Abstract
Alzheimer's disease (AD) has characteristic histopathological, molecular, and biochemical abnormalities, including cell loss; abundant neurofibrillary tangles; dystrophic neurites; amyloid precursor protein, amyloid-β (APP-Aβ) deposits; increased activation of prodeath genes and signaling pathways; impaired energy metabolism; mitochondrial dysfunction; chronic oxidative stress; and DNA damage. Gaining a better understanding of AD pathogenesis will require a framework that mechanistically interlinks all these phenomena. Currently, there is a rapid growth in the literature pointing toward insulin deficiency and insulin resistance as mediators of AD-type neurodegeneration, but this surge of new information is riddled with conflicting and unresolved concepts regarding the potential contributions of type 2 diabetes mellitus (T2DM), metabolic syndrome, and obesity to AD pathogenesis. Herein, we review the evidence that (1) T2DM causes brain insulin resistance, oxidative stress, and cognitive impairment, but its aggregate effects fall far short of mimicking AD; (2) extensive disturbances in brain insulin and insulin-like growth factor (IGF) signaling mechanisms represent early and progressive abnormalities and could account for the majority of molecular, biochemical, and histopathological lesions in AD; (3) experimental brain diabetes produced by intracerebral administration of streptozotocin shares many features with AD, including cognitive impairment and disturbances in acetylcholine homeostasis; and (4) experimental brain diabetes is treatable with insulin sensitizer agents, i.e., drugs currently used to treat T2DM. We conclude that the term “type 3 diabetes” accurately reflects the fact that AD represents a form of diabetes that selectively involves the brain and has molecular and biochemical features that overlap with both type 1 diabetes mellitus and T2DM.
Keywords: Alzheimer's disease, central nervous system, diabetes, insulin gene expression, insulin signaling
Introduction
Alzheimer's disease (AD) can only be diagnosed with certainty by postmortem demonstration of abundant neurofibrillary tangles and neuritic plaques with accompanying accumulation of amyloid precursor protein, amyloid-β (APP-Aβ) deposits in plaques and vessel walls in selected regions of the brain. Dementia-associated structural lesions are caused by neuronal cytoskeletal collapse and accumulation of hyperphosphorylated and polyubiquitinated microtubule-associated proteins, such as tau, resulting in the formation of neurofibrillary tangles, dystrophic neuritis, and neuropil threads.1–3 Progressive loss of fibers and cells and disconnection of synaptic circuitry mediate the cerebral atrophy that worsens over time. The biochemical, molecular, and cellular abnormalities that precede or accompany AD neurodegeneration, including increased activation of prodeath genes and signaling pathways, impaired energy metabolism, mitochondrial dysfunction, chronic oxidative stress, and DNA damage, are virtually stereotypical,4–11 yet they lack a clear etiology. For nearly three decades of relatively intense research on AD, the inability to interlink this constellation of abnormalities under a single primary pathogenic mechanism resulted in the emergence and propagation of various heavily debated theories, each of which focused on how one particular component of AD could trigger a cascade that contributes to the development of all other known abnormalities. However, reevaluation of the older literature revealed that impairments in cerebral glucose utilization and energy metabolism represent very early abnormalities that precede or accompany the initial stages of cognitive impairment12–14 and led us to the concept that impaired insulin signaling has an important role in the pathogenesis of AD and the proposal that AD represents “type 3 diabetes.”5
Characteristic features of diabetes mellitus syndromes include impairments in insulin actions and signaling that result in chronic hyperglycemia, irrespective of subtype, etiology, pathogenesis, or insulin availability. Type 1 diabetes mellitus (T1DM) is caused by destruction (usually autoimmune) of pancreatic islet beta cells and attendant insulin deficiency. Type 2 diabetes mellitus (T2DM), the most common form, is caused by insulin resistance in peripheral tissues and is most frequently associated with aging, a family history of diabetes, obesity, and failure to exercise. Individuals with T2DM have hyperglycemia and hyperinsulinemia. Insulin resistance in T2DM is partly mediated by reduced insulin receptor expression, insulin receptor tyrosine kinase activity, insulin receptor substrate (IRS) type 1 expression, and/or phosphatidyl-inositol-3 (PI3) kinase activation in skeletal muscle and adipocytes.15 Gestational diabetes is pregnancy associated and caused by insulin deficiency and hyperglycemia. Nonalcoholic steatohepatitis (NASH), or metabolic syndrome, is associated with hepatic insulin resistance but overlaps with T2DM.16–18 Type 3 diabetes mellitus (T3DM, discussed later) corresponds to a chronic insulin resistance plus insulin deficiency state that is largely confined to the brain but, like NASH, can overlap with T2DM. We have proposed that T3DM represents a major pathogenic mechanism of AD neurodegeneration.5,10
Interest in clarifying the roles of T2DM, insulin resistance, and hyperinsulinemia in relation to cognitive impairment, AD-associated neuronal cytoskeletal lesions, or APP-Aβ deposits in the brain began around 2000,4,8,14,19–24 but since around 2005, this field literally exploded with new information and a new concept, i.e., that primary brain insulin resistance and insulin deficiency mediate cognitive impairment and AD.5,10,25–29 This idea was fueled by evidence that tau gene expression and phosphorylation are regulated through insulin and insulin-like growth factor (IGF) signaling cascades.23,24 In addition, research performed in our laboratory demonstrated that many key aspects of the central nervous system (CNS) degeneration that occur in AD can be effectuated by impaired insulin signaling.30–33
By way of review, insulin and IGF-1 mediate their effects by activating complex intracellular signaling pathways starting with ligand binding to cell surface receptors, followed by autophosphorylation and activation of the intrinsic receptor tyrosine kinases.34–36 Insulin/IGF-1 receptor tyrosine kinases phosphorylate IRS molecules,34,37–39 which transmit signals downstream by activating the extracellular signal-related kinase/mitogen-activated protein kinase (ERK/MAPK) and PI3 kinase/Akt pathways, and inhibit glycogen synthase kinase 3β (GSK-3β). Major biological responses to signaling through IRS molecules include increased cell growth; survival, energy metabolism, and cholinergic gene expression; and inhibition of oxidative stress and apoptosis.39–46 These very same signaling pathways are activated in various cell types, tissues, and target organs that express insulin and IGF receptors and therefore are practically universal. Moreover, these pathways are phylogenetically conserved and have critical roles in regulating development, growth, survival, senescence, carcinogenesis, and neurodegeneration.
Potential Roles of Obesity and Type 2 Diabetes Mellitus in Alzheimer's Disease Pathogenesis
There is an ongoing debate about the degree to which T2DM and, more recently, T1DM contribute to AD pathogenesis. This concept has been fueled by the rising prevalence rates of obesity, T2DM, and AD over the past several decades. Moreover, an interrelationship among these entities is suggested by (1) increased risk of developing mild cognitive impairment (MCI), dementia, or AD in individuals with T2DM47,48 or obesity/dyslipidemic disorders;49 (2) progressive brain insulin resistance and insulin deficiency in AD;5,10,26,27 (3) cognitive impairment in experimental animal models of T2DM and/or obesity;50,51 (4) AD-type neurodegeneration and cognitive impairment in experimentally induced brain insulin resistance and insulin deficiency;29,52–55 (5) improved cognitive performance in experimental models and humans with AD or MCI after treatment with insulin sensitizer agents or intranasal insulin;28,56–62 and (6) shared molecular, biochemical, and mechanistic abnormalities in T2DM and AD.47,63–67 The urgency of this problem is spotlighted by the estimated 24 million people in the world with dementia and the expectation that, if current trends continue,68 prevalence rates of AD are likely to double every 20 years in the future. While aging is clearly the strongest risk factor for AD, emerging data suggest that T2DM and dyslipidemic states can contribute substantially to the pathogenesis of AD either directly or as cofactors.68
Epidemiologic studies provide convincing evidence for a significant association between T2DM and MCI or dementia and furthermore suggest that T2DM is a significant risk factor for developing AD.47,69–73 However, those findings are not without controversy,74 and in a longitudinal survey, investigators found that although borderline diabetics had a significantly increased risk for future development of diabetes, dementia, or AD, the risk effects were independent rather than linked.75 What this means is that insulin resistance, i.e., impaired ability to respond to insulin stimulation, can vary among target organs and be present in just one or two organs and not in others, a phenomenon that could explain the lack of complete overlap between T2DM and AD. Correspondingly, the finding that obesity (body mass index [BMI] > 30) without T2DM produces a three-fold increase in risk for subsequently developing AD whereas overweight, but nonobese, subjects (BMI 25–30) experience a two-fold increase in risk for AD76 calls into question the specific effects of obesity and T2DM versus a yet unknown associated factor in relation to AD pathogenesis.
Mechanistically, the increased risk of dementia in T2DM and obesity could be linked to chronic hyperglycemia, peripheral insulin resistance, oxidative stress, accumulation of advanced glycation end products, increased production of pro-inflammatory cytokines, and/orcerebral microvascular disease.73 The potential role of cerebral microvascular disease as a complicating, initiating, or accelerating component of AD has been recognized for years.77 However, a magnetic resonance imaging study demonstrated that older adults with T2DM have a moderately increased risk for developing lacunes and hippocampal atrophy and that the severity of those lesions increases with the duration and progression of T2DM.78 Another study showed that T2DM and impaired fasting glucose occur significantly more frequently in AD than in non-AD controls.79 However, since diffuse and neuritic plaques were similarly abundant in T2DM and control brains, and since neurofibrillary tangles, one of the hallmarks and correlates of dementia in AD, were not increased in T2DM,79 the results suggest that T2DM can enhance progression but may not be sufficient to cause AD. Therefore, what remains unclear is the net contribution of T2DM or obesity to the pathogenesis of AD-type neurodegeneration. To address this question, we utilized an established experimental model of chronic high-fat diet (HFD) feeding of C57BL/6 mice to examine the degree to which obesity/T2DM was sufficient to produce histopathological, molecular, and/or biochemical brain abnormalities of AD-type neurodegeneration, i.e., T3DM.
