Abstract
P granules are non-membrane-bound organelles found in the germ-line cytoplasm throughout Caenorhabditis elegans development. Like their “germ granule” counterparts in other animals, P granules are thought to act as determinants of the identity and special properties of germ cells, properties that include the unique ability to give rise to all tissues of future generations of an organism. Therefore, understanding how P granules work is critical to understanding how cellular immortality and totipotency are retained, gained, and lost. Here we report on a genomewide RNAi screen in C. elegans, which identified 173 genes that affect the stability, localization, and function of P granules. Many of these genes fall into specific classes with shared P-granule phenotypes, allowing us to better understand how cellular processes such as protein degradation, translation, splicing, nuclear transport, and mRNA homeostasis converge on P-granule assembly and function. One of the more striking phenotypes is caused by the depletion of CSR-1, an Argonaute associated with an endogenous siRNA pathway that functions in the germ line. We show that CSR-1 and two other endo-siRNA pathway members, the RNA-dependent RNA polymerase EGO-1 and the helicase DRH-3, act to antagonize RNA and P-granule accumulation in the germ line. Our findings strengthen the emerging view that germ granules are involved in numerous aspects of RNA metabolism, including an endo-siRNA pathway in germ cells.
GERM granules are large, non-membrane-bound, ribonucleoprotein (RNP) organelles found in the germ-line cytoplasm of most, if not all, animals (Eddy 1975; Saffman and Lasko 1999). The term “germ granule” encompasses what are known as P granules in Caenorhabditis elegans, polar granules in Drosophila melanogaster, germinal granules in Xenopus laevis, and the perinuclear nuage in mouse and human germ cells. These large RNP complexes contain a heterogeneous mixture of RNAs and proteins. To date, most of the known germ granule proteins across species, and all of the known P-granule components in C. elegans, are associated with RNA metabolism, which suggests that a main function of germ granules is post-transcriptional regulation (Strome 2005; Seydoux and Braun 2006). Germ cells are unique in their ability to give rise to all tissues of future generations of an organism. Consequently, germ cells are considered to be both totipotent and immortal. The widespread presence of germ granules in germ cells across species and the ability of germ granule transplantation to induce functional germ cells suggest that germ granules are key determinants of the identity and special properties of germ cells (Smith 1966; Illmensee and Mahowald 1974; Ephrussi and Lehmann 1992). As more components and regulators of germ granules are identified, we will better understand their role in conferring germ cell identity and properties.
One of the earliest identified constitutive components of C. elegans P granules is PGL-1 (Kawasaki et al. 1998). Identification of genes whose loss alters the level and/or distribution of PGL-1 offers an avenue to identify other P-granule components and components that regulate P-granule assembly and stability. For example, the C. elegans VASA homolog GLH-1, another constitutive P-granule component, acts “upstream” of PGL-1; in glh-1 loss-of-function mutants, PGL-1 is not properly localized to P granules and instead a significant portion of PGL-1 is diffusely distributed in the germ-line cytoplasm (Kawasaki et al. 1998; Spike et al. 2008a). More recently another constitutive P-granule component, DEPS-1, was identified in a forward genetic screen for mutants that display PGL-1 localization defects similar to those seen in glh-1(lf) mutants (Spike et al. 2008b). Targeted studies have demonstrated that mutation or RNAi of other genes also disrupts PGL-1 in the germ line. These include genes encoding the nuclear transportins IMB-2, IMB-3, IMB-5, and IMA-3 (J. Ahringer, personal communication; Geles and Adam 2001); the maternal zinc finger protein MEX-1 (Mello et al. 1992; Guedes and Priess 1997); many of the Sm spliceosome components (Barbee et al. 2002); the eIF-5A homolog IFF-1 (Hanazawa et al. 2004); the germ-line enriched protein MEG-1 (Leacock and Reinke 2008); and the oocyte maturation factors OMA-1 and OMA-2 (Shimada et al. 2006). Of these, MEX-1, MEG-1, OMA-1, and the Sm proteins are also P-granule components. This led us to predict that additional genes that affect the composition and behavior of P granules could be identified through a genomewide RNAi screen for PGL-1 accumulation and localization defects. Unlike the forward mutagenesis that identified deps-1, an RNAi screen would not require mutants to be viable and fertile. In addition, the variable expressivity of RNAi phenotypes could be used to discover P-granule defects accompanying the incomplete loss of essential genes.
In this article we report the identification of 173 genes required for the normal assembly and localization of PGL-1. Many of these components fall into specific gene classes with shared P-granule phenotypes, allowing us to better understand how cellular processes converge on P-granule assembly and function. We looked closely at the striking phenotype, accumulation of enlarged P granules, caused by RNAi depletion of CSR-1, an Argonaute that was previously shown to target its mRNA slicing activity through secondary siRNAs (Yigit et al. 2006; Aoki et al. 2007). Loss of other predicted members of a secondary siRNA pathway, the RNA-dependent RNA polymerase EGO-1 and the DExH helicase DRH-3, causes a similar phenotype. We propose that CSR-1(+), EGO-1(+), and DRH-3(+) antagonize the growth and accumulation of P granules through an endogenous siRNA pathway that functions in the germ line.
MATERIALS AND METHODS
Strains and culture:
C. elegans strains were maintained as described by Brenner (1974). Strains used for this study include N2(Bristol) as wild type (WT), SS747 bnIs1(pie-1∷GFP∷PGL-1; unc-119(+)) (Cheeks et al. 2004), ZT3 csr-1(fj54)IV/nT1[qIs51](IV:V), FX892 csr-1(tm892)/unc-24 IV, NL2098 rrf-1(pk1417)I, NL2099 rrf-3(pk1426)II, EL500 ego-1(om84)I/hT2G(I:III), and SS889 rde-3(r459)I/hT2G(I:III).
GFP∷PGL-1 RNAi screen:
SS747 worms carrying a GFP∷PGL-1 transgene inserted into LGI were maintained at 24°. The following screen protocol was followed for 10 weeks:
Day 1. Worms were washed off of 8 recently starved plates into 100 ml of S medium with 2 g of HT115 bacteria and grown at 24° with shaking (150 rpm) (Lewis and Fleming 1995). Three liters of NGM medium + 60 μg/ml Carbenicillin + 1 mm IPTG (Kamath et al. 2001) were poured into 80 24-well plates (1 ml/well).
Day 2. Five 384-well plates from the Ahringer RNAi library (Kamath et al. 2003) were replicated onto Nunc plates containing LB agar + 50 μg/ml tetracycline + 50 μg/ml ampicillin and incubated overnight at 37°.
Day 3. The 5 spotted Nunc plates were used to inoculate LB medium + 60 μg/ml Carbenicillin cultures in 5 384-well plates (50 μl/well), which were grown overnight at 37°. glh-1 RNAi feeding bacteria were inoculated into 4 empty wells of the 384-well plates each week as a positive control.
Day 4. Worms were collected from the liquid culture (started on day 1) by centrifugation. Embryos, prepared by bleach treatment of adults, were transferred to 3 NGM plates without food and allowed to hatch overnight at 24°. RNAi bacteria from the 50-μl cultures were seeded onto the 80 24-well plates and incubated overnight at 37°.
Day 5. The 80 seeded 24-well plates were placed at room temperature. Hatched L1s were washed off the unseeded plates into 100 ml of S media + 2 g of bacteria and incubated at 24° with shaking.
Day 6. After 30–33 hr of growth, the synchronized worms (mainly L3/L4 larvae) were washed and distributed onto the 80 24-well plates using a COPAS Biosort (Union Biometrica) to deliver 12–15 worms/well. Worms were incubated on RNAi plates at 24°.
Day 7. Approximately 28 hr after sorting, worms were placed at 15° to slow growth. All observable P0 adults and F1 embryos in each well were screened for GFP∷PGL-1 phenotypes using a Zeiss SV11 fluorescence dissecting microscope fitted with a stereo and 10× compound objective. Secondary and tertiary screens were performed using a Leica MZ16F fluorescence dissecting microscope fitted with a stereo and 5× compound objective.
Immunocytochemistry:
Embryos and worms were fixed using methanol/acetone (Strome and Wood 1983). Antibody dilutions were 1:30,000 rabbit anti-PGL-1 (Kawasaki et al. 1998), 1:5000 mouse anti-NPC MAb414 (Covance) (Blobel 1985), 1:5000 rat anti-PGL-3 (Kawasaki et al. 2004), 1:1000 rabbit anti-GLH-1 (Gruidl et al. 1996; Kawasaki et al. 2004), 1:400 Alexa Fluor 488 goat anti-rabbit IgG, 1:400 Alexa Fluor 594 goat anti-mouse IgG, and 1:400 Alexa Fluor 594 goat anti-rat IgG (Molecular Probes, Eugene, OR). Images were acquired with a Volocity spinning disk confocal system (Perkin-Elmer/Improvision, Norwalk, CT) fitted on a Nikon Eclipse TE2000-E inverted microscope.
Quantitative RT–PCR:
Four biological replicates of N2 worms fed empty vector or csr-1 RNAi bacteria at 24° for 30 hr were washed and frozen prior to RNA isolation using Trizol (Invitrogen, Carlsbad, CA). cDNA was synthesized using Superscript III First Strand synthesis (Invitrogen). Quantitative PCR was performed using a 2× SYBR green mix (Roche, Indianapolis) on a Roche LightCycler 480. pgl-1, pgl-3, and glh-1 amplicons were normalized to act-2. Primers were as follows: pgl-1, f-tgttgttggagtcgcgaag; pgl-1, r-tccgcaatggctcgtctt; pgl-3, f-ctcgagcagtgcttttctca; pgl-3, r-tttcgttgttcaactcgcttt; glh-1, f-actctggttttggggaagga; glh-1, r-gtcactcgatcgatgtcctg; act-2, f-cgtcatcaaggagtcatggtc; and act-2, r-catgtcgtcccagttggtaa.
SYTO14 staining of RNA:
Gonads from N2 worms 3 days posthatching were dissected in 118 mm NaCl, 48 mm KCl, and 5 um SYTO14 (Schisa et al. 2001), incubated for 10 min, and then imaged with a confocal microscope.
RNAi conditions:
RNAi depletion of csr-1, ego-1, and drh-3 was performed by feeding bacteria from the Ahringer library (Kamath et al. 2003) seeded on agar containing 50 μg/ml carbenicillin and 1 mm IPTG. Worms at the L3/L4 stage were fed RNAi bacteria for 30 hr before imaging or fixation. Embryos or worms labeled as wild type were fed HT115 bacteria containing the L4440 empty vector as a control. To enhance gene product depletion, worms used for SYTO14 staining were fed either control or RNAi bacteria starting at the L2/L3 stage for 45 hr before dissection. On the basis of sequence data from the Ahringer lab (J. Ahringer, personal communication), we resequenced RNAi clones that had been flagged as targeting the wrong sequence. Correct gene targets are as follows: JA:B0464.7 targets atx-2, JA:Y55F3A_739.a targets cct-8, JA:Y65B4B_13.b targets imb-5, JA:ZK1058.2 targets rpn-1, JA:Y47D9A.d targets rps-3, JA:C24H12.6 targets an AT-rich area with multiple targets, JA:T05H10.3 targets F15E11.5, and JA:ZK675.1 targets F59E12.11.
Western blot analysis:
L3 larvae were fed control or csr-1(RNAi) bacteria for 30 hr. Forty adult worms boiled in SDS–PAGE sample buffer were loaded in each lane. Blots were probed with 1:50,000 rabbit anti-PGL-1 and 1:1000 mouse anti-tubulin [Sigma (St. Louis) DM 1a] for a loading control. Normalized ratios of PGL-1 were compared between six samples of control and csr-1(RNAi) lysates.
RESULTS
Multiple components are required for the proper organization and localization of PGL-1:
To identify effectors of P-granule assembly, stability, and function, we used the Ahringer RNAi feeding library (Kamath et al. 2003), which targets 16,757 genes (87% of the C. elegans genome), to screen for abnormal accumulation and/or localization of GFP-tagged PGL-1 in adult germ lines and their embryonic progeny (Figure 1A). Historically, PGL-1 antibody and GFP∷PGL-1 have been used to identify and characterize P granules, so often the terms P granules and PGL-1 granules are used interchangeably; however, the phenotypes observed in our screen rely solely on PGL-1 localization unless otherwise stated. Our visual GFP screen allowed us to observe patterns that deviated from normal GFP∷PGL-1 patterns, including differences in GFP intensity, subcellular and organismal distribution, and granule shape throughout adult germ-line and embryonic development (Table 1; Figure 1, B and C). We initially identified 234 genes whose depletion by RNAi caused GFP∷PGL-1 phenotypes. These initial positives were taken through two additional rounds of screening, which narrowed our list of positives to 173 genes whose depletion consistently caused aberrant GFP∷PGL-1 phenotypes (Table 1). GFP∷PGL-1 phenotypes observed under the fluorescence dissecting scope were placed in broad classes of high, low, or undetectable (und.) GFP∷PGL-1 accumulation (Figure 1B, Table 1). GFP∷PGL-1 dispersed in the cytoplasm instead of in perinuclear aggregates was classified as “diffuse”; this category included dispersed small granules and PGL-1 signal homogeneously distributed in the cytoplasm. Altered GFP distributions were scored as throughout the embryo or restricted to the germ-line blastomeres. Examples of wild-type GFP∷PGL-1 accumulation, diffuse GFP∷PGL-1 in glh-1(RNAi), and high GFP∷PGL-1 accumulation in csr-1(RNAi) are shown in Figure 1C.
