Abstract
In mammals, puberty onset typically occurs earlier in females than in males, but the explanation for sexual differentiation in the tempo of pubertal development is unknown. Puberty in both sexes is a brain-dependent phenomenon and involves alterations in the sensitivity of neuronal circuits to gonadal steroid feedback as well as gonadal hormone-independent changes in neuronal circuitry. Kisspeptin, encoded by the Kiss1 gene, plays an essential but ill-defined role in pubertal maturation. Neurokinin B (NKB) is coexpressed with Kiss1 in the arcuate nucleus (ARC) and is also important for puberty. We tested whether sex differences in the timing of pubertal development are attributable to sexual differentiation of gonadal hormone-independent mechanisms regulating hypothalamic Kiss1/NKB gene expression. We found that, in juvenile females, gonadotropin secretion and expression of Kiss1 and NKB in the ARC increased immediately following ovariectomy, suggesting that prepubertal females have negligible gonadal hormone-independent restraint on their reproductive axis. In contrast, in similarly aged juvenile males, no changes occurred in LH levels or Kiss1 or NKB expression following castration, suggesting that gonadal hormone-independent mechanisms restrain kisspeptin/NKB-dependent activation of the male reproductive axis before puberty. Notably, adult mice of both sexes showed comparable rapid increases in Kiss1/NKB expression and LH secretion following gonadectomy, signifying that sex differences in the regulation of ARC Kiss1/NKB neurons are manifest only during peripubertal development. Our findings demonstrate that the mechanisms controlling pubertal activation of reproduction in mice are different between the sexes and suggest that gonadal hormone-independent central restraint on pubertal timing involves Kiss1/NKB neurons in the ARC.
Keywords: kisspeptin, Kiss1r, G protein-coupled receptor 54, neurokinin B, gonadotropin-releasing hormone, development, reproduction
puberty comprises the physiological and behavioral changes that occur during the transition from juvenile life into sexual maturity and reproductive competence. Puberty onset is usually defined as the activation (or in some species, reactivation) of the neuroendocrine reproductive axis (11, 35, 37), which typically occurs earlier in females than in males (12, 13, 19). Although the molecular mechanisms underlying the onset of puberty remain unknown, the guiding mechanisms include an apparent reduction in the sensitivity of hypothalamic circuits to the feedback actions of gonadal steroids (estradiol or testosterone) along with gonadal hormone-independent changes in the activity of inhibitory or stimulatory neural networks within the brain (11, 22, 35, 37).
Despite the knowledge that both the gonadal hormone-dependent and -independent processes underlying puberty contain neural components, the identity of the neuronal factors and substrates involved in these mechanisms is unknown. Kisspeptin, a neuropeptide encoded by the Kiss1 gene, is a potent secretagogue for gonadotropin-releasing hormone (GnRH) in mammals (reviewed in Refs. 25 and 38) and may be a critical component of pubertal maturation based on several observations. First, humans and mice with disabling mutations or transgenic disruptions of the kisspeptin receptor gene fail to progress through puberty and remain infertile as adults (9, 14, 27, 46, 47). Second, kisspeptin administration to prepubertal rats and monkeys induces precocious gonadotropin secretion (30, 48). Finally, in rodents and primates, the hypothalamic expression of both Kiss1 and its receptor increase in association with pubertal maturation (6, 21, 29, 48). Collectively, these observations suggest that kisspeptin-induced activation of GnRH neurons serves as a gating mechanism for puberty; however, the identity of the hormonal or molecular trigger that activates Kiss1 neurons at puberty remains unknown.
In rodents, Kiss1 is expressed in the hypothalamic arcuate nucleus (ARC) and the anteroventral periventricular nucleus/periventicular nucleus continuum (AVPV/PeN) (18, 26, 42, 48, 49); in adulthood, the Kiss1 population in the AVPV/PeN, but not the ARC, is sexually dimorphic, with females having greater numbers of Kiss1 neurons in the AVPV/PeN than males (23, 26). Moreover, Kiss1 expression in the AVPV/PeN is stimulated by gonadal steroids, whereas Kiss1 neurons in the ARC are inhibited by gonadal steroids (and, conversely, stimulated by gonadectomy) (26, 50, 51). This differential steroidal regulation of Kiss1 in the AVPV/PeN and ARC may underlie the cellular mechanisms of steroid-mediated positive and negative feedback control of GnRH secretion, respectively (25, 38, 49). However, the physiological role of these two phenotypically unique populations of Kiss1 neurons in pubertal maturation is unclear.
In addition to kisspeptin, neurokinin B (NKB), a tachykinin encoded by the Tac2 gene, has recently been implicated in the control of puberty and reproduction. In rodents, sheep, and primates, NKB is expressed in the ARC, where it colocalizes with kisspeptin neurons (2, 17, 31, 40). In addition, the NKB receptor NK3 is expressed on GnRH neurons (28, 54). Moreover, like Kiss1, NKB mRNA in the ARC increases following gonadectomy (8, 10, 40, 43, 45) in conjunction with a rise in luteinizing hormone (LH) secretion, suggesting that NKB signaling is stimulatory to the reproductive axis. Indeed, humans lacking NKB/NK3 signaling have delayed puberty onset and impaired fertility (55). Despite the implication that NKB plays a role in regulating puberty and reproduction, there is little insight concerning its mechanism of action in the brain.
In the present study, we investigated the role of gonadal hormones in the regulation of Kiss1 and NKB in the ARC during the peripubertal period and examined whether sex differences in the activation of Kiss1/NKB neurons might explain sex differences in the tempo of pubertal maturation. First, we argued that if activation of Kiss1/NKB neurons in the ARC is an essential component of the pubertal process, then these neurons should be regulated by the same gonadal hormone-dependent and/or -independent factors that govern puberty onset. We postulated that if pubertal activation of reproductive circuits, including Kiss1/NKB neurons, is regulated entirely by changes in sensitivity to gonadal hormone feedback, then removal of gonadal steroids before puberty should induce a precocious activation of both Kiss1/NKB neurons and gonadotropin secretion. However, if the peripubertal reproductive axis is also regulated by gonadal hormone-independent restraint mechanisms, then removal of gonadal hormone feedback before puberty may not induce early activation of either Kiss1/NKB circuits or LH secretion (because central restraining processes are still present). Second, we hypothesized that earlier puberty onset in females compared with males reflects sex differences in the gating of Kiss1/NKB reproductive circuits. Specifically, we hypothesized that there is greater or longer-lasting inhibition (or lack of activation) of Kiss1/NKB circuitry in juvenile males compared with juvenile females. We present evidence demonstrating that 1) the mechanisms controlling pubertal activation of reproduction in mice are different between the sexes such that gonadal hormone-independent restraint on pubertal timing is more prevalent (or longer lasting) in juvenile males than in females and 2) this gonadal hormone-independent restraint mechanism acts on Kiss1/NKB neurons in the ARC, thereby delaying activation of sexual maturation in developing males compared with females.
MATERIALS AND METHODS
Animals.
Adult C57Bl6 male and female mice were purchased from Jackson Laboratories and allocated into breeding pairs. Beginning 19 days after pairing, litters were assessed daily, and day of birth was considered postnatal day (PND) 1. In experiments in which prepubertal mice were studied, animals between the ages of PND 14 and PND 16 were used to ensure that they were not already undergoing the pubertal transition (which is evidenced in females by vaginal opening on PND 24–28 but may actually initiate several days beforehand). All animals were housed on a 12:12-h light-dark cycle (lights off at 1800) with food and water available ad libitum. All experiments were conducted in accordance with the National Institutes of Health (NIH) Animal Care and Use Guidelines and with approval of the Animal Care and Use Committee of the University of Washington.
