Abstract
A major challenge for developing effective therapeutic vaccines against cancer is overcoming immunological tolerance to tumor-associated antigens that are expressed on both malignant cells and normal tissues. Herein, we describe a novel vaccination approach, TriVax that utilizes synthetic peptides representing CD8 T cell epitopes, Toll-like receptor (TLR) agonists that function as a potent immunological adjuvants and costimulatory anti-CD40 antibodies to generate large numbers of high avidity antigen-reactive T cells capable of recognizing and killing tumor cells. Our results show that TriVax induced huge numbers of long-lasting antigen specific CD8 T cells that displayed significant anti-tumor effects in vivo. The administration of a TriVax formulation containing a CD8 T cell epitope derived from a melanosomal antigen (Trp2180-188) elicited antigen-specific CD8 T cells that induced systemic autoimmunity (vitiligo). More important, TriVax immunization was effective in eliciting potent protective anti-tumor immunity as well as remarkable therapeutic effects against established B16 melanoma. This therapeutic effect was mediated by CD8 T cells via perforin-mediated lysis and required the participation of type I-IFN but not IFNγ. These results suggest that similar strategies would be applicable for the design of effective vaccination for conducting clinical studies in cancer patients.
Introduction
There is substantial evidence that the immune system and in particular CD8 T cells have the ability to recognize and in some instances eliminate tumor cells (1–3). CD8 T cells recognize tumor antigens of the surface of tumor cells as complexes formed by small peptides that associate with MHC class I molecules (4). These T cell epitopes are derived from the processing of proteins synthesized by the tumor cells (5, 6). Numerous factors will determine whether a peptide can act as a tumor-reactive T cell epitope and function as a tumor rejection antigen. Most importantly is whether the peptide is produced by the endogenous antigen-processing pathway and can bind with sufficient strength to MHC class I molecules to be expressed on the surface of the tumor cell to interact with CD8 T cells. In addition, peptides derived from non-mutated proteins expressed on normal tissues may not be very immunogenic due to their propensity to induce immunological tolerance. Nevertheless, it appears that the mechanisms governing immune tolerance are not flawless and that in many instances auto-reactive CD8 T cell precursors can escape deletion or immune suppression and that when activated appropriately, potent autoimmune or anti-tumor responses can be generated. Several investigators including our group have considered using synthetic peptides representing CD8 T cell epitopes as vaccines for treating established tumors. Numerous studies that have evaluated synthetic peptide vaccines for cancer both in humans and in animal models have yielded rather disappointing results with respect to their anti-tumor effects indicating that peptides per se, are not very immunogenic (7–9). This conclusion is supported by the measurements of antigen-specific CD8 T cell responses induced by these vaccines, where in the majority of cases less than 1% of all CD8 T cells were shown to be antigen-specific. The inadequate performance of synthetic peptide-based vaccines may be due to the use of suboptimal immune adjuvants, which in most instances fail to activate the immune system resulting in T cell ignorance or worse, in the induction of T cell anergy (10, 11). The present studies were originated from the presumption that in order to achieve highly effective anti-tumor effects, peptide vaccines must be capable of generating CD8 T cell responses of a magnitude similar to those responses observed during acute infections where 10–50% of all CD8 T cells are antigen-specific. We present here evidence that with the appropriate combination of Toll-like receptor ligands (TLR-Ls) and costimulatory agonists it is possible to achieve these high levels of CD8 T cell responses using synthetic peptides as antigen. The intensity and duration of T cell responses reported here have not been observed with the use of any type of subunit vaccine and can only be compared to the results obtained with adoptive T cell therapy, which in many cases still requires subsequent vaccination and high dose IL-2 to achieve anti-tumor effects (12, 13). More importantly, our results show that this vaccination strategy was highly effective in a mouse model of melanoma where the employed CD8 T cell epitope represents a peptide derived from a tissue differentiation product expressed by normal melanocytes. We believe that the results from these preclinical studies open a new venue to developing effective peptide vaccination strategies in human patients.
Materials and Methods
Mice and cell lines
Female C57BL/6 (B6), IFNγ-deficient (IFNγ−/−) and perforin-deficient (Prf−/) mice were purchased from The Jackson Laboratory (Bar Harbor, ME). IFNαβ receptor-deficient (IFNαβR−/−) were a generous gift of Dr. Philippa Marrack (National Jewish Medical and Research Center, Denver, CO). All animal care and experiments were conducted according to our institutional animal care and use committee guidelines. The murine melanoma cell line B16F10 was provided by Dr. Jose Guevara (University of Chicago, Chicago, IL). The B16F10-Kb− cell line is a variant of B16F10 previously selected in our laboratory, which does not express H-2Kb molecules (but expresses H-2Db) even after treatment with IFNγ (using 100 ng/ml for 24 h). EL4 and its Ova-transfected variant EG7 were obtained from the American Type Culture Collection (ATCC, Manassas, VA).
Peptides, antibodies, and tetramers
Synthetic peptides representing H-2Kb-restricted CD8 T cell epitopes, Trp2180–188 (SVYDFFVWL), Ova257–264 (SIINFEKL) and Ova55–63 (KVVRFDKL) were purchased from A&A Labs (San Diego, CA). Rat anti-mouse CD40 monoclonal antibody (anti-CD40 mAb), was prepared from the FGK45.5 hybridoma. PE-conjugated H-2Kb/Ova257–264 tetramer was purchased from Beckman Coulter (Fullerton, CA). H-2Kb/Trp2180–188 tetramer was provided by the NIAID Tetramer Facility (Atlanta, GA).
Immunizations
For TriVax immunizations mice were immunized i.v. (unless otherwise noted) with a mixture of 200 μg peptide, 50 μg anti-CD40 mAb and a TLR-L (50 μg poly-IC, 100 μg CpG-1826, 30 μg LPS, 100 μg Gardiquimod (GDQ), or 25 μg FSL1). CpG-1826 was prepared by the Mayo Clinic Molecular Core Facility. Poly-IC (Hiltonol™, a clinical grade stabilized formulation containing poly-L-lysine and carboxymethyl cellulose) was provided by Dr. Andres Salazar (Oncovir, Inc., Washington, DC). LPS, GDQ, and FSL1 were purchased from InvivoGen, (San Diego, CA). In some instances mice received s.c. vaccination with peptide in PBS or emulsified in IFA in addition with anti-CD40 mAb and poly-IC, which were also injected s.c. in close proximity to the peptide.
