Abstract
Studies have shown that the length-tension (L-T) relationships in airway and vascular smooth muscles are dynamic and can adapt to length changes over a period of time. Our prior studies have shown that the passive L-T relationship in rabbit detrusor smooth muscle (DSM) is also dynamic and that DSM exhibits adjustable passive stiffness (APS) characterized by a passive L-T curve that can shift along the length axis as a function of strain history and activation history. The present study demonstrates that the active L-T curve for DSM is also dynamic and that the peak active tension produced at a particular muscle length is a function of both strain and activation history. More specifically, this study reveals that the active L-T relationship, or curve, does not have a unique peak tension value with a single ascending and descending limb, but instead reveals that multiple ascending and descending limbs can be exhibited in the same DSM strip. This study also demonstrates that for DSM strips not stretched far enough to reveal a descending limb, the peak active tension produced by a maximal KCl-induced contraction at a short, passively slack muscle length of 3 mm was reduced by 58.6 ± 4.1% (n = 15) following stretches to and contractions at threefold the original muscle length, 9 mm. Moreover, five subsequent contractions at the short muscle length displayed increasingly greater tension; active tension produced by the sixth contraction was 91.5 ± 9.1% of that produced by the prestretch contraction at that length. Together, these findings indicate for the first time that DSM exhibits length adaptation, similar to vascular and airway smooth muscles. In addition, our findings demonstrate that preconditioning, APS and adaptation of the active L-T curve can each impact the maximum total tension observed at a particular DSM length.
Keywords: bladder, lower urinary tract, smooth muscle mechanics, length adaptation, preconditioning, strain softening, adjustable passive stiffness, passive force, active force
bladder smooth muscle cells can undergo a sevenfold length change during filling (47) and must be prepared to contract efficiently and void throughout this range. How the bladder prepares and positions its actomyosin cross bridges to contract efficiently over this broad length range during the filling phase remains to be determined. Uvelius (47, 48) and Longhurst et al. (27) showed that detrusor smooth muscle (DSM) produces a nonlinear passive length-tension (L-Tp) curve with greater tension at longer lengths and a somewhat parabolic active L-T (L-Ta) curve with ascending and descending limbs very similar to those produced by striated (15) and other smooth muscles (16, 22, 39).
The passive and active L-T relationships for skeletal muscle are considered to be static, with a single passive tension value and a single maximum active tension value for each muscle length (15). Until the 1990s, the L-Tp and L-Ta relationships in smooth muscle were thought to be static, although a few earlier studies showed that Ta and/or Tp at a particular muscle length could differ if tension measurements were made during muscle unloading vs. loading (22, 42). Beginning in the 1990s, several studies showed that airway and vascular smooth muscles can adapt to changes in muscle length over time, producing shifts in the L-Ta (19, 20, 35) and the L-Tp (29, 49) curves (reviewed in Refs. 4, 5, and 11). These studies challenge the static model, and several models for length adaptation of the L-Ta relationship in these smooth muscles have emerged (2, 3, 7, 13, 18, 19, 21, 23, 25, 26, 31, 36, 37, 40, 41). Surprisingly, adaptation of the active L-Ta curve has not been explored in the bladder; however, because the length range over which DSM operates is considerably greater than airway and vascular smooth muscles, it is reasonable to expect that DSM would also undergo length adaptation. The present study examines the L-Ta relationship in DSM and tests the hypothesis that DSM exhibits length adaptation similar to vascular and airway smooth muscles.
In practice, smooth muscle tissues may or may not be preconditioned by cyclic loading and may or may not be actively contracted multiple times at the same muscle length to obtain consistent measurements (14). Regardless of the initiation procedures, the L-Ta curve is derived by subtraction of the L-Tp curve from the length-total tension (L-Tt) curve, under the assumption that the L-Tp curve is static following preconditioning and multiple contractions at a given muscle length. Preconditioning is characterized by reductions toward a steady state in stiffness with each subsequent loading cycle and has been identified in DSM (28). One hypothesis put forth to explain preconditioning is that the loss of stiffness is due to strain-induced stress softening, or strain softening, which is an essentially irreversible loss of stiffness due to structural changes (8, 17).
Our previous studies show that DSM exhibits both reversible, viscoelastic softening and strain softening (43). The latter is irreversible when cross bridges are not cycling to produce active force because tissues are maintained in a Ca2+-free solution. However, the decrease in passive stiffness due to strain softening is reversible upon full muscle activation with KCl or carbachol, and activation at shorter muscle lengths restores more stiffness than activation at longer muscle lengths; i.e., the level of stiffness restored depends upon the muscle length at activation (43). Moreover, restoration of passive stiffness is dependent on RhoA kinase activity (43). Our studies also demonstrate a “dynamic” L-Tp curve for rabbit DSM that shifts to the right as a result of strain softening and to the left following muscle activation at slack length (44). Thus rabbit DSM displays passive stiffness that is adjustable because it is dependent on both strain history and muscle activation history, a characteristic we termed adjustable passive stiffness (APS) (45).
The main objectives of the present study are 1) to test the hypothesis that the L-Ta relationship in rabbit DSM exhibits length adaptation, and 2) to test the hypothesis that preconditioning, APS, and length adaptation each impact the maximum total tension produced at a particular DSM length.
METHODS
Tissue Preparation
All experiments involving animals were conducted within the appropriate animal welfare regulations and guidelines and were approved by the Virginia Commonwealth University Institutional Animal Care and Use Committee. Tissues were prepared as described previously (32, 38). Whole bladders from adult female New Zealand White rabbits (2–4 kg) were removed immediately after death with an overdose of pentobarbital sodium. Bladders were washed, cleaned of adhering tissues, including fat and serosa, and stored in cold (0–4°C) normal physiological salt solution (NPSS). Thin strips (∼0.2 mm thick) of longitudinal DSM free of underlying urothelium and overlying serosa were cut from the bladder wall above the trigone and close to the dome (upper detrusor) by following the natural bundling clearly demarcated when bladders were in ice-cold buffer, as described previously (33).
Solutions
NPSS was composed of (in mM) 140 NaCl, 4.7 KCl, 1.2 MgSO4, 1.6 CaCl2, 1.2 Na2HPO4, 2.0 MOPS (adjusted to pH 7.4 at either 0 or 37°C as appropriate), 0.02 Na2 EDTA, and 5.6 dextrose. A Ca2+-free solution (0Ca) was NPSS in which CaCl2 was not added. Muscle contractions were induced with NPSS modified to include 110 mM KCl, substituted isosmotically for NaCl (KPSS).
Apparatus and Muscle Strip Setup
Each DSM strip was secured by small clips to a micrometer for manual length adjustments and a force transducer (159901a, Radnoti Glass Technology) to record isometric force. Force signals were digitized (PCI-6034E, National Instruments) and stored electronically for analyses. Each tissue was secured such that its initial (cold) zero preload length was ∼3 mm and equilibrated in aerated NPSS at 37°C in a water-jacketed tissue bath for 45 min to permit development of spontaneous contractile rhythm.
