Abstract
Background
We used a mathematical model to estimate the contribution of urinary glucose excretion (UGE) to reported changes in body weight (BW) following oral antihyperglycemic agent (AHA) therapy. This modeling approach was used to gain novel insight into the mechanisms by which oral AHA affects BW.
Methods
Twenty-four hour glucose profiles were used to predict UGE before and after treatment with oral AHA. Modelpredicted changes in BW due to reduced UGE were compared with reported changes in BW to quantify non-UGEdependent effects (fluid retention, food intake, and energy expenditure).
Results
In type 2 diabetes patients [hemoglobin A1c (HbA1c) >7.3%], the energy lost to UGE is predicted to decrease an average of 100 kcal/day for each 1% decrease in HbA1c. This effect, alone, is predicted to increase BW 1.4 kg after 6 months. Differences from this value reported for changes in BW with oral AHA therapy (+1.4 kg for pioglitazone and rosiglitazone; –0.4 kg for glyburide; –0.9 kg for sitagliptin and vildagliptin; –2.3 kg for metformin) are therefore predicted to be due to additional, non-UGE-dependent mechanisms.
Conclusions
Weight gain following thiazolidinedione therapy is predicted to result from both reduced UGE and non-UGE-dependent mechanisms. Reduced UGE alone is predicted to account for most of the weight gain reported following sulfonylurea therapy. Weight loss observed in response to metformin and weight maintenance observed in response to dipeptidyl peptidase-4 inhibitors may result from an increase in satiety, energy expenditure, or both.
Keywords: body weight, diabetes, glyburide, HbA1c, mathematical modeling, metformin, pioglitazone, rosiglitazone, sitagliptin, urinary glucose excretion
Introduction
The increase in body weight (BW) that accompanies many treatments for type 2 diabetes (T2D) is an undesired side effect that limits overall efficacy1,2 and can discourage patient compliance. The relative contribution of food intake, energy expenditure, and glycosuria to changes in BW following treatment with oral antihyperglycemic agents (AHAs) has been challenging to quantify. This is due to the expense and logistical difficulty of measuring the physiological contributors to BW in human subjects. One approach to these challenges is to employ computer modeling. An important advantage of this technique is the ability to estimate the impact of glycosuria on BW while controlling for fluid retention, food intake, or energy expenditure.
We used a mathematical model of human metabolism (the Metabolism PhysioLab® platform) to predict the effects of oral AHAs on changes in BW as a consequence of urinary glucose excretion (UGE). The Metabolism PhysioLab platform was designed to predict the effect of diabetes treatments on 24-hour plasma glucose and hemoglobin A1c (HbA1c). Because the rate of UGE is proportional to plasma glucose concentration in excess of the renal glucose threshold,4 it is possible to calculate changes in UGE from reported changes in plasma glucose with treatment. This allowed us to estimate the changes in BW in response to oral AHA therapy that result from reductions in glycosuria alone. By comparing predicted UGE-dependent changes in BW with reported changes in BW determined through a literature meta-analysis, it is possible to estimate the non-UGE-dependent effects of each therapy.
Research Design and Methods
Estimation of UGE
The amount of glucose filtered into the renal tubules is dependent on both the glomerular filtration rate (GFR) and the plasma glucose concentration. In the nephron, glucose is reabsorbed from the glomerular filtrate such that at plasma glucose concentrations less than ∼180 mg/dl, little glucose is excreted in the urine.4–6 However, in individuals with T2D, plasma glucose concentrations above 180 mg/dl can saturate reabsorption mechanisms in the kidney, leading to significant UGE.
Urinary glucose excretion was calculated using the kidney portion of the Metabolism PhysioLab platform; this submodel calculates UGE as the difference between the amount of glucose entering the proximal tubules and the amount of glucose reabsorbed. Glucose filtered into the proximal tubule is assumed to be proportional to plasma glucose (Figure 1A). Glucose reabsorption was assumed to be equal to the filtration rate for all glucose concentrations below the renal threshold (180 mg/dl) and to saturate at a maximum rate of 330 mg/min (Figure 1A). The following parameter values were derived from the following literature references: GFR of 125 ml/min/1.73 m,2,3 threshold for glucose excretion of 180 mg/dl,4,5 and maximal rate of glucose reabsorption of 330 mg/min.7,8 A quantitative comparison between model predictions and experimental data5 (Figure 1B) demonstrates that predicted UGE rates are within one quartile of reported median values.
