Abstract.
Many neuropsychiatric disorders are considered to be related to the dysregulation of brain serotonergic neurotransmission. Tryptophan hydroxylase-2 (TPH2) is the neuronal-specific enzyme that controls brain serotonin synthesis. There is growing genetic evidence for the possible involvement of TPH2 in serotonin-related neuropsychiatric disorders; however, the degree of genetic variation in TPH2 and, in particular, its possible functional consequences remain unknown. In this short review, we will summarize some recent findings with respect to the functional analysis of TPH2.
Key words. Brain serotonin, tryptophan hydroxylase-2 (TPH2), single nucleotide polymorphism (SNP), congenic mice, PC12 cells
Introduction
The neurotransmitter serotonin (5-hydroxytryptamine, 5-HT) has been implicated in various physiological functions in both peripheral and central nervous systems (CNS). Many neuropsychiatric disorders, including depression [1–4], schizophrenia [4–6], aggression and suicidal behavior [1, 6], attention-deficit/hyperactivity disorder (ADHD) [7–9], obsessive-compulsive disorder [10] and autism [11] have been suggested to be related to the dysregulation of brain serotonergic neurotransmission. Tryptophan hydroxylase (TPH) is the rate-limiting enzyme in 5-HT synthesis and belongs to the superfamily of aromatic amino acid hydroxylase, which also includes tyrosine hydroxylase (TH) and phenylalanine hydroxylase (PAH). TPH, TH and PAH share considerable structural similarity and require the same co-factor (Fe2+) and cosubstrate (tetrahydrobiopterin, BH4) for function [12]. While TH and PAH are each encoded by a single gene, there has been evidence suggesting that two genes may encode TPH [13, 14]. The recent identification of a TPH2 gene [15] has demonstrated that this is indeed the case. The well-characterized TPH1 was identified 4 decades ago [16] and has recently been shown to be essentially peripheral, being expressed predominantly in the pineal gland and enterochromaffin cells of the gut [15, 17–19]. In contrast to the results of RNase protection assay [15] and in situ hybridization study [19], Zill et al. [20] recently reported the expression of TPH1 messenger RNA (mRNA) in several brain regions in post-mortem human brains. However, further studies may be needed to examine whether such TPH1 expression represents the pineal projections in these brain regions [21], as serotonin Nacetyltransferase (AANAT), the rate-limiting enzyme in melatonin synthesis, has been shown to be expressed also in certain brain regions [21]. TPH2, however, is expressed predominantly in serotonergic neurons of the raphe nuclei [15, 19] and in the peripheral myenteric neurons in the gut [18]. To date, TPH2 has been identified and cloned in human, mice, rat, chicken, zebrafish, torafugu and fruit fly (gene accession numbers: NM_173353 [human], NM_173391 [mouse], NM_173839 [rat], NM_001001301 [chicken], NM_214795 [zebrafish], AY616189 [torafugu]) [15, 22, 23]. This paper will summarize some recent findings about TPH2, and focus on the functional analysis of TPH2 and its potential contribution to the dysregulation of brain serotonin homeostasis associated with the pathophysiology of neuropsychiatric disorders.
TPH polymorphisms and neuropsychiatric disorders
TPH1 has long been considered the sole rate-limiting enzyme for the synthesis of 5-HT. Two common intronic (non-coding) single nucleotide polymorphisms (SNPs), A218C and A779C, in TPH1 have been extensively studied for their possible associations with neuropsychiatric conditions, including suicidal behavior, unipolar depression, bipolar depression and schizophrenia with conflicting outcomes (reviewed in [1]). However, the recent discovery of the neuronal-specific TPH2 [15] that is critical for brain serotonin synthesis [24] has redefined our perspective of the serotonergic systems. These findings have suggested that TPH2, rather than its peripheral counterpart (TPH1), should be investigated as a candidate gene for 5-HT-related neuropsychiatric conditions. In fact, a number of studies have already indicated the possible association between several TPH2 polymorphisms and major depression [25–28], suicidal behavior [28, 29], ADHD [30, 31], autism [32], bipolar disorder [33] and obsessive-compulsive disorder [34]. In contrast, other studies have documented in different cohorts a lack of association between TPH2 polymorphisms and suicidal behavior, schizophrenia, bipolar disorder or depression [35–37]. Discrepancies in such studies are difficult to reconcile, but not unexpected given the different cohorts used and perhaps the limitations in diagnostic criteria [38]. It is also noteworthy that most of the SNP in TPH1 and TPH2 identified in the above-mentioned studies are located in the introns or promoter region, thus corresponding to noncoding SNPs without clear functional consequences. These observations underscore the importance of determining the functional consequences of any given SNP in order to address whether such SNP(s) would affect TPH2 gene expression (e.g., transcriptional regulation, splicing) and/or function (e.g., enzyme kinetics, protein folding, protein stability). As an example of the importance of the functional consequences of genetic variants, the polymorphism in the serotonin transporter (SERT) promoter region, which is associated with anxiety and depression [39–41], has been functionally characterized and shown to result in decreased transcriptional efficiencies.
