Summary
Although it is well established that Six3 is a crucial regulator of vertebrate eye and forebrain development, it is unknown whether this homeodomain protein has a role in the initial specification of the anterior neural plate. In this study, we show that exogenous Six3 can expand the anterior neural plate in both Xenopus and zebrafish, and that this occurs in part through Six3-dependent transcriptional regulation of the cell cycle regulators cyclinD1 and p27Xic1, as well as the anti-neurogenic genes Zic2 and Xhairy2. However, Six3 can still expand the neural plate in the presence of cell cycle inhibitors and we show that this is likely to be due to its ability to repress the expression of Bmp4 in ectoderm adjacent to the anterior neural plate. Furthermore, exogenous Six3 is able to restore the size of the anterior neural plate in chordino mutant zebrafish, indicating that it has the ability to promote anterior neural development by antagonising the activity of the BMP pathway. On its own, Six3 is unable to induce neural tissue in animal caps, but it can do so in combination with Otx2. These results suggest a very early role for Six3 in specification of the anterior neural plate, through the regulation of cell proliferation and the inhibition of BMP signalling.
Keywords: Six3, Cell proliferation, Cell fate, Bmp4, Xenopus, Zebrafish
Introduction
A complex and well-coordinated sequence of cell movements and inductive signals orchestrate the specification of dorsal ectoderm to form the anterior neural plate (Kuroda et al., 2004). The BMP subfamily of TGFβ proteins plays a key role in specification and patterning of the ectoderm. High levels of BMP signalling drives unspecified ectodermal cells towards an epidermal rather than neural fate, whereas neural tissue differentiates in regions of low BMP activity (Munoz-Sanjuan and Hemmati-Brivanlou, 2002). Cells at the neural plate boundary are likely to be exposed to intermediate levels of BMP activity and differentiate as neural crest, placodes and cement gland (Morgan and Sargent, 1997; Barth et al., 1999; Nguyen et al., 2000; Aybar and Mayor, 2002).
Specification and patterning of the anterior neural plate is crucially dependent upon the spatial localisation of BMP signalling activity. At early stages, BMP genes are expressed throughout the ectoderm, but, before the beginning of gastrulation, transcription is downregulated dorsally, where the neural plate is going to form (Wilson and Edlund, 2001; Stern, 2002; Kuroda et al., 2004). Two subsequent steps appear to regulate BMP expression and activity in the dorsal ectoderm. First, at blastula stages, Wnt and FGF signalling is crucial to suppress the transcription of BMP genes in the dorsal ectoderm (Baker et al., 1999; Bally-Cuif and Hammerschmidt, 2003; Kuroda et al., 2004). Second, by the late blastula stages, BMP antagonists, such as Noggin and Chordin secreted by the organizer, interact with BMPs and prevent their binding to receptors. Bmp4 transcription is maintained by an autoregulatory loop, where BMP4 protein bound to its receptor stimulates transcription of the Bmp4 gene (Jones et al., 1992; Hammerschmidt et al., 1996; Piccolo et al., 1997). The activity of BMP antagonists can interrupt this positive-feedback loop.
How different signalling pathways such as Wnt, FGF and BMP are integrated during early ectodermal development is still poorly understood. In anamniotes, it is likely that the activity of Wnt/βcatenin induces dorsal activation of genes that modulate FGF signalling; once activated, FGFs mediate an early restriction of BMP expression and activate the expression of BMP antagonists in the organizer (Furthauer et al., 2004; Tsang et al., 2004; Kudoh et al., 2004). Despite the early repression of BMP activity in presumptive neural territories, BMP signalling maintains the ability to inhibit expression of anterior neural plate genes even during neurulation (Hartley et al., 2001). This is of particular relevance considering that the anterior neural plate is surrounded by non-neural ectoderm and is underlain by anterior mesendoderm, both of which are sources of BMPs. Persistent suppression of BMP transcription in the anterior neural plate may be maintained by specific transcriptional repressors activated after neural induction (Hartley et al., 2001). This is indeed the case for XBF2 and Xiro1, which act as Bmp4 transcriptional repressors at early neurula stage, thus ensuring proper neural fate acquisition (Mariani and Harland, 1998; Gomez-Skarmeta et al., 2001).
In addition to suppression of BMP signalling, the rostral neural plate must be protected from the activity of caudalising signals for it to establish anterior forebrain character. Among the signals that promote posterior neural identity are Wnts, and a variety of Wnt antagonists ensure that Wnt activity is suppressed rostrally. The neural plate is patterned along its anteroposterior axis by the graded activity of Wnts, Wnt antagonists and other signals (Wilson and Houart, 2004). This is established by the interplay of Wnt antagonists secreted by the anteriormost neuroectoderm and the underlying mesendoderm, and local sources of Wnt signals in the posterior neuroectoderm, midbrain, diencephalon and mesendodermal tissues (Heisenberg et al., 2001; Kiecker and Niehrs, 2001; Houart et al., 2002). An early event in neural plate patterning is the generation of an anterior region that comprises the presumptive telencephalon, diencephalon and retina. The repression of both Wnt and BMP signalling, together with an enhanced proliferative activity, are crucial for the formation of the anterior neural plate (Zuber et al., 2003; Wilson and Houart, 2004). In fact, embryos with blocked cell proliferation or exaggerated Wnt or BMP signalling display anterior deficiencies (Hammerschmidt et al., 1996; Kim et al., 2000; Houart et al., 2002; Andreazzoli et al., 2003; Zakin and De Robertis, 2004).
Downstream of the signals that subdivide the ectoderm, a variety of transcription factors mediate neural plate patterning. Among these Bf1, Bf2, Rx, Six3 and Otx2, expressed in the anterior neural plate, are crucially involved in the formation of anterior CNS structures (Acampora et al., 1995; Andreazzoli et al., 1997; Mathers et al., 1997; Bourguignon et al., 1998; Mariani and Harland, 1998; Andreazzoli et al., 1999; Loosli et al., 2001; Carl et al., 2002; Lagutin et al., 2003). However, although there is some evidence that links the activity of these transcription factors with those of the Wnt and BMP pathways in the formation of the CNS (Braun et al., 2003; Lagutin et al., 2003), their exact interplay is still scarcely understood.
In this study, we show that Six3 displays the characteristics expected from an effector of neural inducers involved in specifying and maintaining anterior neural plate properties. Xsix3 overexpression promotes cell proliferation and inhibits neurogenesis at early neurula stage by activating Xhairy2, Zic2, Xrx1 and Xbf1 and regulating the expression of p27Xic1 and cyclinD1. Furthermore, Six3 represses BMP expression in both Xenopus and zebrafish, and is able to rescue the anterior neural plate defects of chordino mutants. The effect of Xsix3 on Bmp4 appears to be direct, as suppression occurs even in the absence of protein synthesis and Xsix3 can bind the Bmp4 promoter in vitro. Although Xsix3 efficiently suppresses Bmp4 expression, we observed that it is unable to induce neural tissue, requiring Xotx2 for this activity. Taken together with the recent observation that Six3 is able to repress Wnt expression (Braun et al., 2003; Lagutin et al., 2003), these data indicate Six3 as a crucial factor for anterior neural plate specification.
Materials and methods
Animals
To obtain embryos, induction of ovulation of pigmented Xenopus females, in vitro fertilisation and embryo culture were carried out as described by Newport and Kirschner (Newport and Kirschner, 1982). Embryos were staged according to Nieuwkoop and Faber (Nieuwkoop and Faber, 1967). Zebrafish embryos were obtained by natural spawning from wild-type and dinott250 mutant fish (Schulte-Merker et al., 1997).
In situ hybridization
Whole-mount in situ hybridization experiments on Xenopus and zebrafish embryos, and on animal caps, were carried out essentially as described previously (Harland, 1991; Barth and Wilson, 1995). Bleaching of Xenopus pigmented embryos was carried out following colour reaction as described by Mayor et al. (Mayor et al., 1995). The templates for the production of in situ hybridization probes for Xag (Bradley et al., 1996), Xcg (Sive et al., 1989), Xsix3 (Zuber et al., 2003), Xrx1 (Casarosa et al., 1997), rx3 (Chuang et al., 1999), Zic2 (Brewster et al., 1998), N-tubulin (Hartenstein et al., 1989), elrC (Perron et al., 1999), Xash3 (Turner and Weintraub, 1994), Xbf1 (Bourgouignon et al., 1998), Xdelta1 and Xngnr1 (Ma et al., 1996), Xnotch (Chitnis and Kintner, 1996), Xsix1 (Pandur and Moody, 2000), p27Xic1 (Hardcastle and Papalopulu, 2000), Xhairy2 (Koyano-Nakagawa et al., 2000), Sox9 (Spokony et al., 2002), Xslug (Mayor et al., 1995), Xnrp1 (Knecht et al., 1995), Bmp4 (Fainsod et al., 1994), bmp4 (Nikaido et al., 1997), dlx3 (Akimenko et al., 1994), Sox2 (Misuzeki et al., 1998), sox3 (Kudoh et al., 2001), Xk81 (Jonas et al., 1985), Xwnt8 (Christian et al., 1991), Xotx2 (Pannese et al., 1995), otx2 (Li et al., 1994), hoxb1b (Eisen and Weston, 1993) and cyclinD1 (Vernon et al., 2003) have been described previously.
Mitotic inhibition with hydroxyurea and aphidicolin treatment
Xenopus embryos were devitellinized at stage 9 and allowed to recover for 30 minutes in 2% Ficoll in 0.1×Marc's modified Ringer (MMR). Stage 10 embryos were then incubated in 0.1×MMR containing 20 mM hydroxyurea and 150 μM aphidicolin (HUA) (Harris and Hartenstein, 1991; Hardcastle and Papalopulu, 2000) until fixation. The effect of hydroxyurea and aphidicolin treatment on cell division was examined as described previously (Andreazzoli et al., 2003).
Embryo microinjections and BrdU incorporation
Capped VP16-Six3 RNA was generated from a construct containing the full-length Xsix3 cDNA cloned into Cla1/Xhp1 sites of CS2-VP16 vectors (Kessler, 1997). To make the GR-Xsix3 expression construct, the open reading frame of Xsix3 was PCR amplified using the primers 5′-GCAGATATCATGGTGTTCAGGTCCCCTC-3′ and 5′-GTCCTCGAGTCATACGTCACATTCAGAGTCAC-3′ and inserted into the EcoRV/Xho1 site of pCS2+/GR kindly provided by Thomas Sargent (see Kodjabachian and Lemaire, 2001). RNAs encoding for Xsix3 (20, 40 pg), GR-Xsix3 (65 pg), Xchh (1 ng) (Ekker et al., 1995), Xngnr1 (40 pg) (Ma et al., 1996), tBR (250 pg) and Bmp4 (300 pg) (Kazanskaya et al., 2000) were generated by in vitro transcription using the message machine kit (Ambion, Austin, TX) and co-injected with lacZ RNA into one blastomere at the two-cell stage (Xchh and Xngnr1), or into a dorsal animal blastomere at the four- to eight-cell stage (Xsix3, tBR, Bmp4). β-Galactosidase staining was performed on embryos injected with 200 pg of lacZ RNA as previously described (Turner and Weintraub, 1994); X-Gal and salmon gal substrates were used for blue and red staining, respectively (Roche, Biosinth-AG). The optimal concentration of each batch of RNA was identified through the injection of various doses followed by analysis of either the phenotype or the expression of specific markers. Three different Xsix3 antisense morpholinos were used (Gene Tools): MoXsix3 targeted against the two pseudoallelic genes of Xsix3, Xsix3.1 and Xsix3.2 (5′-ACCTGAACACCATGGGATGGCCGG-3′) (Ghanbari et al., 2001); MoXsix3.2 targeted against Xsix3.2, the gene that we used for overexpression experiments (5′-CAGCAAAACTAGCGACAGCGACAGC-3′); and MoSix3.1 targeted against Six3.1 (5′-TGAAAGAAGCGGCAGCAACACTAGC-3′). Differences in the efficiency of morpholinos were observed; whereas injection of 0.2 mM of MoSix3.1 at the two-cell stage does not induce abnormal development, injection of 0.2 mM of both MoXsix3 and MoXsix3.2 leads to a Xsix3 loss-of-function phenotype. Given that the frequency of affected embryos in the case of MoXsix3 (95%) injection was higher than with MoXsix3.2 (45%), we decided to use MoXsix3 for our studies.
