Abstract
Activation of brain nociceptin/orphanin FQ (NOP) receptors leads to attenuation of μ-opioid receptor (MOP receptor)-mediated antinociception. Buprenorphine, a high-affinity partial MOP receptor agonist also binds to NOP receptors with 80 nM affinity. The buprenorphine-induced inverted U-shaped dose-response curve for antinociception may be due to NOP receptor activation, given that, in the presence of the NOP receptor antagonist, 1-[(3R,4R)-1-cyclooctylmethyl-3-hydroxymethyl-4-piperidyl]-3-ethyl-1,3-dihydro-2H-benzimidazol-2-one (J113397), or in NOP receptor knockout mice, buprenorphine has a steeper dose-response curve and acts as a full agonist. To further explore the involvement of the direct activation of NOP receptors by buprenorphine and other compounds that activate both NOP and MOP receptors, the antinociceptive effects of 1-(1-(2,3,3α,4,5,6-hexahydro-1H-phenalen-1-yl)piperidin-4-yl)-indolin-2-one. (SR16435), 3-ethyl-1-(1-(4-isopropylcyclohexyl)piperidin-4-yl)-indolin-2-one (SR16507), buprenorphine, pentazocine, and morphine, compounds with varying levels of MOP and NOP receptor affinity and efficacy, were assessed in mice using the tail-flick assay. The ability of the selective NOP receptor antagonist (−)-cis-1-methyl-7-[[4-(2,6-dichlorophenyl)piperidin-1-yl]methyl]-6,7,8,9-tetrahydro-5H-benzocyclohepten-5-ol (SB-612111) to potentiate antinociception induced by the above compounds was examined to investigate whether activation of NOP receptors leads to attenuation of MOP receptor-mediated antinociception. SB-612111 potentiated antinociception induced by buprenorphine and the other mixed NOP/MOP receptor agonists SR16435 and SR16507. However, SB-612111 had no effect on pentazocine or morphine antinociception, two compounds with no NOP receptor-binding affinity. These results further support the hypothesis that activation of NOP receptors can lead to attenuation of MOP receptor-mediated antinociception elicited by mixed NOP/MOP receptor compounds such as buprenorphine, SR16435, and SR16507 and that, although buprenorphine has low efficacy in vitro, it has significant NOP receptor agonist activity in vivo.
Buprenorphine is a potent analgesic that is widely used both in the clinic and in the laboratory because of its high therapeutic index. In analgesia experiments in rodents, buprenorphine has been demonstrated to have a very flat, or even an inverted U-shaped dose-response curve, under appropriate experimental conditions (Cowan et al., 1977; Lutfy et al., 2003). Buprenorphine is also approved as a medication for heroin abuse. Buprenorphine's approval as an addiction medication was based on many years of preclinical experiments and clinical trials demonstrating that a daily dose of buprenorphine attenuates heroin abuse while maintaining the patient in a condition suitable for daily life (Jasinski et al., 1978). Because of its safety, buprenorphine has replaced methadone maintenance in a large number of methadone-maintained addicts. Although it is not approved for the treatment of cocaine or alcohol abuse, buprenorphine has been shown to decrease the self-administration of both of those drugs as well in rodents, non-human primates, and people (Martin et al., 1983; Mello et al., 1989; June et al., 1998). However, the exact mechanism of this action is not known.
Buprenorphine is a long-lasting, high-affinity opiate with partial agonist activity at MOP receptors (Lewis, 1985). It also has high affinity to the other opioid receptors, and is a partial agonist at δ-opioid receptors and an antagonist at κ-opioid receptors (Spagnolo et al., 2008). More recently, it was reported that buprenorphine also binds to the NOP receptors (Wnendt et al., 1999; Huang et al., 2001; Spagnolo et al., 2008). The affinity of buprenorphine for NOP receptors (Ki = 80–100 nM) is not as high as its affinity for the other opioid receptors, at which the Ki is in the low nanomolar range (Toll et al., 1998). It is thought that the activity of buprenorphine at the NOP receptor contributes to its shallow dose-response curve for antinociception.
NOP receptors, like the opioid receptors, are involved in a large number of central nervous system and peripheral actions. However, nociceptin/orphanin FQ (N/OFQ), the endogenous ligand for NOP receptors, in general does not have actions similar to the opioid peptides. In fact, when delivered intracerebroventricularly, N/OFQ is not analgesic, it blocks opioid analgesic activity (Meunier et al., 1995; Reinscheid et al., 1995), and it is not rewarding (Devine et al., 1996). Furthermore, N/OFQ blocks the reward induced by opiates and many other drugs of abuse (Murphy et al., 1999; Kotlinska et al., 2003; Ciccocioppo et al., 2004; Sakoori and Murphy, 2004).
Despite its moderate to low affinity at NOP receptors, there are reports of agonist or partial agonist activity of buprenorphine at NOP receptors, with EC50 values ranging from 8 to 100 nM, in a variety of in vitro activity assays (Wnendt et al., 1999; Hashimoto et al., 2000; Huang et al., 2001; Lutfy et al., 2003). However, other reports have shown a lack of any intrinsic activity for buprenorphine at NOP receptor (Lester and Traynor, 2006; Spagnolo et al., 2008). Based on the agonist activity of buprenorphine at NOP receptors, Lutfy et al. (2003) hypothesized that the NOP receptor agonist activity of buprenorphine attenuated its own antinociceptive activity, and this was the reason for the partial agonist activity of buprenorphine and the inverted U-shaped dose-response curve in the tail-flick test. Experiments using NOP receptor knockout mice, and a selective NOP receptor antagonist, J113397, demonstrated full agonist activity for buprenorphine, in the tail-flick test, under conditions when NOP receptor agonist activity would be absent, supporting their hypothesis (Lutfy et al., 2003).
Certainly, the most straightforward explanation for the high-dose activity of buprenorphine is that it is activating NOP receptors to attenuate its MOP receptor-mediated antinociceptive activity. However, there are reasons to question this hypothesis. First, the affinity of buprenorphine for NOP receptors is far less than its affinity for any of the opioid receptors. Nevertheless, in the tail-flick experiments, its activity was potentiated by the NOP receptor antagonist J113397, even at the lowest doses of buprenorphine (Lutfy et al., 2003). Second, it is a weak partial agonist, at best, at NOP receptor in most in vitro assays. It is conceivable that other properties could explain the behavior discussed above. To further test the hypothesis that buprenorphine acts at NOP receptors to attenuate it own antinociceptive activity, we conducted experiments by use of agonists with varying affinities and efficacies at NOP and MOP receptors, and determined whether their antinociceptive activity could be potentiated by the selective high-affinity NOP receptor antagonist SB-612111 (Zaratin et al., 2004).