Serotonin-related neuropsychiatric disorders are complex heterogeneous disorders with markedly different clinical profiles and responses to drugs [3, 4], and may also represent disorders with different endophenotypes [2]. Genetic, environmental as well as biochemical influences are thought to represent the contributing factors for these polygenic disorders which are associated with polymorphism(s) in several genes, including the serotonin transporter, serotonin receptors, the vesicular monoamine transporter, monoamine oxidases, as well as TPH1 and TPH2 [1–3]. Two hypotheses have been suggested to explain the genetic basis of polygenic disorders: the common disease/common variant (CD/CV) hypothesis, where disease susceptibility variants are common in the population, and the common disease/rare variant (CD/RV) hypothesis, with large numbers of rare variants at many loci [42]. The multiple functional polymorphisms in PAH (see below) and their association with phenylketonuria (PKU), a metabolic disease with an incidence of 1 per 15,000 in the United States [43] is one example supporting the CD/RV hypothesis. Similarly, more than 100 missense mutations in superoxide dismutase-1 (SOD1) have been identified in amyotrophic lateral sclerosis (ALS), a disorder with a frequency of 4–6 per 100,000, yet only 2–3% of ALS patients carry SOD1 mutations [44]. With regard to the TPH2 gene, our laboratory has recently identified a G1463A SNP in human TPH2 resulting in a severe loss-of-function in TPH2, where a R441H mutation translates to an ∼80% decrease in 5-HT production when assessed in cell culture systems [27]. The G1463A SNP was identified in a cohort of unipolar depression subjects who were resistant to various forms of pharmacological treatments. Among the 9 subjects carrying the G1463A SNP in a cohort of 87 subjects, 5 subjects were treated with electroconvulsive therapy (ECT), while only 16 subjects in this cohort received ECT treatment.
TPH2 mutations
To date, over 500 SNPs have been identified in the TPH2 gene among different species, including more than 300 SNPs in human (http://www.ncbi.nlm.nih.gov). Among these SNPs, there are 6 coding non-synonymous SNPs (L36P, P206S, A328V, R441H, D479E in humans and P447R in mice) and 3 coding synonymous SNPs (P312P, L327L, A375A in humans) [24, 27, 45]. Although the coding sequence represent only ∼1.5% of the TPH2 gene (93, 600 bp), the prevalence of SNPs (9 to this point) in coding regions of TPH2 is reminiscent of the genetic variants in the PAH gene [46, 47]. At present, ∼500 mutations, including more than 300 missense mutations, have been identified in patients with symptoms ranging from mild hyperphenylalaninemia to severe PKU [46, 47]. Importantly, the amino acids in PAH whose mutations either cause protein misfolding or affect substrate and/or BH4 binding [46, 48] are identical in TPH2 (fig. 1). As an example, the R441H mutation in TPH2 identified in the unipolar major depression patients that severely impairs 5-HT production when expressed in cell culture systems [27] is identical to Arg408 in PAH whose mutation (R408W) is the most prevalent and severe pathogenic mutation in PAH [43, 46, 47] Based on the considerable structural and sequence similarities between TPH2 and PAH [12], it is likely that additional rare functional mutations in TPH2 will be identified, and that the presence of multiple susceptibility genes and/or multiple mutations in a single gene may contribute to the complex polygenic nature and wide range of clinical profiles of 5-HT-related neuropsychiatric conditions. It is also possible that the existence of distinct SNPs in TPH2 with different degrees of TPH2 dysfunction could contribute to the development of phenotypes associated with different neuropsychiatric conditions. Therefore, functional PAH mutations could provide an important roadmap for understanding the potential mechanisms of TPH2 dysfunction.
