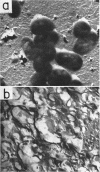
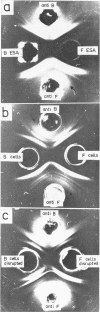
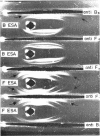
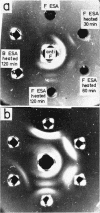
Abstract
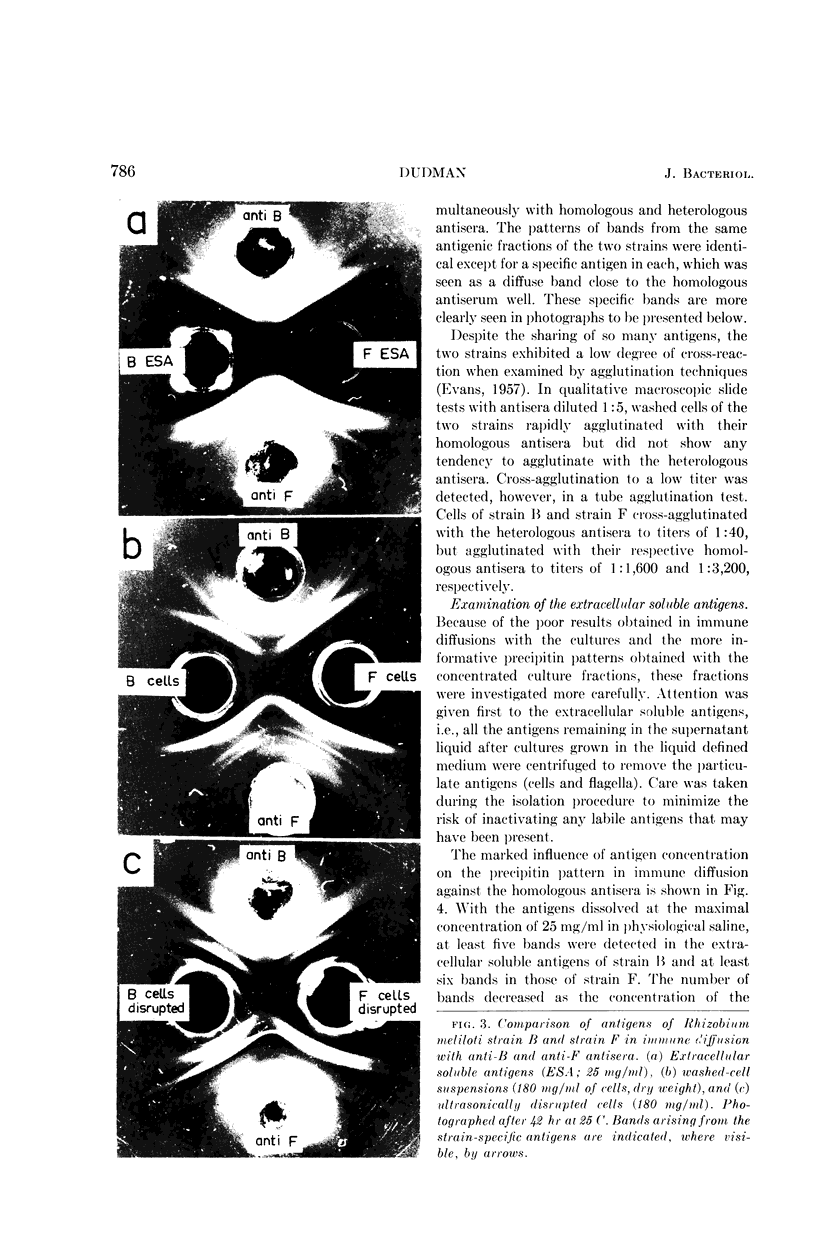
Dudman, W. F. (Commonwealth Scientific and Industrial Research Organization, Canberra, Australia). Immune diffusion analysis of the extracellular soluble antigens of two strains of Rhizobium meliloti. J. Bacteriol. 88:782–794. 1964.—Immune diffusion techniques applied to cultures of two strains of Rhizobium meliloti grown in liquid defined medium showed the presence of multiple antigens. Improved resolution of precipitin patterns was obtained with concentrated antigens separated from the cultures as the extracellular soluble fraction or as suspensions of washed cells. The extracellular fraction contained the same diffusible antigens as the washed cells, but additional antigens were found in the cells after ultrasonic disruption. The extracellular soluble antigens were shown by analysis to contain polysaccharide and protein components. In immune diffusion systems, they gave rise to three groups of precipitin bands, two of which were characterized as polysaccharides by their susceptibility to periodate oxidation, and the third as protein by its lability to heat. All the