Gonadectomies and blood and tissue collection.
Mice were anesthetized with isoflurane and bilaterally gonadectomized (GDX) or left intact. Briefly, the animal's ventral skin was shaved and cleaned, and small incisions were made in the skin and abdominal musculature. The gonads were identified and excised, the abdominal muscle was sutured, and the skin incision was closed with surgical clips (adults) or surgical thread (juveniles). Animals that were left intact received both ventral incisions, but no tissue was removed.
Blood collections were obtained via retroorbital bleeding (∼100 μl) of animals given isoflurane anesthesia. Blood was centrifuged and the serum stored at −20°C until it was assayed for LH or testosterone (T) via radioimmunoassay. All hormone assays were performed by the University of Virginia Ligand Assay Laboratory (Charlottesville, VA), as described previously (24, 27). Brains of euthanized animals were collected and immediately frozen on dry ice and stored at −80°C. Frozen brains were sectioned on a cryostat into 5 sets, 20 microns/section, and the sections were thaw-mounted onto slides. Slides were stored at −80°C until processing via in situ hybridization. Because Kiss1 expression and hormone levels are influenced by circadian signaling (41), all blood and tissue collections were obtained within a 3-h time window between 1000 and 1300.
In situ hybridization.
In situ hybridization for Kiss1 or NKB mRNA was performed as described previously (18, 31, 50). Briefly, slide-mounted brain sections were fixed in 4% paraformaldehyde, pretreated with acetic anhydride, rinsed in 2× standard saline citrate (SSC; sodium citrate, sodium chloride), delipidated in chloroform, dehydrated in graded ethanols, and air-dried. Radiolabeled riboprobe (0.03–0.05 pmol/ml, depending on the probe) was combined with 1/20 volume yeast tRNA (Roche Biochemicals, Indianapolis, IN) in Tris-EDTA (0.1 M Tris-0.01 M EDTA, pH 8.0), and this mixture was heat denatured in boiling water for 3 min, iced for 5 min, added to hybridization buffer, and applied to each slide (100 μl/slide). Slides were coverslipped and placed in humidity chambers at 55°C for 16 h. Following hybridization, the slides were washed in 4× SSC and then placed into RNAse [37 mg/ml RNase A (Roche Biochemicals) in 0.15 M sodium chloride, 10 mM Tris, and 1 mM EDTA, pH 8.0] for 30 min at 37°C and then in RNase buffer without RNase at 37°C for 30 min. After a wash in 2× SSC at room temperature, slides were washed twice in 0.1× SSC at 62°C and then dehydrated in graded ethanols and air-dried. Slides were then dipped in Kodak NTB emulsion, air-dried, and stored at 4°C for 7–10 days (depending on the assay). Slides were then developed, dehydrated in ethanols, cleared in Citrasolv (Fisher Scientific), and coverslipped with Permaslip (Sigma).
Experiment 1: effects of gonadectomy on LH and Kiss1 expression in prepubertal males and females.
Experiment 1A assessed whether removal of gonadal hormones before the normal onset of puberty (which is typically ∼PND 24–28 and PND 32–36 for female and male mice, respectively) results in an early activation of gonadotropin secretion and a corresponding stimulation of Kiss1 neurons in the ARC. We reasoned that if pubertal activation of reproductive circuits is regulated predominantly by changes in sensitivity to gonadal hormone-dependent feedback, then early removal of hormone feedback should result in precocious stimulation of LH secretion and perhaps ARC Kiss1 expression. If, however, the peripubertal reproductive axis is regulated in part (or entirely) by gonadal hormone-independent central circuits, then early removal of gonadal hormones may not induce early activation. Juvenile (PND 14) male and female mice were GDX or left intact and then euthanized 2 or 4 days later (PND 16 and 18, respectively). Blood and brains were collected at the time of euthanization. Blood serum was assayed for LH levels as an indicator of activation (or not) of the neuroendocrine reproductive axis. Brains were processed with in situ hybridization for Kiss1 mRNA levels in the ARC; the number of Kiss1-expressing cells, as well as the relative amount of Kiss1 mRNA per cell (determined by number of silver grains), was determined for each animal. Kiss1 expression in males and females was assayed separately on the basis of experimental limitations on the maximum number of slides per assay.
Results from experiment 1A suggested a possible sex difference in the regulation of gonadotropin secretion and ARC Kiss1 neurons after prepubertal gonadectomy. In experiment 1B, an alternate set of brain sections for PND 18 males and females (GDX and intact for each sex) was analyzed together in the same in situ hybridization assay to allow for direct intersex comparisons of ARC Kiss1 expression in juvenile animals. Moreover, we have shown previously that Kiss1 gene expression in the rat AVPV/PeN is sexually dimorphic in adulthood, but this has not been examined before puberty (or in mice). Thus, for comparison of possible prepubertal sex differences in Kiss1 neurons between hypothalamic regions, experiment 1B also evaluated Kiss1 mRNA levels in the AVPV/PeN of PND 18 males and females (GDX and intact); n = 10–16 animals/group for both experiment 1A and 1B.
Experiment 2: effects of castration on serum T levels in juvenile males.
In experiment 1, we found that GDX juvenile male mice do not display increased LH or Kiss1 levels 2 or 4 days later. These findings raised the possibility that gonadectomy did not effectively reduce T levels. Therefore, we next determined whether 4 days postcastration is long enough to fully remove circulating T from the juvenile system. Juvenile PND 14 male mice (n = 7–9/group) were GDX or left intact and euthanized 4 days later on PND 18. Blood serum from these males was assayed for T levels via RIA to determine whether prepubertal gonadectomy actually removes all endogenous circulating T.
Experiment 3: effects of gonadectomy on LH and Kiss1 mRNA levels in adult males and females.
Another possible reason that males in experiment 1 did not show an effect of castration is that a 4-day duration following gonadectomy is not sufficient for the male system to display changes in LH or Kiss1 expression independent of the animal's age. In this experiment, GDX adults of both sexes were studied to determine whether the sex difference in the regulation of Kiss1 neurons observed after 4 days in GDX juvenile animals is also present (or not) in adulthood. Adult (8 wk) male and female mice were GDX or left intact and then euthanized 4 days later. To control for stage of cycle, all intact females were euthanized on the morning of diestrus (as determined with daily vaginal smears). Blood and brains were collected at the time of euthanization and assayed for LH levels and Kiss1 mRNA in the ARC, respectively; n = 6–7 animals/group.
Experiment 4: assessment of Kiss1 expression in adult males that were GDX as juveniles.
In the previous experiments, we observed a sex difference in the regulation of ARC Kiss1 neurons and gonadotropin secretion in juvenile, but not adult, mice such that GDX prepubertal males display no changes in either LH secretion or ARC Kiss1 expression 2 and 4 days later. This experiment determined whether GDX juvenile males eventually show increased LH and Kiss1 expression if allowed to age beyond the normal period of pubertal onset into adulthood or whether prepubertal GDX chronically impairs the proper development of neuroendocrine reproductive circuits (including ARC Kiss1 neurons). PND 14 male mice were GDX or left intact and then euthanized 3.5 wk later in adulthood at PND 45 (after the time of normal puberty, which is normally ∼PND 35 in males). Blood and brains were collected at the time of euthanization and assayed for LH levels and Kiss1 expression in the ARC, respectively; n = 6–8 animals/group.
Experiment 5: effects of gonadectomy on NKB expression in the ARC of juvenile males and females.