Figure 1.
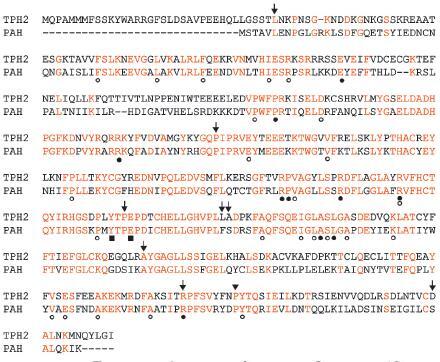
Sequence alignment of TPH2 and PAH in human. Coding synonymous and coding non-synonymous mutations in human TPH2 are shown with arrows. The coding non-synonymous mutation in mouse Tph2 is shown with an arrowhead. PAH mutations that affect substrate binding, protein folding and BH4 binding are indicated with filled boxes, closed circles and open circles, respectively (gene accession numbers: NM_173353 [TPH2], NM_000277 [PAH]).
Mutations identified in TPH2 may generate missense and nonsense mutations (coding mutations), splicing variants (intronic mutations) or other regulatory variants to affect gene function. Therefore, in order to develop a better understanding of the relationships between mutations and functions, it is important to functionally characterize TPH2 mutations and correlate them to 5-HT-related neuropsychiatric disorders. Clearly, a straightforward approach is to using TPH2 complementary DNA (cDNA) to assess 5-HT production of coding non-synonymous SNPs in TPH2, whose potential molecular mechanisms could be predicted based on previous studies of loss-of-function mutations in PAH. Non-coding SNPs of TPH2 (i.e., SNPs in promoter regions, untranslated regions, introns) may potentially regulate TPH2 gene functions by transcriptional regulation and splicing. It should also be mentioned that, while coding synonymous SNPs are typically called ‘silent mutations’, more and more evidence indicates that some of these ‘silent mutations’ may play an important role in regulating premRNA splicing as a mechanism of gene regulation [49]. For example, an exon-skipping mutation caused by a coding synonymous SNP in PAH was characterized from a previously categorized ‘silent mutation’ [50]. Thus, it is necessary to perform detailed studies of TPH2 gene function using both cDNA and genomic DNA.
PC12 cells as a model system for characterizing TPH2
Although expression of recombinant protein in Escherichia coli is indispensable and has been widely used to obtain purified TPH1 [51, 52], TPH2 [53] and PAH [46] in large quantity for further detailed structural and functional analysis, mammalian cell culture systems can provide conditions for protein expression which are closer to the in vivo environment. We have established the pheochromocytoma PC12 cells as a model system to study 5-HT production [24, 27]. PC12 cells are neuroen-docrine cells that endogenously synthesize dopamine and norepinephrine, but not 5-HT [54], indicating that PC12 cells possess essential elements (e.g., BH4 and aromatic amino acid decarboxylase [AADC]) for 5-HT synthesis and are capable of synthesizing 5-HT when TPH2 is exogenously expressed. Moreover, the presence of endogenous dopamine in PC12 cells, which is easily detectable along with 5-HT, provides an ideal internal control for quantification. Walther et al. [15] have previously expressed TPH2 in COS7 cells, which do not express AADC, and measured 5-HTP production as an approach to effectively monitor the rate of TPH2 synthesis. Regulation in 5-HT production (e.g., phosphorylation [53]) and interaction between TPH2 and accessory proteins (e.g., 14-3-3 protein [53]) can also be investigated in mammalian cell culture systems. TPH2, as well as other members of the superfamily of aromatic amino acid hydroxylases, forms homotetramers [12]. Previous studies using recombinant TPH1 [51] and PAH [47] revealed a tendency of the purified proteins to form aberrant oligomers and aggregates, a phenomenon which may or may not occur in vivo. Therefore, expression of TPH2 in mammalian cell systems can provide a rapid functional analysis mimicking the in vivo condition to identify potential mutation(s) that affect protein folding and stability in vivo.