Active and Passive Tension Measurements
Following equilibration, DSM strips were subjected to one of four L-T curve or adaptation protocols that are described below in L–T Curve Protocols. Each of these protocols involved passive and total tension measurements, and Fig. 1A is a sketch that shows the measurement protocol that was performed at each muscle length. DSM strips were incubated for 2 min in 0Ca to eliminate spontaneous contractile rhythm (active tone) (24), stretched or released to the muscle length of interest if necessary, and incubated for an additional 2 min in 0Ca, incubated in NPSS for 3 min, and then incubated in KPSS for 1 min to induce a contraction. Additional time in the protocol (several seconds), not shown in Fig. 1A, was used to change solutions in the four-bath apparatus and to stretch the muscle strips.
Fig. 1.
A: tension measurement protocol diagram. Detrusor smooth muscle (DSM) strips were incubated for 2 min in 0Ca, stretched or released to the muscle length of interest, if necessary; incubated for an additional 2 min in 0Ca; incubated in normal physiological salt solution (NPSS) for 3 min, and then incubated in NPSS modified to include 110 mM KCl, substituted isosmotically for NaCl (KPSS) for 1 min to induce a contraction. “Passive” tension (Tp) was taken to be the lower value of either the final tension in NPSS (Tp1, path 1, solid line) or, if active tension developed during the 3-min period in NPSS, the final tension in 0Ca (Tp2, path 2, dashed line). Active tension (Ta1 or Ta2) was calculated by subtracting the passive tension (either Tp1 or Tp2) from the peak total KPSS-induced tension (Tpeak). B: tension data from 2 DSM strips illustrating the tension measurement protocol. Strip 1 (black line) exhibited less tension in NPSS (F) than in 0Ca (C), while strip 2 (gray line) exhibited more tension in NPSS (G) than in 0Ca (D) because it developed spontaneous contractile rhythm in NPSS (E).
We have previously shown that stress relaxation in a DSM strip can take an hour or more to approach equilibrium (45). Our primary objective was not to determine a fully stress- relaxed Tp value but rather to obtain a reliable measure of Tp at the time of activation, regardless of whether the tissue had completely stress-relaxed to a minimum Tp value. Therefore, Tp was measured immediately before stimulation with KPSS. The time for each segment of the protocol in Fig. 1A was selected to provide the tissues with time to stress-relax toward (but not always completely to) a steady-state value and to enable the complete experiment to be performed in a reasonable amount of time. Tension data for a pair of DSM strips are provided in Fig. 1B to illustrate the protocol. Strip 1 (black line) exhibited less tension in NPSS than in 0Ca, indicating additional stress relaxation while in NPSS. In contrast, the spontaneous contractile rhythm exhibited by strip 2 (gray line) resulted in greater tension in NPSS than in 0Ca, indicating that active tone exceeded any additional stress relaxation while in NPSS. To account for the potential for tension development while tissues were incubated in NPSS, “passive” tension was taken as the lowest value either in NPSS or 0Ca (Fig. 1A). Ta was calculated by subtracting Tp from Tt, the peak total KCl-induced tension. The typical sample data in Fig. 1B demonstrate that Tp should be measured during incubation in 0Ca to eliminate active tone and that 2 min in 0Ca and 3 min in NPSS appear sufficient to allow stress relaxation to achieve a pseudo-steady state.
To determine whether tissues contracted more forcefully when exposed to NPSS, compared with 0Ca, before stimulation with KPSS, we measured Tp and Ta for three isometric contractions performed at 9 mm using the timing from Fig. 1; however, NPSS was replaced with 0Ca before first and third contractions. Therefore, the DSM strips were incubated in 0Ca for 7 min total before the first and third contractions and in 0Ca for 4 min and then NPSS for 3 min before the second contraction. As shown in Fig. 2A, Tp in NPSS was not significantly different from Tp in 0Ca. However, Ta induced by KPSS following incubation in NPSS was significantly greater than following incubation in 0Ca. Therefore, to achieve maximal KCl-induced contractions in our protocols, we incubated tissues in NPSS for 3 min after exposure to 0Ca and before each contraction. Thus the tension measurement cycle used in all of the L-T protocols in this study consisted of 4 min in 0Ca, 3 min in NPSS, and 1 min in KPSS, as shown in Fig. 1. The only exception was a protocol variation described in Adaptation of the L-Ta Curve at Lengths Greater Than Lo, in which the 4 min in 0Ca was replaced with 4 min in NPSS.
Fig. 2.
Rationale for 3-min NPSS treatment following incubation of tissues in 0Ca and preceding stimulation in KPSS. A and B: Tp (A) and KCl-induced Ta (B) values for 3 isometric KCl-induced contractions performed at 9 mm using the timing from Fig 1, except that tissues were not incubated in NPSS before stimulation in KPSS for contractions 1 and 3. Therefore, the DSM strips were incubated in 0Ca for 7 min total before the and contractions 1 and 3 (white bars) and in 0Ca for 4 min and then NPSS for 3 min before the contraction 2 (gray bars). Data are normalized to the maximum (optimal) active tension (To) for that tissue (means ± SE; n = 3). *P < 0.05 compared with 0Ca.
L-T Curve Protocols
Effect of preconditioning on the L-Ta curve.
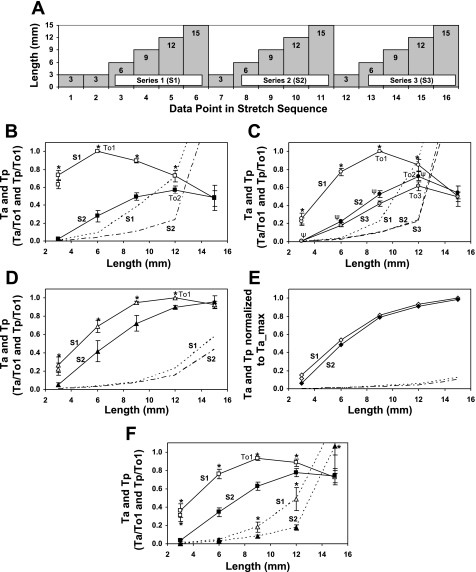
The L-T curve protocol shown in Fig. 3A was designed to test the hypothesis that preconditioning shifts the L-Ta curve. This protocol consists of three series (S1–S3) of five L-T measurements at increasing lengths from 3 to 15 mm. We chose 15 mm as the maximum length because it is fivefold of the initial slack length of 3 mm and incorporates much of the sevenfold-length range over which DSM can operate (47). Relatively large, 3-mm length steps were used to avoid any potential adaptation due to multiple contractions near the same muscle length (49). Series S1 was designed to precondition the tissues to the maximum length in the protocol, 15 mm, which we expected to 1) remove any “kinks” in the tissue (12); 2) establish any permanent damage that would occur due to stretching, especially local damage at the clamps; 3) remove the amount of Tp due to APS that would be lost due to strain-softening (44, 45); and 4) initiate any length adaptation caused by stretching to the maximum length. Furthermore, we expected that if the L-Ta curve exhibited length adaptation, as in airway and vascular smooth muscles, then stretching to 15 mm in S1 would result in a shift to the right for the L-Ta curves for S2 and S3.
Fig. 3.