Figure 1.
Comparison of simulation predictions and published data for plasma glucose concentrations, hemoglobin A1c (A1C) levels, and UGE. (A) Simulations of normal renal function. At plasma glucose concentrations less than about 180 mg/dl, glucose reabsorption is predicted to be essentially complete and no glucose is excreted in the urine. As the plasma glucose concentration increases, the rate of glucose reabsorption is predicted to saturate, resulting in increased urine glucose excretion. (B) A comparison of predicted UGE (dashed line) and experimental data (x symbols).5 Thin bars show the range of data, thick bars show the two middle quartiles (25–50 and 50–75%), and x indicates the median values. (C) Comparison of the average predicted plasma glucose profile (dashed line) of virtual patients matching those used in the study and reported mean data (solid line) for type 2 diabetes patients ± SEM.33 (D) Predicted effects of chronic sitagliptin treatment (100 mg every day, 24 weeks) on HbA1c levels (thick line without symbols) compared to reported clinical data (thin lines with symbols).14,15,20,34 Two different virtual patient cohorts were selected to match the average baseline HbA1c for the patients enrolled in these clinical trials. (E) Published results of an oral glucose tolerance test given before (solid lines) and after (dashed lines) sitagliptin therapy compared with simulation results. (F) Sitagliptin (100 mg every day, 24 weeks) treatment was simulated in virtual patients and the predicted reductions in HbA1c (open circles) were plotted as a function of baseline HbA1c. These results were compared with published sitagliptin trial data (filled circles). A linear regression line was fit to each data set by minimizing the sum of squared differences between it and all the individual points for virtual patients (dashed line) and reported data (solid line).
Estimation of Oral AHA Effects on Plasma Glucose and HbA1c
The Metabolism PhysioLab platform (Entelos, Foster City, CA), a mathematical model of metabolic regulation, was used to predict dynamic plasma glucose concentrations and HbA1c before and after administration of oral AHA therapy. The platform is based on organ-level metabolic processes that have been well quantified in the clinical literature. Because these processes vary from person to person, multiple “virtual patients” are created to represent the ranges of individual characteristics reported in the literature (e.g., the National Health and Nutrition Examination Survey).9
A cohort of 137 virtual patients was created to represent a broad range of individuals with T2D by modeling insulin resistance in muscle, liver, and adipose tissue, as well as a range of β-cell function impairment. Consequently, the virtual patient cohort exhibits variability in response to treatment comparable to that reported for subpopulations of clinical patients. The virtual patients had fasting plasma glucose values that varied from 120 to 247 mg/dl (average: 173 mg/dl) and baseline HbA1c that varied from 5.9 to 10.8% (average: 8.5%).
To validate model predictions of plasma glucose concentration before administration of oral AHA therapy, the reported mean 24-hour plasma glucose profile for T2D patients and the predicted average plasma glucose profile of virtual patients with matching fasting plasma glucose concentrations were compared (Figure 1C). The average values over 24 hours for the virtual patient cohort are within 1 standard error of the mean (SEM) of the experimental measurements at the same time points.
To validate model predictions of the effects of oral AHA therapies on postprandial glucose and HbA1c, simulation predictions were compared with published clinical data. For example, the predicted effects of chronic sitagliptin therapy (100 mg every day, 24 weeks) are within one standard deviation (SD) of clinical data (Figures 1D and 1E). In addition, the predicted relationship between sitagliptin efficacy and disease severity, across a range of baseline HbA1c values, is consistent with the relationship identified by conducting a meta-analysis of the literature (Figure 1F). Similar comparisons were conducted to validate the model's representations of rosiglitazone, pioglitazone, glyburide, and metformin. As was the case for sitagliptin, the predicted efficacy for each therapy across a range of baseline HbA1c values is clinically indistinguishable from published clinical trial data.
Meta-analysis of Oral AHA Therapies
A search of the PubMed database (www.ncbi.nlm.nih.gov) was performed to identify all English-language articles describing unique, randomized controlled T2D clinical trials of sitagliptin, vildagliptin, metformin, glyburide, pioglitazone, and rosiglitazone. Studies were included if they were published between January 1999 and October 2007, specified a study period of 4–6 months, and included reported changes in HbA1c and BW. See the online supplemental material on page 1240 for a list of the studies analyzed for this article. The change in HbA1c and BW from baseline for each trial was determined and an arithmetic mean from all trials was calculated for each therapy.