extracellular antigens of both strains were shared except a fast-diffusing polysaccharide, which was specific for each strain. Despite the sharing of all but one of their antigens, cells of these strains showed only a low degree of cross-agglutination, suggesting that their surfaces are dominated by the specific polysaccharide. No differences could be found in the composition of the polysaccharides in the unfractionated extracellular antigens of the two strains, the main components of which were glucose (66%) and galactose (12%) in the presence of several other unidentified sugars in smaller amounts. The pattern of precipitin bands produced in immune diffusion systems by the extracellular soluble fraction could be changed by altering the cultural conditions.
Full text
PDF



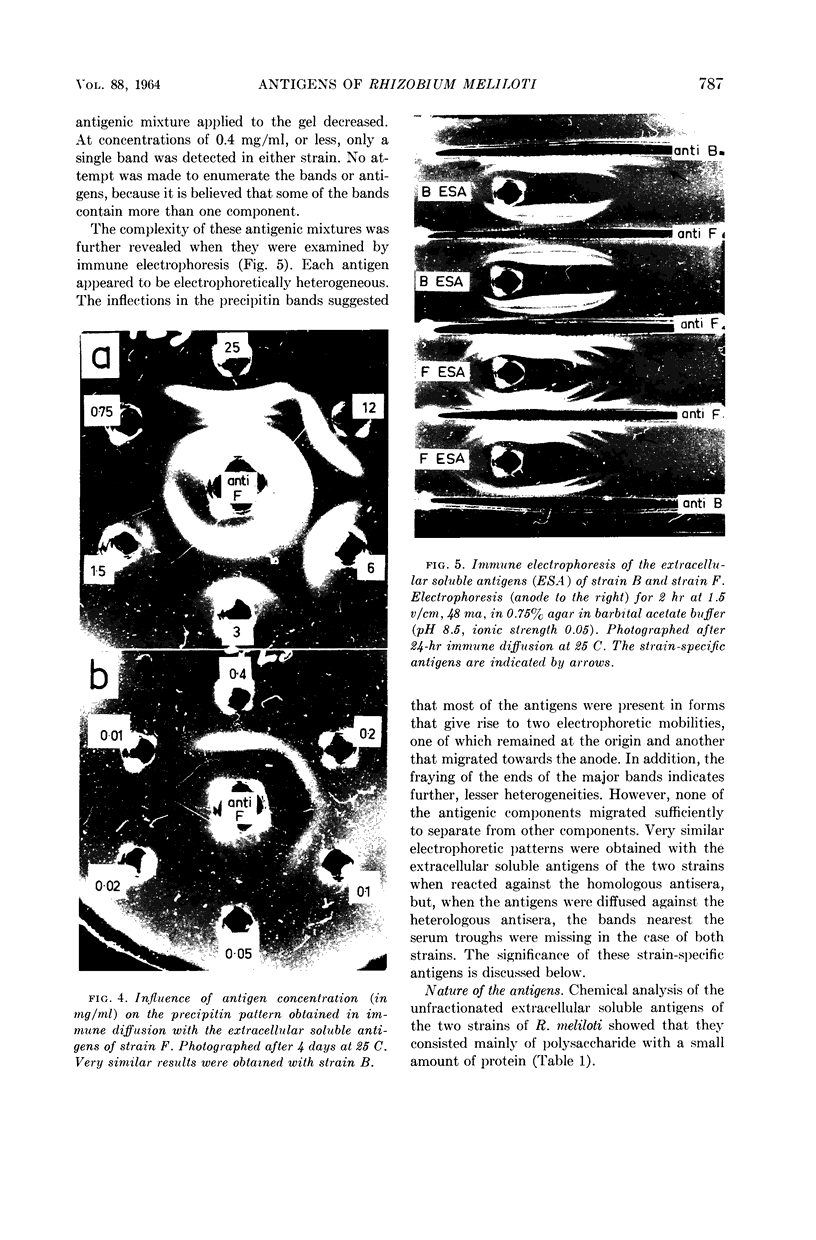
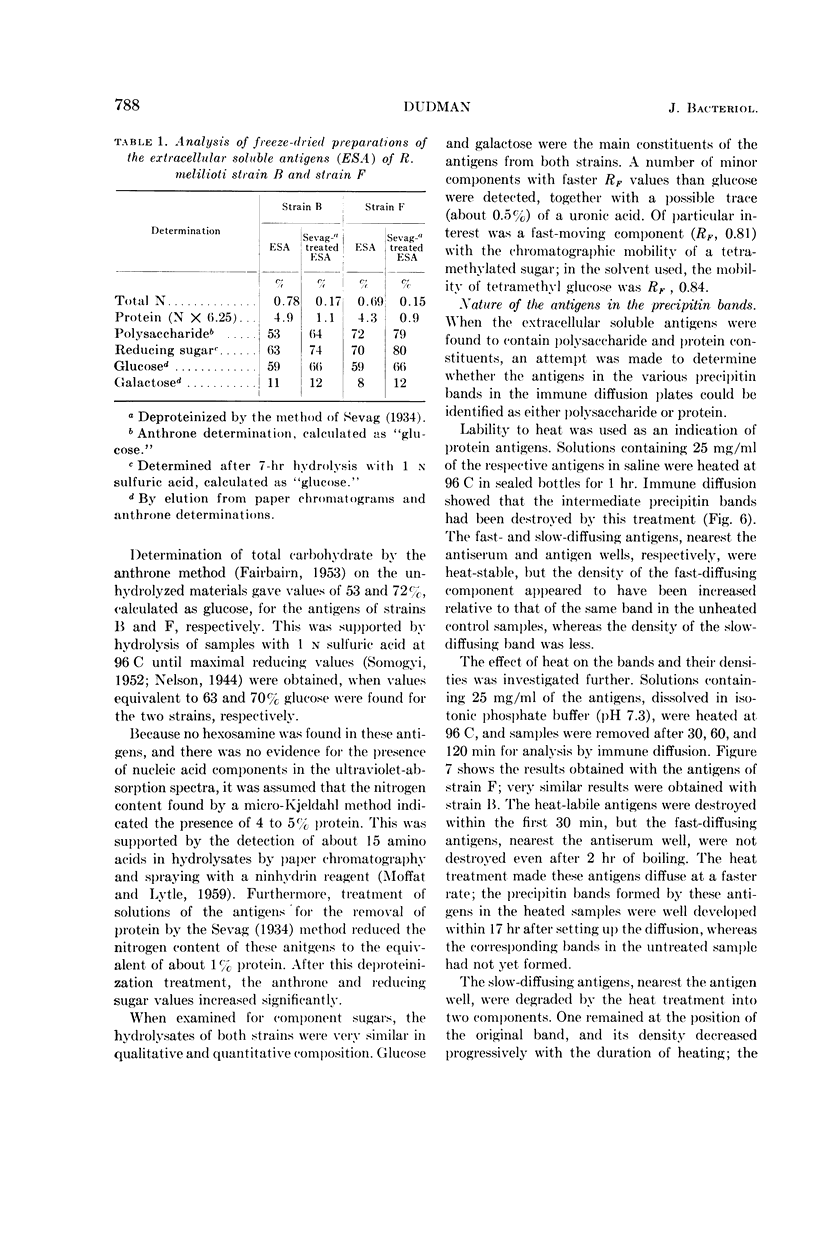






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ARONSSON T., GRONWALL A. Improved separation of serum proteins in paper electrophoresis: a new electrophoresis buffer. Scand J Clin Lab Invest. 1957;9(4):338–341. doi: 10.1080/00365515709079983. [DOI] [PubMed] [Google Scholar]