A: length-tension (L-T) curve protocol for tissues that were not preconditioned. Protocol consisted of 3 series (S1–S3) of 5 measurements at increasing lengths from 3 to 15 mm. B–F: L-Ta curves (means ± SE) for S1 (white symbols), S2 (black symbols), and S3 (gray symbols in C only) and corresponding L-Tp curves (S1, dotted line; S2, dashed-dotted line; S3, dashed line in C only). Data from 12 experiments were grouped according to Lo1 and normalized to To1 (B–D) or Ta_max (E). For S1–S3, peak (optimal) Ta values are labeled To1–To3 and correspond to optimal lengths Lo1 (6 mm, B; 9 mm, C; 12 mm, D; and ≥15 mm, E); Lo2 (12 mm, B and C, ≥15 mm, D and E); and Lo3 (12 mm, C). B: L-Ta and L-Tp curves for S1 and S2 for tissues with Lo1 at 6 mm (n = 3). C: L-Ta and L-Tp curves for S1–S3 for tissues with Lo1 at 9 mm (n = 3). D: L-Ta and L-Tp curves for S1 and S2 for tissues with Lo1 at 12 mm (n = 4). E: L-Ta and L-Tp curves for S1 and S2 for tissues with Lo1 ≥15 mm (n = 2). F: L-Ta and L-Tp curves for S1 and S2 for all 12 tissues (n = 12). B–F: *P < 0.05, Ta for S2 was significantly less than Ta for S1 at that particular length (paired t-test). C: ΨP < 0.05, Ta for S3 was significantly less than Ta for S2 at that particular length (paired t-test).
Effect of preconditioning on the L-Tt curve.
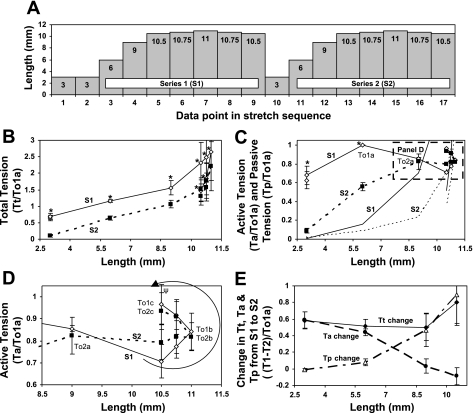
The L-T curve protocol illustrated in Fig. 4A was designed to determine whether the L-Tt curve shifts due to preconditioning and whether any shift was a result of a change in Tp, Ta, or both. The protocol consisted of two series (S1 and S2) of measurements at lengths between 3 and 11 mm, as shown in Fig. 4A. We took smaller length steps at longer muscle lengths to better identify any peak in the L-Ta curve in this region. We expected to find that changes in both Tp and Ta shifted the L-Tt curve.
Fig. 4.
A: L-T curve protocol consisting of 2 series measurements (S1 and S2) to determine if the L-Tt curve shifts due to preconditioning. B: L-Tt curves for S1 (solid line, open symbols) and S2 (dashed line, solid symbols). *P < 0.05 Tt for S2 was significantly less than Tt for S1 at that particular length (paired t-test, n = 4). C and D: full (C) and zoomed (D, box from C) L-Ta curves. Localized peak Ta values for S1 and S2 are labeled To1a (C), To1b (D), and To1c (D) and To2a (C and D), To2b (D), and To2c (D). C: L-Tp curves for S1 (solid line) and S2 (dotted line). *P < 0.05 Ta for S2 was significantly less than Ta for S2 at a particular length <9 mm (paired t-test, n = 4). D: at 10.75 and 11 mm, Ta values for S1 were not different from the corresponding values for S2 (paired t-test, P > 0.05, n = 4). The second Ta values at 10.5 mm were significantly greater than the first for both S1 and S2 (ΨP < 0.05, paired t-test, n = 4), forming counterclockwise loops (arrow) suggesting adaptation to that length region. E: changes in Tt (solid line), Ta (dashed line), and Tp (dashed-dotted line) between S1 and S2 for lengths between 3 and 10.5 mm. The change in Tt between S1 and S2 was due to the change in Ta at shorter lengths and due to the change in Tp at longer lengths.
Effect of multiple contractions on the L-Ta curve at short lengths.
The L-T protocol shown in Fig. 5A was designed to determine whether the reduction in Ta at short muscle lengths following preconditioning identified with a previous protocol (Fig. 3C and see Effect of Preconditioning on the L-Ta Curve) could be restored by multiple contractions at a short length. This protocol consisted of two series of stretches and tension measurements between 3 and 9 mm (S1 and S2) followed by a series of six tension measurements at 3 mm and for some tissues measurements at 6 and 9 mm (S3). We chose to study length adaptation at a short muscle length, 3 mm, where Tp was relatively small (Fig. 3, B–E, and see results) to minimize any changes in Tt due to shifts in the L-Tp curve due to APS. This protocol was designed to determine whether any reduction in Ta at a short length due to preconditioning to a longer length could be restored by multiple contractions at the short length and could therefore be attributed to length adaptation and not to tissue damage.
Fig. 5.
A: protocol to identify adaptation of the L-Ta curve at 3 mm consisting of 2 series of stretches and tension measurements between 3 and 9 mm (S1 and S2) followed by a series of 6 tension measurements at 3 mm and for some tissues measurements at 6 and 9 mm (S3). B–D: L-Ta (solid lines) and L-Tp (dotted lines) curves for S1 (diamonds), S2 (triangles), and S3 (squares) (±SE at 6 and 9 mm). Data were categorized according to the shape of the L-Ta curve produced by S1 and normalized to Ta_max. B: data for tissues producing at least 10% greater Ta at 9 than at 6 mm during S1 were categorized as tissues with a “more steep ascent 6–9 mm” (n = 9 for data points 1–13 and n = 8 for data points 14 and 15). C: data for tissues producing <10% greater Ta at 9 than at 6 mm during S1 were categorized as tissues with a “less steep ascent 6–9 mm” (n = 6, data points 1–13 only). D: data for tissues producing greater Ta at 6 than at 9 mm during S1 were categorized as tissues with a “descent 6–9 mm” (n = 5 for data points 1–13 and n = 3 for data points 14 and 15). D: data for tissues incubated in NPSS for 7 min during each tension measurement cycle, instead of 4 min in 0Ca and 3 min in NPSS (see Figs 1–2) were categorized as tissues with an “ascent 6–9 mm, no 0Ca” (n = 4). F: Ta values (±SE) for contractions at 3 mm, corresponding to the data in B–E, for S1 (dark gray), S2 (white), and S3 (light gray), along with the increase in Ta from contractions 8 to 13 (T_adapt, “13–8”, calculated as Ta for 13 minus Ta for 8). F: * NS indicates whether Ta for contraction 8 or 13 at 3 mm was (*) or was not (NS) significantly different from the Ta value for contraction 2 at 3 mm for that category (ANOVA, Newman-Keuls multiple comparison test), and Ψ indicates T_adapt (“13–8”) was not different from T_adapt for the other categories (ANOVA, Newman-Keuls multiple comparison test).
Effect of multiple contractions vs. time in NPSS on isometric Ta.