Results
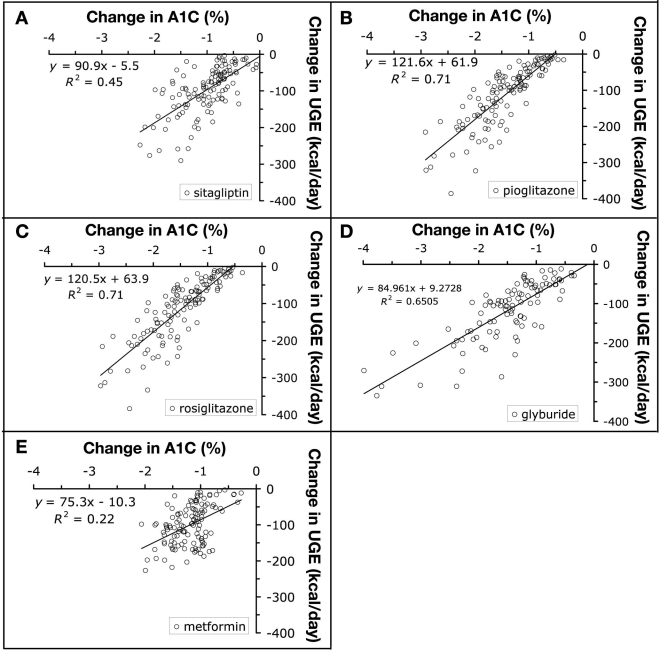
Baseline UGE is predicted to increase by ∼100 kcal per day for every 1% increase in HbA1c above 7.25%. T2D patients with an initial HbA1c of 7.25% or less are not predicted to excrete measurable glucose in the urine. Clinical trials corresponding to meta-analysis selection criteria were simulated in the Metabolism PhysioLab platform to determine the predicted effects of oral AHA therapies on HbA1c, UGE, and BW. Representative compounds from each of the four major classes of oral AHA were assessed: sitagliptin [dipeptidyl peptidase-4 (DPP-4) inhibitor], pioglitazone and rosiglitazone (thiazolidinedione, TZD), glyburide (sulfonylurea), and metformin (biguanide). Approved doses of each compound were simulated. Doses were titrated by increasing dose until the minimum predicted daytime glucose concentration dropped below 70 mg/dl, fasting plasma glucose dropped below 120 mg/dl, or 2-hour postprandial glucose dropped below 140 mg/dl. The final adjusted dose was maintained for the remainder of the trial simulation. After 24 weeks of simulated treatment, the predicted change in HbA1c and UGE from baseline was determined for each virtual patient (Figure 2). For every 1% reduction in predicted HbA1c, UGE was predicted to decrease 75 kcal/day for metformin, 85 kcal/day for glyburide, 91 kcal/day for sitagliptin, 120 kcal/day for rosiglitazone, and 122 kcal/day for pioglitazone. On average across the therapies evaluated, UGE is predicted to decrease by ∼100 kcal per day for every 1% reduction in HbA1c. This estimate is consistent with the measured changes in T2D patients treated with metformin and insulin for 1 year.10 Since the food intake and energy expenditure of each virtual patient were kept fixed at pretreatment values during the simulations, the reduced UGE increases BW. To estimate the effect that caloric retention would have on BW, we simulated a 24-week trial in which a positive energy balance of 100–300 kcal/day was imposed, corresponding to a 1–3% reduction in HbA1c. The average increase in BW, due to changes in UGE alone, was calculated to be 1.4 kg for every 1% reduction in HbA1c. It varied from a low of 1 kg for metformin to a maximum of 1.6 kg for pioglitazone and rosiglitazone.
Figure 2.
Predicted relationship between UGE and hemoglobin A1c (A1C). Treatment of T2D virtual patients for 24 weeks with sitagliptin (A), pioglitazone (B), rosiglitazone (C), glyburide (D), and metformin (E) is predicted to reduce HbA1c and UGE. The equation for the line of best fit and its correlation coefficient (R2) are shown.
