Abstract
Background: Ischemic necrosis of the femoral head can be induced surgically in the piglet. We used this model to assess femoral head deformation and repair in vivo by sequential magnetic resonance imaging and by correlating end-stage findings with histologic assessments.
Methods: Ischemic necrosis of the femoral head was induced in ten three-week-old piglets by tying a silk ligature around the base of the femoral neck (intracapsular) and cutting the ligamentum teres. We used magnetic resonance imaging with the piglets under general anesthesia to study the hips at forty-eight hours and at one, two, four, and eight weeks. Measurements on magnetic resonance images in the midcoronal plane of the involved and control sides at each time documented the femoral head height, femoral head width, superior surface cartilage height, and femoral neck-shaft angle. Histologic assessments were done at the time of killing.
Results: Complete ischemia of the femoral head was identified in all involved femora by magnetic resonance imaging at forty-eight hours. Revascularization began at the periphery of the femoral head as early as one week and was underway in all by two weeks. At eight weeks, magnetic resonance imaging and histologic analysis showed deformation of the femoral head and variable tissue deposition. Tissue responses included (1) vascularized fibroblastic ingrowth with tissue resorption and cartilage, intramembranous bone, and mixed fibro-osseous or fibro-cartilaginous tissue synthesis and (2) resumption of endochondral bone growth. At eight weeks, the mean femoral head measurements (and standard error of the mean) for the control compared with the ligated femora were 10.4 ± 0.4 and 4.8 ± 0.4 mm, respectively, for height; 26.7 ± 0.8 and 31.2 ± 0.8 mm for diameter; 1.1 ± 0.1 and 2.3 ± 0.1 mm for cartilage thickness; and 151° ± 2° and 135° ± 2° for the femoral neck-shaft angle. Repeated-measures mixed-model analysis of variance revealed highly significant effects of ligation in each parameter (p < 0.0001).
Conclusions: Magnetic resonance imaging allows for the assessment of individual hips at sequential time periods to follow deformation and repair. There was a variable tissue response, and histologic assessment at the time of killing was shown to correlate with the evolving and varying magnetic resonance imaging signal intensities. Femoral head height on the ischemic side from one week onward was always less than the initial control value and continually decreased with time, indicating collapse as well as slowed growth. Increased femoral head width occurred relatively late (four to eight weeks), indicating cartilage model overgrowth concentrated at the periphery.
Clinical Relevance: Femoral head deformation and internal tissue deposition patterns can be followed accurately in vivo by magnetic resonance imaging.
Intracapsular surgical ligation at the base of the femoral neck and sectioning of the ligamentum teres is an excellent model for inducing ischemia and necrosis in the immature piglet femoral head1-4. While the development of femoral head deformity is a well-recognized finding with necrosis, the contributions of collapse, slowed growth, and asymmetric growth to deformation remain uncertain. After induction of ischemic necrosis with this piglet model, osteoclastic resorption of necrotic tissue has been recognized as contributing to the deformation of the femoral head1,2, and biomechanical indentation studies of the femoral head have demonstrated progressive structural weakness5. Magnetic resonance imaging with gadolinium enhancement demonstrates age-related vascular changes in the developing epiphysis and metaphysis of normal piglet femora6. Magnetic resonance imaging is also capable of demonstrating femoral head ischemia in piglets in vivo after immobilization of the hip in the widely abducted position7 and after surgical ligation at the base of the femoral neck3. In this study, we report the use of the femoral neck ligation-ligamentum teres sectioning model in three-week-old piglets with assessments by qualitative and quantitative sequential magnetic resonance imaging and end-stage histologic assessment in each to demonstrate the deformation and repair responses of the femoral heads to the vascular injury with time. This investigation of the piglet femoral head differs from previous studies, providing meaningful observations by (1) using magnetic resonance imaging to follow the development of deformity in vivo, assessing the same hips through various time stages; (2) quantifying the changing parameters of deformity by magnetic resonance imaging to help to clarify the mechanism of deformation; (3) using gadolinium-enhanced magnetic resonance imaging to assess the vascular status in necrosis and repair; (4) showing the variability of tissue responses by histologic analysis; and (5) correlating magnetic resonance imaging and histologic tissue differentiation patterns.
Materials and Methods
Operative Technique
The study was approved by the institution's Animal Care and Use Committee. Operations were performed on ten three-week-old piglets. Anesthesia was induced with use of an intramuscular injection of 40 mg/kg of midazolam hydrochloride (Baxter, Deerfield, Illinois) and 20 mg/kg of ketamine hydrochloride (Ketalar; Parke-Davis, Morris Plains, New Jersey). A second dose of anesthetic, 20 mg/kg of ketamine hydrochloride and 5 mg/kg of xylazine (Rompun; Mobay, Shawnee, Kansas), was given thirty minutes later. Continuous intravenous infusion of 1% Diprivan (propofol; AstraZeneca, Wilmington, Delaware) diluted in 5% dextrose in water at a dose of 0.002 mg/kg per minute was used for maintenance. An oral airway was placed, and blow-by oxygen was given at a rate of 4 L per minute. Sterile technique was used.
The right hip was approached with use of a lateral incision with the piglet lying on its left side. A linear 6-cm incision, parallel to the femur, was centered over the tip of the greater trochanter. An anterior capsulotomy and longitudinal traction on the lower extremity allowed for subluxation of the femoral head from the acetabulum. A long, curved scissors was used to cut the ligamentum teres. Ischemia was induced with use of a doubled, number-2 silk ligature placed circumferentially and tied tightly around the base of the femoral neck within the hip joint capsule (Fig. 1, A). The capsule, musculotendinous structures, subcutaneous tissues, and skin were closed with absorbable sutures. Skin dressings and postoperative splinting were not used. The operation was always performed on the right hip, and the left was the control.
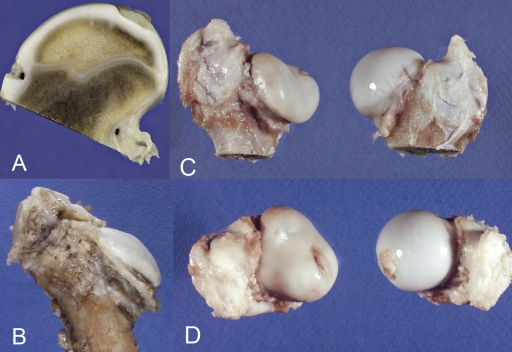
Fig. 1.
A: Midcoronal plane photograph of a decalcified femoral head and neck in a three-week and two-day-old piglet showing the location of the intracapsular circumferential suture. The piglet was killed forty-eight hours postoperatively after magnetic resonance imaging demonstrated complete femoral head ischemia. The intracapsular suture is the black dot on the medial and lateral surfaces of the neck and is placed at the lowest part of the neck adjacent to the capsular attachment. Note the large size of the secondary ossification center at this age. B: Photograph illustrating the gross appearance of the proximal end of a femur at eight weeks after surgery. The head and neck are shorter than normal, and the femoral head is markedly deformed. C: Photographs showing the operatively treated and nonoperatively treated proximal parts of femora from another piglet at eight weeks. The operatively treated femur (left) has a shorter neck, a relative increase of coxa vara, and a nonspherical head with flattening and obliquity of the superior surface. The medial third of the femoral head is least deformed and retains its sphericity best. The normal femoral head is at right. D: Looking down onto the superior surfaces of the same femoral heads, the misshapen head on the involved side is at the left, and the normal, noninvolved head is at the right. The involved head is asymmetric in shape along both the mediolateral and anteroposterior planes.