Materials and Methods
Cell Culture
All receptors were in CHO cells transfected with human receptor cDNA. The cells were grown in Dulbecco's modified Eagle's medium with 10% fetal bovine serum, in the presence of 0.4 mg/ml G418 and 0.1% penicillin/streptomycin, in 100-mm plastic culture dishes. For binding assays, the cells were scraped off the plate at confluence.
Receptor Binding
Binding to cell membranes was conducted in a 96-well format, as described previously (Dooley et al., 1997). Cells were removed from the plates by scraping with a rubber policeman, homogenized in 50 mM, Tris pH 7.5, with use of a Polytron homogenizer (Kinematica, Littau-Lucerne, Switzerland), then centrifuged once, and washed by an additional centrifugation at 27,000g for 15 min. The pellet was resuspended in Tris, and the suspension incubated with [3H]DAMGO (51 Ci/mmol, 1.6 nM) or [3H]N/OFQ (120 Ci/mmol, 0.2 nM) for binding to MOP receptor and NOP receptor, respectively. Nonspecific binding was determined with 1 μM unlabeled DAMGO and N/OFQ, respectively. Total volume of incubation was 1.0 ml, and samples were incubated for 60 min at 25°C. The amount of protein in the binding assay was 15 μg. The reaction was terminated by filtration by use of a Tomtec 96 harvester (Tomtec, Orange, CT) through glass fiber filters. Bound radioactivity was counted on a Pharmacia Biotech β-plate liquid scintillation counter (PerkinElmer Life and Analytical Sciences, Waltham, MA) and expressed in counts per minute. IC50values were determined by use of at least six concentrations of each compound and calculated with Prism (GraphPad Software, Inc., La Jolla, CA). Ki values were determined by the method of Cheng and Prusoff (1973).
[35S]GTPγS Binding
[35S]GTPγS binding was conducted basically as described previously (Toll et al., 1998). Cells were scraped from tissue culture dishes into 20 mM HEPES, 1 mM EDTA, then centrifuged at 500g for 10 min. Cells were resuspended in this buffer and homogenized by use of a Polytron homogenizer (Kinematica). The homogenate was centrifuged at 27,000g for 15 min, and the pellet was resuspended in buffer A, containing: 20 mM HEPES, 10 mM MgCl2, 100 mM NaCl, pH 7.4. The suspension was recentrifuged at 27,000g and suspended once more in buffer A. For the binding assay, membranes (8–15 μg of protein) were incubated with [35S]GTPγS (50 pM), GDP (10 μM), and the appropriate compound, in a total volume of 1.0 ml, for 60 min at 25°C. Samples were filtered over glass fiber filters and counted as described for the binding assays. Percentage of stimulation was determined, in each experiment, with respect to the full agonist N/OFQ or DAMGO. Statistical analysis was conducted by use of the program Prism.
Assessing Acute Thermal Nociception
Animals.
Male ICR mice weighing 25 to 30g at the start of the experiment were used. Animals were group-housed under standard laboratory conditions and were kept on a 12:12-h day/night cycle (lights on at 7:00 AM). Animals were handled for 3 to 4 days before the experiments were conducted. On the test day, animals were transported to the testing room and acclimated to the environment for 1 h. Mice were maintained in accordance with the guidelines of SRI International and of the Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research (National Research Council, 2003).
Tail-Flick Assay.
Acute nociception was assessed by use of the tail-flick assay with an analgesia instrument (Stoelting, Wood Dale, IL) that uses radiant heat. This instrument is equipped with an automatic quantification of tail-flick latency, and a 15-s cutoff to prevent damage to the animal's tail. During testing, the focused beam of light was applied to the lower half of the animal's tail, and tail-flick latency was recorded. Baseline values for tail-flick latency were determined before drug administration in each animal. The mean basal tail-flick latency was 5.03 ± 0.3 S.E.M.
After baseline measures, animals received a subcutaneous injection of their assigned dose of drug and were tested for tail-flick latencies at 10-, 30-, and 60-min after injection. Controls received an injection of vehicle before testing.
Drug Regimen.
Animals (n = 8–14/group) received subcutaneous injections of buprenorphine (0.3–3.0 mg/kg), morphine (1.0–10.0 mg/kg), pentazocine (3.0–30.0 mg/kg), SR16435 (3.0–30.0 mg/kg), SR16507 (0.3–1.0 mg/kg), or SB-612111 (3 or 10 mg/kg). After assessment of baseline values for tail-flick latency, animals received an injection of SB-612111 (3 or 10 mg/kg) or vehicle, and 10 min later they received an injection of buprenorphine, morphine, pentazocine, SR16435, or SR16507. Testing occurred at 10, 30, and 60min after the final drug injection. A group of animals also served as vehicle controls.
Statistical Analyses.
Antinociception (percentage of maximum possible effect, %MPE) was quantified by the following formula: %MPE = 100 * [(test latency − baseline latency)/(15 − baseline latency)]. If the animal did not respond before the 15-s cutoff, the animal was assigned a score of 100%. Behavioral results were analyzed by use of repeated-measures ANOVAs with drug treatment as between-group variables and post–drug-injection time (10, 30, and 60 min) as the repeated measure followed by Student Newman-Keuls post hoc tests where appropriate. The level of significance was set at P < 0.05.
Results
In Vitro Findings.
Binding affinity and in vitro functional activity were determined for the prototypical MOP receptor agonist morphine (which does not bind to NOP receptors), the nonselective MOP receptor partial agonists buprenorphine and pentazocine, and the mixed MOP/NOP receptor compounds SR16435 and SR16507. As seen in Table 1, each of the compounds tested has high affinity for the MOP receptor, whereas only SR16435 and SR16507 have high affinity for the NOP receptor. Buprenorphine has moderate affinity for the NOP receptor (80 nM), whereas neither morphine nor pentazocine has any affinity for NOP receptors, even when tested up to 10 μM.
TABLE 1.