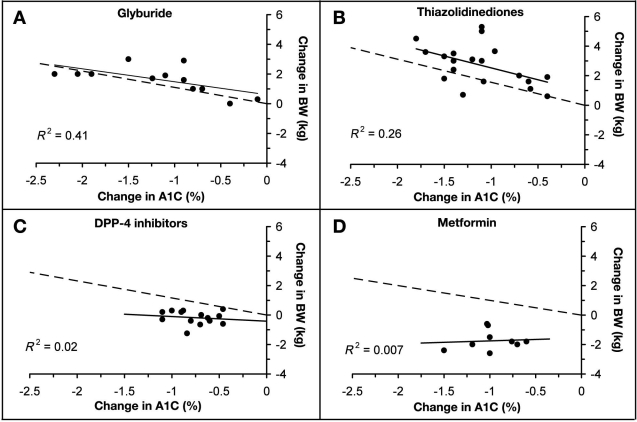
We next compared the predicted UGE-dependent BW changes derived from the model to observed changes reported in the literature after initiation of oral AHA therapy. The change in HbA1c and BW from baseline was determined from the results of clinical trials and an average value was calculated for each therapy. The observed relationship between reductions in HbA1c and BW is shown in Figure 3. Each therapy was effective in reducing HbA1c, but the change in BW varied significantly among therapies. Increases in BW were observed following treatment with the sulfonylurea glyburide (1.4, SD = 1 kg) and the thiazolidinediones pioglitazone and rosiglitazone (2.9, SD = 1.3 kg). Treatment with the DPP-4 inhibitors sitagliptin and vildagliptin did not affect BW (–0.2, SD = 0.5 kg), whereas metformin treatment resulted in BW loss (–1.6, SD = 0.6 kg). Predicted changes in BW based on reduced UGE alone (dashed line) for each therapy are shown in Figure 3. Comparison of this line with the treatment regression line (solid line) allows a visual comparison of overall treatment effects on BW with predicted changes due to reductions in UGE.
Figure 3.
Comparison of the reported changes in BW with predictions of UGE-dependent increases in BW. The observed relationship between changes in HbA1c and BW with marketed oral AHA therapies is shown (filled circles). The regression line of best fit for these data (solid line) and its correlation coefficient (R2) are shown. Model-based predictions of weight gain resulting from reductions in UGE are indicated by the dashed line.
Although BW gain with TZD treatment appears to be related to UGE (Figure 3B), other factors also contribute. The average weight gain reported in most trials exceeds the amount predicted from reductions in UGE alone.11–13 In contrast (Figure 3C), BW is not affected by treatment with DPP-4 inhibitors,14–16 indicating that the positive energy balance predicted to occur with reduced UGE is offset by other factors affecting energy balance (e.g., food intake, energy expenditure). This is also the case with metformin treatment (Figure 3D), which is associated with BW loss.17Because predicted BW gain due to UGE varies from reported changes in BW for all therapies except glyburide, the nonurinary glucose-dependent effects on BW are predicted to differ across therapies. The estimated effect of these factors on BW is equal to the difference between predicted increases in BW due to reduced glycosuria and the changes in BW observed in clinical trials. In patients treated with pioglitazone and rosiglitazone, the average increase in BW totaled 2.9 (SD = 1.3 kg), reductions in UGE are predicted to account for 1.5 (SD = 1 kg) of the total, and therefore 1.4 (SD = 1.4 kg) of the weight gain can be attributed to non-UGE-dependent mechanisms. In patients treated with glyburide, the observed increase in BW totaled 1.4 (SD = 1 kg), whereas changes in UGE are predicted to increase BW by 1.8 (SD = 0.8 kg). The non-UGE-dependent factors are predicted to be –0.4 (SD = 0.6 kg). In patients treated with the DPP-4 inhibitors sitagliptin and vildagliptin, there was no appreciable change in BW (–0.2, SD = 0.5 kg), whereas changes in UGE are predicted to increase BW by 0.8 (SD = 0.4 kg); therefore the non-UGE-dependent factors are predicted to be –0.9 (SD = 0.4 kg). In patients treated with metformin, a decrease in BW is observed (–1.6, SD = 0.6 kg), changes in UGE are predicted to increase BW by 0.6 (SD = 0.6 kg), and therefore the non-UGE-dependent factors are predicted to be –2.3 (SD = 1 kg).