Morpholino oligonucleotides targeted against zebrafish chordin, reliably phenocopying the mutant chordino phenotype, were injected into one-cell stage embryos at concentrations of 0.15 mM (Nasevicius and Ekker, 2000). In vitro transcription of zebrafish six3 RNA and injection of 19-74 pg was performed as described (Kobayashi et al., 1998).
S-phase cells were labelled with BrdU essentially according to the protocol of Hardcastle and Papalopulu (Hardcastle and Papalopulu, 2000). To distinguish the injected from the control side of the embryos, we combined immunohistochemistry with in situ hybridization using Zic2 expression, a gene strongly upregulated upon Xsix3 overexpression (see below). In these experiments the number of BrdU-positive cells in the injected side was compared with that of the uninjected control side, taking into account also their anteroposterior distribution. Whole-mount TUNEL staining was performed at stage 13, as described by Hensey and Gautier (Hensey and Gautier, 1998).
Animal cap assay and cycloheximide treatment
For Xenopus animal cap experiments, capped synthetic chordin (150 pg per blastomere) (Sasai et al., 1995), tBR (600 pg) (Wylie et al., 1996), Bmp4 (1 ng), Xotx2 (250 pg) (Vignali et al., 2000), Xotx2-GR (50 pg) (Gammill and Sive, 1997), Xsix3 (500 pg-2 ng) and GR-Xsix3 (500 pg-1ng) RNA was injected into one-cell stage embryos and animal caps dissected at stage 9. When sibling control embryos reached stage 14, animal caps were fixed and stored in ethanol at −20°C. To inhibit protein synthesis, animal caps were isolated from GR-Xsix3 or Xotx2-GR injected embryos at stage 9 and aged in high-salt Modified Barth's Saline (MBS). Stage 10.5 caps where then incubated in high-salt containing 10 μM of cycloheximide (CHX). Glucocorticoid receptor (GR) fusion proteins were activated at stage 11, after a 30-minute CHX pre-incubation step, by adding DEX 10 μM into the medium containing CHX. Animal caps were then fixed at stage 15.
Gel mobility-shift assay
GST-Xsix3 protein purification was performed as described (Tessmar et al., 2002). A 315-bp PCR fragment of Bmp4 promoter was 32P-end-labelled by T4 kinase (Roche). Binding conditions and electrophoresis were as described by Gomez-Skarmeta et al. (Gomez-Skarmeta et al., 2001).
Results
Xsix3 promotes cell proliferation in the neural plate
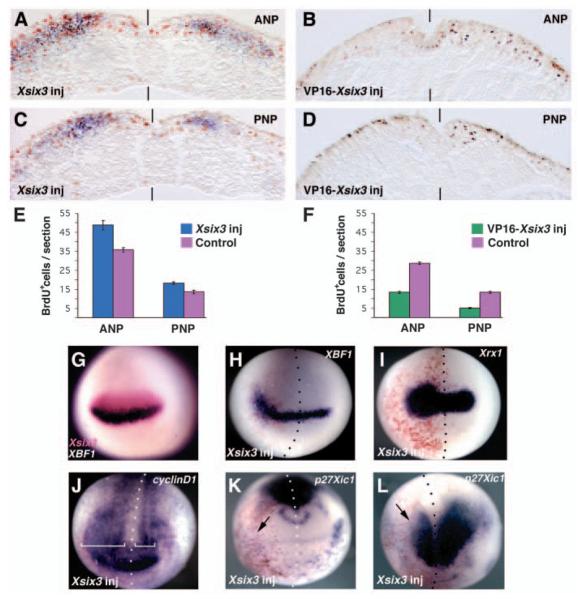
Overexpression of Six3 induces forebrain enlargement in zebrafish (Kobayashi et al., 1998), and the expansion and ectopic appearance of retina primordia in Xenopus (Bernier et al., 2000) and medaka (Loosli et al., 1999). Additionally, Six3 influences cell proliferation (Loosli et al., 1999; Bernier et al., 2000; Carl et al., 2002), possibly at a non-transcriptional level (Del Bene et al., 2004). To clarify how Six3 influences these events, we analysed the consequences of expressing exogenous Xsix3 on early neural plate development. To gain further insight into the role of Xsix3 in the control of cell proliferation, embryos overexpressing Xsix3 were analysed for BrdU incorporation. We found that the anterior neural plate of Xsix3-injected embryos displayed a 36% increase in the number of BrdU-positive cells on the injected side compared with the control side (an average of 48.7 positive cells per section in the injected side, n=755 BrdU-positive nuclei, versus 35.7 cells per section in the control side, n=460; P<0.001; Fig. 1A,E). Although at this stage Xsix3 is expressed exclusively in the anterior neural plate, the posterior neural plate displays a similar significant increase in the rate of BrdU incorporation (34.7%; an average of 18.2 positive cells per section in the injected side, n=473 versus 13.5 cells per section in the control side, n=315; P<0.001; Fig. 1C,E). Thus, the entire neural plate is competent to respond to Xsix3 proliferative activity.
Fig. 1.
Xsix3 promotes cell proliferation in the neural plate. (A-D) Transverse sections of stage 13 embryos injected with either Xsix3 (A,C) or VP16-Xsix3 (B,D), processed simultaneously for BrdU incorporation (brown nuclear staining), and Zic2 (A,C, blue staining) or lacZ (B,D, red staining) expression. Sections are at the level of the anterior (A,B) or posterior neural plate (C,D). ANP, anterior neural plate; PNP, posterior neural plate. (E,F) The average number of BrdU-positive cells per section in either the control side (magenta) or the side injected with Xsix3 (blue) or VP16-Xsix3 (green). Error bars indicate s.e.m. (G) Double whole-mount in situ hybridization of a stage 13 embryo shows that the Xsix3 (magenta) and Xbf1 (purple) expression domains overlap. (H-J) Stage 13 (H,I) and stage 14 (J) embryos injected with Xsix3 display expansion of the Xbf1 (H), Xrx1 (I) and cyclinD1 (J) expression domains. White brackets indicate the anterior expression domain of cyclinD1 in the control (right) and injected (left) side. (K,L) Xsix3 overexpression represses p27Xic1 both in the anterior, trigeminal ganglion (K, arrow) and in its posterior expression domain (L, arrow) in stage 13 embryos. (G-K) Frontal views, dorsal towards the top; (L) dorsal view, anterior towards the bottom. Red staining represents expression of a co-injected lacZ lineage tracer. The injected side of the embryos (to the left of vertical bars or dotted lines representing the midline) is indicated (inj).
To analyse the requirement for Xsix3 function in the control of cell proliferation, we used a loss-of-function approach. In agreement with the work of Kobayashi and colleagues (Kobayashi et al., 2001), we found that a transcriptional activator form of Xsix3 (VP16-Xsix3) acts as a dominant negative. Very low amounts of VP16-Xsix3 RNA (20-40 pg) injected at the four-cell stage dramatically reduced, and sometimes eliminated, eye formation (94%, n=500; data not shown). This phenotype is efficiently rescued by co-injection of Xsix3 RNA, indicating that VP16-Xsix3 specifically antagonises Xsix3 function (90%, n=150; data not shown). Expression analysis of molecular markers (Xbf1, Xrx1, Xpax6, Xotx2, Xen2 and Xkrox20) recapitulated the anterior forebrain truncation phenotypes previously described for Six3 loss of function in medaka and mouse, or inactivation by dominant-negative variants in zebrafish (data not shown) (Kobayashi et al., 2001; Carl et al., 2002; Lagutin et al., 2003). Moreover, both the anterior and posterior neural plate (ANP and PNP) of VP16-Xsix3 injected embryos shows a significant decrease of BrdU-positive cells in the injected side compared with the control side (53% and 60% respectively; ANP: an average of 28.5 cells per section in the control side, n=820, versus 13 cells per section in the injected side, n=380; PNP: an average of 13 cells per section in the control side, n=383, versus 5.6 cells per section in the injected side, n=129; P<0.001; Fig. 1B,D,F).
To gain insight into how Six3 may act on proliferation in the anterior neural plate, we analyzed the expression of putative Six3 target genes known to be involved in cell proliferation control that are expressed rostrally at early neurula stage. We found that Xbf1 and Xrx1 are both ectopically activated in the anterior neural plate (Xbf1: 86%, n=45; Xrx1: 72%, n=90; Fig. 1H,I) by exogenous Xsix3. CyclinD1, a positive regulator of the cell cycle expressed in the eye field, is ectopically activated by Xsix3 (stage 14, 82%, n=34; Fig. 1J), whereas p27Xic1, which encodes a cell cycle inhibitor, is repressed by Xsix3 (100%, n=39; Fig. 1K,L). CyclinD1 and p27Xic1 expression is modulated both in the anterior and posterior neural plate (Fig. 1J-L). Thus, Xrx1, Xbf1 and cyclinD1, genes encoding factors promoting cell proliferation, are activated, and p27Xic1, which inhibits cell proliferation, is repressed by Xsix3 at early neurula stage; this strongly suggests that Xsix3 is able to act on the cell cycle machinery already at this early developmental stage.
Ectopic expression of Xsix3 enlarges the neural plate at the expense of adjacent non-neural tissue
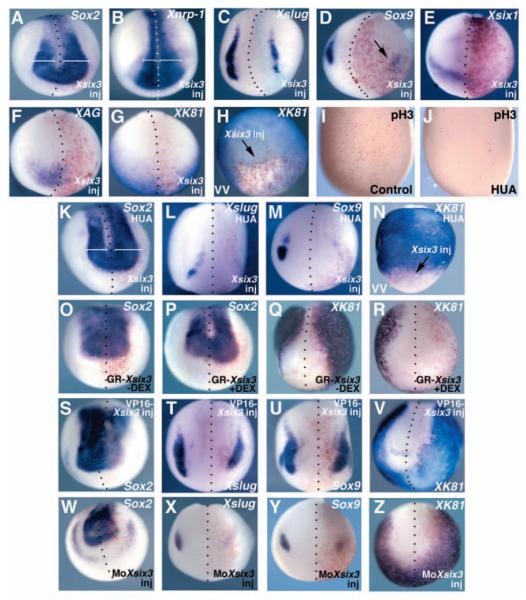
Xsix3 is expressed in the anterior neural plate at early neurula stage, raising the possibility that, besides controlling cell proliferation, it might participate in neuroectoderm specification (Zuber et al., 2003). To test this hypothesis, we analysed the expression of neural plate markers in Xsix3-injected embryos at early neurula stage. We observed that the general neural markers Sox2 and Xnrp1 (Knecht et al., 1995; Kishi et al., 2000) are expanded on the injected side of the embryos (Sox2: 77%, n=135; Xnrp1: 80%, n=23; Fig. 2A,B). Notably, ectopic expression of neural markers in the lateral ectoderm was always contiguous to the neural plate.