Magnetic Resonance Imaging
Technique
Magnetic resonance imaging was performed at 1.5 T on a GE Medical Systems scanner (Milwaukee, Wisconsin) with use of a pair of 3-in (7.6-cm) receive-only surface coils. Each study was performed with the piglet under general anesthesia with use of a single intramuscular injection of 20 mg/kg of ketamine hydrochloride and 5 mg/kg of xylazine followed by a continuous intravenous infusion of 1% Diprivan in 5% dextrose at a dose of 0.002 mg/kg per minute. The studies were performed with the piglets lying in the lateral decubitus position with the noninvolved hip down. In all animals, conventional magnetic resonance images, including T1, T2, and spoiled gradient recalled echo images, were acquired with 2.5-mm section thickness, 0.625-mm in-plane resolution, and 20-cm field of view. T1-weighted images were acquired with a repetition time of 500 msec, an echo time of 9 msec, and one signal acquired. T2-weighted images were acquired with a repetition time of 2000 msec, an echo time of 60 msec, and two signals acquired. Depending on the age and size of the piglet, usually six to nine images per femoral head were acquired in the coronal plane. Gadopentetate dimeglumine (Magnevist; Berlex, Wayne, New Jersey), an intravenous contrast agent, was also used at 0.2 mmol/kg injected manually in a rapid bolus into an ear vein ten seconds after beginning dynamic gadolinium-enhanced magnetic resonance imaging. Enhancement was evaluated by using a spoiled gradient-echo sequence (repetition time, 200 msec; echo time, 2 msec; flip angle, 60°; section thickness, 3 mm; in-plane resolution, 0.625 mm; and field of view, 20 cm). Five images were acquired per section. Spin-echo T1-weighted images (repetition time of 500 msec and echo time of 9 msec) after administration of contrast medium were also acquired at two-minute intervals to evaluate femoral head perfusion. Signal intensity changes on T1 and T2-weighted images were compared from the two femoral heads in each animal and recorded as increased, decreased, or unchanged from the control side.
Number of Assessments
Ten piglets underwent the study. Magnetic resonance imaging was performed after surgery, with the piglets under general anesthesia, at forty-eight hours and at one, two, four, and eight weeks. All piglets recovered from surgery without problem and, at forty-eight hours, all demonstrated completely ischemic and well-located femoral heads. One was killed at forty-eight hours after magnetic resonance imaging to document the position of the circumferential suture at the base of the neck (Fig. 1, A). Sequential magnetic resonance imaging studies were performed on the ten piglets, with images for quantification available from nine piglets since data from one were lost on computer system transfer. One piglet was killed at two weeks to document early changes. One had limping, discomfort, and an infection develop on the involved side and was killed at four weeks, with analyses used only from forty-eight hours and one and two-week time periods when clinical and magnetic resonance appearances were unremarkable. The remaining seven piglets were assessed until eight weeks after surgery.
Quantification
Proximal femoral measurements of the magnetic resonance images were made from the noninvolved, control side and from the ligated side at forty-eight hours and at one, two, four, and eight weeks. These included femoral head height, femoral head diameter, thickness of the articular and underlying epiphyseal cartilage, and femoral neck-shaft angle (as a measurement of proximal femoral varus angulation). Three measurements were done for each parameter per femoral head by two observers, and average values were used. Measurements were made with use of the Advantage workstation 4.2 (GE Medical Systems). The femoral head height was measured in millimeters on magnetic resonance images in the midcoronal plane from the midpoint of the superior articular surface of the femoral head to the top of the physis below the midpoint. The femoral head diameter was measured in millimeters along the midcoronal plane to determine the widest diameter from the articular surface of the medial part of the head to the head-physeal junction laterally. The cartilage thickness included the articular cartilage and the immediately adjacent epiphyseal cartilage and was measured in the midcoronal plane from the midpoint of the superior surface of the articular cartilage to the most peripheral part of the underlying bone or fibrovascular repair tissue of the secondary ossification center. The angle of the midlongitudinal axis of the head-neck region in relation to the longitudinal axis of the shaft was measured to serve as an index of proximal femoral varus.
Statistical Analysis
Statistical analysis was performed on the magnetic resonance imaging measurements8,9. Two-way mixed-model repeated-measures analysis of variance was used to compare height, diameter, angle, and cartilage growth measurements of deformity between ligated hips and contralateral, control hips at each of five time points (forty-eight hours and one, two, four, and eight weeks). This statistical approach efficiently accounts for the two hips per animal over the time course (i.e., correlated data) and handles the missing data problem unlike paired t tests or conventional repeated-measures analysis of variance models. A compound symmetry correlation structure was chosen to fit the model to account for a ligated and a contralateral hip from each of the same five piglets measured and provided good fit as judged by the Akaike information criterion10. Time, group (ligated compared with control), and time-by-group interaction were treated as fixed repeated-measures factors, and animal was treated as a random effect. At each time point, the F test was used to determine differences in height, diameter, angle, and cartilage between groups. An F value of ≥3.84 on one degree of freedom was used as the criterion for significance between groups. On the basis of pairwise comparisons, 95% confidence intervals were constructed for ligated and control hips at each time point. All reported p values are two-tailed.
Structural Studies
After the final magnetic resonance imaging, the piglet remained anesthetized and was killed with an intracardiac injection of Fatal-Plus (pentobarbital sodium; Vortech Pharmaceuticals, Dearborn, Michigan). The proximal aspects of both femora were removed at the subtrochanteric level, inspected, and photographed. For histologic assessment, the proximal femoral specimens were fixed in 10% neutral buffered formalin for two to four weeks followed by decalcification in 25% formic acid until soft. The trochanters were removed. The head-neck regions were cut first in the midcoronal plane followed by additional coronal plane cuts to allow for inspection and photographs from anterior to posterior regions of the head. Processing continued in two ways: (1) paraffin embedding, in which tissues were placed in increasing concentrations of alcohol, infiltrated and embedded in paraffin, cut at 7-μm thickness, and stained with 1% toluidine blue or hematoxylin and eosin, and (2) plastic embedding, in which tissues were infiltrated in JB-4 medium (Polysciences, Warrington, Pennsylvania) for two weeks, embedded in JB-4 plastic, cut at 1-μm thickness, and stained with 1% toluidine blue.
Source of Funding
This study was supported by National Institutes of Health grant 2 R01 AR042396-09.
