Abstract
Ageing is associated with a decline in immune function, which predisposes the elderly to a higher incidence of infections. Information on the mechanism of the age-related increase in susceptibility to Salmonella enterica serovar Typhimurium (S. Typhimurium) is limited. In particular, little is known regarding the involvement of the immune response in this age-related change. We employed streptomycin (Sm)-pretreated C57BL/6 mice to develop a mouse model that would demonstrate age-related differences in susceptibility and immune response to S. Typhimurium. In this model, old mice inoculated orally with doses of 3×108 or 1×106 c.f.u. S. Typhimurium had significantly greater S. Typhimurium colonization in the ileum, colon, Peyer's patches, spleen and liver than young mice. Old mice had significantly higher weight loss than young mice on days 1 and 2 post-infection. In response to S. Typhimurium infection, old mice failed to increase ex vivo production of IFN-γ and TNF-α in the spleen and mesenteric lymph node cells to the same degree as observed in young mice; this was associated with their inability to maintain the presence of neutrophils and macrophages at a ‘youthful’ level. These results indicate that Sm-pretreated C57BL/6 old mice are more susceptible to S. Typhimurium infection than young mice, which might be due to impaired IFN-γ and TNF-α production as well as a corresponding change in the number of neutrophils and macrophages in response to S. Typhimurium infection compared to young mice.
INTRODUCTION
Epidemiological, clinical and animal studies demonstrate that aged hosts have a higher incidence and severity of, and mortality from infections than younger adult hosts (Pinner et al., 1996; Han et al., 2000; Yoshikawa, 2000; Gay et al., 2006). Statistics from the World Health Organization demonstrate a 400-fold increase in mortality attributed to gastrointestinal infections in the elderly in comparison to the young adult population (Schmucker et al., 1996). Salmonella infection is one of the most common food-borne infections worldwide. It is estimated that 200 million to 1.3 billion cases of intestinal disease, including 3 million deaths, owing to non-typhoidal Salmonella occur each year worldwide. An estimated 1.4 million non-typhoidal Salmonella infections result in 168,000 visits to physicians, 15,000 hospitalizations and 580 deaths annually in the US (World Health Organization, 2005).
Salmonella enterica serovar Typhimurium (S. Typhimurium) is a facultative intracellular pathogen capable of surviving and replicating within macrophages (Mφ) and dendritic cells (DC). After oral infection, S. Typhimurium colonizes the intestine and invades M cells and epithelial cells, and then colonizes the Peyer's patch (PP), mesenteric lymph node (MLN) and disseminates to the spleen and liver (Jones & Falkow, 1996; Neutra et al., 1996). S. Typhimurium can evoke non-systemic enterocolitis in humans and cattle. However, in Slc11a1-deficient mice (such as C57BL/6 and BALB/c mice), S. Typhimurium causes a typhoid-like disease, characterized by rapid multiplication of bacteria in the liver and spleen, but little intestinal pathology (Santos et al., 2001). However, animal models in which S. Typhimurium produces a disease similar to that in humans are not suitable for studying the effect of age on S. Typhimurium resistance as sufficiently elderly animals in such models are not available. Barthel et al. (2003) showed that 2- and 6-week-old C57BL/6 mice that were pre-treated with streptomycin (Sm) and then exposed to 107 or 108 c.f.u. S. Typhimurium exhibited pathological changes that were similar to the intestinal inflammation caused by S. Typhimurium in humans. C57BL/6 mice are most commonly used in ageing studies, so we chose this strain to determine the suitability of an Sm-pretreated mouse model for investigating the impact of ageing on host susceptibility to S. Typhimurium infection.
It is well known that the immune response, particularly the response mediated by T cells, is impaired with ageing (Engwerda et al., 1996; Schmucker et al., 2001; Haynes et al., 2003). However, to date, there appears to be no information available regarding the effect of age on resistance to S. Typhimurium infection in a mouse model or the age-specific differences in the immune response to S. Typhimurium. This information is required to determine the underlying mechanisms of the age-related increase in susceptibility to S. Typhimurium as well as development of strategies to combat it. Thus, the objective of this study was to develop a mouse model of S. Typhimurium infection for age-related studies, which will allow us to obtain further insights into the mechanisms of age-related differences. We hypothesized that aged mice would have lower resistance to oral challenge with S. Typhimurium and that the difference in immune response between young and old mice would contribute to this reduced resistance. In the present study, we used an Sm-pretreated mouse model (Barthel et al., 2003) to characterize age-related differences in early stages of oral S. Typhimurium infection as well as assessing the immune response.
METHODS
Animals and bacterial strains.
Young adult (4–6 months) and old (22–24 months) C57BL/6 male mice were obtained from National Institute on Ageing colonies (Harlan Sprague Dawley). Animals were housed individually at a constant temperature of 23 °C with a 12 h light–dark cycle. Body weight was recorded daily. Animals were sacrificed by CO2 narcosis, followed by exsanguination at the indicated time points post-infection (p.i.). The study protocol was approved by the Animal Care and Use Committee of the Jean Mayer USDA Human Nutrition Research Center on Ageing at Tufts University.
The wild-type strain S. Typhimurium SL1344 is naturally resistant to Sm. For inoculation, S. Typhimurium was grown with aeration in Luria–Bertani (LB) broth overnight for 16 h at 37 °C with shaking. The concentration of bacteria was estimated using a spectrophotometer. Bacteria were washed, resuspended and used to infect mice. The effective doses of bacteria administered were determined by plating serial dilutions of the inoculum onto LB agar plates.
