Abstract
Engagement of surface receptors contributes to the antimicrobial activity of human immune cells. We show here that infection of human monocyte-derived macrophages (MDM) with live Mycobacterium avium induced the expression of CD23 on their membrane. Subsequent cross-linking of surface CD23 by appropriate ligands induced a dose-dependent antibacterial activity of MDM and the elimination of most infected cells. The stimulation of inducible nitric oxide synthase-dependent generation of NO from MDM after CD23 activation played a major role during their anti-M. avium activity. CD23 activation also induced tumor necrosis factor alpha (TNF-α) production from MDM. Mycobacteria reduction was partially inhibited by the addition of neutralizing anti-TNF-α antibody to cell cultures without affecting NO levels, which suggested the role of this cytokine for optimal antimicrobial activity. Finally, interleukin-10, a Th2 cytokine known to downregulate CD23 pathway, is shown to decrease NO generation and mycobacteria elimination by macrophages. Therefore, (i) infection with M. avium promotes functional surface CD23 expression on human macrophages and (ii) subsequent signaling of this molecule contributes to the antimicrobial activity of these cells through an NO- and TNF-α-dependent pathway. This study reveals a new human immune response mechanism to counter mycobacterial infection involving CD23 and its related ligands.
Disseminated Mycobacterium avium complex infection is a severe complication encountered among immunocompromised patients such as patients with AIDS (3, 22). Like other pathogenic mycobacteria in humans (e.g., M. tuberculosis and M. leprae), M. avium is phagocytosed by macrophages after entry into the body (10, 23). Uptake of pathogenic mycobacteria occurs via the complement receptors CR3b and CR1 (7, 42), mannosyl-fucosyl receptor (7), fibronectin receptor (36), and/or αvβ3 integrin receptor (7). The phagosomal compartments of nonactivated macrophages containing mycobacteria failed to mature to phagolysosomes but retain the cellular markers associated with early endosomes (18, 39). This feature explains why M. avium, like M. tuberculosis, is not destroyed by nonactivated macrophages but is able to survive and multiply within their phagosomes. The mechanism(s) whereby pathogenic mycobacteria are able to preclude phagosomal maturation is not well understood, but the ability to survive and multiply within macrophages is a key component of mycobacterial pathogenesis.
Signaling through Toll-like-receptor 2 (TLR-2) appears to be essential for the induction of costimulatory molecules and the secretion of cytokines which are essential for the macrophage-mediated antimicrobial activity and antigen-presentation to T lymphocytes (41, 44). Most TLRs recruit the MyD88 adaptor molecule, which leads to the activation of NF-κB and mitogen-activated protein kinases, resulting in the induction of inflammatory mediators (45, 50). After M. avium infection, macrophages produce various effector molecules essential for infection control including tumor necrosis factor alpha (TNF-α), interleukin-12 (IL-12), and reactive oxygen intermediates (2, 46). Inducible nitric oxide synthase (iNOS)-derived NO was also reported as a powerful and necessary antimycobacterial defense mechanism in mouse models (9), and compromised handling of infection was observed in mice lacking the iNOS gene (1). Although iNOS has been detected in macrophages from disease lesions (31), it has not been possible to consistently demonstrate an NO-mediated mycobacterial killing phenomenon in human macrophages (38, 44). TLR-induced antimicrobial activity in human monocytes/macrophages was not found to be dependent on iNOS activity, nor could NO be detected in these cells (44). These data suggested that human macrophages have an NO-independent antimicrobial mechanism without excluding a role for NO in human host defense.
Upon infection, monocyte-derived macrophages (MDM) fulfill their maturation and acquire or increase their surface differentiation markers such as CD14 and CD23 (21, 44). However, whereas CD14/TLR-4 expression has been well documented (44), no data were available on CD23 expression after mycobacterial infection. In the present study, we found that infection with live M. avium subsp. avium induced CD23 expression on the surface of MDM. The role of this molecule during antimicrobial activity of macrophages was then investigated by using various CD23 agonistic ligands (35, 48). Our data indicate that surface CD23 mediates antimicrobial activity of human macrophages, in part, through the induction of NO and TNF-α production.
MATERIALS AND METHODS
Bacterial cultures.
M. avium subsp. avium strains were obtained from the American Type Culture Collection (ATCC 35713 and ATCC 35716) and grown at 37°C in 5% CO2 in Middlebrook 7H9 medium with 10% oleic acid-albumin-dextrose complex (OADC; Difco, Sparks, MD). M. avium subsp. avium samples were serially diluted and counted by determining the optical density (OD) absorbance at 600 nm or on a bacterial hemacytometer. Before macrophage infection, bacterial suspensions were vigorously vortexed to disperse clumps and obtain a consistent OD reading, followed by dilution in phosphate-buffered saline. Heat-inactivated mycobacteria were obtained after incubation at 90°C for 10 min and quantified by determining the OD.
Human cells and infection.