An additional protocol was designed to determine whether the increase in Ta during successive isometric contractions on the ascending limb of the L-Ta curve identified with a previous protocol (Fig. 5B and see Adaptation of the L-Ta curve at Lengths Greater Than Lo) could also be achieved by incubating tissues in NPSS for an equivalent period of time. Following an initial contraction at 3 mm, tissues were stretched in 0Ca to 12 mm, and tension measurements were made to determine the Tp/Ta ratio. If necessary, tissue lengths were adjusted and the Tp/Ta ratio was measured again until it was 18 ± 4%, corresponding to the Tp/Ta ratio at the initial optimal tension-generating length (Lo) identified using the protocol in Fig. 3A (see Effect of Preconditioning on the Tp/Ta Ratio at Lo). Using this length as a reference length, Lref, tissues were subjected to the protocol in Fig. 6A. To adapt tissues back and forth between Lref and 0.8Lref, control tissues were subjected to three sets of three tension measurement cycles (see Fig. 1A) at Lref alternated with three sets of four contraction cycles at 0.8Lref (Fig. 6A). To determine whether tissues would adapt as well during incubation in NPSS as during multiple KCl-induced contractions, the first three 8-min tension measurement cycles in the second set of measurements at 0.8Lref (Fig. 6A, points 12–14, Test) were replaced with an equivalent duration of 24 min of incubation in NPSS. Similarly, the second and third measurement cycles of the final set of measurements at 0.8Lref (Fig. 6A, points 20 and 21, Test) were replaced with 16 min of incubation in NPSS.
Fig. 6.
A: protocol to determine if Ta increased as much following incubation in NPSS as following multiple KCl-induced contractions. Control tissues were subjected to 3 sets of 3 8-min tension measurement cycles at Lref alternated with 3 sets of 4 cycles at 0.8Lref. For “test” tissues, cycles 12–14 and 20–21 at 0.8Lref were replaced with 24- and 16-min incubations in NPSS, respectively. B: Ta values for the first and fourth cycles of each series of measurements at 0.8Lo were normalized to Ta for the first contraction at 0.8Lo, Ta_5 (data point 5, ±SE). Ta increased during initial control contractions in both groups (8 vs. 5, *P < 0.05 compared with 1). Ta did not increase following incubation in NPSS for 24 min (15 vs. 5, test, n = 5), but did increase following 3 isometric contractions (15 vs. 12 and 5, control, n = 4). C: Ta adapted (increased) following initial control contractions in both groups (8 minus 5, ΩP < 0.05 compared with 0). However, Ta did not significantly adapt following 16 min in NPSS but did adapt following KCl-induced contractions (22 minus 19, test vs. control, ΩP < 0.05 compared with 0).
Statistics
A one-way analysis of variance with a Student-Newman-Keuls post hoc test or a t-test with the Bonferroni correction was used to determine significance, and the null hypothesis was rejected at P < 0.05. The population sample size (n value) refers to the number of animals, not the number of tissues. Statistical analyses were performed using Excel (2003, Microsoft) or Prism (5.0, GraphPad Software).
RESULTS
Effect of Preconditioning on the L-Ta Curve
The protocol in Fig. 3A was designed to determine whether the L-Ta curve displays stronger Ta values at short lengths before preconditioning compared with after preconditioning and therefore shifts the L-Ta curve to the right. The L-Ta and L-Tp curves for three series (S1–S3) of five L-T measurements at increasing lengths from 3 to 15 mm are shown in Fig. 3, B–E. Data were grouped according to the optimal length for contraction, Lo1, corresponding to the optimal (maximum) Ta value, To1, for S1. Data for tissues with Lo1 values at 6, 9, 12, and 15 mm are shown separately in Fig. 3, B–E, respectively. The length axis was intentionally not normalized to Lo to demonstrate variability and the challenge of identifying a reference length for a particular muscle strip, especially if the tissue was not preconditioned. These data indicate that although we took great care in hanging the tissue strips to ensure uniformity in muscle strip size and initial length, there was significant variation in Lo. This was likely due to several sources: 1) variation in tissue damage during stretching, especially local damage at the tissue clamps; 2) variation in the “kinks” in the tissue established while exposed to cold buffer; 3) variation in the percentage of smooth muscle relative to connective tissue in the muscle strip due to variation between animals or variation in the location from which the strip was excised; and 4) variation in the tension in the strip when it was clamped. Note that Tp was approximately zero at the initial length of 3 mm for all tissues. Ideally, the tissue was just slack, but it could have been slightly over slack or slightly stretched and stress-relaxed, and small differences in the initial degree of stretch when placed in the tissue clips likely would expand to larger differences at longer lengths. This variability increased the challenge of designing and performing experiments; however, in this case the variability enabled us to quantify the effect of preconditioning on strips stretched well beyond the initial Lo (Fig. 5, B and C), just beyond Lo (Fig. 5D), and not quite to Lo (Fig. 5E) using a single protocol. When data from all 12 experiments were grouped together, the resulting curves still display a shift in the L-Ta and L-Tp curves (Fig. 3F).
In a comparison of S2 to S1 in Fig. 3B for tissues with Lo1 at 6 mm, Ta was significantly less at shorter lengths, and To2 was less than To1 and was shifted to the right from 6 to 12 mm (Lo2). Similarly, in a comparison of S2 to S1 in Fig. 3C for tissues with Lo1 at 9 mm, Ta was significantly less at shorter lengths, and To2 was less than To1 and was shifted to the right from 9 to 12 mm (Lo2). For tissues with Lo1 at 12 mm in Fig. 3D, Ta for S2 was significantly less than Ta for S1 at shorter lengths, and To2 was shifted to the right from 12 to ∼15 mm or beyond. However, in contrast to the tissues with Lo1 at 6 or 9 mm (Fig. 3, B and C), Ta at 15 mm for S2 was not significantly different from To1 at 12 mm (Fig. 3D, n = 4, P > 0.05). Furthermore, although Ta was significantly reduced in S2 compared with S1 at all shorter lengths, Ta values produced at the maximum length in the protocol, 15 mm, were not different for S1 and S2, regardless of the Lo1 value (Fig. 3, B–D). Together, the data in Fig. 3, B–E, reveal that preconditioning collapsed the ascending limb of the L-Ta curve when tissues were stretched beyond Lo1 and that the farther the tissue was stretched and contracted beyond Lo, the greater the increase (rightward shift) in Lo2 and the greater the decrease in To2. Furthermore, the reduction in Ta due to preconditioning was greater at shorter lengths compared with longer lengths. Most importantly, these data demonstrate that Lo can shift significantly upon preconditioning, and therefore preconditioning should be carefully considered during experiment design.
To determine whether a subsequent series of stretches and contractions would reveal additional changes in the L-Ta curves, we examined S3 (Fig. 3C) for tissues with Lo1 at 9 mm. In a comparison of S2 with S3 in Fig. 3C, Lo was not shifted to a longer length, and compared with the decrease in Ta from S1 to S2 at 3, 6, and 9 mm, the decrease from S2 to S3 was considerably less.