Figure 1.—
Genomewide GFP∷PGL-1 screen. (A) GFP∷PGL-1 screen strategy. L3/L4 stage worms were placed on bacteria from the Ahringer RNAi feeding library for 30 hr at 24°. GFP∷PGL-1 phenotypes were examined in the adult (P0) germ line of living animals and their embryonic progeny (F1) under a fluorescence dissecting microscope. (B) RNAi depletion of 173 different genes caused GFP∷PGL-1 phenotypes in F1 embryos and/or phenotypes in P0 germ lines. The range of phenotypes included diffuse, high, low, and undetectable (und.) GFP∷PGL-1 accumulation. “Diffuse” GFP∷PGL-1 included both dispersed small granules and more homogeneously distributed GFP∷PGL-1. Diffuse GFP∷PGL-1 in embryos was either throughout the embryo (see glh-1) or restricted to germ-line progenitor (P) cells. (C) GFP∷PGL-1 accumulation in the P0 germ line (top) and F1 embryos (bottom) of wild-type, glh-1, and csr-1 RNAi worms showing wild-type, diffuse, and high-accumulation phenotypes, respectively. Wild-type and control embryos display small GFP∷PGL-1 granules in somatic cells from the ∼24- to ∼200-cell stage. Small somatic GFP∷PGL-1 granules are much more prevalent in glh-1 and csr-1 RNAi embryos. csr-1 RNAi embryos also often display large perinuclear GFP∷PGL-1 granules in multiple cells (also see Figure 7D). Bars, 25 μm.
TABLE 1.
GFP∷PGL-1 screen positives
| Gene name | Wormbase ID | Gene class codec | GFP∷PGL-1 in the P0 germ line |
GFP∷PGL-1 in F1 embryos |
PGL-1 phenotype preceded by general embryo defects? | Endogenous PGL-1 |
||||
|---|---|---|---|---|---|---|---|---|---|---|
| Description | Altered level | Altered pattern | Altered leveld | Altered pattern | Stained | Same as GFP | ||||
| adr-1 | H15N14.1 | RNA adenosine deaminase | Abnormal | Yes | ||||||
| air-1 | K07C11.2 | Aurora kinase | C | Low | Diffuse | Yes | ||||
| apc-2 | K06H7.6 | Anaphase-promoting complex 2 | U | Low | Diffuse | Yes | ||||
| arl-1 | F54C9.10 | GTP-binding ADP-ribosylation factor | G | Diffuse | Yes | |||||
| atx-2 | D2045.1a | Ataxin-related protein | Low | Diffuse | No | X | X | |||
| bir-1 | T27F2.3 | Aurora B kinase complex | C | Low | No | X | X | |||
| cct-2 | T21B10.7 | Cytosolic “T complex” chaperonin | T | Diffuse | No | X | X | |||
| cct-3 | F54A3.3 | Cytosolic T complex chaperonin | T | Low | Diffuse | Yes | ||||
| cct-7 | T10B5.5 | Cytosolic T complex chaperonin | T | Low | Yes | |||||
| cct-8 | Y55F3AR.3ab | Cytosolic T complex chaperonin | T | Low | Diffuse | Yes | X | X | ||
| cdc-25.1 | K06A5.7 | Cell division cycle 25.1 phosphatase | C | Low | Diffuse. Puncta in multiple cells | No | X | X | ||
| cdc-42 | R07G3.1 | Cell division cycle 42 Rho GTPase | C | Low | Diffuse | No | X | |||
| cdk-1 | T05G5.3 | Cyclin-dependent kinase 1 | C | Low | Diffuse | Yes | ||||
| cdk-9 | H25P06.2 | Cyclin-dependent kinase 9 | C | Low or und. | Yes | X | X | |||
| ceh-21 | T26C11.6 | ONECUT class homeobox protein | Low | No | X | |||||
| cgh-1 | C07H6.5 | Germ-line DEAD-box RNA helicase | Low | No | X | X | ||||
| cpf-2 | F56A8.6 | Cleavage and polyadenylation factor | S | Low | Diffuse in P cells | No | X | |||
| cpl-1 | T03E6.7 | Cathepsin L-like cysteine protease 1 | Low | Diffuse | Yes | |||||
| csr-1 | F20D12.1 | Secondary RNA slicing Argonaute | High | Large granules | High | No | X | X | ||
| cyb-3 | T06E6.2 | Cyclin B3 protein | Low | Diffuse | Yes | |||||
| cyk-4 | K08E3.6 | Rho GTPase-activating protein (GAP) | C | Und. | Yes | |||||
| cyp-31A3 | Y17G9B.3 | CYP7B1 homolog —vitamin D deficiency | Low | Abnormal | No | X | X | |||
| dhc-1 | T21E12.4 | Dynein heavy chain | C | Diffuse | Yes | |||||
| dlc-1 | T26A5.9 | Dynein light chain | N | Low | Yes | |||||
| dnc-1 | ZK593.5 | Dynactin/GLUED orthologs | C | Low | Yes | X | X | |||
| dom-6 | C09G4.3 | Cks/Suc1 ortholog. promotes cyclin B degradation | C | Low | Diffuse | Yes | X | X | ||
| dpf-1 | T23F1.7 | Prolyl oligopeptidase | Low | Diffuse | No | X | ||||
| dyci-1 | C17H12.1 | Dynein cytoplasmic intermediate chain | Diffuse | Yes | ||||||
| eft-1 | ZK328.2 | Tu domain containing elongation factor | High | Diffuse | High in P cells | Diffuse in soma | No | X | X | |
| egg-3 | F44F4.2 | Protein–tyrosine phosphatase | C | Low | Diffuse | Yes | ||||
| egl-45 | C27D11.1 | Eukaryotic translation initiation factor 3 subunit 10 | R | Low or und. | No | X | X | |||
| eif-3.F | D2013.7 | Eukaryotic translation initiation factor 3 subunit 5 | R | Low | No | X | X | |||
| elc-1 | Y82E9BR.15 | Elongin C | U | Undetectable | Und. | Yes | X | X | ||
| emb-9 | K04H4.1 | Collagen | Low | No | X | |||||
| fbxa-81 | T24C2.4 | F-box domain | U | Low | No | X | ||||
| fbxb-51 | K05F6.3 | F-box domain | U | High | High in P cells | Large granules | No | X | X | |
| fbxb-60 | F55C9.7 | F-box domain | U | High | No | X | ||||
| flp-13 | F33D4.3 | FMRF-like amide neurotransmitter | High | High in P cells | Diffuse in soma | No | X | |||
| glb-27 | W01C9.5 | Unknown | Low | No | X | |||||
| gon-1 | F25H8.3 | Secreted metalloprotease (MPT) | Low | Diffuse | No | X | X | |||
| gsk-3 | Y18D10A.5 | Glycogen synthase kinase required for polarization of EMS and P2 blastomeres | C | Low | Diffuse in multiple cells | Yes | ||||
| his-40 | K03A1.1 | Similarity to histones (pseudogene?) | Diffuse in P cells | Yes | ||||||
| his-48 | B0035.8 | H2B histone | Low | Yes | ||||||
| hsp-6 | C37H5.8 | HSP70-like mitochondrial chaperone | Low | No | X | |||||
| ify-1 | C27A2.3 | Ligand of Fizzy protein. Securin? | C | Diffuse | Yes | |||||
| imb-4 | ZK742.1 | Exportin-1/CRM1 nuclear exportin | N | Diffuse in P cells | No | X | X | |||
| imb-5 | Y48G1A.5ab | CSE1/CAS-like nuclear importin | N | Diffuse in P cells | No | X | X | |||
| inx-8 | ZK792.2 | Innexin/GAP junction component | Low | No | X | |||||
| let-70 | M7.1 | E2 ubiquitin-conjugating enzyme | U | Low | Diffuse | Yes | ||||
| let-711 | F57B9.2 | NOT1 deadenylation component | High | Diffuse | High | No | X | X | ||
| let-92 | F38H4.9 | Catalytic subunit of PP2A phosphatase | Diffuse | Yes | X | X | ||||
| let-99 | K08E7.3 | DEP domain. Spindle orientation | C | Low | No | X | X | |||
| lin-41 | C12C8.3 | RING finger B box coiled-coil protein | Low in some | No | X | |||||
| lmn-1 | DY3.2 | Nuclear type B lamin | N | Diffuse in P cells | No | X | X | |||
| mag-1 | R09B3.5 | Mago nashi. Exon–exon junction factor | S | Low in some | No | X | ||||
| mbk-2 | F49E11.1 | Minibrain DYRK2 kinase | C | Low | No | X | X | |||
| mcm-2 | Y17G7B.5 | MCM licensing factor 2 | M | Low | Diffuse in P cells | Yes | X | X | ||
| mcm-4 | Y39G10AR.14 | MCM licensing factor 4 | M | Low | Diffuse in P cells, puncta in soma | Yes | X | X | ||
| mcm-5 | R10E4.4 | MCM licensing factor 5 | M | Diffuse in P cells | Yes | X | X | |||
| mcm-6 | ZK632.1 | MCM licensing factor 6 | M | Diffuse in P cells, high puncta in soma | Yes | X | X | |||
| mcm-7 | F32D1.10 | MCM licensing factor 7 | M | Diffuse in P cells | Yes | X | X | |||
| mdt-6 | Y57E12AL.5 | Transcriptioinal mediator Med6-like | Low | Diffuse | Yes | |||||
| mel-26 | ZK858.4 | Adaptor of the E3 ubiquitin ligase | U | Diffuse | Yes | |||||
| mix-1 | M106.1 | SMC2-like condensing complex factor | Low | No | X | |||||
| nmy-2 | F20G4.3 | Nonmuscle myosin II | C | Low | Diffuse | Low | Diffuse | Yes | X | X |
| npp-10 | ZK328.5 | NUP98-like nucleoporin | N | Diffuse in P cells | No | X | X | |||
| npp-20 | Y77E11A.13 | Nucleoporin | N | Low | Diffuse | Yes | X | X | ||
| npp-6 | F56A3.3 | NUP160-like nucleoporin | N | Diffuse in P cells | No | X | X | |||
| npp-7 | T19B4.2 | Nucleoporin | N | Diffuse in P cells | No | X | X | |||
| npp-9 | F59A2.1 | Nucleoporin | N | Diffuse in P cells | No | X | X | |||
| paa-1 | F48E8.5 | Regulatory subunit of PP2A phosphatase | Diffuse | Yes | X | X | ||||
| par-1 | H39E23.1 | Serine/threonine kinase | C | Low or und. | Diffuse | No | X | X | ||
| par-2 | F58B6.3 | C3HC4-type RING-finger protein | C | Low or und. | Yes | |||||
| par-5 | M117.2 | 14-3-3-containing protein | C | Low | Diffuse | No | X | X | ||
| par-6 | T26E3.3 | PDZ-domain-containing protein | C | Low or und. | Diffuse | Yes | ||||
| pas-4 | C36B1.4 | α4 20S proteasome core subunit | P | Low or und. | Yes | X | X | |||
| pas-5 | F25H2.9 | α5 20S proteasome core subunit | P | Low or und. | Yes | |||||
| pbs-1 | K08D12.1 | β1 20S proteasome core subunit | P | Low | Diffuse | Yes | X | X | ||
| pbs-3 | Y38A8.2 | β3 20S proteasome core subunit | P | Und. in some | Yes | |||||
| pbs-4 | T20F5.2 | β4 20S proteasome core subunit | P | Diffuse | Yes | |||||
| pbs-5 | K05C4.1 | β5 20S proteasome core subunit | P | Diffuse | Low | Diffuse | Yes | X | X | |
| pbs-6 | C02F5.9 | β6 20S proteasome core subunit | P | Diffuse | Yes | |||||
| pbs-7 | F39H11.5 | β7 20S proteasome core subunit | P | Diffuse | Yes | |||||
| pgl-1 | ZK381.4 | RGG box motifs. | Und. | No | ||||||
| phi-10 | Y46G5A.4 | DEAD-box RNA helicase BRR2 | S | High | High | No | X | X | ||
| phi-12 | M03F8.3 | HAT repeat. mRNA splicing | S | Low | No | X | X | |||
| phi-21 | T05H4.6 | Translation release factor | Low | Diffuse | No | X | X | |||
| phi-6 | K02F2.3 | snRNP-associated factor | S | High | High in P cells | Diffuse in soma | No | X | X | |
| phi-62 | F49C12.12 | Unknown | Low | No? | X | X | ||||
| phi-7 | D1081.8 | mRNA splicing protein CDC5 (Myb) | S | High | Diffuse | High in P cells | No | X | X | |
| plk-1 | C14B9.4 | Serine/threonine polo-like kinase | C | Low | Diffuse | Yes | X | X | ||
| pri-1 | F58A4.4 | DNA Pol α-primase subunit D | Low | No | X | X | ||||
| prp-8 | C50C3.6 | U5 snRNA-associated splicing factor | S | High | Diffuse | High | Diffuse | No | X | X |
| prs-1 | T20H4.3 | Proline tRNA ligase | Low | No? | X | X | ||||
| puf-3 | Y45F10A.2 | PUF family RNA-binding protein | Diffuse | No | X | X | ||||
| ran-1 | K01G5.4 | RAN-associated GTP-binding protein | N | Low | Diffuse | No | X | X | ||
| ran-4 | R05D11.3 | Nuclear transport factor 2 (NTF2) | N | Low | Diffuse in P cells | No | X | X | ||
| rnp-4 | R07E5.14 | Exon–exon junction complex factor | S | Diffuse in P cells | Yes | |||||