Peripheral blood samples pretested for the absence of human immunodeficiency virus or hepatitis B and C virus infection were obtained from healthy volunteers (Immunology Department, Pitié-Salpêtrière Hospital, Paris, France). The present study was carried out in compliance with the Helsinki Declaration. Peripheral blood-derived mononuclear leukocytes were obtained by Ficoll gradient separation and suspended in McCoy 5A modified culture medium supplemented with 100 U of penicillin/ml, 100 μg of streptomycin/ml, 25 mM HEPES, 0.1 mM 2-mercaptoethanol, 2 mM sodium pyruvate, 0.2 mM l-cysteine, 5 μg of polymyxin B/ml, and 10% fetal calf serum (FCS; all from Invitrogen, Paisley, United Kingdom). All of the above culture media, chemicals, and FCS were endotoxin-free and tested for the absence of direct activation effects on human monocytes (CD23 expression and TNF-α production as activation markers). Monocytes were subsequently separated from other leukocytes by adherence to FCS-coated culture flasks and reincubated for an additional 5 days in the same culture conditions. After these procedures, >95% of cells expressed CD14 antigen and displayed the cytochemical characteristics of MDM (48). Cells were collected and reincubated (2 × 105/ml) 24 h with live or heat-inactivated M. avium subsp. avium at a ratio of 10 bacilli per cell. Extracellular bacteria were eliminated by washing with culture medium, and cells were reincubated for an additional 24 h. This led to the infection of 50 to 70% MDM, as counted after Ziehl-Neelsen carbolfuchsin staining and microscopic examination of acid-fast bacteria (AFB). To measure MDM viability and apoptosis, externalization of membrane phosphatidylserine was analyzed by using annexin V-fluorescein isothiocyanate (FITC) and a propidium iodide kit (Immunotech, Marseille, France). For simultaneous bacteria and apoptosis staining, cells were cultured on four-well chamber slides (Lab-Tek; Nunc, Brumath, France). After exposure to various culture conditions, the slides were labeled with annexin-propidium iodide (Immunotech) and intracellular AFB staining. The percentage of apoptotic and/or infected cells was obtained after counting 200 cells in each well under fluorescence microscopy.
Human cell activation.
Cell surface expression of CD23 was measured by flow cytometry, using FITC-labeled anti-CD23 (Beckman-Coulter, Fullerton, CA). Infected MDM were directly activated through CD23 pathway, while CD23 cell surface expression was induced in uninfected MDM in the presence of 10 ng of recombinant IL-4/ml, as described elsewhere (48). CD23-positive cells were incubated in the presence of cross-linking anti-CD23 monoclonal antibody (anti-CD23 MAb; clone 135, immunoglobulin G1κ [IgG1κ], 20 μg/ml; Novartis, Geneva, Switzerland) or human IgE/anti-IgE (10 μg of each/ml; Nordic, Tilburg, The Netherlands) immune complexes as detailed elsewhere (6, 48). MDM cultures were supplemented with the chemical NO donor, SNAP (S-nitroso-N-acetylpenicillamine; Coger, Paris, France), recombinant human IL-10, TNF-α, or neutralizing anti-human TNF-α (Genzyme, Cambridge, MA).
TNF-α and NO measurement.
Cell supernatants were harvested 72 h after cell activation for the quantification of TNF-α by a specific enzyme-linked immunosorbent assay (R&D, Paris, France). They were also tested for the levels of nitrites (NO2−), the stable end product of NO, using the Griess reaction modified as detailed elsewhere (25). This method gave a sensitivity limit of 0.2 μM if McCoy 5A modified culture medium was used. An inhibitory analog of l-arginine, l-NMMA (NG-monomethyl-l-arginine; Coger), was used to inhibit iNOS activity, while d-NMMA was used as a negative control.
CFU count and data analysis.
To collect intracellular bacteria, macrophages were lysed inside culture wells by the addition of saponin (1 mg/ml, final concentration; Sigma-Aldrich, St. Louis, MO), washed, and cultured on 7H9 agar plates supplemented with OADC. This procedure took into account both the intracellular and the extracellular CFU in each well. Colony counts were performed after 3 weeks as described previously (24). The data are expressed as the mean percentage ± the standard deviation. The differences in the patterns between uninfected and M. avium subsp. avium-infected cultures were assessed by analysis of variance. The differences between other paired data were examined by the Student t test or correlation test. P values of <0.04 were considered statistically significant.