Binding affinity and in vitro functional activity at NOP and MOP receptors
Binding was conducted as described in Materials and Methods. Values shown represent mean ± S.D. of at least two experiments conducted in triplicate.
| Receptor Binding Ki |
[35S]GTPγS NOPr |
[35S]GTPγS MOPr |
||||
|---|---|---|---|---|---|---|
| NOPr | MOPr | EC50 | Stim | EC50 | Stim | |
| nM | nM | % | nM | % | ||
| SR 16435 | 8.0 ± 7.5 | 2.7 ± 0.5 | 28.7 ± 0.6 | 45.0 ± 5 | 29.5 ± 10.3 | 30 ± 0.2 |
| SR16507 | 5.22 ± 0.65 | 1.07 ± 0.17 | 8.5 ± 0.8 | 95 ± 12 | 5.2 ± 1.6 | 47 ± 1.5 |
| Buprenorphine | 77.4 ± 16 | 1.5 ± 0.8 | 251.2 ± 94.0 | 15.5 ± 5.8 | 10.2 ± 2.21 | 28.7 ± 1.05 |
| Morphine | >10,000 | 1.1 ± 0.1 | 0 | 15.6 ± 0.5 | 93 ± 2.8 | |
| Pentazocine | >10,000 | 3.9 ± 0.7 | 0 | 39.5 ± 0.25 | 20.2 ± 3.4 | |
| SB-612111 | 1.42 ± 0.12 | 674 ± 43.9 | 0 | 0 | ||
NOPr, NOP receptor; MOPr, MOP receptor; Stim, stimulation.
[35S]GTPγS binding studies, in most respects, were consistent with published data. Buprenorphine is a weak partial agonist at the MOP receptor. However, in our CHO cells buprenorphine's ability to stimulate [35S]GTPγS binding at NOP receptor varied from 0 to 17% stimulation in membrane preparations. We have concluded that, in our cells that overexpress NOP receptors, buprenorphine is an extremely weak partial agonist. At MOP receptors, the binding and functional results of SR16435 were similar to those of buprenorphine. At NOP receptors, SR16435 was more potent and had higher efficacy than buprenorphine. SR16507 is a very potent partial agonist at MOP receptors, and a potent full agonist at NOP receptors. In our CHO cells, similar to previous reports, morphine is a full MOP receptor-agonist whereas pentazocine is a partial MOP receptor agonist. Neither of these compounds demonstrated stimulation of [35S]GTPγS binding at NOP receptors.
Antinociceptive Effects of Buprenorphine Alone or Coadministered with SB-612111.
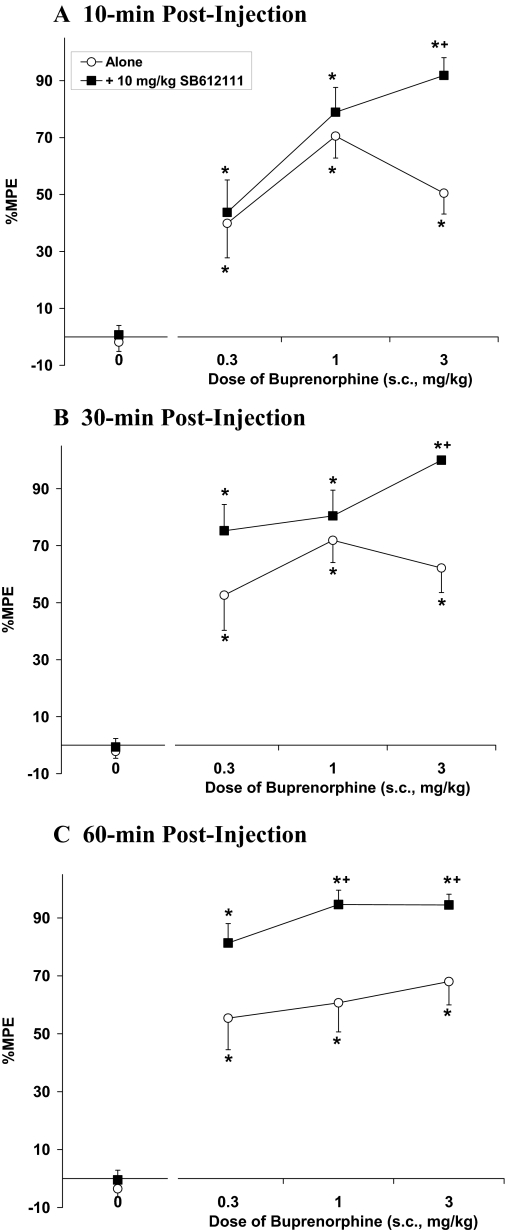
Acute antinociceptive activity of these compounds was measured in the tail-flick assay using ICR mice. As reported previously (Lutfy et al., 2003), the antinociceptive effect of buprenorphine displays a shallow dose-response curve and never attains maximal levels (Fig. 1). Antinociceptive activity begins at 0.1 mg/kg (data not shown; 25% MPE at 30-min; 44% MPE at 60-min after injection). The ANOVA indicated a main effect of buprenorphine [F(3,37) = 14.80, P < 0.05], such that 0.3 to 3.0 mg/kg doses produced a significant increase in tail-flick latency relative to controls at the three post–drug-injection times (10, 30, and 60 min). When animals were pretreated with the NOP receptor antagonist SB-612111, the 3 mg/kg dose of SB-612111 showed a very small potentiation of antinociceptive activity of buprenorphine, and therefore, only the 10 mg/kg data are presented. With use of 10 mg/kg SB-612111, the overall ANOVA indicated a main effect of SB-612111 [F(1,69) = 15.1, P < 0.05] as well as buprenorphine [F(3,69) = 60.6, P < 0.05] such that the effects of buprenorphine were potentiated by SB-612111, although SB-612111 had no effect of its own on tail-flick latency. Specifically, animals that received SB-612111 and 3 mg/kg buprenorphine exhibited an increase in tail-flick latency relative to animals that received buprenorphine alone at 10-, 30-, and 60-min postinjection time points (P < 0.05). At the 60-min time point the 1 mg/kg dose of buprenorphine coadministered with SB-612111 also produced an increase in tail-flick latency relative to buprenorphine alone.
Fig. 1.
Acute thermal antinociceptive effect of buprenorphine alone or coadministered with SB-612111 by use of the tail-flick assay at 10 min (A), 30 min (B), and 60 min (C) after injection. Data are mean %MPE (± S.E.M.). An asterisk (*) represents a significant difference from their respective vehicle controls, whereas a plus sign (+) represents a significant difference from buprenorphine alone (P < 0.05).
Antinociceptive Effects of Mixed NOP/MOP Receptor Agonists SR16435 and SR16507 Alone or Coadministered with SB-612111.