| rnr-1 | T23G5.1 | Ribonucleotide reductase | Low or und. | Diffuse in P cells | No | X | X | |||
| rnr-2 | C03C10.3 | Ribonucleotide reductase | Diffuse in P cells | No | X | X | ||||
| rpa-1 | F18A1.5 | Large subunit of replication protein A | Diffuse in P cells, high puncta in soma | Yes | ||||||
| rpb-2 | C26E6.4 | RNA Pol II β-subunit | Diffuse in P cells | Yes | X | X | ||||
| rpl-12 | JC8.3 | Ribosomal protein large subunit 12 | R | Low | No | X | ||||
| rpl-18 | Y45F10D.12 | Ribosomal protein large subunit 18 | R | Low | Yes | X | X | |||
| rpl-20 | E04A4.8 | Ribosomal protein large subunit 18a | R | Low | No | X | X | |||
| rpl-36 | F37C12.4 | Ribosomal protein large subunit 36 | R | Low | No | X | X | |||
| rpn-1 | T22D1.9a | Proteasome regulatory particle | P | Diffuse | Yes | |||||
| rpn-11 | K07D4.3 | Proteasome regulatory particle | P | Diffuse in P cells | Yes | |||||
| rpn-3 | C30C11.2 | Proteasome regulatory particle | P | Diffuse | Yes | |||||
| rps-0 | B0393.1 | Ribosomal protein small subunit 2A | R | Low | No | X | X | |||
| rps-20 | Y105E8A.16 | Ribosomal protein small subunit 20 | R | Low | Yes | |||||
| rps-22 | F53A3.3 | Ribosomal protein small subunit 15a | R | Low | No | X | X | |||
| rps-24 | T07A9.11 | Ribosomal protein small subunit 24 | R | Low | No | X | ||||
| rps-3 | C23G10.3a | Ribosomal protein small subunit 3 | R | Low | No | X | ||||
| rps-5 | T05E11.1 | Ribosomal protein small subunit 5 | R | Low | No | X | ||||
| rps-8 | F42C5.8b | Ribosomal protein small subunit 8 | R | Low | Diffuse | Yes | ||||
| rps-9 | F40F8.10 | Ribosomal protein small subunit 9 | R | Low | No | X | X | |||
| rpt-1 | C52E4.4 | Proteasome regulatory particle | P | Low | Diffuse | Yes | ||||
| rpt-3 | F23F12.6 | Proteasome regulatory particle | P | Diffuse | Yes | |||||
| rpt-5 | F56H1.4 | Proteasome regulatory particle | P | Diffuse | Yes | |||||
| rsp-7 | D2089.1 | SR protein (splicing factor) 7 | S | Low | Diffuse in P cells | No | X | X | ||
| ruvb-1 | C27H6.2 | RuvB-like DNA helicase | Low | No | X | |||||
| ruvb-2 | T22D1.10 | RuvB-like DNA helicase | Low | No | X | |||||
| scp-1 | D2013.8 | WD repeat, sreb cleavage protein | G | Low in some | No | X | ||||
| skp-1 | T27F2.1 | SKI-binding protein (SKIP) orthologs | S | High | High | Diffuse in soma | No | X | X | |
| snr-2 | W08E3.1 | Core splicesome component | S | Low | Yes | |||||
| spd-5 | F56A3.4 | Dynein heavy chain 1 | C | Diffuse | Yes | |||||
| sqv-6 | Y50D4C.4 | Xylosyltransferase | C | Low or und. | Yes | |||||
| srh-81 | W03F9.7 | Serpentine receptor class H | Low | No | X | |||||
| sri-25 | C41G6.10 | Serpentine receptor class I | Low | No | X | |||||
| srj-6 | F31F4.8 | Serpentine receptor class J | Low | No | X | |||||
| sru-41 | Y51H7BR.6 | Serpentine receptor class U | Low | No | X | |||||
| srx-38 | C03A7.5 | Serpentine receptor class X | Low | No | X | |||||
| tag-198 | F09G8.2 | Putative DNase II | Low | No | X | |||||
| tag-49 | AC7.1 | G-protein receptor | Low | No | X | X | ||||
| tbb-2 | C36E8.5 | β-Tubulin | Diffuse | Yes | X | X | ||||
| tbg-1 | F58A4.8 | γ-Tubulin | C | Low | Yes | |||||
| ttr-14 | T05A10.3 | Transthyretin-related family domain | Low | No | X | |||||
| uba-1 | C47E12.5 | Ubiquitin UBA1/UBE1 related | U | Low | Diffuse | Yes | X | X | ||
| ubl-1 | H06I04.4 | Ribosomal S31 fused to ubiquitin | U | Low | Diffuse | Low | Diffuse | No | X | X |
| unc-112 | C47E8.7 | PH domain mitogen-inducible gene-2 | Low | No | X | |||||
| unc-32 | ZK637.8 | Vacuolar ATPase subunit | V | Low | Yes | |||||
| vha-13 | Y49A3A.2 | Vacuolar ATPase subunit | V | Low | Yes | X | X | |||
| vha-19 | Y55H10A.1 | Vacuolar ATPase subunit | V | Low | Yes | |||||
| vha-2 | R10E11.2 | Vacuolar ATPase subunit | V | Low | No | X | X | |||
| vps-11 | R06F6.2 | Vacuolar protein sorting factor | V | Low | No | X | ||||
| zen-4 | M03D4.1 | Plus-end kinesin-like motor protein | C | Und. | Yes | |||||
| zyg-9 | F22B5.7 | Microtubule-association protein | C | Low | Diffuse | Yes | ||||
| AT-rich areaa | Low | No | X | |||||||
| B0361.10 | SNARE synaptobrevin/VAMP | G | Low | No | X | |||||
| C01G10.14 | Major sperm protein (MSP) domain | Low | No | X | ||||||
| C06A5.1 | Putative integrator complex subunit | S | Low in late embryo | Yes | X | X | ||||
| C34D1.2 | DM DNA-binding domain | Low | No | X | ||||||
| C35A5.4 | Major sperm protein (MSP) domain | Low | No | X | ||||||
| C41G7.3 | TSPO homolog. Putative cholesterol transporter | High in P cells | Large granules in early embryo | No | X | X | ||||
| C42D4.13 | Unknown | Low | No | X | ||||||
| F01G4.3 | DEAD/DEAH box helicase domain | Low | Diffuse | No | X | |||||
| F15E11.5a | Unknown | Low | No | X | ||||||
| F22B5.10 | TMCO1 homolog | C | Und. in some | No | X | X | ||||
| F29G6.1 | Serine protease inhibitor domain | Low | Diffuse | No | X | |||||
| F55C5.7 | Protein kinase, MIT, PX domains | Low | No | X | ||||||
| F59D6.7 | Calcium-binding pH regulator | Low | No | X | ||||||
| F59E10.3 | ζ-Subunit of coatomer (COPI) | G | Low | Diffuse | Low | No | X | |||
| F59E12.11a | Unknown | Diffuse | No | X | X | |||||
| K06H7.1 | Serine/threonine protein kinase | Low | Diffuse | Yes | ||||||
| K12H4.3 | rRNA maturation of 60S ribosome | Low | No | X | ||||||
| T03G6.3 | Phosphodiesterase | Low | No | X | ||||||
| T04A8.7 | Glucosidase acid β-orthologs | Low or und. | No | X | X | |||||
| T06E6.1 | Possible 60S ribosomal Nop7 factor | Low | No | X | ||||||
| T12D8.6 | Calmodulin containing | Low or und. | Yes | |||||||
| T19C3.6 | Unknown | Low | No | X | ||||||
| Y47D3A.29 | DNA polymerase primase | Low | Diffuse in P cells | Yes | ||||||
|
ZC477.4 |
Unknown |
Low |
No |
X |
||||||
Targeted gene originally misannotated in Ahringer RNAi Library. Correct sequenced target is shown.
Gene obtained more than once in the screen.
Gene class letter code (see Table 2).
Und., undetectable.
We wanted to know if GFP∷PGL-1 defects reflect defects associated with endogenous PGL-1 or instead are specific to the GFP-tagged transgenic protein. The GFP∷PGL-1 transgene was integrated into the genome through particle bombardment (Cheeks et al. 2004) and as a consequence should be present at low copy number and shielded from transgene silencing effects. However, since the reporter uses the germ-line-specific pie-1 promoter (Cheeks et al. 2004), it is possible that some RNAi depletions specifically affect expression of the transgene and not endogenous PGL-1. It is also possible that the post-transcriptional processing, expression, or stability of transgenic PGL-1 is affected by the GFP tag. Therefore, we repeated RNAi against 126 screen positives in wild-type (not transgenic) worms, stained with antibody against PGL-1, and examined the P0 germ line and F1 embryos using both widefield fluorescence and spinning disk confocal microscopy (Table 1). In the majority of RNAi depletions that we tested (76 of 126), endogenous PGL-1 recapitulated the GFP∷PGL-1 phenotypes. Furthermore, the improved resolution of fixed and stained samples allowed us to examine the PGL-1 phenotypes in greater detail. However, subtle differences in PGL-1 levels were harder to see in fixed and stained embryos than by GFP imaging. Fifty of 126 RNAi depletions that caused reduced levels of GFP∷PGL-1 did not cause noticeably reduced levels of endogenous PGL-1. This set of 50 genes may specifically affect expression of the GFP transgene or may result in changes in PGL-1 protein levels or distribution that are more easily seen by GFP than by antibody staining.
As expected, the list of screen positives is significantly enriched for genes expressed in the germ line. Seventy-four of the 173 genes identified are in the set of 3144 genes with germ-line-enriched expression defined by Reinke et al. (2004), which is a 2.7-fold enrichment over the number expected by chance. One hundred twenty-seven of the 173 genes identified are in the set of 4699 genes expressed in dissected gonads as detected using SAGE (Wang et al. 2009), which is a 3.5-fold enrichment over the number expected by chance.
The majority of screen positives were grouped into large gene classes that display common phenotypes (Table 2). These classes are discussed below, followed by more in-depth analysis of an argonaute gene identified in our screen.
TABLE 2.
Most prominent PGL-1 phenotypes associated with each gene class
| Gene class (Table 1 code) | Genesb | Most common PGL-1 phenotype in class |
|---|---|---|
| Cell cycle/polarity/cell division(C) | air-1, bir-1, cdc-25.1, cdc-42, cdk-1, cdk-9, cyk-4, dhc-1, dnc-1, dom-6, egg-3, gsk-3, ify-1, let-99, mbk-2, nmy-2, par-1, par-2, par-5, par-6, plk-1, spd-5, sqv-6, tbb-2, tbg-1, zen-4, zyg-9, F22B5.10 | Low PGL-1 level, likely caused by segregation defects in early F1 embryos |
| Proteasome/ubiquitin(P/U) | Proteasome: pas-4, pas-5, pbs-1, pbs-3, pbs-4, pbs-5, pbs-6, pbs-7, rpn-1, rpn-3, rpn-11, rpt-1, rpt-3, rpt-5. Ubiquitin: apc-2, elc-1, fbxa-81, fbxb-51, fbxb-60, let-70, mel-26, uba-1, ubl-1 | Diffuse PGL-1 in F1 embryos |
| Ribosome(R) | egl-45, eif-3.f, rpl-12, rpl-18, rpl-20, rpl-36, rps-0, rps-3, rps-5, rps-8c, rps-9, rps-20, rps-22, rps-24 | Low PGL-1 in F1 embryos |
| Splicing(S) | cpf-2, mag-1, phi-6, phi-7, phi-10, phi-12, prp-8, rnp-4, rsp-7, skp-1, snr-2, C06A5.1 | High PGL-1 in P0 germ lines and F1 embryos |
| Nuclear pore/envelope/transport(N) | dlc-1, imb-4, imb-5c, lmn-1, npp-6, npp-7, npp-9, npp-10, npp-20, ran-1, ran-4 | PGL-1 granules not associated with the nuclear envelope, and PGL-1 diffuse in the cytoplasm of P cells |
| MCM licensing(M) | mcm-2, mcm-4, mcm-5, mcm-6, mcm-7 | Diffuse PGL-1 in the cytoplasm of P cells; associated with severe embryonic defects |
| Vacuole(V) | unc-32, vha-2, vha-13, vha-19, vps-11 | Low PGL-1 in F1 embryos |
| Chaperonin containing TCP-1 (CCT)(T) | cct-2, cct-3, cct-7, cct-8c | Low and diffuse PGL-1 in F1 embryos |
| Golgi/ER(G) | arl-1, scp-1, B0361.10, F59E10.3 | Low PGL-1 in F1 embryos |
| Othera | 59 genes | |
| Unknown |
8 genes |
Groups with fewer than three genes not shown.