S. Typhimurium infection.
To determine age-related difference in susceptibility to S. Typhimurium, young and old mice (11 per group) were pre-administered Sm alone (uninfected animals), or pretreated with Sm and then administered S. Typhimurium by gavage as described by Barthel et al. (2003). Briefly, water and food were withdrawn 4 h before oral administration of 20 mg Sm in 150 μl sterile water. Afterwards, animals were supplied with water and food immediately. Twenty hours after Sm treatment, water and food were withdrawn again for 4 h before the mice were administered with either 3.4×108 or 1×106 c.f.u. S. Typhimurium (200 μl suspension in PBS) orally. Thereafter, drinking water and food were offered ad libitum. Mice were monitored every day. Any animal that exhibited severe clinical abnormalities, became moribund, or lost 20 % of initial body weight was sacrificed by CO2 narcosis followed by exsanguination. To determine the age-related difference in the immune response to S. Typhimurium infection, Sm-pretreated mice (14–18 per group) were infected with 1×106 c.f.u. S. Typhimurium as described above. This lower dose was chosen for assessment of the immune response as there was no statistically significant difference in colonization between the two doses.
Analysis of bacterial load in the intestinal contents, PP, MLN, spleen and liver.
For S. Typhimurium infection, mice were sacrificed by CO2 narcosis followed by exsanguination at days 1, 2 or 4 p.i. MLN, PP, spleen and liver were harvested and placed into pre-weighed tubes with 1 ml sterile PBS containing 15 % glycerol. Ileum, caecum and colon were aseptically removed and cut into pieces approximately 1 inch long. The content of each section was gently squeezed into the collection tubes using forceps. Intestinal tract content, MLN, PP, spleen, and liver were weighed and then mechanically homogenized using a Tissue Tearor apparatus (Biospec). Diluted homogenates in sterile PBS were plated on LB plates containing 200 μg Sm ml−1 to quantify S. Typhimurium as c.f.u. (g tissue)−1.
Lactoferrin assay.
Faecal samples were collected just before mice were treated with Sm, 24 h prior to S. Typhimurium infection. This time point was designated day −1 of infection. We then collected faeces 24 h after Sm treatment and before the mice were infected with S. Typhimurium. This time point was designated day 0 of infection. In addition we collected faecal samples at days 3 and 4 following S. Typhimurium infection and these time points were designated days 3 and 4 p.i., respectively. Faecal samples were put into PBS containing 1 mg deoxycholate ml−1 and protease inhibitors, and homogenized as described above. Lactoferrin levels were measured in faecal samples as described by Logsdon & Mecsas (2006). Levels of lactoferrin are expressed as ng lactoferrin (g faeces)−1.
Isolation of lymphocytes from spleen and MLN.
To characterize the immune response, spleens and MLN were aseptically removed and placed in sterile complete RPMI 1640, which consists of RPMI 1640 (BioWhittaker) medium supplemented with 25 mmol HEPES l−1 (Invitrogen Gibco), 2 mmol glutamine l−1 (Gibco), 1×105 units penicillin l−1, 100 mg Sm l−1 (Gibco) and 50 μg gentamicin ml−1 (Sigma-Aldrich). Single-cell suspensions of MLN were prepared by gently forcing the tissue through a 70 μm nylon cell strainer (BD Biosciences). Splenocytes were prepared by gently disrupting spleens between two sterile frosted-glass slides, then isolated via centrifugation (300 g). Red blood cells were lysed using Gey's reagent. Cell preparations were washed twice with complete RPMI and total viable cell number was determined by trypan blue exclusion. The cells were suspended in complete RPMI containing heat-inactivated fetal bovine serum (Gibco) at appropriate densities for different experiments (see below). All the experiments were conducted at 37 °C in an atmosphere of 5 % CO2 and 95 % humidity, unless indicated otherwise.
Determination of different cell types in spleen and MLN.
The percentages of major cell types in spleen and MLN were determined by FACS analysis using the following anti-mouse antibodies: anti-CD3 (T cells), anti-NK-1.1 (NK cells), F4/80 (anti-BM8, Mφ), anti-Ly-6G (Gr-1, neutrophils), anti-CD11c (N418, DC). All mAbs were obtained from eBioscience. For surface staining, 106 cells were stained with marker-specific fluorescein-5-isothiocyanate (FITC), phycoerythrin (Guerrant et al., 1992), allophycocyanin (APC)-conjugated antibodies, or appropriate isotype control antibodies for 30 min at room temperature. Cells were washed twice with FACS buffer (PBS containing 2 % fetal bovine serum) and resuspended in FACS buffer. All samples were analysed on a FACS Calibur (BD Biosciences) using Summit 4.0 software (DakoCytomation).
Ex vivo cytokine production.
Lymphocytes from spleen and MLN (5×106 cells per well) of control and S. Typhimurium-infected mice were cultured in 24-well culture plates (Becton Dickinson) in the presence or absence of heat-killed S. Typhimurium (HKS, 65 °C for 1 h, at 2×108 c.f.u. ml−1), 5 mg concanavalin A (ConA; Sigma) l−1 (for IFN-γ production) and 5 mg LPS (Sigma) l−1 (for TNF-α production) for 48 h. Cell-free supernatants were collected and stored at −70 °C for later analysis of cytokines by ELISA (BD PharMingen). Owing to limitation in the number of cells available from MLN, ex vivo cytokine production was not evaluated in response to HKS or medium alone.
Statistical analysis.
Colony-forming units were transformed to their log10 values prior to statistical analysis and are expressed graphically as log10[c.f.u. (g tissue)−1]. Bacterial counts less than 1 were given a value of 1 c.f.u. g−1 for inclusion in the analysis. Data which were not normally distributed (bacterial colonization and lactoferrin) were analysed for age differences by the non-parametric Kruskal–Wallis test. Immunological data were analysed for age difference by Student's t test. Significant differences were set at P<0.05.