Results
Femoral Head Deformation and Macroscopic Coronal Plane Hemisections
One piglet was killed at forty-eight hours to show the normal proximal femoral anatomy on coronal plane hemisection and the position of the circumferential intracapsular neck ligature (Fig. 1, A). In piglets killed at eight weeks, gross examination revealed deformation of the femoral head and neck in all involved femora, but a somewhat variable appearance compared with the normal, noninvolved side (Fig. 1, B, C, and D). A shortened head and neck with normal trochanteric growth led to coxa vara. The deformed head was oval to partially flat in shape and wider than normal. The articular surface was always intact but uneven with localized flattening and depression most prominent in the central region along the midcoronal plane. The shape of the medial portion of the femoral head was always better preserved than that of the central and lateral parts. Deformation was seen in both the coronal (mediolateral) and sagittal (anteroposterior) dimensions. Macroscopic examination of coronal plane hemisections after decalcification revealed increased thickness of the articular and underlying epiphyseal cartilage, increased epiphyseal cartilage vascularity, and variable tissue accumulations in the epiphysis, usually involving fibrocartilage interspersed with marrow and bone. Necrotic bone segments and relatively large accumulations of repair cartilage were seen on occasion. When multiple coronal plane sections from anterior to posterior regions of the same femoral head were examined, there was a marked difference in appearance from section to section.
Radiographic and Histologic Appearance of the Proximal Part of the Contralateral, Normal Femur
The proximal part of the femur in the piglets had a well-developed secondary ossification center at three weeks of age (Fig. 1, A) when the operation was done. Radiographic and histologic images of the normal, noninvolved femoral head at the time of killing at eight weeks postoperatively (eleven weeks of age) are shown in the Appendix.
Magnetic Resonance Imaging and Histologic Assessments of Operatively Treated Femoral Heads
Magnetic resonance images and histologic sections from the same femoral head are presented together to correlate the two methods of assessment. The magnetic resonance imaging of the noninvolved femoral head always showed a normal vascular signal (Fig. 2, A), and histologic analysis showed normal marrow and trabecular bone with osteocytes and surface osteoblasts (Fig. 2, B). Complete ischemia in the involved femoral heads was documented by lack of epiphyseal gadolinium enhancement in all ten piglets at forty-eight hours (Fig. 2, C). Histologic analysis showed that the marrow was avascular and fibrous with no surface osteoblasts on the bone trabeculae (Fig. 2, D).
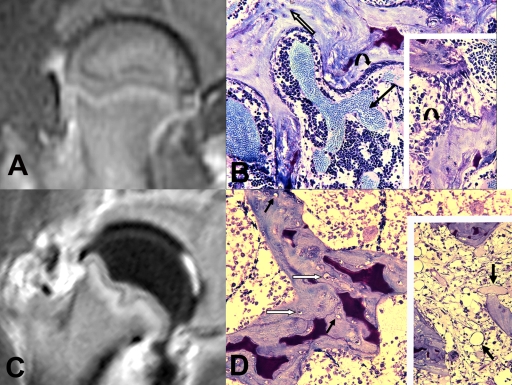
Fig. 2.
A: Magnetic resonance imaging of the normal, noninvolved femoral head at forty-eight hours showing normal signal intensity of the epiphysis and metaphysis on a T1-weighted sequence after gadolinium enhancement. B: Photomicrograph of a portion of the ossification center of the normal femoral head showing bone trabeculae with osteocytes in their lacunae (open arrow), surface osteoblasts (curved arrow) and osteoclasts, and well-vascularized marrow (solid straight arrow). The marrow also contains hematopoietic and mesenchymal cells. The red blood cells are within thin-walled sinusoidal vessels. In this tissue preparation, the red blood cells are circular and stained light blue. The dark purple accumulations are the remaining fragments of calcified cartilage from the endochondral sequence. The inset shows new bone trabeculae with osteocytes, surface osteoblasts (curved arrow), and mesenchymal cells differentiating to osteoblasts between the two trabeculae. An osteoclast is seen at the lower right adjacent to both a cartilage remnant and woven bone. This corresponds to the magnetic resonance imaging in A (paraffin-embedded section, 1% toluidine blue stain). C: Magnetic resonance imaging at forty-eight hours after surgery showing complete ischemia of the involved femoral head with a complete lack of epiphyseal gadolinium enhancement (T1-weighted sequence after gadolinium enhancement). The intra-articular region of high signal intensity is the cut ligamentum teres and surrounding synovial reaction. D: Photomicrograph illustrating a portion of the femoral head ossification center after induction of ischemia. The surfaces of the bone trabeculae are completely devoid of osteoblasts, and many of the lacunae are either empty (black arrows) or show contracted pyknotic osteocytes (white arrows). The marrow is acellular or hypocellular, and the sinusoidal vessels (inset; arrows) are empty of red blood cells. Revascularization has not yet occurred, so the bone trabeculae remain intact (paraffin-embedded section, 1% toluidine blue stain).
Assessments from One Piglet at Forty-eight Hours and at One, Four, and Eight Weeks
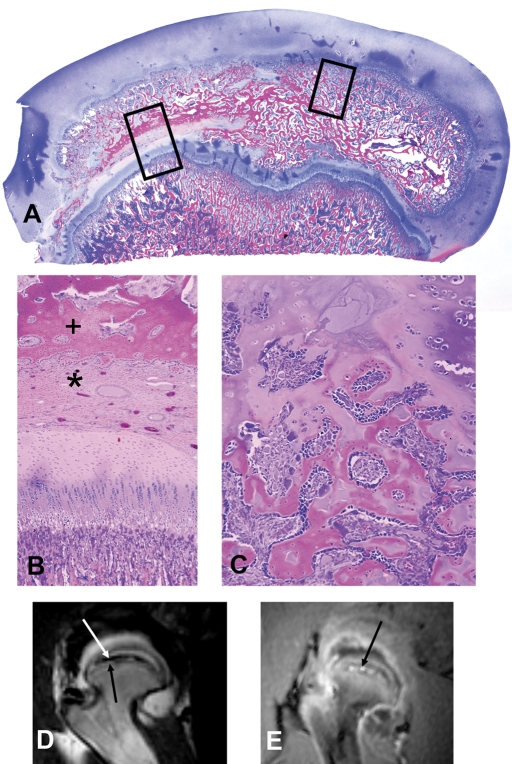
Magnetic resonance images (Fig. 3, A through F) and histologic images (Fig. 4, A through D) from one piglet illustrate the characteristic repair response. At forty-eight hours, the operatively treated femoral head showed lack of signal intensity with gadolinium (Fig. 3, A) while the noninvolved head showed normal vascular signal (Fig. 3, B). At one week, there was early revascularization as peripheral vessels reentered the epiphyseal cartilage and secondary ossification center bone medially and laterally (Fig. 3,C). At four weeks, revascularization of the secondary center was highlighted by high signal intensity (Fig. 3, D). By eight weeks, the signal intensity response was variable within the head and deformation was seen. Two different coronal sections illustrate the variable tissue responses within the same femoral head (Fig. 3, E and F). The fibrovascular invasion is of high signal intensity, dense repair bone is of low signal intensity, and fibrocartilage and endochondral bone are of intermediate signal intensity. Figure 4 illustrates the histologic findings that correspond with the magnetic resonance images at eight weeks at the time of killing.
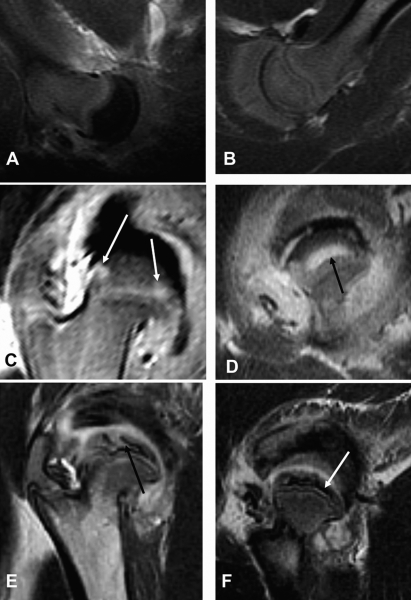
Fig. 3.
