Abstract
Dexamethasone and progesterone have been found to accelerate the time of initiation and enhance the rate of myelin synthesis in Schwann cell/neuronal cocultures. The expression of mRNA for cytochrome P450scc (converts cholesterol to pregnenolone), 3β-hydroxysteroid dehydrogenase (converts pregnenolone to progesterone), and the progesterone receptor were detected and markedly induced during peak myelin formation in the cocultures. The mRNA for the glucocorticoid receptor was detected, but was found to be constituitively expressed. In addition, the specific activity of 3β-hydroxysteroid dehydrogenase was measured and found to increase by 10-fold. The mRNA for cytochrome P450scc and 3β-hydroxysteroid dehydrogenase also were found to be induced during the differentiation of O-2A precursor cells to oligodendrocytes. Fibroblast growth factor and platelet-derived growth factor were found to have proliferative effects on Schwann cells, but they had no effect on the initiation or the rate of myelin formation. These results demonstrate that myelin-forming cells have inducible enzymes responsible for steroid biosynthesis and suggest a critical role for endogenous steroid hormones in signaling the initiation and enhancing the rate of myelin formation.
The myelin sheath is a unique component of the nervous system that functions to maximize the efficiency and velocity of action potentials transmitted through nerve cells. Myelin is an extension of the plasma membrane of Schwann cells in the peripheral nervous system and oligodendrocytes in the central nervous system. In contrast to myelin formation by oligodendrocytes, Schwann cells develop a one-to-one interaction with an internode rather than forming multiple internodes (1). The neuronal participation in signaling myelination has been examined using Schwann cell/neuronal cocultures, and axonal contact has been found to be essential for peripheral nervous system myelination (2). Schwann cells multiply rapidly when in contact with axons and elongate to either ensheath or myelinate axons of approximately 1 μm in diameter or larger. Contrary to the case for Schwann cells, the presence of growth factors and chemically defined media has been found to be sufficient in producing myelin-like sheets in cultured oligodendrocytes, independent of neurons (3–6). The process of myelination has been extensively studied and a number of factors are known to influence the overall process (7); however, the factors that specifically signal the initiation, regulate the rate of formation, and regulate the extent of myelin formation are still unknown.
Light microscopy and electron microscopy have provided valuable information concerning the amount of myelin deposited and the number of wraps of compact myelin during development (8, 9). Antibodies have also been used to chart the appearance of various myelin-specific lipids and proteins during the formation of myelin (10, 11). A method has been developed that allows the continuous monitoring of a single internode throughout the myelination process. Using the fluorescent ceramide analogue, N-[5-(5,7-dimethyl BODIPY)-1-pentanoyl]d-erythrosphingosine (C5-DMB-ceramide), cocultures of dorsal root ganglia neurons and Schwann cells were observed to metabolize the fluorescent ceramide and incorporate it into myelin lipids (12). The rate of incorporation increased substantially when lipid synthesis was induced during active myelin formation and, consequently, this is a measure of the actual rate of myelin formation. In the present study, a fluorescently labeled free fatty acid (C5-DMB-hexadecanoic acid) was used and biosynthetically incorporated into lipids of the myelin sheath. Although the rates obtained with the C5-DMB-ceramide for premyelinating, active myelinating, and fully myelinated internodes were proportional to rates using the C5-DMB-hexadecanoic acid, the changes in fluorescence intensities were greater when using the free fatty acid. This allowed for a more sensitive discrimination of factors that may effect the rates. The rate of fluorescent lipid incorporation was measured for individual internodes during the myelination process using fluorescence digital imaging microscopy. Although previous research concerning the regulation of differentiation and myelin formation in the presence of growth factors and hormones (e.g., fibroblast growth factor, platelet-derived growth factor, progesterone, thyroxine) has been extensive (13–23), it has not been possible to distinguish effects on proliferation, initiation of myelin formation, or the actual rate of myelin formation. With this method, the effects and functions of a variety of factors may be determined on these steps in the overall myelination process. The results reported in this paper suggest that endogenous glucocorticoids and progestins play a critical role in signaling myelin formation.
MATERIALS AND METHODS
Dorsal Root Ganglia Neuron/Schwann Cell Cocultures.
Purified neuronal and Schwann cell cultures were prepared using methods described by Eldridge et al. (24). Briefly, neuronal cultures were established from dorsal root ganglia neurons obtained from 15-day gestation Sprague Dawley rat embryos (Harlan). Twenty-five-millimeter glass coverslips in 35-mm dishes were coated with 1 ml of 100 μg/ml collagen for 12 hr. The collagen was then removed, and the plates were allowed to dry thoroughly. The dorsal root ganglia neurons were dissociated with 0.25% trypsin and plated onto the collagen-coated coverslips. Nonneuronal cells were eliminated by cycling with a fluorodeoxyuridine-containing medium. Neurons were then maintained for 1 wk in a medium consisting of 10% fetal bovine serum in MEM and 200 ng/ml nerve growth factor (M1 medium).
