein particle size and subclass concentrations in children. The most insulin-resistant children, irrespective of race or sex, had smaller LDL particles and higher concentrations of small dense LDL compared with their more insulin-sensitive peers. A previous study showed that the prevalence of small dense LDL was 10% in children with insulin resistance syndrome (IRS) in contrast with 1% in those without IRS (7). However, IRS was defined based on fasting insulin. Small dense LDL particles are predictive of coronary heart disease (5,24). Conversely, large HDL particles have an inverse relationship with coronary heart disease, whereas small HDL has a positive association (25). Our data demonstrate that in both races, insulin resistance was associated with small HDL particle size, increased small HDL concentration, and low large HDL concentration. Finally, VLDL particles differ in atherogenicity, with some investigations suggesting that large particles are most strongly related to arterial disease and obesity (25). Our data demonstrate that the more insulin-resistant white and black children had higher concentrations of large VLDL particles and bigger VLDL particle size. Last, in multiple regression analyses, both VAT and insulin sensitivity were significant determinants of LDL and HDL particle size, whereas VAT and race were significant for VLDL particle size.
In summary, our data confirm prior observations of favorable lipoprotein profiles in black youth compared with white youth and advance them by showing the role of the lower visceral adiposity in blacks. Sex affects the extent of these differences. Moreover, for both blacks and whites, the most insulin-resistant youth exhibit small dense LDL, small HDL, and large VLDL profiles similar to the atherogenic lipoprotein pattern in adults with coronary artery disease (10,11). Such data underscore the need to initiate therapeutic interventions early in childhood to lessen abdominal obesity and insulin resistance and improve the associated adverse alterations in lipoprotein profile, irrespective of race or sex, and reduce the potential risk of atherosclerotic cardiovascular changes.
Supplementary Material
Acknowledgments
This work was supported by the National Institutes of Health (Grants R01-HD-27503 to S.A.A., K24-HD-01357 to S.A.A., and UL1-RR-024153 CTSA [previously M01-RR-00084]).
No potential conflicts of interest relevant to this article were reported.
Parts of this study were presented in abstract form at the 69th Scientific Sessions of the American Diabetes Association, New Orleans, Louisiana, 5–9 June 2009.
We express our gratitude to the children and their parents who volunteered to participate in this study. We are grateful to the nursing staff of the Pediatric Clinical and Translational Research Center for their outstanding care of the participants and meticulous attention to the research and to Drs. Neslihan Gungor and Fida Bacha and all past endocrine fellows for their assistance with some of the clamp experiments.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
References
- 1. Howard BV, Mayer-Davis EJ, Goff D, Zaccaro DJ, Laws A, Robbins DC, Saad MF, Selby J, Hamman RF, Krauss RM, Haffner SM: Relationships between insulin resistance and lipoproteins in nondiabetic African Americans, Hispanics, and non-Hispanic whites: the Insulin Resistance Atherosclerosis Study. Metabolism 1998;47:1174–1179 [DOI] [PubMed] [Google Scholar]
- 2. Albers JJ, Marcovina SM, Imperatore G, Snively BM, Stafford J, Fujimoto WY, Mayer-Davis EJ, Petitti DB, Pihoker C, Dolan L, Dabelea DM: Prevalence and determinants of elevated apolipoprotein B and dense low-density lipoprotein in youths with type 1 and type 2 diabetes. J Clin Endocrinol Metab 2008;93:735–742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lee S, Gungor N, Bacha F, Arslanian S: Insulin resistance: link to the components of the metabolic syndrome and biomarkers of endothelial dysfunction in youth. Diabetes Care 2007;30:2091–2097 [DOI] [PubMed] [Google Scholar]
- 4. Pérez-Méndez O, Torres-Tamayo M, Posadas-Romero C, Vidaure Garcés V, Carreón-Torres E, Mendoza-Pérez E, Medina Urrutia A, Huesca-Gómez C, Zamora-González J, Aguilar-Herrera B: Abnormal HDL subclasses distribution in overweight children with insulin resistance or type 2 diabetes mellitus. Clin Chim Acta 2007;376:17–22 [DOI] [PubMed] [Google Scholar]
- 5. Packard CJ: LDL subfractions and atherogenicity: an hypothesis from the University of Glasgow. Curr Med Res Opin 1996;13:379–390 [DOI] [PubMed] [Google Scholar]
- 6. Freedman DS, Bowman BA, Otvos JD, Srinivasan SR, Berenson GS: Levels and correlates of LDL and VLDL particle sizes among children: the Bogalusa Heart Study. Atherosclerosis 2000;152:441–449 [DOI] [PubMed] [Google Scholar]
