Abstract
In our previous study, we have demonstrated that peak inspiratory pressure over birth weight (PIP/kg) and mean airway pressure over birth weight (MAP/kg) were more significant risk factors for the development of neonatal chronic lung disease (CLD) than PIP and MAP. We aimed to develop a scoring method using the modified respiratory variables (SMUMRV) to predict CLD at early postnatal period. From 1997 to 1999, a retrospective review was performed for 197 infants <1,500 g for the development of the SMUMRV based on statistical analysis. From 2000 to 2001, calculated scores on day 4, 7 and 10 of life were obtained prospectively for 107 infants <1,500 g. Predictive values and the area under the receiver operator characteristic curve (AUC) were determined and compared with the result of the previous regression model. Gestational age, birth weight, 5 min Apgar score, PIP/kg at 12 hr of age, fractional inspired oxygen (FiO2), MAP/kg, modified oxygenation index and ventilatory mode were selected as parameters of SMUMRV. No significant differences of AUCs were found between the SMUMRV and the Yoder model. It is likely that our scoring method provides reliable values for predicting the development of CLD in very low birth weight infants.
Keywords: Bronchopulmonary Dysplasia; Infant, Premature; Predictive Value of Tests; ROC Curve; Scoring Method; Epidemiologic Research Design
INTRODUCTION
Neonatal chronic lung disease (CLD) (1-3) is one of the serious sequelae of very low birth weight infants. Inspite of various therapies such as fluid restriction (4), corticosteroids (5), vitamin A (6), low positive inspiratory pressure (PIP) and permissive hypercapnea (7), high-frequency ventilation (HFV) (8) and inhaled nitrogen oxide (9) have been and are still being tried to prevent the development of neonatal CLD, there is no specific treatment for CLD once it is established, indicating the importance of early detection in decreasing the incidence of neonatal CLD.
Many regression models have been developed to predict the development of neonatal CLD (10-19), but some limitations were present; 1) most of them were investigated prior to the use of surfactant replacement or HFV, 2) logistic regression equations used were too difficult and highly complicated for bedside application, and 3) the prospective populations for validation were different from retrospective populations, suggesting problems in accuracy and reliability. Therefore a simple, modern and appropriate-population-based clinical method was needed to acquire information which patients would be at risk of neonatal CLD in the future.
In our previous study, we found that PIP over birth weight (PIP/kg) at 12 hr of age, mean airway pressure (MAP) over birth weight (MAP/kg) and modified oxygenation index [MAP/kg×a fractional inspired oxygen (FiO2)÷partial pressure of oxygen in arterial blood (PaO2)] were significant risk factors for the development of CLD than traditionally used PIP, MAP and oxygenation index (20). We, therefore, hypothesized that a scoring method using those modified respiratory parameters would be able to better predict the development of CLD than other methods using conventional respiratory variables.
The aims of this study were to develop a simple clinical scoring method for predicting neonatal CLD using modified respiratory variables, and to determine and compare the predictive values of our scoring system.
MATERIALS AND METHODS
Of the 203 infants with birth weight <1,500 g admitted to the neonatal intensive care unit (NICU) at Asan Medical Center (Seoul, Korea) between January 1997 and December 1999 (period I), 197 infants were enrolled and their medical records reviewed retrospectively. Patients with life-threatening congenital malformation (n=3) and incomplete medical records (n=3) were excluded. Recorded data were divided into 3 groups: maternal factors, neonatal factors and ventilator-associated factors. Maternal factors include maternal age, mode of delivery, duration of premature ruptured membrane, antenatal corticosteroids, white blood cell (WBC) count in amniotic fluid, placental pathology, maternal WBC count and maternal serum C-reactive protein (CRP) level. Neonatal factors include birth weight, gestational age, small for gestational age, sex, 1 min and 5 min Apgar score, patent ductus arteriosus, respiratory distress syndrome (RDS), surfactant supplementation, amount of fluid administered within 3 days of life, blood WBC count, and serum level of CRP and total immunoglobulin M. Ventilator-associated factors included FiO2, PaO2, a partial pressure of carbon dioxide in arterial blood (PaCO2), PIP/kg at 12 hr of age, MAP/kg and the modified oxygenation index. Those ventilator-associated factors were recorded on day 4, 7 and 10 of life, respectively.