Schwann cells were isolated from the sciatic nerve of 4-day-old rat pups (25). The sciatic nerves were dissociated with trypsin and collagenase. The dissociated Schwann cells were maintained for 3–5 days in a medium consisting of 10% fetal bovine serum in DMEM with 50 μg/ml gentamicin and 10 μM cytosine arabinoside to eliminate fibroblasts. The purified Schwann cells were then used to seed the purified neuronal cells and establish cocultures. The purified neuronal cultures of approximately 70,000 cells were seeded with approximately 100,000 Schwann cells. Cocultures were then maintained in MEM with the addition of 10% charcoal-filtered fetal calf serum (delipidated) and 200 ng/ml nerve growth factor. After the Schwann cells fully populated and ensheathed the axons, myelination was induced with the addition of ascorbate (50 μg/ml), which was replenished when feeding every third day.
Oligodendrocyte Cell Culture Conditions.
Oligodendrocyte precursor cells were established from brains of 2-day-old Sprague Dawley rat pups (Harlan) using a modified method of McKinnon et al. (18) and Asou et al. (26). Briefly, after the removal of the meninges, cells were dissociated by mechanical disruption through 18-, 20-, and 25-gauge needles. The cells were plated in 75-cm2 Falcon flasks and maintained in DMEM containing 10% fetal bovine serum. After 12–18 days in culture, the O-2A cells had spread over a continuous layer of astrocytes. These cells were detached by overnight shaking (200 rpm) at 35.5°C (27) and washed with DMEM. The isolated cells were centrifuged and allowed to adhere to 100-mm dishes for 30 min at 35.5°C. During this differential adhesion step, astrocytes and microglia adhered to the plastic dishes, whereas the O-2A cells did not. These cells were washed and seeded onto poly l-lysine coated coverslips or 35- and 60-mm dishes.
O-2A precursors were maintained in a proliferative state in DMEM supplemented with 50 μg/ml transferrin, 30 nM sodium selenite, 30 nM triiodothyronine (28), 50 ng/ml insulin, 0.5% fetal bovine serum, 5 ng/ml basic fibroblast growth factor, and 10 ng/ml human platelet-derived growth factor-AA (Sigma). O-2A cells were pushed to differentiate into oligodendrocytes by the removal of the growth factors and astrocytes were generated by the addition of DMEM containing 20% fetal bovine serum (6).
Labeling of Cocultures with C5-DMB-Hexadecanoic Acid.
Cells were labeled using essentially the conditions of Bilderback et al. (12). Briefly, 250 nmol of the C5-DMB-hexadecanoic acid was dissolved in 200 μl of ethanol and injected into 10 ml of 1.7 mg/ml delipidated BSA in Hepes-buffered DMEM. The complex was dialyzed overnight against 1 liter of Hepes-buffered DMEM at 4°C. The cocultures were then placed into a temperature-controlled perfusion chamber (Medical Systems Corporation, Chicago, IL) with a final concentration of 0.75 μM C5-DMB-hexadecanoic acid. The fluorescence intensity was monitored using a fluorescence digital imaging microscope described previously (29). The labeled cocultures were excited at wavelengths between 420 and 480 nm, whereas the emission was monitored at wavelengths greater than 515 nm. The fluorescence intensity of an internode was corrected for background fluorescence and the mean intensity of the internode was calculated (12). The fluorescence intensity and the internode diameters were measured using the IPLab Spectrum program (Signal Analytics Corporation, Vienna, VA). Internodes were randomly measured throughout the myelination process and at least 10 internodes were followed for each time point. Generally, three to five internodes were followed on each coverslip. In addition, the ratio of fluorescent lipid to total lipid was determined to be <1:6000 during the time course when rate measurements were made. No significant self-quenching was observed at levels below 1:1000 when BODIPY phosphatidylcholine vesicles were examined (30). The cultures were incubated at 35.5°C in Hepes-buffered DMEM with 10% charcoal-filtered fetal calf serum and 200 ng/ml nerve growth factor.
Oligonucleotides.
Primer pairs were designed in conservative regions from sequences available in the GenBank/European Molecular Biology Laboratory database. Primer pairs for the progesterone receptor and the internal standard were based on primers designed by Park-Sarge et al. (31). Two 21-mer nucleotides (5′-CTGAAGGTCAAAGGGAATGTG-3′ and 5′-GGACAGAGTCTTGATGATCTC-3′) were used to amplify a 195-bp region of ribosomal protein L19 as an internal control (32), whereas two 22-mer nucleotides flanking the hormone-binding domain of the rat progesterone receptor (5′-CCCACAGGAGTTTGTCAAGCTC-3′ and 5′-TAACTTCAGACATCATTTCCGG-3′) were used to amplify the 325-bp product (31). Primers for cytochrome P450scc and 3β-hydroxysteroid dehydrogenase (3β-HSD) were based on primers designed by Sanne and Krueger (33). Primers for cytochrome P450scc (5′-GAGACACCACCCTCAAATGCCCC-3′ and 5′-CCTGATGCCTGAGAAGCCTATC-3′) were designed to amplify a 150-bp product with three AluI restriction sites. Primers for 3β-HSD (5′-CTGAGCAAGGAATGGGGCCTCCGC-3′ and 5′-GCTATTTGACAGTGTGACCAAGTG-3′) were constructed to amplify a 159-bp product, which was based on nucleotide sequences of types I and II rat 3β-HSD (34) with two AluI restriction sites. Two 21-mer nucleotides flanking the hormone-binding domain of the rat glucocorticoid receptor (5′-GAGCAGAGAATGTCTCTACCC-3′ and 5′-GACGATGGCTTTTCCTAGCTC-3′) were constructed to amplify a 216-bp product with two SapI sites.