RESULTS
M. avium promotes functional CD23 expression by human macrophages.
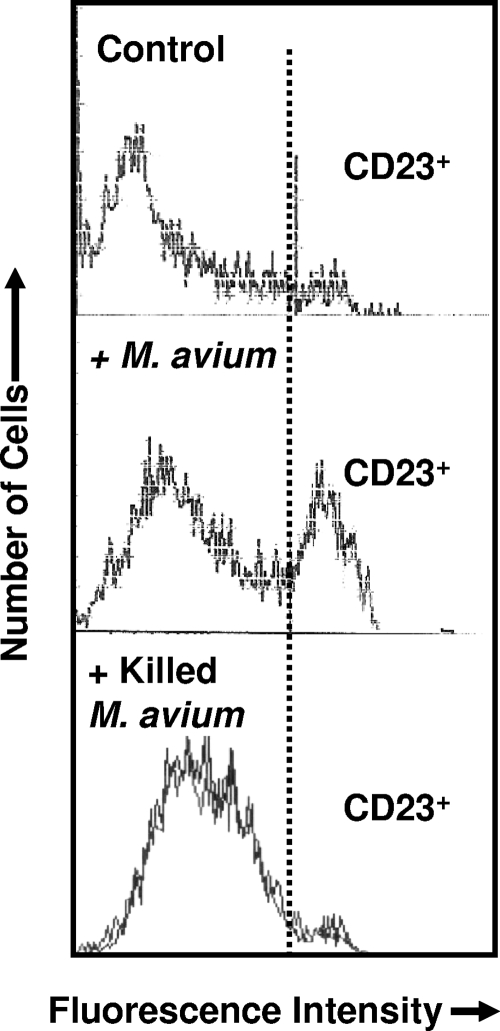
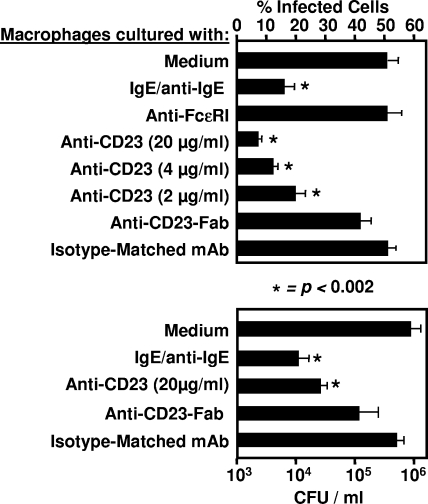
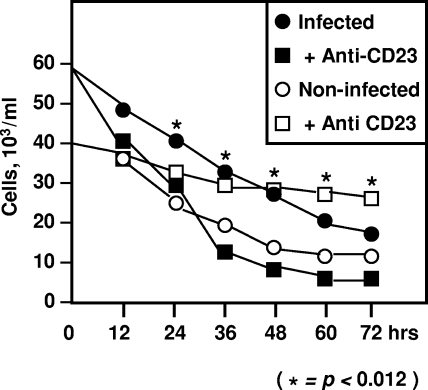
We investigated the expression of CD23 in in vitro MDM cultures with live or heat-inactivated M. avium subsp. avium. We first showed that a significant percentage of macrophages acquired CD23 surface expression after 48-h cell incubation with live M. avium subsp. avium (from 5% ± 3% in uninfected cells to 34% ± 8% after infection, P < 0.0002, Fig. 1). In contrast, MDM incubation with heat-inactivated M. avium subsp. avium failed to promote CD23 expression (Fig. 1). Since CD23 mediates MDM activation, we sought to determine whether CD23 engagement led to the activation of antimicrobial activity of these cells. To induce CD23 related signaling, IgE/anti-IgE immune complexes or a cross-linking anti-CD23 antibody were used (48). After 3 days of incubation, we counted the percentage of AFB-containing infected cells and lysed macrophages to evaluate the total CFU. Upon infection, engagement of CD23 by both ligands reduced the number of infected cells in a dose-dependent manner (Fig. 2). Isotype-matched control (anti-CD8, IgG1κ) or anti-FcɛRI antibodies failed to induce antimicrobial effects. Interestingly, Fab fragments of anti-CD23, which bound CD23 without cross-linking, had no effect in this respect (Fig. 2). We also determined the intra- and extramacrophagic growth of the M. avium subsp. avium after CD23 ligation, and our results confirmed a reduction of bacterial CFU by this pathway (Fig. 2, lower panel). In addition, good correlation was found between the percentage of infected cells and the number of CFU in these cultures (correlation coefficient of 97%). Relationship between M. avium subsp. avium infection and MDM survival was also analyzed with or without CD23 cross-linking. Our data (Fig. 3) showed that M. avium subsp. avium infection induced clear elimination of both infected and uninfected macrophages in the same cultures. Upon CD23 activation, infected cell death was accelerated, while the number of uninfected cells remained stable (Fig. 3) and could be maintained in culture for 10 to 14 days, the usual survival delay for activated macrophages in vitro (48). Therefore, in addition to microbiostasis, the CD23 pathway rescues uninfected cells from M. avium subsp. avium-induced mortality.
FIG. 1.
Mycobacterial infection promotes CD23 expression on human macrophages. Cells were infected with live or heat-inactivated M. avium subsp. avium for 24 h, washed, and reincubated for an additional 24 h. They were then labeled with FITC-conjugated anti-CD23 antibody and analyzed by flow cytometry. Representative data from one donor cell, out of three tested, are shown.
FIG. 2.
CD23 engagement mediates microbicidal activity of human macrophages. M. avium subsp. avium-infected MDM were incubated in the presence of IgE/anti-IgE immune complexes, cross-linking anti-CD23 MAb or non-cross-linking Fab fragments of anti-CD23 MAb (20 μg/ml). Anti-human CD8 MAb (20 μg/ml) was used as an isotype-matched control. After 72 h of incubation, the percentage of infected macrophages was assessed (upper panel). In parallel, cells were lysed, and whole CFU content in culture wells was analyzed after 3 weeks (lower panel). The results show the means ± the standard deviations for data from five distinct MDM preparations, each performed in duplicate. Asterisks indicate significance compared to cells cultured in medium alone. A good correlation coefficient (>97%) was obtained between the percentage of infected macrophages and the number of CFU under these culture conditions.
FIG. 3.
Effect of infection and CD23 activation on macrophage viability. MDM were incubated with M. avium subsp. avium for 48 h, washed, and reincubated with medium alone (circles) or in the presence of cross-linking anti-CD23 MAb (20 μg/ml, squares). In each well, the number of viable cells that were infected (filled symbols) or uninfected (empty symbols) was determined by simultaneous labeling with annexin V and propidium iodide. For each culture condition, 200 cells were counted. Asterisks indicate significance between anti-CD23 MAb-treated cell cultures and untreated cells. The mean from three experiments is shown (standard deviation < 21%).