RESULTS
Effect of age on tissue colonization following oral infection with S. Typhimurium
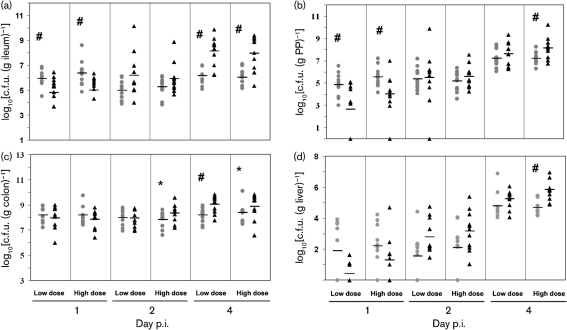
Young adult and aged mice were orally administered with a low dose of 1×106 c.f.u. and a high dose of 3.4×108 c.f.u. S. Typhimurium. Bacterial colonization of the intestinal contents from the ileum, caecum and colon, as well as in PP, MLN, spleen and liver cells were determined on days 1, 2 and 4 p.i. Except for two old mice and one young mouse infected with the high dose, all mice survived the course of infection. Overall, bacterial levels in the ileum were lower than those in the colon throughout the course of the infection. There was no significant effect of bacterial dose on bacterial colonization (Fig. 1). At day 1 p.i., young mice had 13- and 22-fold higher colonization than old mice at low and high S. Typhimurium doses in the ileum, respectively (P<0.05). However, at day 4 p.i., infected old mice had 96- and 81-fold higher levels of colonization than young mice at low and high doses in the ileum, respectively (P<0.01) (Fig. 1a), indicating that old mice had reduced ability to control the bacteria multiplying than young mice day 4 p.i. in the small intestine. Similar to the observation in the ileum, old mice infected with a high bacterial dose exhibited 9-fold higher colonization in PP cells at day 4 p.i. (P<0.05), although young mice infected with either low or high doses had significantly higher levels of mean colonization than old mice in PP cells at day 1 p.i. (P<0.05) (Fig. 1b).
Fig. 1.
Kinetic analysis of S. Typhimurium colonization in different tissues of streptomycin-pretreated young and old mice. Data represent c.f.u. (g tissue)−1 in ileum (a), PP (b), colon (c), and liver cells (d) of mice infected with low (1×106 c.f.u.) or high doses (3.4×108 c.f.u.) of S. Typhimurium for 1, 2, or 4 days. Grey circle, young mice; black triangle, old mice; level line, mean of each group; # indicates that the number of c.f.u. recovered from young mice was significantly different from c.f.u. recovered from old mice at P<0.05; * indicates a similar trend at P<0.1 by the non-parametric Kruskal–Wallis test (n=11 per group) from three independent experiments.
Consistent colonization in the colon was observed at days 1, 2 and 4 p.i. [range from 6.0 to 10.1 log(c.f.u. g−1)] except for two young mice that were infected with the low bacterial dose at day 1 p.i. As in the ileum, old mice infected with the low dose had significantly higher (7-fold) levels of colonization in the colon than young mice at day 4 p.i. (P<0.01). A similar trend was observed for the high bacterial dose at days 2 and 4 p.i., but the differences did not reach statistical significance (P=0.06 and 0.09, respectively) (Fig. 1c). No significant age-related difference was observed in bacterial colonization in the caecum (data not shown).
The level of bacterial colonization in MLN cells increased as the infection progressed in both young and old mice at either dose. However, there were no statistically significant age-related differences in the mean bacterial colonization in MLN cells at either dose or at any time point p.i. (data not shown).
The liver exhibited increased colonization as the infection progressed. At day 4 p.i., all livers exhibited colonization. The levels of colonization in old mice infected with the high bacterial dose were 14-fold higher than those of young mice (P<0.01) (Fig. 1d).
The levels of colonization in the spleen of both young and old mice increased from day 1 to day 4 p.i., and the spleen of old mice infected with the low bacterial dose exhibited 10-fold higher bacterial levels at day 2 p.i. than those of young mice (P<0.05) (data not shown).
Overall, these data indicate that young mice are colonized at higher levels in the ileum and PP cells at day 1 p.i., but these mice are more efficient at controlling bacterial multiplication than old mice as the infection progresses. Similar results were observed at day 4 p.i., with colonization of the colon and deeper tissues, such as the liver, indicating higher systemic infection in old mice compared to young mice.
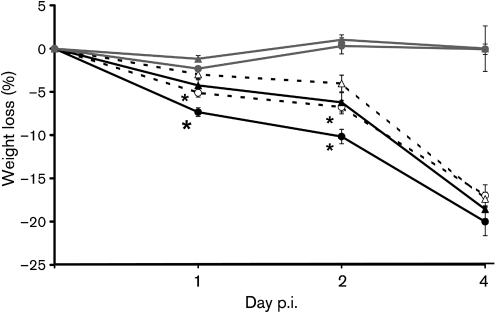
Effect of age on S. Typhimurium infection-induced weight loss
Changes in percentage body weight were monitored during the experiment to determine if the age-related difference observed in colonization is reflected in the health status of the mice. As expected, an overall decrease in percentage body weight was observed as the infection progressed. By days 2 and 4 p.i., all the infected groups had significantly greater weight loss than both uninfected young and old mice. However, at days 1 and 2 p.i., old mice infected with low or high doses of S. Typhimurium lost significantly more weight than the young mice infected with low or high doses of S. Typhimurium, respectively (P<0.05) (Fig. 2). Old mice infected with a high dose also had a significantly greater percentage weight loss than those infected with a low dose (P<0.05). In summary, old mice lost significantly more weight during the early stage of infection (on days 1 and 2 p.i.) compared to young mice infected with either dose, which is consistent with the idea that old mice could not control bacterial colonization as well as young mice.
Fig. 2.
Effect of S. Typhimurium infection on weight loss in young and old mice. Percentage weight loss in streptomycin-pretreated young and old mice infected with 3.4×108 or 1×106 c.f.u. S. Typhimurium at days 1, 2, and 4 p.i. (n=11 per group; data from three independent experiments). Blank circle, old mice infected with the low dose; blank triangle, young mice infected with the low dose; solid circle, old mice infected with the high dose; solid triangle, young mice infected with the high dose; grey circle, old uninfected mice; grey triangle, young uninfected mice; * indicates significantly greater percentage weight loss than infected young mice of the same dose group at P<0.05.