The previous experiments identified a unique developmental sex difference in the regulation of Kiss1 gene expression in the ARC of juvenile, but not adult, mice. This experiment determined whether this juvenile sex difference extends to NKB, another reproduction-related gene also expressed in the ARC. PND 14 male and female mice were GDX or left intact and then euthanized 4 days later (PND 18). Brains were collected and assayed for NKB expression in the ARC using in situ hybridization assay. For comparison with the juvenile situation, NKB expression in the ARC was also assessed in adult male and female mice that were GDX or left intact (in adulthood) and then euthanized 4 days later as well as in males that were GDX prepubertally on PND 14 and then euthanized in adulthood on PND 45 (alternate sets of tissue from experiments 3 and 4); n = 8–12 animals/group.
Data analysis and statistics.
In situ hybridization slides were analyzed with an automated image-processing system by a person blind to the treatment group of each slide. Custom grain-counting software was used to unilaterally count the number of cell clusters and the number of silver grains over each cell (a semiquantitative index of mRNA content per cell), as described previously (18, 26, 50). Cells were considered Kiss1 (or NKB)-positive when the number of silver grains in a cluster exceeded that of background by threefold. In each experiment, differences in group means of LH or mRNA levels were assessed via ANOVA, with post hoc analysis determined by Fisher's least significant difference test. Due to nonhomogeneity in the variance of AVPV Kiss1 cell numbers between groups, these data were log transformed prior to statistical analysis. For all comparisons, statistical significance was set at P < 0.05.
RESULTS
Experiment 1: effects of gonadectomy on LH and Kiss1 expression in prepubertal males and females.
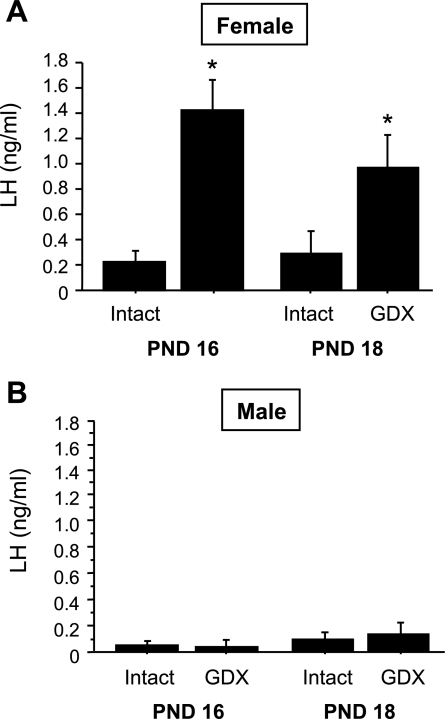
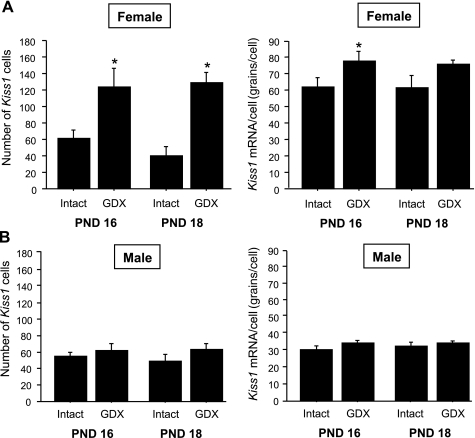
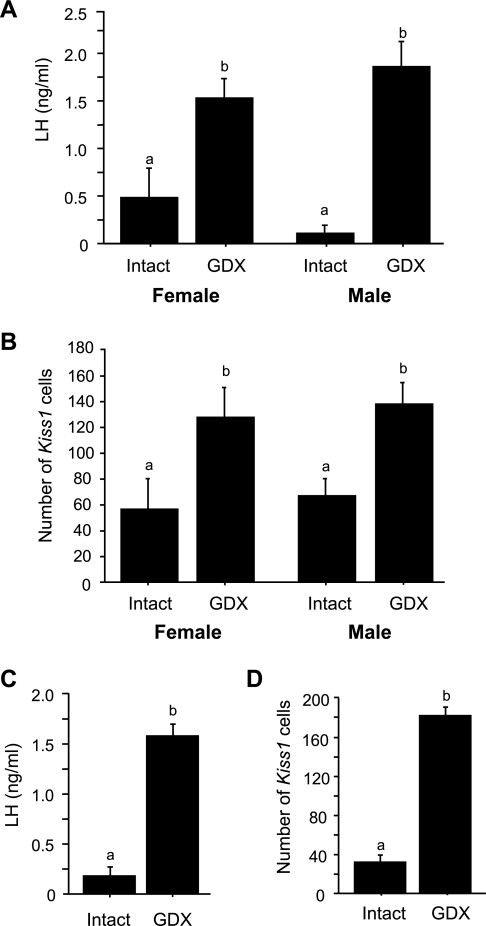
In experiment 1A, we evaluated whether the removal of gonadal hormones on PND 14, prior to normal puberty onset, triggers early increases in gonadotropin secretion and/or Kiss1 gene expression in the ARC. In juvenile GDX females, serum LH levels were significantly elevated at 2 and 4 days postgonadectomy (PND 16 and 18, respectively) relative to LH levels of intact females (P < 0.05 for each age; Fig. 1A). In contrast, plasma LH levels were unchanged in GDX juvenile males at 2 and 4 days postgonadectomy (P > 0.05 for each age relative to intact males; Fig. 1B). Kiss1 gene expression in the ARC followed a similar sex-specific pattern. Specifically, juvenile GDX females had significantly higher numbers of Kiss1 neurons in the ARC than intact females at both postgonadectomy ages (P < 0.05 for each time; Figs. 2 and 3), whereas GDX juvenile males did not have more detectable ARC Kiss1 neurons than intact males at either postgonadectomy age (Figs. 2 and 3). Silver grains per cell, a semiquantitative measurement of Kiss1 mRNA per cell, followed a pattern similar to the number of Kiss1 cells for each sex (Fig. 3).
Fig. 1.
Mean (+ SE) plasma LH levels in juvenile female (A) and male (B) mice that were gonadectomized (GDX) or left intact on postnatal day (PND) 14 and then euthanized 2 or 4 days later (PND 16 and 18, respectively). GDX female mice displayed elevated LH at all postgonadectomy time points, whereas GDX males did not show any significant increases in LH. *Significantly different from intact animal of same age.
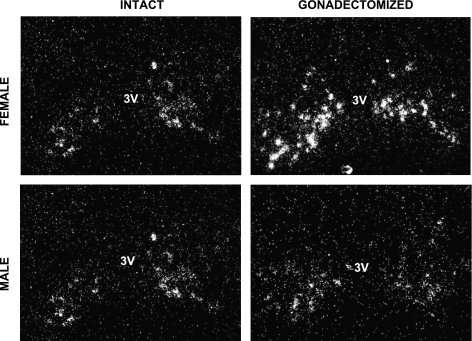
Fig. 2.
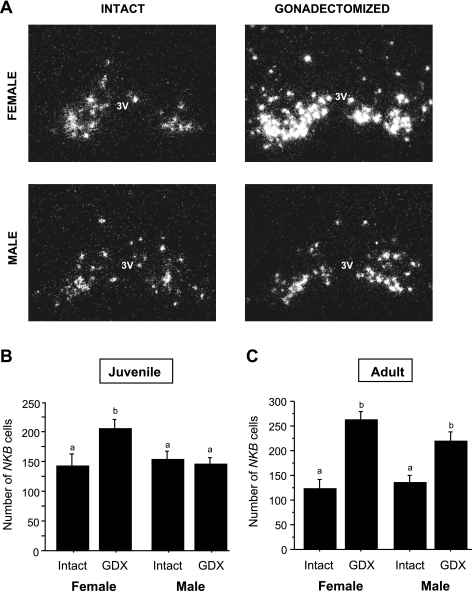
Representative photomicrographs of Kiss1 neurons in the arcuate nucleus (ARC) of prepubertal females and males that were GDX or left intact on PND 14 and then euthanized 2 days later (PND 16). 3V, third ventricle.