Animal models for studying 5-HT-related behaviors
It has been noted for many years that there are substantial behavioral differences among different inbred mouse strains, particularly in behavioral assessments using the tail suspension test, forced swim test, alcohol preference and aggression [55–58]. However, the underlying mechanism governing these differences has been unknown. We recently identified a functional (C1473G) SNP in mouse Tph2 that exhibited an ∼55% reduction in Tph2 activity when expressed in PC12 cells [24]. Importantly, BALB/cJ and DBA/2 inbred mice carrying the homozygous 1473G allele exhibited an ∼50% decrease in the rate of brain 5-HT synthesis and tissue content as compared with C57Bl/6 and 129X1/SvJ inbred mice carrying the homozygous 1473C allele [24]. These results provide potential molecular and genetic explanations for the behavioral differences among these strains of mice. Interestingly, (C1473G) functional polymorphism in mouse Tph2 gene was recently associated with differences in aggressive behavior [59], pre-mRNA editing of the 5-HT2C receptor [60] and responsiveness to selective serotonin reuptake inhibitor (SSRI) [61, 62], but not to certain aspects of impulsive behaviors [63]. These studies have independently confirmed the homozygous 1473C allele in C57Bl/6, 129X1/SvJ strains and the homozygous 1473G allele in BALB/cJ, DBA/2 strains. In addition, it has been shown that other inbred strains carry either the homozygous 1473C allele (ARK/J, C3H/HeJ, CBA/Lac, DD/He, PT/Y, YT/Y) or the homozygous 1473G allele (A/He, A/J, CBA/Ca, CC57BR/Mv) [59, 62, 63]. It should be emphasized that the behavioral studies mentioned above have used these different inbred mouse strains for comparison; thus the influence of potential variations in other genes cannot be ruled out. Therefore, to precisely address the role of functional polymorphism(s) in behavioral studies, it is imperative to generate congenic mice carrying either the 1473C or 1473G allele using a backcross breeding strategy (fig. 2). Congenic mice carrying 1473C/C, 1473C/G or 1473G/G alleles will enable assessment of the role of brain 5-HT while minimizing the potential contribution of other modifier genes. An alternative strategy would be to generate Tph2 knock-out mice or knock-in mice carrying functional mutations of TPH2. Such transgenic mice, along with the congenic mice described above, may recapitulate behavioral manifestations of 5-HT-related neuropsychiatric conditions and provide valuable tools to test therapeutic pharmacological interventions.
Figure 2.
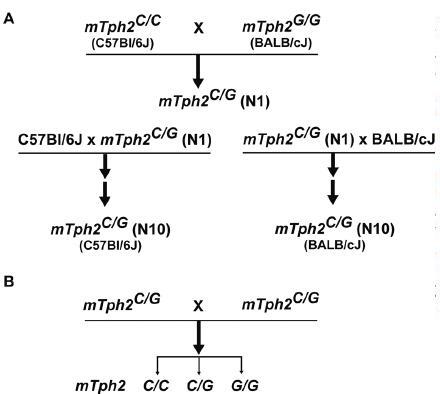
Strategy to generate congenic mouse lines carrying either 1473C or 1473G allele in Tph2. (A) Backcross breeding of C57Bl/6J carrying homozygous 1473C and BALB/cJ carrying homozygous 1473G in Tph2. (B) Using C57Bl/6J or BALB/cJ mice carrying heterozygous 1473C/G congenic mice to generate congenic mice carrying 1473C/C, 1473C/G and 1473G/G alleles in Tph2.
Summary
Brain 5-HT plays an important role in regulating complex behaviors, and dysregulation of brain 5-HT homeostasis may contribute to many neuropsychiatric disorders. The identification of the role of TPH2 in brain 5-HT synthesis has opened a new area to explore the molecular and genetic mechanisms of 5-HT-related conditions. Functional and genetic analyses are two powerful, indispensable and complementary tools to address the role of TPH2. These studies should also pave the way for the development of animal models for behavioral and pharmacological studies. Therefore, characterization of TPH2 and its functional SNPs may ultimately provide important insights into the pathophysiology of 5-HT-related neuropsychiatric disorders.