Reverse Transcription-PCR.
RNA from cultured cells was isolated using RNAgents Total Isolation System (Promega). The concentration and purity of total RNA was determined by measuring the optical density at 260 and 280 nm. The RNA was subjected to DNase treatment (DNase I, FPLCpure; Pharmacia Biotech, Piscataway, NJ) at 37°C for 15 min to remove residual genomic DNA. The RNA was reverse transcribed (33) at 37°C for 90 min in the presence of oligo(dT) primers. Residual RNA was then digested at 37°C with 1 unit of RNase H. Aliquots of cDNA were subjected to 30 cycles of amplification using a minicycler (MJ Research, Watertown, MA). The amplification mixture contained the cDNA, the oligonucleotide primers (0.2 μg each), 200 μM deoxy-NTPs with the addition of [α-32P]deoxy-CTP (4 mCi) at 3000 Ci/mmol, 1.5 mM MgCl2, 2.5 units of Taq DNA polymerase, the total volume was brought to 100 μl with 1× PCR buffer [10 mM Tris (pH 8.3) and 50 mM KCl], and the amplification mixture was then overlaid with light mineral oil. The amplification included a “hot start” protocol in which the Taq polymerase was withheld until the reaction temperature was at 80°C, to ensure high specificity of the products being synthesized. After the hot start, each cycle consisted of a denaturation step (94°C, 45 sec), an annealing step (55°C, 30 sec), and an elongation step (72°C, 1.5 min), with a final 10-min elongation after the last cycle.
The amplified samples were analyzed using 2% agarose gels and 8% polyacrylamide gels. The gels were subsequently quantified with the IPLab Images software as well as using a PhosphorImager and the ImageQuant software (Molecular Dynamics). The relative levels of gene expression were measured by determining a ratio between the products generated from the target gene and the endogenous internal standard in separate reactions (35). The linear range for amplifying regions of the genes of interest and the gene for the internal standard were determined by serial dilutions of the cDNA. The slopes of the lines for the intensity of the amplified cDNA versus concentration were calculated and the ratios of the gene of interest to the internal standard were compared. The fold increase observed is relative to premyelinating cultures at 3 days after seeding.
Specific Activity of 3β-HSD.
The specific activity for 3β-HSD was measured at 37°C at various stages during the myelination process in Schwann cell/neuronal cocultures using methods previously described by Zhao et al. (36) and Ishii-Ohba et al. (37). The cocultures were homogenized in phosphate buffer (50 mM KH2PO4, 20% glycerol, 1 mM EDTA, pH 7.5) with the addition of 1 mM phenylmethylsulfonyl fluoride. The 1-ml reaction mixture contained 100 mM phosphate buffer (pH 7.5), 1.5 mM NAD+, and 3 mM pregnenolone. Using a SLM 8000 fluorometer, the activity of crude coculture homogenate was assayed by monitoring the time-dependent increase in NADH fluorescence using an exitation wavelength of 340 nm and an emission wavelength of 459 nm. The specific activity was expressed as nmoles of NADH formed per minute per milligram of total protein. Protein concentrations were measured using the BCA kit (Sigma).
RESULTS
Rate of Myelin Formation.
Fluorescence digital imaging microscopy was employed to investigate the process of myelin formation by Schwann cells in neuronal cocultures. Using the fluorescent-labeled free fatty acid (C5-DMB-hexadecanoic acid), fluorescent lipid was incorporated into the myelin sheath. C5-DMB-hexadecanoic acid (0.75 μM) was added directly to the cocultures, and the rate of fluorescent lipid incorporation at various stages in the myelination process was continuously measured. By monitoring the synthesis of myelin-specific lipids (galactocerebroside, sulfatide, and sphingomyelin) by TLC, active myelin formation was found to occur between 4 and 7 days after induction with ascorbate (12). By measuring the fluorescent lipid incorporation throughout the myelination process by fluorescence digital imaging microscopy, the peak rate of myelin formation was also found to occur between 4 and 7 days after induction. The fluorescence intensity (radiance value) was measured for individual internodes during myelin formation in a temperature-controlled (35°C) perfusion chamber. For example, changes in the fluorescence intensity of an internode were observed at 5, 10, 13, 16, and 22 hr after labeling, as shown in Fig. 1. As the Schwann cell actively formed new myelin membrane, the rate of fluorescent lipid incorporation (radiance value increase in the internode per hour) increased by approximately 5-fold (Table 1). This increase was comparable to results obtained using the fluorescently labeled ceramide analogue (12).