Fig. 2.
Alterations of the size of the neural plate in Xsix3 gain- and loss-of-function experiments. (A-H,K-Z) Embryos injected unilaterally with Xsix3 (A-H,K-N), GR-Xsix3 (O-R), VP16-Xsix3 (S-V) or MoXsix3 (W-Z), and analysed at early neurula stage. The probes used are indicated in each panel. Red staining in A,B,D-H,K-Z and turquoise staining in C represent expression of the co-injected lacZ lineage tracer. The injected side of the embryos (to the right of dotted lines representing the midline) is indicated (inj). (A-G,K-M,O-Z) Frontal views, dorsal towards the top; (H,N) ventral views (VV), anterior towards the bottom. (H,N) Arrows indicate Xk81 repression caused by Xsix3 overexpression. (I,J) Embryos untreated (I) or treated (J) with HUA, brown nuclear staining representing anti-phosphorylated histone H3 staining; dorsal views, anterior towards the bottom. (K-N) Embryos injected with Xsix3 and treated with HUA. (A,B,K) White brackets indicate the anterior expression of Sox2 (A,K) or Xnrp1 (B) in Xsix3-injected (right) and control (left) sides.
To examine whether the expression of non-neural markers at the border of the neural plate was also affected, we analysed the expression of the neural crest markers Xslug and Sox9 (Sasai et al., 2001), the cranial placode marker Xsix1 surrounding the anterolateral neural plate (Pandur and Moody, 2000), and the cement-gland specific marker Xag (Sive and Bradle, 1996), which is expressed in the most anterior part of the non-neural ectoderm in a region partially overlapping with Xsix3 (data not shown). Xsix3 injection resulted in the reduction or loss of the expression of all of these markers (Xslug: 96%, n=46; Sox9: 93%, n=29; Xsix1: 93%, n=40; Xag: 82%, n=29; Fig. 2C-F). Moreover, when present, the expression domains of the markers were displaced to a more ventrolateral position (Fig. 2C,D and data not shown). These changes were probably due to the enlargement of the neural plate. This suggests that the expansion of the neural plate occurs, at least in part, by converting non-neural cells at the neural plate border to a neural fate. Complementing the ability of Xsix3 to promote neural fate, expression of the epidermal marker Xk81 (Jonas et al., 1989) was also severely blocked in the Xsix3 injected area (80%, n=68; Fig. 2G,H). In contrast to what was observed for the neural markers, Xsix3-dependent suppression of Xk81 occurred also in isolated areas of ventral ectoderm non contiguous with the neural plate (Fig. 2H). These results indicate that Xsix3 is sufficient to repress epidermal fate but requires a competent territory in order to promote a neural fate.
To test whether the ability of Xsix3 to expand the anterior neural plate at the expense of non-neural tissue might be a consequence of its proliferative activity, we overexpressed Xsix3 in embryos in which cell division was blocked by HUA treatment. HUA treatment severely reduced anti-phosphorylated histone H3 (H3P) staining, a marker of cells in mitosis (Fig. 2I,J). Even under these conditions, Xsix3 is still able to expand Sox2 expression and to repress the expression of Xslug, Sox9 and Xk81 (Sox2: 69%, n=87; Xslug: 73%, n=29; Sox9: 90%, n=19; Xk81: 76%, n=34; Fig. 2K-N). Taken together, these results suggest that the effects of Xsix3 on markers of neural patterning are at least in part independent of proliferation.
To rule out the possibility that the observed effects on neural plate specification might be a secondary consequence of an early repression of Wnt gene expression by Xsix3 (Lagutin et al., 2003), we injected a Dexamethasone (DEX)-inducible form of the Xsix3 construct. In these experimental conditions, activation of GR-Xsix3 at mid-gastrula stages is able to promote Sox2 and to repress Xk81 expression (Sox2: 100%, n=91; Xk81: 90%, n=90; Fig. 2P,R). In the absence of DEX, the fusion protein GR-Xsix3 is inactive or only slightly active, and embryos do not display significant Sox2 and Xk81 expression alterations (Sox2: 89% normal expression, 11% slightly expanded, n=80; Xk81: 85% normal expression, 15% slightly reduced, n=59; Fig. 2O,Q). These results suggest that Xsix3 has a role in the initial specification of the neural plate.
To further analyse the requirement for Xsix3 function in anterior neural plate specification, we used two different loss-of-function strategies. In VP16-Xsix3-injected embryos, we observed repression of Sox2 and expansion of Xk81 in the prospective anterior neural plate (Sox2: 72%, n=42; Xk81: 81%, n=32; Fig. 2S,V). The neural crest markers analysed are still expressed, although they are localized closer to the midline (Xslug: 77%, n=27; Sox9: 83%, n=31; Fig. 2T,U). Thus, conversion of presumptive neural plate cells towards an epidermal fate is likely to contribute to the Six3 loss-of-function phenotype.
As an independent loss-of-function approach, we injected an antisense morpholino (MoXsix3). Virtually all the MoXsix3-injected embryos show eye and anterior head defects similar to those observed in VP16-Xsix3-injection experiments (95%, n=130, data not shown). However, the penetrance of MoXsix3 phenotypic alterations is milder than that observed with VP16-Xsix3, as we never observed a complete loss of eye structures in MoXsix3-injected embryos. The phenotype is efficiently rescued by co-injection of Xsix3 RNA (data not shown and Fig. 5Q). As for VP16-Xsix3, overexpression of MoXsix3 reduces Sox2 and expanded Xk81 expression at early neurula stages (Sox2: 100%, n=25; Xk81: 80%; n=75; Fig. 2W,Z). Similarly, the expression of the neural crest markers Sox9 and Xslug is delocalised in a more dorsal position, even though in this case a reduced expression is also observed (Xslug: 100, n=35; Sox9: 100%, n=28; Fig. 2X,Y). Altogether, these results indicate that Xsix3 acts as a transcriptional repressor in the control of anterior neural plate specification.
Fig. 5.
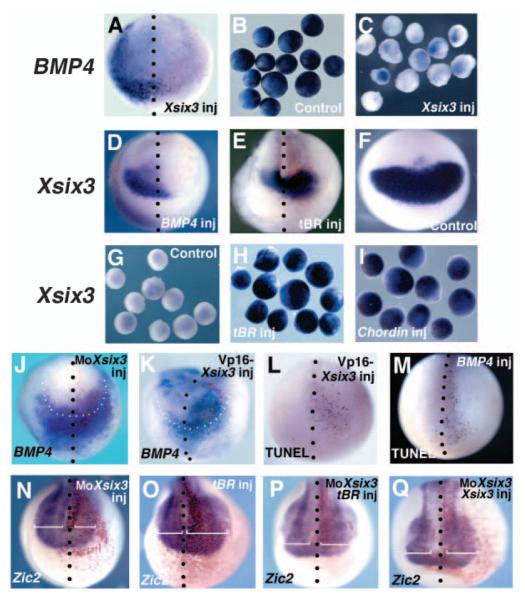
Xsix3 and Bmp4 repress each other. (A-Q) Control and injected embryos, and animal caps, were analysed at stage 13 for the expression of the genes indicated. (A-C) Xsix3 overexpression represses Bmp4 expression in embryos (A) and animal caps (C), whereas uninjected caps (B) strongly express Bmp4. (D) Xsix3 is repressed in embryos injected with Bmp4. (E) tBR injection expands the Xsix3 expression domain. (G-I) Animal caps injected with tBR (H) or Chordin (I) strongly express Xsix3, whereas uninjected caps (G) do not express Xsix3. (J,K) MoXsix3- and VP16-Xsix3-injected embryos display anterior expansion of Bmp4 (the white dots indicate the anterior neural plate border). (L,M) VP16-Xsix3 and Bmp4 injection induces apoptosis (TUNEL staining) in the anterior neural plate. (N,O) MoXsix3 reduces (N) and tBR expands (O) the domain of Zic2 expression. (P,Q) Zic2 repression is rescued in embryos co-injected either with MoXsix3 and tBR (P, compare with N), or with MoXix3 and Xsix3 (Q, compare with N).
Xsix3 suppresses differentiation of primary neurons
In Xenopus, a subset of neuroectodermal cells leave the cell cycle and start to differentiate at the end of gastrulation, giving rise to primary neurons (Hartenstein, 1989). As Xsix3 promotes neural plate expansion, we tested its involvement in the specification of primary neurons.
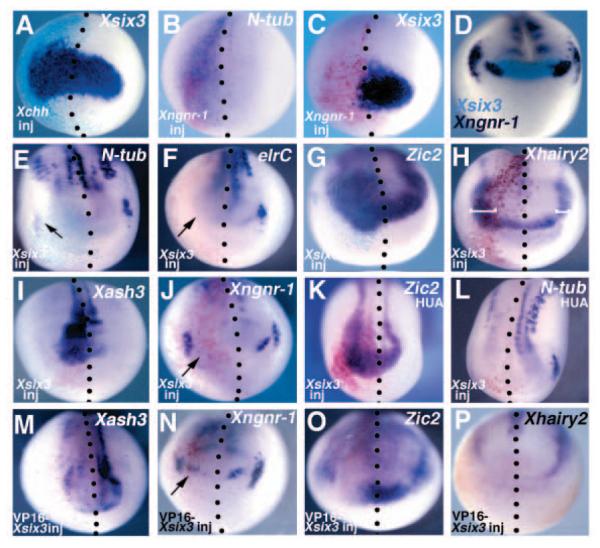
First, we investigated the effects of positive and negative regulators of cell differentiation on Xsix3 expression. Cephalic hedgehog (chh) induces proliferation and delays differentiation in Xenopus anterior neural plate (Franco et al., 1999). Injection of Xchh strongly expands the Xsix3 expression domain at the early neurula stage (75%, n=45; Fig. 3A). Conversely, Xngnr1, which encodes a factor promoting neurogenesis in the anterior neural plate (Ma et al., 1996) – as shown by its ability to induce ectopic N-tubulin expression (100%, n=29; Fig. 3B) – strongly repressed Xsix3 (100%, n=24; Fig. 3C).
Fig. 3.
Xsix3 regulates the expression of genes that control cell differentiation, and its expression is controlled by Hedgehog and Neurogenin signalling. (A-C) Embryos injected unilaterally with Xchh (A) and Xngnr1 (B,C) display ectopic expression of Xsix3 (A) and N-tubulin (B), and repression of Xsix3 (C). (D) Comparison of the expression of Xsix3 (light blue) with that of Xngnr1 (purple) in a stage 13 embryo. (E-K) Embryos injected with Xsix3 display repression of N-tubulin (E, the arrow indicates the repressed expression in the trigeminal ganglion), elrC (F) and Xngnr1 (J), and ectopic expression of Zic2 (G), Hairy2 (H) and Xash3 (I). (K,L) Embryos injected with Xsix3 and treated with HUA show expansion of Zic2 (K) and repression of N-tubulin (L). (M,O,P) Embryos injected with VP16-Xsix3 display reduction of the Xash3 (M), Zic2 (O) and Xhairy2 (P) expression domains. (J,N) Arrows indicate the anterior boundaries of Xngnr1 in the injected side of the embryo. (A-P) Frontal views, dorsal towards the top. Red staining in B,C,F-P and turquoise staining in A,E represent expression of the co-injected lacZ lineage tracer. The injected side of the embryos (to the left of dotted lines representing the midline) is indicated (inj). (H) White brackets indicate the anterior expression of Xhairy2 in Xsix3-injected (left) and control (right) sides.