Evaluation of cellular immune responses
For tetramer staining, either blood, or splenocytes were stained with FITC-conjugated MHC class II, PerCP Cy5.5-conjugated CD8a (both from eBioscience, San Diego, CA), and PE-conjugated Kb/peptide tetramers for 40 min in ice. Fuorescence was evaluated using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA). For detection of cytokine-secreting CD8 T cells, enzyme-linked immunosorbent spot (EliSpot) assays (IFNγ or TNFα) were performed as described previously (14) using purified CD8 T cells. For cytotoxicity determinations, conventional 5-h chromium release assays were performed using freshly isolated CD8 T cells (effectors) against various target cells that were labelled with 51Cr in triplicate using various effectors to target ratios.
Anti-tumor effects
To assess the efficacy of vaccination in the prophylactic setting, previously immunized mice were challenged with 3 × 105 B16F10 cells injected through the tail vein 12 days after the last vaccination. To study the therapeutic effects of vaccination, mice first received an i.v. tumor inoculation (3 × 104, 1 × 105 or 3 × 105 B16F10 cells) and 3 to 7 days later (depending on the specific experiment) the mice received their first immunization. Anti-tumor effects were evaluated by the examination and measurements of lung tumor nodules and lung weights approximately 4 weeks after tumor injections (when the mice in the unvaccinated control group started to appear ill). In some experiments the anti-tumor effects were evaluated by survival measurements (time to euthanasia). In all instances groups of mice consisted of 8 animals per group. For in vivo cell depletions (NK, CD4 or CD8 T cells), mice received the following mAbs via i.p. injections: Anti-NK (clone PK136) 300 μg/injection, anti-CD4 (clone GK1.5) 300 μg/injection and anti-CD8 (clone 2.43) 500 μg/injection. Depletions were confirmed by analysis of blood samples using flow cytometry (data not presented). In some instances, survivor mice were re-challenged s.c. in the posterior flank with either B16F10 or B16F10-Kb− cells (5 × 105/mouse). Tumor growth was monitored every 2–4 days in individual tagged mice by measuring 2 opposing diameters with a set of callipers. Results are presented as the mean tumor size (area in mm2) ± SD for every treatment group at various time points until the termination of the experiment.
Statistical analyses
Statistical significance to assess numbers of antigen specific CD8 T cells (EliSpot or tetramer analyses), absolute number of lung tumor nodules, lung weights, and tumor sizes were performed using Student’s t tests or two-way ANOVA. Statistical significance for survival was established by Kaplan-Mayer curves using log-rank tests. All analysis and graphics were done using GraphPad Prism (San Diego, CA).
Results
Peptide immunization can induce large numbers of long-lasting and functional antigen specific CD8 T cells
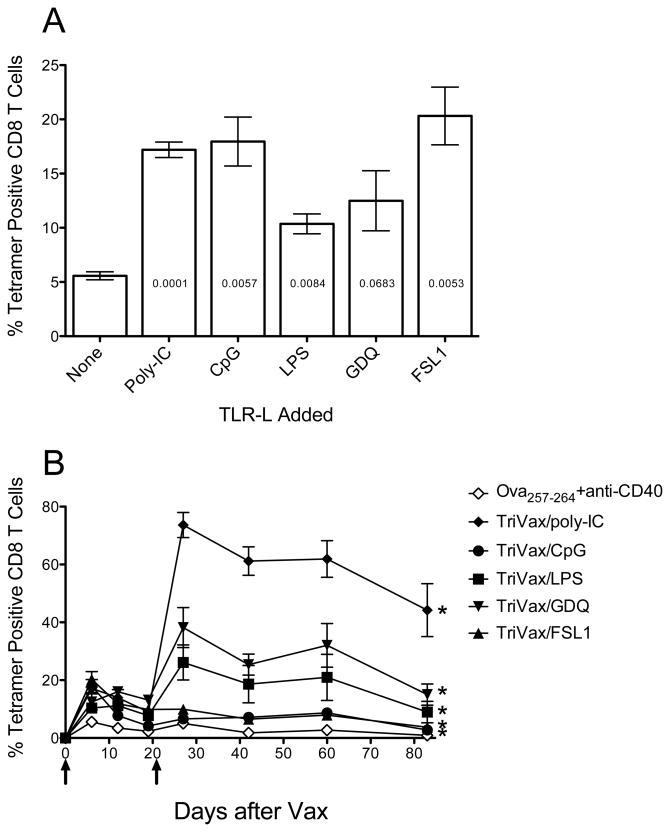
It has been reported that the use of agonistic monoclonal antibodies (mAb) specific for CD40 is capable of increasing T cell responses resulting from various types of immunizations including peptide vaccines (15–17). It is believed that anti-CD40 mAb functions as an adjuvant by preventing the induction of T cell anergy that peptides can induce when presented to CD8 T cells by non-professional APC. However, we have observed that the CD8 T cell responses generated in mice immunized with soluble peptide and anti-CD40 mAb exhibit clear signs of T cell anergy since a subsequent booster peptide immunization, even in the presence of anti-CD40 mAb, was unable to elicit a significant secondary immune response (H. Cho, unpublished). In view of this, we evaluated whether the use of Toll-like receptor ligands (TLR-Ls) would improve the immune response of peptide vaccination administered with anti-CD40 mAb. As shown in Figure 1A, the addition of a TLR-L to a vaccine consisting of peptide Ova257–264 and anti-CD40 mAb increased from 2–3 fold induced the levels of antigen-specific CD8 T cells observed in blood 6 days after the administration of the vaccine. Vaccination with peptide alone or peptide with either of these TLR-Ls w/o anti-CD40 mAb generated less than 0.5% tetramer positive cells (data not shown). An identical booster immunization was applied on day 21 and the levels of antigen-specific CD8 T cells in blood were monitored every ~20 days. The data in Figure 1B indicate that only 3 of the 5 TLR-Ls (poly-IC, GDQ and LPS) were able to elicit secondary CD8 T cell responses. These results indicate that peptide immunization with a vaccine containing 3 components: synthetic peptide anti-CD40 mAb and poly-IC, from hereafter referred to as TriVax generates antigen-specific CD8 T cells that are capable of dramatically expanding upon a booster immunization.