Induction of bacteriostasis by CD23 activation involves NO pathway.
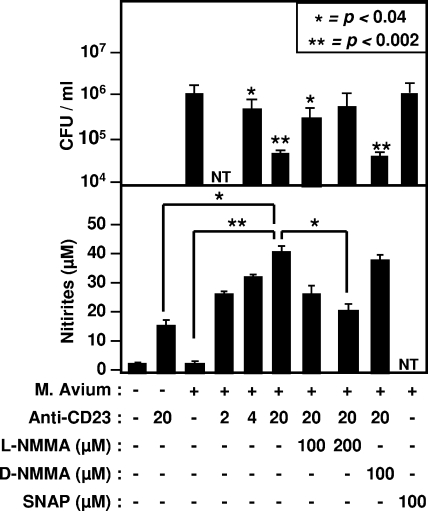
In human macrophages, CD23 ligation is well known to promote gene expression and the secretion of various inflammatory cytokines, including TNF-α, IL-1, IL-6, and IL-8, among others (29, 35). The CD23 pathway also promotes the generation of superoxide, the transcription of iNOS gene, and subsequent production of NO from human macrophages, eosinophils, and epithelial cells (4, 6, 48). Since NO mediates microbiostatic effect, we sought to determine whether the NO pathway had a role in the inhibition of M. avium CFU after CD23 activation. The levels of nitrites, the final metabolites of NO, were quantified in cell supernatants. The data in Fig. 4 indicate that CD23-mediated CFU inhibition correlated with increased NO levels produced by MDM. The addition of an iNOS inhibitor, l-NMMA, significantly reversed microbiostatic activity (P < 0.007), whereas d-NMMA had no effect. SNAP, a chemical NO donor (28), mimicked the CD23-mediated effect. The results also indicate that, upon activation, infected MDM produced higher NO levels than uninfected cells (P < 0.008, Fig. 4), which suggests that infection promoted CD23 signaling, as has been observed after parasitic infection of human macrophages (48). These data clearly support the critical role for NO during CD23-mediated antimycobacterial activity.
FIG. 4.
Involvement of iNOS pathway during CD23-mediated activation of microbicidal activity of human macrophages. Uninfected or infected MDM were incubated in the presence of a cross-linking anti-CD23 MAb, a chemical NO donor (SNAP), an iNOS inhibitor (l-NMMA), or a negative control (d-NMMA). After 72 h of incubation, the number of CFU was assessed and correlated to the levels of nitrites recovered in cell supernatants. The results show means ± the standard deviation for data from five distinct MDM preparations, each performed in duplicate. Asterisks in the upper panel indicate significance compared to untreated cells. NT, not tested.
Role of TNF-α and IL-10 during CD23-mediated inhibition of M. avium growth.
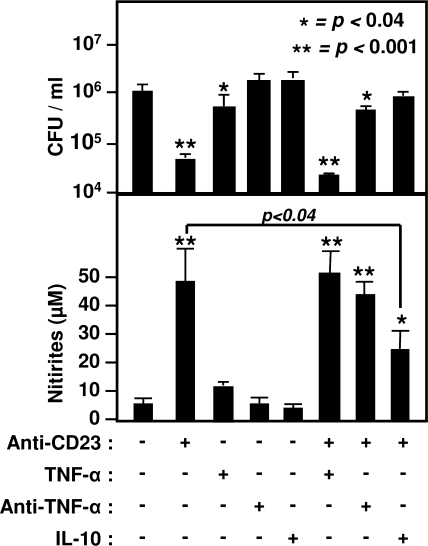
We next investigated the role of TNF-α during CD23-mediated M. avium subsp. avium killing because this cytokine has been suggested as an antimycobacterial mediator (2, 17). After CD23 activation, a significant increase in TNF-α levels was observed in activated MDM cell supernatants compared to controls (from 32 ± 14 pg/ml to 588 ± 154 pg/ml, P < 0.0001, means ± the standard deviations of five distinct MDM preparations). In nonactivated MDM, exogenous TNF-α induced low but significant CFU inhibition (Fig. 5), whereas the simultaneous addition of neutralizing anti-TNF-α antibody during CD23 activation reduced bacteriostatic activity (P < 0.006). This suggests a role for this cytokine during CD23 engagement and may explain the inability of iNOS inhibitor to completely inhibit CD23 effect. However, anti-TNF-α failed to significantly inhibit NO generation from human macrophages, which suggested that, in infected cells, NO pathway is independent of TNF-α production. Finally, the regulation of microbiostatic effects of CD23 pathway by IL-10 was investigated. We have previously shown that this Th2 cytokine, produced during various inflammatory responses (32), downregulated CD23 pathway in human cells (5, 47). Accordingly, the addition of recombinant IL-10 during CD23 engagement significantly inhibited antimicrobial activity and reduced NO generation from human macrophages (Fig. 5).
FIG. 5.
Regulation of CD23-mediated antimicrobial activity of human macrophages. M. avium subsp. avium-infected MDM were incubated in the presence of a cross-linking anti-CD23 MAb (20 μg/ml), recombinant TNF-α (100 IU/ml), IL-10 (10 ng/ml), or anti-TNF-α MAb (20 μg/ml). After 72 h of incubation, the number of CFU in culture wells was obtained and correlated to the levels of nitrites recovered in cell supernatants. The results show the means ± the standard deviations from five distinct MDM preparations, each performed in duplicate. Asterisks indicate significance compared to untreated cells.