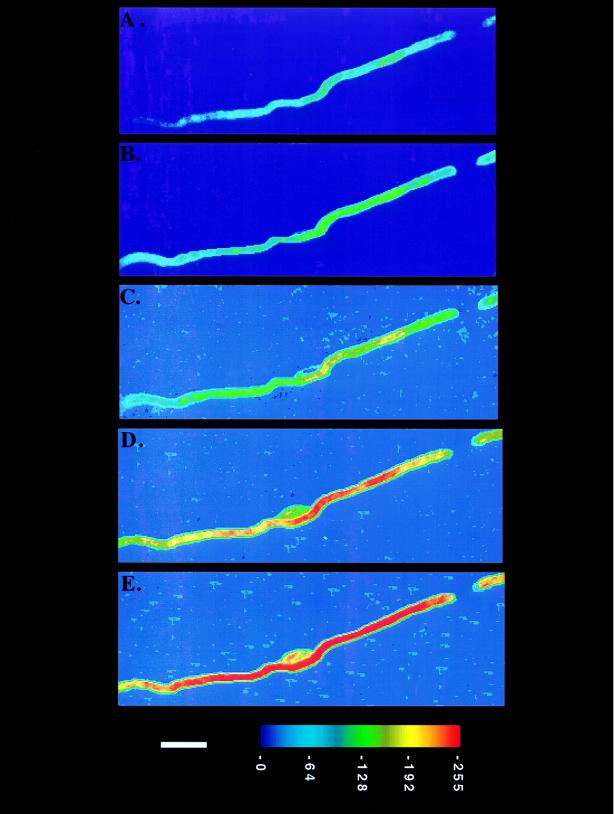
Figure 1.
Time course of myelin formation by a single Schwann cell on an axon. C5-DMB-hexadecanoic acid (0.75 μM) was added directly to the coculture and its incorporation was monitored for 30 hr. The coculture was placed in a temperature-controlled (35°C) perfusion chamber and viewed with a fluorescence digital imaging microscope. Changes in the fluorescence intensity of the internode were observed at 5 hr (A), 10 hr (B), 13 hr (C), 16 hr (D), and 22 hr (E) after labeling. The cell body of the Schwann cell is visible in the middle of the internode in the last three frames. The background fluorescence attributable to the node of Ranvier is detected in the upper right corner along with the tip of the adjacent internode. The axon is not visible. The applied pseudocolor scheme and a 10-μm bar appear at the bottom.
Table 1.
Rate of myelin formation and the diameters for different internodes were measured during premyelination and myelination periods by Schwann cells in neuronal cocultures
| Rate of myelin formation
|
|||
|---|---|---|---|
| (Radiance value change/hr) | Diameter, μm | ||
| Dorsal root ganglia axons alone | 0.9 ± 0.2 | 0.8 ± 0.1 | |
| Premyelinating internodes (0–2 days after induction) | Day 0 | Day 2 | |
| Control | 1.1 ± 0.2 | 0.8 ± 0.2 | 1.2 ± 0.3 |
| Myelinating internodes (3–5 days after induction) | Day 3 | Day 5 | |
| Control | 5.1 ± 0.9 | 1.9 ± 0.1 | 3.0 ± 0.4 |
| +Progesterone | 10.5 ± 1.2 | 2.2 ± 0.2 | 2.9 ± 0.6 |
| +RU486 | 2.4 ± 0.9 | 1.6 ± 0.1 | 1.8 ± 0.3 |
| +R5020 | 10.0 ± 1.6 | 2.0 ± 0.1 | 3.1 ± 0.4 |
| +Dexamethasone | 11.2 ± 1.7 | 1.9 ± 0.2 | 3.0 ± 0.4 |
| +Basic fibroblast growth factor | 5.4 ± 1.0 | 1.8 ± 0.3 | 3.2 ± 0.3 |
| +Platelet-derived growth factor | 5.0 ± 1.2 | 2.0 ± 0.4 | 2.9 ± 0.3 |
| Mature myelin internodes (7–10 days after induction) | |||
| Control | 1.0 ± 0.2 | 3.0 ± 0.4 | |
| +Progesterone | 1.1 ± 0.5 | 2.9 ± 0.6 | |
| +Dexamethasone | 1.2 ± 0.3 | 3.0 ± 0.4 | |
Initially, several compounds were added to the cocultures that were reported to effect the overall myelination process. Although fibroblast growth factor and platelet-derived growth factor, for example, were found to have proliferative effects on Schwann cells, they did not affect the time of initiation or rate of myelin formation (Table 1). To investigate the influence of progesterone on the rate of myelination, exogenous progesterone (100 nM) was added to the cocultures. The Schwann cells appeared to differentiate earlier (ensheath axons) and increased the peak rate of myelin formation by more than 2-fold. Similarly, dexamethasone, a glucocorticoid analogue (100 nM), was also found to accelerate differentiation and increased the peak rate by more than 2-fold (Table 1). In the presence of RU-486 (mifepristone, 100 nM), a competitive antagonist of progestins and glucocorticoids, the Schwann cells seemed to ensheath the axons normally but the peak rate of myelination decreased by 4-fold as compared with the cultures with progesterone or dexamethasone. Internode diameters were measured using fluorescence microscopy during various stages of myelin formation. The average internode diameters were measured by counting the number of pixels across the internode. Progesterone and dexamethasone did not seem to affect the total extent of myelin, as determined by the final diameter of myelin internodes. RU-486 inhibited the formation of myelin and no increase in internode diameters was detected. R-5020 (promegestone, 10 nM), an agonist of progesterone, was also applied to the cocultures. The rate of myelination increased more than 4-fold, as compared with cultures in the presence of RU-486. The extent of myelin formation in the presence of R-5020 was similar to that with dexamethasone or progesterone (Table 1).
Initiation of Myelination.