Magnetic resonance images from one piglet are shown at forty-eight hours (A, involved side and B, noninvolved side), one week (C), four weeks (D), and eight weeks (E and F). The femoral head underwent the common pattern of repair and deformation, which was seen in six of eight hips with long-term follow-up data. A: Magnetic resonance imaging of the involved femoral head at forty-eight hours showing absent signal intensity in the entire epiphysis with no enhancement with gadolinium, which is a sign of complete ischemia, while the normally perfused neck shows signal intensity indicative of persisting vascularization (T1-weighted sequence after gadolinium enhancement). B: Magnetic resonance imaging of the contralateral, noninvolved hip at forty-eight hours showing normal signal intensity indicating good vascularization of the femoral head and neck (T1-weighted sequence after gadolinium enhancement). C: Magnetic resonance imaging at one week after surgery showing high signal intensity at the point of entrance of the medial and lateral epiphyseal vessels into the femoral head (arrows) to initiate the repair response (T1-weighted sequence after gadolinium enhancement). D: Magnetic resonance imaging at four weeks after surgery showing the area of high signal intensity (arrow) in the central part of the femoral head, indicative of fibrovascular and/or vascularized cartilage repair tissue (T1-weighted sequence after gadolinium enhancement). E and F: Two T2-weighted magnetic resonance images in the coronal plane illustrating different depths of the same femoral head at eight weeks after surgery. The altered and deformed shape of the femoral head is evident. The tissue accumulations of differing signal intensities in both images are indicative of the spatially variable tissue repair. In E, the high signal intensity (arrow) entering the lateral portion of the head is the fibrovascular repair response. The low signal intensity surrounding the fibrovascular tissue represents dense repair bone, and the tissue above with intermediate signal intensity is cartilage undergoing endochondral ossification. In F, the femoral head has a greater proportion of tissue with low signal uptake, which represents accumulations of dense repair bone (arrow).
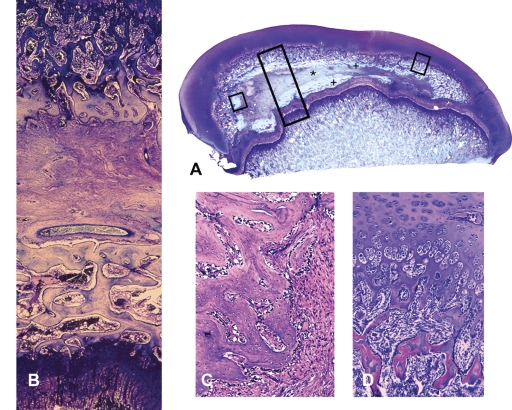
Fig. 4.
Histologic images corresponding to magnetic resonance images at eight weeks from the same femoral head illustrated in Figure 3, E and F, are shown. The figures are oriented in the same coronal plane as the magnetic resonance imaging scans. A: Photomicrograph of a coronal plane histologic section of the entire femoral head and physis (epiphysis) and the adjacent neck (metaphysis) are shown. The medial part of the head is at the right. The articular and epiphyseal cartilage thickness is increased from the normal (compare with Fig. E-1, B). The fibrovascular invasion (*) from a lateral point of entrance has passed in an arc-like fashion across the femoral head into the medial segment. Dense intramembranous bone formation (+) is seen at the upper and lower margins of the fibrovascular tissue. Endochondral bone formation resumes at the undersurface of the continuous articular-epiphyseal cartilage. This orientation corresponds to the magnetic resonance images in Figure 3, E and F, with the fibrovascular tissue of high signal intensity, the dense intramembranous bone of low signal intensity, and the endochondral tissue of intermediate signal intensity. Tissue within the long rectangular box is seen at higher magnification in B, from the smaller box at left in C, and from the box at the upper right in D (paraffin-embedded tissue, 1% toluidine blue stain). B: The prominent band of fibrovascular tissue is seen in the middle portion of the histologic section along with a large transversely oriented vessel. Below this accumulation, tissue differentiation by means of the intramembranous mechanism has formed woven bone on which lamellar bone is being synthesized. Endochondral bone has not formed at this site. Above the fibrovascular tissue, there is also new intramembranous bone synthesis and, above that, the endochondral bone sequence from the epiphyseal cartilage has reestablished itself. The intact physis is seen at bottom (paraffin-embedded tissue, 1% toluidine blue stain). C: Higher-magnification photomicrograph shows intramembranous woven bone formation, at left, from osteoblasts, which have differentiated from the fibrovascular tissue invasion. Osteoblasts rim the surface of the woven bone (paraffin-embedded section, 1% toluidine blue stain). D: Higher-magnification photomicrograph of tissue response at the upper right region of the medial part of the femoral head showing continuation and/or resumption of endochondral bone formation (paraffin-embedded section, 1% toluidine blue stain).
Imaging Sequences Highlighting Variability of Repair
Revascularization, indicated by increased signal intensity on T1-weighted images after gadolinium enhancement, was seen at the periphery of the femoral head as early as one week in some piglets (Fig. 3, C) and in all by two weeks. In a femoral head with a characteristic pattern of repair and deformation at eight weeks, magnetic resonance imaging sequences showed differing intensities (Fig. 5). Sites of fibrovascular invasion with T2-weighted sequences had intermediate signal intensity, and sites of dense repair bone with woven and lamellar bone had low signal intensity (Fig. 5, D). Focal vascularized endochondral repair and fibrocartilage repair regions had high signal intensity on T1-weighted sequences after gadolinium enhancement (Fig. 5, E). The corresponding histologic variability is illustrated in Figure 5.
Fig. 5.
Histologic and magnetic resonance images from a different piglet than that shown in Figures 3 and 4 illustrate other aspects of the common response to ischemia at eight weeks. This response was predominant in six of eight hips followed to eight weeks. A: Photomicrograph of another entire femoral head is shown. Fibrous tissue ingrowth is seen at the lower part of the secondary ossification center laterally above the physis. The cartilage surface is much thicker than normal. Bone in the medial third of the femoral head is relatively normal-appearing endochondral bone, but most bone centrally and in the lateral third is intramembranous bone from the new tissue ingrowth. Tissue from the box at left is shown at higher magnification in B, and tissue from the box at right is shown at higher magnification in C (paraffin-embedded section, hematoxylin and eosin stain). B: The characteristic fibrovascular invasion tissue (*) is seen centrally with large and small blood vessels prominent. Below, cartilage tissue persists, while, above (+), intramembranous bone synthesis from the fibrovascular component has occurred (paraffin-embedded section, hematoxylin and eosin stain). C: Tissue at the upper right showing endochondral bone formation emanating not from a physeal structure but as differentiation with vascularization of preceding cartilage repair tissue. Osteoclasts are prominent in the remodeling repair tissue (paraffin-embedded section, hematoxylin and eosin stain). D: Magnetic resonance imaging along the same plane and orientation as A, highlights the low-signal-intensity tissue (upper arrow), which is dense woven and lamellar intramembranous bone. Immediately below (inferior), the fibrovascular tissue has intermediate signal intensity (lower arrow). Note also the oval shape of the femoral head and its diminished height (T2-weighted sequence). E: Magnetic resonance image at a different depth but still in the coronal plane showing high-signal-intensity (arrow) accumulations, which represent fibrovascular tissue in greater accumulation at this site (T1-weighted sequence after gadolinium enhancement).