Genes in boldface type exhibit the most common PGL-1 phenotype in their class (described in right column).
Genes represented multiple times in the screen.
Cell cycle/polarity/cell division:
The largest group of screen positives can be classified by their role in the cell cycle, establishing cell polarity, and the segregation of cellular components during early cell divisions. This group includes 28 genes of the 173 total screen positives (Table 2). In wild-type animals, P granules are evenly distributed in oocytes and newly fertilized zygotes, but prior to each P-cell division the granules are segregated to the cytoplasm destined for the next P-cell daughter (Strome and Wood 1982). P granules left behind in somatic daughters are disassembled or degraded (Hird et al. 1996; Saffman and Lasko 1999; DeRenzo et al. 2003; Spike and Strome 2003). RNAi depletions that cause early cell cycle arrest or altered cell division patterns and eventually embryonic lethality may lead to a diffuse or “missegregated” P-granule pattern. In fact, P granules have been used as a marker to study cell polarity for >20 years, and some examples of P-granule missegregation have been intensively studied (e.g., Gonczy and Rose 2005; Cowan and Hyman 2007). In our secondary and tertiary screens, we more closely examined whether a defective GFP∷PGL-1 phenotype appeared to be correlated with abnormal nuclear morphology, embryo patterning defects, and/or early arrest (Table 1). PGL-1 patterning defects appeared to precede general embryo defects for only 8 of the 28 genes in this class (bir-1, cdc-25.1, cdc-42, let-99, mbk-2, par-1, par-5, and F22B5.10).
As shown in Figure 2, RNAi depletion of genes in the cell cycle/polarity/cell division class tended to show two types of phenotypes. As exemplified by par-1 and cdk-9, RNAi depletion caused PGL-1 to be present in all cells of early embryos and reduced or absent from all cells of later embryos. This pattern likely reflects both missegregation of P granules and failure to properly separate germ-line and somatic lineages, with their different abilities to retain (germ-line) vs. disassemble (somatic) P granules (Kemphues et al. 1988). As exemplified by let-99, par-5, and cdc-25.1, RNAi depletion caused PGL-1 to be present in all cells of early embryos and to persist in numerous cells of later embryos (Rose and Kemphues 1998; Morton et al. 2002). We suspect that the extra PGL-1-containing cells in later embryos are in most cases a consequence of the earlier segregation defects, but another interesting possibility is that they arise by extra divisions of the P-cell lineage.
Figure 2.—
Cell cycle/cell division/polarity class. Antibody-stained PGL-1 (grayscale, green in merge) and nuclear pores (red) and DAPI-stained DNA (blue) in fixed embryos at the 4-, ∼30-, and ∼150-cell stages of development are shown. PGL-1 segregation defects are shown at the 4-cell stage of par-1, cdk-9, cdc-25.1, par-5, and let-99 RNAi embryos. In later embryos, the absence of germ-line progenitors (par-1) or improper P-granule segregation accompanied by general embryo defects (cdk-9) caused PGL-1 to be low or undetectable. Embryos depleted of cdc-25.1, par-5, and let-99 have multiple P-granule-containing cells later in development. Bar, 10 μm.
Proteasome/ubiquitin:
The next largest group of screen positives contains 23 protein degradation components from the proteasome and ubiquitin pathways (Table 2). RNAi depletion of the majority of genes in this class resulted in a characteristic GFP∷PGL-1 phenotype: the uterus was filled with early embryos that exhibit a diffuse PGL-1 distributed throughout the embryo (Table 1, Figure 3A). Often the embryonic phenotype was accompanied by diffuse PGL-1 distribution in the P0 germ line. The PGL-1 phenotype displayed by this class is likely a result of compromised protein degradation mechanisms, which may stabilize PGL-1, and by extension P-granule proteins, in both the P0 germ line and in embryos (DeRenzo et al. 2003; Spike and Strome 2003). This phenotype is probably compounded by the early embryonic arrest associated with the depletion of all but 4 of the 23 genes. These 4 include ubl-1(RNAi), whose RNAi depletion caused PGL-1 granules to persist in multiple cells prior to embryonic arrest, and the F-box-containing genes fbxa-81, fbxb-51, and fbxb-60, whose depletion did not cause embryonic lethality under our experimental conditions. This class emphasizes the importance of protein degradation in regulating P-granule patterns in germ lines and embryos.
Figure 3.—
Proteasome/ubiquitin and ribosome classes. GFP∷PGL-1 (green) in live worms (A) and PGL-1 (green), nuclear pores (red), and DNA (blue) in fixed embryos (B) are shown. Dashed boxes indicate the location of zoomed images on the right. (A) Proteasome/ubiquitin class. Depletion of proteasome (e.g., pbs-5) and ubiquitin components often causes early embryonic arrest with diffuse PGL-1 throughout the embryo. Arrested embryos accumulate in the uterus (bracketed bar) after proteasome or ubiquitin depletion, which creates a distinctive GFP∷PGL-1 phenotype. (B) Ribosome class. Four-cell embryos are shown. Depletion of core ribosomal components such as rps-0, rpl-20, and rpl-36 results in reduced PGL-1 and smaller granules (arrowheads). Bars: (A) 50 μm and (B) 10 μm.
Ribosome:
We obtained 14 genes that encode ribosomal components in our screen (Table 2). In contrast to the groups mentioned above, most PGL-1 phenotypes in this class (11/14) preceded apparent cell/embryo death (egl-45, eif-3.f, rpl-12, rpl-20, rpl-36, rps-0, rps-3, rps-5, rps-9, rps-22, and rps-24) (Table 1). RNAi depletion of all 14 of these genes resulted in reduced PGL-1 signal and small PGL-1 aggregates (Figure 3B). While compromised translation of GFP∷PGL-1 or endogenous PGL-1 may account for this phenotype, it is also possible that the accumulation of P granules is dependent upon active translation. In comparison to somatic cell differentiation, which relies heavily on transcriptional regulation, this “ribosome” class emphasizes the crucial role of post-transcriptional regulation in the identity and development of the germ line. Indeed, recent transcript profiling from dissected germ lines revealed that a relatively large percentage of germ-line expressed mRNAs are involved in translation and ribosome structure and biogenesis (Wang et al. 2009). P-granule integrity and distribution may be sensitive to the translational status of germ cells and early embryos.
Splicing:
Our screen identified 12 genes that encode known or putative splicing factors (Table 2). The PGL-1 phenotypes of 9 of these genes appeared to precede cell/embryo death (cpf-2, mag-1, phi-6, phi-7, phi-10, phi-12, prp-8, rsp-7, and skp-1) (Table 1). Several PGL-1 phenotypes were shared by multiple members of the splicing class of genes (Figure 4). RNAi depletion of 5 genes (phi-6, phi-7, phi-10, prp-8, and skp-1) resulted in increased PGL-1 signal in the germ line of P0 adults and in F1 embryos. RNAi depletion of 9 genes (cpf-2, phi-6, phi-7, phi-10, phi-12, prp-8, rnp-4, rsp-7, and skp-1) resulted in some diffuse PGL-1 in the embryonic germ-line blastomeres and PGL-1 granules that are dissociated from the nuclear periphery. In wild-type embryos, between the 4- and 8-cell stages, P granules start to become perinuclear in the germ-line blastomere, while embryos depleted for splicing factors often contained PGL-1 granules dispersed in the cytoplasm, which persisted until the embryos arrested around the 100-cell stage. This arrest was accompanied by failure of the primordial germ cell (P4) to migrate internally.
Figure 4.—
Splicing class. Antibody-stained PGL-1 (green), nuclear pores (red), and DAPI-stained DNA (blue) in fixed embryos are shown. Dashed boxes indicate the location of zoomed images on the right. (A) Approximately 16-cell (left) and ∼150-cell (right) embryos. In wild-type embryos, PGL-1 granules are attached to the periphery of the nucleus in the primordial germ cell P4 (left embryo, ∼16 cells) and its daughters Z2 and Z3 (right embryo, ∼150 cells). P4, Z2, and Z3 nuclei are marked with asterisks. phi-12, phi-10, prp-8, phi-6, and skp-1 RNAi embryos contain P granules that are detached from the periphery of the nucleus (arrowheads). Higher levels of diffuse PGL-1 in the cytoplasm of Z2/Z3 cells of phi-12, phi-10, and prp-8 embryos can be observed in the areas circumscribed by dashed lines, and Z2/Z3 fail to migrate internally. (B) SF3b complex components phi-6, phi-11, and sap-49 RNAi embryos contain PGL-1 granules that are detached from the nucleus, and Z2/Z3 fail to migrate internally. Bar, 10 μm.
Sm components of the splicesome have been shown to localize to germ plasm in multiple species, and previous RNAi depletion of the C. elegans Sm proteins disrupted P-granule patterns in embryos (Moussa et al. 1994; Barbee et al. 2002; Chuma et al. 2003; Bilinski et al. 2004; Barbee and Evans 2006). However, previous RNAi depletion of other splicing components did not disrupt P-granule patterns in embryos (Barbee et al. 2002), suggesting that P-granule integrity and perinuclear localization in embryonic cells do not depend on pre-mRNA splicing. Our screen demonstrated that at least some pre-mRNA splicing factors are necessary for proper PGL-1 localization to granules at the nuclear periphery in embryos. Since the embryonic germ-line blastomeres are not actively engaged in transcription (Seydoux et al. 1996), the requirement for proper splicing regulation likely reflects that maternally generated mRNAs must be properly spliced for P granules to localize normally in embryos. Whether properly spliced mRNAs or the process of splicing affect P-granule patterning directly or indirectly remains to be determined.
We noted that several splicing factors obtained in our screen represent one subunit of multisubunit splicing complexes. This finding raised two possibilities: the other subunits in the complex do not participate in regulating P-granule distributions or the other subunits were missed in our single-pass RNAi screen. To distinguish between these possibilities, we tested all subunits of the SF3b complex, which is absolutely essential for pre-mRNA splicing (Will et al. 1999). This complex contains seven proteins, of which six have clear homologs in worms, encoded by the genes W03F9.10, phf-5, C46F11.4, sap-49, phi-11, and phi-6 (supporting information, Table S1). Only phi-6 was identified in our screen. Of the six worm genes, only phi-6, phi-11, sap-49, and C46F11.4 are targeted by the Ahringer RNAi library. We depleted these four genes by RNAi and found that in addition to phi-6, loss of both phi-11 and sap-49 resulted in diffuse PGL-1 in the embryonic germ-line blastomeres, PGL-1 granules that are dissociated from the nuclear periphery, and failure of P4 to migrate internally (Figure 4B, Table S1). Late embryonic arrest was used as a measure of RNAi effectiveness, and unlike phi-6, phi-11, and sap-49, depletion of C46F11.4 did not result in late embryonic arrest, diffuse PGL-1, or a P4 migration defect. Interestingly, the Ahringer RNAi clone targeting C46F11.4 contains a short sequence that is present in numerous copies throughout the genome, potentially diluting the effectiveness of this RNAi clone. Taken together, our results suggest that the SF3b complex and at least certain aspects of pre-mRNA splicing are essential for proper PGL-1 localization. These results exemplify the limitations of RNAi screens (incomplete gene coverage, ineffective RNAi, and missing phenotypes in single-pass screens) but also illustrate how RNAi screen positives can lay the foundation for more targeted approaches.
Nuclear pore/envelope/transport:
We identified 11 nuclear pore or nuclear pore/envelope associated factors in our screen (Table 2). RNAi depletion of 9 of these resulted in a distinctive phenotype that appeared to precede cell death in early embryos (imb-4, imb-5, lmn-1, npp-6, npp-7, npp-9, npp-10, ran-1, and ran-4). In contrast to the perinuclear association of PGL-1 in the more than four-cell stage wild-type embryos, most PGL-1-containing granules in embryos depleted for nuclear pore components remained detached from the nuclear envelope (Figure 5A). The finding that >75% of nuclear pores are overlaid by P granules in the adult germ line (Pitt et al. 2000) predicted that loss of nuclear pore components might compromise association of P granules with the nuclear periphery.