- Bushnell O. A., Sarles W. B. Investigations upon the Antigenic Relationships among the Root-Nodule Bacteria of the Soybean, Cowpea, and Lupine Cross-Inoculation Groups. J Bacteriol. 1939 Oct;38(4):401–410. doi: 10.1128/jb.38.4.401-410.1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CARLISLE H. N., HINCHLIFFE V., SASLAW S. Immunodiffusion studies with Pasteurella tularensis antigen-rabbit antibody systems. J Immunol. 1962 Nov;89:638–644. [PubMed] [Google Scholar]
- DUDMAN W. F. GROWTH AND EXTRACELLULAR POLYSACCHARIDE PRODUCTION BY RHIZOBIUM MELILOTI IN DEFINED MEDIUM. J Bacteriol. 1964 Sep;88:640–645. doi: 10.1128/jb.88.3.640-645.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DYER J. R. Use of periodate oxidations in biochemical analysis. Methods Biochem Anal. 1956;3:111–152. doi: 10.1002/9780470110195.ch5. [DOI] [PubMed] [Google Scholar]
- ELLNER P. D., GREEN S. S. SEROLOGY OF THE SOLUBLE ANTIGENS OF THE PATHOGENIC CLOSTRIDIA. J Bacteriol. 1963 Nov;86:1084–1097. doi: 10.1128/jb.86.5.1084-1097.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleczkowski A., Thornton H. G. A Serological Study of Root Nodule Bacteria from Pea and Clover Inoculation Groups. J Bacteriol. 1944 Dec;48(6):661–672. doi: 10.1128/jb.48.6.661-672.1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OUCHTERLONY O. Diffusion-in-gel methods for immunological analysis. Prog Allergy. 1958;5:1–78. [PubMed] [Google Scholar]