Acknowledgement
This work was supported by grants from the National Institutes of Health (MH-40159 and NS-19576). M.G.C. is the recipient of a Distinguished Investigator Award from NARSAD. J.-M.B is recipients of a Young Investigator Award from NARSAD and fellowships from the Human Frontier Research Program and the Canadian Institutes of Health Research.
Footnotes
Received 12 September 2005; received after revision 25 October 2005; accepted 31 October 2005
References
- 1.Arango V., Huang Y. Y., Underwood M. D., Mann J. J. Genetics of the serotonergic system in suicidal behavior. J. Psychiatr. Res. 2003;37:375–386. doi: 10.1016/S0022-3956(03)00048-7. [DOI] [PubMed] [Google Scholar]
- 2.Hasler G., Drevets W. C., Manji H. K., Charney D. S. Discovering endophenotypes for major depression. Neuropsychopharmacology. 2004;29:1765–1781. doi: 10.1038/sj.npp.1300506. [DOI] [PubMed] [Google Scholar]
- 3.Lesch K. P. Gene-environment interaction and the genetics of depression. J. Psychiatry Neurosci. 2004;29:174–184. [PMC free article] [PubMed] [Google Scholar]
- 4.Malhotra A. K., Murphy G. M., Kennedy J. L. Pharmacogenetics of psychotropic drug response. Am. J. Psychiatry. 2004;161:780–796. doi: 10.1176/appi.ajp.161.5.780. [DOI] [PubMed] [Google Scholar]
- 5.Miyamoto S., LaMantia A. S., Duncan G. E., Sullivan P., Gilmore J. H., Lieberman J. A. Recent advances in the neurobiology of schizophrenia. Mol. Interv. 2003;3:27–39. doi: 10.1124/mi.3.1.27. [DOI] [PubMed] [Google Scholar]
- 6.Courtet P., Jollant F., Castelnau D., Buresi C., Malafosse A. Suicidal behavior: relationship between phenotype and serotonergic genotype. Am. J. Med. Genet. 2005;133C:25–33. doi: 10.1002/ajmg.c.30043. [DOI] [PubMed] [Google Scholar]
- 7.Gainetdinov R. R., Wetsel W. C., Jones S. R., Levin E. D., Jaber M., Caron M. G. Role of serotonin in the paradoxical calming effect of psychostimulants on hyperactivity. Science. 1999;283:397–401. doi: 10.1126/science.283.5400.397. [DOI] [PubMed] [Google Scholar]
- 8.Quist J. F., Kennedy J. L. Genetics of childhood disorders: XXIII. ADHD, Part 7: The serotonin system. J. Am. Acad. Child Adolesc. Psychiatry. 2001;40:253–256. doi: 10.1097/00004583-200102000-00022. [DOI] [PubMed] [Google Scholar]
- 9.Bobb A. J., Castellanos F. X., Addington A. M., Rapoport J. L. Molecular genetic studies of ADHD: 1991 to 2004. Am. J. Med. Genet. 2005;132B:109–125. [PubMed] [Google Scholar]
- 10.Blier P., Abbott F. V. Putative mechanisms of action of antidepressant drugs in affective and anxiety disorders and pain. J. Psychiatry Neurosci. 2001;26:37–43. [PMC free article] [PubMed] [Google Scholar]
- 11.Veenstra-VanderWeele J., Cook E. H. Molecular genetics of autism spectrum disorder. Mol. Psychiatry. 2004;9:819–832. doi: 10.1038/sj.mp.4001505. [DOI] [PubMed] [Google Scholar]
- 12.Fitzpatrick P. F. Tetrahydropterin-dependent amino acid hydroxylases. Annu. Rev. Biochem. 1999;68:355–381. doi: 10.1146/annurev.biochem.68.1.355. [DOI] [PubMed] [Google Scholar]
- 13.Kim K. S., Wessel T. C., Stone D. M., Carver C. H., Joh T. H., Park D. H. Molecular cloning and characterization of cDNA encoding tryptophan hydroxylase from rat central serotonergic neurons. Brain Res. Mol. Brain Res. 1991;9:277–283. doi: 10.1016/0169-328x(91)90073-7. [DOI] [PubMed] [Google Scholar]