Discussion
Results predict that the effects of DPP-4 inhibitors, thiazolidinediones, sulfonylureas, and metformin on UGE are similar, but predicted nonurinary glucose-dependent effects vary dramatically. Treatment of T2D patients with oral AHA improves glycemic control and results in the retention of calories that were excreted previously in the urine before therapy.18 Our model predictions suggest that this effect alone would increase BW 1–1.6 kg for every 1% reduction in HbA1c. Comparison of these predictions with a meta-analysis of literature reports for these therapies indicates that these therapies do not increase BW uniformly. TZD and sulfonylureas increase BW, metformin decreases BW, and DPP-4 inhibitors are generally weight neutral. This suggests that non-UGE-dependent effects on BW (e.g., fluid retention, food intake, and energy expenditure) differ dramatically among these therapies.
Body weight is relatively unaffected by treatment with the DPP-4 inhibitors sitagliptin or vildagliptin (Figure 3).19,20 This suggests that the positive energy balance as a consequence of reduced UGE is balanced by reduced food intake or increased energy expenditure. Literature suggests that both effects are possible. Sitagliptin treatment results in an approximate twofold increase in active glucagon-like peptide 1 (GLP1) concentrations.21 Increases in GLP1 result in satiety,22,23 which could mediate a decrease in food intake following DPP-4 inhibitor treatment. Moreover, Pannacciulli and colleagues24 reported a positive correlation between resting energy expenditure and fasting GLP1 concentration.24 In the study by Pannacciulli and associates,24 an increase in total fasting GLP1 was associated with an increase in resting energy expenditure of approximately 130 kcal/day. A change of this magnitude is sufficient to compensate for the predicted positive energy balance due to reduced UGE. Thus the maintenance of BW after treatment with DPP-4 inhibitors could be due to either a decrease in food intake or an increase in resting energy expenditure.
In patients with moderate and severe diabetes (HbA1c >8%), glyburide causes an increase in BW that is consistent with that predicted for a reduction in UGE alone (Figure 3).25,26 This suggests that the effect of glyburide to increase BW can be attributed solely to the reduced glycosuria that occurs with improvements in glycemic control. The changes in UGE predicted in this study (90 kcal/day) are consistent with values reported previously for sulfonylurea therapy (105 kcal/day).27
Body weight has consistently been reported to increase in response to thiazolidinedione therapy, and potential contributing mechanisms have been reviewed.28 A number of these mechanisms are independent of UGE and can be subdivided into those with a direct effect on BW and those with indirect effects that occur through the partitioning of metabolite disposal away from oxidation and toward storage. Examples of such indirect mechanisms include increased differentiation of adipocytes, inhibition of lipolysis, and stimulation of lipogenesis. Examples of mechanisms with a direct effect on weight gain include increased food intake, decreased energy expenditure, and increased fluid retention. In the meta-analysis conducted for this study, the average increase in BW with thiazolidinedione therapy was 2.9 kg. Predicted reductions in glycosuria alone were insufficient to account for the increase in BW observed, with approximately half of the BW increase predicted to be because of nonurinary glucose-dependent mechanisms. Therefore, the effective use of thiazolidinediones to treat diabetes may require imposition of a low-calorie diet to ameliorate some of the mechanisms of weight gain.26
Treatment with metformin typically results in decreases in BW.29,30 This suggests that substantial nonurinary glucose-dependent effects on energy balance must occur to compensate for the predicted increases in BW due to reduced glycosuria. Although treatment with metformin is not reported to affect energy expenditure,31 decreased food intake is a well-known effect of metformin therapy.10,32
In this study, a meta-analysis of the literature was combined with a model-based prediction of UGE to predict the effects of several oral AHAs on changes in BW due to reduced glycosuria. This approach allowed us to evaluate the impact of physiological mechanisms contributing to BW regulation that were not measured directly in the meta-analysis studies. This modeling approach was substantially more time and cost-effective than a clinical trial and allowed control for the individual variations in food intake and activity that often confound the interpretation of clinical trial results. A potential limitation of this modeling approach is the possibility that important interactions and feedback loops are not represented in the model and not all possible patient phenotypes may be represented. In addition, we calculated UGE as a function of plasma glucose, but do not have direct literature support to verify or refute our predicted UGE versus patient HbA1c. However, it should be noted that the results predicted using virtual patients are consistent with values reported in the literature for 24-hour glucose, postprandial glucose, and reduction in HbA1c following treatment with sitagliptin for 24 weeks (Figures 1C–1F).