Fig. 3.
Mean (+ SE) number of Kiss1 cells or Kiss1 mRNA per cell in the ARC of prepubertal females (A) and males (B) that were GDX or left intact on PND 14 and then euthanized 2 or 4 days later (i.e., on PND 16 or PND 18, respectively). Kiss1 gene expression in the ARC was significantly higher in GDX than in intact females on PND 16. In contrast, there was no significant difference in ARC Kiss1 expression between intact and GDX juvenile males. *Significantly different from intact animal of same age (P < 0.05).
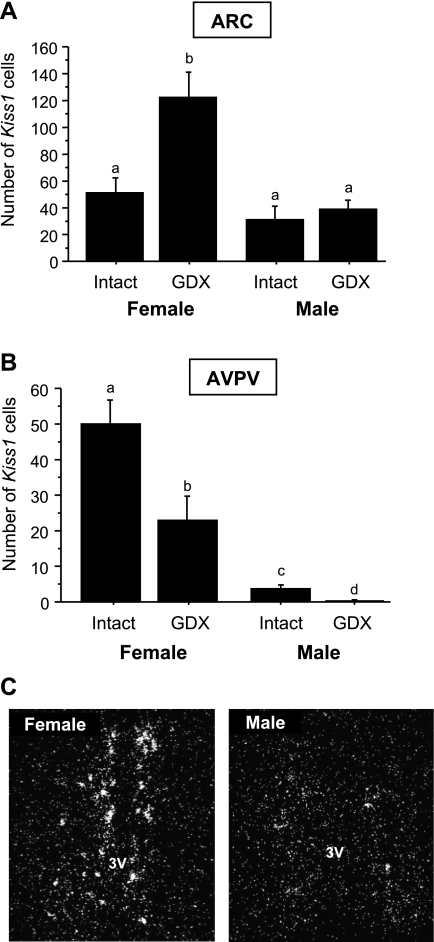
In experiment 1B, brain sections of PND 18 males and females were assayed together. In the ARC, the number of Kiss1 neurons was not significantly different between intact PND 18 males and females (Fig. 4A). However, in PND 18 GDX males and females (4 days after gonadectomy) there was a robust sex difference, with GDX juvenile females having significantly more Kiss1 neurons in the ARC than GDX juvenile males (P < 0.05; Fig. 4A). The number of ARC Kiss1 neurons in GDX PND 18 males was not significantly different from intact males and females (Fig. 4A). These findings indicate a sex difference in prepubertal ARC Kiss1 neurons that is observed only after removal of gonadal hormones. In the AVPV/PeN, a region with previously documented sex differences in Kiss1 expression in adult rodents (females > males), we found a significant sex difference in the number of Kiss1 neurons in PND 18 mice in both intact and GDX scenarios. PND 18 females had significantly more Kiss1 neurons (and Kiss1 mRNA/cell; data not shown) than PND 18 males, regardless of gonadal status (P < 0.05; Fig. 4B), indicating that the AVPV/PeN Kiss1 system is sexually differentiated as early as PND 18, and this sex difference is independent of the circulating hormonal milieu. Intact PND 18 males had low numbers of Kiss1 neurons in the AVPV/PeN but still significantly more than GDX PND 18 males (P < 0.05; Fig. 4B), indicating that the AVPV/PeN Kiss1 system in juvenile males responds to gonadectomy, unlike ARC Kiss1 neurons.
Fig. 4.
Sex differences in the mean number of Kiss1 cells in the ARC (A) or anteroventral periventricular nucleus/periventicular nucleus continuum (AVPV/PeN; B) of prepubertal males and females that were GDX or left intact on PND 14 and euthanized 4 days later on PND 18. Brain tissue from animals of each sex was assayed together to directly assess sex differences in Kiss1 expression. Bars with different letters differ significantly from each other (P < 0.05). A direct comparison between GDX and intact males revealed a small but significant difference in the number of AVPV/PeN Kiss1 cells (P < 0.05). C: representative photomicrographs of Kiss1 neurons in the AVPV/PeN of prepubertal females and males that were left intact on PND 14 and then euthanized 4 days later (PND 18).
Experiment 2: effect of castration on serum T levels in juvenile males.
In experiment 1, we determined that both gonadotropin secretion and ARC Kiss1 gene expression are elevated 4 days after gonadectomy in juvenile females but not males. Here, we assessed whether plasma T levels were significantly different between intact and GDX juvenile mice 4 days postcastration (on PND 18). In general, serum T levels were low in intact PND 18 males given their young age. Even so, GDX PND 18 males had significantly lower, in most cases undetectable, T levels compared with intact PND 18 males (0.10 ± 0.05 vs. 0.27 ± 0.05 ng/ml, P < 0.05), confirming reduced levels of T at 4 days postcastration. Thus, the absence of elevated LH and Kiss1 expression in PND 18 GDX males is not due to the presence of residual circulating T.
Experiment 3: effects of gonadectomy on levels of LH and Kiss1 mRNA in adult males and females.
In experiment 1, both gonadotropin secretion and ARC Kiss1 gene expression were elevated in juvenile females but not juvenile males 4 days after gonadectomy. Experiment 3 determined whether 4 days postcastration is long enough to evoke changes in LH secretion or Kiss1 expression in adult males and whether the sex difference in the regulation of ARC Kiss1 neurons that was observed prepubertally is also present in adulthood. We found that, unlike the pattern observed in juvenile animals, serum LH levels were significantly elevated in adults of both sexes 4 days after gonadectomy (P < 0.05 vs. intact adults; Fig. 5A). Similarly, unlike the juvenile situation, Kiss1 expression in the ARC was significantly increased to the same degree in both adult males and females 4 days after gonadectomy (P < 0.05 relative to intact adults; Fig. 5B), indicating that the sex difference in Kiss1 regulation in the ARC is present before puberty but not in adulthood.
Fig. 5.
Mean (+ SE) serum LH levels and number of Kiss1 cells in the ARC of adult mice that were either GDX or left intact as adults and euthanized 4 days later (A and B) or GDX prepubertally on PND 14 and then euthanized later in adulthood on PND 45 (C and D). Both LH levels and the number of Kiss1 cells in the ARC were elevated in adult mice of both sexes. Bars with different letters differ significantly from each other (P < 0.05).
Experiment 4: Kiss1 expression and LH secretion in adult males that were GDX as juveniles.
The previous experiments determined that male mice do not display activation of either LH secretion or ARC Kiss1 gene expression 4 days after gonadectomy as juveniles but do so after gonadectomy as adults. Here, we tested whether the reproductive axis (including Kiss1 neurons in the ARC) of juvenile GDX males eventually becomes activated as they age into adulthood in the absence of developmental changes in gonadal hormones. If so, it would indicate that the gonadal hormone-independent restraint on male reproductive circuits is eventually overcome as males reach adulthood and that the juvenile ARC Kiss1 system can develop the adult phenotype in the absence of gonadal hormones. We found that males that were GDX as juveniles on PND 14 and euthanized 31 days later in adulthood (on PND 45) had serum LH levels that were significantly elevated above that of intact adult males (P < 0.05; Fig. 5C). Similarly, the number of Kiss1 neurons in the ARC was significantly higher in adult males that had been GDX as juveniles compared with adult males that were left intact as juveniles (P < 0.05; Fig. 5D). Likewise, Kiss1 mRNA per cell was significantly greater in adult males GDX as juveniles (P < 0.05 vs. intact adult males; data not shown).