All descriptive variables were categorized and scored within the limits. A range of values was selected for each variable, and points were initially awarded as -1, 0, 1, 2 and 3 for each parameter based on the measured value. Those variables categorized were entered into logistic regression analysis. Categorized variables not independently significantly associated with CLD were removed from the model. After its removal, the logistic regression was repeated until only significantly related variables remained. Based on the results of regression analysis, gestational age, FiO2 and MAP/kg were selected. Five variables including birth weight, 5 min Apgar score, PIP/kg at 12 hr of age, modified oxygenation index and ventilatory modality, based on our clinical experiences, were also adopted for development of the scoring system. All of them were statistically highly significant in univariate analysis. The total score was determined by the subtotals of the parameters in the scoring table. The primary outcome variable was CLD diagnosed at 36 weeks of corrected age (CA). Using the total score and outcome data, receiver operator characteristic (ROC) curves were developed, and predictive values and area under the ROC curve (AUC) were determined. We were unable to use maternal factors in the development of our model since data for many mothers were missing.
From January 2000 to August 2001 (period II), 107 infants with birth weight <1,500 g admitted at the same center were evaluated prospectively for cross-validation, utilizing the results of the period I patients. Prospective data in period II were collected to validate the usefulness of sensitivity, specificity, and predictive values obtained from the regression model generated from retrospective data in period I. Calculated score on day 4, 7 and 10 of life and its true outcome data were matched and plotted over ROC curve to obtain their AUCs. The results of AUC in period II were compared with those in period I.
After determination of an optimal regression model for predicting CLD, other methods for predicting CLD were compared with our model. Yoder et al. (12) developed respiratory failure score (Yoder model) for infants <32 weeks' gestation for prediction of CLD in 1999. To our knowledge, Yoder model was the first scoring method to predict the development for premature infants. We evaluated the infants on day 4, 7 and 10 days of life using the two published method.
Mechanical ventilation using modes of continuous positive end-expiratory pressure (CPAP), intermittent mandatory ventilation/synchronous intermittent mandatory ventilation (IMV/SIMV), assisted/control (A/C) and HFV was initiated based on clinical manifestations and results of blood gas analysis. Indications for CPAP included frequent recurrent apnea, chest retraction with or without grunting, and PaO2 <50 mmHg in 40% oxygen. Indications for mechanical ventilation included CPAP failure, PaCO2 >50 mmHg in 60% oxygen, and persistent respiratory acidosis (21). Nasal CPAP was also regarded as a mode of CPAP. CLD was diagnosed if the infants had prolonged oxygen requirements at 36 weeks of CA (3). HFV was applied as rescue therapy during the study periods and the MAP was adjusted to 2.0 cmH2O higher than that of a conventional mode of ventilation (8, 22). Patients without mechanical ventilation were given the lowest scores. Chest radiographic findings within 10 days of age were excluded from the analysis because we found no significant association with the development of CLD.
Data were analyzed using SPSS 10.0 software (SPSS. Inc., Chicago, IL, U.S.A.). Categorical variables were compared by using Fischer's exact test or the chi-square test, and continuous variables were compared by ANOVA, Kruskal-Wallis test or Wilcoxon rank sum test. After selecting the significant variables in the univariate analysis, they were entered into the stepwise selection method to make a predictive logistic model. Differences between ROC curves were compared using MedCalc 7.2 software (MedCalc Software, Belgium). The level of significance was set at p<0.05.