Figure 1. Effect of TLR-Ls on peptide vaccination with anit-CD40 mAb.
(A), B6 mince (3 per group) were immunized i.v. with Ova 257–264 peptide and anti-CD40 mAb and one of the following TLR-L: poly-IC (TLR3-L), CpG (TLR9-L), Lps (TLR4-L), GDQ (TLR7-L), FSL1 (TLR6/2-L). Seven days later, antigen-specific CD8 T cell responses were measured in blood by using tetramer analysis. Results represent the means and SD for each group where p values compared to the no TLR group are shown inside of each bar. (B), The mice received a second identical immunization (on day 21) and the frequency of tetramer-specific CD8 T cells in peripheral blood was followed in individual mice throughout various time points. Results correspond to the man and SD for each group. Arrows, times of vaccination. P values were calculated using two-way ANOVA test (*p < 0.0001 for TriVax versus peptide+anti-CD40 mAb).
Evaluation of TriVax using a peptide representing a CD8 tumor epitope
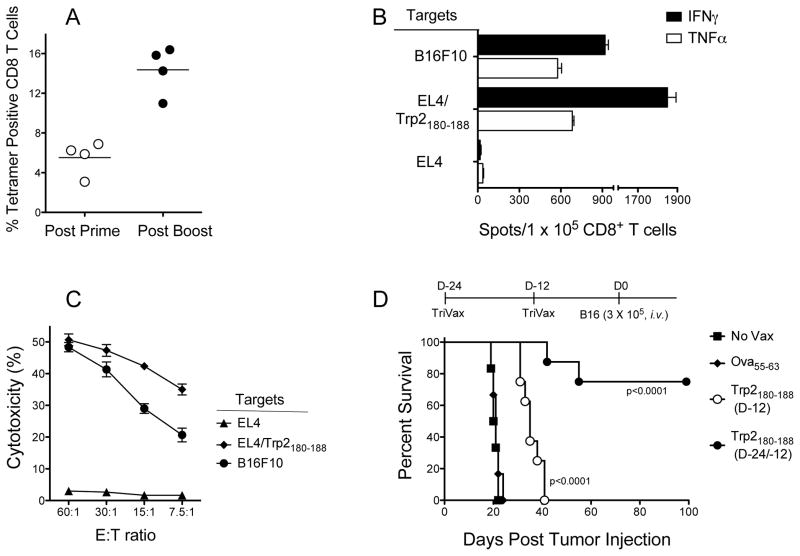
Next, we evaluated TriVax using a CD8 peptide epitope derived from the melanosomal tyrosinase-related protein 2 (Trp2, also known as dopachrome tautomerase). Trp2180–188 functions as an H-2Kb restricted CD8 T cell epitope expressed by B16 melanoma (18, 19). Trp2180–188-TriVax immunization induced a strong primary CD8 T cell response that could be increased by ~3-fold with a subsequent TriVax boost (Fig. 2A). The functional activity of CD8 T cells isolated from spleens was demonstrated in EliSpot assays (Fig. 2B). These responses were evident not only against peptide-pulsed APC but also towards B16 melanoma cells. Freshly isolated spleen CD8 T cells (w/o further peptide restimulation) were shown to high levels of cytotoxic activity (Fig. 2C). The levels of antigen-specific CD8 T cells in blood could be further increased (to 40–60% tetramer positive) when the mice received additional immunizations, remaining high for several months and presumably were responsible for the generation of extensive vitiligo (supplementary Fig. S1).
Figure 2. Immunization with Trp2180-–88-TriVax induces strong anti-tumor Cd8 T cell responses.
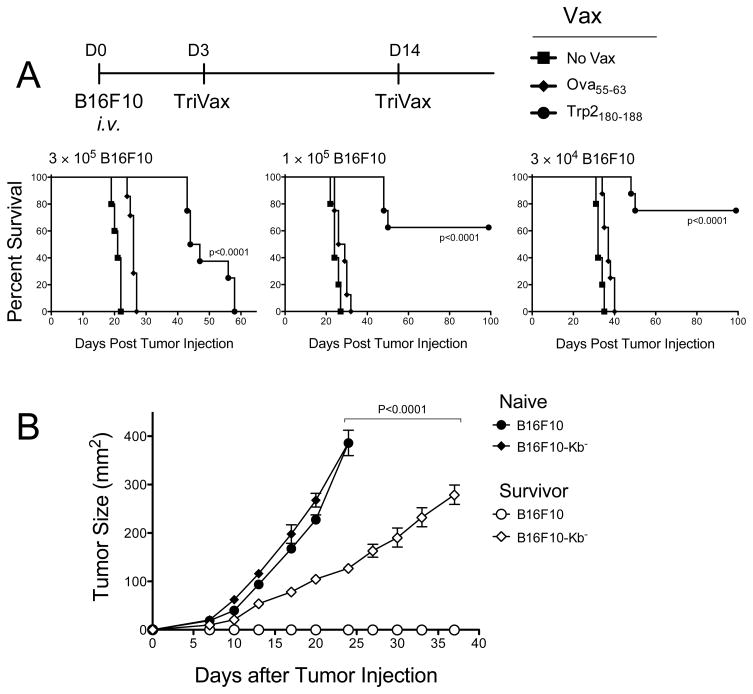
B6mice were vaccinated i.v. on days 0 and 14 with Trp2180–188-TriVax/poly-IC.(A), On days 7 and 22, blood samples were evaluated by tetramer analysis. Each point represents the value for each individual mouse and horizontal line represents the average value of the group. (B), Eight days after the boost, CD8 T cells were purified from pooled splenocytes and antigen-induced IFNγ and TNFα secretion was evaluated by EliSpot. (C), Cytolytic activity of freshly isolated CD8 T cells, assessed by a 51Cr-release assay. (D), B6 mice (8 per group) were immunized on days −24 and −12 or only on day −12 with Trp2180–188-TriVax/poly-IC. Twelve days after the last immunization, the mice received 3 × 105 B16F10 melanoma cells i.v. Survival is compared against unvaccinated group (No Vax) or group that received Ova55–63-TriVax/poly-IC (negative control). Kaplan–Mayer survival curves for all groups of mice are shown. P values were determined by log-rank tests as compared to the negative control group.