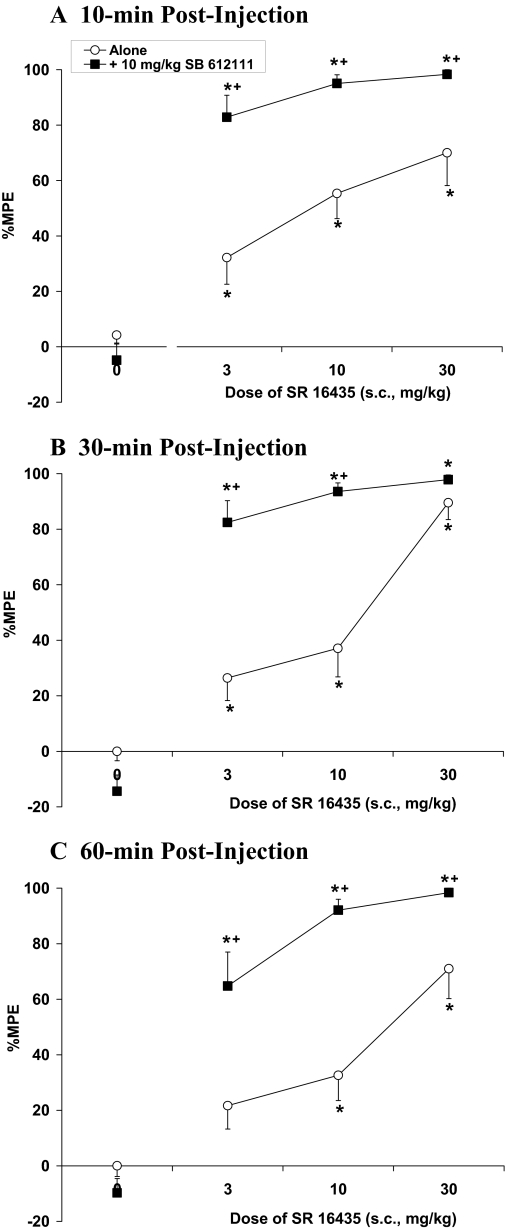
The bifunctional NOP receptor/MOP receptor partial agonist SR16435 also demonstrated antinociceptive activity ([F(3,46) = 15.1, P < 0.05]). The 10 and 30 mg/kg doses of SR16435 produced an increase in tail-flick latency 10, 30, and 60 min after injection (Fig. 2), whereas the 3 mg/kg produced a significant increase in %MPE at the earlier time points of 10 and 30 min after injection (P < 0.05). Unlike buprenorphine, SR16435 had a steep dose-response curve and approached maximal inhibition of tail-flick (15-s cutoff) at 30 mg/kg SR16435. In the presence of 10 mg/kg SB-612111, the overall ANOVA indicated a significant interaction effect [F(3,90) = 9.3, P < 0.05]. SB-612111 significantly potentiated the tail-flick latency of 3 to 10 mg/kg SR16435 at all of the time points tested, whereas with the 30 mg/kg dose %MPE was potentiated at 10 and 60 min after injection, because at 30 min, SR16435%MPE was close to the ceiling effect of 100% in the absence of the NOP receptor antagonist and could not be greatly potentiated in its presence.
Fig. 2.
Acute thermal antinociceptive effect of mixed NOP receptor/MOP receptor partial agonist SR16435 alone or coadministered with SB-612111 by use of the tail-flick assay at 10 min (A), 30 min (B), and 60 min (C) after injection. Data are mean %MPE (± S.E.M.). An asterisk (*) represents a significant difference from their respective vehicle controls, whereas a plus sign (+) represents a significant difference from SR16435 alone (P < 0.05).
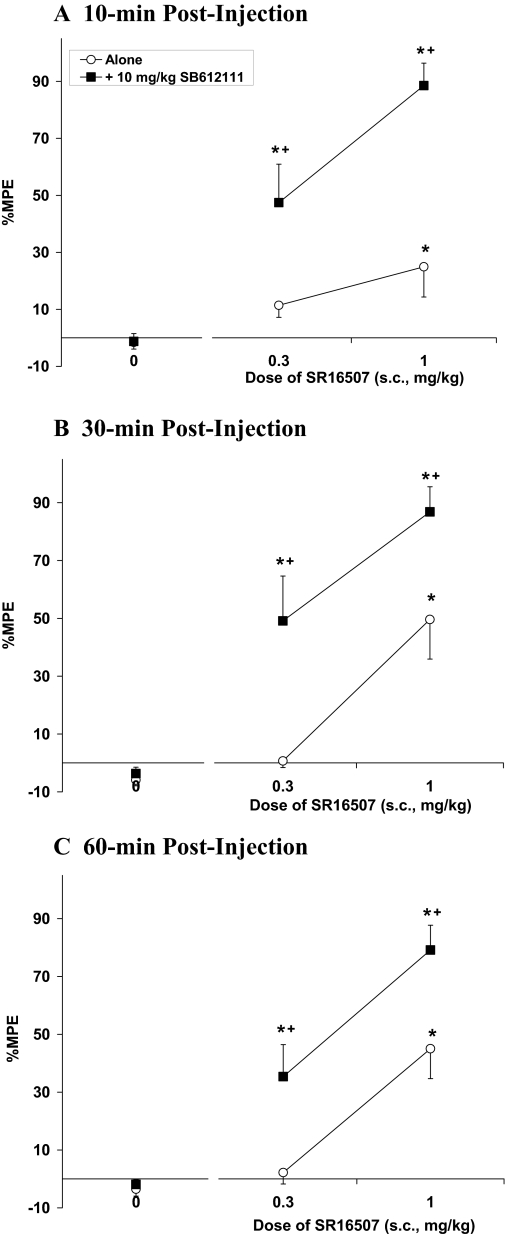
The mixed NOP receptor full agonist/MOP receptor partial agonist SR16507 also produced an increase in tail-flick latency relative to controls [F(2,22) = 15.8, P < 0.05], where the 1 mg/kg dose produced an increase in tail-flick latency at 10, 30, and 60 min after injection (Fig. 3) and the 0.3 mg/kg dose alone was ineffective. SR16507 did not produce maximal effects possibly because of the increase in efficacy at NOP receptor. When SB-612111 was given before SR16507, the ANOVA indicated a significant interaction [F(2,43) = 7.0, P < 0.05], such that SB-612111 potentiated the antinociceptive activity of SR16507. SB-612111 not only potentiated the effects of 1.0 mg/kg SR16507, which alone produced an increase in antinociception, but also produced an increase in tail-flick latency when coadministered with the 0.3 mg/kg dose that alone was ineffective. Because the antinociceptive activity of both SR16435 and SR16507 is blocked by naloxone (Khroyan et al., 2007; Toll et al., submitted), these studies indicate that the analgesic activity of these mixed NOP/MOP receptor agonists is mediated by the MOP receptor, and that NOP receptor agonist activity attenuates the analgesic activity.
Fig. 3.