DISCUSSION
CD23 is an activation/differentiation antigen expressed by most mature macrophages. During various parasitic infections, increased expression of this molecule has been observed. Its role in the generation of microbicidal mediators from macrophages and in intracellular and extracellular killing of pathogens has been described (29, 40, 48). Since both gamma interferon (IFN-γ) and IL-4 are able to promote CD23 gene expression, this pathway could be involved during both Th1 and Th2 responses (20, 35). We clearly show here that infection with live M. avium subsp. avium promoted surface CD23 expression in human MDM. With heat-inactivated M. avium subsp. avium, we failed to detect a CD23 increase. CD23 expression may be due to the ability of mycobacteria to activate mitogen-activated protein kinase and NF-κB, potent factors for the activation of macrophage gene expression (21). Intracellular cell signaling and cell activation via CD23 require the presence of appropriate physiologic ligands. In addition to its ability to bind IgE (FcɛRII), CD23 displays various other physiologic ligands such as CD21, CD11b/c, CD47-vitronectin, and mannose-containing proteins (20, 29). CD23 cross-linking by its ligands promotes the secretion of IL-1, IL-6, TNF-α, superoxide, and NO from various human cells (4, 6, 29). In the present study, we show that CD23 ligation promotes antimicrobial activity of human macrophages, in part through the generation of NO and TNF-α, two potent antimycobacterial mediators. CD23 also accelerated infected cell death (Fig. 3), while uninfected MDM survived and may continue their antimicrobial activity through the release of various mediators. The mechanism of cell death remains to be clarified and may be due to autophagy-dependent antimicrobial activity of macrophages, recently reported in a similar model (43). Fab fragments of anti-CD23 MAb (clone 135), which bind CD23 without cross-linkage, blocked cell activation and reversed antimicrobial effect. Therefore, the acquisition of surface CD23 and the availability of its physiological ligands may play an important role during human immune response to M. avium subsp. avium infections.
CD23-mediated NO generation seems to play a major role during the antimicrobial activity of MDM. NO is a diffusible free radical, soluble in both lipids and water, and able to highly react with oxygen and oxygen intermediates to form NO2, NO2−, NO3−, N2O3, and the highly mycobactericidal ONOO− (30). iNOS is a tightly regulated enzyme and the major generator of NO in immune cells (49). Studies investigating defense against NO derivatives in mycobacteria have demonstrated that several mycobacterial proteins are produced against reactive nitrogen intermediates in order to rescue bacteria from the immune response (11, 27). Recently, it was reported that M. tuberculosis inhibits trafficking of iNOS to the mycobacterial phagosome to escape immune response (12). In the present study we show that CD23 engagement, which mediates increased transcription of iNOS in human macrophages, induced NO generation after M. avium subsp. avium infection in a dose-dependent manner (Fig. 2). We consistently failed to detect spontaneous NO production after M. avium subsp. avium infection as reported previously (14). In addition to the generation of highly mycobactericidal ONOO−, NO may inhibit M. avium subsp. avium growth through its ability to reduce iron levels. We have previously shown that NO-mediated antitumoral activity is in part due to its ability to decrease intracellular ferric iron levels in target cells (15). This may explain why intracellular iron elevation increased mycobacterial resistance and human susceptibility to these infections (8).
Among cytokines, TNF-α plays a critical role in host defense against intracellular mycobacterial infections (2, 17). Researchers have reported that M. avium subsp. avium infection produces lower levels of TNF-α and NO compared to infections with nonpathogenic mycobacteria (37), contributing to the persistence of infection. CD23 engagement significantly induced TNF-α generation by infected MDM and the addition of anti-TNF-α during CD23 activation partially decreased antimicrobial activity, supporting a role for this cytokine. However, whereas TNF-α was previously suggested to be necessary for optimal NO production from human infected macrophages (13), CD23-mediated NO generation was not significantly affected after the addition of anti-TNF-α to our cultures, which suggests that early TNF-α generation following CD23 ligation (33) was not required for iNOS induction in infected macrophages. Meanwhile, the presence of both factors seems to be required for optimal antimicrobial activity. Therefore, human CD23 pathway differs from TLR-mediated antimycobacterial activity in mice (19), where the role of oxidative radicals and TNF-α is marginal.
During antigen-specific immune response, activation of infected macrophages and the control of mycobacteria multiplication is essentially dependent on IFN-γ produced by Th1 lymphocytes (26). We have previously shown that IFN-γ is a potent inducer of CD23 gene transcription and surface expression (34) and may therefore potentiate antimicrobial activity via CD23 pathway without IgE requirement. As with most Th1-like responses, IL-10 inhibits CD23-mediated activities (5, 47), and transgenic mice expressing human IL-10 showed increased susceptibility to infection with M. avium subsp. avium associated with decreased macrophage effector function and apoptosis (16). Similarly, we here show that CD23 activation and NO generation were significantly decreased after the addition of exogenous IL-10 to human macrophages. In conclusion, CD23 expression after mycobacterial infection, its engagement, and the production of antimicrobial mediators may contribute to our understanding of human immune response to these infections.
Acknowledgments
We thank M. Nacher, F. Ouaaz, and V. Jarlier for helpful scientific advice.