In summary, we used predictions of UGE and a literature meta-analysis to analyze the effects of oral AHA on BW. Improvements in glycemic control reduce UGE and predispose patients to appreciable increases in BW. The reported BW gain in moderate to severe diabetes following treatment with sulfonylureas is consistent with that predicted for reduced UGE alone. BW gain following thiazolidinedione therapy is likely due to urinary glucose-dependent and nonurinary glucose-dependent mechanisms, including increased fluid retention, increased food intake, and/or reduced energy expenditure. Results suggest that weight loss following metformin treatment and weight maintenance following therapy with DPP-4 inhibitors are due to increases in satiety or energy expenditure that exceed the effect of reduced glycosuria on BW. The lack of weight gain with these therapies may be helpful in the long term and result in improved efficacy (increased BW decreases insulin sensitivity) and patient compliance (therapies that increase BW can be discouraging for T2D patients).
Acknowledgements
This study was supported by Merck & Co., Inc., Whitehouse Station, New Jersey. The authors thank Mike Cole for his editorial assistance and helpful discussions during the preparation of this article.
Abbreviations
- AHA
antihyperglycemic agent
- BW
body weight
- DPP-4
dipeptidyl peptidase-4
- GFR
glomerular filtration rate
- GLP1
glucagon-like peptide 1
- HbA1c
hemoglobin A1c
- SD
standard deviation
- SEM
standard error of the mean
- T2D
type 2 diabetes
- TZD
thiazolidinedione
- UGE
urinary glucose excretion
References
- 1.Pi-Sunyer FX. The obesity epidemic: pathophysiology and consequences of obesity. Obes Res. 2002;10(Suppl 2):97S–104S. doi: 10.1038/oby.2002.202. [DOI] [PubMed] [Google Scholar]
- 2.Pi-Sunyer FX. The obesity epidemic: pathophysiology and consequences of obesity. Obes Res. 2002;10(Suppl 2):97S–104S. doi: 10.1038/oby.2002.202. [DOI] [PubMed] [Google Scholar]
- 3.Heller S. Weight gain during insulin therapy in patients with type 2 diabetes mellitus. Diabetes Res Clin Pract. 2004;65(Suppl 1):S23–S27. doi: 10.1016/j.diabres.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 4.Guyton AC, Hall JE. Textbook of medical physiology. 9 ed. Philadelphia: W.B. Saunders; 1996. Urine formation by the kidneys. [Google Scholar]
- 5.Butterfield WJ, Keen H, Whichelow MJ. Renal glucose threshold variations with age. Br Med J. 1967;4(5578):505–507. doi: 10.1136/bmj.4.5578.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ruhnau B, Faber OK, Borch-Johnsen K, Thorsteinsson B. Renal threshold for glucose in non-insulin-dependent diabetic patients. Diabetes Res Clin Pract. 1997;36(1):27–33. doi: 10.1016/s0168-8227(97)01389-2. [DOI] [PubMed] [Google Scholar]
- 7.Keller DM. Glucose excretion in man and dog. Nephron. 1968;5(1):43–66. doi: 10.1159/000179616. [DOI] [PubMed] [Google Scholar]
- 8.Brodehl J, Oemar BS, Hoyer PF. Renal glucosuria. Pediatr Nephrol. 1987;1(3):502–508. doi: 10.1007/BF00849261. [DOI] [PubMed] [Google Scholar]
- 9.Deetjen P, Baeyer HV, Drexel H. Renal handling of D-glucose and other sugars. In: Massry SG, Glassoch KJ, editors. The textbook of nephrology. Baltimore, MD: Williams & Wilkins; 1995. [Google Scholar]
- 10.Asao K, Kao WH, Baptiste-Roberts K, Bandeen-Roche K, Erlinger TP, Brancati FL. Short stature and the risk of adiposity, insulin resistance, and type 2 diabetes in middle age: the Third National Health and Nutrition Examination Survey (NHANES III), 1988-1994. Diabetes Care. 2006;29(7):1632–1637. doi: 10.2337/dc05-1997. [DOI] [PubMed] [Google Scholar]