Effect of S. Typhimurium infection on faecal lactoferrin levels
Lactoferrin is abundantly expressed and secreted from glandular epithelial cells and is a prominent component of the secondary granules of neutrophils (Levay & Viljoen, 1995; Baveye et al., 1999). During intestinal inflammation, neutrophils infiltrate the mucosa, resulting in an increased concentration of lactoferrin in faeces (Guerrant et al., 1992; Kane et al., 2003). Therefore we measured the levels of faecal lactoferrin as an indicator of intestinal inflammation.
There was no significant difference in lactoferrin levels between young and old mice at baseline (24 h prior to administration of Sm). Twenty-four hours after oral administration of Sm, lactoferrin levels in both young and old uninfected (control) mice dropped significantly, with no significant age-related difference. Lactoferrin levels increased following inoculation with S. Typhimurium in both young and old mice at days 3 and 4 p.i. However, old mice infected with a low dose of S. Typhimurium had significantly higher lactoferrin levels in their faeces compared to the infected young mice at day 4 p.i. (186±1.2 ng g−1 in old vs 72±1.4 ng g−1 in young mice, P<0.05), suggesting higher inflammation induced by S. Typhimurium in old mice compared to young mice (data not shown).
Cytokine response to S. Typhimurium infection
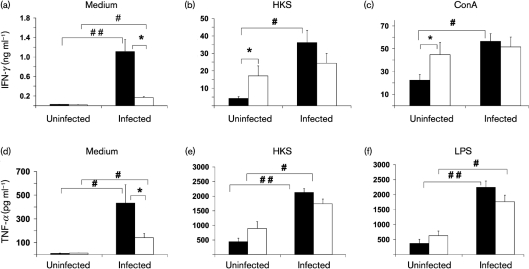
Because ageing is associated with dysregulation of the immune response, we examined the levels of IFN-γ and TNF-α in spleen and MLN cells to explore the mechanism of age-related difference in S. Typhimurium infection. The cytokines were selected as they have been shown to play an important defensive role in response to S. Typhimurium infection (Tite et al., 1991). Splenocytes and lymphocytes in MLN from uninfected or infected mice at day 4 p.i. were cultured for 48 h in the presence or absence of HKS, LPS or ConA for measurement of secreted cytokines in cultures using ELISA. Due to the limited number of lymphocytes obtained from MLN, cytokine production was only measured in response to ConA or LPS. There was no significant difference in the levels of IFN-γ from non-stimulated splenocytes between uninfected young and old mice, but splenocytes from infected old mice had significantly lower levels of non-stimulated IFN-γ production compared to splenocytes from infected young mice (Fig. 3a). While infection increased non-stimulated IFN-γ production in both young and old mice, the infection-induced increase in INF-γ production was much higher in young compared to old mice (40-fold vs 9.5-fold in young vs old mice) (Fig. 3a). Fig. 3(b, c) show ex vivo IFN-γ production of splenocytes from uninfected and infected mice in response to HKS and ConA, respectively. Uninfected old mice showed a significantly higher production of IFN-γ in response to both stimuli than uninfected young mice. However, infected old mice failed to significantly increase IFN-γ production following S. Typhimurium infection, while infected young mice demonstrated significantly greater IFN-γ production in response to HKS or ConA (1.4- and 1.1-fold higher in infected old mice compared to old uninfected mice vs 8.6- and 2.5-fold higher IFN-γ production in infected young mice compared to young uninfected mice in response to HKS and ConA, respectively). Similar results were observed in splenocyte TNF-α production in response to medium alone, HKS or LPS (Fig. 3d, e, f, respectively).
Fig. 3.
Effect of S. Typhimurium infection on ex vivo cytokine production by splenocytes of streptomycin-pretreated young and old mice. IFN-γ (a, b, c) and TNF-α (d, e, f) production by splenocytes from young and old mice, uninfected or infected with 1×106 c.f.u. S. Typhimurium at day 4 p.i. is shown. Splenocytes were stimulated with medium, HKS, ConA or LPS for 48 h. Supernatants were collected and cytokines were measured by ELISA. (a) Non-stimulated IFN-γ; (b) HKS-stimulated IFN-γ; (c) ConA-stimulated IFN-γ; (d) non-stimulated TNF-α; (e) HKS-stimulated TNF-α; (f) LPS-stimulated TNF-α. Solid bars, young mice; blank bars, old mice; * indicates significant age-related difference in uninfected or infected mice at P<0.05 using Student's t test; # and ## indicate significant differences between uninfected and infected mice in their respective age groups at P<0.05 and <0.01, respectively, using Student's t test. Data are means±sem (n=14−18 per group) from two independent experiments.
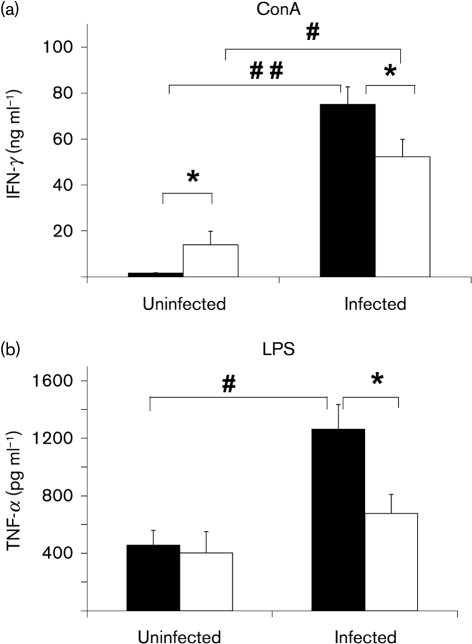
Similar to the observation in spleen tissue, MLN cells from uninfected old mice had a significantly higher level of IFN-γ compared to young mice in response to ConA (Fig. 4a). While infection increased production of both cytokines in both young and old mice, the infected old mice produced significantly lower levels of IFN-γ and TNF-α than young mice (Fig. 4a, b). In summary, old mice are significantly less capable of elevating IFN-γ and TNF-α production in their splenocytes and MLN cells compared to young mice following infection with S. Typhimurium.