- PREER J. R., Jr A quantitative study of a technique of double diffusion in agar. J Immunol. 1956 Jul;77(1):52–60. [PubMed] [Google Scholar]
- RIBI E., HASKINS W. T., MILNER K. C., ANACKER R. L., RITTER D. B., GOODE G., TRAPANI R. J., LANDY M. Physicochemical changes in endotoxin associated with loss of biological potency. J Bacteriol. 1962 Oct;84:803–814. doi: 10.1128/jb.84.4.803-814.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RUDBACH J. A., JOHNSON A. G. Changes in serologic reactivity of endotoxin induced by fraction IV-1 (Cohn) of normal human serum. Proc Soc Exp Biol Med. 1962 Dec;111:651–655. doi: 10.3181/00379727-111-27882. [DOI] [PubMed] [Google Scholar]
- RUEDE E., GOEBEL W. F. Colicine K. V. The somatic antigen of a non-colicinogenic variant of E. coli K235. J Exp Med. 1962 Jul 1;116:73–100. doi: 10.1084/jem.116.1.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCHEIDEGGER J. J. Une micro-méthode de l'immuno-electrophorèse. Int Arch Allergy Appl Immunol. 1955;7(2):103–110. [PubMed] [Google Scholar]
- SMOGYI M. Notes on sugar determination. J Biol Chem. 1952 Mar;195(1):19–23. [PubMed] [Google Scholar]
- TREVELYAN W. E., PROCTER D. P., HARRISON J. S. Detection of sugars on paper chromatograms. Nature. 1950 Sep 9;166(4219):444–445. doi: 10.1038/166444b0. [DOI] [PubMed] [Google Scholar]