High-fat diet feeding for 16 weeks doubled mean body weight, caused T2DM, and marginally reduced mean brain weight.80 Those effects were associated with significantly increased levels of tau, IGF-1 receptor, IRS-1, IRS-4, ubiquitin, glial fibrillary acidic protein (GFAP), and 4-hydroxynonenal and decreased expression of β actin. Importantly, HFD feeding also caused brain insulin resistance manifested by reduced top-level (Bmax) insulin receptor binding and modestly increased brain insulin gene expression. However, HFD fed mouse brains did not exhibit AD histopathology or increases in APP-Aβ or phospho-tau, nor were there impairments in IGF signaling, which typically occurs in AD.10 In essence, although the chronic obesity with T2DM model exhibited mild brain atrophy with insulin resistance, oxidative stress, and cytoskeleton degradation, the effects were modest compared with AD5,10 and other more robust experimental models of T3DM,28,29 and most of the molecular, biochemical, and histopathological features that typify AD were not present. Therefore, T2DM and obesity may contribute to, i.e., serve as cofactors of AD but by themselves are probably not sufficient to cause AD. Moreover, the findings in the T2DM/obesity model indicate the unlikelihood that brain insulin resistance is sufficient to cause AD and that additional significant abnormalities, such as ongoing DNA damage and mitochondrial dysfunction, are required.
Alzheimer's Disease is Type 3 Diabetes: Evidence from Human Studies
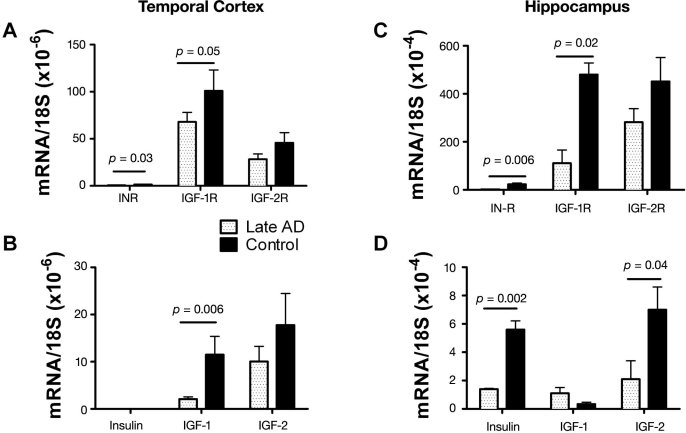
This hypothesis was directly investigated by first examining postmortem cases of advanced AD and determining if the neurodegeneration was associated with significant abnormalities in the expression of genes encoding insulin, IGF-1, and IGF-2 peptides, their receptors, and downstream signaling mechanisms.5 In that study, we demonstrated advanced AD to be associated with strikingly reduced levels of insulin and IGF-1 polypeptide and receptor genes in the brain (Figure 1). In addition, all the signaling pathways that mediate insulin and IGF-1-stimulated neuronal survival, tau expression, energy metabolism, and mitochondrial function were perturbed in AD. This study carries additional significance because it established that, like all other pancreatic and intestinal polypeptide genes, the insulin gene was also expressed in the adult human brain. Moreover, the results taught us that endogenous brain deficiencies in insulin, IGF-1, IGF-2, and their corresponding receptors, in the absence of T2DM or obesity, could be linked to the most common form of dementia-associated neurodegeneration in the Western hemisphere. Since the abnormalities identified in the brain were quite similar to the effects of T1DM or T2DM (though none of the patients had either of these diseases), including abnormalities in IGFs,81–83 which are important for islet cell function,84,85 we proposed the concept that AD may represent a brain-specific form of diabetes mellitus and coined the term “type 3 diabetes.”
Figure 1.
Impaired insulin and IGF (A, C) receptor and (B, D) polypeptide gene expression in late/end-stage AD (A, B) temporal cortex and (C, D) hippocampus.5 Gene expression was measured by qRT-PCR using RNA isolated from the temporal cortex or hippocampus from postmortem histopathologically confirmed cases of severe AD or normal aging. We reverse transcribed mRNA, and the resulting cDNA was PCR amplified. The products were detected continuously with a BIO-RAD iCycler Multi-Color RealTime PCR Detection System. Gene expression was normalized to 18S rRNA measured in the same samples. Graphs depict the mean ± standard deviation of results obtained from 28 AD and 26 control cases. Data were analyzed using Student's t-tests. Significant p-values are indicated over the bars. Note that insulin gene expression was not detected in temporal cortex.
Even before the initial study had been published, it was realized that if brain insulin/IGF resistance and insulin/IGF deficiency were causal in the pathogenesis of AD, the related abnormalities should be detectable in the early stages of disease and possibly worsen as disease progresses. The investigations were extended to examine the brains of patients with different degrees, i.e., Braak stages,86,87 of AD.10 In that study, we measured the expression of genes encoding insulin, IGF-1, IGF-2 polypeptides, and their corresponding receptors as well as tau and amyloid precursor protein (APP). In addition, we used competitive equilibrium and saturation binding assays to further characterize the degree to which growth factor-transmitted signaling was impaired in the brains with different severities of AD. Finally, the study included the measurement of steady-state levels of adenosine triphosphate and genes regulating acetylcholine homeostasis and energy metabolism.
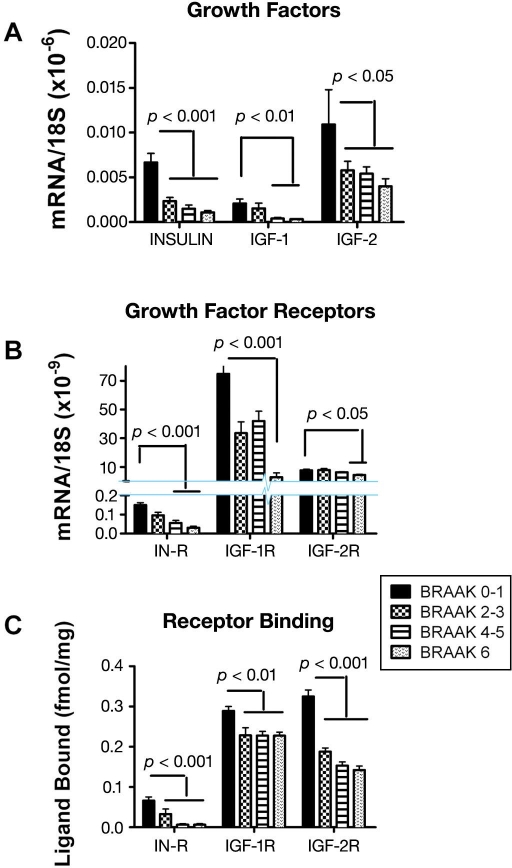
Using the previously mentioned approaches, we demonstrated progressive AD Braak stage-dependent reductions in insulin, IGF-1, and IGF-2 receptor expression, with more pronounced deterioration in insulin and IGF-1 compared with IGF-2 receptors, and the lowest levels of gene expression in brains with AD Braak Stage 6 (Figure 2). Therefore, loss of insulin and IGF-1 receptor-bearing neurons begins early and progresses with disease such that, in the advanced stages, the deficits are severe and global. These results provided further evidence that the abnormalities in AD are not restricted to insulin signaling pathways, as they also involve IGF-1 and IGF-2 stimulated mechanisms. Analysis of growth-factor polypeptide genes also revealed AD Braak stage-dependent impairments in insulin, IGF-1, and IGF-2 polypeptide expression, corresponding with progressive trophic factor withdrawal (Figure 2). Again, the results support the hypothesis that abnormalities in insulin and IGF signaling mechanisms begin early in the course of AD and are therefore likely have an important role in its pathogenesis.
Figure 2.
Brain insulin and IGF deficiency and resistance increase with progression of AD.10 Postmortem histopathological studies categorized the brains as having normal aging (Braak 0–1), or mild (Braak 2–3), moderate to severe (Braak 4–5), or end-stage (Braak 6) AD. We used mRNA isolated from fresh frozen frontal lobe tissue to measure insulin, IGF-1, or IGF-2 (A) polypeptide or (B) receptor gene expression by qRT-PCR. Results were normalized to 18S rRNA measured in the same samples. (C) For the competitive equilibrium binding assays, frontal lobe membrane protein extracts were incubated with [125I]-labeled insulin, IGF-1, or IGF-2 in the presence or absence of excess cold ligand. Radioactivity present in membrane protein precipitates was measured in a gamma counter. Specific binding (fmol/mg) was calculated using the GraphPad Prism 4 software. All graphs depict the mean ± standard deviation of results obtained from 9–12 cases per group. Intergroup comparisons were made using analysis of variance (ANOVA) with post hoc Tukey–Kramer significance tests. Significant p-values are indicated over the bars. Note axis break in Panel B.
The eventual paucity of local growth-factor gene expression could substantially impair growth-factor signaling and produce a state of growth-factor withdrawal, which is a well-established mechanism of neuronal death. Therefore, to complement the molecular data, we performed competitive equilibrium and saturation binding assays to determine if reduced levels of growth factor receptor expression were associated with and perhaps mediated by impaired ligand-receptor binding as occurs with insulin/IGF resistance. Those investigations demonstrated progressive declines in equilibrium (Figure 2) and top-level binding (Bmax) to the insulin, IGF-1, and IGF-2 receptors but either unchanged or increased binding affinity, suggesting that impaired insulin/IGF actions in AD brains were mediated by decreased polypeptide and receptor gene expression due to cell loss.
Through a series of in vitro and in vivo experiments performed by several groups, including our own, we have been able to draw the conclusion that neuronal and oligodendroglial cell survival and function are integrally related to the integrity of insulin and IGF signaling mechanisms in the brain.10,28,29,31,33,88,89 Similarly, impairments in insulin/IGF signaling lead to deficits in energy metabolism with attendant increased oxidative stress, mitochondrial dysfunction, proinflammatory cytokine activation, and APP expression.4,10,28,89 Correspondingly, the reduced expression of neuronal and oligodendroglial specific genes and the increased expression of astrocytic and microglial inflammatory genes in AD were attributed to progressive brain insulin/IGF deficiency and resistance. Although this point requires the generation of experimental models to demonstrate proof of principle, the finding that microglial, astrocytic, and APP mRNA levels are all increased in the early stages of neurodegeneration supports the inflammatory hypothesis of AD.6 Previous studies demonstrated that microglial activation promotes APP-Aβ accumulation90–92 and that APP gene expression and cleavage increase with oxidative stress.93 Therefore, the mechanism we propose is that impaired insulin/ IGF signaling leads to increased oxidative stress and mitochondrial dysfunction,32,94,95 which induces APP gene expression and cleavage.93 The attendant APP-Aβ accumulations cause local neurotoxicity96–98 and further increase in oxidative stress-induced APP expression and APP-Aβ deposition.