Figure 5.—
Nuclear pore/envelope/transport and MCM licensing classes. PGL-1 (green), nuclear pores (red), and DNA (blue) in fixed ∼30-cell embryos are shown. Dashed boxes indicate the location of zoomed images on the right. (A) Nuclear pore/envelope/transport class. Depletion of nuclear pore and nuclear pore-associated components results in the dispersal of PGL-1 granules from the nuclear periphery. (B) MCM licensing class. Depletion of MCM licensing factors also causes PGL-1 granules to be distributed throughout the cytoplasm; this phenotype is accompanied by severe embryonic defects. Bars, 10 μm.
Other labs have previously demonstrated a requirement for the nuclear transportins IMB-2, IMB-3, IMB-5, and IMA-3 to maintain P-granule integrity or perinuclear localization (J. Ahringer, personal communication; Geles and Adam 2001). There are three imb-5 RNAi clones in the Ahringer RNAi library (two correctly and one incorrectly annotated); our screen identified all three clones as causing PGL-1 granules to be detached from the nuclear periphery. However, our screen did not identify imb-2, imb-3, or ima-3. In targeted retests of those three RNAi clones, depletion of imb-2 and imb-3 resulted in detachment of PGL-1 from the nuclear periphery of embryos (Table S2), while depletion of ima-3 did not. We note that a stronger RNAi regime by Geles and Adam resulted in diffuse P granules in the adult germ line but, similar to our results, not in embryos (Geles and Adam 2001).
Our screen identified 5 of the 20 NPP proteins in C. elegans. To determine if only a subset of NPPs participates in tethering PGL-1 granules to the nuclear periphery, or if some NPPs were missed by our screen, we more closely examined PGL-1 patterns after RNAi depleting individual NPPs. The results support both scenarios. Thirteen of the 15 remaining NPPs are covered in the Ahringer RNAi library. Of these 13, RNAi depletion of 4 (npp-1, npp-3, npp-8, and npp-19) caused PGL-1 to be detached from the nuclear periphery in a high enough proportion of embryos that they should have emerged as screen positives (Table S2). Thus, our screen was successful in identifying 5 of 9 of the genes whose depletion results in high penetrance detachment of PGL-1 from the nuclear periphery. We missed 4 genes. Interestingly, RNAi depletion of 3 genes (npp-14, npp-15, and npp-16) caused late embryonic lethality, demonstrating the effectiveness of RNAi, but did not cause any apparent defects in PGL-1 patterns. These findings suggest that the localization of PGL-1 granules to the nuclear periphery depends on many but not all NPPs.
Our results contribute to the emerging view that the localization of P granules to the nuclear periphery relies heavily upon underlying nuclear pores and nuclear transport. It will be intriguing to determine if this localization depends on a direct interaction between P-granule and nuclear pore components, mRNA trafficking through nuclear pores, or both. We are currently investigating the former possibility and the hypothesis that the GLH components of P granules interact via their phenylalanine–glycine (FG) domains with the FG domains of nuclear pore proteins (see discussion).
MCM licensing:
One class of screen positives contains all five MCM licensing factors represented in the RNAi library (Table 2). MCM licensing factors ensure that DNA replication occurs only once per cell cycle, so this group could have been included in the cell cycle class of positives. We chose to put the MCM licensing factors in their own class because depletion of all five factors caused a similar phenotype: PGL-1 granules were no longer perinuclear in the embryonic P cells (Figure 5B). RNAi of these five MCM factors caused severe abnormalities, so at this time we cannot deduce whether the PGL-1 phenotype is a secondary effect of embryonic death due to compromised DNA replication or whether the MCM licensing factors serve a DNA replication-independent function that facilitates P-granule attachment to the nuclear periphery. In yeast and mammalian cells, MCMs vastly outnumber replication origins and their function is not restricted to DNA synthesis (Blow and Dutta 2005). In Xenopus, MCMs associate with RNA polymerase II holoenzyme (Yankulov et al. 1999) and were recently found to be required for RNA polymerase II-mediated transcription (Snyder et al. 2009). Therefore, the PGL-1 phenotype in MCM RNAi embryos may be independent of replication and dependent on transcription or transcriptional regulation of other specific factors. It is worth noting that RNAi depletion of subunits of DNA polymerase δ and DNA polymerase α-primase can delay early cell divisions and cause missegregation of P granules (Encalada et al. 2000). We found that RNAi depletion of another DNA polymerase α-primase subunit, PRI-1, the large subunit of replication protein A, RPA-1, or the catalytic subunit of DNA polymerase alpha, Y47D3A.29, resulted in a diffuse cytoplasmic distribution of PGL-1 in early germ-line blastomeres. It would be interesting to investigate whether any treatment that slows the timing of cell divisions alters P-granule distribution and segregation or whether the factors discussed above have more direct effects on P granules.
Other small classes of screen positives:
Another small class of screen positives includes the actin/microtubule chaperonins cct-2, cct-3, cct-7, and cct-8 (Table 2). Depletion of these CCT proteins resulted in low and diffuse levels of PGL-1 in embryos. Severe embryonic defects prevented further characterization of the PGL-1 phenotype, but we predict that PGL-1 mislocalization (data not shown) stems from compromised actin-based P-granule segregation in these mutants. Interestingly, it was recently demonstrated that RNAi depletion of other components of this chaperonin complex, cct-4 and cct-6, causes ectopic expression of PGL-1 in the intestine and hypodermis of adult worms, which may contribute to their long-lived phenotype (Curran et al. 2009).
Remaining screen positives can be placed into a handful of smaller categories. Some genes encode components of subcellular organelles including vacuoles, Golgi, and ER. RNAi depletion typically resulted in decreased PGL-1 signal (data not shown). We were unable to assign 59 screen positives to a class larger than three members, and the functions of 8 positives are not yet predictable (Table 2).
Accumulation of P granules mediated through mRNA homeostasis:
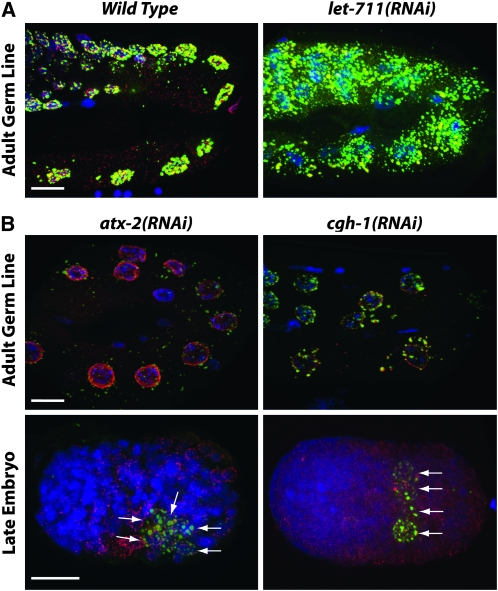
In embryos, clusters of poly(A)+ RNAs colocalize with P granules (Seydoux and Fire 1994). In adult germ lines and embryos, SL1 sequences, which are trans-spliced onto the 5′ end of most mRNAs (Zorio et al. 1994), also colocalize with P granules (Pitt et al. 2000; Schisa et al. 2001). These and other results suggest that P granules transiently retain developmentally regulated mRNAs as they exit the nucleus (Schisa et al. 2001). P granules may regulate associated mRNAs in various ways, including control of mRNA stability and/or translation, and delivery of mRNAs to the germ-line blastomeres and eventually the primordial germ cells in embryos (Strome 2005). In somatic cells, mRNA deadenylation, decapping, and degradation occur in cytoplasmic RNP aggregates called P bodies, which share many components with P granules. For example, C. elegans P granules have been shown to contain the CCR4/Not1 deadenylase component CCF-1, as well as multiple decapping coactivators including PATR-1, DCAP-1, DCAP-2, and CGH-1 (Navarro et al. 2001; Lall et al. 2005; Squirrell et al. 2006; Gallo et al. 2008). Our screen identified several mRNA degradation components as being required for regulation of PGL-1 accumulation. Interestingly, their depletion caused different PGL-1 phenotypes (Figure 6). RNAi depletion of the Not1 component LET-711 resulted in increased PGL-1 accumulation in the adult germ line (Figure 6A) and in progeny embryos, while RNAi depletion of the decapping factors CGH-1 and ATX-2 and the predicted SKI2 exosome homolog F01G4.3 resulted in reduced PGL-1 levels (Table 1; Figure 6B). Depletion of CGH-1 and ATX-2 also caused PGL-1 to persist in numerous cells of later embryos (Figure 6B). These findings extend the list of mRNA degradation components that are likely to be shared between P bodies and P granules and support the view that the accumulation of P granules is closely connected to mRNA levels.
Figure 6.—
mRNA degradation components are required for proper P-granule accumulation. Antibody-stained PGL-1 (green) and nuclear pores (red) and DAPI-stained DNA (blue) in fixed germ lines and embryos are shown. (A) In the germ lines of animals depleted of the Not1 deadenylase LET-711, PGL-1 granules are larger and more numerous and more dispersed than in wild type. (B) After depletion of decapping coactivators ATX-2 and CGH-1, low levels of PGL-1 are observed in the adult germ line, and PGL-1 is often observed in multiple cells of late embryos (arrows). Bars, 10 μm.
CSR-1 downregulates RNA and P-granule accumulation through an endo-siRNA pathway in the germ line:
The most striking PGL-1 phenotype observed in the P0 germ line was caused by RNAi of csr-1. Depletion of csr-1 resulted in very intense PGL-1 staining and large PGL-1 aggregates distributed throughout the cytoplasm of the adult germ line and embryos (Figures 1C and 7A). To determine if the large PGL-1 aggregates in csr-1(RNAi) germ lines are bona fide P granules, we tested for whether they contain other constitutive components of P granules, such as the PGL-1 paralog PGL-3 (Kawasaki et al. 2004) and the VASA homolog GLH-1 (Gruidl et al. 1996). PGL-3 and GLH-1 colocalize with the large PGL-1 aggregates in csr-1(RNAi) adult germ lines (Figure 7C). Quantitative PCR of pgl-1, pgl-3, and glh-1 transcripts in adult hermaphrodites showed a modest increase after csr-1 RNAi (1.69-fold for pgl-1, P = 0.0053; 1.12-fold for pgl-3, P = 0.0390; 2.17-fold for glh-1, P = 0.0010). While GFP∷PGL-1 reporter and antibody-stained endogenous PGL-1 appear dramatically brighter in csr-1(RNAi) worms (Figures 1C and 7A), Western blot analysis revealed a modest 1.4-fold increase in PGL-1 protein in csr-1(RNAi) worms compared to wild type (Figure S1), consistent with the ∼1.7-fold change in pgl-1 transcript noted above. The brighter, larger P granules observed in csr-1(RNAi) embryos may reflect a combination of modestly elevated levels of P-granule proteins and enhanced aggregation of those proteins into granules. We noted an additional phenotype in csr-1(RNAi) embryos. In many RNAi embryos (40%, n = 70), PGL-1 and PGL-3 granules were found in somatic blastomeres (Figure 7D). Interestingly, GLH-1 did not colocalize with the PGLs in these ectopic granules (Figure 7D). This is reminiscent of the partial P granules (containing PGL-1 and PGL-3; lacking GLH-1) observed in mutant embryos defective in autophagy (Zhang et al. 2009), raising the possibility that CSR-1 participates in autophagocytic removal of P-granule proteins from somatic cells.
Figure 7.—
PGL-1 and RNA accumulation in animals depleted of the Argonaute CSR-1. (A) Endogenous PGL-1 staining (green) in wild-type and csr-1(RNAi) germ lines. Depletion of CSR-1 results in the accumulation of large PGL-1 aggregates. (B) RNA staining with SYTO14 shows that RNA accumulates in the syncytial cytoplasm of the adult germ line after CSR-1 is depleted. (C) Endogenous PGL-3 (green) and GLH-1 (red) colocalize in large P-granule aggregates after CSR-1 is depleted. (D) In csr-1(RNAi) embryos (∼150-cell stage shown), PGL-3 accumulates in somatic cells, whereas GLH-1 is restricted to germ-line blastomeres. Bars, 10 μm.
CSR-1 is an Argonaute protein with endonucleolytic mRNA cleavage (slicing) activity (Yigit et al. 2006; Aoki et al. 2007). The slicing activity of Argonautes mediates siRNA silencing of mRNAs, which are then degraded by exonucleases in the exosome and decapping pathways (Orban and Izaurralde 2005). To determine if the formation of large P granules in csr-1(RNAi) animals is accompanied by an increase in RNA accumulation, we dissected hermaphrodite gonads in the presence of SYTO14, a nucleic acid stain that is used to visualize nucleolar and cytoplasmic RNA (Schisa et al. 2001). Both control(RNAi) and csr-1(RNAi) worms showed comparable SYTO14 staining in germ-line nucleoli. However, we observed a much higher level of RNA accumulation in the central cytoplasmic “rachis” of csr-1(RNAi) animals than in control worms fed empty RNAi vector (Figure 7B, gonads with high levels of RNA accumulation: control, 1 of 16 gonad arms; csr-1, 12 of 18 gonad arms). Our several attempts to concomitantly image cytoplasmic RNA accumulations and P granules were unsuccessful. Thus, we do not know if they colocalize. The different locations and morphologies of cytoplasmic RNA accumulations and enlarged P granules in csr-1(RNAi) suggest that P granules are not the sites of excessive RNA accumulation. Previous studies have shown that depletion of sperm and arrested ovulation cause RNA to accumulate in the rachis; this phenotype is regulated by the major sperm protein pathway (Schisa et al. 2001; Jud et al. 2008). csr-1(RNAi) animals exhibited no obvious defects in fertilization or ovulation, suggesting that RNA accumulation in csr-1(RNAi) animals is not caused by loss of the major sperm protein pathway. We hypothesize that the accumulation of cytoplasmic RNA and of enlarged P granules observed after depletion of CSR-1 is due to reduced slicing and degradation of mRNAs.