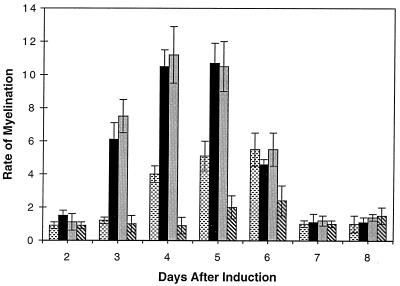
To determine the role of progesterone and dexamethasone on the initiation of myelin formation, the steroids were added on the day of induction of myelination by ascorbate and were replenished every 2 days with new media. Progesterone and dexamethasone were found to accelerate the time of initiation of myelin formation by 24 hr (Fig. 2) and shift the peak rate of myelin formation to 3–5 days. In the presence of RU-486, the Schwann cells seemed to ensheath the axons normally, but the initiation and rate of myelination were drastically reduced. RU-486 was found to inhibit myelin formation as determined by diameter measurements (Table 1).
Figure 2.
The rate of myelination measured at various stages during the myelination process in Schwann cell/neuronal cocultures. The rate of myelin formation was measured in the presence of progesterone ( ), dexamethasone (■), RU-486 (
), dexamethasone (■), RU-486 ( ), and in control cultures (
), and in control cultures ( ).
).
Relative mRNA Expression in Schwann Cell/Neuronal Cocultures.
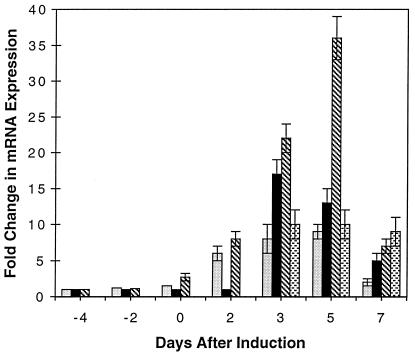
The rate-limiting step in progesterone biosynthesis is the conversion of cholesterol to pregnenolone by cytochrome P450scc. This reaction includes the hydroxylation of cholesterol and cleavage of the C20–C22 bond to form pregnenolone and isocaproic acid (38). 3β-HSD is responsible for the isomerization and oxidation of pregnenolone to progesterone. The mRNA expression of cytochrome P450scc, 3β-HSD, the progesterone receptor, and the glucocorticoid receptor were examined in cocultures at various stages of myelination. Because of the small amounts of message for cytochrome P450scc and 3β-HSD in nonsteroidogenic tissues (33, 38), reverse transcription-PCR was used. The cDNA prepared from cocultures was subjected to 30 cycles of amplification and the relative levels of gene expression were measured by determining a ratio between the products generated from the target gene and an endogenous internal standard gene (31) in separate reactions (35). The linear range for amplifying regions of the genes of interest and the gene for the internal standard were determined by serial dilutions of the cDNA. The slopes of the lines for the intensity of the amplified cDNA versus concentration were calculated and the ratios of the gene of interest to the internal standard were compared. The fold change in expression given in Fig. 3 is relative to premyelinating cultures at 3 days after seeding.
Figure 3.
The relative change in the mRNA expression at various stages of the myelination process in Schwann cell/neuronal cocultures. The relative mRNA expression for myelin basic protein ( ), cytochrome P450scc (■), 3β-HSD (
), cytochrome P450scc (■), 3β-HSD ( ), and the progesterone receptor (
), and the progesterone receptor ( ) were measured during various stages of the myelination process. The fold change in mRNA expression is relative to premyelinating cultures at 3 days after seeding. Schwann cells were induced to myelinate 7 days after seeding (day 0) by the addition of ascorbic acid.
) were measured during various stages of the myelination process. The fold change in mRNA expression is relative to premyelinating cultures at 3 days after seeding. Schwann cells were induced to myelinate 7 days after seeding (day 0) by the addition of ascorbic acid.
A 17-fold increase in mRNA expression of cytochrome P450scc was observed between 3 and 5 days after induction of myelination with ascorbate. 3β-HSD and the progesterone receptor were also found to increase by 35- and 10-fold, respectively, between 3 and 5 days (Fig. 3). As mentioned previously, active myelin formation was found to occur between 4 and 7 days as determined by monitoring the synthesis of myelin-specific lipids (12) and measuring the rate of fluorescent lipid incorporation. In addition, the myelin basic protein mRNA expression was found to increase by 9-fold and coincide with cytochrome P450scc, 3β-HSD, and the progesterone receptor messages (Fig. 3). These results demonstrate that the increase in mRNA for the enzymes and the progesterone receptor in Schwann cell/neuronal cocultures coincide or slightly precede myelin formation. The expression of the glucocorticoid receptor message was also detected, and the level of expression did not change during the myelination process (data not shown).
Relative mRNA Expression in Cultured Oligodendrocytes.
Oligodendrocyte precursor cells (O-2A), type 2 astrocytes, and oligodendrocytes were analyzed for cytochrome P450scc, 3β-HSD, the progesterone receptor, and the glucocorticoid receptor. Using different culturing conditions, O-2A cells were influenced to differentiate into oligodendrocytes, type 2 astrocytes, or remain in a proliferative state (6, 18, 20, 39). Using reverse transcription-PCR, a 20-fold increase in mRNA expression of cytochrome P450scc was observed as O-2A cells differentiated into mature oligodendrocytes. 3β-HSD was also found to increase by approximately 10-fold, the progesterone receptor was barely detected, whereas the glucocorticoid receptor was constituitively expressed (data not shown). The induction of cytochrome P450scc and 3β-HSD was found to coincide with the differentiation of O-2A cells into myelin-forming oligodendrocytes. Induction of the mRNA for the enzymes responsible for steroid biosynthesis was not observed when O-2A cells differentiated into type 2 astrocytes.