To test the effects of Xsix3 on neurogenesis, we analyzed the expression of Xngnr1 and the neuronal differentiation markers N-tubulin and elrC in Xsix3-injected embryos. Xsix3 overexpression represses Xngnr1 in the most anterior semicircular stripes of expression, corresponding to the presumptive olfactory placodes and part of the telencephalon (38%, n=53, Fig. 3J, arrow), and in the midbrain-hindbrain boundary (100%, n=53, Fig. 3J). At early neurula stage, Ntubulin and elrC are expressed in primary neurons organized into three longitudinal domains on each side of the posterior neural plate, which will give rise to motoneurons, interneurons and sensory neurons, and in the trigeminal ganglia (Chitnis et al., 1999; Perron et al., 1999). Xsix3 injection causes a downregulation of N-tubulin (90%, n=50; Fig. 3E) and elrC (91%, n=23; Fig. 3F), both in the trigeminal ganglion (Fig. 3E,F, arrows) and in the posterior stripes. To understand which step of the neurogenesis cascade can be affected by Xsix3, we examined the effects of Xsix3 injection on the antineurogenic genes Zic2 (Brewster et al., 1998), Xhairy2 (Dawson et al., 1995) and Xash3 (Chitnis and Kintner, 1996). Xsix3-injected embryos showed an expansion of Zic2 (83%, n=108; Fig. 3G), Xhairy2 (82%, n=43; Fig. 3H) and Xash3 (69%, n=26; Fig. 3I) along the anterior lateral neural plate. These results suggest that Xsix3 reduces primary neuron formation by upregulating the prepattern antineurogenic genes Zic2 and Xhairy2. The fact that Xsix3 overexpression results in the repression of N-tubulin, and the expansion of the Zic2 and Xhairy2 expression domains, even in embryos in which cell division was blocked (Zic2: 78%, n=75; N-tubulin: 80%, n=20; Fig. K,L and data not shown) suggests that the effect of Xsix3 on neuronal differentiation is not dependent on cell proliferation. These data indicate that Xsix3 inhibits neuronal differentiation through the activation of the antineurogenic factors Zic2, Xhairy2 and Xash3, and/or through the repression of neurogenic factors such as Xngnr1.
To determine the requirement for Xsix3 function in the control of neurogenesis, we investigated the expression of the same markers in VP16-Xsix3-injected embryos. In the case of Xash3, Zic2 and Xhairy2, we observed a repression of their expression domains (100%; Xash3: n=25; Zic2: n=69; Xhairy2: n=35; Fig. 3M,O,P). Despite this, Xngnr1 expression in VP16-Xsix3-injected embryos was not significantly increased (Fig. 3N). These results suggest that the effects observed in Xsix3 loss of function are mainly independent of precocious neurogenesis.
Xsix3 inhibits epidermal fate but requires Xotx2 to induce neural markers in animal caps
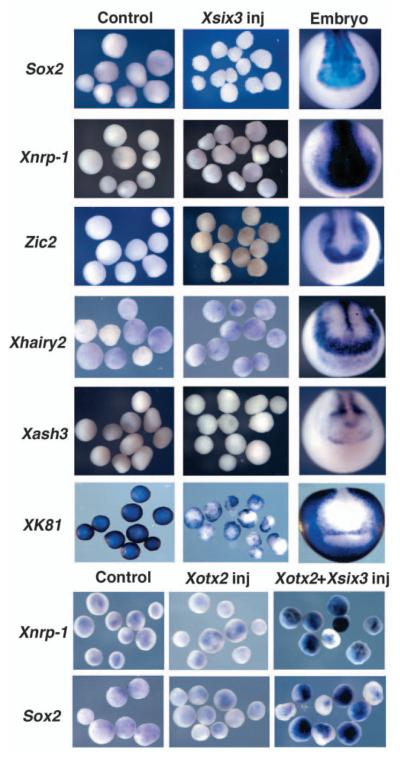
As Xsix3 can expand the neural plate, we tested whether Xsix3 is able to induce neural tissue independently of mesoderm by performing animal cap assays. Control uninjected caps showed no expression of Sox2, Xnrp1, Xash3, Xhairy2 and Zic2 (0% in all caps; Sox2: n=79; Xnrp1: n=25; Xash3: n=27; Zic2: n=55; Xhairy2: n=60; Fig. 4), whereas they did express Xk81 (100%, n=61; Fig. 4). None of these markers was induced in Xsix3-injected caps (Sox2: n=129; Xnrp1: n=30; Xash3: n=35; Zic2: n=90; Xhairy2: n=90; Fig. 4). By contrast, Xsix3 is able to strongly suppress expression of the epidermal marker Xk81 (83%, n=78; Fig. 4). These results indicate that Xsix3 negatively regulates the expression of the epidermal marker Xk81 and that it requires additional factors, absent in animal caps, to activate neural markers in the ectoderm. Among these, Xotx2, encoding a factor crucial for anterior neural plate formation, is co-expressed with Xsix3 in the presumptive anterior neuroectoderm at early neurula stage (stage 12.5) (Zuber et al., 2003). Similar to Xsix3, overexpression of Xotx2 in animal caps strongly suppresses the expression of the epidermal marker Xk81, but it does not activate (or only weakly activates) expression of general neural markers (100%; Sox2: n=20; Xnrp1: n=35; Fig. 4) (Vignali et al., 2000). However, co-injection of Xsix3 and Xotx2 mRNA strongly activates the expression of both Sox2 and Xnrp1 (Sox2: 88%, n=60; Xnrp1: 82%, n=60; Fig. 4). These results suggest that Xotx2 and Xsix3 may function in a synergistic way in anterior neural plate specification.
Fig. 4.
Xsix3 represses epidermal fate but requires Xotx2 for the induction of neural markers in animal caps. Animal caps from control uninjected embryos (Control) or embryos injected with either Xsix3, Xotx2, or a mixture of the two RNAs, as indicated at the top, were dissected at stage 9, cultured to stage 14 and analysed for the expression of the genes indicated to the left. The column to the right (Embryo) shows expression of the analysed genes in control embryos.
Mutual antagonism between Xsix3 and Bmp4
As Xsix3 inhibits the expression of the epidermal marker Xk81, we asked whether this effect may result from a downregulation of the expression of the epidermalizing factor BMP4 (Sasai et al., 1995; Wilson and Hemmati-Brivanlou, 1995). At blastula stage, Bmp4 is ubiquitously expressed and becomes restricted to the ventrolateral regions of the embryo during gastrulation. At this stage, Bmp4 and Xsix3 display mainly complementary expression domains, with Xsix3 being activated in the anterior neural plate concomitant with repression of Bmp4 in the same domain.
Overexpression of Xsix3 resulted in repression of Bmp4 expression both in the embryo (71%, n=49; Fig. 5A) and in animal caps (71%, n=138; Fig. 5C). Control uninjected caps displayed normal Bmp4 expression (100%, n=39; Fig. 5B).
To elucidate whether Bmp4 and Xsix3 might antagonise each other, we analysed the effects that the overexpression of each of these genes exert on the other. We observed that Bmp4 overexpression leads to a strong reduction of Xsix3 expression (88%, n=26; Fig. 5D). Conversely, interfering with BMP signalling by injection of either tBR, a dominant-negative BMP receptor, or chordin mRNA (Weinstein and Hemmati-Brivanlou, 1999) induced a strong activation of Xsix3 both in animal caps (control: 0%, n=30; chordin: 100%, n=39; tBR: 100%, n=45; Fig. 5G,H,I) and in the anterior neural plate of the embryo (tBR: 92%, n=26; Fig. 5E). Conversely, both VP16-Xsix3 and MoXsix3 injection leads to expansion of Bmp4 expression in the presumptive anterior neural plate (VP16-Xsix3: 45%, n=50; MoXsix3: 77%, n=27; Fig. 5J,K). Additionally, TUNEL analysis showed that both Bmp4-and VP16-Xsix3-injected embryos displayed an anterior accumulation of apoptotic nuclei (Fig. 5L,M).
To analyse whether the effects of Xsix3 loss of function are a consequence of BMP4 expansion in the anterior neural plate, we tested whether interfering with BMP signalling can counteract the reduction of the anterior neural plate in MoXsix3-injected embryos. To achieve this, we analysed the expression of Zic2 (a gene expressed both in the anterior and posterior neural plate that is strongly modulated by Xsix3), in MoXsix3/tBR co-injected embryos. Injection of MoXsix3 alone repressed anterior Zic2 expression (81%, n=73; Fig. 5N). Conversely, MoXsix3/tBR co-injected embryos showed a complete or partial rescue of the Zic2 expression domain (65%, normal expression; 10% slightly reduced expression, 25% expanded expression; n=124; Fig. 5P). None of the co-injected embryos showed the strong expansion of Zic2 seen for tBR alone (100%, n=60; Fig. 5O). As a control, a similar rescue is observed when MoXsix3 is co-injected with Xsix3 (83%, normal expression; 17% slightly expanded; n=68; Fig. 5Q). Taken together, these results indicate a mutual antagonism between Xsix3 and Bmp4.
six3 rescues the anterior alterations in zebrafish chordino mutants
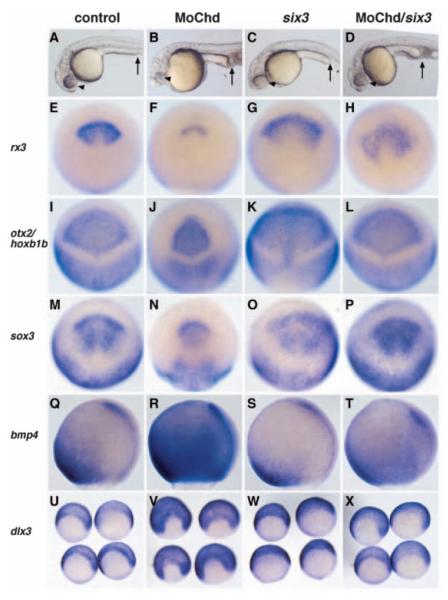
To analyse whether six3 could counteract the effects of BMP dependent modulation of the neural plate size in vivo, we took advantage of zebrafish chordino mutants/morphants. These mutants (Hammerschmidt et al., 1996) carry a mutation in the chordin gene, a known antagonist of BMP signalling. The mutation causes the enlargement of tail structures, alterations in trunk development and a reduction of anterior central nervous system structures, including the eye (Fig. 6B) (Schulte-Merker et al., 1997). Analysis of chordino mutants/morphants at early neurula stages (90% epiboly) shows a reduced anterior neural plate (rx3, otx2, sox3) and expanded epidermal (bmp4, dlx3) markers, a phenotype similar to VP16-Xsix3-injected frog embryos (Fig. 6F,J,N,R,V).