This study was supported by grants from the Conseil Régional d'Aquitaine and the Ministère de l'Industrie. The fund providers had no role in study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
Editor: J. L. Flynn
Footnotes
Published ahead of print on 5 October 2009.
REFERENCES
- 1.Adams, L. B., M. C. Dinauer, D. E. Morgenstern, and J. L. Krahenbuhl. 1997. Comparison of the roles of reactive oxygen and nitrogen intermediates in the host response to Mycobacterium tuberculosis using transgenic mice. Tuberc. Lung Dis. 78:237-246. [DOI] [PubMed] [Google Scholar]
- 2.Akaki, T., K. Sato, T. Shimizu, C. Sano, H. Kajitani, S. Dekio, and H. Tomioka. 1997. Effector molecules in expression of the antimicrobial activity of macrophages against Mycobacterium avium complex: roles of reactive nitrogen intermediates, reactive oxygen intermediates, and free fatty acids. J. Leukoc. Biol. 62:795-804. [DOI] [PubMed] [Google Scholar]
- 3.Ammann, A. J., M. J. Cowan, D. W. Wara, P. Weintrub, S. Dritz, H. Goldman, and H. A. Perkins. 1983. Acquired immunodeficiency in an infant: possible transmission by means of blood products. Lancet i:956-958. [DOI] [PubMed] [Google Scholar]
- 4.Arock, M., L. Le Goff, P. A. Becherel, B. Dugas, P. Debre, and M. D. Mossalayi. 1994. Involvement of Fc epsilon RII/CD23 and l-arginine dependent pathway in IgE-mediated activation of human eosinophils. Biochem. Biophys. Res. Commun. 203:265-271. [DOI] [PubMed] [Google Scholar]
- 5.Becherel, P. A., L. LeGoff, C. Frances, O. Chosidow, J. J. Guillosson, P. Debre, M. D. Mossalayi, and M. Arock. 1997. Induction of IL-10 synthesis by human keratinocytes through CD23 ligation: a cyclic adenosine 3′,5′-monophosphate-dependent mechanism. J. Immunol. 159:5761-5765. [PubMed] [Google Scholar]
- 6.Becherel, P. A., M. D. Mossalayi, F. Ouaaz, L. Le Goff, B. Dugas, N. Paul-Eugene, C. Frances, O. Chosidow, E. Kilchherr, J. J. Guillosson, P. Debre, and M. Arock. 1994. Involvement of cyclic AMP and nitric oxide in immunoglobulin E-dependent activation of Fc epsilon RII/CD23+ normal human keratinocytes. J. Clin. Investig. 93:2275-2279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bermudez, L. E., L. S. Young, and H. Enkel. 1991. Interaction of Mycobacterium avium complex with human macrophages: roles of membrane receptors and serum proteins. Infect. Immun. 59:1697-1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Boelaert, J. R., S. J. Vandecasteele, R. Appelberg, and V. R. Gordeuk. 2007. The effect of the host's iron status on tuberculosis. J. Infect. Dis. 195:1745-1753. [DOI] [PubMed] [Google Scholar]
- 9.Chan, J., Y. Xing, R. S. Magliozzo, and B. R. Bloom. 1992. Killing of virulent Mycobacterium tuberculosis by reactive nitrogen intermediates produced by activated murine macrophages. J. Exp. Med. 175:1111-1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cosma, C. L., D. R. Sherman, and L. Ramakrishnan. 2003. The secret lives of the pathogenic mycobacteria. Annu. Rev. Microbiol. 57:641-676. [DOI] [PubMed] [Google Scholar]
- 11.Darwin, K. H., S. Ehrt, J. C. Gutierrez-Ramos, N. Weich, and C. F. Nathan. 2003. The proteasome of Mycobacterium tuberculosis is required for resistance to nitric oxide. Science 302:1963-1966. [DOI] [PubMed] [Google Scholar]
- 12.Davis, A. S., I. Vergne, S. S. Master, G. B. Kyei, J. Chua, and V. Deretic. 2007. Mechanism of inducible nitric oxide synthase exclusion from mycobacterial phagosomes. PLoS Pathog. 3:e186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Denis, M. 1991. Interferon-gamma-treated murine macrophages inhibit growth of tubercle bacilli via the generation of reactive nitrogen intermediates. Cell. Immunol. 132:150-157. [DOI] [PubMed] [Google Scholar]
- 14.Dumarey, C. H., V. Labrousse, N. Rastogi, B. B. Vargaftig, and M. Bachelet. 1994. Selective Mycobacterium avium-induced production of nitric oxide by human monocyte-derived macrophages. J. Leukoc. Biol. 56:36-40. [DOI] [PubMed] [Google Scholar]
- 15.Feger, F., H. Ferry-Dumazet, M. Mamani Matsuda, J. Bordenave, M. Dupouy, A. K. Nussler, M. Arock, L. Devevey, J. Nafziger, J. J. Guillosson, J. Reiffers, and M. D. Mossalayi. 2001. Role of iron in tumor cell protection from the proapoptotic effect of nitric oxide. Cancer Res. 61:5289-5294. [PubMed] [Google Scholar]