Fig. 4.
Effect of S. Typhimurium infection on ex vivo cytokine production in MLN cells. IFN-γ (a) and TNF-α (b) production in MLN cells from streptomycin-pretreated young and old mice, uninfected or infected with 1×106 c.f.u. S. Typhimurium at day 4 p.i. is shown. Lymphocytes in MLN were stimulated with ConA or LPS for 48 h. Supernatants were collected and cytokines were measured by ELISA. (a) ConA-stimulated IFN-γ; (b) LPS-stimulated TNF-α. Solid bars, young mice; blank bars, old mice; * indicates significant age-related difference in uninfected or infected mice at P<0.05 using Student's t test; # and ## indicate significant differences between uninfected and infected mice in their respective age groups at P<0.05 and <0.01, respectively, using Student's t test. Data are means±sem (n=14−18 per group) from two independent experiments.
Changes in lymphoid cell populations in spleen and MLN cells in response to S. Typhimurium infection
To determine whether changes in TNF-α and IFN-γ production could reflect changes in the number of cells that produce these cytokines, we analysed cell populations in spleen and MLN. Although earlier studies have indicated that IFN-γ is primarily produced by NK and T cells in early and late stages of S. Typhimurium infection, and that TNF-α is primarily produced by Mφ (Nauciel & Espinasse-Maes, 1992; Lalmanach & Lantier, 1999), a later study indicated that dominant sources of IFN-γ and TNF-α were Mφ and neutrophils in the early stage of S. Typhimurium infection (Kirby et al., 2002). Therefore, we determined the percentage of Mφ, neutrophils, T cells, and NK cells in spleens and MLN from uninfected and infected mice (Table 1, and data not shown). Given the size variation in the spleens of old mice, which ranged from 0.04 to 0.29 g, the percentage of cells per spleen was used rather than total numbers.
Table 1.
Effect of age and S. Typhimurium infection on the percentage of spleen and MLN cell subpopulations
Streptomycin-pretreated young and old mice were administered with PBS or infected with 1×106 c.f.u. S. Typhimurium in PBS at day 4 p.i. The percentage of neutrophils and Mφ in spleen and MLN cells from uninfected and infected mice was analysed by FACS. Data are mean values (%)±sem (n=11 per group) from two independent experiments.
| Cell type | Spleen (%) | MLN (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Young | Old | Young | Old | |||||
| Uninfected | Infected | Uninfected | Infected | Uninfected | Infected | Uninfected | Infected | |
| Neutrophil | 2.8±0.5 | 4.6±0.4* | 5.4±0.9† | 6.4±0.6‡ | 2.5±0.2 | 4.2±0.4* | 9.6±1.9‡ | 11.5±1.4‡ |
| Mφ | 2.8±0.3 | 2.7±0.3 | 6.2±1.0‡ | 4.1±0.5*‡ | 1.1±0.5 | 1.4±0.3 | 2.2±0.8 | 1.1±0.2* |
*Significant difference between uninfected and infected mice in their respective age groups at P<0.05 using Student's t test.
†, ‡Significant age-related differences in uninfected or infected mice at P<0.05 and <0.01, respectively, using Student's t test.
Spleens and MLN from both uninfected and infected old mice had a significantly higher percentage of neutrophils compared to their respective groups in young mice (Table 1). However, while young mice showed a significant increase in the percentage of neutrophils in response to S. Typhimurium infection in both spleen and MLN (2.8±0.5 vs 4.6±0.4 in spleen, and 2.5±0.2 vs 4.2±0.4 in MLN of uninfected and infected mice, respectively, P<0.05), there was no significant increase in the percentage of neutrophils in spleen or MLN of old mice (5.4±0.9 vs 6.4±0.6 in spleen, and 9.6±1.9 vs 11.5±1.4 in MLN of uninfected and infected mice, respectively) (Table 1). Similar results were observed in the percentage of Mφ in spleen. Spleens of old mice had a significantly higher percentage of Mφ than those of young mice in both uninfected and infected mice (Table 1). However, while the percentage of Mφ in the spleens of young mice did not change in response to S. Typhimurium infection, spleens from old mice infected with S. Typhimurium had a significantly lower percentage of Mφ compared to uninfected mice (2.8±0.3 vs 2.7±0.3 in uninfected and infected young mice, and 6.2±1.0 vs 4.1±0.5 in uninfected and infected old mice, respectively, P<0.05). In MLN, there was no significant age-related difference in the percentage of Mφ in uninfected or infected mice (Table 1). However, while no significant change in the percentage of MLN Mφ was observed in young mice (1.1±0.5 vs 1.4±0.3 in uninfected and infected young mice, respectively), MLN of old mice infected with S. Typhimurium had a significantly lower percentage of Mφ compared to uninfected mice (2.2±0.8 vs 1.1±0.2 in uninfected and infected mice, respectively, P<0.05) (Table 1). Similar results were obtained when the total number of Mφ and neutrophils in spleen and MLN were examined (data not shown).
No significant age-related difference in the percentage of T and NK cells in spleen or MLN was observed in uninfected or infected mice, except that infected old mice had a higher percentage of NK cells in MLN than infected young mice (data not shown). These data suggest that the inability of old mice to increase or maintain the percentage of neutrophils and Mφ in response to S. Typhimurium in their immune system organs might contribute to their reduced ability to increase TNF-α and IFN-γ production in response to S. Typhimurium compared to those of young mice.