The response to ischemia in two femoral heads was considerably different from the common pattern (see Appendix). In one, the central and lateral parts of the head contained a large necrotic bone segment with no vascular invasion at eight weeks. Histologic analysis revealed a large central and lateral area of necrotic secondary ossification center bone, without evidence of vascularization or cellular repair, separated from the rest of the epiphyseal cartilage secondary ossification center bone by a rim of fibrous tissue. This was evident on the coronal plane hemisection of decalcified femoral head from which the histologic section was subsequently made. Magnetic resonance imaging showed delayed central and superior femoral head revascularization at two weeks and a clear contrast between persisting normal medial bone with signal and the adjacent large necrotic fragment with low signal intensity on gadolinium-enhanced T1-weighted images at eight weeks. In the other femoral head, cartilage repair tissue predominated at central and lateral regions. At higher magnification, the tissue was vascularized fibrocartilage. On magnetic resonance imaging, there were wide areas of high signal intensity (gadolinium enhancement) and intermediate signal intensity representing the cartilaginous repair, which correlated well with the histologic findings.
At the lateral peripheral part of the head, histologic sections of the surface cartilage often showed vascularized fibrous tissue overgrowth (pannus) and areas of chondrocyte cloning, which varied from a few clustered cells to highly structured circular accumulations with the cells at the periphery (see Appendix). This was not detected by magnetic resonance imaging. Increased vessels within the lateral epiphyseal cartilage were frequently seen, many of which eventually became associated with ectopic foci of endochondral ossification. These findings were highlighted by gadolinium-enhanced magnetic resonance imaging. The physes showed variable thickness and an undulating pathway, but transphyseal bone bridges were seen infrequently. When present, they were usually focal. Magnetic resonance imaging demonstrated marrow signal continuity across the physis between epiphyseal and metaphyseal regions, and histologic sections revealed repopulation of the necrotic epiphysis with vascularized marrow and bone trabeculae.
Quantitative Magnetic Resonance Imaging Assessments
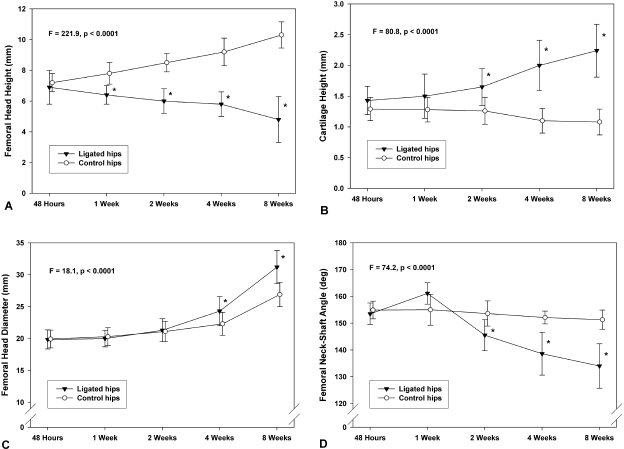
Magnetic resonance imaging measurements of the proximal part of the femur on the ligated and control sides were done on eight piglets to determine femoral head height (Fig. 6, A), cartilage height (Fig. 6, B), femoral head diameter (Fig. 6, C), and femoral neck-shaft angle (Fig. 6, D) during the eight-week time course. At eight weeks, the mean femoral head measurements (and standard error of the mean) for the control compared with the ligated femora were 10.4 ± 0.4 and 4.8 ± 0.4 mm, respectively, for height; 26.7 ± 0.8 and 31.2 ± 0.8 mm for diameter; 1.1 ± 0.1 and 2.3 ± 0.1 mm for cartilage thickness; and 151° ± 2° and 135° ± 2° for the femoral neck-shaft angle. Repeated-measures mixed-model analysis of variance revealed highly significant effects of ligation in each parameter (p < 0.0001).
Fig. 6.
Figures illustrating magnetic resonance imaging measurements in the involved (ligated) and noninvolved (control) sides analyzed by repeated-measures analysis of variance, with significant group differences denoted by asterisks. The error bars denote 95% confidence intervals. A: Femoral head height. At four weeks, the mean femoral head height was 9.3 mm for the control side and 5.8 mm for the ligated side (mean difference, 3.5 mm; 95% confidence interval for difference, 2.5 to 4.3 mm). At eight weeks, the mean femoral head height was 10.4 mm for the control side and 4.8 mm for the involved side (mean difference, 5.6 mm; 95% confidence interval for difference, 4.7 to 6.5 mm). Therefore, the effect of ligation was an average 3.5-mm reduction at four weeks and a 5.6-mm reduction at eight weeks of follow-up (p < 0.0001). B: Cartilage height, measuring articular cartilage and underlying epiphyseal cartilage. At four weeks, the mean cartilage height was 1.1 mm for the control side and 2.0 mm for the ligated side (mean difference, 0.9 mm; 95% confidence interval for difference, 0.6 to 1.2 mm). At eight weeks, the mean cartilage height was 1.1 mm for the control side and 2.3 mm for the involved side (mean difference, 1.2 mm; 95% confidence interval for difference, 0.8 to 1.5 mm). Thus, the average effect of ligation on cartilage height was an increase of 0.9 mm at four weeks and 1.2 mm at eight weeks of follow-up (p < 0.0001). C: Femoral head diameter. At eight weeks, the mean femoral head diameter was 26.7 mm for the control side and 31.2 mm for the involved side (mean difference, 4.5 mm; 95% confidence interval for difference, 3.2 to 5.6 mm). The effect of ligation was an average increase of 4.5 mm in femoral head diameter (p < 0.0001). D: Femoral neck-shaft angle. At eight weeks, the mean femoral neck-shaft angle was 151° for the control side and 135° for the involved side (mean difference, 16°; 95% confidence interval for difference, 11° to 21°). Therefore, the effect of ligation was a mean reduction of 16° in the femoral neck-shaft angle at eight weeks of follow-up (p < 0.0001).
Repeated-measures analysis of variance revealed a highly significant effect of ligation on femoral head height (F = 221.9, p < 0.0001) (Fig. 6, A). The involved femoral heads had progressively less height than the noninvolved, control sides with significant differences in height between ligated and control hips at one week (F = 16.4, p < 0.0001), two weeks (F = 46.1, p < 0.0001), four weeks (F = 59.4, p < 0.0001), and eight weeks (F = 158.4, p < 0.0001). No group difference was detected at forty-eight hours (p = 0.36). Highly significant differences were found between the ligated and control hips regarding the rate of change from baseline to eight weeks (F = 23.7, p < 0.0001).
Repeated-measures analysis of variance indicated a highly significant overall effect of ligation on femoral head cartilage height (F = 80.8, p < 0.0001) (Fig. 6, B). Greater cartilage height was observed in the ligated group compared with contralateral, control femoral heads at two weeks (F = 9.6, p = 0.003), four weeks (F = 33.4, p < 0.0001), and eight weeks (F = 55.7, p < 0.0001). The rate of change over the eight-week time period was significantly faster for ligated compared with control femoral heads (F = 9.5, p < 0.001).