A critical goal in these investigations was to draw connections between brain insulin/IGF deficiency and resistance and the major dementia-associated structural and biochemical abnormalities in AD. In this regard, the postmortem studies demonstrated that the Braak stage-associated declines in tau mRNA paralleled the progressive reductions in insulin and IGF-1 receptor expression in AD. In addition, the studies demonstrated AD Braak stage-associated declines in choline acetyltransferase (ChAT) expression with reduced colocalization of ChAT with insulin or IGF-1 receptor immunoreactivity in cortical neurons. These results correspond with experimental data demonstrating that neuronal tau and ChAT gene expression are regulated by IGF-1 and insulin stimulation.88 Therefore, brain insulin and IGF deficiency and resistance could account for the cytoskeletal collapse, neurite retraction, synaptic disconnection, loss of neuronal plasticity, and deficiencies in acetylcholine production, all of which correlate with cognitive decline and dementia in AD. Altogether, the studies utilizing postmortem human brain tissue provide solid evidence that AD is associated with fundamental abnormalities in insulin/IGF signaling mechanisms that are highly correlated with development and progression of structural, molecular, and biochemical lesions that correlate with dementia. Although the abnormalities noted in AD share features in common with T1MD and T2MD, they are nonetheless distinguished by the dual presence of trophic factor deficiencies and trophic factor receptor resistance, ergo the term “type 3 diabetes.”
Alzheimer's Disease Is Type 3 Diabetes: Experimental Animal Model Results
The human postmortem brain studies linked many of the characteristic molecular and pathological features of AD to the reduced expression of the insulin and IGF genes and their corresponding receptors. However, without direct experimentation that generates cause–effect data, conclusions drawn from human studies would remain correlative rather than mechanistic. Consequently, we utilized experimental models to demonstrate that diabetes mellitus-type molecular and biochemical abnormalities could be produced in CNS neurons and brain by exposure to streptozotocin (STZ). Streptozotocin is 2-Deoxy-2{[methyl-nitrosoamino)carbonyl]amino}D-glucopyranose, i.e., a nitrosamide methylnitrosourea linked to the C2 position of D glucose. Once metabolized, the N nitrosoureido is liberated and causes DNA damage through generation of reactive oxygen species such as superoxide, hydrogen peroxide, and nitric oxide.99,100 Streptozotocin causes diabetes because it is taken up by insulin-producing cells, such as beta cells, in pancreatic islets.
We treated rats with a single intracerebral injection of STZ (ic-STZ) and allowed the rats to grow older for 2 to 8 weeks. The rats were subjected to Morris water maze tests of spatial learning and memory, and their brains were examined for histopathological, biochemical, and molecular indices of AD-type neurodegeneration.
Although a similar model had been generated much earlier by other investigators,101–104 and it was noted that the ic-STZ treatments reduced cerebral glucose utilization104 and oxidative metabolism,101 it inhibited insulin receptor function,95 and it caused progressive deficits in learning, memory, cognitive behavior, and cerebral energy balance,94,103 efforts were not made to connect these effects of ic-STZ to AD by characterizing the neuropathology, molecular pathology, abnormalities in genes expression pertinent to insulin and IGF-1 signaling in brain or by evaluating the integrity of the pancreas. Our goal in generating the model was to demonstrate that AD-type neurodegeneration with features of T3DM could be produced in the absence of either T1DM or T2DM.
The ic-STZ-injected rats did not have elevated blood glucose or insulin levels, and pancreatic architecture and insulin immunoreactivity were similar to control, yet their brains were atrophied and had striking evidence of neurodegeneration with cell loss, gliosis, and increased immunoreactivity for p53, activated GSK-3β, phospho-tau, ubiquitin, and APP-Aβ.28,29 Moreover, quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) studies demonstrated that the ic-STZ-treated brains had significantly reduced expression of genes corresponding to neurons (Hu), oligodendroglia [myelin-associated glycoprotein-1 (MAG-1)], and ChAT and to increased expression of genes encoding GFAP, microglia-specific proteins [allograft inflammatory factor-1 (AiF-1)]), acetylcholinesterase (AChE), tau, and APP.28,29 Increased p53 and decreased Hu and MAG-1 expression in ic-STZ-treated brains suggest that neuronal and oligodendroglial cell loss and cerebral atrophy were mediated by apoptosis. These findings correspond well with previous studies demonstrating increased expression of various proapoptosis molecules, including p53,105,106 colocalization of increased p53 immunoreactivity in neurons and white matter glia, and reduced levels of Hu and MAG-1 mRNA in human brains with AD. Loss of oligodendroglia could contribute to the early white matter degeneration107 and synaptic disconnection108–111in AD.
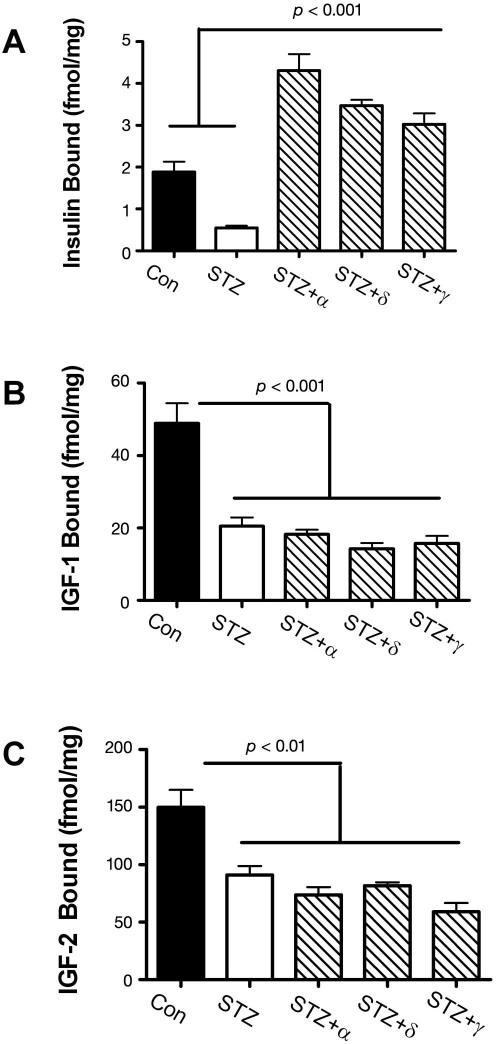
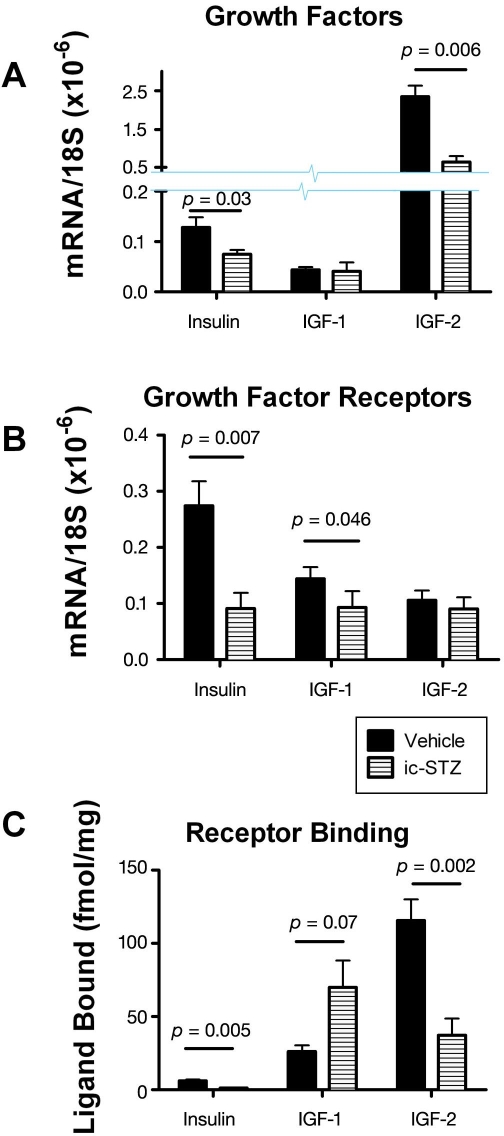
The previously mentioned adverse effects of ic-STZ were associated with reduced expression of genes encoding insulin, IGF-2, insulin receptor, IGF-1 receptor, and IRS-1 and reduced ligand binding to the insulin and IGF-2 receptors (Figure 3). Note that most of these effects were also detected in brains with sporadic AD5 and were found to increase with disease progression.10 The reduced levels of IRS-1 mRNA observed in both AD and rats treated with ic-STZ were reminiscent of the murine IRS-1 and insulin receptor knock-out models, which exhibit reduced brain and body weights due to impaired insulin stimulated growth and survival signaling.23,112,113 The combined effects of reduced insulin/IGF polypeptide gene expression, receptor expression, receptor binding, and IRS expression all point toward failure of insulin/IGF signaling mechanisms in the brain as a major consequence of ic-STZ treatment. Importantly, many molecular abnormalities that characteristically occur in AD and were produced by ic-STZ, including increased GSK-3β activation, increased tau phosphorylation, and decreased neuronal survival, could be mediated by downstream effects of impaired insulin and IGF signaling in the CNS. Again, similar results have been reported by other investigators using this experimental model of neurodegeneration.114–117 Therefore, the ic-STZ experimental animal model recapitulates many of the characteristic features of AD-type neurodegeneration/T3DM.
Figure 3.
Effects of intracerebral ic-STZ treatment on CNS expression of insulin and IGF (A) genes and (B) receptors and (C) ligand binding to the insulin, IGF-1, or IGF-2 receptors in temporal lobe tissue. Rat pups were given 50 mg/kg ic-STZ or vehicle and sacrificed 14 days later. Temporal lobe mRNA was used to measure gene expression by qRT-PCR, and results were normalized to 18S rRNA measured in the same samples.29 Note axis break in Panel A. (C) Competitive equilibrium binding assays were used to measure specific binding to the insulin, IGF-1, or IGF-2 receptors as described in Figure 2. Graphs depict the mean ± standard error of the mean of results. Data were analyzed statistically using Student's t-tests. Significant p-values are indicated over the bar graphs.