- 14.Haycock J. W., Kumer S. C., Lewis D. A., Vrana K. E., Stockmeier C. A. A monoclonal antibody to tryptophan hydroxylase: applications and identification of the epitope. J. Neurosci. Methods. 2002;114:205–212. doi: 10.1016/S0165-0270(01)00530-1. [DOI] [PubMed] [Google Scholar]
- 15.Walther D. J., Peter J. U., Bashammakh S., Hortnagl H., Voits M., Fink H., et al. Synthesis of serotonin by a second tryptophan hydroxylase isoform. Science. 2003;299:76. doi: 10.1126/science.1078197. [DOI] [PubMed] [Google Scholar]
- 16.Grahame-Smith D. G. Tryptophan hydroxylation in brain. Biochem. Biophys. Res. Commun. 1964;16:586–592. doi: 10.1016/0006-291X(64)90197-4. [DOI] [PubMed] [Google Scholar]
- 17.Yu P. L., Fujimura M., Okumiya K., Kinoshita M., Hasegawa H., Fujimiya M. Immunohistochemical localization of tryptophan hydroxylase in the human and rat gastrointestinal tracts. J. Comp. Neurol. 1999;411:654–665. doi: 10.1002/(SICI)1096-9861(19990906)411:4<654::AID-CNE9>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 18.Cote F., Thevenot E., Fligny C., Fromes Y., Darmon M., Ripoche M. A., et al. Disruption of the nonneuronal tph1 gene demonstrates the importance of peripheral serotonin in cardiac function. Proc. Natl. Acad. Sci. USA. 2003;100:13525–13530. doi: 10.1073/pnas.2233056100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Patel P. D., Pontrello C., Burke S. Robust and tissue-specific expression of TPH2 versus TPH1 in rat raphe and pineal gland. Biol. Psychiatry. 2004;55:428–433. doi: 10.1016/j.biopsych.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 20.Zill P., Buttner A., Eisenmenger W., Moller H. J., Ackenheil M. and Bondy B. (2005) Analysis of tryptophan hydroxylase I and II mRNA expression in the human brain: a post-mortem study. J. Psychiatr. Res. [doi:10.1016/j.jpsychires.2005.05.004] [DOI] [PubMed]
- 21.Coon S. L., Mazuruk K., Bernard M., Roseboom P. H., Klein D. C., Rodriguez I. R. The human serotonin N-acetyltransferase (EC 2.3.1.87) gene (AANAT): structure, chromosomal localization and tissue expression. Genomics. 1996;34:76–84. doi: 10.1006/geno.1996.0243. [DOI] [PubMed] [Google Scholar]
- 22.Teraoka H., Russell C., Regan J., Chandrasekhar A., Concha M. L., Yokoyama R., et al. Hedgehog and Fgf signaling pathways regulate the development of tphR-expressing serotonergic raphe neurons in zebrafish embryos. J. Neurobiol. 2004;60:275–288. doi: 10.1002/neu.20023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Coleman C. M., Neckameyer W. S. serotonin synthesis by two distinct enzymes in Drosophila melanogaster . Arch. Insect Biochem. Physiol. 2005;59:12–31. doi: 10.1002/arch.20050. [DOI] [PubMed] [Google Scholar]
- 24.Zhang X., Beaulieu J. M., Sotnikova T. D., Gainetdinov R. R., Caron M. G. Tryptophan hydroxylase-2 controls brain serotonin synthesis. Science. 2004;305:217. doi: 10.1126/science.1097540. [DOI] [PubMed] [Google Scholar]
- 25.Peters E. J., Slager S. L., McGrath P. J., Knowles J. A., Hamilton S. P. Investigation of serotonin-related genes in antidepressant response. Mol. Psychiatry. 2004;9:879–889. doi: 10.1038/sj.mp.4001502. [DOI] [PubMed] [Google Scholar]