RESULTS
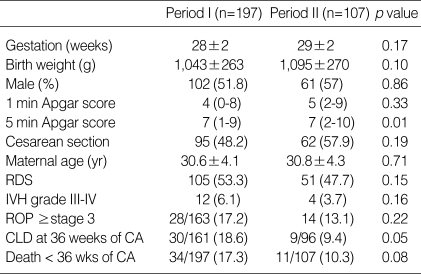
Of the 197 very low birth weight (VLBW) infants who were recruited for retrospective study during period I, 164 (83.2%) were survived to 36 weeks of CA, 87 (44.2%) were weighed ≤1,000 g, 123 (62.4%) were ≤28 weeks' gestation, and 105 (53.3%) had been treated for RDS. Of the 107 VLBW infants during period II, 96 (89.7%) survived to 36 weeks of CA, 43 (40.2%) weighed ≤1,000 g, 53 (49.5%) were ≤28 weeks' gestation, and 51 (47.7%) had been treated for RDS. Mean gestational age and birth weight (±SD) were 28.2±1.9 weeks and 1,043.3±262.6 g in period I, and 28.5±1.9 weeks and 1,095.4±270.1 g in period II, respectively. Table 1 demonstrates demographic data of the study subjects enrolled for statistical analysis during each time period. There were no significant differences except for the 5 min Apgar score in demographic data between the two periods. The incidence of CLD at 36 weeks of CA was 18.6% (30/161) in period I, and 9.4% (9/96) in period II, respectively (p=0.05).
Table 1.
Patient characteristics during each time period
Values are means±SD, median (range) or number (%).
IVH, intraventricular hemorrhage; ROP, retinopathy of prematurity.
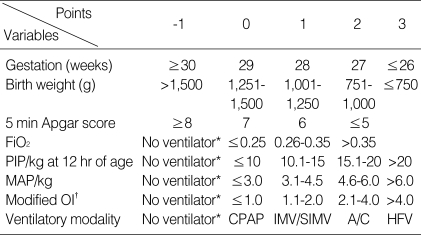
Table 2 demonstrates the selected eight variables based on the results of regression analysis and our clinical experience. Each was given a value and entered into a scoring table on day 4, 7 and 10 of life. According to the scoring table, total scores were determined and matched with the primary outcome, the incidence of CLD at 36 weeks of CA.
Table 2.
Scoring table for prediction of neonatal chronic lung disease
FiO2, fractional inspired oxygen; PIP/kg, peak inspiratory pressure divided by birth weight; MAP/kg, mean airway pressure divided by birth weight; CPAP, continuous positive airway pressure; IMV/SIMV, intermittent mandatory ventilation/synchronized intermittent mandatory ventilation; A/C, assisted and controlled mode of ventilation; OI, oxygenation index; HFV, high-frequency ventilation.
*No ventilatory therapy regardless of oxygen supplementation; †MAP is replaced by MAP/kg in calculation of oxygenation index.
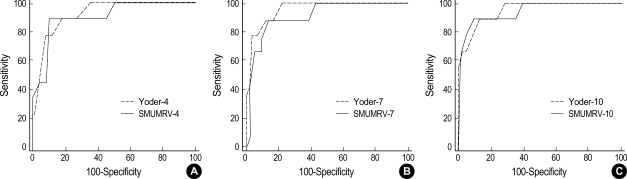
A series of ROC curves was developed to compare the predictivity of each study period. Areas under the constructed ROC curves on day 4, 7 and 10 of life were 0.76 (SEM, 0.05), 0.84 (SEM, 0.05) and 0.86 (SEM, 0.05) in period I, and 0.90 (SEM, 0.07), 0.91 (SEM, 0.07) and 0.94 (SEM, 0.05) in period II. And then, we compared the AUCs of our model (SMUMRV) with those of Yoder model on our study subjects in period II. Fig. 1 shows the comparison of ROC curves and their AUCs for each method tested. Areas under the ROC curves of Yoder model and the p values of statistical results compared with AUCs of SMUMRV were as follows; 0.92 (SEM, 0.06) on day 4 (p=0.73), 0.96 (SEM, 0.05) on day 7 (p=0.38), and 0.95 (SEM, 0.05) on day 10 (p=0.85).
Fig. 1.
Comparison of ROC curves of predictive models at day 4 (A), 7 (B) and 10 (C) of life for neonatal chronic lung disease [SMUMRV (solid line) vs. Yoder model (dotted line)]. CLD is defined at 36 weeks of corrected age. Area under the curve: (A) SMUMRV, 0.90 (SEM, 0.07); Yoder, 0.92 (SEM, 0.06); p=0.73, (B) SMUMRV, 0.91 (SEM, 0.07); Yoder, 0.96 (SEM, 0.05); p=0.38, (C) SMUMRV, 0.94 (SEM, 0.05); Yoder, 0.95 (SEM, 0.05); p=0.85.
