Abstract
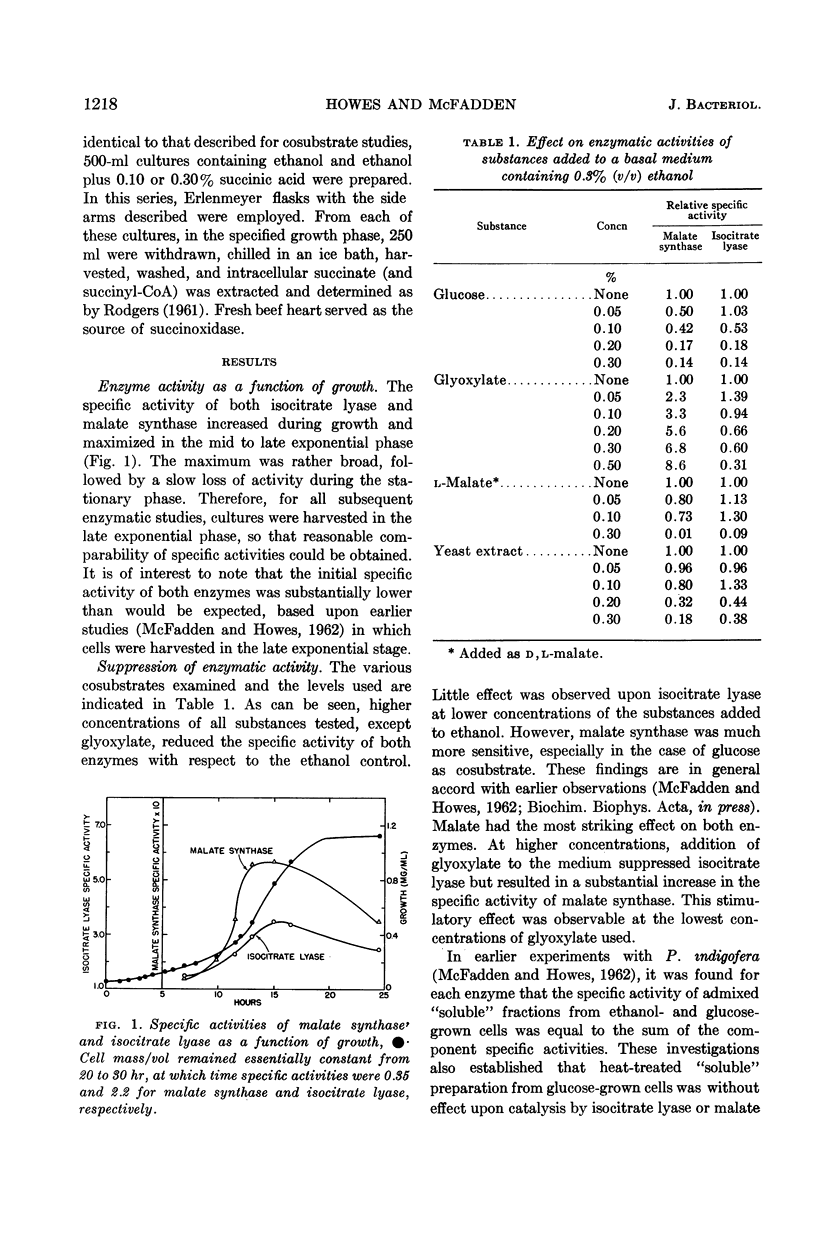
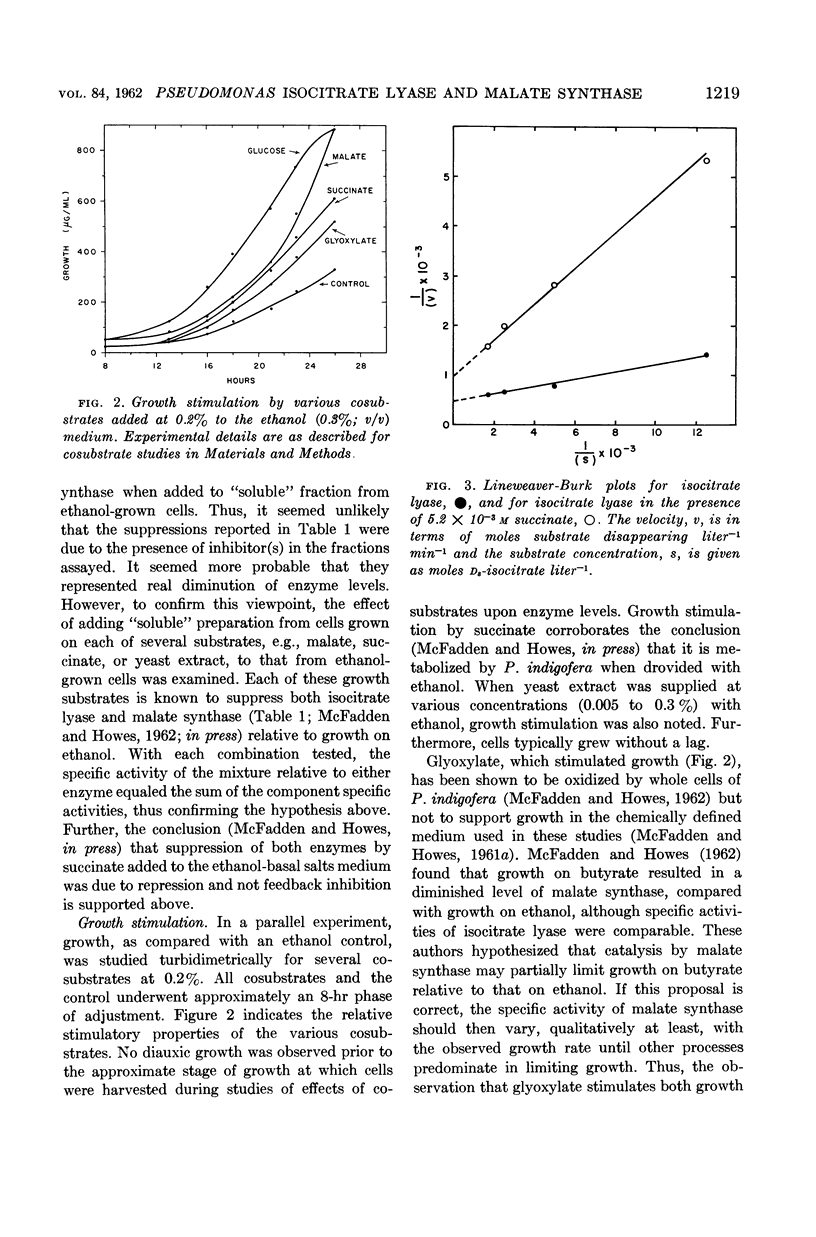
Howes, William V. (Washington State University, Pullman) and Bruce A. McFadden. Isocitrate lyase and malate synthase in Pseudomonas indigofera. I. Suppression and stimulation during growth. J. Bacteriol. 84:1216–1221. 1962.—Specific activities of isocitrate lyase and malate synthase from Pseudomonas indigofera were studied as a function of growth. Each activity maximized in the mid to late exponential phase. Effects upon levels of isocitrate lyase and malate synthase of each of several compounds supplied with ethanol to P. indigofera were next investigated. Compounds tested were glucose, glyoxylate, malate, and yeast extract. Each was added at various concentrations to ethanol. At higher concentrations, glucose, malate, and yeast extract suppressed both enzymatic activities. Malate synthase was more sensitive to suppression. Glyoxylate, at higher concentrations, suppressed isocitrate lyase but substantially stimulated malate synthase. There was no evidence for diauxic growth. Each substrate furnished with ethanol stimulated growth. The possibility that in vivo control of isocitrate lyase activity was partially exerted through feedback inhibition by succinate was examined. Succinate at 5.2 × 10−3m inhibited the enzyme in “soluble” preparations. Intracellular succinate concentrations did not vary significantly at each of three growth phases when exogenous succinate was supplied. Thus, regulation of isocitrate lyase activity through feedback inhibition by succinate seems unlikely.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- JACOB F., MONOD J. Genetic regulatory mechanisms in the synthesis of proteins. J Mol Biol. 1961 Jun;3:318–356. doi: 10.1016/s0022-2836(61)80072-7. [DOI] [PubMed] [Google Scholar]