Analysis of variance indicated a significant effect of ligation on femoral head diameter measurements compared with control hips (F = 18.1, p < 0.0001) (Fig. 6, C). The involved femoral heads became wider than the controls. Significant group differences were found at four weeks (F = 4.2, p = 0.045) and eight weeks (F = 49.1, p < 0.0001). No group difference was found at forty-eight hours (p = 0.91), one week (p = 0.51), or two weeks (p = 0.66). Significant differences were detected in the rate of change from baseline to eight weeks between the two groups (F = 10.4, p < 0.001).
Repeated-measures analysis of variance indicated a highly significant effect of ligation on the femoral neck-shaft angle measurements (F = 74.2, p < 0.0001) (Fig. 6, D). Significantly smaller angles (indicative of a coxa vara deformity) were demonstrated in ligated femora than control sides at two weeks (F = 17.5, p < 0.001), four weeks (F = 25.4, p < 0.0001), and eight weeks (F = 44.0, p < 0.0001). Although no difference was observed at forty-eight hours (p = 0.48), marginally significant differences were detected at one week (F = 4.0, p = 0.05). The slope test revealed significant differences in the rate of change over the eight weeks between the ligated and control sides (F = 7.2, p < 0.001).
Discussion
The Model
In a previous work, placement of intracapsular circumferential silk ligatures around the base of the femoral neck in young piglets caused whole femoral head ischemia as defined by magnetic resonance imaging with gadolinium enhancement at six and ninety-six hours3. The longer-term study described in the present report added sectioning of the ligamentum teres to induce femoral head ischemia and necrosis, a technique used successfully in other studies1,2,4. The blood supply of the proximal part of the femur in the piglet appears to be the same as in the human11. Repair in the piglet model is characterized by fibrovascular invasion1,2, resorption of necrotic tissue, and variable tissue synthesis, whereas adult femoral head osteonecrosis is characterized by new bone formation on persisting necrotic trabeculae12. Other animal models13-15 do not produce the extensive femoral head deformity seen in the piglet.
Contribution of Quantitative Magnetic Resonance Imaging to an Understanding of Femoral Head Deformation
At the moment when the femoral head becomes ischemic, there is no deformity since structure is maintained by the intact cartilage and osseous trabeculae. Deformity occurs in association with the repair response to ischemia and necrosis, which is initiated by revascularization of the epiphyseal cartilage and secondary ossification center bone and allows fibrovascular tissue to provide undifferentiated mesenchymal cells for repair and osteoclasts to resorb both necrotic and early repair tissue.
Quantitative magnetic resonance studies help to define the mechanism of femoral head deformation in piglet ischemic necrosis by measuring changes in shape in vivo in the same hip over a relatively extended period of time. The studies show continuing growth in the height of the normal, control femoral head from a mean of 7.2 mm at the initial assessment forty-eight hours postoperatively to 10.4 mm at eight weeks, while the ligated side not only failed to grow but actually decreased in height at each time period, from a mean of 6.9 mm at one week to 4.8 mm at eight weeks (Fig. 6, A). The mean final height is considerably less than the control height at the start of the experiment, indicating actual collapse of the head in the midcoronal plane—not just an absence of growth or a slowed rate of continuing growth. Absent or slowed growth alone cannot account for a decreasing height beyond the starting level since these occurrences alone would leave the height either the same or only slightly increased from its value at the time of vascular insult.
The diameter of the control femoral head increases with time, but the diameter on the ligated side increases at an even greater rate and is wider than the normal side at eight weeks. This finding is consistent with the magnetic resonance and histologic findings of early and prominent revascularization at the femoral head periphery. There is no significant change in diameter over the first two weeks on either side, with an increased diameter on the ligated side starting after two weeks and the major and significant width change occurring between four and eight weeks after surgery (Fig. 6, C). Since the femoral head height is less than normal as early as one and two weeks, and actually less than it was at the time of induction of ischemia, while the width at one and two weeks is the same as the width of the noninvolved side, there is clearly no reciprocal squeezing or balloon effect change in the shape of the head, whereby collapse at one site immediately induces width change at another. We interpret the considerable width increase starting at four weeks on the ischemic side to indicate that width change is, to a great extent, due to asymmetric growth rather than to shape change with collapse. If collapse alone caused deformation of the femoral head in terms of widening it in one dimension and shortening it in another, the two events would occur simultaneously.
Measurement of the other parameters also provides information regarding femoral head deformation. On the control side, the superior surface cartilage thickness stays almost the same throughout the period of study with only a slow decrease from a mean of 1.3 mm to a mean of 1.1 mm at eight weeks. Outward growth of the articular cartilage is in balance with cartilage to bone conversion at the hemispheric physeal periphery of the secondary ossification center16. The surface articular-epiphyseal cartilage thickness, however, increases significantly on the ligated side from 1.4 mm to 2.3 mm at eight weeks (Fig. 6, B), progressively increasing its size differential with time. It increases slowly compared with the control side in the first two weeks after surgery but then increases at an even greater rate from four to eight weeks after surgery. Synovial fluid diffusing from the joint mediates continuing cartilage growth14,17, while bone ischemia internally prevents transformation of epiphyseal cartilage to secondary ossification center bone.
The femoral neck-shaft angle shows a slight decrease with time from a mean of 155° to 151° on the control side, while collapse of the head and diminished growth on the ligated side decrease the angle into more varus from a mean of 154° to 135° at eight weeks (Fig. 6, D). Greater trochanteric growth continues normally bilaterally since its blood supply remains intact.
Contribution of Magnetic Resonance Imaging and Histologic Analysis to an Understanding of Repair and Deformation After Ischemia
In this study, ischemia, necrosis, repair, and femoral head deformities were effectively induced in all cases, but there is a considerable range of histologic responses. The variability occurs from animal to animal and also in different parts of the same femoral head. Magnetic resonance imaging and correlative histologic findings in previous studies have demonstrated differing signal characteristics in the various regions of the developing ends of the bone6,18-20. For all imaging sequences, low signal intensity (black image) is similar to dense cortical bone, intermediate signal intensity is similar to the image of muscle (non-fat-suppressed), and high signal intensity (light image) is similar to fluid (on a fluid-sensitive sequence such as T2). Gadolinium enhancement with T1-weighted imaging is particularly valuable in demonstrating normal vascularity (high signal intensity)6 and ischemia of the proximal femoral epiphyses (low signal intensity)3,4,7. With the application of different sequences, specific tissues can be highlighted, although not all tissue groups are demonstrated well on each sequence. Absolute correlation of histologic specificity with magnetic resonance imaging is not possible at all times. Magnetic resonance image thickness is in the range of 2.5 mm, while histologic section thickness is generally 7 μm, such that the magnetic resonance tissue slice is markedly thicker. Tissue variability within a narrow range can lead to decreased magnetic resonance image resolution. Despite these considerations, differing signal intensities with use of specific sequences can be correlated, often with high accuracy, with the actual tissue as confirmed by histologic analysis.