DISCUSSION
Ageing is associated with higher morbidity and mortality from S. Typhimurium infection; however, the underlying mechanism of this age-related difference is not well understood. In particular, little is known regarding the role of the immune response in this regard. This is in part due to the lack of an appropriate rodent model for which old age groups are available to study age-related differences. In this study, we employed Sm-pretreated C57BL/6 mice to develop a mouse model that demonstrates age-related differences in susceptibility to S. Typhimurium. Furthermore, we explored the underlying mechanisms as they relate to the immune response.
A normal murine intestine infected by S. Typhimurium is resistant to colonization (Santos et al., 2001), but Sm-resistant S. Typhimurium can trigger severe acute diffuse inflammation of the caecum and colon (colitis) in the Sm-pretreated murine model, which more closely resembles the symptoms observed in humans (Barthel et al., 2003). This model has been used to study S. Typhimurium infection in young mice (2 months old), and the results showed that intestinal inflammation and measurable tissue colonization at day 2 p.i. could be induced using S. Typhimurium at approximately 107 c.f.u. (Barthel et al., 2003). Based on this, we used doses of 106 and108 c.f.u. in the present study and found significant colonization at both doses, suggesting that Sm treatment increased the susceptibility to orally induced S. Typhimurium infection. Using the Sm-treated mouse model, we have demonstrated that old mice have significantly higher morbidity compared to young mice as demonstrated by higher tissue colonization and higher weight loss following S. Typhimurium infection. Thus, this animal model can be used to determine the underlying mechanisms of age-related differences in susceptibility to S. Typhimurium as well as for development of strategies to combat it.
To explore the underlying mechanism for the differences in infectivity of S. Typhimurium between young and old mice, we examined the immune response in spleen and MLN cells from uninfected or infected mice. After oral infection, the first cells encountered by S. Typhimurium are intestinal epithelial cells, DC and Mφ. Interaction with these cells leads to the synthesis of proinflammatory cytokines and chemokines resulting in an influx of neutrophils, Mφ and immature DC. IFN-γ acts in synergy with TNF-α to trigger efficient bactericidal activity of the Mφ, and connects the innate immunity with the acquired immune response mediated by T and B cells (Nauciel & Espinasse-Maes, 1992; Lalmanach & Lantier, 1999). However, there is no information available regarding cytokine production in old mice infected with S. Typhimurium. Our data showed that the levels of non-stimulated TNF-α and IFN-γ from spleen cells were not significantly different between uninfected young and old mice. However, old mice were not able to increase their ex vivo production of non-stimulated IFN-γ or TNF-α in response to S. Typhimurium infection to the same level as young mice, so that splenocytes of infected old mice showed significantly less production of IFN-γ and TNF-α compared to young mice. Similarly, old mice exhibited significantly less ability to increase ex vivo stimulated IFN-γ and TNF-α production by splenocytes and MLN cells compared to those of young mice after they were infected with S. Typhimurium. The inability of old mice to increase TNF-α and IFN-γ production in response to S. Typhimurium could contribute to the higher level of bacterial colonization in old mice. TNF-α in infected mice could enhance bactericidal activity synergistically with IFN-γ and trigger nitric oxide production (Tite et al., 1991). The lower level of TNF-α in the infected old mice could reflect the reduced ability to defend against infection, resulting in higher susceptibility of old mice to S. Typhimurium as indicated by higher bacterial colonization in many tissues. No other information on age-related changes in cytokine levels in response to S. Typhimurium is available; however, the impaired immune response to S. Typhimurium infection in aged mice in this study is consistent with observations by others of decreased T helper 1 cytokine production in old mice following Candida albicans (Murciano et al., 2006) and influenza infection (Han et al., 2000).
To determine the underlying causes of age-related reduction in TNF-α and IFN-γ, we determined age-associated alterations in immune cells known to be the producers of these cytokines. The number of immune cells can directly or indirectly influence immune function and alter the outcome of infection. Earlier reports have indicated that NK and T cells are the main producers of IFN-γ in response to S. Typhimurium infection. However, we did not observe a significant age-related difference in the percentage of T and NK cells in spleens, and infected old mice had a slightly higher percentage of NK cells in MLN than young mice, suggesting that the age-related difference in IFN-γ production was not due to changes in the percentage of NK and T cells with age. One explanation, based on a report by Kirby et al. (2002), is that in this model, the NK and T cells are not the predominant source of IFN-γ production compared to neutrophils and Mφ. This latter observation suggests that the inability of old mice to increase IFN-γ production to youthful levels following S. Typhimurium infection might be due to their inability to mount a significant change in the percentage of neutrophils and Mφ in response to this pathogen. Another possibility is that age-related functional changes, but not the change in the percentage of NK and T cells, might contribute to the inability of old mice to mount an effective IFN-γ response following S. Typhimurium infection. Further studies are needed to determine the contribution of functional changes in NK and T cells to age-related altered IFN-γ production in response to S. Typhimurium infection.