Next, we assessed whether the CD8 T cells generated with TriVax immunization would be capable of displaying in vivo anti-tumor effects. Mice received various modes of TriVax and 12 days after the last immunization they were challenged with B16 cells. We also compared the anti-tumor efficacy of a single immunization versus a prime/boost. The results indicate that TriVax with Trp2180–188 offered significant protection against B16 and that the prime/boost protocol was more effective than a single immunization (Fig. 2D).
Therapeutic effects of TriVax against established B16 melanoma
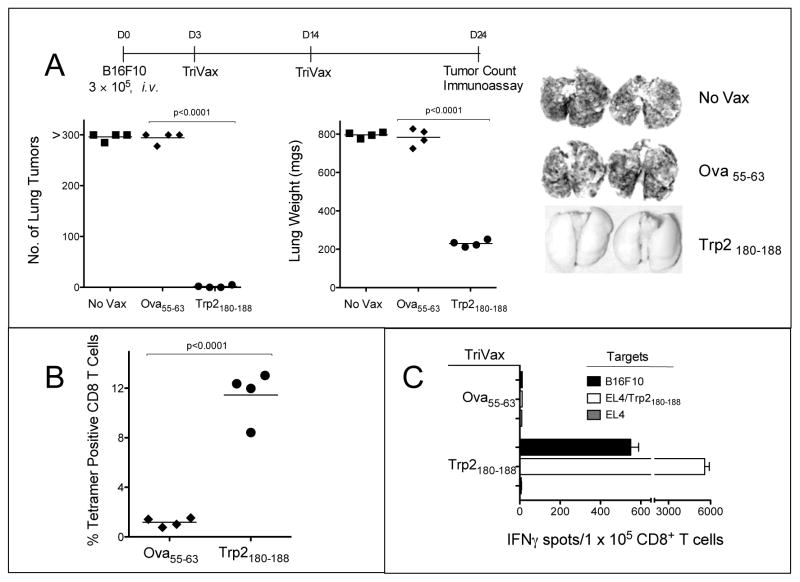
Next, we studied whether TriVax would offer a therapeutic benefit against established B16 tumors. Mice received an intravenous injection of B16F10 cells and 3 days later the vaccinations were initiated. On day 24, when the control mice appeared sick, all mice were euthanatized and their lungs were evaluated for the presence of tumors. As shown in Figure 3A, the lungs from mice that received Trp2180–188-TriVax had little evidence of disease as compared to the control mice. Furthermore, the mice that received the Trp2180–188-TriVax had high levels of antigen-specific CD8 T cells while the mice that received the Ova55–63-TriVax did not have any detectable Trp2180–188-specific CD8 T cells (Figs. 3B and 3C). We also evaluated the therapeutic effects of Trp2180–188-TriVax against established (7-day) s.c. B16 tumors. Although no tumor rejections were obtained in this more challenging model, a significant reduction in the tumor growth rate was observed (supplementary Fig. S2). The anti-tumor effects of TriVax were evaluated against more established tumors. Significant anti-tumor effects were still observed when mice received their first immunization either 5 or 7 days after the i.v. tumor inoculation (supplementary Fig. S3). However, the lungs from the Trp2180–188-TriVax mice on day 7 had some evidence of small tumor nodules, even though these mice exhibited large numbers of antigen-specific CD8 T cells (supplementary Fig. S3).
Figure 3. Therapeutic anti-tumor effects of Trp2180–188-TriVax.
(A), B6 mice (4 per group) received 3 × 105 B16Fl0 cells i.v., and 3 and 11 days later the mice were vaccinated with Trp2180–188-TriVax or Ova55–63-TriVax. A non-vaccinated group (No Vax) was also included. On Day 24 (when the control mice appeared sick), the presence of B16 pulmonary nodules was evaluated. Results are presented as “Numbers of Lung Tumors” and “Lung Weights” for individual mice. Representative photographs of lungs of 2 mice from each group are shown. Splenocytes from mice of the 2 vaccinated groups in “A” were evaluated for Trp2-specific CD8 T cells by tetramer analysis (B), and antigen-induced IFNγ production in EliSpot assays (C). P values were calculated using unpaired Student’s test.