Acute thermal antinociceptive effect of mixed NOP receptor/MOP receptor full agonist SR16507 alone or coadministered with SB-612111 by use of the tail-flick assay at 10 min (A), 30 min (B), and 60 min (C) after injection. Data are mean %MPE (± S.E.M.). An asterisk (*) represents a significant difference from their respective vehicle controls, whereas a plus sign (+) represents a significant difference from SR16507 alone (P < 0.05).
Antinociceptive Effects of MOP Receptor Agonists Pentazocine and Morphine Alone or Coadministered with SB-612111.
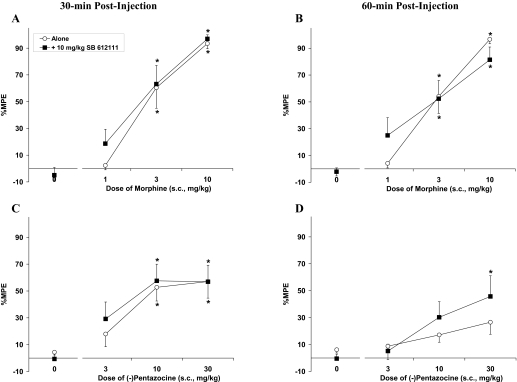
Morphine, has a steep dose-response curve [F(3,39) = 62.5, P < 0.05] with nearly 100% MPE at 10 mg/kg (Fig. 4, A and B), whereas the MOP receptor partial agonist pentazocine demonstrates a very shallow dose-response curve [F(3,31) = 6.7, P < 0.05], never approaching 100% MPE even up to 30 mg/kg (Fig. 4, C and D). Unlike buprenorphine, SR16435, and SR16507, which bind to both MOP receptor and NOP receptor, morphine and pentazocine are selective for MOP receptor. Probably because of the lack of NOP receptor agonist activity of morphine and pentazocine, SB-612111 was unable to potentiate the antinociceptive activity of morphine and only potentiated pentazocine at a single dose (30 mg/kg) and at only the 60 min time point.
Fig. 4.
Acute thermal antinociceptive effect of MOP receptor full agonist morphine (A, B) and partial agonist pentazocine (C, D) alone or coadministered with SB-612111 using the tail-flick assay at 30 and 60 min after injection. Data are mean %MPE (± S.E.M.). An asterisk (*) represents a significant difference from their respective vehicle controls (P < 0.05).
Discussion
There is considerable evidence that N/OFQ and the NOP receptor are involved in a multitude of opioid actions. N/OFQ, given intracerebroventricularly, has been shown to block morphine analgesia (Mogil et al., 1996); however, it has antinociceptive activities when administered intrathecally (Tian et al., 1997). Although selective peptide antagonists have antinociceptive activity when given intracerebroventricularly (Calo et al., 2000, 2005), selective small-molecule antagonists are not analgesic (Ozaki et al., 2000; Zaratin et al., 2004). The analgesic action of small-molecule agonists, when given systemically, is also unclear. Initial publications suggested that the selective agonist Ro 64-6198 is not analgesic, and attenuates morphine analgesia (Jenck et al., 2000). However, recent studies have shown that Ro 64-6198 has antinociceptive activity in mice using the hotplate but not the tail-flick test (Reiss et al., 2008), and in monkeys when measuring tail withdrawal (Ko et al., 2009). In both of these latter experiments, the antinociceptive activity of Ro 64-6198 was not naloxone reversible. Clearly, the actions of the NOP receptor system on acute pain are still not well understood.
Recently, it was discovered that the opioid partial agonist buprenorphine has affinity for the NOP receptor. In vitro, the affinity of buprenorphine for NOP receptors is considerably lower than for the opioid receptors (see Table 1). In addition, in functional assays, buprenorphine ranges from having no agonist efficacy for stimulation of [35S]GTPγS binding to dog brain membranes or transfected cells (Lester and Traynor, 2006; Spagnolo et al., 2008), partial agonist activity for stimulation of [35S]GTPγS binding in transfected cells (Huang et al., 2001), to being a full agonist for stimulation of mitogen-activated protein kinase (Lutfy et al., 2003) or when using the NOP reporter gene assay in transfected cells (Wnendt et al., 1999). Because the efficacy of a partial agonist depends on the receptor reserve and the particular assay being performed, it is possible that buprenorphine could display NOP receptor agonist activity in vivo.
In this regard, Lutfy et al. (2003) have demonstrated that the antinociceptive activity of buprenorphine in the tail-flick assay was potentiated by the NOP receptor antagonist J113397, and at higher heat intensity, the inverted U-shaped dose-response curve for buprenorphine straightened out in the presence of the antagonist or in NOP receptor KO mice (Lutfy et al., 2003). These results suggest that the shallowness of the dose-response curve is, at least to some extent, due to NOP receptor agonist activity attenuating the MOP receptor-mediated antinociceptive activity of buprenorphine.
In addition to modulating opiate analgesic activity, N/OFQ modulates the reward induced by a variety of drugs of abuse (Murphy et al., 1999; Kotlinska et al., 2003; Sakoori and Murphy, 2004), and the small-molecule NOP receptor agonists Ro 64-6198 (Shoblock et al., 2005) and SR16835 (Toll et al., 2009) both attenuate morphine place preference. Ciccocioppo and colleagues demonstrated that buprenorphine increased alcohol consumption at low doses, but attenuated alcohol consumption at higher nonsedating doses in Sardinian alcohol-preferring rats (Ciccocioppo et al., 2007). The reduction in alcohol consumption at high buprenorphine doses was blocked by the NOP antagonist UFP-101. It was concluded that buprenorphine acts through MOP receptors at low doses, but at higher doses NOP receptor activity counteracts the opioid-mediated effects, resulting in the overall attenuation of alcohol consumption.
Although it is possible that the NOP receptor component of buprenorphine results in a shallow dose-response curve (for antinociception) or a decrease in reward (decrease in alcohol intake), there were several reasons why this seemed unlikely. Buprenorphine is a potent opiate, having antinociceptive activity at less than 0.3 mg/kg. Its affinity for NOP receptors is only approximately 80 to 100 nM. Furthermore, in most paradigms, buprenorphine is only a partial NOP receptor agonist, at best, even in CHO cells that overexpress NOP receptors. In addition, other opioid partial agonists, such as pentazocine, that do not bind NOP receptor also have very shallow or inverted U-shaped dose-response curves (Cowan et al., 1977). Finally, other MOP/NOP receptor agonists, such as SR16435, that have a considerably greater NOP receptor component have potent analgesic activity (Khroyan et al., 2007). Thus, although it is plausible that the NOP receptor system plays a role in attenuating opioid-mediated actions of buprenorphine, this attenuation could be caused by some other phenomenon, such as inhibition of endogenous N/OFQ, rather than direct interaction of buprenorphine with the NOP receptor.