- 16.Feng, C. G., M. C. Kullberg, D. Jankovic, A. W. Cheever, P. Caspar, R. L. Coffman, and A. Sher. 2002. Transgenic mice expressing human interleukin-10 in the antigen-presenting cell compartment show increased susceptibility to infection with Mycobacterium avium associated with decreased macrophage effector function and apoptosis. Infect. Immun. 70:6672-6679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Flynn, J. L., M. M. Goldstein, J. Chan, K. J. Triebold, K. Pfeffer, C. J. Lowenstein, R. Schreiber, T. W. Mak, and B. R. Bloom. 1995. Tumor necrosis factor-alpha is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity 2:561-572. [DOI] [PubMed] [Google Scholar]
- 18.Frehel, C., C. de Chastellier, T. Lang, and N. Rastogi. 1986. Evidence for inhibition of fusion of lysosomal and prelysosomal compartments with phagosomes in macrophages infected with pathogenic Mycobacterium avium. Infect. Immun. 52:252-262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gomes, M. S., S. Sousa Fernandes, J. V. Cordeiro, S. Silva Gomes, A. Vieira, and R. Appelberg. 2008. Engagement of Toll-like receptor 2 in mouse macrophages infected with Mycobacterium avium induces non-oxidative and TNF-independent anti-mycobacterial activity. Eur. J. Immunol. 38:2180-2189. [DOI] [PubMed] [Google Scholar]
- 20.Gould, H. J., and B. J. Sutton. 2008. IgE in allergy and asthma today. Nat. Rev. Immunol. 8:205-217. [DOI] [PubMed] [Google Scholar]
- 21.Guha, M., and N. Mackman. 2001. LPS induction of gene expression in human monocytes. Cell Signal. 13:85-94. [DOI] [PubMed] [Google Scholar]
- 22.Horsburgh, C. R., Jr. 1991. Mycobacterium avium complex infection in the acquired immunodeficiency syndrome. N. Engl. J. Med. 324:1332-1338. [DOI] [PubMed] [Google Scholar]
- 23.Houben, E. N., L. Nguyen, and J. Pieters. 2006. Interaction of pathogenic mycobacteria with the host immune system. Curr. Opin. Microbiol. 9:76-85. [DOI] [PubMed] [Google Scholar]
- 24.Kent, P. T., and G. P. Kubica. 1985. Public health mycobacteriology: a guide for the level III laboratory. Centers for Disease Control and Prevention, Atlanta, GA.
- 25.Kolb, J. P., N. Paul-Eugene, C. Damais, K. Yamaoka, J. C. Drapier, and B. Dugas. 1994. Interleukin-4 stimulates cGMP production by IFN-γ-activated human monocytes: involvement of the nitric oxide synthase pathway. J. Biol. Chem. 269:9811-9816. [PubMed] [Google Scholar]
- 26.Liu, P. T., and R. L. Modlin. 2008. Human macrophage host defense against Mycobacterium tuberculosis. Curr. Opin. Immunol. 20:371-376. [DOI] [PubMed] [Google Scholar]
- 27.Miller, B. H., R. A. Fratti, J. F. Poschet, G. S. Timmins, S. S. Master, M. Burgos, M. A. Marletta, and V. Deretic. 2004. Mycobacteria inhibit nitric oxide synthase recruitment to phagosomes during macrophage infection. Infect. Immun. 72:2872-2878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moncada, S., and E. A. Higgs. 1995. Molecular mechanisms and therapeutic strategies related to nitric oxide. FASEB J. 9:1319-1330. [PubMed] [Google Scholar]
- 29.Mossalayi, M. D., M. Arock, and P. Debre. 1997. CD23/Fc epsilon RII: signaling and clinical implication. Int. Rev. Immunol. 16:129-146. [DOI] [PubMed] [Google Scholar]
- 30.Nathan, C. 2006. Role of iNOS in human host defense. Science 312:1874-1875. [DOI] [PubMed] [Google Scholar]
- 31.Nicholson, S., G. Bonecini-Almeida Mda, J. R. Lapa e Silva, C. Nathan, Q. W. Xie, R. Mumford, J. R. Weidner, J. Calaycay, J. Geng, N. Boechat, C. Linhares, W. Rom, and J. L. Ho. 1996. Inducible nitric oxide synthase in pulmonary alveolar macrophages from patients with tuberculosis. J. Exp. Med. 183:2293-2302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nylen, S., and D. Sacks. 2007. Interleukin-10 and the pathogenesis of human visceral leishmaniasis. Trends Immunol. 28:378-384. [DOI] [PubMed] [Google Scholar]
- 33.Ouaaz, F., B. Sola, F. Issaly, J. P. Kolb, F. Davi, F. Mentz, M. Arock, N. Paul-Eugene, M. Korner, B. Dugas, et al. 1994. Growth arrest and terminal differentiation of leukemic myelomonocytic cells induced through ligation of surface CD23 antigen. Blood 84:3095-3104. [PubMed] [Google Scholar]
- 34.Paul-Eugene, N., D. Mossalayi, M. Sarfati, K. Yamaoka, J. P. Aubry, J. Y. Bonnefoy, B. Dugas, and J. P. Kolb. 1995. Evidence for a role of Fc epsilon RII/CD23 in the IL-4-induced nitric oxide production by normal human mononuclear phagocytes. Cell. Immunol. 163:314-318. [DOI] [PubMed] [Google Scholar]