Corresponding with the findings in AD,5 the ic-STZ-treated brains had increased levels of activated GSK-3β, phospho-tau, ubiquitin, APP and APP-Aβ and decreased levels of tau protein. These results are consistent with previous studies demonstrating that tau is regulated by insulin/IGF-1 stimulation88,118 and that tau phosphorylation and ubiquitination increase with oxidative stress and activation of GSK-3β.93 Similarly, APP mRNA increases with oxidative stress and is a feature of sporadic AD.5,10 Increased APP gene expression could account for APP-Aβ accumulation in AD and ic-STZ-treated brains. Potential sources of oxidative stress in AD and the ic-STZ model include (1) mitochondrial dysfunction;6,53,95 (2) microglial cell activation with increased cytokine release; and (3) impaired insulin/IGF signaling through PI3 kinaseAkt, leading to increased levels of GSK-3β activity.
A crucial step was to determine whether ic-STZ could cause disturbances in acetylcholine homeostasis and cognitive impairment as they occur in AD. QRT-PCR and immunohistochemistry detected reduced levels of ChAT and increased levels of AChE mRNA and protein in icSTZ-treated brains relative to control brains. Note that energy metabolism leads to production of Acetyl-CoA, which is needed to make acetylcholine. Since the ChAT gene is responsive to insulin and IGF-1 stimulation, deficits in insulin/IGF signaling and energy metabolism push in the direction of cholinergic deficiency mediated by impaired energy metabolism and decreased expression of ChAT, which are key features in AD. In addition, increased levels of AChE expression in the ic-STZ brains could result in increased degradation of acetylcholine, thereby exacerbating the acetylcholine deficits caused by reduced ChAT expression. The significance of these results is highlighted by the prominent learning and memory deficits detected in ic-STZ-treated rats.28,29
Type 3 Diabetes May Be Treatable, Preventable, or Curable with Antidiabetes Drugs
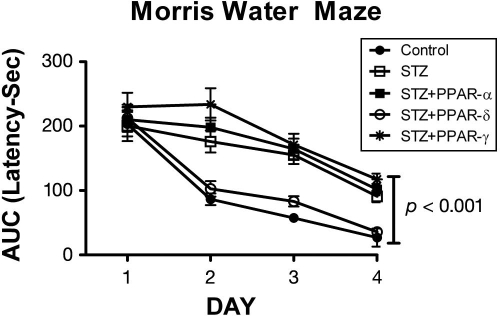
The findings that (1) pronounced insulin/IGF deficiency and resistance develop early in the course of AD; (2) insulin/IGF signaling abnormalities progress with severity of neurodegeneration;5,10 and (3) an experimental animal model with features closely mimicking the molecular, biochemical, and neuroanatomical pathologies of AD could be generated by intracerebral delivery of a drug that causes T1DM or T2DM led us to test the hypothesis that AD-type neurodegeneration and cognitive could be reduced or prevented by early treatment with insulin-sensitizer antidiabetes agents such as peroxisome proliferator-activated receptor (PPAR) agonists. Peroxisome proliferator-activated receptor agonists function at the level of the nucleus to activate insulin-responsive genes and signaling mechanisms. PPAR-α, PPAR-δ, and PPAR-γ are all expressed in adult human brains, including AD, but PPAR-δ is the most abundant of the three isoforms.6 The experimental design involved treating rats with ic-STZ, followed by a single intraperitoneal injection of saline, a PPAR-α (GW7647; 25 µg/kg), PPAR-δ (L-160,043; 2 µg/kg), or PPAR-γ (F-L-Leu; 20 µg/kg) activator (CalBiochem, Carlsbad, CA).28 The doses used were considerably lower than those routinely given to treat T2DM. The major effects of the PPAR agonist treatments were to prevent brain atrophy, preserve insulin and IGF-2 receptor bearing CNS neurons, and particularly with regard to the PPAR-δ agonist, prevent ic-STZ-induced deficits in learning and memory.28 Since the ic-STZ-mediated losses of insulin and IGF-expressing cells were not prevented by the PPAR agonist treatments, the PPAR agonists probably functioned by preserving insulin and IGF responsive (receptor-bearing) cells, including neurons and oligodendroglia. In support of this concept was finding that insulin receptor expression and binding were increased by the PPAR agonist treatments (Figure 4). Peroxisome proliferator-activated receptor agonist mediated preservation of insulin/IGF responsive neurons was associated with increased expression of ChAT, which has an important role in cognition, as cholinergic neuron deficits are a fundamental feature of AD.119–122 Importantly, the PPAR-δ agonist mediated increases in insulin binding, and ChAT were associated with significant improvements in learning and spatial memory tasks as demonstrated using Morris water maze tests28 (Figure 5). These effects of the PPAR agonist treatments are consistent with the facts that ChAT expression is regulated by insulin/IGF88,118 and insulin/IGF resistance mediates cognitive impairment in AD. The PPAR-mediated increases in MAG-1 expression, corresponding to oligodendroglia, were of particular interest because previous research demonstrated that one of the earliest AD lesions was white matter atrophy and degeneration with loss of oligodendroglial cells.107 Within the context of the present discussion, white matter atrophy in AD can now be interpreted as a manifestation of CNS insulin/IGF resistance since oligodendroglia require intact insulin/IGF signaling mechanisms for survival and function, including myelin synthesis.123,124 Besides preserving insulin and IGF receptor-bearing CNS cells and signaling mechanisms germane to survival, energy metabolism, and neurotransmitter functions, the PPAR agonists rescued the ic-STZ model by lowering critical AD-associated indices of oxidative stress, including microglial and astrocyte activation, p53, nitric oxide synthase and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase gene expression, lipid peroxidation, DNA damage, APP expression, and tau phosphorylation.6,28,29,91,92,125,126
Figure 4.
Treatment with PPAR agonists restores brain insulin receptor binding in ic-STZ-treated rats.28 Long Evans rat pups were treated with 50 mg/kg ic-STZ or vehicle and sacrificed 30 days later to examine brains for insulin and IGF polypeptide and receptor gene expression and insulin and IGF receptor binding. Temporal lobe membrane protein extracts were used in competitive equilibrium binding assays to measure specific binding to the (A) insulin, (B) IGF-1, or (C) IGF-2 receptors as described in Figure 2. Graphs depict the mean ± standard error of the mean of results. Data were analyzed using ANOVA with the Tukey–Kramer post hoc significance test. Significant p-values are shown within each panel.
Figure 5.
Peroxisome proliferator-activated receptor-δ agonist treatment preserves visual-spatial learning and memory in ic-STZ-treated rats.28 Long Evans rat pups were treated with 50 mg/kg ic-STZ or vehicle, followed by a single intraperitoneal injection of a PPAR-α (GW7647; 25 µg/kg), PPAR-δ (L-160,043; 2 µg/kg), or PPAR-γ (F-L-Leu; 20 µg/kg) agonist (n = 8 rats per group). Four weeks later, the rats were subjected to Morris water maze testing, in which the latency required to locate the hidden platform was measured for 3 independent trials on 4 consecutive days. Area under the curve (AUC) was computed for the 3 daily trials. Graphs depict the mean AUC ± standard error of the mean for latency (seconds) in each group. Data were analyzed using ANOVA with the Tukey–Kramer post hoc significance test. Performance in the control and ic-STZ + PPAR-δ groups were similar, and on Days 2, 3, and 4, their mean latencies required to locate the hidden platform were significantly shorter than in the other 3 groups.