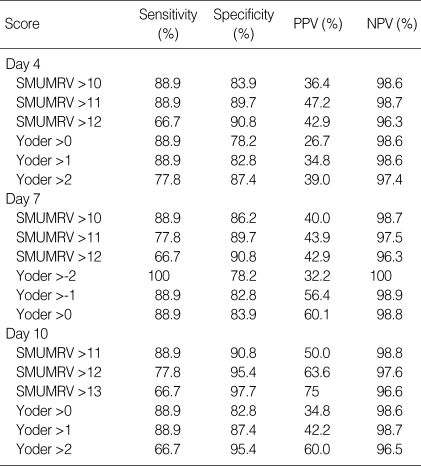
With the establishment of ROC curves, the cutoff values showing the best equilibrium between sensitivity and specificity were 4 on day 4, 1 on day 7, and 1 on day 10 in period I, and 11 on day 4, 10 on day 7, and 11 on day 10 in period II. Table 3 demonstrates the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) that were determined on day 4, 7 and 10 of life according to the specific cut-off values in SMUMRV and Yoder's model. As a single value to predict risk for CLD, only the SMUMRV value >13 on day 10 of age reached 75% with sufficient levels of sensitivity and specificity.
Table 3.
Comparison of predictive values for CLD at 36 weeks of corrected age using the scoring method
PPV, positive predictive value; NPV, negative predictive value; CLD, chronic lung disease.
DISCUSSION
In our previous study (20), we found that PIP/kg, MAP/kg were more significantly related to the development of CLD rather than conventional PIP and MAP. These findings suggest that infants who require higher inspiratory pressure over their birth weights to maintain PaO2 within normal ranges have a higher tendency to develop CLD. We also found that a significant portion of patients with CLD has been more dependent upon highly advanced mechanical ventilatory techniques (such as HFV or assist/control mode of ventilation) at their early ages of life than those without CLD. Using the modified respiratory variables, we attempted to predict the development of neonatal CLD at an early age of life by means of a simple and powerful clinical tool at the bedside.
Our scoring method for predicting neonatal CLD has some advantages over current methods. Data can be obtained by simple calculations at the bedside, not requiring complicated logistic equation. All the study subjects were admitted to the same unit with same therapeutic strategy during the study periods from data collection to cross-validation. Combination of categorized variables makes it easier to compare the severity of disease among patients who cannot be distinguished by the numerical disparities only. For example, we can compare two theoretical patients. Patient A who requires increased oxygen supplementation, from 0.3 to 0.6 of FiO2, without artificial mechanical ventilation, and patient B who is a newly set up on a mechanical ventilator with 0.3 of FiO2 and 14 mmHg of PIP. The respiratory condition of patient B is not necessarily more severe than that of patient A, because physiologic PIP will not be zero even if the patient has a severe respiratory problem and it is hard to precisely estimate the additional PIP needed to maintain the airway pressure. Therefore, more than one factor will be helpful to evaluate the progression of disease or to compare the severity in respiratory illness. Scoring method might help to meet the purpose.
Yoder et al. (12) firstly developed the respiratory failure score for infants of <32 weeks' gestation to predict neonatal CLD at 36 weeks of CA. Although the Yoder model was a simple scoring method for clinical application, the pressure amplitude of HFV was compared with the PIP of conventional ventilation, and the studies for validation were conducted at different centers having different therapeutic strategies. To overcome those drawbacks, we compared MAP of HFV with that of conventional ventilation, and validated our model prospectively at the same center in which it was developed from retrospective data. During the periods of our study, there were no significant changes in the main clinical practices, except that fluid therapy was made more restrictively since the last half of 1998.