- 7. Stan S, Levy E, Devlin EE, Hanley JA, LaMarche B, O'Loughlin J, Paradis G, Lambert M: Distribution of LDL particle size in a population-based sample of children and adolescents and relationship with other cardiovascular risk factors. Clin Chem 2005;51:1192–1200 [DOI] [PubMed] [Google Scholar]
- 8. Freedman DS, Bowman BA, Srinivasan SR, Berenson GS, Otvos JD: Distribution and correlates of high-density lipoprotein subclasses among children and adolescents. Metabolism 2001;50:370–376 [DOI] [PubMed] [Google Scholar]
- 9. Kang H-S, Gutin B, Barbeau P, Litaker MS, Allison J, Le NA: Low-density lipoprotein particle size, central obesity, cardiovascular fitness, and insulin resistance syndrome markers in obese youths. Int J Obes 2002;26:1030–1035 [DOI] [PubMed] [Google Scholar]
- 10. Austin MA, King MC, Vranizan KM, Krauss RM: Atherogenic lipoprotein phenotype: a proposed genetic marker for coronary heart disease risk. Circulation 1990;82:495–506 [DOI] [PubMed] [Google Scholar]
- 11. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002;106:3143–3421 [PubMed] [Google Scholar]
- 12. Arslanian SA, Saad R, Lewy V, Danadian K, Janosky J: Hyperinsulinemia in African-American children: decreased insulin clearance and increased insulin secretion and its relationship to insulin sensitivity. Diabetes 2002;51:3014–3019 [DOI] [PubMed] [Google Scholar]
- 13. Gower BA, Granger WM, Franklin F, Shewchuk RM, Goran MI: Contribution of insulin secretion and clearance to glucose-induced insulin concentration in African-American and Caucasian children. J Clin Endocrinol Metab 2002;87:2218–2224 [DOI] [PubMed] [Google Scholar]
- 14. Herd SL, Gower BA, Dashti N, Goran MI: Body fat, fat distribution and serum lipids, lipoproteins and apolipoproteins in African-American and Caucasian-American prepubertal children. Int J Obes 2001;25:198–204 [DOI] [PubMed] [Google Scholar]
- 15. Bacha F, Saad R, Gungor N, Janosky J, Arslanian SA: Obesity, regional fat distribution, and syndrome X in obese black versus white adolescents: race differential in diabetogenic and atherogenic risk factors. J Clin Endocrinol Metab 2003;88:2534–2540 [DOI] [PubMed] [Google Scholar]
- 16. Sumner AE, Finley KB, Genovese DJ, Criqui MH, Boston RC: Fasting triglyceride and the triglyceride-HDL cholesterol ratio are not markers of insulin resistance in African Americans. Arch Intern Med 2005;165:1395–1400 [DOI] [PubMed] [Google Scholar]
- 17. Lee S, Bacha F, Gungor N, Arslanian SA: Waist circumference is an independent predictor of insulin resistance in black and white youths. J Pediatr 2006;148:188–194 [DOI] [PubMed] [Google Scholar]
- 18. Matthews KA, Kuller LH, Wing RR, Meilann EN, Plantinga P: Prior to use of estrogen replacement therapy are users healthier than nonusers. Am J Epidemiol 1996;143:971–978 [DOI] [PubMed] [Google Scholar]
- 19. Otvos JD, Jeyarajah EJ, Bennett DW, Krauss RM: Development of a proton nuclear magnetic resonance spectroscopic method for determining plasma lipoprotein concentrations and subspecies distributions from a single, rapid measurement. Clin Chem 1992;38:1632–1638 [PubMed] [Google Scholar]
- 20. Strong JP, Malcom GT, McMahan CA, Tracy RE, Newman WP, 3rd, Herderick EE, Cornhill JF: Prevalence and extent of atherosclerosis in adolescents and young adults: implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA 1999;281:727–735 [DOI] [PubMed] [Google Scholar]
- 21. Sumner AE, Vega GL, Genovese DJ, Finley KB, Bergman RN, Boston RC: Normal triglyceride levels despite insulin resistance in African Americans: role of lipoprotein lipase. Metabolism 2005;54:902–909 [DOI] [PubMed] [Google Scholar]
- 22. Lee S, Kuk JL, Hannon TS, Arslanian SA: Race and gender differences in the relationships between anthropometrics and abdominal fat in youth. Obesity 2008;16:1066–1071 [DOI] [PubMed] [Google Scholar]
- 23. Friday KE, Srinivasan SR, Elkasabany A, Dong C, Wattigney WA, Dalferes E, Jr, Berenson GS: Black-white differences in postprandial triglyceride response and postheparin lipoprotein lipase and hepatic triglyceride lipase among young men. Metabolism 1999;48:749–754 [DOI] [PubMed] [Google Scholar]
- 24. Stampfer MJ, Krauss RM, Ma J, Blanche PJ, Holl LG, Sacks FM, Hennekens CH: A prospective study of triglyceride level, low-density lipoprotein particle diameter, and risk of myocardial infarction. JAMA 1996;276:882–888 [PubMed] [Google Scholar]
- 25. Freedman DS, Otvos JD, Jeyarajah EJ, Barboriak JJ, Anderson AJ, Walker J: The measurement of lipoprotein subclasses with proton nuclear magnetic resonance spectroscopy: associations with the extent of documented coronary artery disease. Arterioscler Thromb Vasc Biol 1998;18:1046–1053 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.