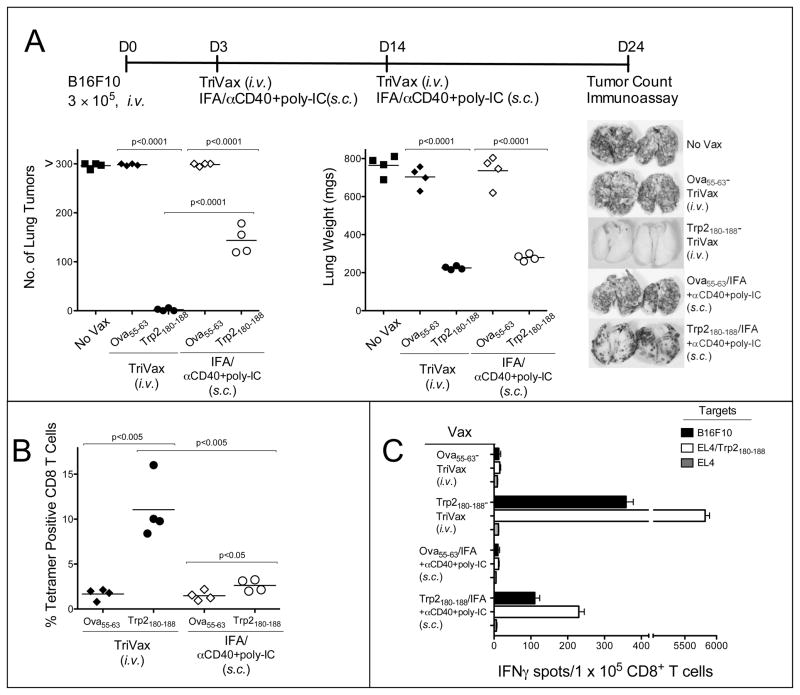
Vaccination with peptides can induce T cell anergy, especially when the peptides are administered in soluble form via an i.v. route. To avoid this problem, peptides are administered via a s.c. route, formulated in a water-in-oil emulsions such as incomplete Freund’s adjuvant (IFA). Thus, we compared the therapeutic anti-tumor responses of the conventional TriVax i.v. immunization with a TriVax modification where the peptide was injected s.c. in IFA and where the poly-IC and anti-CD40 mAb were also injected in close proximity to the peptide emulsion. The s.c. administration of Trp2180–188/IFA-TriVax exhibited some therapeutic benefit but was not as effective as the i.v. Trp2180–188-TriVax (Fig. 4A). These results may be explained by the differences of the Trp2180–188-specific CD8 T cell responses observed with these vaccines (Figs. 4B and 4C). Suboptimal anti-tumor effects were also obtained when TriVax formulated with soluble peptide (no IFA) was administered via a s.c. route as compared to the i.v. route (supplementary Fig. S4), indicating that systemic administration of the vaccine was important to generate a therapeutic effect.
Figure 4. Soluble peptide is more effective than peptide emulsified in IFA in TriVal.
(A), B6 mice (4 per group) received 3 × 105 B16F10 cells i.v. and 3 and 11 days later were vaccinated either i.v. or s.c. with Trp2180–188-TriVax or Ova55–63-TriVax. Peptides were emulsified in IFA for the s.c. vaccines. A non-vaccinated group (No Vax) was included. On day 24 all the mice were the presence of B16 pulmonary tumors was evaluated as described in Figure 3A. Splenocytes from individual mice were evaluated for Trp2-specific CD8 T cells by tetramer analysis (B) or EliSpot assaya (C) P values were calculated using unpaired Student’s test.
TriVax was evaluated for its survival benefit of in the pulmonary B16 tumor model. The results shown in Figure 5A demonstrate that with a high tumor dose, TriVax increased the medium survival by ~25 days, but by day 58 all the mice perished. However, the survival advantage of TriVax was enhanced when the tumor dose was decreased. Interestingly, post-mortem examination of the Trp2180-188-TriVax immunized mice that died in these experiments revealed only few, if any, lung tumor nodules (supplementary Fig. S5). However, in some instances tumors developed in the peritoneal cavity or in the pericardium (not shown). To assess the presence of long-term immunity, the surviving TriVax immunized mice were rechallenged with a s.c. B16 injection. Half of the mice received B16F10 cells and the other half were challenged with a B16F10 tumor variant that does not express the H-2Kb molecule (B16F10-Kb−). None of the immune mice developed tumors when rechallenged s.c. with B16F10 cells (Fig. 5B). On the other hand, the mice challenged with the B16F10-Kb− cells developed tumors but the growth rate was significantly slower than the control group that was challenged with the same tumor suggesting that these mice may have acquired additional anti-tumor immune mechanisms (antibodies, CD4 T cell responses or H-2Db-restricted CD8 T cell epitope) capable of decreasing growth of tumors not expressing the H-2Kb-restricted Trp2180–188 CD8 epitope.
Figure 5. Therapeutic TriVax results in increased survival.
(A), B6 mice (8 per group) received 3 different doses of B16F10 cells i.v. (as indicated) and 3 days later the mice were immunized with either Trp2180–188-TriVax, Ova55–63-TriVax or left unvaccinated (No Vax). P values were determined by log-rank tests. (B), At the termination of the experiment presented in (A), survivor mice were pooled, randomized and received s.c. tumor challenge with either B16F10 or B16F10-kb− cells (5 × 105 cell/mouse). Naï ve, unvaccinated mice inoculated with the same number of tumore cells were included as control. Tumore sizes were determined in individual mice by measurements of 2 opposing diameters and are presented as tumore areas in mm2. Each data point corresponds to the means and SD for each group of mice (5 per group). P value was calculated using two-ways ANOVA test.