Conclusions
Altogether, the results from these studies provide strong evidence in support of the hypothesis that AD represents a form of diabetes mellitus that selectively afflicts the brain. Positive data stemmed from (1) direct analysis of postmortem human brains with documented AD; (2) an experimental animal model in which brain diabetes with cognitive impairment and molecular and pathological features that mimic AD was produced by intracerebral administration of a drug that is commonly used to produce T1DM or T2DM; and (3) a study showing that PPAR agonists, which are used to treat T2DM, prevent many of the AD-associated neurodegenerative effects of ic-STZ. The data are supported by abundant in vitro experiments that demonstrated essentially the same or similar effects of STZ or oxidative stress treatments of neuronal cells. The human and experimental animal model studies also showed that CNS impairments in insulin/IGF signaling mechanisms can occur in the absence of T1DM or T2DM. Finally, we demonstrated that although obesity with T2DM causes brain insulin resistance with some features of AD-type neurodegeneration, the effects are relatively modest, not associated with significant histopathological lesions, and lack most of the critical abnormalities that typify AD. Therefore, T2DM was deemed not sufficient to cause AD, although it could possibly serve as a cofactor in its pathogenesis or progression. Altogether, the data provide strong evidence that AD is intrinsically a neuroendocrine disease caused by selective impairments in insulin and IGF signaling mechanisms, including deficiencies in local insulin and IGF production. At the same time, it is essential to recognize that T2DM and T3DM are not solely the end results of insulin/IGF resistance and/or deficiency, because these syndromes are unequivocally accompanied by significant activation of inflammatory mediators, oxidative stress, DNA damage, and mitochondrial dysfunction, which contribute to the degenerative cascade by exacerbating insulin/ IGF resistance. Referring to AD as T3DM is justified, because the fundamental molecular and biochemical abnormalities overlap with T1DM and T2DM rather than mimic the effects of either one. Some of the most relevant data supporting this concept have emerged from clinical studies demonstrating cognitive improvement and/or stabilization of cognitive impairment in subjects with early AD following treatment with intranasal insulin or a PPAR agonist.58,60,127–130
Abbreviations
- AChE
acetylcholinesterase
- AD
Alzheimer's disease
- ANOVA
analysis of variance
- AAP
amyloid precursor protein
- APP-Aβ
amyloid precursor protein, amyloid-β
- AUC
area under the curve
- BMI
body mass index
- ChAT
choline acetyltransferase
- CNS
central nervous system
- GFAP
glial fibrillary acidic protein
- GSK-3β
glycogen synthase kinase 3β
- HFD
high-fat diet
- ic-STZ
intracerebral injection of streptozotocin
- IGF
insulin-like growth factor
- IRS
insulin receptor substrate
- MAG-1
myelin-associated glycoprotein
- MCI
mild cognitive impairment
- NASH
nonalcoholic steatohepatitis
- PI3
phosphatidyl-inositol-3
- PPAR
peroxisome proliferator-activated receptor
- qRT-PCR
quantitative reverse transcriptase polymerase chain reaction
- STZ
streptozotocin
- T1DM
type 1 diabetes mellitus
- T2DM
type 2 diabetes mellitus
- T3DM
type 3 diabetes mellitus
References
- 1.Jalbert JJ, Daiello LA, Lapane KL. Dementia of the Alzheimer Type. Epidemiol Rev. 2008 doi: 10.1093/epirev/mxn008. [Epub ahead of print.] [DOI] [PubMed] [Google Scholar]
- 2.Jellinger KA. Neuropathological aspects of Alzheimer disease, Parkinson disease and frontotemporal dementia. Neurodegener Dis. 2008;5(3-4):118–121. doi: 10.1159/000113679. [DOI] [PubMed] [Google Scholar]
- 3.Wang XP, Ding HL. Alzheimer's disease: epidemiology, genetics, and beyond. Neurosci Bull. 2008;24(2):105–109. doi: 10.1007/s12264-008-0105-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de la Monte SM, Wands JR. Review of insulin and insulin-like growth factor expression, signaling, and malfunction in the central nervous system: relevance to Alzheimer's disease. J Alzheimers Dis. 2005;7(1):45–61. doi: 10.3233/jad-2005-7106. [DOI] [PubMed] [Google Scholar]
- 5.Steen E, Terry BM, Rivera EJ, Cannon JL, Neely TR, Tavares R, Xu XJ, Wands JR, de la Monte SM. Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer's disease—is this type 3 diabetes? J Alzheimers Dis. 2005;7(1):63–80. doi: 10.3233/jad-2005-7107. [DOI] [PubMed] [Google Scholar]
- 6.de la Monte SM, Wands JR. Molecular indices of oxidative stress and mitochondrial dysfunction occur early and often progress with severity of Alzheimer's disease. J Alzheimers Dis. 2006;9(2):167–181. doi: 10.3233/jad-2006-9209. [DOI] [PubMed] [Google Scholar]
- 7.Moreira PI, Santos MS, Seiça R, Oliveira CR. Brain mitochondrial dysfunction as a link between Alzheimer's disease and diabetes. J Neurol Sci. 2007;257(1-2):206–214. doi: 10.1016/j.jns.2007.01.017. [DOI] [PubMed] [Google Scholar]
- 8.Hoyer S. The brain insulin signal transduction system and sporadic (type II) Alzheimer disease: an update. J Neural Transm. 2002;109(3):341–360. doi: 10.1007/s007020200028. [DOI] [PubMed] [Google Scholar]
- 9.Nixon RA. The calpains in aging and aging-related diseases. Ageing Res Rev. 2003;2(4):407–418. doi: 10.1016/s1568-1637(03)00029-1. [DOI] [PubMed] [Google Scholar]
- 10.Rivera EJ, Goldin A, Fulmer N, Tavares R, Wands JR, de la Monte SM. Insulin and insulin-like growth factor expression and function deteriorate with progression of Alzheimer's disease: link to brain reductions in acetylcholine. J Alzheimers Dis. 2005;8(3):247–268. doi: 10.3233/jad-2005-8304. [DOI] [PubMed] [Google Scholar]
- 11.Revill P, Moral MA, Prous JR. Impaired insulin signaling and the pathogenesis of Alzheimer's disease. Drugs Today (Barc) 2006;42(12):785–790. doi: 10.1358/dot.2006.42.12.1032059. [DOI] [PubMed] [Google Scholar]
- 12.Iwangoff P, Armbruster R, Enz A, Meier-Ruge W. Glycolytic enzymes from human autoptic brain cortex: normal aged and demented cases. Mech Ageing Dev. 1980;14(1-2):203–209. doi: 10.1016/0047-6374(80)90120-7. [DOI] [PubMed] [Google Scholar]
- 13.Sims NR, Bowen DM, Smith CC, Flack RH, Davison AN, Snowden JS, Neary D. Glucose metabolism and acetylcholine synthesis in relation to neuronal activity in Alzheimer's disease. Lancet. 1980;1(8164):333–336. doi: 10.1016/s0140-6736(80)90884-3. [DOI] [PubMed] [Google Scholar]
- 14.Hoyer S. Causes and consequences of disturbances of cerebral glucose metabolism in sporadic Alzheimer disease: therapeutic implications. Adv Exp Med Biol. 2004;541:135–152. doi: 10.1007/978-1-4419-8969-7_8. [DOI] [PubMed] [Google Scholar]
- 15.Virkamäki A, Ueki K, Kahn CR. Protein-protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. J Clin Invest. 1999;103(7):931–943. doi: 10.1172/JCI6609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alvarez-Martínez H, Pérez-Campos E. [Non-alcoholic steatohepatitis] Rev Gastroenterol Mex. 2002;67(2):118–125. [PubMed] [Google Scholar]
- 17.Solís Herruzo JA, García Ruiz I, Pérez Carreras M, Muñoz Yagüe MT. Non-alcoholic fatty liver disease. From insulin resistance to mitochondrial dysfunction. Rev Esp Enferm Dig. 2006;98(11):844–874. doi: 10.4321/s1130-01082006001100006. [DOI] [PubMed] [Google Scholar]
- 18.Saito T, Misawa K, Kawata S. 1 Fatty liver and non-alcoholic steatohepatitis. Intern Med. 2007;46(2):101–103. doi: 10.2169/internalmedicine.46.1784. [DOI] [PubMed] [Google Scholar]
- 19.Craft S, Asthana S, Cook DG, Baker LD, Cherrier M, Purganan K, Wait C, Petrova A, Latendresse S, Watson GS, Newcomer JW, Schellenberg GD, Krohn AJ. Insulin dose-response effects on memory and plasma amyloid precursor protein in Alzheimer's disease: interactions with apolipoprotein-E genotype. Psychoneuroendocrinology. 2003;28(6):809–822. doi: 10.1016/s0306-4530(02)00087-2. [DOI] [PubMed] [Google Scholar]
- 20.Craft S, Asthana S, Schellenberg G, Baker L, Cherrier M, Boyt AA, Martins RN, Raskind M, Peskind E, Plymate S. Insulin effects on glucose metabolism, memory, and plasma amyloid precursor protein in Alzheimer's disease differ according to apolipoprotein-E genotype. Ann N Y Acad Sci. 2000;903:222–228. doi: 10.1111/j.1749-6632.2000.tb06371.x. [DOI] [PubMed] [Google Scholar]
- 21.Farris W, Mansourian S, Leissring MA, Eckman EA, Bertram L, Eckman CB, Tranzi RE, Selkoe DJ. Partial loss-of-function mutations in insulin-degrading enzyme that induce diabetes also impair degradation of amyloid beta-protein. Am J Pathol. 2004;164(4):1425–1434. doi: 10.1016/s0002-9440(10)63229-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hoyer S. Glucose metabolism and insulin receptor signal transduction in Alzheimer disease. Eur J Pharmacol. 2004;490(1-3):115–125. doi: 10.1016/j.ejphar.2004.02.049. [DOI] [PubMed] [Google Scholar]