Fig. 6.
six3 rescues chordin loss of function. (A-D) Lateral view of 24 hpf zebrafish embryos. Arrowheads indicate the eye/brain size in control (A), and in embryos injected with MoChd (B), six3 (C), or with both six3 and MoChd (D). Arrows indicate the posterior region, which is expanded in embryos injected with MoChd, or co-injected with MoChd and six3. (E-X) Early neurula embryos (90% epiboly) treated as in A-D and analysed for expression of the genes indicated. (E-P,U-X) Dorsal-anterior views, vegetal towards the bottom; (Q-T) lateral views, anterior towards the top.
To investigate whether Six3 activity can compensate for the loss of Chordin function, we overexpressed six3 in wild-type and chordino mutant/morphant embryos. Injection of six3 mRNA into wild-type zebrafish embryos results in a consistent expansion of both brain (Kobayashi et al., 1998) and eye structures, as reported for Xenopus and medaka (Bernier et al., 2000; Loosli et al., 1999) (Fig. 6C, Table 1). The development of posterior structures remained unaffected (Fig. 6C). At early neurula stages, six3 misexpression resulted in the anterior lateral and ventral expansion of the neural markers rx3, otx2 and sox3, together with a mild reduction of the epidermal fate markers bmp4 and dlx3, whereas the expression of the posterior neural plate marker hoxb1b remained largely unaffected (Fig. 6G,K,O,S,W). These data suggest that six3 is able to specifically expand the anterior neural plate without affecting anteroposterior patterning.
Table 1.
Concentration-dependent rescue of loss of chordin function by Six3 overexpression
| Wild type |
Small eye |
Big eye/brain |
Anterior rescue |
Others | n | |
|---|---|---|---|---|---|---|
| MoChd (1 nl; 0.15 mM) | 5% | 85% | 0% | – | 10% | 40 |
| Six3: 19 pg | 90% | 0% | 0% | – | 10% | 49 |
| + MoChd (1 nl; 0.15 mM) | 9% | 38% | 3% | 38% | 12% | 34 |
| Six3: 37 pg | 16% | 0% | 77% | – | 7% | 57 |
| + MoChd (1 nl; 0.15 mM) | 13% | 22% | 0% | 48% | 17% | 23 |
| Six3: 74 pg | 25% | 0% | 57% | – | 18% | 67 |
| + MoChd (1 nl; 0.15 mM) | 10% | 10% | 48% | 15% | 17% | 52 |
| + MoChd (2 nl; 0.15 mM) | 2% | 7% | 54% | 18% | 19% | 107 |
| Six3: 74 pg in dino mutant | 0% | 26% | 63% | 11% | 0% | 19 |
In chordino mutants/morphants, six3 injection resulted in a highly specific rescue of anterior defects in a concentration-dependent manner, whereas the trunk maintained the ventralized morphology typical for the loss of chordin gene function (Fig. 6D, Table 1). At early neurula stages, we observed the rescue of rx3, otx2 and sox3 expression, whereas hoxb1b expression in the posterior neural plate remained largely unaffected. Moreover, six3 was able to suppress the expanded expression of bmp4 and dlx3 caused by the lack of chordin function in the anterior region of co-injected embryos (Fig. 6H,L,P,T,X). These data indicate that six3 can act independently of the BMP inhibitor Chordin, in repressing epidermal fate anteriorly, to specifically promote anterior neural plate development.
Xsix3 directly represses Bmp4 transcription
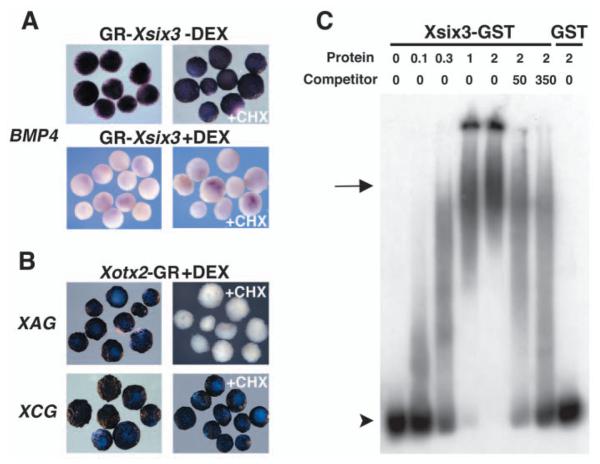
Because Xsix3 works as a transcriptional repressor, we investigated whether Bmp4 might be a direct target of Xsix3 and could therefore be repressed by GR-Xsix3 in the absence of protein synthesis. To achieve this, the protein synthesis inhibitor cycloheximide (CHX) was added to GR-Xsix3-injected animal caps before DEX activation.
Injection of GR-Xsix3 resulted in repression of Bmp4 expression in animal caps treated with DEX (88%, n=62; Fig. 7A), as well as in animal caps treated with both CHX and DEX (90%, n=120; Fig. 7A). By contrast, control caps injected with GR-Xsix3 in the absence of DEX displayed normal Bmp4 expression (100%, n=60; Fig. 7A). Similar results were observed in GR-Xsix3-injected caps following CHX but not DEX treatment, suggesting that CHX alone had no effect on Bmp4 expression (100%, n=65; Fig. 7A). As a positive control for the efficiency of CHX treatment, we injected Xotx2-GR and analysed the expression of Xcg and Xag, which are known to be direct and indirect Xotx2 targets, respectively (Gammill and Sive, 1997). As expected, although DEX treatment of injected embryos activated both Xcg and Xag, addition of DEX and CHX led to the activation of Xcg but not of Xag (Fig. 7B).
Fig. 7.
Xsix3 directly represses Bmp4 transcription. (A,B) Animal caps were analysed at stage 13 for the expression of the genes indicated to the left. (A) Bmp4 repression in GR-Xsix3-injected animal caps treated either with dexamethasone (DEX) alone, or with both DEX and cycloheximide (CHX), compare with control with no DEX treatment shown above. (B) Control of CHX treatment. Xotx2-GR-injected animal caps treated with DEX alone, or with both DEX and CHX. (C) Xsix3 binds to a Bmp4 promoter fragment containing homeodomain-binding sites. Xsix3-GST, but not GST alone, binds to the labelled Bmp4 promoter to form various complexes in a concentration-dependent manner. Addition of an excess of unlabelled Bmp4 promoter fragment (50- to 350-fold) significantly inhibits the binding. The arrow indicates the major complex formed with the highest protein concentration. The arrowhead indicates the free probe. The amount of added protein is indicated in micrograms.
To directly test the interaction of Xsix3 with the regulatory regions of the Bmp4 gene, we performed an electrophoretic mobility shift assay (EMSA; Fig. 7C). We found that the Xsix3-GST fusion protein binds to a 32P-labelled 315 bp fragment of the Bmp4 promoter, which displays putative binding sites for homeodomain-containing proteins and was previously shown to interact with Xiro1 (Gomez-Skarmeta et al., 2001). The Xsix3-GST protein binds to the Bmp4 promoter fragment in a concentration-dependent manner forming several complexes. This binding, which is not observed with the GST protein alone, is specifically competed by an excess of the unlabelled fragment (Fig. 7C). Thus both in vivo and biochemical assays suggest that Xsix3 directly represses Bmp4 transcription.
Discussion
In this study, we show that exogenous Six3 expands the anterior neural plate in zebrafish and Xenopus. In part, this is due to increased proliferation of neural progenitor cells. However, we show that Six3 can still induce neural plate expansion in the presence of cell cycle inhibitors. Accordingly, we indicate that Xsix3 acts as a transcriptional repressor in the specification of the anterior neural plate and we identify Bmp4 as a direct downstream target of Six3. Gain- and loss-of-function experiments show that Six3 represses Bmp4 expression, thus promoting neural fate at the expense of neural crest and epidermis. Altogether, these data indicate that Six3 plays a crucial role in controlling cell fate specification and cell proliferation in the anterior neural plate.
Xsix3 promotes cell proliferation and delays cell differentiation in the neural plate
The ability of Six3 to induce proliferation and inhibit neuronal differentiation may involve interactions with different cofactors in the neural plate. Despite the anterior restricted expression of Xsix3, both anterior and posterior regions of the neural plate are competent to respond to Xsix3. This suggests that cofactors required for Xsix3 proliferative activity are expressed throughout the neural plate. The recently isolated Six3 cofactors can be grouped into two categories underscoring different mechanisms of action for Six3. One group includes the groucho family, NeuroD and ATH3/ATH5, which contribute to the specificity of Six3 transcriptional activity (Kobayashi et al., 2001; Zhu et al., 2002; Tessmar et al., 2002; Lopez-Rios et al., 2003). The second category of Six3 interactors is represented by Geminin. Six3 displaces the DNA replication-inhibitor Geminin from Cdt1 resulting in activation of cell proliferation in a transcriptional-independent manner (Del Bene et al., 2004). Both types of interactors are expressed along the entire neural plate thus being potentially available for cooperation with injected Xsix3. Although the Six3/Geminin complex may contribute to the enhanced cell proliferation elicited by Six3, this protein is likely to control cell proliferation and neurogenesis by also acting as a transcription factor. Indeed, Xsix3 misexpression expands the expression of Zic2, Xhairy2 and cyclinD1, while repressing that of p27Xic1, N-tubulin and elrC. These effects are unlikely to be explained as a consequence of cell proliferation induced by Six3/Geminin interaction. Indeed, Xsix3 is able to regulate the expression of the same genes even when cell proliferation is blocked (Fig. 3K,L and data not shown). Reduction of cell proliferation in VP16-Xsix3-injected embryos provides additional evidence that Xsix3 regulates cell proliferation at the transcriptional level.
Six3 represses Bmp4 expression
A prerequisite for dorsal ectoderm to acquire neural fate is the inhibition of BMP signalling, and one of the ways in which this is accomplished is through the transcriptional inhibition of BMP genes in the nascent neural plate (reviewed by Bally-Cuif and Hammerschmidt, 2003). Prior to gastrulation, Bmp4 is expressed throughout the embryo, but begins to be cleared from the prospective neural plate soon after Xsix3 activation at gastrula stage, which would be consistent with a role for Xsix3 in repressing Bmp4 at these early stages (Fainsod et al., 1994; Hemmati-Brivanlou and Thomsen, 1995; Zuber et al., 2003). Indeed, our overexpression experiments in both embryos and animal caps provide compelling evidence for a mutual antagonism between Six3 and Bmp4. In addition, the reduction of neural plate size elicited by loss of Xsix3 function is significantly alleviated by attenuation of BMP signalling. Moreover, our study in zebrafish shows that six3 is able to compensate for the lack of the BMP antagonist chordin, to specify the size of the anterior neural plate. Because chordin is not required for six3 induction (data not shown), it is likely that these two genes act in parallel to exclude Bmp4 from the anterior neural plate.