Experiment 5: effects of gonadectomy on NKB expression in juvenile males and females.
Here, we tested whether NKB, which is coexpressed in ARC Kiss1 cells (17, 31), shows similar sex-specific changes in gene expression as Kiss1 following removal of gonadal hormones during the prepubertal period. We found that, similar to Kiss1, the number of NKB-expressing neurons in the ARC was significantly upregulated 4 days after gonadectomy in PND 18 GDX females but not PND 18 GDX males (compared with PND 18 intact animals, P < 0.05 for female comparison; Fig. 6, A and B). Similarly, PND 18 GDX females had significantly more (P < 0.05) NKB mRNA per cell compared with the other three PND 18 groups [121.2 ± 3.5 (GDX female) vs. 101.6 ± 7.0 (intact female), 99.5 ± 5.3 (GDX male), and 93.9 ± 8.2 (intact male) grains/cell]. Also like Kiss1, the sex difference in NKB regulation was no longer manifest in adulthood, since both males and females showed similar increased NKB gene expression 4 days after gonadectomy as adults (P < 0.05 for both sexes relative to intact adults; Fig. 6C). Moreover, as with Kiss1, the number of NKB-expressing neurons in the ARC was significantly higher in adult males that had previously been GDX as juveniles compared with adult males that had been left intact as juveniles (261.5 ± 15.4 vs. 140.7 ± 30.6 NKB cells, P < 0.01). NKB mRNA per cell in the ARC followed a similar pattern, being higher in adult males that had been GDX as juveniles than those left intact (P < 0.01; data not shown).
Fig. 6.
Representative photomicrographs (A) and mean (+ SE) number of neurokinin B (NKB) cells in the ARC (B) of mice that were GDX prepubertally (PND 14) and euthanized 4 days later on PND 18. Like Kiss1 (see Fig. 3), NKB gene expression in the ARC was elevated in GDX females but not in GDX males on PND 18. Bars with different letters differ significantly from each other (P < 0.05). C: mean (+ SE) number of NKB cells in the ARC of adult mice that were GDX in adulthood and euthanized 4 days later. Similarly to Kiss1 expression (see Fig. 5), NKB expression in the ARC was elevated in all GDX adults compared with intact animals. Bars with different letters differ significantly from each other (P < 0.05).
DISCUSSION
Puberty reflects the physiological processes that guide the awakening of the adult neuroendocrine reproductive axis (11, 35, 37). Despite awareness that gonadal hormone-dependent and hormone-independent mechanisms may guide the tempo of pubertal development (11, 22, 35, 37), the cellular and molecular processes that mediate this event are poorly understood, as is the enigma that females usually mature earlier than males (12, 13, 19). Recently, neuronal kisspeptin and NKB signaling have each been implicated in puberty onset, as highlighted by findings that humans lacking either a functional kisspeptin receptor or NKB receptor (or NKB itself) display salient deficits in pubertal onset and fertility (9, 14, 27, 46, 47, 55). Our present study provides further evidence that Kiss1/NKB neurons, located in the ARC, are linked to the timing of developmental activation of the reproductive axis and that the regulation of these neurons is sexually differentiated. Specifically, gonadectomy during the juvenile period stimulates Kiss1/NKB expression in the ARC and concurrently increases gonadotropin secretion in female but not male mice, suggesting that the neuronal circuits that activate the peripubertal reproductive axis are regulated in a sex-specific manner. On the basis of our findings, we infer that activation (or disinhibition) of Kiss1/NKB neurons in the ARC constitutes a critical element of the puberty-triggering mechanism. Furthermore, we deduce that during juvenile life in females, Kiss1/NKB neuronal activity in the ARC is constrained primarily by gonadal hormones, whereas in juvenile males, ARC Kiss1/NKB signaling is gated by gonadal hormone-independent factors (and, likely concurrently, by gonadal hormones as well). Thus, sex differences in the timing of puberty onset may reflect fundamentally different neuronal mechanisms (or differences in their regulation) that govern the tempo of sexual maturation in males and females.
We have shown previously that the Kiss1 population in the ARC is not sexually dimorphic in adult rodents, regardless of sex steroid milieu (26). However, our present study identifies a robust sex difference in the regulation of ARC Kiss1 neurons during the prepubertal period. We also report for the first time a prepubertal sex difference in the regulation of NKB expression in the ARC. The factor(s) regulating Kiss1/NKB neurons in the ARC of juvenile animals has not previously been identified. However, our findings suggest that, in juvenile females, at PND 16–18, ovarian hormones (presumably E2) are key regulators of ARC Kiss1/NKB neurons, because gonadectomy induces striking increases in Kiss1 and NKB expression. Thus, we infer that E2-mediated inhibition of Kiss1/NKB neurons in the ARC represents a primary mechanism for maintaining the quiescent state of the reproductive axis in intact prepubertal females. Indeed, estrogen receptors are coexpressed in virtually all Kiss1 and NKB neurons in the ARC of adult mice (10, 50), lending support to this argument. In contrast to females, juvenile GDX male mice do not display increases in Kiss1, NKB, or LH levels at PND 16–18. Thus, although gonadal hormones likely inhibit the reproductive axis in juvenile males (as in juvenile females), other regulatory inputs, apart from gonadal hormones, must either impose active restraint on or fail to activate Kiss1/NKB expression and LH secretion during prepubertal life.
NKB has recently been implicated in the control of puberty and reproduction. Humans lacking NKB or its receptor have delayed puberty onset and impaired fertility (55), suggesting a role for NKB signaling in the initiation of puberty. Although intracerebroventricular infusions of NKB agonist diminish LH secretion in adult female ovariectomized E-primed rats (44), site-specific NKB agonist infusions in discrete hypothalamic regions (rather than the ventricles) stimulate LH secretion in sheep (32), suggesting that NKB's effects are site specific. Indeed, NKB most likely acts at several places in the brain, including GnRH neurons in the medial preoptic area and Kiss1/NKB neurons in the ARC, both of which express NK3 (28, 31, 54). Our results here implicate NKB in the ARC as a critical component of the circuits underlying activation of the neuroendocrine reproductive axis.
Our results signify that the brains of juvenile male and female mice, including Kiss1/NKB neurons in the ARC, are sexually differentiated such that prepubertal females aged PND 14 can initiate activation of the reproductive axis after gonadectomy, whereas similarly aged males cannot. These findings suggest that sex differences in how and when Kiss1/NKB neurons in the ARC are regulated may be instrumental to explaining sex differences in puberty onset. Although sex differences in sexual maturation are well documented, the basis for the differential timing of puberty between males and females is unclear and may reflect either fundamentally different mechanisms operating in each sex or, alternatively, a common regulatory process that is initiated at different times in the two sexes (i.e., earlier in females than in males). Both of these possibilities could include either the presence of additional (or longer-lasting) restraining factors in PND 16–18 male vs. female brains or the absence of additional (or later induction of) activating circuits in PND 16–18 males that are already present in females at the same prepubertal age. In either case, these additional restraining/activating factors appear to be gonadal hormone independent in nature, as is their developmental onset/offset, as evidenced by that fact that LH and Kiss1/NKB in GDX males were not elevated at PND 18 but did eventually increase by PND 45 in the absence of any changes in gonadal hormone levels. The identity of the gonadal hormone-independent factor(s) that regulates prepubertal Kiss1/NKB neurons is currently unknown. Previous studies in primates and rodents have implicated several factors that may regulate pubertal circuits, including neuropeptide Y, leptin, GABA, enhanced at puberty 1, and glial factors (3, 4, 34, 35, 37). Any one or more of these nongonadal factors may provide critical input into prepubertal Kiss1/NKB neurons and perhaps even explain the sex differences in Kiss1/NKB expression in the ARC after prepubertal gonadectomy, and revealing their identity will be an important objective for future studies.