- 23.Schubert M, Brazil DP, Burks DJ, Kushner JA, Ye J, Flint CL, Farhang-Fallah J, Dikkes P, Warot XM, Rio C, Corfas G, White MF. Insulin receptor substrate-2 deficiency impairs brain growth and promotes tau phosphorylation. J Neurosci. 2003;23(18):7084–7092. doi: 10.1523/JNEUROSCI.23-18-07084.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schubert M, Gautam D, Surjo D, Ueki K, Baudler S, Schubert D, Kondo T, Alber J, Galldiks N, Küstermann E, Arndt S, Jacobs AH, Krone W, Kahn CR, Brüning JC. Role for neuronal insulin resistance in neurodegenerative diseases. Proc Natl Acad Sci U S A. 2004;101(9):3100–3105. doi: 10.1073/pnas.0308724101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Craft S. Insulin resistance and cognitive impairment: a view through the prism of epidemiology. Arch Neurol. 2005;62(7):1043–1044. doi: 10.1001/archneur.62.7.1043-a. [DOI] [PubMed] [Google Scholar]
- 26.Craft S. Insulin resistance syndrome and Alzheimer disease: pathophysiologic mechanisms and therapeutic implications. Alzheimer Dis Assoc Disord. 2006;20(4):298–301. doi: 10.1097/01.wad.0000213866.86934.7e. [DOI] [PubMed] [Google Scholar]
- 27.Craft S. Insulin resistance and Alzheimer's disease pathogenesis: potential mechanisms and implications for treatment. Curr Alzheimer Res. 2007;4(2):147–152. doi: 10.2174/156720507780362137. [DOI] [PubMed] [Google Scholar]
- 28.de la Monte SM, Tong M, Lester-Coll N, Plater M, Jr, Wands JR. Therapeutic rescue of neurodegeneration in experimental type 3 diabetes: relevance to Alzheimer's disease. J Alzheimers Dis. 2006;10(1):89–109. doi: 10.3233/jad-2006-10113. [DOI] [PubMed] [Google Scholar]
- 29.Lester-Coll N, Rivera EJ, Soscia SJ, Doiron K, Wands JR, de la Monte SM. Intracerebral streptozotocin model of type 3 diabetes: relevance to sporadic Alzheimer's disease. J Alzheimers Dis. 2006;9(1):13–33. doi: 10.3233/jad-2006-9102. [DOI] [PubMed] [Google Scholar]
- 30.de la Monte SM, Ganju N, Banerjee K, Brown NV, Luong T, Wands JR. Partial rescue of ethanol-induced neuronal apoptosis by growth factor activation of phosphoinositol-3-kinase. Alcohol Clin Exp Res. 2000;24(5):716–726. [PubMed] [Google Scholar]
- 31.de la Monte SM, Neely TR, Cannon J, Wands JR. Ethanol impairs insulin-stimulated mitochondrial function in cerebellar granule neurons. Cell Mol Life Sci. 2001;58(12-13):1950–1960. doi: 10.1007/PL00000829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.de la Monte SM, Wands JR. Chronic gestational exposure to ethanol impairs insulin-stimulated survival and mitochondrial function in cerebellar neurons. Cell Mol Life Sci. 2002;59(5):882–893. doi: 10.1007/s00018-002-8475-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xu J, Yeon JE, Chang H, Tison G, Chen GJ, Wands J, de la Monte S. Ethanol impairs insulin-stimulated neuronal survival in the developing brarole of PTEN phosphatase. J Biol Chem. 2003;278(29):26929–26937. doi: 10.1074/jbc.M300401200. [DOI] [PubMed] [Google Scholar]
- 34.Myers MG, Sun XJ, White MF. The IRS-1 signaling system. Trends Biochem Sci. 1994;19(7):289–293. doi: 10.1016/0968-0004(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 35.O'Hare T, Pilch PF. Intrinsic kinase activity of the insulin receptor. Int J Biochem. 1990;22(4):315–324. doi: 10.1016/0020-711x(90)90132-m. [DOI] [PubMed] [Google Scholar]
- 36.Ullrich A, Bell JR, Chen EY, Herrera R, Petruzzelli LM, Dull TJ, Gray A, Coussens L, Liao YC, Tsubokawa M, et al. Human insulin receptor and its relationship to the tyrosine kinase family of oncogenes. Nature. 1985;313(6005):756–761. doi: 10.1038/313756a0. [DOI] [PubMed] [Google Scholar]
- 37.Sun XJ, Rothenberg P, Kahn CR, Backer JM, Araki E, Wilden PA, Cahill DA, Goldstein BJ, White MF. Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein. Nature. 1991;352(6330):73–77. doi: 10.1038/352073a0. [DOI] [PubMed] [Google Scholar]
- 38.White MF, Maron R, Kahn CR. Insulin rapidly stimulates tyrosine phosphorylation of a Mr-185,000 protein in intact cells. Nature. 1985;318(6042):183–186. doi: 10.1038/318183a0. [DOI] [PubMed] [Google Scholar]
- 39.Sun XJ, Crimmins DL, Myers MJ, Jr, Miralpeix M, White MF. Pleiotropic insulin signals are engaged by multisite phosphorylation of IRS-1. Mol Cell Biol. 1993;13(12):7418–7428. doi: 10.1128/mcb.13.12.7418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Burgering BM, Coffer PJ. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature. 1995;376(6541):599–602. doi: 10.1038/376599a0. [DOI] [PubMed] [Google Scholar]
- 41.Delcommenne M, Tan C, Gray V, Rue L, Woodgett J, Dedhar S. Phosphoinositide-3-OH kinase-dependent regulation of glycogen synthase kinase 3 and protein kinase B/AKT by the integrin-linked kinase. Proc Natl Acad Sci U S A. 1998;95(19):11211–11216. doi: 10.1073/pnas.95.19.11211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dudek H, Datta SR, Franke TF, Birnbaum MJ, Yao R, Cooper GM, Segal RA, Kaplan DR, Greenberg ME. Regulation of neuronal survival by the serine-threonine protein kinase Akt. Science. 1997;275(5300):661–665. doi: 10.1126/science.275.5300.661. [See comments.] [DOI] [PubMed] [Google Scholar]
- 43.Kulik G, Klippel A, Weber MJ. Antiapoptotic signalling by the insulin-like growth factor I receptor, phosphatidylinositol 3-kinase, and Akt. Mol Cell Biol. 1997;17(3):1595–1606. doi: 10.1128/mcb.17.3.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lam K, Carpenter CL, Ruderman NB, Friel JC, Kelly KL. The phosphatidylinositol 3-kinase serine kinase phosphorylates IRS 1. Stimulation by insulin and inhibition by Wortmannin. J Biol Chem. 1994;269(32):20648–20652. [PubMed] [Google Scholar]
- 45.Mill JF, Chao MV, Ishii DN. Insulin, insulin-like growth factor II, and nerve growth factor effects on tubulin mRNA levels and neurite formation. Proc Natl Acad Sci U S A. 1985;82(20):7126–7130. doi: 10.1073/pnas.82.20.7126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Puro DG, Agardh E. Insulin-mediated regulation of neuronal maturation. Science. 1984;225(4667):1170–1172. doi: 10.1126/science.6089343. [DOI] [PubMed] [Google Scholar]
- 47.Pasquier F, Boulogne A, Leys D, Fontaine P. Diabetes mellitus and dementia. Diabetes Metab. 2006;32(5 Pt 1):403–414. doi: 10.1016/s1262-3636(07)70298-7. [DOI] [PubMed] [Google Scholar]
- 48.Verdelho A, Madureira S, Ferro JM, Basile AM, Chabriat H, Erkinjuntti T, Fazekas F, Hennerici M, O'Brien J, Pantoni L, Salvadori E, Scheltens P, Visser MC, Wahlund LO, Waldemar G, Wallin A, Inzitari D. LADIS Study. Differential impact of cerebral white matter changes, diabetes, hypertension and stroke on cognitive performance among non-disabled elderly. The LADIS study. J Neurol Neurosurg Psychiatry. 2007;78(12):1325–1330. doi: 10.1136/jnnp.2006.110361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Martins IJ, Hone E, Foster JK, Sünram-Lea SI, Gnjec A, Fuller SJ, Nolan D, Gandy SE, Martins RN. Apolipoprotein E, cholesterol metabolism, diabetes, and the convergence of risk factors for Alzheimer's disease and cardiovascular disease. Mol Psychiatry. 2006;11(8):721–736. doi: 10.1038/sj.mp.4001854. [DOI] [PubMed] [Google Scholar]
- 50.Winocur G, Greenwood CE. Studies of the effects of high fat diets on cognitive function in a rat model. Neurobiol Aging. 2005;26(Suppl 1):46–49. doi: 10.1016/j.neurobiolaging.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 51.Winocur G, Greenwood CE, Piroli GG, Grillo CA, Reznikov LR, Reagan LP, McEwen BS. Memory impairment in obese Zucker rats: an investigation of cognitive function in an animal model of insulin resistance and obesity. Behav Neurosci. 2005;119(5):1389–1395. doi: 10.1037/0735-7044.119.5.1389. [DOI] [PubMed] [Google Scholar]
- 52.Biju MP, Paulose CS. Brain glutamate dehydrogenase changes in streptozotocin diabetic rats as a function of age. Biochem Mol Biol Int. 1998;44(1):1–7. doi: 10.1080/15216549800201002. [DOI] [PubMed] [Google Scholar]
- 53.Hoyer S, Lannert H, Nöldner M, Chatterjee SS. Damaged neuronal energy metabolism and behavior are improved by Ginkgo biloba extract (EGb 761) J Neural Transm. 1999;106(11-12):1171–1188. doi: 10.1007/s007020050232. [DOI] [PubMed] [Google Scholar]
- 54.Nitta A, Murai R, Suzuki N, Ito H, Nomoto H, Katoh G, Furukawa Y, Furukawa S. Diabetic neuropathies in brain are induced by deficiency of BDNF. Neurotoxicol Teratol. 2002;24(5):695–701. doi: 10.1016/s0892-0362(02)00220-9. [DOI] [PubMed] [Google Scholar]
- 55.Weinstock M, Shoham S. Rat models of dementia based on reductions in regional glucose metabolism, cerebral blood flow and cytochrome oxidase activity. J Neural Transm. 2004;111(3):347–366. doi: 10.1007/s00702-003-0058-y. [DOI] [PubMed] [Google Scholar]
- 56.Haan MN. Therapy insight: type 2 diabetes mellitus and the risk of late-onset alzheimer's disease. Nat Clin Pract Neurol. 2006;2(3):159–166. doi: 10.1038/ncpneuro0124. [DOI] [PubMed] [Google Scholar]
- 57.Landreth G. PPARgamma agonists as new therapeutic agents for the treatment of Alzheimer's disease. Exp Neurol. 2006;199(2):245–248. doi: 10.1016/j.expneurol.2006.04.006. [DOI] [PubMed] [Google Scholar]
- 58.Landreth G. Therapeutic use of agonists of the nuclear receptor PPARgamma in Alzheimer's disease. Curr Alzheimer Res. 2007;4(2):159–164. doi: 10.2174/156720507780362092. [DOI] [PubMed] [Google Scholar]