The most common repair pattern, induced by the fibrovascular ingrowth and synovial fluid diffusion from the joint surface, is intramembranous bone synthesis above the physis in the lateral and central parts of the head, resumption of endochondral bone formation in the epiphyseal cartilage closer to the articular surface centrally and medially (Figs. 4 and 5), and increased surface cartilage thickness. In some instances, large necrotic bone segments persist, walled off from the surrounding secondary ossification center by thick fibrous tissue, while in others the response can be predominantly cartilaginous or fibrocartilaginous with bone formation delayed (see Appendix). Magnetic resonance imaging documents in vivo the initial ischemia, the pattern of revascularization, the onset and progression of femoral head deformity, and the variable tissue responses to repair.
In the ischemic femoral heads at forty-eight hours, the entire head is avascular (Fig. 2, C) with lack of signal on gadolinium-enhanced T1-weighted sequences. At one week, revascularization is noted medially and laterally from the periphery (Fig. 3, C; high signal intensity) and, at two weeks, the head is substantially revascularized with the central and superior regions slowest to revascularize. Large necrotic segments continue with low signal intensity until revascularization occurs. The fibrovascular response can be illustrated on gadolinium-enhanced T1-weighted images with high signal intensity. The synthesis of woven and lamellar bone is demonstrated by intraepiphyseal regions of low signal intensity both on gadolinium-enhanced T1-weighted images and on T2-weighted images. Endochondral tissue is generally of intermediate intensity. It is not possible to differentiate fibrovascular tissue from well-vascularized fibrocartilage since both are of high signal intensity due to the fluid vascularity. Babyn et al. used magnetic resonance imaging in piglets with femoral head ischemia to assess the resected femoral heads after the piglets were killed, with some high-resolution imaging done on narrow core biopsies21. Our study documents the response to ischemic insult in vivo. For the future, imaging with a high-field-strength 3-T magnet is capable of producing images of higher spatial resolution and thinner sections without sacrificing signal-to-noise ratio.
Similarities of the Piglet Model to Childhood Legg-Calvé-Perthes Disease
The piglet model leads to femoral head deformity similar to Legg-Calvé-Perthes disease1,2,4,5,22-24, with histologic changes characteristic of human cases25-33. Studies from humans with the disease have recognized the changes to be intermingling of necrosis and repair25,27, new bone formation by intramembranous ossification30, increased thickness of the surface cartilage31, retained shape and internal structure of the most medial portion of the head, and (in the central and lateral aspects of the head) a callus-like fibrocartilage, vascular granulation tissue, new bone formation on necrotic trabeculae, and chondroid tissue32,33. Magnetic resonance imaging has been used over the past two decades to assess Legg-Calvé-Perthes disease, but the studies lack histologic correlation34-37. The tissue responses in human specimens and in this model are similar to bone repair induced by fracture in an environment of variable vascularity and micromotion38. The model differs in two important ways from Legg-Calvé-Perthes disease: (1) it induces complete ischemia rapidly by a mechanism different from that in humans, in whom periosseous vascular constriction has not been implicated, and (2) it involves a single event to induce ischemia, while there is evidence that at least some humans experience two or more episodes of ischemia on the basis of the histologic finding of necrosis of repair tissue39,40. The model has been helpful in demonstrating that femoral head deformation can be minimized by preventing osteoclastic bone resorption with use of bisphosphonates systemically22 or by intraosseous injection23 or by inducing osteoclast inhibition by other means24. This study demonstrates the ability of magnetic resonance imaging to assess the model in vivo and can be applied to gain further information about the development and prevention of deformity following ischemic insults.
Supplementary Material
Acknowledgments
Note: The authors thank Jim Koepfler for his invaluable assistance with all figures.
Appendix
The figures showing normal proximal femoral histologic findings, two other femoral heads with magnetic resonance imaging and corresponding histologic assessment, and specific histologic findings are available with the electronic versions of this article, on our web site at jbjs.org (go to the article citation and click on “Supplementary Material”) and on our quarterly CD/DVD (call our subscription department, at 781-449-9780, to order the CD or DVD). 
Disclosure: In support of their research for or preparation of this work, one or more of the authors received, in any one year, outside funding or grants in excess of $10,000 from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health. Neither they nor a member of their immediate families received payments or other benefits or a commitment or agreement to provide such benefits from a commercial entity.
Investigation performed at Orthopaedic Research Laboratory and Department of Radiology, Children's Hospital Boston, and Department of Radiology, Massachusetts General Hospital, Boston, Massachusetts
References
- 1.Kim HK, Su PH, Qiu YS. Histopathologic changes in growth-plate cartilage following ischemic necrosis of the capital femoral epiphysis. An experimental investigation in immature pigs. J Bone Joint Surg Am. 2001;83:688-97. [DOI] [PubMed] [Google Scholar]
- 2.Kim HK, Su PH. Development of flattening and apparent fragmentation following ischemic necrosis of the capital femoral epiphysis in a piglet model. J Bone Joint Surg Am. 2002;84:1329-34. [DOI] [PubMed] [Google Scholar]
- 3.Jaramillo D, Connolly SA, Vajapeyam S, Robertson RL, Dunning PS, Mulkern RV, Hayward A, Maier SE, Shapiro F. Normal and ischemic epiphysis of the femur: diffusion MR imaging study in piglets. Radiology. 2003;227:825-32. [DOI] [PubMed] [Google Scholar]
- 4.Menezes NM, Connolly SA, Shapiro F, Olear EA, Jimenez RM, Zurakowski D, Jaramillo D. Early ischemia in growing piglet skeleton: MR diffusion and perfusion imaging. Radiology. 2007;242:129-36. [DOI] [PubMed] [Google Scholar]
- 5.Pringle D, Koob TJ, Kim HK. Indentation properties of growing femoral head following ischemic necrosis. J Orthop Res. 2004;22:122-30. [DOI] [PubMed] [Google Scholar]
- 6.Jaramillo D, Villegas-Medina OL, Doty DK, Rivas R, Strife K, Dwek JR, Mulkern RV, Shapiro F. Age-related vascular changes in the epiphysis, physis, and metaphysis: normal findings on gadolinium-enhanced MRI of piglets. AJR Am J Roentgenol. 2004;182:353-60. [DOI] [PubMed] [Google Scholar]
- 7.Jaramillo D, Villegas-Medina OL, Doty DK, Dwek JR, Ransil BJ, Mulkern RV, Shapiro F. Gadolinium-enhanced MR imaging demonstrates abduction-caused hip ischemia and its reversal in piglets. AJR Am J Roentgenol. 1996;166:879-87. [DOI] [PubMed] [Google Scholar]
- 8.Laird NM, Ware JH. Random-effects models for longitudinal data. Biometrics. 1982;38:963-74. [PubMed] [Google Scholar]
- 9.Vittinghoff E, Glidden DV, Shiboski SC, McCulloch CE. Regression methods in biostatistics: linear, logistic, survival, and repeated measures models. New York: Springer; 2005. p 253-89.