Effect of Preconditioning on the Tp/Ta Ratio at Lo
The L-Tp curves for S2 in Fig. 3, B–D, exhibited a shift to the right compared with S1 that corresponded to the shift in the L-Ta curves. Before preconditioning beyond Lo1 in S1, the passive-to-active ratio at Lo1 (Tp at To1)/To1 was 18.7 ± 4.0% (n = 10, data from Fig. 3, B–D). After preconditioning in S2, Tp at Lo1 was reduced (Fig. 3, B–D); however, because of the shift to the right from Lo1 to Lo2 and the reduction in Ta from To1 to To2, the passive-to-active ratio at Lo2 (Tp at To2)/To2 increased to 39.5 ± 5.0% (n = 6, data from Fig. 3, B and C). Thus, while Tp at a given length was reduced by preconditioning, the combined shifting of the L-Ta and L-Tp curves due to preconditioning produced a higher Tp/Ta ratio at Lo2.
Effect of Preconditioning on the L-Tt Curve
The L-T curve protocol illustrated in Fig. 4A produced the two L-Tt curves in Fig. 4B. Tt for S2 was significantly lower at each length, revealing that preconditioning from S1 shifted the L-Tt curve for S2 downward and to the right. Ta was reduced more at shorter than longer lengths, while Tp was reduced more at longer than shorter lengths (Fig. 4C). Thus the change in Tt between S1 and S2 was due to the change in Ta at shorter lengths (Fig. 4E, on the ascending limb of the S1 L-Ta curve in Fig. 4C) and due to the change in Tp at longer lengths (Fig. 4E, on the descending limb of the S1 L-Ta curve in Fig. 4C).
Adaptation of the L-Ta Curve at Lengths Greater Than Lo
Figure 4D shows zoomed L-Ta curves from the boxed region of Fig. 4C. In this region, the Ta values at 10.75 and 11 mm for S1 were not different from the corresponding values for S2, indicating that preconditioning did not significantly alter Ta near the maximum length in the protocol, 11 mm (Fig. 4D, n = 4, P > 0.05). However, the second Ta values measured at 10.5 mm (during unloading) were significantly greater than the first (during loading) for both S1 and S2 (Fig. 4D), forming counterclockwise loops, suggesting adaptation of Ta to that length region. Furthermore, the To1c and To2c values at 10.5 mm were not significantly different from the To1a value at 6 mm (Fig. 4, C and D), indicating that for both S1 and S2, the tissues were able to adapt back to their initial To1 values while at 175% of Lo1.
L-Ta Curves with Multiple Ascending and Descending Limbs
In Fig. 4C, the stretches and contractions in S1 produced an L-Ta curve with an ascending limb that reached To1a at 6 mm and a descending limb between 6 and 10.5 mm. A second ascending limb was produced between 10.5 and 11 mm, with peak To1b at 11 mm, and a second descending limb was produced during unloading between 11 and 10.5 mm with peak value To1c at 10.5 mm (Fig. 4D). Thus the L-Ta curve for S1 has two ascending limbs and two descending limbs. The L-Ta curve for S2 follows a similar pattern with multiple ascending and descending limbs and three peak values, To2a–To2c. The presence of multiple ascending and descending limbs indicates that there is not a single, static L-Ta curve for DSM.
Adaptation of the L-Ta curve at Lengths Less Than Lo
Data from tissues subjected to the L-T protocol in Fig. 5A were categorized according to the shape of the L-Ta curve produced by S1. Data for tissues producing at least 10% greater Ta at 9 than at 6 mm during S1 are shown in Fig. 5B and were categorized as tissues with a “more steep ascent 6–9 mm.” Data for tissues producing <10% greater Ta at 9 than at 6 mm during S1 are shown in Fig. 5C and were categorized as tissues with a “less steep ascent 6–9 mm.” Data for tissues producing greater Ta at 6 than at 9 mm during S1 are shown in Fig. 5D and were categorized as tissues with a “descent 6–9 mm.” Finally, data from a protocol in which 0Ca was replaced with NPSS are shown in Fig. 5E and categorized as tissues with an “ascent 6–9 mm, no 0Ca.”
For each of the four categories, Ta for contraction 8 was significantly less than Ta for contraction 2 at 3 mm (Fig. 5F, compare data points 2 and 8 for each category), following the stretches to and contractions at 6 and 9 mm (Fig. 5A, points 3–7). For tissues not stretched to the descending limb of the L-Ta curve, Ta at 3 mm was reduced by 55.8 ± 4.9% for the more steep ascent group and by 62.9 ± 7.2% for the less steep ascent group following the stretches to and contractions at 6 and 9 mm (Fig. 5F, compare data points 2 and 8). For the two ascent groups combined, Ta at 3 mm was reduced by 58.6 ± 4.1% (n = 15). For tissues stretched to the descending limb of the L-Ta curve, Ta was reduced by 84.5 ± 2.8% at 3 mm, following the stretches to and contractions at 6 and 9 mm (Fig. 6F, compare data points 2 and 8).
For each of the four categories, the multiple contractions at 3 mm in S3 produced increasingly greater Ta (Fig. 5F, data points 8–13). For the more steep ascent group, Ta produced by the sixth consecutive contraction at 3 mm reached 103.3 ± 12.3% of, and was not significantly different from, the second prestretch contraction at that length (Fig. 5F, compare data points 2 and 13). For the less steep ascent group, Ta produced by the sixth consecutive contraction at 3 mm reached 73.9 ± 10.8% of the second prestretch contraction at that length (Fig. 5F, compare data points 2 and 13). For the two ascent groups combined, the sixth contraction in S3 produced 91.5 ± 9.1% of the Ta produced by the second prestretch contraction at 3 mm in S1. Together, these data show that multiple contractions at 3 mm enabled Ta to adapt to a greater value and in some cases back to its prestretch value at that length.
For tissues stretched to the descending limb of the L-Ta curve, multiple contractions at 3 mm enabled Ta to adapt, but Ta only returned to 47.8 ± 7.2% of the prestretch Ta value at that length by the sixth contraction (Fig. 5F, descent 6–9 mm, compare data points 2 and 13). However, the level of adaptation of Ta for S3, T_adapt [(Ta of first minus Ta of last contraction in S3)/Ta_max)] was not significantly different for the four categories (“13–8”, Fig. 5F). These results indicate that a significant portion of the change in Ta at short lengths due to preconditioning was due to adaptation and not irreversible tissue damage. Furthermore, the adaptation could be reversed by KCl-induced contractions at a short length. Finally, replacing 0Ca with NPSS in the protocol did not change the trends in the data (Fig. 5, E and F, Ascent 6–9 mm, no 0Ca), indicating that adaptation was not an artifact of the 0Ca solution.