- 59.Pedersen WA, McMillan PJ, Kulstad JJ, Leverenz JB, Craft S, Haynatzki GR. Rosiglitazone attenuates learning and memory deficits in Tg2576 Alzheimer mice. Exp Neurol. 2006;199(2):265–273. doi: 10.1016/j.expneurol.2006.01.018. [DOI] [PubMed] [Google Scholar]
- 60.Reger MA, Watson GS, Frey WH, 2nd, Baker LD, Cholerton B, Keeling ML, Belongia DA, FIshel MA, Plymate SR, Schellenberg GD, Cherrier MM, Craft S. Effects of intranasal insulin on cognition in memory-impaired older adults: modulation by APOE genotype. Neurobiol Aging. 2006;27(3):451–458. doi: 10.1016/j.neurobiolaging.2005.03.016. [DOI] [PubMed] [Google Scholar]
- 61.Reger MA, Watson GS, Green PS, Wilkinson CW, Baker LD, Cholerton B, Fishel MA, Plymate SR, Breitner JC, DeGroodt W, Mehta P, Craft S. Intranasal insulin improves cognition and modulates beta-amyloid in early AD. Neurology. 2008;70(6):440–448. doi: 10.1212/01.WNL.0000265401.62434.36. [DOI] [PubMed] [Google Scholar]
- 62.Watson GS, Bernhardt T, Reger MA, Cholerton BA, Baker LD, Peskin ER, Asthana S, Plymate SR, Frölich L, Craft S. Insulin effects on CSF norepinephrine and cognition in Alzheimer's disease. Neurobiol Aging. 2006;27(1):38–41. doi: 10.1016/j.neurobiolaging.2004.11.011. [DOI] [PubMed] [Google Scholar]
- 63.Marchesini G, Marzocchi R. Metabolic syndrome and NASH. Clin Liver Dis. 2007;11(1):105–117. ix. doi: 10.1016/j.cld.2007.02.013. [DOI] [PubMed] [Google Scholar]
- 64.Nicolls MR. The clinical and biological relationship between type II diabetes mellitus and Alzheimer's disease. Curr Alzheimer Res. 2004;1(1):47–54. doi: 10.2174/1567205043480555. [DOI] [PubMed] [Google Scholar]
- 65.Papandreou D, Rousso I, Mavromichalis I. Update on non-alcoholic fatty liver disease in children. Clin Nutr. 2007;26(4):409–415. doi: 10.1016/j.clnu.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 66.Pessayre D. Role of mitochondria in non-alcoholic fatty liver disease. J Gastroenterol Hepatol. 2007;22(Suppl 1):S20–S27. doi: 10.1111/j.1440-1746.2006.04640.x. [DOI] [PubMed] [Google Scholar]
- 67.Yeh MM, Brunt EM. Pathology of nonalcoholic fatty liver disease. Am J Clin Pathol. 2007;128(5):837–847. doi: 10.1309/RTPM1PY6YGBL2G2R. [DOI] [PubMed] [Google Scholar]
- 68.Qiu C, De Ronchi D, Fratiglioni L. The epidemiology of the dementias: an update. Curr Opin Psychiatry. 2007;20(4):380–385. doi: 10.1097/YCO.0b013e32816ebc7b. [DOI] [PubMed] [Google Scholar]
- 69.Haan MN, Wallace R. Can dementia be prevented? Brain aging in a population-based context. Annu Rev Public Health. 2004;25:1–24. doi: 10.1146/annurev.publhealth.25.101802.122951. [DOI] [PubMed] [Google Scholar]
- 70.Launer LJ. Diabetes and brain aging: epidemiologic evidence. Curr Diab Rep. 2005;5(1):59–63. doi: 10.1007/s11892-005-0069-1. [DOI] [PubMed] [Google Scholar]
- 71.Luchsinger JA, Mayeux R. Cardiovascular risk factors and Alzheimer's disease. Curr Atheroscler Rep. 2004;6(4):261–266. doi: 10.1007/s11883-004-0056-z. [DOI] [PubMed] [Google Scholar]
- 72.Luchsinger JA, Reitz C, Patel B, Tang MX, Manly JJ, Mayeux R. Relation of diabetes to mild cognitive impairment. Arch Neurol. 2007;64(4):570–575. doi: 10.1001/archneur.64.4.570. [DOI] [PubMed] [Google Scholar]
- 73.Whitmer RA. Type 2 diabetes and risk of cognitive impairment and dementia. Curr Neurol Neurosci Rep. 2007;7(5):373–380. doi: 10.1007/s11910-007-0058-7. [DOI] [PubMed] [Google Scholar]
- 74.Cicconetti P, Riolo N, Priami C, Tafaro L, Ettore E. [Risk factors for cognitive impairment] Recenti Prog Med. 2004;95(11):535–545. [PubMed] [Google Scholar]
- 75.Xu W, Qiu C, Winblad B, Fratiglioni L. The effect of borderline diabetes on the risk of dementia and Alzheimer's disease. Diabetes. 2007;56(1):211–216. doi: 10.2337/db06-0879. [DOI] [PubMed] [Google Scholar]
- 76.Whitmer RA, Gunderson EP, Quesenberry CP, Jr, Zhou J, Yaffe K. Body mass index in midlife and risk of Alzheimer disease and vascular dementia. Curr Alzheimer Res. 2007;4(2):103–109. doi: 10.2174/156720507780362047. [DOI] [PubMed] [Google Scholar]
- 77.Etiene D, Kraft J, Ganju N, Gomez-Isla T, Gemelli B, Hyman BT, Hedley-Whyte ET, Wands JR, de la Monte SM. Cerebrovascular pathology contributes to the heterogeneity of alzheimer's disease. J Alzheimers Dis. 1998;1(2):119–134. doi: 10.3233/jad-1998-1205. [DOI] [PubMed] [Google Scholar]
- 78.Korf ES, White LR, Scheltens P, Launer LJ. Brain aging in very old men with type 2 diabetes: the Honolulu-Asia Aging Study. Diabetes Care. 2006;29(10):2268–2274. doi: 10.2337/dc06-0243. [DOI] [PubMed] [Google Scholar]
- 79.Janson J, Laedtke T, Parisi JE, O'Brien P, Petersen RC, Butler PC. Increased risk of type 2 diabetes in Alzheimer disease. Diabetes. 2004;53(2):474–481. doi: 10.2337/diabetes.53.2.474. [DOI] [PubMed] [Google Scholar]
- 80.Moroz N, Tong M, Longato L, Xu H, de la Monte SM. Limited Alzheimer-type neurodegeneration in experimental obesity and type 2 diabetes mellitus. J Alzheimers Dis. 2008;15(1):29–44. doi: 10.3233/jad-2008-15103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ezzat VA, Duncan ER, Wheatcroft SB, Kearney MT. The role of IGF-1 and its binding proteins in the development of type 2 diabetes and cardiovascular disease. Diabetes Obes Metab. 2008;10(3):198–211. doi: 10.1111/j.1463-1326.2007.00709.x. [DOI] [PubMed] [Google Scholar]
- 82.Ichinose K, Kawasaki E, Eguchi K. Recent advancement of understanding pathogenesis of type 1 diabetes and potential relevance to diabetic nephropathy. Am J Nephrol. 2007;27(6):554–564. doi: 10.1159/000107758. [DOI] [PubMed] [Google Scholar]
- 83.Ringholm Nielsen L, Juul A, Pedersen-Bjergaard U, Thorsteinsson B, Damm P, Mathiesen ER. Lower levels of circulating IGF-1 in type 1 diabetic women with frequent severe hypoglycaemia during pregnancy. Diabet Med. 2008;25(7):826–833. doi: 10.1111/j.1464-5491.2008.02495.x. [DOI] [PubMed] [Google Scholar]
- 84.Calderari S, Gangnerau MN, Thibault M, Meile MJ, Kassis N, Alvarez C, Portha B, Serradas P. Defective IGF2 and IGF1R protein production in embryonic pancreas precedes beta cell mass anomaly in the Goto-Kakizaki rat model of type 2 diabetes. Diabetologia. 2007;50(7):1463–1471. doi: 10.1007/s00125-007-0676-2. [DOI] [PubMed] [Google Scholar]
- 85.Liu JL. Does IGF-1 stimulate pancreatic islet cell growth? Cell Biochem Biophys. 2007;48(2-3):115–125. doi: 10.1007/s12013-007-0016-7. [DOI] [PubMed] [Google Scholar]
- 86.Braak H, Braak E. Diagnostic criteria for neuropathologic assessment of Alzheimer's disease. Neurobiol Aging. 1997;18(4 Suppl):S85–S88. doi: 10.1016/s0197-4580(97)00062-6. [DOI] [PubMed] [Google Scholar]
- 87.Nagy Z, Yilmazer-Hanke DM, Braak H, Braak E, Schultz C, Hanke J. Assessment of the pathological stages of Alzheimer's disease in thin paraffin sections: a comparative study. Dement Geriatr Cogn Disord. 1998;9(3):140–144. doi: 10.1159/000017038. [DOI] [PubMed] [Google Scholar]
- 88.de la Monte SM, Chen GJ, Rivera E, Wands JR. Neuronal thread protein regulation and interaction with microtubule-associated proteins in SH-Sy5y neuronal cells. Cell Mol Life Sci. 2003;60(12):2679–2691. doi: 10.1007/s00018-003-3305-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Soscia SJ, Tong M, Xu XJ, Cohen AC, Chu J, Wands JR, de la Monte SM. Chronic gestational exposure to ethanol causes insulin and IGF resistance and impairs acetylcholine homeostasis in the brain. Cell Mol Life Sci. 2006;63(17):2039–2056. doi: 10.1007/s00018-006-6208-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Blasko I, Stampfer-Kountchev M, Robatscher P, Veerhuis R, Eikelenboom P, Grubeck-Loebenstein B. How chronic inflammation can affect the brain and support the development of Alzheimer's disease in old age: the role of microglia and astrocytes. Aging Cell. 2004;3(4):169–176. doi: 10.1111/j.1474-9728.2004.00101.x. [DOI] [PubMed] [Google Scholar]
- 91.Eikelenboom P, van Gool WA. Neuroinflammatory perspectives on the two faces of Alzheimer's disease. J Neural Transm. 2004;111(3):281–294. doi: 10.1007/s00702-003-0055-1. [DOI] [PubMed] [Google Scholar]
- 92.Tuppo EE, Arias HR. The role of inflammation in Alzheimer's disease. Int J Biochem Cell Biol. 2005;37(2):289–305. doi: 10.1016/j.biocel.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 93.Chen GJ, Xu J, Lahousse SA, Caggiano NL, de la Monte SM. Transient hypoxia causes Alzheimer-type molecular and biochemical abnormalities in cortical neurons: potential strategies for neuroprotection. J Alzheimers Dis. 2003;5(3):209–228. doi: 10.3233/jad-2003-5305. [DOI] [PubMed] [Google Scholar]
- 94.Hoyer S, Lannert H. Inhibition of the neuronal insulin receptor causes Alzheimer-like disturbances in oxidative/energy brain metabolism and in behavior in adult rats. Ann N Y Acad Sci. 1999;893:301–303. doi: 10.1111/j.1749-6632.1999.tb07842.x. [DOI] [PubMed] [Google Scholar]
- 95.Hoyer S, Lee SK, Löffler T, Schliebs R. Inhibition of the neuronal insulin receptor. An in vivo model for sporadic Alzheimer disease? Ann N Y Acad Sci. 2000;920:256–258. doi: 10.1111/j.1749-6632.2000.tb06932.x. [DOI] [PubMed] [Google Scholar]