- 11.Makimattila S, Nikkila K, Yki-Jarvinen H. Causes of weight gain during insulin therapy with and without metformin in patients with Type II diabetes mellitus. Diabetologia. 1999;42(4):406–412. doi: 10.1007/s001250051172. [DOI] [PubMed] [Google Scholar]
- 12.Goldberg RB, Kendall DM, Deeg MA, Buse JB, Zagar AJ, Pinaire JA, Tan MH, Khan MA, Perez AT, Jacober SJ GLAI Study Investigators. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care. 2005;28(7):1547–1554. doi: 10.2337/diacare.28.7.1547. [DOI] [PubMed] [Google Scholar]
- 13.Raskin P, Rendell M, Riddle MC, Dole JF, Freed MI, Rosenstock J. A randomized trial of rosiglitazone therapy in patients with inadequately controlled insulin-treated type 2 diabetes. Diabetes Care. 2001;24(7):1226–1232. doi: 10.2337/diacare.24.7.1226. [DOI] [PubMed] [Google Scholar]
- 14.St John-Sutton M, Rendell M, Dandona P, Dole JF, Murphy K, Patwardhan R, Patel J, Freed M. A comparison of the effects of rosiglitazone and glyburide on cardiovascular function and glycemic control in patients with type 2 diabetes. Diabetes Care. 2002;25(11):2058–2064. doi: 10.2337/diacare.25.11.2058. [DOI] [PubMed] [Google Scholar]
- 15.Nauck MA, Meininger G, Sheng D, Terranella L, Stein PP. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab. 2007;9(2):194–205. doi: 10.1111/j.1463-1326.2006.00704.x. [DOI] [PubMed] [Google Scholar]
- 16.Charbonnel B, Karasik A, Liu J, Wu M, Meininger G. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes Care. 2006;29(12):2638–2643. doi: 10.2337/dc06-0706. [DOI] [PubMed] [Google Scholar]
- 17.Rosenstock J, Baron MA, Dejager S, Mills D, Schweizer A. Comparison of vildagliptin and rosiglitazone monotherapy in patients with type 2 diabetes: a 24-week, double-blind, randomized trial. Diabetes Care. 2007;30(2):217–223. doi: 10.2337/dc06-1815. [DOI] [PubMed] [Google Scholar]
- 18.ADOPT Study Group. Kahn SE, Haffner SM, Heise MA, Herman WH, Holman RR, Jones NP, Kravitz BG, Lachin JM, O'Neill MC, Zinman B, Viberti G. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med. 2006;355(23):2427–2443. doi: 10.1056/NEJMoa066224. [DOI] [PubMed] [Google Scholar]
- 19.Geldermans CA, Terpstra J, Krans HM. The effect of phenformin-HCl on patients with diabetes mellitus, studied under strict balance conditions. Diabetologia. 1975;11(5):475–482. doi: 10.1007/BF00429918. [DOI] [PubMed] [Google Scholar]
- 20.Ristic S, Byiers S, Foley J, Holmes D. Improved glycaemic control with dipeptidyl peptidase-4 inhibition in patients with type 2 diabetes: vildagliptin (LAF237) dose response. Diabetes Obes Metab. 2005;7(6):692–698. doi: 10.1111/j.1463-1326.2005.00539.x. [DOI] [PubMed] [Google Scholar]
- 21.Sitagliptin Study 021 Group. Aschner P, Kipnes MS, Lunceford JK, Sanchez M, Mickel C, Williams-Herman DE. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care. 2006;29(12):2632–2637. doi: 10.2337/dc06-0703. [DOI] [PubMed] [Google Scholar]
- 22.Herman GA, Bergman A, Stevens C, Kotey P, Yi B, Zhao P, Dietrich B, Golor G, Schrodter A, Keymeulen B, Lasseter KC, Kipnes MS, Snyder K, Hilliard D, Tanen M, Cilissen C, De Smet M, de Lepeleire I, Van Dyck K, Wang AQ, Zeng W, Davies MJ, Tanaka W, Holst JJ, Deacon CF, Gottesdiener KM, Wagner JA. Effect of single oral doses of sitagliptin, a dipeptidyl peptidase-4 inhibitor, on incretin and plasma glucose levels after an oral glucose tolerance test in patients with type 2 diabetes. J Clin Endocrinol Metab. 2006;91(11):4612–4619. doi: 10.1210/jc.2006-1009. [DOI] [PubMed] [Google Scholar]