Bmp4 transcriptional repression is not sufficient to elicit neural induction
Although Six3 is able to expand the expression domains of the neural markers Xsox2, Xnrp1, otx2 and sox3, we never observed ectopic expression of these genes in the ventral ectoderm, where Six3 represses the epidermal marker Xk81. Furthermore, we show that in animal caps Xsix3 does not activate the expression of neural markers, despite being able to repress epidermal markers. This suggests that Xsix3 activity alone is not sufficient to respecify non-neural ectoderm to a neural fate. Indeed, we found that neural markers are induced only if Xotx2 is co-injected with Xsix3, indicating that these two genes cooperate in neural fate determination. These data suggest that an additional step besides Bmp4 transcriptional inhibition might be required to drive unspecified ectodermal cells towards a neural fate. A number of genes expressed in the presumptive neuroectoderm and involved in promoting neural fate (e.g. Xanf1, Opl and Sox2) have been described (Kuo et al., 1998; Mizuseki et al., 1998; Ermakova et al., 1999). These genes share the inability to induce neural tissue in animal caps. Because for most of these genes their effect on Bmp4 expression has not been tested, at the moment it is not clear how common is the ability of repressing Bmp4 without inducing neural fate in competent ectoderm. However, it still remains to be clarified whether the ability of factors like Xiro2, Soxd and Zic3 to neuralise competent ectoderm is simply the result of the suppression of Bmp4 expression, as has been previously reported for Geminin and Xiro1 (Nakata et al., 1997; Gomez-Skarmeta et al., 1998; Gomez-Skarmeta et al., 2001; Mizuseki et al., 1998; Kroll et al., 1998). Moreover, even though Xiro1 can directly repress Bmp4, Gomez-Skarmeta and co-workers suggested that this protein may repress additional uncharacterized factors to neuralise the ectoderm (Gomez-Skarmeta et al., 2001).
Xsix3 controls anterior neural fate, at least in part, independently from Geminin
The recently demonstrated interaction between Six3 and Geminin opens the possibility that Xsix3 may play a role in neural fate determination in a Geminin-dependent way. However, Six3 and Geminin activities in the neural plate appear to be distinct. Geminin is a coiled-coil protein, with two separable functional domains, one of which neuralises ectoderm, whereas the other is involved in the inhibition of DNA replication (Kroll et al., 1998; McGarry and Kirschner, 1998). Thus, Geminin and Six3 have antagonistic functions in the control of cell proliferation; by contrast, they share the ability to repress Bmp4 and to promote neural plate expansion in Xenopus. However, unlike Xsix3, Geminin has the ability to induce neural tissue in animal caps. Moreover, differently from the anterior restricted effects of Xsix3, Geminin induces neural tissue of posterior but not anterior character in animal caps. Finally, although the DNA-binding activity of Six3 is dispensable for the interaction with Geminin, mutagenesis of Six3/Groucho binding sites, which impairs the transcriptional repressor activity of Six3, completely abolishes its in vivo function (Kobayashi et al., 2001; Zhu et al., 2002). Altogether, these data indicate that Six3 may function in different pathways either acting through the interaction with Geminin, or controlling the transcription of key regulators of proliferation and anterior neural plate specification.
A crucial role for Six3 in anterior neural plate specification
Six3 plays a dual role in the anterior neural plate controlling proliferation and neurogenesis, and protecting the anterior neuroectoderm from the ventralizing activity of BMPs. The fate of amphibian presumptive neuroectoderm is reversible during early gastrula stages and can be changed to epidermis by transplantation to the ventral side (Spemann, 1938). Although by the end of gastrulation, the neuroectoderm has little competence left to form epidermis in transplants, epidermalising factors surrounding the neural plate maintain their ability to inhibit neural plate genes (Hartley et al., 2001). Six3 is activated by the neural inducers Noggin, Chordin and β-catenin, and begins to be expressed at a high level at mid-gastrula stage, when it is likely to modulate the responsiveness of neuroectodermal cells rather than the initial fate decisions of uncommitted ectodermal cells (Bernier et al., 2000; Zuber et al., 2003; Kuroda et al., 2004) (this work). Thus, by repressing Bmp4 expression, Xsix3 might maintain the competence of neuroectodermal cells to form the anterior neural plate.
These data, together with the observation that Six3 also counteracts Wnt signalling (Lagutin et al., 2003) and promotes cell proliferation, suggests that Six3 links cell specification and proliferation to maintain and refine anterior identity.
Acknowledgments
We thank W. A. Harris, T. D. Sargent, S. Moody, I. Dawid, J. L. Gomez-Skarmeta, J. Wittbrodt and M. Perron for sharing plasmids, and T. Kudoh for chordino mutants. We are indebted to M. Fabbri, G. De Matienzo for technical assistance, and S. Di Maria and C. Wilson and her team for animal care. We gratefully acknowledge S. Banfi and his group for Xsix3 cDNA, E. Landi for help with GST protein purification and L. Pitto for instruction on EMSA. This work was supported by grants from M.I.U.R., Telethon (grant no. GGP04268) and from E.C. Quality of life and Management of Living Resources Program, Contract no. QLG3-CT-2001-01460. Work in S.W.W.'s group is additionally supported by the BBSRC and Wellcome Trust. M.C. is a EMBO Fellow and S.W.W. was a Wellcome Trust Senior Research Fellow.
References
- Acampora D, Mazan S, Lallemand Y, Avantaggiato V, Maury M, Simeone A, Brulet P. Forebrain and midbrain regions are deleted in Otx2−/− mutants due to a defective anterior neuroectoderm specification during gastrulation. Development. 1995;121:3279–3290. doi: 10.1242/dev.121.10.3279. [DOI] [PubMed] [Google Scholar]
- Akimenko MA, Ekker M, Wegner J, Lin W, Westerfield M. Combinatorial expression of three zebrafish genes related to distalless: part of a homeobox gene code for the head. J. Neurosci. 1994;14:3475–3486. doi: 10.1523/JNEUROSCI.14-06-03475.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreazzoli M, Pannese M, Boncinelli E. Activating and repressing signals in head development: the role of Xotx1 and Xotx2. Development. 1997;124:1733–1743. doi: 10.1242/dev.124.9.1733. [DOI] [PubMed] [Google Scholar]
- Andreazzoli M, Gestri G, Angeloni D, Menna E, Barsacchi G. Role of Xrx1 in Xenopus eye and anterior brain development. Development. 1999;126:2451–2460. doi: 10.1242/dev.126.11.2451. [DOI] [PubMed] [Google Scholar]
- Andreazzoli M, Gestri G, Cremisi F, Casarosa S, Dawid IB, Barsacchi G. Xrx1 controls proliferation and neurogenesis in Xenopus anterior neural plate. Development. 2003;130:5143–5154. doi: 10.1242/dev.00665. [DOI] [PubMed] [Google Scholar]
- Aybar MJ, Mayor R. Early induction of neural crest cells: lessons learned from frog, fish and chick. Curr. Opin. Genet. Dev. 2002;12:452–458. doi: 10.1016/s0959-437x(02)00325-8. [DOI] [PubMed] [Google Scholar]
- Baker JC, Beddington RS, Harland RM. Wnt signaling in Xenopus embryos inhibits bmp4 expression and activates neural development. Genes Dev. 1999;13:3149–3159. doi: 10.1101/gad.13.23.3149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bally-Cuif L, Hammerschmidt M. Induction and patterning of neuronal development, and its connection to cell cycle control. Curr. Opin. Neurobiol. 2003;13:16–25. doi: 10.1016/s0959-4388(03)00015-1. [DOI] [PubMed] [Google Scholar]
- Barth KA, Wilson SW. Expression of zebrafish nk2.2 is influenced by sonic hedgehog/vertebrate hedgehog-1 and demarcates a zone of neuronal differentiation in the embryonic forebrain. Development. 1995;121:755–768. doi: 10.1242/dev.121.6.1755. [DOI] [PubMed] [Google Scholar]
- Barth KA, Kishimoto Y, Rohr KB, Seydler C, Schulte-Merker S, Wilson SW. Bmp activity establishes a gradient of positional information throughout the entire neural plate. Development. 1999;126:4977–4987. doi: 10.1242/dev.126.22.4977. [DOI] [PubMed] [Google Scholar]
- Bernier G, Panitz F, Zhou X, Hollemann T, Gruss P, Pieler T. Expanded retina territory by midbrain transformation upon overexpression of Six6 (Optx2) in Xenopus embryos. Mech. Dev. 2000;93:59–69. doi: 10.1016/s0925-4773(00)00271-9. [DOI] [PubMed] [Google Scholar]
- Bourguignon C, Li J, Papalopulu N. XBF-1, a winged helix transcription factor with dual activity, has a role in positioning neurogenesis in Xenopus competent ectoderm. Development. 1998;125:4889–4900. doi: 10.1242/dev.125.24.4889. [DOI] [PubMed] [Google Scholar]
- Bradley L, Wainstock D, Sive H. Positive and negative signals modulate formation of the Xenopus cement gland. Development. 1996;122:2739–2750. doi: 10.1242/dev.122.9.2739. [DOI] [PubMed] [Google Scholar]
- Braun MM, Etheridge A, Bernard A, Robertson CP, Roelink H. Wnt signaling is required at distinct stages of development for the induction of the posterior forebrain. Development. 2003;130:5579–5587. doi: 10.1242/dev.00685. [DOI] [PubMed] [Google Scholar]
- Brewster R, Lee J, Ruiz i Altaba A. Gli/Zic factors pattern the neural plate by defining domains of cell differentiation. Nature. 1998;393:579–583. doi: 10.1038/31242. [DOI] [PubMed] [Google Scholar]
- Carl M, Loosli F, Wittbrodt J. Six3 inactivation reveals its essential role for the formation and patterning of the vertebrate eye. Development. 2002;129:4057–4163. doi: 10.1242/dev.129.17.4057. [DOI] [PubMed] [Google Scholar]
- Casarosa S, Andreazzoli M, Simeone A, Barsacchi G. Xrx1, a novel Xenopus homeobox gene expressed during eye and pineal gland development. Mech. Dev. 1997;61:187–198. doi: 10.1016/s0925-4773(96)00640-5. [DOI] [PubMed] [Google Scholar]
- Chitnis AB. Control of neurogenesis – lessons from frogs, fish and flies. Curr. Opin. Neurobiol. 1999;9:18–25. doi: 10.1016/s0959-4388(99)80003-8. [DOI] [PubMed] [Google Scholar]