Specific Activity of 3β-HSD.
The specific activity of 3β-HSD was measured at various stages during myelin formation in Schwann cell/neuronal cocultures (Table 2). The activity of the crude coculture homogenate was assayed by monitoring the time-dependent increase in NADH fluorescence using an excitation wavelength of 340 nm and an emission wavelength of 459 nm (37) in the presence of NAD+ and substrate (pregnenolone). The specific activity for 3β-HSD increased by 10-fold between 3 and 5 days after induction. The peak activity coincided with the increased mRNA expression for cytochrome P450scc, 3β-HSD, the progesterone receptor, myelin basic protein, and myelin formation. Thus, the mRNA for 3β-HSD was translated into active enzyme during this period.
Table 2.
Specific activity for 3β-HSD at various stages during the myelination process in Schwann cell/neuronal cocultures
| Days after induction | Specific activity, nmol/min/mg |
|---|---|
| −4 | 0.42 ± 0.03 |
| −2 | 1.02 ± 0.01 |
| 0 | 1.55 ± 0.30 |
| 3 | 4.25 ± 0.50 |
| 6 | 3.59 ± 0.11 |
DISCUSSION
Recently, the presence and actions of steroids and hormones on the central and peripheral nervous systems have received widespread attention. Estradiol (40), retinoic acid (41), and thyroid hormone (14, 21, 42, 43) have all been implicated in regulating the differentiation of glial cells in culture, suggesting an interaction of various hormones in the myelination process. Corticosteroids have played an important role in the diagnosis and therapy of demyelinating diseases (44–46) and have been found to modulate myelin proteins in oligodendrocytes (14, 19). In addition, the synthesis of progesterone and the presence of the receptor have been reported in Schwann cells and oligodendrocytes. Progesterone has also been implicated in increasing the expression of myelin proteins (13, 47, 48), but the direct effects and functions are not known. Research concerning the proliferation and differentiation of glial cells in the presence of growth factors has been extensive (15, 17, 18, 20), but it has also been difficult to elucidate the factors directly responsible for the initiation, rate of myelin formation, and extent of myelin formation.
We have developed a method that allows the continuous monitoring of a single internode throughout the myelination process. Using a fluorescently labeled free fatty acid, fluorescent lipid was incorporated into the myelin sheath and the intensity (radiance value) was measured for individual internodes. Progesterone and dexamethasone were found to accelerate the time of initiation of myelin formation by approximately 24 hr and increase the peak rate by more than 2-fold. Thus, the rate of myelin formation is not just induced to a certain value, but can be modulated depending on the conditions. There were no noticeable differences in the extent of myelin formation, as determined by diameter measurements. RU-486 was found to inhibit the initiation of myelin formation and to decrease the rate of myelin formation and the extent of the formation of the myelin sheath. Since RU-486 is an antagonist of both glucocorticoids and progestins, the effects of the steroids cannot be solely attributed to either receptor. Although the effects of R-5020, a progesterone agonist, are specifically directed at the progesterone receptor, the functions of the glucocorticoid receptor may nevertheless be significant. The inhibition of RU-486 and the enhancement of R-5020 demonstrate that the actions of progesterone and dexamethasone are specific at the level of the receptor.
Regulation of Myelin Formation.
The mRNA for cytochrome P450scc, 3β-HSD, and the progesterone receptor were found to be induced during active myelin formation in Schwann cell/neuronal cocultures, whereas the glucocorticoid receptor was found to be constitutively expressed. The induction of 3β-HSD enzyme activity was also found to occur during myelin formation, demonstrating the translation of the message during this period. The mRNA for the enzymes responsible for steroid biosynthesis cannot be detected in appreciable amounts in cultured Schwann cells or in cultured neurons alone. The induction of these enzymes occurs during myelin formation in the cocultures. Since approximately 10–15% of the axons in the cultures are myelinated, the change in mRNA expression in individual neurons may be significantly greater.
Cultured oligodendrocytes also have been found to express cytochrome P450scc, 3β-HSD, the progesterone receptor, and the glucocorticoid receptor independently of neurons. Although neuronal participation is essential for Schwann cells to differentiate and make myelin, oligodendrocytes present a different system of differentiation. In vitro, O-2A progenitor cells display a phenotypic plasticity seen in various multipotential precursors. Cultured oligodendrocytes are capable of forming myelin-like sheets independent of neurons, although the interactions with neurons are still essential in producing proper myelin internodes. O-2A cells appear to generate myelinating oligodendrocytes but not type 2 astrocytes when cocultured in the presence of neurons (49). Thus, the induction of the enzymes involved in steroid biosynthesis in oligodendrocytes independent of neurons does not exclude the possibility that interactions between the two cells will change the pattern of expression. In summary, these results demonstrate that myelin-forming cells have inducible enzymes responsible for steroid biosynthesis and suggest a critical role for endogenous steroid hormones in signaling the initiation and enhancing the rate of myelin formation. By elucidating the factors that regulate neuronal–glial interactions and the myelination process, new therapeutic strategies into myelin repair and demyelinating diseases may be possible.