Effect of Multiple Contractions vs. Time in NPSS on Isometric Ta
Following the determination of a reference length (11.66 ± 0.10 mm, means ± SE, n = 9) at which the Tp/Ta ratio (18.4 ± 0.7%, range 15–22%) corresponded to the Tp/Ta ratio at Lo1 for the data in Fig. 3 (18.7 ± 4%, see Effect of Preconditioning on the Tp/Ta Ratio at Lo), tissues were subjected to the protocol in Fig. 6A to determine whether Ta adapted as much during incubation in NPSS as during multiple KCl-induced contractions. Ta increased during initial control contractions in both the test and control groups (8 vs. 5, *). However, Ta did not increase following incubation in NPSS for 24 min (Fig. 6B, 15 vs. 5, test) but did increase following three isometric contractions (Fig. 6B, 15 vs. 12 and 5, control). The degree of adaptation for control tissues was 17.5 ± 2.6 and 15.1 ± 1.7%, respectively, for the first and third sets of contractions at 0.8Lref (Fig. 6C, 8−5 and 22−19). Although the average value of Ta increased following 16 min in NPSS, this apparent increase was not statistically significant (Fig. 6B, 22 vs. 5). Moreover, the degree of adaptation following 16 min in NPSS was not greater than zero (Fig. 6C, 22−5, test). These results show that 16–24 min of incubation in NPSS did not, whereas three KCl-induced contractions did, permit tissues to adapt at 0.8Lref.
DISCUSSION
L-T Curves From Other Studies
In Fig. 7A, L -Ta and L-Tp curves from the present study were compared with curves approximated from studies by Uvelius (47) and Longhurst et al. (27) and normalized to To and Lo. When normalized to To1 and Lo1, our data from S1 in Fig. 3C (Fig. 7A, ♦) were consistent with those of Uvelius (squares) and those of Longhurst et al. (circles).
Fig. 7.
Normalized L-Ta and L-Tp curves. A: data from S1 in Fig 3C normalized to To1 and Lo1 (Ta/To1, ♦), data approximated from Ref. 47 normalized to To and Lo (□), and data approximated from Ref. 27 normalized to To and Lo (○). B: data from S1 in Fig 3C normalized to To1 and Lo1 (Ta/To1, ♦) and data from S2 in Fig 3C normalized to To1 and Lo1 (Ta/To1, ●, dashed line) and normalized to To2 and Lo2 (Ta/To2, ▲).
In Fig. 7B, the data from S1 and S2 in Fig. 3C were normalized to To1 and Lo1 (S1, ♦; S2, ●), and the L-Ta and L-Tp curves for S2 were clearly shifted to the right compared with S1, as previously described. However, when the data from S2 were normalized to To2 and Lo2 (Fig. 7B, ▲), they were much more consistent with the data from S1 (♦) and the data from other studies in Fig. 7A. Furthermore, when normalized to To1 and Lo1, the ascending limb of the S2 L-Ta curve was less steep compared with that the S1 curve, but when normalized to To2 and Lo2, the ascending limb of the S2 L-Ta curve was more steep than that of the S1 curve. Thus, if the data in S1 were not recorded, as might be the case if tissues were preconditioned before the L-Ta curve was produced, then only the data from S2 would have been observed, and the significant shift from the S1 curve to the S2 curve (Fig. 7B, compare circles to triangles) would not have been observed. Moreover, if normalized to Lo and To, the S2 curve would appear consistent with tissues that were not preconditioned, S1, and the effect of preconditioning would be missed (compare triangles to diamonds). Three key observations from these comparisons are that 1) DSM strips clearly produce an L-Ta curve with ascending and descending limbs, 2) a single DSM strip can produce two very different L-Ta curves, and 3) normalization of the L-Ta curve to Lo and To may hide important characteristics, like length adaptation, by making two very different curves appear similar.
Shifting of the L-Ta and L-Tp Curves due to Preconditioning, APS, and Length Adaptation
One important contribution provided by our current study is strong evidence that the L-Ta relationship in DSM can best be described by a series of curves over a broad range of muscle lengths (see Fig. 3C) rather than by a unique parabolic curve. We believe that this is the first study to identify length adaptation of the L-Ta curve in DSM. The data in Figs. 3, B–D, and 4C show that the L-Ta and L-Tp curves shifted down and to the right following a series of contractions at progressively longer lengths, indicating that preconditioning caused the L-T curves to shift to longer lengths. These data also indicate that the farther a tissue strip was stretched beyond Lo, the greater the shift in the L-Ta curve (compare Figs. 3, B with D, and 5, B with D), but if a tissue strip was not stretched beyond Lo, there was no significant change in Ta at intermediate lengths (Fig. 5B, Ta at 6 mm, n = 8, P > 0.05; and trend in Fig. 3E). Our results in Fig. 4E demonstrate a decrease in Tt at short lengths that was due to a decrease in Ta upon preconditioning, and Fig. 5, B–F, shows that at least some of this decrease in Ta could be reversed by adaptation resulting from one or more additional contractions at that short length. Neither the increase in Ta at 3 mm (Fig. 5, B–F) nor the decrease in Ta at 3 mm following preconditioning (Figs. 3, B and C, 4C, and 5, B–E) resulted from a decrease in Tp due to stress relaxation across the repeated contraction cycles because Tp was essentially zero at this length and Tp was measured before each contraction. In Fig. 5B, the reduction in Ta at the short length following preconditioning was completely restored, indicating that the reduction was due to adaptation and not irreversible preconditioning. However, for tissues preconditioned beyond the initial Lo, the data in Fig. 5D indicate that approximately half of the reduction in Ta upon preconditioning was reversible and attributable to adaptation, while the other half appeared irreversible. Thus preconditioning can induce both a reversible and an irreversible shift in the L-Ta curve. The reversible portion of the rightward shift in the L-Ta curve following preconditioning is due to length adaptation. Moreover, Fig. 4, C and D, reveals that in addition to the adaptation due to preconditioning in S1, multiple contractions at longer muscle lengths in S2 adapted the optimal length from Lo2a to Lo2c. In summary, we have identified length adaptation of the L-Ta relationship in DSM which is characterized by a decrease in Ta due to subsequent stretches to and contractions at progressively longer lengths and by an increase in Ta as a result of one or more contractions at or near a particular muscle length.
Another important contribution of this study is evidence that the L-Tt curve is shifted by changes in the L-Tp curve in addition to length adaptation of the L-Ta curve. Figure 4E demonstrates a decrease in Tt at longer lengths that is due to a decrease in Tp resulting from preconditioning. Together, the data in Figs. 3–5 demonstrate that the L-Ta and L-Tp relationships in rabbit DSM are dynamic functions of both strain and activation history.
Choosing a Reference Length for DSM Strips
Given evidence that both the L-Tp and L-Ta curves in DSM are dynamic and can shift as a function of strain and activation history, the choice of a suitable reference length becomes quite challenging. Initially, all tissues in the present study were essentially slack with Tp ≈ 0 at 3 mm; however, Ta_max occurred at 6 mm in some tissues and at 15 mm or beyond in others (Fig. 3, B–E). Therefore, passive slack length may not be a suitable reference length. Furthermore, Lo shifted from 6 to 11 and then to 10.5 mm in some tissues (Fig. 4, C and D); therefore, Lo may also not be a suitable reference length. Thus, for studies involving DSM strips, the dynamic nature of the L-Ta and L-Tp curves should be considered when a reference length is chosen. In the present study, we stretched tissues from an initial length of 3 mm to a maximum length of 9–15 mm and then grouped our data based on trends in the L-Ta curve and the length at which the first peak Ta value was identified.