- Chitnis A, Kintner C. Sensitivity of proneural genes to lateral inhibition affects the pattern of primary neurons in Xenopus embryos. Development. 1996;122:2295–2301. doi: 10.1242/dev.122.7.2295. [DOI] [PubMed] [Google Scholar]
- Christian JL, McMahon JA, McMahon AP, Moon RT. Xwnt-8, a Xenopus Wnt-1/int-1-related gene responsive to mesoderm-inducing growth factors, may play a role in ventral mesodermal patterning during embryogenesis. Development. 1991;111:1045–1055. doi: 10.1242/dev.111.4.1045. [DOI] [PubMed] [Google Scholar]
- Chuang JC, Mathers PH, Raymond PA. Expression of three Rx homeobox genes in embryonic and adult zebrafish. Mech. Dev. 1999;84:195–198. doi: 10.1016/s0925-4773(99)00077-5. [DOI] [PubMed] [Google Scholar]
- Dawson SR, Turner DL, Weintraub H, Parkhurst SM. Specificity for the hairy/enhancer of split basic helix-loop-helix (bHLH) proteins maps outside the bHLH domain and suggests two separable modes of transcriptional repression. Mol. Cell. Biol. 1995;15:6923–6931. doi: 10.1128/mcb.15.12.6923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Bene F, Tessmar-Raible K, Wittbrodt J. Direct interaction of geminin and Six3 in eye development. Nature. 2004;427:745–749. doi: 10.1038/nature02292. [DOI] [PubMed] [Google Scholar]
- Eisen JS, Weston JA. Development of the neural crest in the zebrafish. Dev. Biol. 1993;159:50–59. doi: 10.1006/dbio.1993.1220. [DOI] [PubMed] [Google Scholar]
- Ermakova GV, Alexandrova EM, Kazanskaya OV, Vasiliev OL, Smith MW, Zaraisky AG. The homeobox gene, Xanf-1, can control both neural differentiation and patterning in the presumptive anterior neurectoderm of the Xenopus laevis embryo. Development. 1999;126:4513–4523. doi: 10.1242/dev.126.20.4513. [DOI] [PubMed] [Google Scholar]
- Ekker SC, McGrew LL, Lai CJ, Lee JJ, von Kessler DP, Moon RT, Beachy PA. Distinct expression and shared activities of members of the hedgehog gene family of Xenopus laevis. Development. 1995;121:2337–2347. doi: 10.1242/dev.121.8.2337. [DOI] [PubMed] [Google Scholar]
- Fainsod A, Steinbeisser H, de Robertis EM. On the function of BMP-4 in patterning the marginal zone of the Xenopus embryo. EMBO J. 1994;13:5015–5125. doi: 10.1002/j.1460-2075.1994.tb06830.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco PG, Paganelli AR, Lopez SL, Carrasco AE. Functional association of retinoic acid and hedgehog signaling in Xenopus primary neurogenesis. Development. 1999;126:4257–4265. doi: 10.1242/dev.126.19.4257. [DOI] [PubMed] [Google Scholar]
- Furthauer M, van Celst J, Thisse C, Thisse B. Fgf signalling controls the dorsoventral patterning of the zebrafish embryo. Development. 2004;131:2853–2864. doi: 10.1242/dev.01156. [DOI] [PubMed] [Google Scholar]
- Gammill LS, Sive H. Identification of otx2 target genes and restrictions in ectodermal competence during Xenopus cement gland formation. Development. 1997;124:471–481. doi: 10.1242/dev.124.2.471. [DOI] [PubMed] [Google Scholar]
- Ghanbari H, Seo HC, Fjose A, Brändli AW. Molecular cloning and embryonic expression of Xenopus Six homeobox genes. Mech. Dev. 2001;101:271–277. doi: 10.1016/s0925-4773(00)00572-4. [DOI] [PubMed] [Google Scholar]
- Gomez-Skarmeta JL, Glavic A, de la Calle-Mustienes E, Modolell J, Mayor R. Xiro, a Xenopus homolog of the Drosophila Iroquois complex genes, controls development at the neural plate. EMBO J. 1998;2:181–190. doi: 10.1093/emboj/17.1.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Skarmeta J, de la Calle-Mustienes E, Modolell J. The Wnt-activated Xiro1 gene encodes a repressor that is essential for neural development and downregulates Bmp4. Development. 2001;128:551–560. doi: 10.1242/dev.128.4.551. [DOI] [PubMed] [Google Scholar]
- Hammerschmidt M, Serbedzija GN, McMahon AP. Genetic analysis of dorsoventral pattern formation in the zebrafish: requirement of a BMP-like ventralizing activity and its dorsal repressor. Genes Dev. 1996;10:2452–2461. doi: 10.1101/gad.10.19.2452. [DOI] [PubMed] [Google Scholar]
- Hardcastle Z, Papalopulu N. Distinct effects of XBF-1 in regulating the cell cycle inhibitor p27(XIC1) and imparting a neural fate. Development. 2000;127:1303–1314. doi: 10.1242/dev.127.6.1303. [DOI] [PubMed] [Google Scholar]
- Harland RM. In situ hybridization: an improved whole-mount method for Xenopus embryos. Methods Cell Biol. 1991;36:685–695. doi: 10.1016/s0091-679x(08)60307-6. [DOI] [PubMed] [Google Scholar]
- Harris WA, Hartenstein V. Neuronal determination without cell division in Xenopus embryos. Neuron. 1991;6:499–515. doi: 10.1016/0896-6273(91)90053-3. [DOI] [PubMed] [Google Scholar]
- Hartenstein V. Early neurogenesis in Xenopus: the spatio-temporal pattern of proliferation and cell lineages in the embryonic spinal cord. Neuron. 1989;3:399–411. doi: 10.1016/0896-6273(89)90200-6. [DOI] [PubMed] [Google Scholar]
- Hartley KO, Hardcastle Z, Friday RV, Amaya E, Papalopulu N. Transgenic Xenopus embryos reveal that anterior neural development requires continued suppression of BMP signaling after gastrulation. Dev. Biol. 2001;238:168–184. doi: 10.1006/dbio.2001.0398. [DOI] [PubMed] [Google Scholar]
- Heisenberg CP, Houart C, Take-Uchi M, Rauch GJ, Young N, Coutinho P, Masai I, Caneparo L, Concha ML, Geisler R, et al. A mutation in the Gsk3-binding domain of zebrafish Masterblind/Axin1 leads to a fate transformation of telencephalon and eyes to diencephalon. Genes Dev. 2001;15:1427–1434. doi: 10.1101/gad.194301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemmati-Brivanlou A, Thomsen GH. Ventral mesodermal patterning in Xenopus embryos: expression patterns and activities of BMP-2 and BMP-4. Dev. Genet. 1995;17:78–89. doi: 10.1002/dvg.1020170109. [DOI] [PubMed] [Google Scholar]
- Hensey C, Gautier J. Programmed cell death during Xenopus development: a spatio-temporal analysis. Dev. Biol. 1998;203:36–48. doi: 10.1006/dbio.1998.9028. [DOI] [PubMed] [Google Scholar]
- Houart C, Caneparo L, Heisenberg C, Barth K, Take-Uchi M, Wilson S. Establishment of the telencephalon during gastrulation by local antagonism of Wnt signaling. Neuron. 2002;35:255–265. doi: 10.1016/s0896-6273(02)00751-1. [DOI] [PubMed] [Google Scholar]
- Jonas E, Sargent TD, Dawid IB. Epidermal keratin gene expressed in embryos of Xenopus laevis. Proc. Natl. Acad. Sci. USA. 1985;82:5413–5417. doi: 10.1073/pnas.82.16.5413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonas EA, Snape AM, Sargent TD. Transcriptional regulation of a Xenopus embryonic epidermal keratin gene. Development. 1989;106:399–405. doi: 10.1242/dev.106.2.399. [DOI] [PubMed] [Google Scholar]
- Jones CM, Lyons KM, Lapan PM, Wright CV, Hogan BL. DVR-4 (bone morphogenetic protein-4) as a posterior-ventralizing factor in Xenopus mesoderm induction. Development. 1992;115:639–647. doi: 10.1242/dev.115.2.639. [DOI] [PubMed] [Google Scholar]
- Kazanskaya O, Glinka A, Niehrs C. The role of Xenopus dickkopf1 in prechordal plate specification and neural patterning. Development. 2000;122:4981–4992. doi: 10.1242/dev.127.22.4981. [DOI] [PubMed] [Google Scholar]
- Kessler DS. Siamois is required for formation of Spemann's organizer. Proc. Natl. Acad. Sci. USA. 1997;94:13017–13022. doi: 10.1073/pnas.94.24.13017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiecker C, Niehrs C. A morphogen gradient of Wnt/beta-catenin signalling regulates anteroposterior neural patterning in Xenopus. Development. 2001;128:4189–4201. doi: 10.1242/dev.128.21.4189. [DOI] [PubMed] [Google Scholar]
- Kim CH, Oda T, Itoh M, Jiang D, Artinger KB, Chandrasekharappa SC, Driever W, Chitnis AB. Repressor activity of Headless/Tcf3 is essential for vertebrate head formation. Nature. 2000;407:913–916. doi: 10.1038/35038097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishi M, Mizuseki K, Sasai N, Yamazaki H, Shiota K, Nakanishi S, Sasai Y. Requirement of Sox2-mediated signaling for differentiation of early Xenopus neuroectoderm. Development. 2000;127:791–800. doi: 10.1242/dev.127.4.791. [DOI] [PubMed] [Google Scholar]
- Knecht AK, Good PJ, Dawid IB, Harland RM. Dorsalventral patterning and differentiation of noggin-induced neural tissue in the absence of mesoderm. Development. 1995;121:1927–1935. doi: 10.1242/dev.121.6.1927. [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Toyama R, Takeda H, Dawid IB, Kawakami K. Overexpression of the forebrain-specific homeobox gene six3 induces rostral forebrain enlargement in zebrafish. Development. 1998;125:2973–2982. doi: 10.1242/dev.125.15.2973. [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Nishikawa K, Suzuki T, Yamamoto M. The homeobox protein Six3 interacts with the Groucho corepressor and acts as a transcriptional repressor in eye and forebrain formation. Dev. Biol. 2001;232:315–326. doi: 10.1006/dbio.2001.0185. [DOI] [PubMed] [Google Scholar]
- Kodjabachian L, Lemaire P. Siamois functions in the early blastula to induce Spemann's organiser. Mech. Dev. 2001;108:71–79. doi: 10.1016/s0925-4773(01)00484-1. [DOI] [PubMed] [Google Scholar]
- Koyano-Nakagawa N, Kim J, Anderson D, Kintner C. Hes6 acts in a positive feedback loop with the neurogenins to promote neuronal differentiation. Development. 2000;127:4203–4216. doi: 10.1242/dev.127.19.4203. [DOI] [PubMed] [Google Scholar]
- Kroll KL, Salic AN, Evans LM, Kirschner MW. Geminin, a neuralizing molecule that demarcates the future neural plate at the onset of gastrulation. Development. 1998;125:3247–3258. doi: 10.1242/dev.125.16.3247. [DOI] [PubMed] [Google Scholar]
- Kudoh T, Tsang M, Hukriede NA, Chen X, Dedekian M, Clarke CJ, Kiang A, Schultz S, Epstein JA, Toyama R, Dawid IB. A gene expression screen in zebrafish embryogenesis. Genome Res. 2001;11:979–987. doi: 10.1101/gr.209601. [DOI] [PubMed] [Google Scholar]
- Kudoh T, Concha ML, Houart C, Dawid IB, Wilson SW. Combinatorial Fgf and Bmp signalling patterns the gastrula ectoderm into prospective neural and epidermal domains. Development. 2004;131:3581–3592. doi: 10.1242/dev.01227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo JS, Patel M, Gamse J, Merzdorf C, Liu X, Apekin V, Sive H. Opl: a zinc finger protein that regulates neural determination and patterning in Xenopus. Development. 1998;125:2867–2882. doi: 10.1242/dev.125.15.2867. [DOI] [PubMed] [Google Scholar]