Acknowledgments
This work was supported in part by Grant RG 2660A1/1 from the Multiple Sclerosis Society. J.P. is a Howard Hughes Medical Institute Predoctoral Fellow.
ABBREVIATION
- 3β-HSD
3β-hydroxysteroid dehydrogenase
References
- 1.Bunge R P. Curr Opin Neurobiol. 1993;3:805–809. doi: 10.1016/0959-4388(93)90157-t. [DOI] [PubMed] [Google Scholar]
- 2.Bunge M B, Williams A K, Woods P M. Dev Biol. 1982;92:449–460. doi: 10.1016/0012-1606(82)90190-7. [DOI] [PubMed] [Google Scholar]
- 3.Knapp P E, Bartlett W P, Skoff R P. Dev Biol. 1987;120:356–365. doi: 10.1016/0012-1606(87)90238-7. [DOI] [PubMed] [Google Scholar]
- 4.Rome L H, Bullock P N, Chiappelli F, Cardwell M, Adinoff A M, Swanson D. J Neurosci Res. 1986;15:49–65. doi: 10.1002/jnr.490150106. [DOI] [PubMed] [Google Scholar]
- 5.Bradel E J, Prince F P. J Neurosci Res. 1983;9:381–392. doi: 10.1002/jnr.490090404. [DOI] [PubMed] [Google Scholar]
- 6.Raff M C, Miller R H, Noble M. Nature (London) 1983;303:390–396. doi: 10.1038/303390a0. [DOI] [PubMed] [Google Scholar]
- 7.Gould R M, Jessen K R, Mirsky R, Tennekoon G. In: The Cell of Schwann: An Update. Myelin: Biology and Chemistry. Martenson R E, editor. Boco Raton, FL: CRC; 1992. pp. 123–171. [Google Scholar]
- 8.Bunge R P, Bunge M B, Bates M. J Cell Biol. 1989;109:273–284. doi: 10.1083/jcb.109.1.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Webster H, deF. J Cell Biol. 1971;48:348–367. doi: 10.1083/jcb.48.2.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fernandez-Valle C, Fregien N, Wood P M, Bunge M B. Development. 1993;119:867–880. doi: 10.1242/dev.119.3.867. [DOI] [PubMed] [Google Scholar]
- 11.Mirsky R, Dubois C, Morgan L, Jessen K R. Development. 1990;109:105–116. doi: 10.1242/dev.109.1.105. [DOI] [PubMed] [Google Scholar]
- 12.Bilderback T, Chan J R, Harvey J, Glaser M. J Neurosci Res. 1997;49:497–507. [PubMed] [Google Scholar]
- 13.Koenig H L, Schumacher M, Ferzaz B, Do Thi A N, Ressouches A, Guennoun R, Jung-Testas I, Robel P, Baulieu E E. Science. 1995;268:1500–1503. doi: 10.1126/science.7770777. [DOI] [PubMed] [Google Scholar]
- 14.Barres B A, Lazar M A, Raff M C. Development. 1994;120:1097–1108. doi: 10.1242/dev.120.5.1097. [DOI] [PubMed] [Google Scholar]
- 15.Watabe K, Fukuda T, Tanaka J, Toyohara K, Sakai O. J Neurosci Res. 1994;39:525–534. doi: 10.1002/jnr.490390504. [DOI] [PubMed] [Google Scholar]
- 16.Tosic M, Torch S, Comte V, Dolivo M, Honegger P, Matthieu J M. J Neurochem. 1992;59:1770–1777. doi: 10.1111/j.1471-4159.1992.tb11009.x. [DOI] [PubMed] [Google Scholar]
- 17.Bogler O, Wren D, Barnett S C, Land H, Noble M. Proc Natl Acad Sci USA. 1990;87:6368–6372. doi: 10.1073/pnas.87.16.6368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McKinnon R D, Matsui T, Dubois-Dalcq M, Aaronson S A. Neuron. 1990;5:603–614. doi: 10.1016/0896-6273(90)90215-2. [DOI] [PubMed] [Google Scholar]
- 19.Kumar S, Cole R, Chiappelli F, de Vellis J. Proc Natl Acad Sci USA. 1989;86:6807–6811. doi: 10.1073/pnas.86.17.6807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Raff M C, Lillien L E, Richardson W D, Burne J F, Noble M D. Nature (London) 1988;333:562–565. doi: 10.1038/333562a0. [DOI] [PubMed] [Google Scholar]
- 21.Almazan G, Honegger P, Matthieu J M. Dev Neurosci. 1985;7:45–54. doi: 10.1159/000112275. [DOI] [PubMed] [Google Scholar]
- 22.Canoll P D, Musacchio J M, Hardy R, Reynolds R, Marchionni M A, Salzer J L. Neuron. 1996;17:229–243. doi: 10.1016/s0896-6273(00)80155-5. [DOI] [PubMed] [Google Scholar]
- 23.Shinar Y, McMorris F A. J Neurosci Res. 1995;42:516–527. doi: 10.1002/jnr.490420410. [DOI] [PubMed] [Google Scholar]