Evaluation of the immune effector mechanisms of TriVax
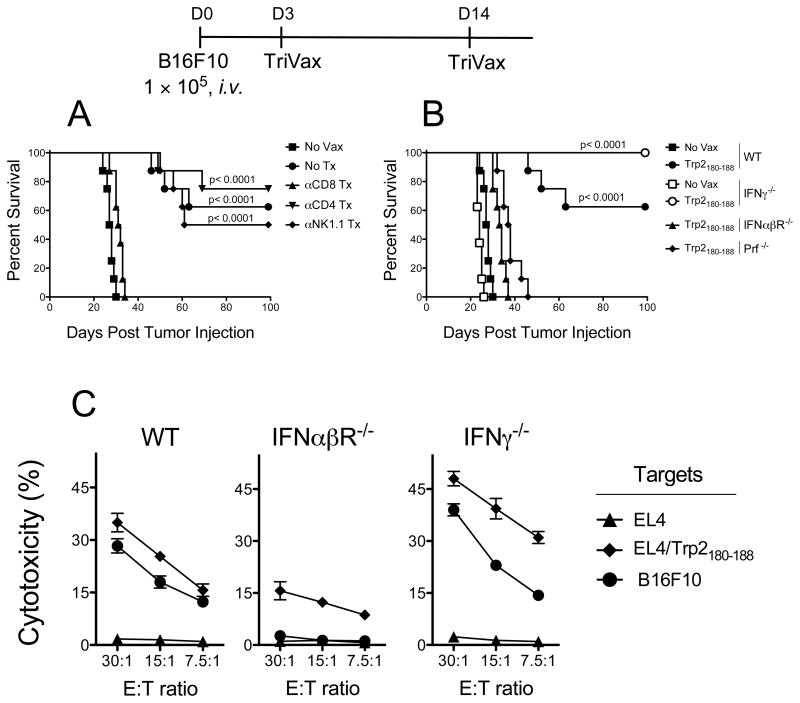
Lastly we assessed the roles of various immune effector cells such as NK and CD4 T cells that could participate in the induction of the CD8 T cell response and/or synergize with CD8 T cells in fighting the established tumors. Depletion of either CD4 T cells or NK cells had no significant deleterious effect in the survival advantage provided by TriVax while depletion of CD8 T cells completely abolished the anti-tumor effects (Fig. 6A). CD8 T cells can exert their anti-tumor effect via numerous effector mechanisms such as perforin mediated cytolysis (20) and IFNγ (21–23). In addition, type-I IFN has been implicated as being critical in CD8 T cell responses (24). Thus, the efficacy of Trp2180–188-TriVax was evaluated in mice deficient for perforin, IFNγ, or type-I IFN receptor. The therapeutic benefit of TriVax was absent in mice that lack perforin or type-I IFN receptor, indicating that these molecules are crucial for the rejection of established tumors (Fig. 6B). Conversely, an increase in the therapeutic effect of Trp2180–188-TriVax was observed in mice deficient for IFNγ, suggesting that this cytokine may somehow inhibit the function of CD8 T cells in this model. Cell-mediated cytotoxicity assays revealed that the IFNαβR−/− mice had decreased CD8 T cell activity as compared to B6 WT mice, while the IFNγ-deficient mice had slightly higher levels of activity (Fig. 6C). A similar pattern was observed when CD8 T cells in these mice were evaluated by tetramer analysis (supplementary Fig. S6). Overall, these results demonstrate that the anti-tumor effect of Trp2180–188-TriVax in this tumor model is mostly derived from perforin-mediated lysis of tumor cells by CD8 T cells.
Figure 6. Effector mechanisms involved in anti−tumor effects of TriVax.
(A), B6 mice (8 per group) received with 1 × 10 5 B16F10 cells i.v. Various subsets of immune effector cells (CD8 T cells, CD4 T cells or NK cells) were depleted using mAb on days −3 and −1 before receiving the TriVax injection. An addition mAb injection was administered 2 days after vaccinatioin. TriVax immunizations were administered on days 3 and 14 after tumor injections. Kaplan–Mayer survival curves for all groups of mice are shown. P values were determined by log-rank tests. (B), Therapeutic effects of Trp2180–188-TriVax in WT B6 mice (WT), IFNγ−/−, IFNαβR−/−, and Prf−/− mice were evaluated a described above. Results were evaluated for statistical significance using log-rank test. (C), In a parallel experiment freshly isolated spleen CD8 T cells from WT, IFNγ−/−, IFNαβR−/−, mice were tested for cytolytic activity in a 5-h 51Cr-release assay as described in Figure 2C.
Discussion
The development of T cell-based immunotherapy for cancer continues to be a major challenge and one of the top priorities for tumor immunologists. The most clear and dramatic example showing that T lymphocytes can eliminate large tumors come from studies using adoptive T cell therapy (12, 25). Adoptive T cell therapy usually requires the administration of high doses of IL-2 and in many instances lymphoablation and a subsequent strong immunization (e.g., recombinant vaccinia) to achieve anti-tumor effects (13). Under these conditions, very high numbers of antigen-specific T cells are achieved, which rarely can be observed with any conventional vaccines w/o the use of adoptive T cell transfers. Numerous homeostatic mechanisms operate in the immune system to ensure that numbers T cells for various antigenic specificities increase (expansion phase) when required to combat disease and later decrease (contraction phase) when the threat subsides and are no longer needed (26). To successfully combat infections the immune system overrides homeostasis allowing huge expansion of pathogen-specific T lymphocytes (27). The majority of the peptide-based vaccines fail to generate the vast CD8 T cell responses obtained by adoptive T cell therapy/lymphoablation or observed during acute infections and therefore it is not surprising that these perform poorly against tumors.
We present here the results of our attempts to design and implement a peptide vaccination strategy that mimics an acute infection generating CD8 T cell responses of the magnitude observed in such infections and achieved by adoptive T cell therapy. We assessed the adjuvant activity of 5 compounds known to stimulate different TLR family members. After primary immunization, all of the TLR-L tested significantly increased the levels of antigen specific CD8 T cells as compared to the use of peptide and anti-CD40 mAb alone (Fig. 1A). Interestingly, only some of the TLR-L were able to generate secondary immune responses (Fig. 1B). The results obtained with poly-IC were most dramatic, where at the peak of the response (6 days post-boost) nearly 80% of the CD8 T cells in blood were antigen specific. To our knowledge this could be the first example where these high levels of CD8 T cells are generated using any kind of non-infectious vaccine and w/o a prior infusion of TCR transgenic T cells. At present we do not know the reasons why the various TLR-L performed so differently as adjuvants in the TriVax immunization strategy. These differences could be due to variations in stability, biodistribution and pharmacokinetics of these compounds or due to their capacity to stimulate different cells of the immune system. In addition, poly-IC stimulates the immune system through other pathways in addition to TLR3 (28) (29, 30). The combination of TLR-L, anti-CD40 mAb and peptide for the generation of CD8 T cell responses has been used by other groups, but for unknown reasons their results differ substantially from ours. Ahonen, et al. first reported the generation of strong CD8 T cell responses to Ova257–263, but subsequent boosters were not effective in generating secondary immune responses (17). The same group applied were unable to elicit CD8 T cell responses to the Trp2180–188 natural epitope (SVYDFFVWL) unless they used an analogue synthetic peptide (31). Moreover, even when the analogue peptide was used, booster immunizations remained ineffective and the anti-tumor effects were suboptimal. In other studies Wells, et al., showed that the use of TLR-L combinations improved significantly the immunogenicity and anti-tumor effectiveness of peptide vaccines administered in combination with anti-CD40 mAb and IFNγ, as compared to the use of a single TLR-L, which for them proved to be suboptimal (32). We do not know what the experimental conditions are between our group and the work of others that may account for the different results. Regardless of the reasons for the different results, we believe that the capacity of a vaccine to induce CD8 T cells capable of further expanding after a subsequent encounter with antigen (e.g., booster immunization) will determine its effectiveness against tumors. Our results clearly show that in the case of B16 melanoma, to achieve effective anti-tumor responses, that magnitude of the antigen-specific T cell response is important.