- 26.Zill P., Baghai T. C., Zwanzger P., Schule C., Eser D., Rupprecht R., et al. SNP and haplotype analysis of a novel tryptophan hydroxylase isoform (TPH2) gene provide evidence for association with major depression. Mol. Psychiatry. 2004;9:1030–1036. doi: 10.1038/sj.mp.4001525. [DOI] [PubMed] [Google Scholar]
- 27.Zhang X., Gainetdinov R. R., Beaulieu J. M., Sotnikova T. D., Burch L. H., Williams R. B., et al. Loss-of-function mutation in tryptophan hydroxylase-2 identified in unipolar major depression. Neuron. 2005;45:11–16. doi: 10.1016/j.neuron.2004.12.014. [DOI] [PubMed] [Google Scholar]
- 28.Zhou Z., Roy A., Lipsky R., Kuchipudi K., Zhu G., Taubman J., et al. Haplotype-based linkage of tryptophan hydroxylase 2 to suicide attempt, major depression and cerebrospinal fluid 5-hydroxyindoleacetic acid in 4 populations. Arch. Gen. Psychiatry. 2005;62:1109–1118. doi: 10.1001/archpsyc.62.10.1109. [DOI] [PubMed] [Google Scholar]
- 29.Zill P., Buttner A., Eisenmenger W., Moller H. J., Bondy B., Ackenheil M. Single nucleotide polymorphism and haplotype analysis of a novel tryptophan hydroxylase isoform (TPH2) gene in suicide victims. Biol. Psychiatry. 2004;56:581–586. doi: 10.1016/j.biopsych.2004.07.015. [DOI] [PubMed] [Google Scholar]
- 30.Sheehan K., Lowe N., Kirley A., Mullins C., Fitzgerald M., Gill M., et al. Tryptophan hydroxylase 2 (TPH2) gene variants associated with ADHD. Mol. Psychiatry. 2005;10:944–949. doi: 10.1038/sj.mp.4001698. [DOI] [PubMed] [Google Scholar]
- 31.Walitza S., Renner T. J., Dempfle A, Konrad K, Wewetzer C, Halbach A. et al. (2005) Transmission disequilibrium of polymorphic variants in the tryptophan hydroxylase-2 gene in attention-deficit/hyperactivity disorder. Mol. Psychiatry [doi:10. 1038/sj.mp.4001734] [DOI] [PubMed]
- 32.Coon H., Dunn D., Lainhart J., Miller J., Hamil C., Battaglia A., et al. Possible association between autism and variants in the brain-expressed tryptophan hydroxylase gene (TPH2) Am. J. Med. Genet. 2005;135B:42–46. doi: 10.1002/ajmg.b.30168. [DOI] [PubMed] [Google Scholar]
- 33.Harvey M., Shink E., Tremblay M., Gagne B., Raymond C., Labbe M., et al. Support for the involvement of TPH2 gene in affective disorders. Mol. Psychiatry. 2004;9:980–981. doi: 10.1038/sj.mp.4001557. [DOI] [PubMed] [Google Scholar]
- 34.Mossner R., Walitza S., Geller F., Scherag A., Gutknecht L., Jacob C. et al. (2005) Transmission disequilibrium of polymorphic variants in the tryptophan hydroxylase-2 gene in children and adolescents with obsessive-compulsive disorder. Int. J. Neuropsychopharmacol. [doi:10.107/sia61145705005997] [DOI] [PubMed]
- 35.De Luca V., Mueller D. J., Tharmalingam S., King N., Kennedy J. L. Analysis of the novel TPH2 gene in bipolar disorder and suicidality. Mol. Psychiatry. 2004;9:896–897. doi: 10.1038/sj.mp.4001531. [DOI] [PubMed] [Google Scholar]
- 36.De Luca V., Voineskos D., Wong G. W., Shinkai T., Rothe C., Strauss J., et al. Promoter polymorphism of second tryptophan hydroxylase isoform (TPH2) in schizophrenia and suicidality. Psychiatry Res. 2005;134:195–198. doi: 10.1016/j.psychres.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 37.Garriock H. A., Allen J. J., Delgado P., Nahaz Z., Kling M. A., Carpenter L., et al. Lack of association of TPH2 exon XI polymorphisms with major depression and treatment resistance. Mol. Psychiatry. 2005;10:976–977. doi: 10.1038/sj.mp.4001712. [DOI] [PubMed] [Google Scholar]