- 24.Eldridge C F, Bunge R P, Wood P M. J Cell Biol. 1987;105:1023–1034. doi: 10.1083/jcb.105.2.1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Brockes J P, Fields K L, Raff M C. Brain Res. 1979;165:105–118. doi: 10.1016/0006-8993(79)90048-9. [DOI] [PubMed] [Google Scholar]
- 26.Asou H, Hamada K, Miyazaki T, Sakota T, Hayashi K, Takeda Y, Marret S, Delpech B, Itoh K, Uyemura K. J Neurosci Res. 1995;40:519–534. doi: 10.1002/jnr.490400411. [DOI] [PubMed] [Google Scholar]
- 27.McCarthy K D, de Vellis J. J Cell Biol. 1980;85:890–902. doi: 10.1083/jcb.85.3.890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eccleston P A, Silberberg D H. Dev Brain Res. 1984;16:1–9. doi: 10.1016/0165-3806(84)90056-7. [DOI] [PubMed] [Google Scholar]
- 29.Yang L, Glaser M. Biochemistry. 1995;34:1500–1506. doi: 10.1021/bi00005a005. [DOI] [PubMed] [Google Scholar]
- 30.Johnson I D, Kang H C, Haugland R P. Anal Biochem. 1991;198:228–237. doi: 10.1016/0003-2697(91)90418-s. [DOI] [PubMed] [Google Scholar]
- 31.Park-Sarge O K, Parmer T G, Gibori G. Endocrinology. 1995;136:1537–1543. doi: 10.1210/endo.136.4.7534703. [DOI] [PubMed] [Google Scholar]
- 32.Chan Y-L, Lin A, McNally J, Pelleg D, Meyuhas O, Wool I. J Biol Chem. 1987;262:1111–1115. [PubMed] [Google Scholar]
- 33.Sanne J L, Krueger K E. J Neurochem. 1995;65:528–536. doi: 10.1046/j.1471-4159.1995.65020528.x. [DOI] [PubMed] [Google Scholar]
- 34.Zhao H F, Labrie C, Simard J, Launoit Y, Trudel C, Martel C, Rheaume E, Dupont E, Luu-The V, Pelletier G, Labrie F. J Biol Chem. 1991;266:583–593. [PubMed] [Google Scholar]
- 35.Horikoshi T, Danenberg K, Volkenandt M, Stadlbauer T, Danenberg P V. In: PCR Protocols. White B A, editor. Clifton, NJ: Humana; 1993. pp. 177–188. [DOI] [PubMed] [Google Scholar]
- 36.Zhao H F, Rheaume E, Trudel C, Couet J, Labrie F, Simard J. Endocrinology. 1990;127:3237–3239. doi: 10.1210/endo-127-6-3237. [DOI] [PubMed] [Google Scholar]
- 37.Ishii-Ohba H, Saiki N, Inano H, Tamaoki B I. J Steroid Biochem. 1986;24:753–760. doi: 10.1016/0022-4731(86)90854-x. [DOI] [PubMed] [Google Scholar]
- 38.Lambeth J D, Pember S O. J Biol Chem. 1983;258:5596–5602. [PubMed] [Google Scholar]
- 39.Dubois-Dalcq M. EMBO J. 1987;6:2587–2595. doi: 10.1002/j.1460-2075.1987.tb02549.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jung-Testas I, Schumacher M, Robel P, Baulieu E E. J Steroid Biochem Mol Biol. 1994;48:145–154. doi: 10.1016/0960-0760(94)90261-5. [DOI] [PubMed] [Google Scholar]
- 41.Laeng P, Decimo D, Pettmann B, Janet T, Labourdette G. J Neurosci Res. 1994;39:613–633. doi: 10.1002/jnr.490390602. [DOI] [PubMed] [Google Scholar]
- 42.Koper J W, Hoeben R C, Hochstenbach F, van Golde L, Lopes-Cardozo M. Biochim Biophys Acta. 1986;887:327–334. doi: 10.1016/0167-4889(86)90162-x. [DOI] [PubMed] [Google Scholar]
- 43.Walters S N, Morell P. J Neurochem. 1981;36:1792–1801. doi: 10.1111/j.1471-4159.1981.tb00433.x. [DOI] [PubMed] [Google Scholar]
- 44.Holsboer F, Grasser A, Friess E, Wiedemann K. Ann NY Acad Sci. 1994;746:345–359. doi: 10.1111/j.1749-6632.1994.tb39255.x. [DOI] [PubMed] [Google Scholar]
- 45.Michelson D, Stone L, Galliven E, Magiakou M A, Chrousos G P, Sternberg E M, Gold P W. J Clin Endocrinol Metab. 1994;79:848–853. doi: 10.1210/jcem.79.3.8077372. [DOI] [PubMed] [Google Scholar]
- 46.Woelk H, Neu I. Fortschr Med. 1980;98:1427–1430. [PubMed] [Google Scholar]
- 47.Jung-Testas I, Schumacher M, Robel P, Baulieu E E. Cell Mol Neurobiol. 1996;16:439–443. doi: 10.1007/BF02088110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Robel P, Baulieu E E. Trends Endocrinol Metab. 1994;5:1–8. doi: 10.1016/1043-2760(94)90114-7. [DOI] [PubMed] [Google Scholar]
- 49.Espinosa de los Monteros A, Zhang M, de Vellis J. Proc Natl Acad Sci USA. 1993;90:50–54. doi: 10.1073/pnas.90.1.50. [DOI] [PMC free article] [PubMed] [Google Scholar]