- 10.Akaike H. Likelihood of a model and information criteria. J Econometrics. 1981;16:3-14. [Google Scholar]
- 11.Trueta J. The normal vascular anatomy of the human femoral head during growth. J Bone Joint Surg Br. 1957;39:358-94. [DOI] [PubMed] [Google Scholar]
- 12.Glimcher MJ, Kenzora JE. The biology of osteonecrosis of the human femoral head and its clinical implications. III. Discussion of the etiology and genesis of the pathological sequelae; comments on treatment. Clin Orthop Relat Res. 1979;140:273-312. [PubMed] [Google Scholar]
- 13.Kenzora JE, Steele RE, Zosipovitch ZH, Glimcher MJ. Experimental osteonecrosis of the femoral head in adult rabbits. Clin Orthop Relat Res. 1978;130:8-46. [PubMed] [Google Scholar]
- 14.Zahir R, Freeman MAR. Cartilage changes following a single episode of infarction of the capital femoral epiphysis in the dog. J Bone Joint Surg Am. 1972;54:125-36. [PubMed] [Google Scholar]
- 15.Sanchis M, Zahir A, Freeman MAR. The experimental simulation of Perthes disease by consecutive interruptions of the blood supply to the capital femoral epiphysis in the puppy. J Bone Joint Surg Am. 1973;55:335-42. [PubMed] [Google Scholar]
- 16.Rivas R, Shapiro F. Structural stages in the development of the long bones and epiphyses: a study in the New Zealand white rabbit. J Bone Joint Surg Am. 2002;84:85-100. [DOI] [PubMed] [Google Scholar]
- 17.McKibbin B, Holdsworth FW. The nutrition of immature cartilage in the lamb. J Bone Joint Surg Br. 1966;48:793-803. [PubMed] [Google Scholar]
- 18.Jaramillo D, Connolly SA, Mulkern RV, Shapiro F. Developing epiphysis: MR imaging characteristics and histologic correlation in the newborn lamb. Radiology. 1998;207:637-45. [DOI] [PubMed] [Google Scholar]
- 19.Connolly SA, Jaramillo D, Hong JK, Shapiro F. Skeletal development in fetal pig specimens: MR imaging of femur with histologic comparison. Radiology. 2004;233:505-14. [DOI] [PubMed] [Google Scholar]
- 20.Jaramillo D, Shapiro F. Growth cartilage: normal appearance, variants and abnormalities. Magn Reson Imaging Clin N Am. 1998;6:455-71. [PubMed] [Google Scholar]
- 21.Babyn PS, Kim HK, Gahunia HK, Lemaire C, Salter RB, Fornasier V, Pritzker KP. MRI of the cartilaginous epiphysis of the femoral head in the piglet hip after ischemic damage. J Magn Reson Imaging. 1998;8:717-23. [DOI] [PubMed] [Google Scholar]
- 22.Kim HK, Randall TS, Bian H, Jenkins J, Garces A, Bauss F. Ibandronate for prevention of femoral head deformity after ischemic necrosis of the capital femoral epiphysis in immature pigs. J Bone Joint Surg Am. 2005;87:550-7. [DOI] [PubMed] [Google Scholar]
- 23.Aya-ay J, Athavale S, Morgan-Bagley S, Bian H, Bauss F, Kim HK. Retention, distribution, and effects of intraosseously administered ibandronate in the infarcted femoral head. J Bone Miner Res. 2007;22:93-100. Erratum in: J Bone Miner Res. 2007;22:642. [DOI] [PubMed] [Google Scholar]
- 24.Kim HK, Morgan-Bagley S, Kostenuik P. RANKL inhibition: a novel strategy to decrease femoral head deformity after ischemic osteonecrosis. J Bone Miner Res. 2006;21:1946-54. [DOI] [PubMed] [Google Scholar]
- 25.Zemansky AP Jr. The pathology and pathogenesis of Legg-Calve-Perthes' disease (osteochondritis juvenilis deformans coxae). Am J Surg. 1928;4:169-84. [Google Scholar]
- 26.Lippmann RK. The pathogenesis of Legg-Calve-Perthes' disease based upon the pathologic findings in a case. Am J Surg. 1929;6:785-91. [Google Scholar]
- 27.Nagasaka K. Histological study of 8 cases of osteochondritis deformans coxae juvenilis (Calve-Legg- Perthes). Fukuoka Acta Med. 1930;23:26. [Google Scholar]
- 28.Jonsater S. Coxa plana: a histo-pathologic and arthrografic study. Acta Orthop Scand Suppl. 1953;12:5-98. [PubMed] [Google Scholar]
- 29.Mizuno S, Hirayama M, Kotani PT, Simazu A. Pathological histology of the Legg-Calve-Perthes disease with a special reference to its experimental production. Med J Osaka Univ. 1966;17:177-209. [PubMed] [Google Scholar]
- 30.Dolman CL, Bell HM. The pathology of Legg-Calve-Perthes disease. A case report. J Bone Joint Surg Am. 1973;55:184-8. [PubMed] [Google Scholar]
- 31.McKibbin B, Ralis Z. Pathological changes in a case of Perthes' disease. J Bone Joint Surg Br. 1974;56:438-47. [PubMed] [Google Scholar]
- 32.Catterall A, Pringle J, Byers PD, Fulford GE, Kemp HB. Perthes' disease: is the epiphyseal infarction complete? J Bone Joint Surg Br. 1982;64:276-81. [DOI] [PubMed] [Google Scholar]
- 33.Catterall A, Pringle J, Byers PD, Fulford GE, Kemp HB, Dolman CL, Bell HM, McKibbin B, Rális Z, Jensen OM, Lauritzen J, Ponseti IV, Ogden J. A review of the morphology of Perthes' disease. J Bone Joint Surg Br. 1982;64:269-75. [DOI] [PubMed] [Google Scholar]
- 34.Ducou le Pointe H, Haddad S, Silberman B, Filipe G, Monroc M, Montagne JP. Legg-Perthes-Calve disease: staging by MRI using gadolinium. Pediatr Radiol. 1994;24:88-91. [DOI] [PubMed] [Google Scholar]
- 35.Jaramillo D, Kasser JR, Villegas-Medina OL, Gaary E, Zurakowski D. Cartilaginous abnormalities and growth disturbances in Legg-Calvé-Perthes disease: evaluation with MR imaging. Radiology. 1995;197:767-73. [DOI] [PubMed] [Google Scholar]
- 36.de Sanctis N, Rega AN, Rondinella F. Prognostic evaluation of Legg-Calve-Perthes disease by MRI. Part I: the role of physeal involvement. J Pediatr Orthop. 2000;20:455-62. [PubMed] [Google Scholar]
- 37.de Sanctis N, Rondinella F. Prognostic evaluation of Legg-Calve-Perthes disease by MRI. Part II: pathomorphogenesis and new classification. J Pediatr Orthop. 2000;20:463-70. [PubMed] [Google Scholar]
- 38.Shapiro F. Bone development and its relation to fracture repair. The role of mesenchymal osteoblasts and surface osteoblasts. Eur Cell Mater. 2008;15:53-76. [DOI] [PubMed] [Google Scholar]
- 39.Inoue A, Freeman MA, Vernon-Roberts B, Mizuno S. The pathogenesis of Perthes' disease. J Bone Joint Surg Br. 1976;58:453-61. [DOI] [PubMed] [Google Scholar]
- 40.Inoue A, Ono K, Takaoka K, Yoshioka T, Hosoya T. A comparative study of histology in Perthes' disease and idiopathic femoral avascular necrosis of the femoral head in adults (IANF). Int Orthop. 1980;4:39-46. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.