- 23.Flint A, Raben A, Astrup A, Holst JJ. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J Clin Invest. 1998;101(3):515–520. doi: 10.1172/JCI990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Flint A, Raben A, Ersboll AK, Holst JJ, Astrup A. The effect of physiological levels of glucagon-like peptide-1 on appetite, gastric emptying, energy and substrate metabolism in obesity. Int J Obes Relat Metab Disord. 2001;25(6):781–792. doi: 10.1038/sj.ijo.0801627. [DOI] [PubMed] [Google Scholar]
- 25.Pannacciulli N, Bunt JC, Koska J, Bogardus C, Krakoff J. Higher fasting plasma concentrations of glucagon-like peptide 1 are associated with higher resting energy expenditure and fat oxidation rates in humans. Am J Clin Nutr. 2006;84(3):556–560. doi: 10.1093/ajcn/84.3.556. [DOI] [PubMed] [Google Scholar]
- 26.Esposito K, Giugliano D, Nappo F, Marfella R Campanian Postprandial Hyperglycemia Study Group. Regression of carotid atherosclerosis by control of postprandial hyperglycemia in type 2 diabetes mellitus. Circulation. 2004;110(2):214–219. doi: 10.1161/01.CIR.0000134501.57864.66. [DOI] [PubMed] [Google Scholar]
- 27.Garber AJ, Donovan DS, Jr., Dandona P, Bruce S, Park JS. Efficacy of glyburide/metformin tablets compared with initial monotherapy in type 2 diabetes. J Clin Endocrinol Metab. 2003;88(8):3598–3604. doi: 10.1210/jc.2002-021225. [DOI] [PubMed] [Google Scholar]
- 28.Welle S, Nair KS, Lockwood D. Effect of a sulfonylurea and insulin on energy expenditure in type II diabetes mellitus. J Clin Endocrinol Metab. 1988;66(3):593–597. doi: 10.1210/jcem-66-3-593. [DOI] [PubMed] [Google Scholar]
- 29.Fonseca V. Effect of thiazolidinediones on body weight in patients with diabetes mellitus. Am J Med. 2003;115(Suppl 8A):42S–48S. doi: 10.1016/j.amjmed.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 30.Tiikkainen M, Hakkinen AM, Korsheninnikova E, Nyman T, Makimattila S, Yki-Jarvinen H. Effects of rosiglitazone and metformin on liver fat content, hepatic insulin resistance, insulin clearance, and gene expression in adipose tissue in patients with type 2 diabetes. Diabetes. 2004;53(8):2169–2176. doi: 10.2337/diabetes.53.8.2169. [DOI] [PubMed] [Google Scholar]
- 31.Pavo I, Jermendy G, Varkonyi TT, Kerenyi Z, Gyimesi A, Shoustov S, Shestakova M, Herz M, Johns D, Schluchter BJ, Festa A, Tan MH. Effect of pioglitazone compared with metformin on glycemic control and indicators of insulin sensitivity in recently diagnosed patients with type 2 diabetes. J Clin Endocrinol Metab. 2003;88(4):1637–1645. doi: 10.1210/jc.2002-021786. [DOI] [PubMed] [Google Scholar]
- 32.Chong PK, Jung RT, Rennie MJ, Scrimgeour CM. Energy expenditure in type 2 diabetic patients on metformin and sulphonylurea therapy. Diabet Med. 1995;12(5):401–408. doi: 10.1111/j.1464-5491.1995.tb00503.x. [DOI] [PubMed] [Google Scholar]
- 33.Stumvoll M, Nurjhan N, Perriello G, Dailey G, Gerich JE. Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N Engl J Med. 1995;333(9):550–554. doi: 10.1056/NEJM199508313330903. [DOI] [PubMed] [Google Scholar]
- 34.Polonsky KS, Given BD, Hirsch LJ, Tillil H, Shapiro ET, Beebe C, Frank BH, Galloway JA, Van Cauter E. Abnormal patterns of insulin secretion in non-insulin-dependent diabetes mellitus. N Engl J Med. 1988;318:1231–1239. doi: 10.1056/NEJM198805123181903. [DOI] [PubMed] [Google Scholar]
- 35.Rosenstock J, Brazg R, Andryuk PJ, Lu K, Stein P Sitagliptin Study 019 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing pioglitazone therapy in patients with type 2 diabetes: a 24-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Clin Ther. 2006;28(10):1556–1568. doi: 10.1016/j.clinthera.2006.10.007. [DOI] [PubMed] [Google Scholar]