Potential Mechanical Models of Length Adaptation in DSM
A number of other models have been developed to account for the dynamic L-T relationship in smooth muscle. Fabry and Fredberg et al. (9, 10, 40, 41) have described smooth muscle as a soft glassy material in which the cytoskeleton can deform, flow, and reorganize. Gunst et al. (18, 19) attributed adaptation to shifting of the sites where actin filaments connect to dense bodies and to actin polymerization. Seow and Ford et al. (31, 37) have developed models in which contractile units are added in series or parallel and a model in which myosin filaments lengthen (13) as a smooth muscle adapts to longer lengths (reviewed in Refs. 3, 5, 11, 34, 36). In addition, Solway et al. (7) proposed that changes in actin filament length and a parallel-to-series transition of the contractile units contribute to smooth muscle plasticity. Our data in Fig. 3C, which show a shift from a taller, narrower L-T curve to a shorter, wider curve, are consistent with this parallel-to-series model. Furthermore, our data in Figs. 4D, 5, B–E, and 6B, which show increased Ta at a particular length following multiple contractions at or near that muscle length, are consistent with the formation of additional cross bridges in parallel and with the lengthening of myosin and/or actin filaments. Moreover, our data in Fig. 6B show that 24 min in NPSS were insufficient to adapt Ta, indicating that the KCl-induced contractions, and not time alone, were responsible for length adaptation. Additional studies are necessary to determine the specific mechanisms responsible for length adaptation in DSM.
Potential Clinical Significance
Overactive bladder (OAB) affects 50–100 million people worldwide, and in the US ranks ahead of diabetes in a list of the 10 most common chronic disorders (1). OAB is a complex disorder that involves involuntary DSM contractions during bladder filling that may be associated with several different conditions (46), including accentuated DSM wall stiffness that could trigger urgency or premature voiding (6). During filling, both passive structures and low-grade rhythmic contractions may contribute to bladder wall tension (30). We speculate that length adaptation plays a significant role in bladder physiology and that pathological alterations in this system could potentially play a role in bladder disorders. Thus understanding the molecular mechanisms responsible for length adaptation in DSM may provide information essential for the timely discovery of therapies to specifically target OAB and other bladder contractile disorders.
GRANTS
This study was supported by a grant from the Edwin Beer Research Program in Urology and Urology Related Fields from the New York Academy of Medicine (to J. E. Speich). Some bladders used for these studies were from rabbits used for vascular smooth muscle studies supported by National Heart, Lung, and Blood Institute Grant R01-HL61320 (to P. H. Ratz).
ACKNOWLEDGMENTS
We gratefully acknowledge the expert technical assistance of Amy S. Miner.
REFERENCES
- 1. Abrams P, Wein AJ. Introduction: overactive bladder and its treatment. Urology 55: 1–2, 2000. 10767441 [Google Scholar]
- 2. Ali F, Chin L, Pare PD, Seow CY. Mechanism of partial adaptation in airway smooth muscle after a step change in length. J Appl Physiol 103: 569–577, 2007 [DOI] [PubMed] [Google Scholar]
- 3. Ali F, Pare PD, Seow CY. Models of contractile units and their assembly in smooth muscle. Can J Physiol Pharmacol 83: 825–831, 2005 [DOI] [PubMed] [Google Scholar]
- 4. Bai TR, Bates JH, Brusasco V, Camoretti-Mercado B, Chitano P, Deng LH, Dowell M, Fabry B, Ford LE, Fredberg JJ, Gerthoffer WT, Gilbert SH, Gunst SJ, Hai CM, Halayko AJ, Hirst SJ, James AL, Janssen LJ, Jones KA, King GG, Lakser OJ, Lambert RK, Lauzon AM, Lutchen KR, Maksym GN, Meiss RA, Mijailovich SM, Mitchell HW, Mitchell RW, Mitzner W, Murphy TM, Pare PD, Schellenberg RR, Seow CY, Sieck GC, Smith PG, Smolensky AV, Solway J, Stephens NL, Stewart AG, Tang DD, Wang L. On the terminology for describing the length-force relationship and its changes in airway smooth muscle. J Appl Physiol 97: 2029–2034, 2004 [DOI] [PubMed] [Google Scholar]
- 5. Bosse Y, Sobieszek A, Pare PD, Seow CY. Length adaptation of airway smooth muscle. Proc Am Thorac Soc 5: 62–67, 2008 [DOI] [PubMed] [Google Scholar]
- 6. de Groat WC. A neurological basis for the overactive bladder. Urology 50: 36–52, 1997 [DOI] [PubMed] [Google Scholar]
- 7. Dulin NO, Fernandes DJ, Dowell M, Bellam S, McConville J, Lakser O, Mitchell R, Camoretti-Mercado B, Kogut P, Solway J. What evidence implicates airway smooth muscle in the cause of BHR? Clin Rev Allergy Immunol 24: 73–84, 2003 [DOI] [PubMed] [Google Scholar]
- 8. Emery JL, Omens JH, McCulloch AD. Strain softening in rat left ventricular myocardium. J Biomech Eng 119: 6–12, 1997 [DOI] [PubMed] [Google Scholar]
- 9. Fabry B, Fredberg JJ. Remodeling of the airway smooth muscle cell: are we built of glass? Respir Physiol Neurobiol 137: 109–124, 2003 [DOI] [PubMed] [Google Scholar]
- 10. Fabry B, Maksym GN, Butler JP, Glogauer M, Navajas D, Fredberg JJ. Scaling the microrheology of living cells. Phys Rev Lett 87: 148102, 2001 [DOI] [PubMed] [Google Scholar]
- 11. Ford LE. Plasticity in airway smooth muscle: an update. Can J Physiol Pharmacol 83: 841–850, 2005 [DOI] [PubMed] [Google Scholar]
- 12. Ford LE, Gilbert SH. The significance of variable passive compliance in smooth muscle. J Appl Physiol 102: 1735–1736, 2007 [DOI] [PubMed] [Google Scholar]
- 13. Ford LE, Seow CY, Pratusevich VR. Plasticity in smooth muscle, a hypothesis. Can J Physiol Pharmacol 72: 1320–1324, 1994 [DOI] [PubMed] [Google Scholar]
- 14. Fung YC. Biomechanics. New York: Springer-Verlag, 1993 [Google Scholar]
- 15. Gordon AM, Huxley AF, Julian FJ. The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J Physiol 184: 170–192, 1966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Gordon AR, Siegman MJ. Mechanical properties of smooth muscle. I. Length-tension and force-velocity relations. Am J Physiol 221: 1243–1249, 1971 [DOI] [PubMed] [Google Scholar]
- 17. Gregersen H, Emery JL, McCulloch AD. History-dependent mechanical behavior of guinea-pig small intestine. Ann Biomed Eng 26: 850–858, 1998 [DOI] [PubMed] [Google Scholar]