To resolve this issue, we determined the in vitro and in vivo actions of five compounds: morphine, a high-efficacy MOP receptor agonist with no affinity for NOP receptors; pentazocine, a MOP receptor partial agonist with no affinity for NOP receptors; buprenorphine, a MOP receptor partial agonist with low affinity for NOP receptors; SR16435, a high-affinity partial agonist at MOP and NOP receptors; and SR16507, a full NOP receptor agonist and partial MOP receptor agonist with high affinity for both receptors. The results indicated that compounds with any measurable NOP receptor activity and affinity, including buprenorphine, SR16435, and SR16507, produced acute antinociception by use of the tail-flick assay that was potentiated by the NOP receptor antagonist SB-612111. Potentiation was greatest for SR16507, which is a potent full agonist at the NOP receptor followed by SR16435, which also has a high affinity for NOP receptors. This is consistent with these compounds having a greater NOP receptor component. However, pentazocine and morphine, which do not bind to NOP receptors, produced antinociception that was not potentiated by SB-612111. These results are consistent with NOP receptor agonist activity within a molecule being required for analgesic activity to be potentiated by a NOP receptor antagonist, and greatly strengthen the argument that NOP receptor agonist activity of buprenorphine mediates a decrease in antinociceptive activity and alcohol consumption.
The fact that SB-612111 and J113397 do not have antinociceptive activity on their own, or even potentiate morphine, is important and suggests that there is no significant NOP receptor tone with respect to acute radiant heat in normal mice. This finding is in contrast to what has been reported with the NOP receptor antagonist JTC-801. JTC-801 is a less selective NOP receptor antagonist, but has antinociceptive activity in both acute and chronic pain models that is not naloxone reversible (Yamada et al., 2002; Suyama et al., 2003). The fact that the other, more selective antagonists do not act as analgesics suggests that JTC-801 may be acting through some other mechanism.
Buprenorphine is a unique compound. In humans it is a very potent long-lasting analgesic that is useful after surgery. Nevertheless, buprenorphine is safer than most opiate analgesics because it has low opioid efficacy resulting in reduced respiratory depression, and has reduced addiction liability (Jasinski et al., 1978; Lewis, 1985). These properties also make it useful for opioid maintenance therapy (Mello and Mendelson, 1980). In addition to these beneficial actions, buprenorphine has been known for many years to attenuate cocaine use (Schottenfeld et al., 1993), and to attenuate alcohol consumption (Mannelli et al., 1993). The mechanism by which it attenuates consumption of abused drugs may very well involve activation of NOP receptors. It is not clear, however, how a compound with such weak NOP receptor agonist activity can be acting through this receptor, in particular, because full agonist activity seems to be required to attenuate morphine conditioned place preference (CPP) by SR16507 and SR16835, two compounds with much higher affinity for the NOP receptor (Toll et al., 2009). The binding affinity of buprenorphine and the potency for stimulation of [35S]GTPγS binding through the MOP receptors are also not particularly impressive, but none-the-less it is a very potent analgesic. It is possible that buprenorphine maintains unusually high in vivo activity at whichever receptor it is activating, because of its lipophilicity and slow dissociation rate (Lewis, 1985). In addition, receptors involved in analgesia might have a very high receptor reserve allowing weak partial agonists, such as buprenorphine, to possess significant agonist activity, whether it be antinociception through MOP receptors or inhibition of antinociception through NOP receptors. Another possibility to explain buprenorphine's NOP receptor-mediated activity is the presence of MOP/NOP heterodimers. Such dimers have been demonstrated to attenuate MOP receptor-mediated activity in transfected HEK cells (Wang et al., 2005). Such a mechanism may also exist in vivo to attenuate buprenorphine's antinociceptive activity.
These experiments also suggest that modulation of the balance of affinity and efficacy of mixed MOP/NOP receptor compounds, such as buprenorphine, SR16435, and SR16507 could lead to the development of novel compounds that may have clinical use as both analgesics and drug abuse medications. Apparently, NOP receptor agonist activity can attenuate the MOP receptor-mediated analgesic activity, but not completely block it. In addition, the NOP receptor agonist activity can attenuate the reward induced by the MOP receptor component. SR16435 is an analgesic, but it induces a conditioned place preference (Khroyan et al., 2007). SR16507 has higher efficacy at NOP receptor, is a potent analgesic, and induces a CPP (Toll et al., 2009). In fact, another novel NOP/MOP receptor compound, SR14150, has lower efficacy at MOP receptor but still has naloxone-reversible antinociceptive activity, and no apparent CPP (Toll et al., 2009). Compounds such as these, or additional NOP/MOP receptor compounds based on a buprenorphine scaffold, as we are now synthesizing, may prove useful as analgesics with reduced addiction liability or potentially as drug abuse medications.
In conclusion, these results are consistent with the hypothesis that the NOP receptor agonist activity of buprenorphine, and other mixed NOP/MOP receptor compounds, is able to attenuate the MOP receptor-mediated antinociceptive activity of this compound. These results further suggest that the development of additional compounds with mixed MOP/NOP receptor agonist activity could be beneficial for discovery of novel analgesics with reduced addiction liability and drug abuse medications.
This work was supported by the National Institutes of Health National Institute on Drug Abuse [Grants DA023281, DA14026] (to L.T. and N.Z., respectively).
This work was presented in part at the International Narcotics Research Conference; 2000 Jul 13–18; Charleston, SC.
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.109.156711
- MOP receptor
- μ-opioid receptor
- CHO cells
- Chinese hamster ovary cells
- N/OFQ
- nociceptin/orphanin FQ
- MPE
- maximum possible effect
- NOP receptor
- nociceptin/orphanin FQ receptor
- J113397
- 1-[(3R,4R)-1-cyclooctylmethyl-3-hydroxymethyl-4-piperidyl]-3-ethyl-1,3-dihydro-2H-benzimidazol-2-one
- SB-612111
- (−)-cis-1-methyl-7-[[4-(2,6-dichlorophenyl)piperidin-1-yl]methyl]-6,7,8,9-tetrahydro-5H-benzocyclohepten-5-ol
- SR16507
- 3-ethyl-1-(1-(4-isopropylcyclohexyl)piperidin-4-yl)-indolin-2-one
- JTC-801
- N-(4-amino-2-methylquinolin-6-yl)-2-(4-ethylphenoxymethyl) benzamide monohydrochloride
- G418
- (2R,3S,4R,5R,6S)-5-amino-6-[(1R,2S,3S,4R,6S)-4,6-diamino-3-[(2R,3R,4R,5R)-3,5-dihydroxy-5-methyl-4-methylaminooxan-2-yl]oxy-2-hydroxycyclohexyl]oxy-2-(1-hydroxyethyl)oxane-3,4-diol
- Ro 64-6198
- (1S,3aS)-8-(2,3,3a,4,5,6-hexahydro-1H-phenalen-1-yl)-1-phenyl-1,3,8-triaza-spiro[4.5]decan-4-one
- UFP-101
- [Nphe1,Arg14,Lys15]nociceptin-NH2
- DAMGO
- (2S)-2-[[2-[[(2R)-2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]propanoyl]amino]acetyl]-methylamino]-N-(2-hydroxyethyl)-3-phenylpropanamide; GTPγS, guanosine 5′-3-O-(thio)triphosphate
- ANOVA
- analysis of variance
- CPP
- conditioned place preference
- SR16835
- 1-(1-(2,3,3α,4,5,6-hexahydro-1H-phenalen-1-yl)piperidin-4-yl)-indolin-2-one.