If loss of CSR-1 slicing activity causes enlarged and disorganized P granules, then loss of proteins that function in the CSR-1 pathway should cause similar defects. CSR-1 preferentially binds to secondary siRNAs, siRNAs that are produced from primary siRNA-targeted mRNAs by RNA-dependent RNA polymerase (RdRP) (Aoki et al. 2007). We tested whether enlarged and disorganized PGL-1 granules are seen in mutants lacking the somatic RdRP RRF-1 or either of the two RdRPs that are known to be active in the germ line, EGO-1 and RRF-3. We observed an accumulation of large PGL-1 granules in ego-1 mutant germ lines (as observed by Vought et al. 2005) but not in rrf-1 or rrf-3 mutant germ lines (Figure 8A). The PGL-1 phenotype in ego-1 and csr-1 mutants was typically more severe than the phenotype caused by RNAi; in mutant germ lines, the large PGL-1 aggregates were less perinuclear than observed following RNAi. Mutant germ lines may have less EGO-1 or CSR-1 than can be achieved by RNAi, or alternatively mutant germ lines may have compromised health and show more severe defects. Enlarged PGL-1 granules were not observed in RNAi-defective mutants such as dcr-1 (Vought et al. 2005) or rde-3 (our observations, Figure 8A), suggesting that the phenotype is not due to a general defect in RNAi. Next we examined GFP∷PGL-1 in drh-3(RNAi) germ lines. drh-3 encodes a DExH-box helicase that is required for RdRP activity and shares several phenotypes with ego-1 and csr-1 (Duchaine et al. 2006; Yigit et al. 2006; Aoki et al. 2007; Rocheleau et al. 2008; She et al. 2009). Depletion of drh-3 phenocopied the PGL-1 phenotype observed in ego-1 and csr-1 RNAi germ lines (Figure 8B), providing further evidence that DRH-3 functions in a pathway with EGO-1 and CSR-1.
Figure 8.—
PGL-1 accumulates in numerous large granules in the germ line after an endogenous siRNA pathway is compromised. (A) Endogenous PGL-1 staining (green) resembles wild type in the germ lines of rde-1, rrf-1, and rrf-3 mutants. Large PGL-1 granules accumulate in the germ lines of ego-1 and csr-1 mutants. Most granules are dissociated from the nuclear periphery. (B) GFP∷PGL-1 accumulation in control(RNAi), drh-3(RNAi), and ego-1(RNAi) germ lines. RNAi depletion of EGO-1 and DRH-3 produces the same large PGL-1 granule phenotype as depletion of CSR-1. Bars, 10 μm.
We wanted to see if elevated RNA accumulation correlates with elevated levels of PGL-1 or if the effect is specific to the depletion of csr-1. We found that, like csr-1, depletion of both ego-1 and drh-3 also resulted in increased cytoplasmic RNA as detected by SYTO14 staining (Table 3). We also examined other factors whose depletion resulted in elevated PGL-1 in the adult germ line (eft-1, let-711, phi-6, phi-7, phi-11, prp-8, and skp-1). In general, increased PGL-1 levels correlated with increased cytoplasmic RNA. Depletion of csr-1, ego-1, and the splicing factors phi-7, phi-11, and skp-1 had particularly strong effects on RNA patterns (Table 3). At this time we do not know if the accumulation of cytoplasmic RNA results from loss of function of CSR-1, EGO-1, DRH-3, and the handful of RNA-binding proteins whose depletion results in elevated PGL-1 or if the accumulation of RNA results from displaced and elevated PGL-1 itself. A third possibility is that P-granule proteins and cytoplasmic RNAs interact with and stabilize each other.
TABLE 3.
Correlation of elevated PGL-1 and elevated RNA in RNAi germ lines
| Target | % penetrance of elevated PGL-1 phenotype (n = 20) | % penetrance of elevated cytoplasmic RNA (n = 10)a |
|---|---|---|
| Control | 0 | 0 |
| csr-1 | 100 | 80 |
| ego-1 | 100 | 70 |
| drh-3 | 80 | 40 |
| eft-1 | 90 | 20 |
| let-711 | 100 | 40 |
| phi-6 | 100 | 10 |
| phi-7 | 100 | 80 |
| phi-11 | 95 | 60 |
| prp-8 | 100 | 30 |
|
skp-1 |
90 |
80 |
Based on SYTO14 staining.
CSR-1, EGO-1, and DRH-3 are thought to work together in a germ-line-specific endogenous siRNA pathway (Maine et al. 2005; Robert et al. 2005; Duchaine et al. 2006; Yigit et al. 2006; Aoki et al. 2007; Rocheleau et al. 2008; She et al. 2009). Argonautes, like CSR-1, have been identified as germ granule components from worms to mammals (Kotaja et al. 2006; Batista et al. 2008; Wang and Reinke 2008). In addition, an RGG domain, like that found in PGL-1 and PGL-3, in the N terminus of CSR-1 also suggests that CSR-1 is likely to associate with P granules. To test that prediction, we attempted to generate good anti-CSR-1 antibodies and informative GFP∷CSR-1 transgenic worms. Although both antibodies and transgenic worms showed P-granule localization, neither passed our tests for specificity. Nevertheless, on the basis of the specific PGL-1 phenotype of CSR-1 pathway mutants and the predicted P-granule localization of CSR-1, we hypothesize that P granules function as a center for endogenous siRNA silencing in the germ line and that disruption of this endo-siRNA pathway results in not only the accumulation in the germ-line syncytium of RNA transcripts that in wild type are silenced and degraded, but also the accumulation of large P granules.
DISCUSSION
Our RNAi screen proved to be an effective method to identify genes whose depletion causes aberrant PGL-1 phenotypes. One advantage of performing an RNAi screen over a forward mutagenesis screen is that mutants are not required to be viable and fertile. This allowed us to identify multiple gene classes, including some essential genes that are required for PGL-1 assembly and localization. One disadvantage of RNAi screens is missing some genes due to ineffective RNAi or incomplete RNAi library coverage. To expedite our screen, RNAi was performed for only 30 hr, and only P0 and F1 progeny were examined. Because of the maternal contribution of many P-granule proteins, some phenotypes, like that caused by DEPS-1 loss, are not observable until the F2 generation after RNAi is started. Our P0/F1 screen did not identify any additional genes whose depletion causes the PGL-1 phenotypes of deps-1 and glh-1 mutants where PGL-1 is dispersed in all cells of normally developing embryos. We do not know if DEPS-1 and GLH-1 are the only proteins whose loss causes that specific phenotype or if an F2 generation screen could identify more genes with that phenotype.
Despite the limitations of RNAi screens, this type of genomewide screen serves as an excellent starting point for identification and in-depth analysis of gene families in particular cellular processes. For example, the 5 NPPs identified in our screen drew our attention to nuclear pores as being involved in P-granule localization. Targeted retests of the remaining 13 NPP genes represented in the RNAi library identified 4 NPP genes that our screen should have identified, 5 NPP genes that have low-penetrance RNAi phenotypes and therefore require other analysis strategies, and 4 NPP genes that may not participate in regulating P-granule localization. Another example is our identification of one subunit of the SF3b splicing complex. Targeted retests identified two additional subunits. Loss of the remaining three SF3b components, which are not covered or inefficiently covered in the RNAi library, is likely to result in PGL-1 phenotypes as well. Finally, a recent report used a similar RNAi screen and identified a single autophagy component, lgg-1, which is required for clearing small P granules from somatic cells. Targeted tests of other autophagy components, by injection of double-stranded RNA to achieve stronger RNAi, revealed their involvement in clearing P granules from somatic cells (Zhang et al. 2009). Certainly, the 173 genes identified in our screen are an underestimate. We expect more stringent RNAi procedures, such as injecting or soaking or feeding for longer times, will identify additional genes that affect P granules in C. elegans.
The majority of genes identified in our screen were grouped into large gene classes. While some classes were likely identified because of P-granule segregation or degradation defects (cell cycle/polarity/cell division and proteasome/ubiquitin), most classes associate P granules with post-transcriptional regulation in the germ line. For example, splicing, nuclear pore, and ribosomal classes emphasize the importance of mRNA maturation, nuclear transport, and translational regulation in controlling P-granule size and location.
Nascent transcripts are processed in the nucleus by spliceosomes prior to their export as mature mRNAs. Sm proteins are core components of nuclear spliceosomes and are also concentrated in the cytoplasmic germ granules of multiple species (Moussa et al. 1994; Chuma et al. 2003; Barbee et al. 2002; Bilinski et al. 2004). In C. elegans, Sm proteins are required for proper P-granule localization to the nuclear periphery; it has been proposed that this role is independent of pre-mRNA splicing (Barbee and Evans 2006). We, too, identified the Sm protein, SNR-2, as required for proper PGL-1 granule assembly. We also identified other splicing and mRNA maturation components. These include the cleavage and polyadenylation factor CPF-2 and the putative integrator complex subunit C06A5.1, both of which are involved in maturation of 3′ ends of mRNA; the putative exon–exon junction factors MAG-1 and RNP-4; possible snRNP associated factor PRP-8 along with SF3b splicesome components PHI-6, PHI-11, and SAP-49; and other factors whose homologs are required for pre-mRNA splicing, including RSP-7, SKP-1, PHI-7, PHI-10, and PHI-12. While the role of Sm proteins in germ granules may still be independent of their role in splicing, our results suggest that proper mRNA maturation in the nucleus affects the assembly and localization of P granules. It will be interesting to determine whether properly assembled P granules require specific features of spliced mRNAs, splicing factors associated with mRNAs, or mRNA export through nuclear pores.
The integrity of nuclear pores appears to be vital for the perinuclear distribution of P granules. At least 14 of 20 nuclear pore components are required for PGL-1's perinuclear distribution, as are nuclear transport factors such as IMB-1, IMB-5, RAN-1, and RAN-4. Nuclear lamin LMN-1 is also required, as is dynein light chain-1, DLC-1. DLC-1's yeast homolog DYN2 is a nucleoporin (Stelter et al. 2007), and interestingly C. elegans DLC-1 was found to interact with PGL-3 in a high-throughput binding assay (Li et al. 2004). Therefore, it is possible that some nucleoporins, like the putative nucleoporin DLC-1, provide a physical linkage between P granules and nuclear pores. On that note, we find it interesting that the P-granule components GLH-1, GLH-2, and GLH-4 contain multiple Phe–Gly (FG) repeat domains, domains that are found in many nucleoporins (Rexach and Blobel 1995). Some FG domains form cohesive hydrophobic interactions with other FG domains in the nuclear pore (Patel et al. 2007). The close association between P granules and nuclear pores, as well as similarities between GLH proteins and nucleoporins, has led to the prediction that P granules serve as an extension of nuclear pores in the germ line (Kuznicki et al. 2000; Pitt et al. 2000; Schisa et al. 2001). We are currently testing whether the FG domains of GLH-1, GLH-2, and GLH-4 interact with each other and with other FG-containing nucleoporins and whether FG proteins in P granules do indeed serve to extend the nuclear pore environment.
As RNAs transcribed in germ nuclei exit through nuclear pores, many transcripts will encounter P granules. Poly(A)+ and developmentally regulated mRNAs have been shown to accumulate in P granules (Seydoux and Fire 1994; Schisa et al. 2001). P granules may act as a scaffold for multiple RNA processing activities, possibly by providing an environment that facilitates germ-line-specific binding interactions and regulates the translation, transport, modification, storage, and/or degradation of RNA. Our screen results support many of these possibilities. For example, we found that multiple ribosomal components are required for proper P-granule accumulation and stability. This finding could reflect a need for synthesis of P-granule proteins or a requirement for ongoing translation during the assembly of P granules.