Mφ have been shown to be the predominant source of TNF-α in response to S. Typhimurium (Conlan, 1996, 1997; Vassiloyanakopoulos et al., 1998). Furthermore, a study by Kirby et al. (2002) showed that neutrophils can also contribute to TNF-α production in the early stages of S. Typhimurium infection. Age-related changes in the profile of different cells of the innate immune response have been reported, including increased monocyte/Mφ cell numbers in mice (Mocchegiani & Malavolta, 2004; Plackett et al., 2004).We have shown that both uninfected and infected old mice have a higher percentage of neutrophils and Mφ in their spleen compared to young mice. MLN of infected old mice had a significantly higher percentage of neutrophils compared to those in young mice. These age-related differences in cell population do not readily explain the higher susceptibility of old mice to S. Typhimurium infection or the lower production of TNF-α. However, on closer inspection we noted that the pattern of change in response to infection is influenced by age and might contribute to the higher susceptibility of old mice to S. Typhimurium. Of particular interest is the observation that in young mice, there is a significant increase in the percentage of neutrophils after infection, while no such increase is observed in old mice. Furthermore, while there was no change in the percentage of Mφ of young mice, old mice exhibited a significant decrease in levels of Mφ following infection. Given the important role that neutrophils and Mφ play in IFN-γ and TNF-α production in the early stages of S. Typhimurium infection, the age-related difference in the pattern of change observed in these cells following S. Typhimurium infection could be a contributing factor to the impaired IFN-γ and TNF-α production in response to S. Typhimurium infection in old mice. These cellular differences could also contribute to higher susceptibility of the old mice to S. Typhimurium through other mechanisms. The impact of ageing on Mφ functions has been well documented. For instance, the age-related deficit in Toll-like receptor (TLR) function as evidenced by reduced cytokine secretion, could be associated with the loss of TLR expression or another component of the receptor complex (Renshaw et al., 2002). However, another study failed to show age-dependent differences in cell-surface expression of TLR4 (Chelvarajan et al., 2005). Reduced phagocytosis, and nitric oxide and superoxide production with age have also been documented (De la Fuente et al., 2000; Rosenberger et al., 2004). In addition, Mφ are cellular hosts to S. Typhimurium during infection, and S. Typhimurium can induce programmed cell death in Mφ (Monack et al., 1996; Hueffer & Galán, 2004), contributing to the reduction in the number of Mφ following infection in old mice.
We observed a higher level of lactoferrin production in faeces from infected old mice compared to those from young mice. This agrees with the higher overall percentage of neutrophils in old mice compared to young mice. It is, however, interesting to note that unlike young mice, old mice did not exhibit a significant increase in the percentage of neutrophils in response to S. Typhimurium infection. This, together with lower levels of TNF-α and IFN-γ, suggests that despite the higher number of neutrophils in old mice, they are not functional and lack the ability to mount an effective response to S. Typhimurium.
In conclusion, we have demonstrated that Sm-treated C57BL/6 mice can be used as an animal model to study the effect of age on resistance to S. Typhimurium infection. Our observations demonstrated that old mice infected with S. Typhimurium had higher bacterial loading in the ileum, colon, PP, spleen and liver, as well as greater weight loss than infected young mice. The higher susceptibility of aged mice to S. Typhimurium infection could be due to their decreased ability to effectively increase their production of IFN-γ and TNF-α in response to S. Typhimurium, as well as maintaining the number of Mφ and neutrophils at a ‘youthful’ level following infection. Further work is required to more definitively determine the role of IFN-γ and TNF-α, as well as Mφ and neutrophils, in the higher susceptibility of old mice to S. Typhimurium. Furthermore, the role of the adaptive immune response in control of the late stage of S. Typhimurium infection in the context of ageing needs to be determined.
Acknowledgments
This work was supported by the US Department of Agriculture, Agriculture Research Service (58-1950-7-707) and National Institutes of Health (AI056068) to J. M. and L. L.
Abbreviations
ConA, concanavalin A
DC, dendritic cells
HKS, heat-killed Salmonella
MLN, mesenteric lymph node(s)
Mφ, macrophage(s)
p.i., post-infection
PP, Peyer's patch
S. Typhimurium, Salmonella enterica serovar Typhimurium
Sm, streptomycin
References
- Barthel, M., Hapfelmeier, S., Quintanilla-Martínez, L., Kremer, M., Rohde, M., Hogardt, M., Pfeffer, K., Rüssmann, H. & Hardt, W. D. (2003). Pretreatment of mice with streptomycin provides a Salmonella enterica serovar Typhimurium colitis model that allows analysis of both pathogen and host. Infect Immun 71, 2839–2858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baveye, S., Elass, E., Mazurier, J., Spik, G. & Legrand, D. (1999). Lactoferrin: a multifunctional glycoprotein involved in the modulation of the inflammatory process. Clin Chem Lab Med 37, 281–286. [DOI] [PubMed] [Google Scholar]
- Chelvarajan, R. L., Collins, S. M., Van Willigen, J. M. & Bondada, S. (2005). The unresponsiveness of aged mice to polysaccharide antigens is a result of a defect in macrophage function. J Leukoc Biol 77, 503–512. [DOI] [PubMed] [Google Scholar]
- Conlan, J. W. (1996). Neutrophils prevent extracellular colonization of the liver microvasculature by Salmonella typhimurium. Infect Immun 64, 1043–1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conlan, J. W. (1997). Critical roles of neutrophils in host defense against experimental systemic infections of mice by Listeria monocytogenes, Salmonella typhimurium, and Yersinia enterocolitica. Infect Immun 65, 630–635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De la Fuente, M., Medina, S., Del Rio, M., Ferrández, M. D. & Hernanz, A. (2000). Effect of aging on the modulation of macrophage functions by neuropeptides. Life Sci 67, 2125–2135. [DOI] [PubMed] [Google Scholar]