We and others have shown previously that the Kiss1 system in the AVPV/PeN of adult rodents is sexually differentiated, with adult females having many more Kiss1 neurons in the AVPV/PeN than males (6, 26). This sex difference in AVPV/PeN Kiss1 neurons may explain the ability of adult females, but not males, to display an E2-induced LH surge (23, 27). Here, we report that Kiss1 expression in the AVPV/PeN of mice is sexually dimorphic as early as PND 18. This is the first demonstration of the sex difference in AVPV/PeN Kiss1 expression in prepubertal animals of any species. [In fact, females have significantly more Kiss1 neurons in the AVPV/PeN than males as early as PND 16 (P < 0.01; Kauffman AS and Steiner RA, unpublished observations)]. Our study also presents the earliest reported age (PND 16) of Kiss1 gene expression in the AVPV/PeN, with a previous study in the mouse documenting detectable kisspeptin protein by immunohistochemistry in the female AVPV/PeN on PND 25 but not on PND 10 (6). It is noteworthy that a direct comparison between intact and GDX PND 18 males revealed a small but significant elevation in Kiss1 cell number in the AVPV/PeN of intact males. Similarly, we have found that, on PND 16, intact males have five times as many Kiss1 cells in the AVPV/PeN than GDX males at this age (P < 0.01; Kauffman AS and Steiner RA, unpublished observations), indicating that Kiss1 neurons in the AVPV/PeN of juvenile males can respond to changes in gonadal hormone status, unlike Kiss1 neurons in the ARC.
Recently, Clarkson et al. (5) suggested a role for the AVPV/PeN Kiss1 system in the initiation of puberty onset on the basis of the finding that ovariectomized prepubertal mice fail to display vaginal opening or elevated kisspeptin protein in the AVPV/PeN later in early adulthood. However, Kiss1 mRNA and kisspeptin protein levels in the AVPV/PeN are highly E2 dependent (upregulated by E2) in the adult female rodent (1, 50, 52), and thus, an important caveat when interpreting the finding of Clarkson et al. (5) is that their GDX juveniles were never replaced with E2 in adulthood. Therefore, it is possible that the low kisspeptin levels observed in the AVPV/PeN of their adult GDX animals reflect absent circulating E2 in adulthood. Moreover, in our study, Kiss1 mRNA levels in the AVPV/PeN clearly decrease in prepubertal females following GDX despite the concurrent increase in LH secretion in these same females. We interpret this to signify that Kiss1 activity in the AVPV/PeN is unlikely to be driving the elevated GnRH/LH secretion in these prepubertal animals (since the AVPV/PeN is producing significantly less kisspeptin after gonadectomy). Notwithstanding, it is certainly possible that the AVPV/PeN Kiss1 system contributes to the enhanced drive to GnRH neurons at some point during puberty; however, this would definitely not exclude an important (even central) role for other neural circuits as well, including Kiss1/NKB neurons in the ARC. Indeed, it is somewhat paradoxical that in the GDX mice lacking vaginal opening in the study conducted by Clarkson et al. (5), kisspeptin levels were also noticeably reduced in the ARC, but the possibility that the diminished ARC kisspeptin levels contributed to these animals' impaired puberty onset was not addressed.
Interestingly, although not yet assessed, there may not be a sex difference in the regulation of ARC Kiss1/NKB neurons in prepubertal rats, since GDX prepubertal rats of both sexes appear to show comparable rapid elevations in LH secretion both 2 and 4 days postsurgery, at least at the ages studied (20, 33, 39). However, adult female rats take much longer to exhibit elevated LH levels following gonadectomy than they do as juveniles (15, 16, 53), indicating that some developmental alteration in central feedback mechanisms occurs in female rats as they age through puberty. Although these earlier observations in prepubertal rats suggest that puberty in rodents is primarily gonadal hormone dependent, our findings argue that gonadal hormone-independent factors are also critical at least in mice and perhaps other rodents as well. Similarly, puberty in primates, including humans, also appears to involve both gonadal hormone-dependent and -independent processes, as evidenced by findings that GDX juvenile monkeys do not display elevated LH before the normal age of puberty (36, 37), nor do humans that lack gonadal hormone secretion before puberty (7, 19). To the extent that such divergent species can be compared (and with caution), puberty in mice may therefore share common features with other species, including humans and nonhuman primates. Therefore, our findings may have implications for the pathophysiology of puberty in humans, in which puberty is not only earlier in girls than in boys, but girls also display a much higher incidence of precocious puberty (10-fold) than boys (37). On the other hand, boys are more likely to have delayed puberty than girls (37). These sex differences in normal puberty onset and disorders in pubertal timing are widely recognized but poorly understood and may involve sex differences in the timing or regulation of Kiss1/NKB signaling in the brain.
In conclusion, we report a novel sex difference in the regulation of LH secretion and Kiss1 and NKB expression in the ARC of juvenile mice such that the developing brains of juvenile males exhibit greater (or longer-lasting) gonadal hormone-independent restraint (or less gonadal hormone-independent activation) on reproductive neuronal circuits, including ARC Kiss1/NKB neurons. This sex difference in Kiss1/NKB regulation may contribute to the differential timing of puberty onset between the sexes. Thus, although gonadal hormones most likely play a critical role in regulating puberty onset in both sexes, our findings indicate that gonadal hormone-independent processes also contribute to timing puberty in mice (as they do in primates). Additional experiments are needed to identify the central factors inhibiting/activating Kiss1/NKB neurons in the ARC and to determine the specific role of these two reproductive neuropeptides.
GRANTS
This research was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD)/NIH through cooperative agreements U54-HD-12629 (to the University of Washington Center for Research in Reproduction and Contraception) and U54-HD-28934 (University of Virginia Ligand Assay Core) as part of the Specialized Cooperative Centers Program in Reproduction and Infertility Research. Additional funding was provided by NICHD Grants HD-056157 (to A. S. Kauffman) and HD-049651 (to R. A. Steiner) and the Fulbright Program of the US Department of State and the Ministerio Español de Ciencia e Innovación (to V. M. Navarro).