- 35.Rambert, J., M. Mamani-Matsuda, D. Moynet, P. Dubus, V. Desplat, T. Kauss, J. l. Dehais, T. Schaeverbeke, K. Ezzedine, D. Malvy, P. Vincendeau, and M. D. Mossalayi. 2009. Molecular blocking of CD23 supports its role in the pathogenesis of arthritis. PLoS ONE 4:e4834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ratliff, T. L., J. A. McGarr, C. Abou-Zeid, G. A. Rook, J. L. Stanford, J. Aslanzadeh, and E. J. Brown. 1988. Attachment of mycobacteria to fibronectin-coated surfaces. J. Gen. Microbiol. 134:1307-1313. [DOI] [PubMed] [Google Scholar]
- 37.Roach, S. K., and J. S. Schorey. 2002. Differential regulation of the mitogen-activated protein kinases by pathogenic and nonpathogenic mycobacteria. Infect. Immun. 70:3040-3052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rockett, K. A., R. Brookes, I. Udalova, V. Vidal, A. V. Hill, and D. Kwiatkowski. 1998. 1,25-Dihydroxyvitamin D3 induces nitric oxide synthase and suppresses growth of Mycobacterium tuberculosis in a human macrophage-like cell line. Infect. Immun. 66:5314-5321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rumsey, J., J. F. Valentine, and S. A. Naser. 2006. Inhibition of phagosome maturation and survival of Mycobacterium avium subspecies paratuberculosis in polymorphonuclear leukocytes from Crohn's disease patients. Med. Sci. Monit. 12:BR130-BR139. [PubMed] [Google Scholar]
- 40.Safeukui, I., R. Vatan, M. Dethoua, H. Agbo, G. Haumont, D. Moynet, D. Malvy, P. Vincendeau, D. Mossalayi, and P. Millet. 2008. A role of IgE and CD23/NO immune pathway in age-related resistance of Lewis rats to Plasmodium berghei Anka? Microbes Infect. 10:1411-1416. [DOI] [PubMed] [Google Scholar]
- 41.Sampaio, E. P., H. Z. Elloumi, A. Zelazny, L. Ding, M. L. Paulson, A. Sher, A. L. Bafica, Y. R. Shea, and S. M. Holland. 2008. Mycobacterium abscessus and M. avium trigger Toll-like receptor 2 and distinct cytokine response in human cells. Am. J. Respir. Cell Mol. Biol. 39:431-439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schlesinger, L. S., C. G. Bellinger-Kawahara, N. R. Payne, and M. A. Horwitz. 1990. Phagocytosis of Mycobacterium tuberculosis is mediated by human monocyte complement receptors and complement component C3. J. Immunol. 144:2771-2780. [PubMed] [Google Scholar]
- 43.Subauste, C. S., R. M. Andrade, and M. Wessendarp. 2007. CD40-TRAF6 and autophagy-dependent anti-microbial activity in macrophages. Autophagy 3:245-248. [DOI] [PubMed] [Google Scholar]
- 44.Thoma-Uszynski, S., S. Stenger, O. Takeuchi, M. T. Ochoa, M. Engele, P. A. Sieling, P. F. Barnes, M. Rollinghoff, P. L. Bolcskei, M. Wagner, S. Akira, M. V. Norgard, J. T. Belisle, P. J. Godowski, B. R. Bloom, and R. L. Modlin. 2001. Induction of direct antimicrobial activity through mammalian Toll-like receptors. Science 291:1544-1547. [DOI] [PubMed] [Google Scholar]
- 45.Underhill, D. M., A. Ozinsky, K. D. Smith, and A. Aderem. 1999. Toll-like receptor-2 mediates mycobacterium-induced proinflammatory signaling in macrophages. Proc. Natl. Acad. Sci. USA 96:14459-14463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vankayalapati, R., B. Wizel, B. Samten, D. E. Griffith, H. Shams, M. R. Galland, C. F. Von Reyn, W. M. Girard, R. J. Wallace, Jr., and P. F. Barnes. 2001. Cytokine profiles in immunocompetent persons infected with Mycobacterium avium complex. J. Infect. Dis. 183:478-484. [DOI] [PubMed] [Google Scholar]
- 47.Vouldoukis, I., P. A. Becherel, V. Riveros-Moreno, M. Arock, O. da Silva, P. Debre, D. Mazier, and M. D. Mossalayi. 1997. Interleukin-10 and interleukin-4 inhibit intracellular killing of Leishmania infantum and Leishmania major by human macrophages by decreasing nitric oxide generation. Eur. J. Immunol. 27:860-865. [DOI] [PubMed] [Google Scholar]
- 48.Vouldoukis, I., V. Riveros-Moreno, B. Dugas, F. Ouaaz, P. Becherel, P. Debre, S. Moncada, and M. D. Mossalayi. 1995. The killing of Leishmania major by human macrophages is mediated by nitric oxide induced after ligation of the Fc epsilon RII/CD23 surface antigen. Proc. Natl. Acad. Sci. USA 92:7804-7808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wong, J. M., and T. R. Billiar. 1995. Regulation and function of inducible nitric oxide synthase during sepsis and acute inflammation. Adv. Pharmacol. 34:155-170. [DOI] [PubMed] [Google Scholar]
- 50.Yadav, M., and J. S. Schorey. 2006. The beta-glucan receptor dectin-1 functions together with TLR2 to mediate macrophage activation by mycobacteria. Blood 108:3168-3175. [DOI] [PMC free article] [PubMed] [Google Scholar]