- 18. Gunst SJ, Meiss RA, Wu MF, Rowe M. Mechanisms for the mechanical plasticity of tracheal smooth muscle. Am J Physiol Cell Physiol 268: C1267–C1276, 1995 [DOI] [PubMed] [Google Scholar]
- 19. Gunst SJ, Tang DD, Opazo Saez A. Cytoskeletal remodeling of the airway smooth muscle cell: a mechanism for adaptation to mechanical forces in the lung. Respir Physiol Neurobiol 137: 151–168, 2003 [DOI] [PubMed] [Google Scholar]
- 20. Gunst SJ, Wu MF. Selected contribution: plasticity of airway smooth muscle stiffness and extensibility: role of length-adaptive mechanisms. J Appl Physiol 90: 741–749, 2001 [DOI] [PubMed] [Google Scholar]
- 21. Gunst SJ, Zhang W. Actin cytoskeletal dynamics in smooth muscle: a new paradigm for the regulation of smooth muscle contraction. Am J Physiol Cell Physiol 295: C576–C587, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Herlihy JT, Murphy RA. Length-tension relationship of smooth muscle of the hog carotid artery. Circ Res 33: 257–283, 1973 [DOI] [PubMed] [Google Scholar]
- 23. Herrera AM, McParland BE, Bienkowska A, Tait R, Pare PD, Seow CY. ‘Sarcomeres’ of smooth muscle: functional characteristics and ultrastructural evidence. J Cell Sci 118: 2381–2392, 2005 [DOI] [PubMed] [Google Scholar]
- 24. Jezior JR, Brady JD, Rosenstein DI, McCammon KA, Miner AS, Ratz PH. Dependency of detrusor contractions on calcium sensitization and calcium entry through LOE-908-sensitive channels. Br J Pharmacol 134: 78–87, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kuo KH, Herrera AM, Wang L, Pare PD, Ford LE, Stephens NL, Seow CY. Structure-function correlation in airway smooth muscle adapted to different lengths. Am J Physiol Cell Physiol 285: C384–C390, 2003 [DOI] [PubMed] [Google Scholar]
- 26. Lambert RK, Pare PD, Seow CY. Mathematical description of geometric and kinematic aspects of smooth muscle plasticity and some related morphometrics. J Appl Physiol 96: 469–476, 2004 [DOI] [PubMed] [Google Scholar]
- 27. Longhurst PA, Kang JS, Wein AJ, Levin RM. Comparative length-tension relationship of urinary bladder strips from hamsters, rats, guinea-pigs, rabbits and cats. Comp Biochem Physiol A 96: 221–225, 1990 [DOI] [PubMed] [Google Scholar]
- 28. Minekus J, van Mastrigt R. Length dependence of the contractility of pig detrusor smooth muscle fibres. Urol Res 29: 126–133, 2001 [DOI] [PubMed] [Google Scholar]
- 29. Naghshin J, Wang L, Pare PD, Seow CY. Adaptation to chronic length change in explanted airway smooth muscle. J Appl Physiol 95: 448–453, 2003 [DOI] [PubMed] [Google Scholar]
- 30. Plum F, Colfelt RH. The genesis of vesical rhythmicity. AMA Arch Neurol 2: 487–496, 1960 [Google Scholar]
- 31. Pratusevich VR, Seow CY, Ford LE. Plasticity in canine airway smooth muscle. J Gen Physiol 105: 73–94, 1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ratz PH. High α1-adrenergic receptor occupancy decreases relaxing potency of nifedipine by increasing myosin light chain phosphorylation. Circ Res 72: 1308–1316, 1993 [DOI] [PubMed] [Google Scholar]
- 33. Ratz PH, Miner AS. Length-dependent regulation of basal myosin phosphorylation and force in detrusor smooth muscle. Am J Physiol Regul Integr Comp Physiol 284: R1063–R1070, 2003 [DOI] [PubMed] [Google Scholar]
- 34. Seow CY. Myosin filament assembly in an ever-changing myofilament lattice of smooth muscle. Am J Physiol Cell Physiol 289: C1363–C1368, 2005 [DOI] [PubMed] [Google Scholar]
- 35. Seow CY. Response of arterial smooth muscle to length perturbation. J Appl Physiol 89: 2065–2072, 2000 [DOI] [PubMed] [Google Scholar]
- 36. Seow CY, Pare PD. Ultrastructural basis of airway smooth muscle contraction. Can J Physiol Pharmacol 85: 659–665, 2007 [DOI] [PubMed] [Google Scholar]
- 37. Seow CY, Pratusevich VR, Ford LE. Series-to-parallel transition in the filament lattice of airway smooth muscle. J Appl Physiol 89: 869–876, 2000 [DOI] [PubMed] [Google Scholar]
- 38. Shenfeld OZ, Morgan CW, Ratz PH. Bethanechol activates a post-receptor negative feedback mechanism in rabbit urinary bladder smooth muscle. J Urol 159: 252–257, 1998 [DOI] [PubMed] [Google Scholar]
- 39. Siegman MJ, Butler TM, Mooers SU, Davies RE. Calcium-dependent resistance to stretch and stress relaxation in resting smooth muscles. Am J Physiol 231: 1501–1508, 1976 [DOI] [PubMed] [Google Scholar]
- 40. Silveira PS, Butler JP, Fredberg JJ. Length adaptation of airway smooth muscle: a stochastic model of cytoskeletal dynamics. J Appl Physiol 99: 2087–2098, 2005 [DOI] [PubMed] [Google Scholar]
- 41. Silveira PS, Fredberg JJ. Smooth muscle length adaptation and actin filament length: a network model of the cytoskeletal dysregulation. Can J Physiol Pharmacol 83: 923–931, 2005 [DOI] [PubMed] [Google Scholar]
- 42. Speden RN. The effect of initial strip length on the noradrenaline-induced contraction of arterial strips. J Physiol 154: 15–25, 1960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Speich JE, Borgsmiller L, Call C, Mohr R, Ratz PH. ROK-induced cross-link formation stiffens passive muscle: reversible strain-induced stress softening in rabbit detrusor. Am J Physiol Cell Physiol 289: C12–C21, 2005 [DOI] [PubMed] [Google Scholar]
- 44. Speich JE, Dosier C, Borgsmiller L, Quintero K, Koo HP, Ratz PH. Adjustable passive length-tension curve in rabbit detrusor smooth muscle. J Appl Physiol 102: 1746–1755, 2007 [DOI] [PubMed] [Google Scholar]
- 45. Speich JE, Quintero K, Dosier C, Borgsmiller L, Koo HP, Ratz PH. A mechanical model for adjustable passive stiffness in rabbit detrusor. J Appl Physiol 101: 1189–1198, 2006 [DOI] [PubMed] [Google Scholar]
- 46. Steers WD. Pathophysiology of overactive bladder and urge urinary incontinence. Rev Urol 4 Suppl 4: S7–S18, 2002 [PMC free article] [PubMed] [Google Scholar]
- 47. Uvelius B. Isometric and isotonic length-tension relations and variations in longitudinal smooth muscle from rabbit urinary bladder. Acta Physiol Scand 97: 1–12, 1976 [DOI] [PubMed] [Google Scholar]
- 48. Uvelius B. Length-tension relations of in vitro urinary bladder smooth muscle strips. J Pharmacol Toxicol Methods 45: 87–90, 2001 [DOI] [PubMed] [Google Scholar]
- 49. Wang L, Pare PD, Seow CY. Plasticity in skeletal, cardiac, and smooth muscle: selected contribution: effect of chronic passive length change on airway smooth muscle length-tension relationship. J Appl Physiol 90: 734–740, 2001 [DOI] [PubMed] [Google Scholar]