- 96.Lorenzo A, Yankner BA. Amyloid fibril toxicity in Alzheimer's disease and diabetes. Ann N Y Acad Sci. 1996;777:89–95. doi: 10.1111/j.1749-6632.1996.tb34406.x. [DOI] [PubMed] [Google Scholar]
- 97.Niikura T, Hashimoto Y, Tajima H, Nishimoto I. Death and survival of neuronal cells exposed to Alzheimer's insults. J Neurosci Res. 2002;70(3):380–391. doi: 10.1002/jnr.10354. [DOI] [PubMed] [Google Scholar]
- 98.Tsukamoto E, Hashimoto Y, Kanekura K, Niikura T, Aiso S, Nishimoto I. Characterization of the toxic mechanism triggered by Alzheimer's amyloid-beta peptides via p75 neurotrophin receptor in neuronal hybrid cells. J Neurosci Res. 2003;73(5):627–636. doi: 10.1002/jnr.10703. [DOI] [PubMed] [Google Scholar]
- 99.Bolzán AD, Bianchi MS. Genotoxicity of streptozotocin. Mutat Res. 2002;512(2-3):121–134. doi: 10.1016/s1383-5742(02)00044-3. [DOI] [PubMed] [Google Scholar]
- 100.Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res. 2001;50(6):537–546. [PubMed] [Google Scholar]
- 101.Duelli R, Schröck H, Kuschinsky W, Hoyer S. Intracerebroventricular injection of streptozotocin induces discrete local changes in cerebral glucose utilization in rats. Int J Dev Neurosci. 1994;12(8):737–743. doi: 10.1016/0736-5748(94)90053-1. [DOI] [PubMed] [Google Scholar]
- 102.Hoyer S, Müller D, Plaschke K. Desensitization of brain insulin receptor. Effect on glucose/energy and related metabolism. J Neural Transm Suppl. 1994;44:259–268. doi: 10.1007/978-3-7091-9350-1_20. [DOI] [PubMed] [Google Scholar]
- 103.Lannert H, Hoyer S. Intracerebroventricular administration of streptozotocin causes long-term diminutions in learning and memory abilities and in cerebral energy metabolism in adult rats. Behav Neurosci. 1998;112(5):1199–1208. doi: 10.1037//0735-7044.112.5.1199. [DOI] [PubMed] [Google Scholar]
- 104.Plaschke K, Hoyer S. Action of the diabetogenic drug streptozotocin on glycolytic and glycogenolytic metabolism in adult rat brain cortex and hippocampus. Int J Dev Neurosci. 1993;11(4):477–483. doi: 10.1016/0736-5748(93)90021-5. [DOI] [PubMed] [Google Scholar]
- 105.de la Monte SM, Sohn YK, Wands JR. Correlates of p53- and Fas (CD95)-mediated apoptosis in Alzheimer's disease. J Neurol Sci. 1997;152(1):73–83. doi: 10.1016/s0022-510x(97)00131-7. [DOI] [PubMed] [Google Scholar]
- 106.Su JH, Deng G, Cotman CW. Bax protein expression is increased in Alzheimer's bracorrelations with DNA damage, Bcl-2 expression, and brain pathology. J Neuropathol Exp Neurol. 1997;56(1):86–93. doi: 10.1097/00005072-199701000-00009. [DOI] [PubMed] [Google Scholar]
- 107.de la Monte SM. Quantitation of cerebral atrophy in preclinical and end-stage Alzheimer's disease. Ann Neurol. 1989;25(5):450–459. doi: 10.1002/ana.410250506. [DOI] [PubMed] [Google Scholar]
- 108.Brun A, Liu X, Erikson C. Synapse loss and gliosis in the molecular layer of the cerebral cortex in Alzheimer's disease and in frontal lobe degeneration. Neurodegeneration. 1995;4(2):171–177. doi: 10.1006/neur.1995.0021. [DOI] [PubMed] [Google Scholar]
- 109.Lassmann H, Fischer P, Jellinger K. Synaptic pathology of Alzheimer's disease. Ann N Y Acad Sci. 1993;695:59–64. doi: 10.1111/j.1749-6632.1993.tb23028.x. [DOI] [PubMed] [Google Scholar]
- 110.Lassmann H, Weiler R, Fischer P, Bancher C. Jellinger K, Floor E, Danielczyk W, Seitelberger F, Winkler H. Synaptic pathology in Alzheimer's disease: immunological data for markers of synaptic and large dense-core vesicles. Neuroscience. 1992;46(1):1–8. doi: 10.1016/0306-4522(92)90003-k. [DOI] [PubMed] [Google Scholar]
- 111.Liu X, Erikson C, Brun A. Cortical synaptic changes and gliosis in normal aging, Alzheimer's disease and frontal lobe degeneration. Dementia. 1996;7(3):128–134. doi: 10.1159/000106867. [DOI] [PubMed] [Google Scholar]
- 112.Doublier S, Duyckaerts C, Seurin D, Binoux M. Impaired brain development and hydrocephalus in a line of transgenic mice with liver-specific expression of human insulin-like growth factor binding protein-1. Growth Horm IGF Res. 2000;10(5):267–274. doi: 10.1054/ghir.2000.0168. [DOI] [PubMed] [Google Scholar]
- 113.Nishiyama T, Shirotani T, Murakami T, Shimada F, Todaka M, Saito S, Hayashi H, Noma Y, Shima K, Makino H, et al. Expression of the gene encoding the tyrosine kinase-deficient human insulin receptor in transgenic mice. Gene. 1994;141(2):187–192. doi: 10.1016/0378-1119(94)90569-x. [DOI] [PubMed] [Google Scholar]
- 114.Frölich L, Blum-Degen D, Bernstein HG, Engelsberger S, Humrich J, Laufer S, Muschner D, Thalheimer A, Türk A, Hoyer S, Zöchling R, Boissl KW, Jellinger K, Riederer P. Brain insulin and insulin receptors in aging and sporadic Alzheimer's disease. J Neural Transm. 1998;105(4-5):423–438. doi: 10.1007/s007020050068. [DOI] [PubMed] [Google Scholar]
- 115.Grünblatt E, Salkovic-Petrisic M, Osmanovic J, Riederer P, Hoyer S. Brain insulin system dysfunction in streptozotocin intracerebroventricularly treated rats generates hyperphosphorylated tau protein. J Neurochem. 2007;101(3):757–770. doi: 10.1111/j.1471-4159.2006.04368.x. [DOI] [PubMed] [Google Scholar]
- 116.Salkovic-Petrisic M, Tribl F, Schmidt M, Hoyer S, Riederer P. Alzheimer-like changes in protein kinase B and glycogen synthase kinase-3 in rat frontal cortex and hippocampus after damage to the insulin signalling pathway. J Neurochem. 2006;96(4):1005–1015. doi: 10.1111/j.1471-4159.2005.03637.x. [DOI] [PubMed] [Google Scholar]
- 117.Tahirovic I, Sofic E, Sapcanin A, Gavrankapetanovic I, Bach-Rojecky L, Salkovic-Petrisic M, Lackovic Z, Hoyer S, Riederer P. Reduced brain antioxidant capacity in rat models of betacytotoxic-induced experimental sporadic Alzheimer's disease and diabetes mellitus. Neurochem Res. 2007;32(10):1709–1717. doi: 10.1007/s11064-007-9410-1. [DOI] [PubMed] [Google Scholar]
- 118.Hong M, Lee VM. Insulin and insulin-like growth factor-1 regulate tau phosphorylation in cultured human neurons. J Biol Chem. 1997;272(31):19547–19553. doi: 10.1074/jbc.272.31.19547. [DOI] [PubMed] [Google Scholar]
- 119.Auld DS, Kornecook TJ, Bastianetto S, Quirion R. Alzheimer's disease and the basal forebrain cholinergic system: relations to beta-amyloid peptides, cognition, and treatment strategies. Prog Neurobiol. 2002;68(3):209–245. doi: 10.1016/s0301-0082(02)00079-5. [DOI] [PubMed] [Google Scholar]
- 120.Dunnett SB, Fibiger HC. Role of forebrain cholinergic systems in learning and memory: relevance to the cognitive deficits of aging and Alzheimer's dementia. Prog Brain Res. 1993;98:413–420. doi: 10.1016/s0079-6123(08)62425-5. [DOI] [PubMed] [Google Scholar]
- 121.Felician O, Sandson TA. The neurobiology and pharmacotherapy of Alzheimer's disease. J Neuropsychiatry Clin Neurosci. 1999;11(1):19–31. doi: 10.1176/jnp.11.1.19. [DOI] [PubMed] [Google Scholar]
- 122.Price DL. New perspectives on Alzheimer's disease. Annu Rev Neurosci. 1986;9:489–512. doi: 10.1146/annurev.ne.09.030186.002421. [DOI] [PubMed] [Google Scholar]
- 123.Chesik D, De Keyser J, Wilczak N. Insulin-like growth factor system regulates oligodendroglial cell behavior: therapeutic potential in CNS. J Mol Neurosci. 2008;35(1):81–90. doi: 10.1007/s12031-008-9041-2. [DOI] [PubMed] [Google Scholar]
- 124.Lopes-Cardozo M, Sykes JE, Van der Pal RH, van Golde LM. Development of oligodendrocytes. Studies of rat glial cells cultured in chemically-defined medium. J Dev Physiol. 1989;12(3):117–127. [PubMed] [Google Scholar]
- 125.Markesbery WR, Carney JM. Oxidative alterations in Alzheimer's disease. Brain Pathol. 1999;9(1):133–146. doi: 10.1111/j.1750-3639.1999.tb00215.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Sayre LM, Zelasko DA, Harris PL, Perry G, Salomon RG, Smith MA. 4-Hydroxynonenal-derived advanced lipid peroxidation end products are increased in Alzheimer's disease. J Neurochem. 1997;68(5):2092–2097. doi: 10.1046/j.1471-4159.1997.68052092.x. [DOI] [PubMed] [Google Scholar]
- 127.Benedict C, Hallschmid M, Hatke A, Schultes B, Fehm HL, Born J, Kern W. Intranasal insulin improves memory in humans. Psychoneuroendocrinology. 2004;29(10):1326–1334. doi: 10.1016/j.psyneuen.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 128.Benedict C, Hallschmid M, Schmitz K, Schultes B, Ratter F, Fehm HL, Born J, Kern W. Intranasal insulin improves memory in humans: superiority of insulin aspart. Neuropsychopharmacology. 2007;32(1):239–243. doi: 10.1038/sj.npp.1301193. [DOI] [PubMed] [Google Scholar]
- 129.Benedict C, Hallschmid M, Schultes B, Born J, Kern W. Intranasal insulin to improve memory function in humans. Neuroendocrinology. 2007;86(2):136–142. doi: 10.1159/000106378. [DOI] [PubMed] [Google Scholar]
- 130.Watson GS, Cholerton BA, Reger MA, Baker LD, Plymate SR, Asthana S, Fishel MA, Kulstad JJ, Green PS, Cook DG, Kahn SE, Keeling ML, Craft S. Preserved cognition in patients with early Alzheimer disease and amnestic mild cognitive impairment during treatment with rosiglitazone: a preliminary study. Am J Geriatr Psychiatry. 2005;13(11):950–958. doi: 10.1176/appi.ajgp.13.11.950. [DOI] [PubMed] [Google Scholar]