We observed PGL-1 phenotypes when RNA deadenlyase, decapping, and degradation components were depleted. Depletion of LET-711, the NOT1 ortholog of the CCR4/NOT1 mRNA deadenylation complex, had previously been reported to cause PGL-1 localization outside of the embryonic germ line (Gallo et al. 2008). We identified LET-711 primarily on the basis of the elevated accumulation of transgenic and endogenous PGL-1 in the adult germ line. This phenotype was observed within 30 hr after initiation of RNAi and before germ-line morphology became altered, suggesting that the PGL-1 phenotype was a direct result of LET-711 depletion. The PGL-1 aggregates in let-711(RNAi) germ lines are smaller than those observed in csr-1 mutants, but as in the case of csr-1, the enhanced accumulation may be correlated with RNA stabilization. The accumulation of P granules may be sensitive to cytoplasmic RNA levels, requiring RNA destabilizing factors like LET-711 and CSR-1 to control RNA levels through exo- or endonucleolytic activity.
One exciting gene that emerged from our screen was csr-1. CSR-1 is an Argonaute that exhibits endonucleolytic or slicing activity on RNA targets, showing a preference for using secondary siRNAs generated by RdRP activity rather than primary siRNAs generated by Dicer (Aoki et al. 2007). Thus far, C. elegans is unique among animals in its large number of Argonaute-encoding genes: 27 in C. elegans compared to 5 in Drosophila and 8 in humans (Hutvagner and Simard 2008). The diversity of Argonautes in C. elegans has proven advantageous for dissecting out the multiple roles of these proteins. C. elegans Argonautes ALG-1 and ALG-2 are essential for miRNA-mediated gene silencing, RDE-1 for exogenous RNAi, ERGO-1 for endogenous RNAi, and PRG-1 and PRG-2 for piRNA-mediated gene silencing, and a handful of other Argonautes are thought to bind secondary siRNAs but lack the ability to slice RNA targets (Grishok et al. 2001; Yigit et al. 2006; Batista et al. 2008; Das et al. 2008; Wang and Reinke 2008). csr-1 was first obtained in a germ-line cosuppression screen, which suggested that it may be involved in silencing repetitive transgene arrays in the germ line (Robert et al. 2005). csr-1 worms are also partially deficient in RNAi (Yigit et al. 2006). It was recently discovered that csr-1 shares multiple phenotypes with ego-1 and drh-3, which encode an RdRP and an RdRP interacting factor required for small endogenous RNAi in the germ line (Aoki et al. 2007; Rocheleau et al. 2008; She et al. 2009).
We discovered that csr-1 worms accumulate large P granules in the adult germ line and ectopic granules in somatic cells of embryos. This phenotype is accompanied by a dramatic increase in RNA in the central cytoplasm of the germ line. While it was not obtained in our original screen, our targeted experiments demonstrated that depletion of EGO-1 and DRH-3 phenocopy the very specific PGL-1 phenotype of csr-1, supporting a role for CSR-1 in a germ-line-specific endo-RNAi pathway. One model proposed previously to explain the irregular P granules in ego-1 mutants is that EGO-1 may promote a chromatin state that regulates nuclear pore distribution and that the clumping of nuclear pore material leads to perturbed P-granule assembly (Vought et al. 2005). This model is strengthened by the observation that EGO-1 is found in nuclear fractions and is required for unpaired chromosomal regions to accumulate high levels of histone H3 lysine K9 dimethylation, a mark associated with heterochromatin assembly and transcriptional silencing (Maine et al. 2005; She et al. 2009). However, we observed a different PGL-1 phenotype when nuclear pore components were depleted (PGL-1 granules dissociated from the nuclear periphery in germ-line blastomeres of developing embryos) than we did when CSR-1, EGO-1, and DRH-3 were depleted (enhanced accumulation of large PGL-1 granules in the adult germ line). Loss of the nuclear envelope component LMN-1, which also results in the clustering of nuclear pores (Liu et al. 2000), causes a P-granule phenotype that is more similar to that caused by depletion of core nuclear pore proteins than to the large P-granule phenotype caused by depletion of CSR-1, EGO-1, and DRH-3. Another simple model is that genes typically silenced by endo-RNAi in the germ line are desilenced when csr-1 is depleted, allowing these genes' transcripts to accumulate in the cytoplasm. In normal germ lines, these transcripts could be silenced at the level of chromatin and regulation of transcription or at the level of exiting the nucleus into P granules where they are sliced by CSR-1 and subsequently degraded. As all known P-granule components are thought to function in RNA metabolism, the accumulation of large P granules in csr-1 animals may be due to increased levels of cytoplasmic RNA in csr-1 germ lines. Another possibility is that transcripts encoding P-granule proteins are normally downregulated by endo-RNAi, and the loss of this downregulation in csr-1 animals leads to the accumulation of large P granules. Profiling germ-line transcripts in csr-1 mutants may reveal which, if any, of these models are correct.
The P-granule components DEPS-1 and PGL-1 are both required for a robust exogenous RNAi response (Robert et al. 2005; Spike et al. 2008b). While PGL-1 has not been implicated in a specific RNAi pathway, deps-1 mutants are thought to resist the effects of RNAi due to a 7- to 10-fold decrease in rde-4 expression (Spike et al. 2008b). RDE-4 binds dsRNA and forms a complex with Dicer to produce siRNAs, and rde-4 mutants are RNAi defective (Tabara et al. 2002). Another component identified in screens for RNAi-defective mutants, RDE-3, is thought to act downstream of RDE-4 and Dicer. Comparison of transcriptional profiles of rde-3 and deps-1 mutants showed that nearly 30% of genes upregulated in deps-1 mutant germ lines are also upregulated in rde-3 worms, suggesting that RDE-3 and DEPS-1 function together to regulate expression of at least their shared target genes (Spike et al. 2008b). As DEPS-1 is a constitutive P-granule component, it likely participates in regulation at a post-transcriptional level. Our results with CSR-1, EGO-1, and DRH-3 strengthen the emerging view that P granules and RNAi are intimately related: P-granule components participate in regulating RNAi, and RNAi factors influence the structure, localization, and perhaps function of P granules. Better defining the RNAi–P-granule relationship will shed light on mechanisms of post-transcriptional gene regulation used by germ cells.
Acknowledgments
We thank Colin Thacker for training provided during the screen, use of equipment including the Copas Biosort, and preparation of RNAi colonies from the Ahringer RNAi library; Robb Cundick for the development and use of a C. elegans RNAi database; Susan Mango for the use of lab space and equipment during the first pass of screening; Julie Ahringer for unpublished information on the imb-2, imb-3, and imb-5 PGL-1 phenotypes; Xingyu She and Eleanor Maine for providing ego-1 strain EL500; Andreas Rechtsteiner for comparison of germ-line data sets; and the National Bioresource Project at Tokyo Women's Medical College for providing the FX892 strain. This work was supported by Ruth Kirchstein National Research Service Award postdoctoral fellowship GM084673 (to D.U.) and National Institutes of Health (NIH) grant GM34059 (to S.S.). Some C. elegans strains were provided by the Caenorhabditis Genetics Center, which is funded by the NIH National Center for Research Resources.
Note added in proof: Since the acceptance of this article for publication, an additional study was published that shows abnormal P granules in csr-1, drh-3, and ego-1 mutants (J. M. Claycomb, P. J. Batista, K. M. Pang, W. Gu, J. J. Vasale et al., 2009, The Argonaute CSR-1 and its 22G-RNA cofactors are required for holocentric chromosome segregation. Cell 139: 123–134). Claycomb et al. also show that CSR-1, DRH-3, and EGO-1 colocalize with P granules.
Supporting information is available online at http://www.genetics.org/cgi/content/full/genetics.109.110171/DC1.
References
- Aoki, K., H. Moriguchi, T. Yoshioka, K. Okawa and H. Tabara, 2007. In vitro analyses of the production and activity of secondary small interfering RNAs in C. elegans. EMBO J. 26 5007–5019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbee, S. A., and T. C. Evans, 2006. The Sm proteins regulate germ cell specification during early C. elegans embryogenesis. Dev. Biol. 291 132–143. [DOI] [PubMed] [Google Scholar]
- Barbee, S. A., A. L. Lublin and T. C. Evans, 2002. A novel function for the Sm proteins in germ granule localization during C. elegans embryogenesis. Curr. Biol. 12 1502–1506. [DOI] [PubMed] [Google Scholar]
- Batista, P. J., J. G. Ruby, J. M. Claycomb, R. Chiang, N. Fahlgren et al., 2008. PRG-1 and 21U-RNAs interact to form the piRNA complex required for fertility in C. elegans. Mol. Cell 31 67–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilinski, S. M., M. K. Jaglarz, B. Szymanska, L. D. Etkin and M. Kloc, 2004. Sm proteins, the constituents of the spliceosome, are components of nuage and mitochondrial cement in Xenopus oocytes. Exp. Cell Res. 299 171–178. [DOI] [PubMed] [Google Scholar]
- Blobel, G., 1985. Gene gating: a hypothesis. Proc. Natl. Acad. Sci. USA 82 8527–8529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blow, J. J., and A. Dutta, 2005. Preventing re-replication of chromosomal DNA. Nat. Rev. Mol. Cell. Biol. 6 476–486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner, S., 1974. The genetics of Caenorhabditis elegans. Genetics 77 71–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheeks, R. J., J. C. Canman, W. N. Gabriel, N. Meyer, S. Strome et al., 2004. C. elegans PAR proteins function by mobilizing and stabilizing asymmetrically localized protein complexes. Curr. Biol. 14 851–862. [DOI] [PubMed] [Google Scholar]
- Chuma, S., M. Hiyoshi, A. Yamamoto, M. Hosokawa, K. Takamune et al., 2003. Mouse Tudor Repeat-1 (MTR-1) is a novel component of chromatoid bodies/nuages in male germ cells and forms a complex with snRNPs. Mech. Dev. 120 979–990. [DOI] [PubMed] [Google Scholar]
- Cowan, C. R., and A. A. Hyman, 2007. Acto-myosin reorganization and PAR polarity in C. elegans. Development 134 1035–1043. [DOI] [PubMed] [Google Scholar]
- Curran, S. P., X. Wu, C. G. Riedel and G. Ruvkun, 2009. A soma-to-germline transformation in long-lived Caenorhabditis elegans mutants. Nature 459 1079–1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das, P. P., M. P. Bagijn, L. D. Goldstein, J. R. Woolford, N. J. Lehrbach et al., 2008. Piwi and piRNAs act upstream of an endogenous siRNA pathway to suppress Tc3 transposon mobility in the Caenorhabditis elegans germline. Mol. Cell 31 79–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeRenzo, C., K. J. Reese and G. Seydoux, 2003. Exclusion of germ plasm proteins from somatic lineages by cullin-dependent degradation. Nature 424 685–689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duchaine, T. F., J. A. Wohlschlegel, S. Kennedy, Y. Bei, D. Conte, Jr. et al., 2006. Functional proteomics reveals the biochemical niche of C. elegans DCR-1 in multiple small-RNA-mediated pathways. Cell 124 343–354. [DOI] [PubMed] [Google Scholar]
- Eddy, E. M., 1975. Germ plasm and the differentiation of the germ cell line. Int. Rev. Cytol. 43 229–280. [DOI] [PubMed] [Google Scholar]
- Encalada, S. E., P. R. Martin, J. B. Phillips, R. Lyczak, D. R. Hamill et al., 2000. DNA replication defects delay cell division and disrupt cell polarity in early Caenorhabditis elegans embryos. Dev. Biol. 228 225–238. [DOI] [PubMed] [Google Scholar]
- Ephrussi, A., and R. Lehmann, 1992. Induction of germ cell formation by oskar. Nature 358 387–392. [DOI] [PubMed] [Google Scholar]
- Gallo, C. M., E. Munro, D. Rasoloson, C. Merritt and G. Seydoux, 2008. Processing bodies and germ granules are distinct RNA granules that interact in C. elegans embryos. Dev. Biol. 323 76–87. [DOI] [PubMed]
- Geles, K. G., and S. A. Adam, 2001. Germline and developmental roles of the nuclear transport factor importin alpha3 in C. elegans. Development 128 1817–1830. [DOI] [PubMed] [Google Scholar]
- Gonczy, P., and L. S. Rose, 2005. Asymmetric cell division and axis formation in the embryo. WormBook 1–20. www.wormbase.org [DOI] [PMC free article] [PubMed]
- Grishok, A., A. E. Pasquinelli, D. Conte, N. Li, S. Parrish et al., 2001. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell 106 23–34. [DOI] [PubMed] [Google Scholar]
- Gruidl, M. E., P. A. Smith, K. A. Kuznicki, J. S. McCrone, J. Kirchner et al., 1996. Multiple potential germ-line helicases are components of the germ-line-specific P granules of Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 93 13837–13842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guedes, S., and J. R. Priess, 1997. The C. elegans MEX-1 protein is present in germline blastomeres and is a P granule component. Development 124 731–739. [DOI] [PubMed] [Google Scholar]