- Engwerda, C. R., Fox, B. S. & Handwerger, B. S. (1996). Cytokine production by T lymphocytes from young and aged mice. J Immunol 156, 3621–3630. [PubMed] [Google Scholar]
- Gay, R. T., Belisle, S., Beck, M. A. & Meydani, S. N. (2006). An aged host promotes the evolution of avirulent coxsackievirus into a virulent strain. Proc Natl Acad Sci U S A 103, 13825–13830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerrant, R. L., Araujo, V., Soares, E., Kotloff, K., Lima, A. A., Cooper, W. H. & Lee, A. G. (1992). Measurement of fecal lactoferrin as a marker of fecal leukocytes. J Clin Microbiol 30, 1238–1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han, S. N., Wu, D., Ha, W. K., Beharka, A., Smith, D. E., Bender, B. S. & Meydani, S. N. (2000). Vitamin E supplementation increases T helper 1 cytokine production in old mice infected with influenza virus. Immunology 100, 487–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haynes, L., Eaton, S. M., Burns, E. M., Randall, T. D. & Swain, S. L. (2003). CD4 T cell memory derived from young naive cells functions well into old age, but memory generated from aged naive cells functions poorly. Proc Natl Acad Sci U S A 100, 15053–15058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hueffer, K. & Galán, J. E. (2004). Salmonella-induced macrophage death: multiple mechanisms, different outcomes. Cell Microbiol 6, 1019–1025. [DOI] [PubMed] [Google Scholar]
- Jones, B. D. & Falkow, S. (1996). Salmonellosis: host immune responses and bacterial virulence determinants. Annu Rev Immunol 14, 533–561. [DOI] [PubMed] [Google Scholar]
- Kane, S. V., Sandborn, W. J., Rufo, P. A., Zholudev, A., Boone, J., Lyerly, D., Camilleri, M. & Hanauer, S. B. (2003). Fecal lactoferrin is a sensitive and specific marker in identifying intestinal inflammation. Am J Gastroenterol 98, 1309–1314. [DOI] [PubMed] [Google Scholar]
- Kirby, A. C., Yrlid, U. & Wick, M. J. (2002). The innate immune response differs in primary and secondary Salmonella infection. J Immunol 169, 4450–4459. [DOI] [PubMed] [Google Scholar]
- Lalmanach, A. C. & Lantier, F. (1999). Host cytokine response and resistance to Salmonella infection. Microbes Infect 1, 719–726. [DOI] [PubMed] [Google Scholar]
- Levay, P. F. & Viljoen, M. (1995). Lactoferrin: a general review. Haematologica 80, 252–267. [PubMed] [Google Scholar]
- Logsdon, L. K. & Mecsas, J. (2006). A non-invasive quantitative assay to measure murine intestinal inflammation using the neutrophil marker lactoferrin. J Immunol Methods 313, 183–190. [DOI] [PubMed] [Google Scholar]
- Mocchegiani, E. & Malavolta, M. (2004). NK and NKT cell functions in immunosenescence. Aging Cell 3, 177–184. [DOI] [PubMed] [Google Scholar]
- Monack, D. M., Raupach, B., Hromockyj, A. E. & Falkow, S. (1996). Salmonella typhimurium invasion induces apoptosis in infected macrophages. Proc Natl Acad Sci U S A 93, 9833–9838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murciano, C., Villamón, E., Yáñez, A., O'Connor, J. E., Gozalbo, D. & Gil, M. L. (2006). Impaired immune response to Candida albicans in aged mice. J Med Microbiol 55, 1649–1656. [DOI] [PubMed] [Google Scholar]
- Nauciel, C. & Espinasse-Maes, F. (1992). Role of gamma interferon and tumor necrosis factor alpha in resistance to Salmonella typhimurium infection. Infect Immun 60, 450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neutra, M. R., Frey, A. & Kraehenbuhl, J. P. (1996). Epithelial M cells: gateways for mucosal infection and immunization. Cell 86, 345–348. [DOI] [PubMed] [Google Scholar]
- Pinner, R. W., Teutsch, S. M., Simonsen, L., Klug, L. A., Graber, J. M., Clarke, M. J. & Berkelman, R. L. (1996). Trends in infectious diseases mortality in the United States. JAMA 275, 189–193. [PubMed] [Google Scholar]
- Plackett, T. P., Boehmer, E. D., Faunce, D. E. & Kovacs, E. J. (2004). Aging and innate immune cells. J Leukoc Biol 76, 291–299. [DOI] [PubMed] [Google Scholar]
- Renshaw, M., Rockwell, J., Engleman, C., Gewirtz, A., Katz, J. & Sambhara, S. (2002). Cutting edge: impaired Toll-like receptor expression and function in aging. J Immunol 169, 4697–4701. [DOI] [PubMed] [Google Scholar]
- Rosenberger, C. M., Gallo, R. L. & Finlay, B. B. (2004). Interplay between antibacterial effectors: a macrophage antimicrobial peptide impairs intracellular Salmonella replication. Proc Natl Acad Sci U S A 101, 2422–2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos, R. L., Zhang, S., Tsolis, R. M., Kingsley, R. A., Adams, L. G. & Bäumler, A. J. (2001). Animal models of Salmonella infections: enteritis versus typhoid fever. Microbes Infect 3, 1335–1344. [DOI] [PubMed] [Google Scholar]
- Schmucker, D. L., Heyworth, M. F., Owen, R. L. & Daniels, C. K. (1996). Impact of aging on gastrointestinal mucosal immunity. Dig Dis Sci 41, 1183–1193. [DOI] [PubMed] [Google Scholar]
- Schmucker, D. L., Thoreux, K. & Owen, R. L. (2001). Aging impairs intestinal immunity. Mech Ageing Dev 122, 1397–1411. [DOI] [PubMed] [Google Scholar]
- Tite, J. P., Dougan, G. & Chatfield, S. N. (1991). The involvement of tumor necrosis factor in immunity to Salmonella infection. J Immunol 147, 3161–3164. [PubMed] [Google Scholar]
- Vassiloyanakopoulos, A. P., Okamoto, S. & Fierer, J. (1998). The crucial role of polymorphonuclear leukocytes in resistance to Salmonella dublin infections in genetically susceptible and resistant mice. Proc Natl Acad Sci U S A 95, 7676–7681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization (2005). Drug-resistant Salmonella. Factsheet no. 139. www.who.int/mediacentre/factsheets/fs139/en/print.html.
- Yoshikawa, T. T. (2000). Epidemiology and unique aspects of aging and infectious diseases. Clin Infect Dis 30, 931–933. [DOI] [PubMed] [Google Scholar]