- KORNBERG H. L., COLLINS J. F., BIGLEY D. The influence of growth substrates on metabolic pathways in Micrococcus denitrificans. Biochim Biophys Acta. 1960 Mar 25;39:9–24. doi: 10.1016/0006-3002(60)90117-7. [DOI] [PubMed] [Google Scholar]
- KORNBERG H. L., ELSDEN S. R. The metabolism of 2-carbon compounds by microorganisms. Adv Enzymol Relat Subj Biochem. 1961;23:401–470. doi: 10.1002/9780470122686.ch8. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MCFADDEN B. A., HOWES W. V. Pseudomonas indigofera. J Bacteriol. 1961 Jun;81:858–862. doi: 10.1128/jb.81.6.858-862.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFadden B. A., Howes W. V. OXIDATIVE METABOLISM AND THE GLYOXYLATE CYCLE IN PSEUDOMONAS INDIGOFERA. J Bacteriol. 1962 Jul;84(1):72–76. doi: 10.1128/jb.84.1.72-76.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PARDEE A. B., PRESTIDGE L. S. The initial kinetics of enzyme induction. Biochim Biophys Acta. 1961 Apr 29;49:77–88. doi: 10.1016/0006-3002(61)90871-x. [DOI] [PubMed] [Google Scholar]
- RODGERS K. Estimation of succinic acid in biological materials. Biochem J. 1961 Aug;80:240–244. doi: 10.1042/bj0800240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SMITH R. A., GUNSALUS I. C. Isocitritase; enzyme properties and reaction equilibrium. J Biol Chem. 1957 Nov;229(1):305–319. [PubMed] [Google Scholar]
- UMBARGER H. E. Evidence for a negative-feedback mechanism in the biosynthesis of isoleucine. Science. 1956 May 11;123(3202):848–848. doi: 10.1126/science.123.3202.848. [DOI] [PubMed] [Google Scholar]