- Kuroda H, Wessely O, de Robertis EM. Neural induction in Xenopus: requirement for ectodermal and endodermal signals via Chordin, Noggin, β-Catenin and Cerberus. PLoS Biol. 2004;2:623–634. doi: 10.1371/journal.pbio.0020092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagutin OV, Zhu CC, Kobayashi D, Topczewski J, Shimamura K, Puelles L, Russell HR, McKinnon PJ, Solnica-Krezel L, Oliver G. Six3 repression of Wnt signaling in the anterior neuroectoderm is essential for vertebrate forebrain development. Genes Dev. 2003;17:368–379. doi: 10.1101/gad.1059403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Allende ML, Finkelstein R, Weinberg ES. Expression of two zebrafish orthodenticle-related genes in the embryonic brain. Mech. Dev. 1994;48:229–244. doi: 10.1016/0925-4773(94)90062-0. [DOI] [PubMed] [Google Scholar]
- Loosli F, Winkler S, Wittbrodt J. Six3 overexpression initiates the formation of ectopic retina. Genes Dev. 1999;13:649–654. doi: 10.1101/gad.13.6.649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loosli F, Winkler S, Burgtorf C, Wurmbach E, Ansorge W, Henrich T, Grabher C, Arendt D, Carl M, Krone A, et al. Medaka eyeless is the key factor linking retinal determination and eye growth. Development. 2001;128:4035–4044. doi: 10.1242/dev.128.20.4035. [DOI] [PubMed] [Google Scholar]
- Lopez-Rios J, Tessmar K, Loosli F, Wittbrodt J, Bovolenta P. Six3 and Six6 activity is modulated by members of the groucho family. Development. 2003;130:185–195. doi: 10.1242/dev.00185. [DOI] [PubMed] [Google Scholar]
- Ma Q, Kintner C, Anderson DJ. Identification of neurogenin, a vertebrate neuronal determination gene. Cell. 1996;87:43–52. doi: 10.1016/s0092-8674(00)81321-5. [DOI] [PubMed] [Google Scholar]
- Mariani FV, Harland RM. XBF-2 is a transcriptional repressor that converts ectoderm into neural tissue. Development. 1998;125:5019–5031. doi: 10.1242/dev.125.24.5019. [DOI] [PubMed] [Google Scholar]
- Mayor R, Morgan R, Sargent MG. Induction of the prospective neural crest of Xenopus. Development. 1995;121:767–777. doi: 10.1242/dev.121.3.767. [DOI] [PubMed] [Google Scholar]
- Mathers PH, Grinberg A, Mahon KA, Jamrich M. The Rx homeobox gene is essential for vertebrate eye development. Nature. 1997;387:603–607. doi: 10.1038/42475. [DOI] [PubMed] [Google Scholar]
- McGarry TJ, Kirschner MW. Geminin, an inhibitor of DNA replication, is degraded during mitosis. Cell. 1998;93:1043–1053. doi: 10.1016/s0092-8674(00)81209-x. [DOI] [PubMed] [Google Scholar]
- Mizuseki K, Kishi M, Matsui M, Nakanishi S, Sasai Y. Xenopus Zic-related-1 and Sox-2, two factors induced by chordin, have distinct activities in the initiation of neural induction. Development. 1998;125:579–587. doi: 10.1242/dev.125.4.579. [DOI] [PubMed] [Google Scholar]
- Morgan R, Sargent MG. The role in neural patterning of translation initiation factor eIF4AII; induction of neural fold genes. Development. 1997;124:2751–2760. doi: 10.1242/dev.124.14.2751. [DOI] [PubMed] [Google Scholar]
- Munoz-Sanjuan I, Brivanlou AH. Neural induction, the default model and embryonic stem cells. Nat. Rev. Neurosci. 2002;3:271–280. doi: 10.1038/nrn786. [DOI] [PubMed] [Google Scholar]
- Nasevicius A, Ekker SC. Effective targeted gene ‘knockdown’ in zebrafish. Nat. Genet. 2000;26:216–220. doi: 10.1038/79951. [DOI] [PubMed] [Google Scholar]
- Nakata K, Nagai T, Aruga J, Mikoshiba K. Xenopus Zic3, a primary regulator both in neural and neural crest development. Proc. Natl. Acad. Sci. USA. 1997;94:11980–11985. doi: 10.1073/pnas.94.22.11980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwkoop PD, Faber J. Normal table of development of Xenopus laevis (Daudin) Elsevier; North-Holland, Amsterdam: 1967. [Google Scholar]
- Newport J, Kirschner M. A major developmental transition in early Xenopus embryos: I. characterization and timing of cellular changes at the midblastula stage. Cell. 1982;30:675–686. doi: 10.1016/0092-8674(82)90272-0. [DOI] [PubMed] [Google Scholar]
- Nguyen VH, Trout J, Connors SA, Andermann P, Weinberg E, Mullins MC. Dorsal and intermediate neuronal cell types of the spinal cord are established by a BMP signaling pathway. Development. 2000;127:1209–1220. doi: 10.1242/dev.127.6.1209. [DOI] [PubMed] [Google Scholar]
- Nikaido M, Tada M, Saji T, Ueno N. Conservation of BMP signaling in zebrafish mesoderm patterning. Mech. Dev. 1997;61:75–88. doi: 10.1016/s0925-4773(96)00625-9. [DOI] [PubMed] [Google Scholar]
- Pandur PD, Moody SA. Xenopus Six1 gene is expressed in neurogenic cranial placodes and maintained in the differentiating lateral lines. Mech. Dev. 2000;96:253–257. doi: 10.1016/s0925-4773(00)00396-8. [DOI] [PubMed] [Google Scholar]
- Pannese M, Polo C, Andreazzoli M, Vignali R, Kablar B, Barsacchi G, Boncinelli E. The Xenopus homologue of Otx2 is a maternal homeobox gene that demarcates and specifies anterior body regions. Development. 1995;121:707–720. doi: 10.1242/dev.121.3.707. [DOI] [PubMed] [Google Scholar]
- Perron M, Furrer MP, Wegnez M, Theodore L. Xenopus elav-like genes are differentially expressed during neurogenesis. Mech. Dev. 1999;84:139–142. doi: 10.1016/s0925-4773(99)00056-8. [DOI] [PubMed] [Google Scholar]
- Piccolo S, Agius E, Lu B, Goodman S, Dale L, de Robertis EM. Cleavage of Chordin by Xolloid metalloprotease suggests a role for proteolytic processing in the regulation of Spemann organizer activity. Cell. 1997;91:407–416. doi: 10.1016/s0092-8674(00)80424-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasai N, Mizuseki K, Sasai Y. Requirement of FoxD3-class signaling for neural crest determination in Xenopus. Development. 2001;128:2525–2536. doi: 10.1242/dev.128.13.2525. [DOI] [PubMed] [Google Scholar]
- Sasai Y, Lu B, Steinbeisser H, de Robertis EM. Regulation of neural induction by the Chd and Bmp-4 antagonistic patterning signals in Xenopus. Nature. 1995;377:757. doi: 10.1038/377757a0. [DOI] [PubMed] [Google Scholar]
- Schulte-Merker S, Lee KJ, McMahon AP, Hammerschmidt M. The zebrafish organizer requires chordino. Nature. 1997;387:862–863. doi: 10.1038/43092. [DOI] [PubMed] [Google Scholar]
- Sive H, Bradley L. A sticky problem: the Xenopus cement gland as a paradigm for anteroposterior patterning. Dev. Dyn. 1996;205:265–280. doi: 10.1002/(SICI)1097-0177(199603)205:3<265::AID-AJA7>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- Sive HL, Hattori K, Weintraub H. Progressive determination during formation of the anteroposterior axis in Xenopus laevis. Cell. 1989;58:171–180. doi: 10.1016/0092-8674(89)90413-3. [DOI] [PubMed] [Google Scholar]
- Spemann H. Embryonic Induction and Development. Yale University Press; New Haven, CT: 1938. [Google Scholar]
- Spokony RF, Aoki Y, Saint-Germain N, Magner-Fink E, Saint-Jeannet JP. The transcription factor Sox9 is required for cranial neural crest development in Xenopus. Development. 2002;129:421–432. doi: 10.1242/dev.129.2.421. [DOI] [PubMed] [Google Scholar]
- Stern CD. Induction and initial patterning of the nervous system–the chick embryo enters the scene. Curr. Opin. Genet. Dev. 2002;12:447–451. doi: 10.1016/s0959-437x(02)00324-6. [DOI] [PubMed] [Google Scholar]
- Tessmar K, Loosli F, Wittbrodt J. A screen for co-factors of Six3. Mech. Dev. 2002;117:103–113. doi: 10.1016/s0925-4773(02)00185-5. [DOI] [PubMed] [Google Scholar]
- Tsang M, Maegawa S, Kiang A, Habas R, Weinberg E, Dawid IB. A role for MKP3 in axial patterning of the zebrafish embryo. Development. 2004;131:2769–2779. doi: 10.1242/dev.01157. [DOI] [PubMed] [Google Scholar]
- Turner DL, Weintraub H. Expression of achaete-scute homolog 3 in Xenopus embryos converts ectodermal cells to a neural fate. Genes Dev. 1994;8:1434–1447. doi: 10.1101/gad.8.12.1434. [DOI] [PubMed] [Google Scholar]
- Vernon AE, Philpott A. The developmental expression of cell cycle regulators in Xenopus laevis. Gene Expr. Patterns. 2003;3:179–192. doi: 10.1016/s1567-133x(03)00006-1. [DOI] [PubMed] [Google Scholar]
- Vignali R, Colombetti S, Lupo G, Zhang W, Stachel S, Harland RM, Barsacchi G. Xotx5b, a new member of the Otx gene family, may be involved in anterior and eye development in Xenopus laevis. Mech. Dev. 2000;96:3–13. doi: 10.1016/s0925-4773(00)00367-1. [DOI] [PubMed] [Google Scholar]
- Weinstein DC, Hemmati-Brivanlou A. Neural induction. Annu. Rev. Cell Dev. Biol. 1999;15:411–433. doi: 10.1146/annurev.cellbio.15.1.411. [DOI] [PubMed] [Google Scholar]
- Wilson PA, Hemmati-Brivanlou A. Induction of epidermis and inhibition of neural fate by Bmp-4. Nature. 1995;376:331–333. doi: 10.1038/376331a0. [DOI] [PubMed] [Google Scholar]
- Wilson SI, Edlund T. Neural induction: toward a unifying mechanism. Nat. Neurosci. 2001;4:1161–1168. doi: 10.1038/nn747. [DOI] [PubMed] [Google Scholar]
- Wilson SW, Houart C. Early steps in the development of the forebrain. Dev. Cell. 2004;6:167–181. doi: 10.1016/s1534-5807(04)00027-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wylie C, Kofron M, Payne C, Anderson R, Hosobuchi M, Joseph E, Heasman J. Maternal beta-catenin establishes a ‘dorsal signal’ in early Xenopus embryos. Development. 1996;122:2987–2996. doi: 10.1242/dev.122.10.2987. [DOI] [PubMed] [Google Scholar]
- Zakin L, de Robertis EM. Inactivation of mouse Twisted gastrulation reveals its role in promoting Bmp4 activity during forebrain development. Development. 2004;131:413–424. doi: 10.1242/dev.00946. [DOI] [PubMed] [Google Scholar]
- Zhu CC, Dyer MA, Uchikawa M, Kondoh H, Lagutin OV, Oliver G. Six3-mediated auto repression and eye development requires its interaction with members of the Groucho-related family of corepressors. Development. 2002;129:2835–2849. doi: 10.1242/dev.129.12.2835. [DOI] [PubMed] [Google Scholar]
- Zuber ME, Gestri G, Viczian AS, Barsacchi G, Harris WA. Specification of the vertebrate eye by a network of eye field transcription factors. Development. 2003;130:5155–5167. doi: 10.1242/dev.00723. [DOI] [PubMed] [Google Scholar]