- Hanazawa, M., I. Kawasaki, H. Kunitomo, K. Gengyo-Ando, K. L. Bennett et al., 2004. The Caenorhabditis elegans eukaryotic initiation factor 5A homologue, IFF-1, is required for germ cell proliferation, gametogenesis and localization of the P-granule component PGL-1. Mech. Dev. 121 213–224. [DOI] [PubMed] [Google Scholar]
- Hird, S. N., J. E. Paulsen and S. Strome, 1996. Segregation of germ granules in living Caenorhabditis elegans embryos: cell-type-specific mechanisms for cytoplasmic localisation. Development 122 1303–1312. [DOI] [PubMed] [Google Scholar]
- Hutvagner, G., and M. J. Simard, 2008. Argonaute proteins: key players in RNA silencing. Nat. Rev. Mol. Cell. Biol. 9 22–32. [DOI] [PubMed] [Google Scholar]
- Illmensee, K., and A. P. Mahowald, 1974. Transplantation of posterior polar plasm in Drosophila. Induction of germ cells at the anterior pole of the egg. Proc. Natl. Acad. Sci. USA 71 1016–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jud, M. C., M. J. Czerwinski, M. P. Wood, R. A. Young, C. M. Gallo et al., 2008. Large P body-like RNPs form in C. elegans oocytes in response to arrested ovulation, heat shock, osmotic stress, and anoxia and are regulated by the major sperm protein pathway. Dev. Biol. 318 38–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamath, R. S., M. Martinez-Campos, P. Zipperlen, A. G. Fraser and J. Ahringer, 2001. Effectiveness of specific RNA-mediated interference through ingested double-stranded RNA in Caenorhabditis elegans. Genome Biol. 2 RESEARCH0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamath, R. S., A. G. Fraser, Y. Dong, G. Poulin, R. Durbin et al., 2003. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421 231–237. [DOI] [PubMed] [Google Scholar]
- Kawasaki, I., Y. H. Shim, J. Kirchner, J. Kaminker, W. B. Wood et al., 1998. PGL-1, a predicted RNA-binding component of germ granules, is essential for fertility in C. elegans. Cell 94 635–645. [DOI] [PubMed] [Google Scholar]
- Kawasaki, I., A. Amiri, Y. Fan, N. Meyer, S. Dunkelbarger et al., 2004. The PGL family proteins associate with germ granules and function redundantly in Caenorhabditis elegans germline development. Genetics 167 645–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemphues, K. J., J. R. Priess, D. G. Morton and N. S. Cheng, 1988. Identification of genes required for cytoplasmic localization in early C. elegans embryos. Cell 52 311–320. [DOI] [PubMed] [Google Scholar]
- Kotaja, N., S. N. Bhattacharyya, L. Jaskiewicz, S. Kimmins, M. Parvinen et al., 2006. The chromatoid body of male germ cells: similarity with processing bodies and presence of Dicer and microRNA pathway components. Proc. Natl. Acad. Sci. USA 103 2647–2652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuznicki, K. A., P. A. Smith, W. M. Leung-Chiu, A. O. Estevez, H. C. Scott et al., 2000. Combinatorial RNA interference indicates GLH-4 can compensate for GLH-1; these two P granule components are critical for fertility in C. elegans. Development 127 2907–2916. [DOI] [PubMed] [Google Scholar]
- Lall, S., F. Piano and R. E. Davis, 2005. Caenorhabditis elegans decapping proteins: localization and functional analysis of Dcp1, Dcp2, and DcpS during embryogenesis. Mol. Biol. Cell 16 5880–5890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leacock, S. W., and V. Reinke, 2008. MEG-1 and MEG-2 are embryo-specific P-granule components required for germline development in Caenorhabditis elegans. Genetics 178 295–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis, J. A., and J. T. Fleming, 1995. Basic culture methods. Methods Cell Biol. 48 3–29. [PubMed] [Google Scholar]
- Li, S., C. M. Armstrong, N. Bertin, H. Ge, S. Milstein et al., 2004. A map of the interactome network of the metazoan C. elegans. Science 303 540–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, J., T. Rolef Ben-Shahar, D. Riemer, M. Treinin, P. Spann et al., 2000. Essential roles for Caenorhabditis elegans lamin gene in nuclear organization, cell cycle progression, and spatial organization of nuclear pore complexes. Mol. Biol. Cell 11 3937–3947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maine, E. M., J. Hauth, T. Ratliff, V. E. Vought, X. She et al., 2005. EGO-1, a putative RNA-dependent RNA polymerase, is required for heterochromatin assembly on unpaired DNA during C. elegans meiosis. Curr. Biol. 15 1972–1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mello, C. C., B. W. Draper, M. Krause, H. Weintraub and J. R. Priess, 1992. The pie-1 and mex-1 genes and maternal control of blastomere identity in early C. elegans embryos. Cell 70 163–176. [DOI] [PubMed] [Google Scholar]
- Morton, D. G., D. C. Shakes, S. Nugent, D. Dichoso, W. Wang et al., 2002. The Caenorhabditis elegans par-5 gene encodes a 14-3-3 protein required for cellular asymmetry in the early embryo. Dev. Biol. 241 47–58. [DOI] [PubMed] [Google Scholar]
- Moussa, F., R. Oko and L. Hermo, 1994. The immunolocalization of small nuclear ribonucleoprotein particles in testicular cells during the cycle of the seminiferous epithelium of the adult rat. Cell Tissue Res. 278 363–378. [DOI] [PubMed] [Google Scholar]
- Navarro, R. E., E. Y. Shim, Y. Kohara, A. Singson and T. K. Blackwell, 2001. cgh-1, a conserved predicted RNA helicase required for gametogenesis and protection from physiological germline apoptosis in C. elegans. Development 128 3221–3232. [DOI] [PubMed] [Google Scholar]
- Orban, T. I., and E. Izaurralde, 2005. Decay of mRNAs targeted by RISC requires XRN1, the Ski complex, and the exosome. RNA 11 459–469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel, S. S., B. J. Belmont, J. M. Sante and M. F. Rexach, 2007. Natively unfolded nucleoporins gate protein diffusion across the nuclear pore complex. Cell 129 83–96. [DOI] [PubMed] [Google Scholar]
- Pitt, J. N., J. A. Schisa and J. R. Priess, 2000. P granules in the germ cells of Caenorhabditis elegans adults are associated with clusters of nuclear pores and contain RNA. Dev. Biol. 219 315–333. [DOI] [PubMed] [Google Scholar]
- Reinke, V., I. S. Gil, S. Ward and K. Kazmer, 2004. Genome-wide germline-enriched and sex-biased expression profiles in Caenorhabditis elegans. Development 131 311–323. [DOI] [PubMed] [Google Scholar]
- Rexach, M., and G. Blobel, 1995. Protein import into nuclei: association and dissociation reactions involving transport substrate, transport factors, and nucleoporins. Cell 83 683–692. [DOI] [PubMed] [Google Scholar]
- Robert, V. J., T. Sijen, J. van Wolfswinkel and R. H. Plasterk, 2005. Chromatin and RNAi factors protect the C. elegans germline against repetitive sequences. Genes Dev. 19 782–787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocheleau, C. E., K. Cullison, K. Huang, Y. Bernstein, A. C. Spilker et al., 2008. The Caenorhabditis elegans ekl (enhancer of ksr-1 lethality) genes include putative components of a germline small RNA pathway. Genetics 178 1431–1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose, L. S., and K. Kemphues, 1998. The let-99 gene is required for proper spindle orientation during cleavage of the C. elegans embryo. Development 125 1337–1346. [DOI] [PubMed] [Google Scholar]
- Saffman, E. E., and P. Lasko, 1999. Germline development in vertebrates and invertebrates. Cell. Mol. Life Sci. 55 1141–1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schisa, J. A., J. N. Pitt and J. R. Priess, 2001. Analysis of RNA associated with P granules in germ cells of C. elegans adults. Development 128 1287–1298. [DOI] [PubMed] [Google Scholar]
- Seydoux, G., and R. E. Braun, 2006. Pathway to totipotency: lessons from germ cells. Cell 127 891–904. [DOI] [PubMed] [Google Scholar]
- Seydoux, G., and A. Fire, 1994. Soma-germline asymmetry in the distributions of embryonic RNAs in Caenorhabditis elegans. Development 120 2823–2834. [DOI] [PubMed] [Google Scholar]
- Seydoux, G., C. C. Mello, J. Pettitt, W. B. Wood, J. R. Priess et al., 1996. Repression of gene expression in the embryonic germ lineage of C. elegans. Nature 382 713–716. [DOI] [PubMed] [Google Scholar]
- She, X., X. Xu, A. Fedotov, W. G. Kelly and E. M. Maine, 2009. Regulation of heterochromatin assembly on unpaired chromosomes during Caenorhabditis elegans meiosis by components of a small RNA-mediated pathway. PLoS Genet. 5 e1000624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada, M., H. Yokosawa and H. Kawahara, 2006. OMA-1 is a P granules-associated protein that is required for germline specification in Caenorhabditis elegans embryos. Genes Cells 11 383–396. [DOI] [PubMed] [Google Scholar]
- Smith, L. D., 1966. The role of a “germinal plasm” in the formation of primordial germ cells in Rana pipiens. Dev. Biol. 14 330–347. [DOI] [PubMed] [Google Scholar]
- Snyder, M., X. Y. Huang and J. J. Zhang, 2009. The mini-chromosome maintenance proteins 2–7 (MCM2–7) are necessary for RNA polymerase II (pol II)-mediated transcription. J. Biol. Chem. 284 13466–13472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spike, C. A., and S. Strome, 2003. Germ plasm: protein degradation in the soma. Curr. Biol. 13 R837–R839. [DOI] [PubMed] [Google Scholar]
- Spike, C., N. Meyer, E. Racen, A. Orsborn, J. Kirchner et al., 2008. a Genetic analysis of the Caenorhabditis elegans GLH family of P-granule proteins. Genetics 178 1973–1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spike, C. A., J. Bader, V. Reinke and S. Strome, 2008. b DEPS-1 promotes P-granule assembly and RNA interference in C. elegans germ cells. Development 135 983–993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Squirrell, J. M., Z. T. Eggers, N. Luedke, B. Saari, A. Grimson et al., 2006. CAR-1, a protein that localizes with the mRNA decapping component DCAP-1, is required for cytokinesis and ER organization in Caenorhabditis elegans embryos. Mol. Biol. Cell 17 336–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stelter, P., R. Kunze, D. Flemming, D. Hopfner, M. Diepholz et al., 2007. Molecular basis for the functional interaction of dynein light chain with the nuclear-pore complex. Nat. Cell Biol. 9 788–796. [DOI] [PubMed] [Google Scholar]
- Strome, S., 2005. Specification of the germ line. WormBook 1–10. www.wormbase.org [DOI] [PMC free article] [PubMed]
- Strome, S., and W. B. Wood, 1982. Immunofluorescence visualization of germ-line-specific cytoplasmic granules in embryos, larvae, and adults of Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 79 1558–1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strome, S., and W. B. Wood, 1983. Generation of asymmetry and segregation of germ-line granules in early C. elegans embryos. Cell 35 15–25. [DOI] [PubMed] [Google Scholar]
- Tabara, H., E. Yigit, H. Siomi and C. C. Mello, 2002. The dsRNA binding protein RDE-4 interacts with RDE-1, DCR-1, and a DExH-box helicase to direct RNAi in C. elegans. Cell 109 861–871. [DOI] [PubMed] [Google Scholar]
- Vought, V. E., M. Ohmachi, M. H. Lee and E. M. Maine, 2005. EGO-1, a putative RNA-directed RNA polymerase, promotes germline proliferation in parallel with GLP-1/notch signaling and regulates the spatial organization of nuclear pore complexes and germline P granules in Caenorhabditis elegans. Genetics 170 1121–1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, G., and V. Reinke, 2008. A C. elegans Piwi, PRG-1, regulates 21U-RNAs during spermatogenesis. Curr. Biol. 18 861–867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, X., Y. Zhao, K. Wong, P. Ehlers, Y. Kohara et al., 2009. Identification of genes expressed in the hermaphrodite germ line of C. elegans using SAGE. BMC Genomics 10 213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Will, C. L., C. Schneider, R. Reed and R. Luhrmann, 1999. Identification of both shared and distinct proteins in the major and minor spliceosomes. Science 284 2003–2005. [DOI] [PubMed] [Google Scholar]
- Yankulov, K., I. Todorov, P. Romanowski, D. Licatalosi, K. Cilli et al., 1999. MCM proteins are associated with RNA polymerase II holoenzyme. Mol. Cell. Biol. 19 6154–6163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yigit, E., P. J. Batista, Y. Bei, K. M. Pang, C. C. Chen et al., 2006. Analysis of the C. elegans Argonaute family reveals that distinct Argonautes act sequentially during RNAi. Cell 127 747–757. [DOI] [PubMed] [Google Scholar]
- Zhang, Y., L. Yan, Z. Zhou, P. Yang, E. Tian et al., 2009. SEPA-1 mediates the specific recognition and degradation of P granule components by autophagy in C. elegans. Cell 136 308–321. [DOI] [PubMed] [Google Scholar]
- Zorio, D. A., N. N. Cheng, T. Blumenthal and J. Spieth, 1994. Operons as a common form of chromosomal organization in C. elegans. Nature 372 270–272. [DOI] [PubMed] [Google Scholar]