Classification criteria of CLD for statistical analysis in our study were different from other predictive models. In attempting to compare our results with those in other studies, we found that each study used its own different analytical method for predicting the development of neonatal CLD. For example, Sinkin et al. (10) performed his study prior to surfactant supplementation and HFV, obtained prospective data for validation from different hospitals and recorded airway pressure in infants without mechanical ventilatory support. Ryan et al. (11) did not mention HFV and classified infants who died after day 4 of life into the non-CLD group, whereas Yoder et al. (12) classified infants who died after 7 days of life into the CLD group. In our study, we excluded the patients who died within 36 weeks of corrected age for the statistical analysis. When we forcibly tried to adopt the respiratory failure score (Yoder model) to our patients, we found that predictive values in both models were still remained lower than 60% even if we included our patients who died within 36 weeks of corrected age into CLD group. Those results were much different from Yoder's one (12), which showed 75% of positive predictive values at 72 hr of life. It may partly be due to discrepancy in prevalence of CLD between the two different studies. The incidence of CLD in SMUMRV was much less than in Yoder model (9.4% vs. 34%), and it could reciprocally affect the predictive values of our screening test.
We also investigated the involvement of several antenatal factors including maternal WBC counts and serum levels of CRP and total immunoglobulin M, placental pathology and WBC counts in the amniotic fluid. However, we were not able to adopt these variables into the scoring method because of incomplete medical records. We were able to obtain placental pathology data and amniocentesis data only in 61.3% and 26.4% of total subject infants, respectively during period I. Future inclusion of antenatal factors in the scoring method would be expected to show a more clear relationship between perinatal inflammation and the development of neonatal CLD.
Despite the limitations above, predictive values and the differences of ROC curves between the two models, based on the area under the curves, did not achieve statistical significance. Although these results were a little different from what we have expected, it could still likely that our scoring method using modified respiratory parameters might be a reliable tool for the early prediction of the development of neonatal CLD for infants born at <32 weeks gestation. Moreover, this method is simple enough to be easily utilized at the bedside. Future research in a larger number of study subjects should be considered to clarify the usefulness of our scoring method for predicting neonatal CLD.
References
- 1.Avery ME, Tooley WH, Keller JB, Hurd SS, Bryan MH, Cotton RB, Epstein MF, Fitzhardinge PM, Hansen CB, Hansen TN. Is chronic lung disease in low birth weight infants preventable? A survey of eight centers. Pediatrics. 1987;79:26–30. [PubMed] [Google Scholar]
- 2.Northway WH, Jr, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. N Engl J Med. 1967;276:357–368. doi: 10.1056/NEJM196702162760701. [DOI] [PubMed] [Google Scholar]
- 3.Shennan AT, Dunn MS, Ohlsson A, Lennox K, Hoskins EM. Abnormal pulmonary outcomes in premature infants: prediction from oxygen requirement in the neonatal period. Pediatrics. 1988;82:527–532. [PubMed] [Google Scholar]
- 4.Tammela OK, Koivisto ME. Fluid restriction for preventing bronchopulmonary dysplasia? Reduced fluid intake during the first weeks of life improves the outcome of low-birth-weight infants. Acta Paediatr. 1992;81:207–212. doi: 10.1111/j.1651-2227.1992.tb12205.x. [DOI] [PubMed] [Google Scholar]
- 5.Halliday HL, Ehrenkranz RA. Early postnatal (<96 hours) corticosteroids for preventing chronic lung disease in preterm infants. Cochrane Database Syst Rev. 2000;(1):CD001146. doi: 10.1002/14651858.CD001146. [DOI] [PubMed] [Google Scholar]