REFERENCES
- 1.Adachi S, Yamada S, Takatsu Y, Matsui H, Kinoshita M, Takase K, Sugiura H, Ohtaki T, Matsumoto H, Uenoyama Y, Tsukamura H, Inoue K, Maeda K. Involvement of anteroventral periventricular metastin/kisspeptin neurons in estrogen positive feedback action on luteinizing hormone release in female rats. J Reprod Dev 53: 367–378, 2007 [DOI] [PubMed] [Google Scholar]
- 2.Chawla MK, Gutierrez GM, Young WS 3rd, McMullen NT, Rance NE. Localization of neurons expressing substance P and neurokinin B gene transcripts in the human hypothalamus and basal forebrain. J Comp Neurol 384: 429–442, 1997 [DOI] [PubMed] [Google Scholar]
- 3.Cheung CC, Thornton JE, Kuijper JL, Weigle DS, Clifton DK, Steiner RA. Leptin is a metabolic gate for the onset of puberty in the female rat. Endocrinology 138: 855–858, 1997 [DOI] [PubMed] [Google Scholar]
- 4.Cheung CC, Thornton JE, Nurani SD, Clifton DK, Steiner RA. A reassessment of leptin's role in triggering the onset of puberty in the rat and mouse. Neuroendocrinology 74: 12–21, 2001 [DOI] [PubMed] [Google Scholar]
- 5.Clarkson J, Boon WC, Simpson ER, Herbison AE. Postnatal development of an estradiol-kisspeptin positive feedback mechanism implicated in puberty onset. Endocrinology 150: 3214–3220, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Clarkson J, Herbison AE. Postnatal development of kisspeptin neurons in mouse hypothalamus; sexual dimorphism and projections to gonadotropin-releasing hormone neurons. Endocrinology 147: 5817–5825, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Conte FA, Grumbach MM, Kaplan SL, Reiter EO. Correlation of luteinizing hormone-releasing factor-induced luteinizing hormone and follicle-stimulating hormone release from infancy to 19 years with the changing pattern of gonadotropin secretion in agonadal patients: relation to the restraint of puberty. J Clin Endocrinol Metab 50: 163–168, 1980 [DOI] [PubMed] [Google Scholar]
- 8.Danzer SC, Price RO, McMullen NT, Rance NE. Sex steroid modulation of neurokinin B gene expression in the arcuate nucleus of adult male rats. Brain Res Mol Brain Res 66: 200–204, 1999 [DOI] [PubMed] [Google Scholar]
- 9.de Roux N, Genin E, Carel JC, Matsuda F, Chaussain JL, Milgrom E. Hypogonadotropic hypogonadism due to loss of function of the KiSS1-derived peptide receptor GPR54. Proc Natl Acad Sci USA 100: 10972–10976, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dellovade TL, Merchenthaler I. Estrogen regulation of neurokinin B gene expression in the mouse arcuate nucleus is mediated by estrogen receptor alpha. Endocrinology 145: 736–742, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Ebling FJ. The neuroendocrine timing of puberty. Reproduction 129: 675–683, 2005 [DOI] [PubMed] [Google Scholar]
- 12.Fechner PY. Gender differences in puberty. J Adolesc Health 30: 44–48, 2002 [DOI] [PubMed] [Google Scholar]
- 13.Foster DL, Jackson LM, Padmanabhan V. Programming of GnRH feedback controls timing puberty and adult reproductive activity. Mol Cell Endocrinol 254–255: 109–119, 2006 [DOI] [PubMed] [Google Scholar]
- 14.Funes S, Hedrick JA, Vassileva G, Markowitz L, Abbondanzo S, Golovko A, Yang S, Monsma FJ, Gustafson EL. The KiSS-1 receptor GPR54 is essential for the development of the murine reproductive system. Biochem Biophys Res Commun 312: 1357–1363, 2003 [DOI] [PubMed] [Google Scholar]
- 15.Gay VL, Hauger RL. A sex-related pattern of gonadotropin secretion in the castrated rat: effects of changing the inhibitory steroid or pituitary LH content. Biol Reprod 16: 527–535, 1977 [PubMed] [Google Scholar]
- 16.Gay VL, Midgley AR., Jr Response of the adult rat to orchidectomy and ovariectomy as determined by LH radioimmunoassay. Endocrinology 84: 1359–1364, 1969 [DOI] [PubMed] [Google Scholar]
- 17.Goodman RL, Lehman MN, Smith JT, Coolen LM, de Oliveira CV, Jafarzadehshirazi MR, Pereira A, Iqbal J, Caraty A, Ciofi P, Clarke IJ. Kisspeptin neurons in the arcuate nucleus of the ewe express both dynorphin A and neurokinin B. Endocrinology 148: 5752–5760, 2007 [DOI] [PubMed] [Google Scholar]
- 18.Gottsch ML, Cunningham MJ, Smith JT, Popa SM, Acohido BV, Crowley WF, Seminara S, Clifton DK, Steiner RA. A role for kisspeptins in the regulation of gonadotropin secretion in the mouse. Endocrinology 145: 4073–4077, 2004 [DOI] [PubMed] [Google Scholar]
- 19.Grumbach MM. The neuroendocrinology of human puberty revisited. Horm Res 57, Suppl 2: 2–14, 2002 [DOI] [PubMed] [Google Scholar]
- 20.Gupta D, Rager K, Zarzycki J, Eichner M. Levels of luteinizing hormone, follicle-stimulating hormone, testosterone and dihydrotestosterone in the circulation of sexually maturing intact male rats and after orchidectomy and experimental bilateral cryptorchidism. J Endocrinol 66: 183–193, 1975 [DOI] [PubMed] [Google Scholar]
- 21.Han SK, Gottsch ML, Lee KJ, Popa SM, Smith JT, Jakawich SK, Clifton DK, Steiner RA, Herbison AE. Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuroendocrine switch for the onset of puberty. J Neurosci 25: 11349–11356, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Herbison AE. Genetics of puberty. Horm Res 68, Suppl 5: 75–79, 2007 [DOI] [PubMed] [Google Scholar]
- 23.Kauffman AS. Sexual differentiation and the Kiss1 system: hormonal and developmental considerations. Peptides 30: 83–93, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kauffman AS, Buenzle J, Fraley GS, Rissman EF. Effects of galanin-like peptide (GALP) on locomotion, reproduction, and body weight in female and male mice. Horm Behav 48: 141–151, 2005 [DOI] [PubMed] [Google Scholar]
- 25.Kauffman AS, Clifton DK, Steiner RA. Emerging ideas about kisspeptin- GPR54 signaling in the neuroendocrine regulation of reproduction. Trends Neurosci 30: 504–511, 2007 [DOI] [PubMed] [Google Scholar]
- 26.Kauffman AS, Gottsch ML, Roa J, Byquist AC, Crown A, Clifton DK, Hoffman GE, Steiner RA, Tena-Sempere M. Sexual differentiation of Kiss1 gene expression in the brain of the rat. Endocrinology 148: 1774–1783, 2007 [DOI] [PubMed] [Google Scholar]
- 27.Kauffman AS, Park JH, McPhie-Lalmansingh AA, Gottsch ML, Bodo C, Hohmann JG, Pavlova MN, Rohde AD, Clifton DK, Steiner RA, Rissman EF. The kisspeptin receptor GPR54 is required for sexual differentiation of the brain and behavior. J Neurosci 27: 8826–8835, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Krajewski SJ, Anderson MJ, Iles-Shih L, Chen KJ, Urbanski HF, Rance NE. Morphologic evidence that neurokinin B modulates gonadotropin-releasing hormone secretion via neurokinin 3 receptors in the rat median eminence. J Comp Neurol 489: 372–386, 2005 [DOI] [PubMed] [Google Scholar]