References
- Calo G, Guerrini R, Bigoni R, Rizzi A, Marzola G, Okawa H, Bianchi C, Lambert DG, Salvadori S, Regoli D. (2000) Characterization of [Nphe(1)]nociceptin(1–13)NH(2), a new selective nociceptin receptor antagonist. Br J Pharmacol 129:1183–1193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calo G, Guerrini R, Rizzi A, Salvadori S, Burmeister M, Kapusta DR, Lambert DG, Regoli D. (2005) UFP-101, a peptide antagonist selective for the nociceptin/orphanin FQ receptor. CNS Drug Rev 11:97–112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y, Prusoff WH. (1973) Relationship between the inhibition constant (Ki) and the concentration of inhibitor which causes 50 percent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108 [DOI] [PubMed] [Google Scholar]
- Ciccocioppo R, Economidou D, Fedeli A, Angeletti S, Weiss F, Heilig M, Massi M. (2004) Attenuation of ethanol self-administration and of conditioned reinstatement of alcohol-seeking behaviour by the antiopioid peptide nociceptin/orphanin FQ in alcohol-preferring rats. Psychopharmacology (Berl) 172:170–178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciccocioppo R, Economidou D, Rimondini R, Sommer W, Massi M, Heilig M. (2007) Buprenorphine reduces alcohol drinking through activation of the nociceptin/orphanin FQ-NOP Receptor System. Biol Psychiatry 61:4–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan A, Lewis JW, Macfarlane IR. (1977) Agonist and antagonist properties of buprenorphine, a new antinociceptive agent. Br J Pharmacol 60:537–545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devine DP, Reinscheid RK, Monsma FJ, Jr., Civelli O, Akil H. (1996) The novel neuropeptide orphanin FQ fails to produce conditioned place preference or aversion. Brain Res 727:225–229 [DOI] [PubMed] [Google Scholar]
- Dooley CT, Spaeth CG, Berzetei-Gurske IP, Craymer K, Adapa ID, Brandt SR, Houghten RA, Toll L. (1997) Binding and in vitro activities of peptides with high affinity for the nociceptin/orphanin FQ receptor, ORL1. J Pharmacol Exp Ther 283:735–741 [PubMed] [Google Scholar]
- Hashimoto Y, Caló G, Guerrini R, Smith G, Lambert DG. (2000) Antagonistic effects of [Nphe1]nociceptin(1–13)NH2 on nociceptin receptor mediated inhibition of cAMP formation in Chinese hamster ovary cells stably expressing the recombinant human nociceptin receptor. Neurosci Lett 278:109–112 [DOI] [PubMed] [Google Scholar]
- Huang P, Kehner GB, Cowan A, Liu-Chen LY. (2001) Comparison of pharmacological activities of buprenorphine and norbuprenorphine: norbuprenorphine is a potent opioid agonist. J Pharmacol Exp Ther 297:688–695 [PubMed] [Google Scholar]
- Jasinski DR, Pevnick JS, Griffith JD. (1978) Human pharmacology and abuse potential of the analgesic buprenorphine: a potential agent for treating narcotic addiction. Arch Gen Psychiatry 35:501–516 [DOI] [PubMed] [Google Scholar]
- Jenck F, Wichmann J, Dautzenberg FM, Moreau JL, Ouagazzal AM, Martin JR, Lundstrom K, Cesura AM, Poli SM, Roever S, et al. ( 2000) A synthetic agonist at the orphanin FQ/nociceptin receptor ORL1: anxiolytic profile in the rat. Proc Natl Acad Sci U S A 97:4938–4943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- June HL, Cason CR, Chen SH, Lewis MJ. (1998) Buprenorphine alters ethanol self-administration in rats: dose-response and time-dependent effects. Psychopharmacology (Berl) 140:29–37 [DOI] [PubMed] [Google Scholar]
- Khroyan TV, Zaveri NT, Polgar WE, Orduna J, Olsen C, Jiang F, Toll L. (2007) SR 16435 [1-(1-(bicyclo[3.3.1]nonan-9-yl)piperidin-4-yl)indolin-2-one], a novel mixed nociceptin/orphanin FQ/mu-opioid receptor partial agonist: analgesic and rewarding properties in mice. J Pharmacol Exp Ther 320:934–943 [DOI] [PubMed] [Google Scholar]
- Ko MC, Woods JH, Fantegrossi WE, Galuska CM, Wichmann J, Prinssen EP. (2009) Behavioral effects of a synthetic agonist selective for nociceptin/orphanin fq peptide receptors in monkeys. Neuropsychopharmacology 34:2088–2096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotlinska J, Rafalski P, Biala G, Dylag T, Rolka K, Silberring J. (2003) Nociceptin inhibits acquisition of amphetamine-induced place preference and sensitization to stereotypy in rats. Eur J Pharmacol 474:233–239 [DOI] [PubMed] [Google Scholar]
- Lester PA, Traynor JR. (2006) Comparison of the in vitro efficacy of mu, delta, kappa and ORL1 receptor agonists and non-selective opioid agonists in dog brain membranes. Brain Res 1073–1074:290–296 [DOI] [PubMed] [Google Scholar]
- Lewis JW. (1985) Buprenorphine. Drug Alcohol Depend 14:363–372 [DOI] [PubMed] [Google Scholar]
- Lutfy K, Eitan S, Bryant CD, Yang YC, Saliminejad N, Walwyn W, Kieffer BL, Takeshima H, Carroll FI, Maidment NT, et al. ( 2003) Buprenorphine-induced antinociception is mediated by mu-opioid receptors and compromised by concomitant activation of opioid receptor-like receptors. J Neurosci 23:10331–10337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannelli P, Janiri L, Tempesta E, Jones RT. (1993) Prediction in drug abuse: cocaine interactions with alcohol and buprenorphine. Br J Psychiatry Suppl Sep:39–45 [PubMed] [Google Scholar]