- 6.Shenai JP, Kennedy KA, Chytil F, Stahlman MT. Clinical trial of vitamin A supplementation in infants susceptible to bronchopulmonary dysplasia. J Pediatr. 1987;111:269–277. doi: 10.1016/s0022-3476(87)80086-0. [DOI] [PubMed] [Google Scholar]
- 7.Mariani G, Cifuentes J, Carlo WA. Randomized trial of permissive hypercapnia in preterm infants. Pediatrics. 1999;104:1082–1088. doi: 10.1542/peds.104.5.1082. [DOI] [PubMed] [Google Scholar]
- 8.HiFi Study Group. High-frequency oscillatory ventilation compared with conventional mechanical ventilation in the treatment of respiratory failure in preterm infants: assessment of pulmonary function at 9 months of corrected age. J Pediatr. 1990;116:933–941. doi: 10.1016/s0022-3476(05)80657-2. [DOI] [PubMed] [Google Scholar]
- 9.Banks BA, Seri I, Ischiropoulos H, Merrill J, Rychik J, Ballard RA. Changes in oxygenation with inhaled nitric oxide in severe bronchopulmonary dysplasia. Pediatrics. 1999;103:610–618. doi: 10.1542/peds.103.3.610. [DOI] [PubMed] [Google Scholar]
- 10.Sinkin RA, Cox C, Phelps DL. Predicting risk for bronchopulmonary dysplasia: selection criteria for clinical trials. Pediatrics. 1990;86:728–736. [PubMed] [Google Scholar]
- 11.Ryan SW, Nycyk J, Shaw BN. Prediction of chronic neonatal lung disease on day 4 of life. Eur J Pediatr. 1996;155:668–671. doi: 10.1007/BF01957150. [DOI] [PubMed] [Google Scholar]
- 12.Yoder BA, Anwar MU, Clark RH. Early prediction of neonatal chronic lung disease: a comparison of three scoring methods. Pediatr Pulmonol. 1999;27:388–394. doi: 10.1002/(sici)1099-0496(199906)27:6<388::aid-ppul5>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 13.Palta M, Gabbert D, Fryback D, Widjaja I, Peters ME, Farrell P, Johnson J. Development and validation of an index for scoring baseline respiratory disease in the very low birth weight neonate. Severity Index Development and Validation Panels and Newborn Lung Project. Pediatrics. 1990;86:714–721. [PubMed] [Google Scholar]
- 14.Goldman SL, Gerhardt T, Sonni R, Feller R, Hehre D, Tapia JL, Bancalari E. Early prediction of chronic lung disease by pulmonary function testing. J Pediatr. 1983;102:613–617. doi: 10.1016/s0022-3476(83)80202-9. [DOI] [PubMed] [Google Scholar]
- 15.Bhutani VK, Abbasi S. Relative likelihood of bronchopulmonary dysplasia based on pulmonary mechanics measured in preterm neonates during the first week of life. J Pediatr. 1992;120:605–613. doi: 10.1016/s0022-3476(05)82491-6. [DOI] [PubMed] [Google Scholar]
- 16.Corcoran JD, Patterson CC, Thomas PS, Halliday HL. Reduction in the risk of bronchopulmonary dysplasia from 1980-1990: results of a multivariate logistic regression analysis. Eur J Pediatr. 1993;152:677–681. doi: 10.1007/BF01955247. [DOI] [PubMed] [Google Scholar]
- 17.Farstad T, Bratlid D. Incidence and prediction of bronchopulmonary dysplasia in a cohort of premature infants. Acta Paediatr. 1994;83:19–24. doi: 10.1111/j.1651-2227.1994.tb12946.x. [DOI] [PubMed] [Google Scholar]
- 18.Ryan SW, Wild NJ, Arthur RJ, Shaw BN. Prediction of chronic neonatal lung disease in very low birthweight neonates using clinical and radiological variables. Arch Dis Child Fetal Neonatal Ed. 1994;71:F36–F39. doi: 10.1136/fn.71.1.f36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hentschel J, Friedel C, Maier RF, Bassir C, Obladen M. Predicting chronic lung disease in very low birthweight infants: comparison of 3 scores. J Perinat Med. 1998;26:378–383. doi: 10.1515/jpme.1998.26.5.378. [DOI] [PubMed] [Google Scholar]
- 20.Kim YD, Kim EA, Kim KS, Pi SY, Lee JJ, Park SJ. Perinatal risk factors for the development of bronchopulmonary dysplasia in premature infants less than 32 weeks' gestation. J Korean Soc Neonatal. 2001;8:78–93. [Google Scholar]
- 21.Goldsmith JP, Karotkin EH. Introduction to assisted ventilation. In: Goldsmith JP, editor. Assisted ventilation of the neonate. 3rd ed. Philadelphia: WB Saunders; 1996. pp. 1–19. [Google Scholar]
- 22.Courtney SE, Durand DJ, Asselin JM, Hudak ML, Aschner JL, Shoemaker CT. High-frequency oscillatory ventilation versus conventional mechanical ventilation for very-low-birth-weight infants. N Engl J Med. 2002;347:643–652. doi: 10.1056/NEJMoa012750. [DOI] [PubMed] [Google Scholar]