The therapeutic anti-tumor response generated by TriVax required the participation of CD8 T cells but not any contribution of CD4 T cells or NK cells (Fig. 6). In addition, our results indicate that tumors were eliminated mostly via perforin-mediated lysis and that production of IFNγ in this tumor model was not only not required, but could also be detrimental for tumor eradication. The increased anti-tumor therapeutic effect of TriVax against B16 melanoma observed in IFNγ-deficient mice has been observed in numerous experiments and even with established s.c. tumors (H. Cho, unpublished). While the exact mechanism by which IFNγ decreases the therapeutic efficacy of TriVax against B16 tumors remains unknown, it is possible that IFNγ may reduce the magnitude of CD8 T cell responses, as previously reported (33–36) and as suggested by our results (Fig. 6C). Furthermore, we have observed that in vitro IFNγ-treatment of B16 cells increases MHC-I expression, but significantly reduces the capacity of the CD8 T cell to recognize the tumors as observed both in EliSpot and cytotoxicity assays (H. Cho, unpublished). We are currently investigating whether this phenomenon extends to other melanosomal CD8 T cell epitopes and if it occurs in other tumor models besides B16 melanoma where the TriVax immunotherapy is being explored.
Supplementary Material
Acknowledgments
The authors would like to thank the NIAID Tetramer Facility for providing valuable reagents. We also wish to thank Dr. Andres Salazar (Oncovir, Inc.) for providing vast amounts of poly-IC (Hiltonol™). Supported in part by NIH grants R01CA80782, R01CA103921, R01CA136828 and by joint developmental funds provided by the Donald A. Adam Comprehensive Melanoma Research Center and the Bankhead Coley Melanoma Pre-SPORE Program of the Moffitt Cancer Center.
Footnotes
Conflict of interest: The authors state that there is no financial interest in this work.
References
- 1.Cavallo F, Offringa R, van der Burg SH, Forni G, Melief CJ. Vaccination for treatment and prevention of cancer in animal models. Adv Immunol. 2006;90:175–213. doi: 10.1016/S0065-2776(06)90005-4. [DOI] [PubMed] [Google Scholar]
- 2.Dougan M, Dranoff G. Immune therapy for cancer. Annu Rev Immunol. 2009;27:83–117. doi: 10.1146/annurev.immunol.021908.132544. [DOI] [PubMed] [Google Scholar]
- 3.Finn OJ. Cancer immunology. N Engl J Med. 2008;358:2704–15. doi: 10.1056/NEJMra072739. [DOI] [PubMed] [Google Scholar]
- 4.Van Der Bruggen P, Zhang Y, Chaux P, et al. Tumor-specific shared antigenic peptides recognized by human T cells. Immunol Rev. 2002;188:51–64. doi: 10.1034/j.1600-065x.2002.18806.x. [DOI] [PubMed] [Google Scholar]
- 5.Groothuis T, Neefjes J. The ins and outs of intracellular peptides and antigen presentation by MHC class I molecules. Curr Top Microbiol Immunol. 2005;300:127–48. doi: 10.1007/3-540-28007-3_6. [DOI] [PubMed] [Google Scholar]
- 6.Trombetta ES, Mellman I. Cell biology of antigen processing in vitro and in vivo. Annu Rev Immunol. 2005;23:975–1028. doi: 10.1146/annurev.immunol.22.012703.104538. [DOI] [PubMed] [Google Scholar]
- 7.Boon T, Coulie PG, Van den Eynde BJ, van der Bruggen P. Human T cell responses against melanoma. Annu Rev Immunol. 2006;24:175–208. doi: 10.1146/annurev.immunol.24.021605.090733. [DOI] [PubMed] [Google Scholar]
- 8.Celis E. Overlapping human leukocyte antigen class I/II binding peptide vaccine for the treatment of patients with stage IV melanoma: evidence of systemic immune dysfunction. Cancer. 2007;110:203–14. doi: 10.1002/cncr.22744. [DOI] [PubMed] [Google Scholar]
- 9.Rosenberg SA, Yang JC, Restifo NP. Cancer immunotherapy: moving beyond current vaccines. Nat Med. 2004;10:909–15. doi: 10.1038/nm1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aichele P, Brduscha-Riem K, Zinkernagel RM, Hengartner H, Pircher H. T cell priming versus T cell tolerance induced by synthetic peptides. J Exp Med. 1995;182:261–6. doi: 10.1084/jem.182.1.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Toes RE, Offringa R, Blom RJ, Melief CJ, Kast WM. Peptide vaccination can lead to enhanced tumor growth through specific T-cell tolerance induction. Proc Natl Acad Sci U S A. 1996;93:7855–60. doi: 10.1073/pnas.93.15.7855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rosenberg SA, Restifo NP, Yang JC, Morgan RA, Dudley ME. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat Rev Cancer. 2008;8:299–308. doi: 10.1038/nrc2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Overwijk WW, Theoret MR, Finkelstein SE, et al. Tumor regression and autoimmunity after reversal of a functionally tolerant state of self-reactive CD8+ T cells. J Exp Med. 2003;198:569–80. doi: 10.1084/jem.20030590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cho HI, Niu G, Bradley N, Celis E. Optimized DNA vaccines to specifically induce therapeutic CD8 T cell responses against autochthonous breast tumors. Cancer Immunol Immunother. 2008;57:1695–703. doi: 10.1007/s00262-008-0465-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Diehl L, den Boer AT, Schoenberger SP, et al. CD40 activation in vivo overcomes peptide-induced peripheral cytotoxic T-lymphocyte tolerance and augments anti-tumor vaccine efficacy. Nat Med. 1999;5:774–9. doi: 10.1038/10495. [DOI] [PubMed] [Google Scholar]