- 29.Navarro VM, Castellano JM, Fernández-Fernández R, Barreiro ML, Roa J, Sanchez-Criado JE, Aguilar E, Dieguez C, Pinilla L, Tena-Sempere M. Developmental and hormonally regulated messenger ribonucleic acid expression of KiSS-1 and its putative receptor, GPR54, in rat hypothalamus and potent luteinizing hormone-releasing activity of KiSS-1 peptide. Endocrinology 145: 4565–4574, 2004 [DOI] [PubMed] [Google Scholar]
- 30.Navarro VM, Fernández-Fernández R, Castellano JM, Roa J, Mayen A, Barreiro ML, Gaytan F, Aguilar E, Pinilla L, Dieguez C, Tena-Sempere M. Advanced vaginal opening and precocious activation of the reproductive axis by KiSS-1 peptide, the endogenous ligand of GPR54. J Physiol 561: 379–386, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Navarro VM, Gottsch ML, Chavkin C, Okamura H, Clifton DK, Steiner RA. Regulation of GnRH Secretion by Kiss1/Dynorphin/Neurokinin B Neurons in the Arcuate Nucleus of the Mouse. J Neurosci In Press [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nestor C, Holaskova I, Goodman R, Billings H. Surge-like release of LH following administration of a neurokinin-3 receptor agonist to the retrochiasmatic area of early follicular phase ewes (Abstract). Presentation No. 707, Society for the Study of Reproduction 41st Annual Meeting, Kailua-Kona, HI, 2008 [Google Scholar]
- 33.Ojeda SR, Kalra PS, McCann SM. Further studies on the maturation of the estrogen negative feedback on gonadotropin release in the female rat. Neuroendocrinology 18: 242–255, 1975 [DOI] [PubMed] [Google Scholar]
- 34.Ojeda SR, Lomniczi A, Mastronardi C, Heger S, Roth C, Parent AS, Matagne V, Mungenast AE. Minireview: the neuroendocrine regulation of puberty: is the time ripe for a systems biology approach? Endocrinology 147: 1166–1174, 2006 [DOI] [PubMed] [Google Scholar]
- 35.Ojeda SR, Skinner MK. Puberty in the rat. In: The Physiology of Reproduction (3rd ed.), edited by Neill JD. San Diego, CA: Academic-Elsevier, 2006, p. 2061–2126 [Google Scholar]
- 36.Plant TM. The male monkey as a model for the study of the neurobiology of puberty onset in man. Mol Cell Endocrinol 254–255: 97–102, 2006 [DOI] [PubMed] [Google Scholar]
- 37.Plant TM, Witchel SM. Puberty in non-human primates and humans. In: Knobil and Neill's Physiology of Reproduction (3rd ed.), edited by Neill JD: Elsevier, 2006, p. 2177–2230 [Google Scholar]
- 38.Popa SM, Clifton DK, Steiner RA. The role of kisspeptins and GPR54 in the neuroendocrine regulation of reproduction. Annu Rev Physiol 70: 213–238, 2008 [DOI] [PubMed] [Google Scholar]
- 39.Rager K, Zarzycki J, Eichner M, Gupta D. Effects of experimental bilateral cryptorchidism and castration on the plasma gonadotropins in male rats during sexual maturation. Res Exp Med (Berl) 165: 55–59, 1975 [DOI] [PubMed] [Google Scholar]
- 40.Rance NE, Bruce TR. Neurokinin B gene expression is increased in the arcuate nucleus of ovariectomized rats. Neuroendocrinology 60: 337–345, 1994 [DOI] [PubMed] [Google Scholar]
- 41.Robertson JL, Clifton DK, de la Iglesia HO, Steiner RA, Kauffman AS. Circadian regulation of Kiss1 neurons: implications for timing the preovulatory gonadotropin-releasing hormone/luteinizing hormone surge. Endocrinology 150: 3664–3671, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rometo AM, Krajewski SJ, Voytko ML, Rance NE. Hypertrophy and increased kisspeptin gene expression in the hypothalamic infundibular nucleus of postmenopausal women and ovariectomized monkeys. J Clin Endocrinol Metab 92: 2744–2750, 2007 [DOI] [PubMed] [Google Scholar]
- 43.Rometo AM, Rance NE. Changes in prodynorphin gene expression and neuronal morphology in the hypothalamus of postmenopausal women. J Neuroendocrinol 20: 1376–1381, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sandoval-Guzman T, Rance NE. Central injection of senktide, an NK3 receptor agonist, or neuropeptide Y inhibits LH secretion and induces different patterns of Fos expression in the rat hypothalamus. Brain Res 1026: 307–312, 2004 [DOI] [PubMed] [Google Scholar]
- 45.Sandoval-Guzman T, Stalcup ST, Krajewski SJ, Voytko ML, Rance NE. Effects of ovariectomy on the neuroendocrine axes regulating reproduction and energy balance in young cynomolgus macaques. J Neuroendocrinol 16: 146–153, 2004 [DOI] [PubMed] [Google Scholar]
- 46.Seminara SB, Messager S, Chatzidaki EE, Thresher RR, Acierno JS, Jr, Shagoury JK, Bo-Abbas Y, Kuohung W, Schwinof KM, Hendrick AG, Zahn D, Dixon J, Kaiser UB, Slaugenhaupt SA, Gusella JF, O'Rahilly S, Carlton MB, Crowley WF, Jr, Aparicio SA, Colledge WH. The GPR54 gene as a regulator of puberty. N Engl J Med 349: 1614–1627, 2003 [DOI] [PubMed] [Google Scholar]
- 47.Semple RK, Achermann JC, Ellery J, Farooqi IS, Karet FE, Stanhope RG, O'rahilly S, Aparicio SA. Two novel missense mutations in g protein-coupled receptor 54 in a patient with hypogonadotropic hypogonadism. J Clin Endocrinol Metab 90: 1849–1855, 2005 [DOI] [PubMed] [Google Scholar]
- 48.Shahab M, Mastronardi C, Seminara SB, Crowley WF, Ojeda SR, Plant TM. Increased hypothalamic GPR54 signaling: a potential mechanism for initiation of puberty in primates. Proc Natl Acad Sci USA 102: 2129–2134, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smith JT. Sex steroid control of hypothalamic Kiss1 expression in sheep and rodents: comparative aspects. Peptides 30: 94–102, 2009 [DOI] [PubMed] [Google Scholar]
- 50.Smith JT, Cunningham MJ, Rissman EF, Clifton DK, Steiner RA. Regulation of Kiss1 gene expression in the brain of the female mouse. Endocrinology 146: 3686–3692, 2005 [DOI] [PubMed] [Google Scholar]
- 51.Smith JT, Dungan HM, Stoll EA, Gottsch ML, Braun RE, Eacker SM, Clifton DK, Steiner RA. Differential regulation of KiSS-1 mRNA expression by sex steroids in the brain of the male mouse. Endocrinology 146: 2976–2984, 2005 [DOI] [PubMed] [Google Scholar]
- 52.Smith JT, Popa SM, Clifton DK, Hoffman GE, Steiner RA. Kiss1 neurons in the forebrain as central processors for generating the preovulatory luteinizing hormone surge. J Neurosci 26: 6687–6694, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tapper CM, Naftolin F, Brown-Grant K. Influence of the reproductive state at the time of operation on the early response to ovariectomy in the rat. J Endocrinol 53: 47–57, 1972 [DOI] [PubMed] [Google Scholar]
- 54.Todman MG, Han SK, Herbison AE. Profiling neurotransmitter receptor expression in mouse gonadotropin-releasing hormone neurons using green fluorescent protein-promoter transgenics and microarrays. Neuroscience 132: 703–712, 2005 [DOI] [PubMed] [Google Scholar]
- 55.Topaloglu AK, Reimann F, Guclu M, Yalin AS, Kotan LD, Porter KM, Serin A, Mungan NO, Cook JR, Ozbek MN, Imamoglu S, Akalin NS, Yuksel B, O'Rahilly S, Semple RK. TAC3 and TACR3 mutations in familial hypogonadotropic hypogonadism reveal a key role for Neurokinin B in the central control of reproduction. Nat Genet 41: 354–358, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]