- Martin A, Pilotto R, Singer G, Oei TP. (1983) The suppression of ethanol self injection by buprenorphine. Pharmacol Biochem Behav 19:985–986 [DOI] [PubMed] [Google Scholar]
- Mello NK, Mendelson JH. (1980) Buprenorphine suppresses heroin use by heroin addicts. Science 207:657–659 [DOI] [PubMed] [Google Scholar]
- Mello NK, Mendelson JH, Bree MP, Lukas SE. (1989) Buprenorphine suppresses cocaine self-administration in rhesus monkeys. NIDA Res Monogr 95:333–334 [PubMed] [Google Scholar]
- Meunier JC, Mollereau C, Toll L, Suaudeau C, Moisand C, Alvinerie P, Butour JL, Guillemot JC, Ferrara P, Monsarrat B, et al. ( 1995) Isolation and structure of the endogenous agonist of opioid receptor-like ORL1 receptor. Nature 377:532–535 [DOI] [PubMed] [Google Scholar]
- Mogil JS, Grisel JE, Zhangs G, Belknap JK, Grandy DK. (1996) Functional antagonism of mu-, delta- and kappa-opioid antinociception by orphanin FQ. Neurosci Lett 214:131–134 [DOI] [PubMed] [Google Scholar]
- Murphy NP, Lee Y, Maidment NT. (1999) Orphanin FQ/nociceptin blocks acquisition of morphine place preference. Brain Res 832:168–170 [DOI] [PubMed] [Google Scholar]
- Ozaki S, Kawamoto H, Itoh Y, Miyaji M, Azuma T, Ichikawa D, Nambu H, Iguchi T, Iwasawa Y, Ohta H. (2000) In vitro and in vivo pharmacological characterization of J-113397, a potent and selective non-peptidyl ORL1 receptor antagonist. Eur J Pharmacol 402:45–53 [DOI] [PubMed] [Google Scholar]
- Reinscheid RK, Nothacker HP, Bourson A, Ardati A, Henningsen RA, Bunzow JR, Grandy DK, Langen H, Monsma FJ, Jr, Civelli O. (1995) Orphanin FQ: a neuropeptide that activates an opioidlike G protein-coupled receptor. Science 270:792–794 [DOI] [PubMed] [Google Scholar]
- Reiss D, Wichmann J, Tekeshima H, Kieffer BL, Ouagazzal AM. (2008) Effects of nociceptin/orphanin FQ receptor (NOP) agonist, Ro64–6198, on reactivity to acute pain in mice: comparison to morphine. Eur J Pharmacol 579:141–148 [DOI] [PubMed] [Google Scholar]
- Sakoori K, Murphy NP. (2004) Central administration of nociceptin/orphanin FQ blocks the acquisition of conditioned place preference to morphine and cocaine, but not conditioned place aversion to naloxone in mice. Psychopharmacology (Berl) 172:129–136 [DOI] [PubMed] [Google Scholar]
- Schottenfeld RS, Pakes J, Ziedonis D, Kosten TR. (1993) Buprenorphine: dose-related effects on cocaine and opioid use in cocaine-abusing opioid-dependent humans. Biol Psychiatry 34:66–74 [DOI] [PubMed] [Google Scholar]
- Shoblock JR, Wichmann J, Maidment NT. (2005) The effect of a systemically active ORL-1 agonist, Ro 64–6198, on the acquisition, expression, extinction, and reinstatement of morphine conditioned place preference. Neuropharmacology 49:439–446 [DOI] [PubMed] [Google Scholar]
- Spagnolo B, Calo G, Polgar WE, Jiang F, Olsen CM, Berzetei-Gurske I, Khroyan TV, Husbands SM, Lewis JW, Toll L, et al. ( 2008) Activities of mixed NOP and mu-opioid receptor ligands. Br J Pharmacol 153:609–619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suyama H, Kawamoto M, Gaus S, Yuge O. (2003) Effect of JTC-801 (nociceptin antagonist) on neuropathic pain in a rat model. Neurosci Lett 351:133–136 [DOI] [PubMed] [Google Scholar]
- Tian JH, Xu W, Fang Y, Mogil JS, Grisel JE, Grandy DK, Han JS. (1997) Bidirectional modulatory effect of orphanin FQ on morphine-induced analgesia: antagonism in brain and potentiation in spinal cord of the rat. Br J Pharmacol 120:676–680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toll L, Khroyan TV, Polgar WE, Jiang F, Olsen C, Zaveri NT. (2009) Comparison of the antinociceptive and antirewarding profiles of novel bifunctional nociceptin receptor/μ-opioid receptor ligands: implications for therapeutic applications. J Pharmacol Exp Ther 331:954–964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toll L, Berzetei-Gurske IP, Polgar WE, Brandt SR, Adapa ID, Rodriguez L, Schwartz RW, Haggart D, O'Brien A, White A, et al. ( 1998) Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications. NIDA Res Monogr 178:440–466 [PubMed] [Google Scholar]
- Wang HL, Hsu CY, Huang PC, Kuo YL, Li AH, Yeh TH, Tso AS, Chen YL. (2005) Heterodimerization of opioid receptor-like 1 and mu-opioid receptors impairs the potency of micro receptor agonist. J Neurochem 92:1285–1294 [DOI] [PubMed] [Google Scholar]
- Wnendt S, Krüger T, Janocha E, Hildebrandt D, Englberger W. (1999) Agonistic effect of buprenorphine in a nociceptin/OFQ receptor-triggered reporter gene assay. Mol Pharmacol 56:334–338 [DOI] [PubMed] [Google Scholar]
- Yamada H, Nakamoto H, Suzuki Y, Ito T, Aisaka K. (2002) Pharmacological profiles of a novel opioid receptor-like1 (ORL(1)) receptor antagonist, JTC-801. Br J Pharmacol 135:323–332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaratin PF, Petrone G, Sbacchi M, Garnier M, Fossati C, Petrillo P, Ronzoni S, Giardina GA, Scheideler MA. (2004) Modification of nociception and morphine tolerance by the selective opiate receptor-like orphan receptor antagonist (−)-cis-1-methyl-7-[[4-(2,6-dichlorophenyl)piperidin-1-yl]methyl]-6,7,8,9- tetrahydro-5H-benzocyclohepten-5-ol (SB-612111). J Pharmacol Exp Ther 308:454–461 [DOI] [PubMed] [Google Scholar]