- 16.Schoenberger SP, Toes RE, van der Voort EI, Offringa R, Melief CJ. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature. 1998;393:480–3. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- 17.Ahonen CL, Doxsee CL, McGurran SM, et al. Combined TLR and CD40 triggering induces potent CD8+ T cell expansion with variable dependence on type I IFN. J Exp Med. 2004;199:775–84. doi: 10.1084/jem.20031591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bloom MB, Perry-Lalley D, Robbins PF, et al. Identification of tyrosinase-related protein 2 as a tumor rejection antigen for the B16 melanoma. J Exp Med. 1997;185:453–9. doi: 10.1084/jem.185.3.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schreurs MW, Eggert AA, de Boer AJ, et al. Dendritic cells break tolerance and induce protective immunity against a melanocyte differentiation antigen in an autologous melanoma model. Cancer Res. 2000;60:6995–7001. [PubMed] [Google Scholar]
- 20.Catalfamo M, Henkart PA. Perforin and the granule exocytosis cytotoxicity pathway. Curr Opin Immunol. 2003;15:522–7. doi: 10.1016/s0952-7915(03)00114-6. [DOI] [PubMed] [Google Scholar]
- 21.Blankenstein T, Qin Z. The role of IFN-gamma in tumor transplantation immunity and inhibition of chemical carcinogenesis. Curr Opin Immunol. 2003;15:148–54. doi: 10.1016/s0952-7915(03)00007-4. [DOI] [PubMed] [Google Scholar]
- 22.Dunn GP, Ikeda H, Bruce AT, et al. Interferon-gamma and cancer immunoediting. Immunol Res. 2005;32:231–45. doi: 10.1385/ir:32:1-3:231. [DOI] [PubMed] [Google Scholar]
- 23.Young HA, Bream JH. IFN-gamma: recent advances in understanding regulation of expression, biological functions, and clinical applications. Curr Top Microbiol Immunol. 2007;316:97–117. doi: 10.1007/978-3-540-71329-6_6. [DOI] [PubMed] [Google Scholar]
- 24.Mescher MF, Curtsinger JM, Agarwal P, et al. Signals required for programming effector and memory development by CD8+ T cells. Immunol Rev. 2006;211:81–92. doi: 10.1111/j.0105-2896.2006.00382.x. [DOI] [PubMed] [Google Scholar]
- 25.June CH. Adoptive T cell therapy for cancer in the clinic. J Clin Invest. 2007;117:1466–76. doi: 10.1172/JCI32446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Surh CD, Sprent J. Homeostasis of naive and memory T cells. Immunity. 2008;29:848–62. doi: 10.1016/j.immuni.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 27.Antia R, Ganusov VV, Ahmed R. The role of models in understanding CD8+ T-cell memory. Nat Rev Immunol. 2005;5:101–11. doi: 10.1038/nri1550. [DOI] [PubMed] [Google Scholar]
- 28.Kawai T, Akira S. Innate immune recognition of viral infection. Nat Immunol. 2006;7:131–7. doi: 10.1038/ni1303. [DOI] [PubMed] [Google Scholar]
- 29.Gitlin L, Barchet W, Gilfillan S, et al. Essential role of mda-5 in type I IFN responses to polyriboinosinic:polyribocytidylic acid and encephalomyocarditis picornavirus. Proc Natl Acad Sci U S A. 2006;103:8459–64. doi: 10.1073/pnas.0603082103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kato H, Takeuchi O, Sato S, et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature. 2006;441:101–5. doi: 10.1038/nature04734. [DOI] [PubMed] [Google Scholar]
- 31.McWilliams JA, McGurran SM, Dow SW, Slansky JE, Kedl RM. A modified tyrosinase-related protein 2 epitope generates high-affinity tumor-specific T cells but does not mediate therapeutic efficacy in an intradermal tumor model. J Immunol. 2006;177:155–61. doi: 10.4049/jimmunol.177.1.155. [DOI] [PubMed] [Google Scholar]
- 32.Wells JW, Cowled CJ, Farzaneh F, Noble A. Combined triggering of dendritic cell receptors results in synergistic activation and potent cytotoxic immunity. J Immunol. 2008;181:3422–31. doi: 10.4049/jimmunol.181.5.3422. [DOI] [PubMed] [Google Scholar]
- 33.Badovinac VP, Tvinnereim AR, Harty JT. Regulation of antigen-specific CD8+ T cell homeostasis by perforin and interferon-gamma. Science. 2000;290:1354–8. doi: 10.1126/science.290.5495.1354. [DOI] [PubMed] [Google Scholar]
- 34.Balashov KE, Khoury SJ, Hafler DA, Weiner HL. Inhibition of T cell responses by activated human CD8+ T cells is mediated by interferon-gamma and is defective in chronic progressive multiple sclerosis. J Clin Invest. 1995;95:2711–9. doi: 10.1172/JCI117973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dalton DK, Pitts-Meek S, Keshav S, Figari IS, Bradley A, Stewart TA. Multiple defects of immune cell function in mice with disrupted interferon-gamma genes. Science. 1993;259:1739–42. doi: 10.1126/science.8456300. [DOI] [PubMed] [Google Scholar]
- 36.Groux H, Monte D, Plouvier B, Capron A, Ameisen JC. CD3-mediated apoptosis of human medullary thymocytes and activated peripheral T cells: respective roles of interleukin-1, interleukin-2, interferon-gamma and accessory cells. Eur J Immunol. 1993;23:1623–9. doi: 10.1002/eji.1830230734. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.