- 38.Krishnan K. R. Psychiatric disease in the genomic era: rational approach. Mol. Psychiatry. 2005;10:978–984. doi: 10.1038/sj.mp.4001723. [DOI] [PubMed] [Google Scholar]
- 39.Lesch K. P., Bengel D., Heils A., Sabol S. Z., Greenberg B. D., Petri S., et al. Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science. 1996;274:1527–1531. doi: 10.1126/science.274.5292.1527. [DOI] [PubMed] [Google Scholar]
- 40.Caspi A., Sugden K., Moffitt T. E., Taylor A., Craig I. W., Harrington H. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science. 2003;301:386–389. doi: 10.1126/science.1083968. [DOI] [PubMed] [Google Scholar]
- 41.Pezawas L., Meyer-Lindenberg A., Drabant E. M., Verchinski B. A., Munoz K. E., Kolachana B. S., et al. 5-HTTLPR polymorphism impacts human cingulate-amygdala interactions: a genetic susceptibility mechanism for depression. Nat. Neurosci. 2005;8:828–834. doi: 10.1038/nn1463. [DOI] [PubMed] [Google Scholar]
- 42.Wright A., Charlesworth B., Rudan I., Carothers A., Campbell H. A polygenic basis for late-onset disease. Trends Genet. 2003;19:97–106. doi: 10.1016/S0168-9525(02)00033-1. [DOI] [PubMed] [Google Scholar]
- 43.Scriver C. R., Hurtubise M., Konecki D., Phommarinh M., Prevost L., Erlandsen H., et al. PAHdb 2003: what a locus-specific knowledgebase can do. Hum. Mutat. 2003;21:333–344. doi: 10.1002/humu.10200. [DOI] [PubMed] [Google Scholar]
- 44.Bendotti C., Carri M. T. Lessons from models of SOD1-linked familial ALS. Trends Mol. Med. 2004;10:393–400. doi: 10.1016/j.molmed.2004.06.009. [DOI] [PubMed] [Google Scholar]
- 45.Breidenthal S. E., White D. J., Glatt C. E. Identification of genetic variants in the neuronal form of tryptophan hydroxylase (TPH2) Psychiatr. Genet. 2004;14:69–72. doi: 10.1097/01.ypg.0000107929.32051.58. [DOI] [PubMed] [Google Scholar]
- 46.Pey A. L., Desviat L. R., Gamez A., Ugarte M., Perez B. Phenylketonuria: genotype-phenotype correlations based on expression analysis of structural and functional mutations in PAH. Hum. Mutat. 2003;21:370–378. doi: 10.1002/humu.10198. [DOI] [PubMed] [Google Scholar]
- 47.Waters P. J. How PAH gene mutations cause hyperphenylalaninemia and why mechanism matters: insights from in vitro expression. Hum. Mutat. 2003;21:357–369. doi: 10.1002/humu.10197. [DOI] [PubMed] [Google Scholar]
- 48.Erlandsen H., Pey A. L., Gamez A., Perez B., Desviat L. R., Aguado C., et al. Correction of kinetic and stability defects by tetrahydrobiopterin in phenylketonuria patients with certain phenylalanine hydroxylase mutations. Proc. Natl. Acad. Sci. USA. 2004;101:16903–16908. doi: 10.1073/pnas.0407256101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cartegni L., Chew S. L., Krainer A. R. Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat. Rev. Genet. 2002;3:285–298. doi: 10.1038/nrg775. [DOI] [PubMed] [Google Scholar]
- 50.Chao H.-K., Hsiao K.-J., Su T.-S. A silent mutation induces exon skipping in the phenylalanine hydroxylase gene in phenylketonuria. Hum. Genet. 2001;108:14–19. doi: 10.1007/s004390000435. [DOI] [PubMed] [Google Scholar]
- 51.Yang X. J., Kaufman S. High-level expression and deletion mutagenesis of human tryptophan hydroxylase. Proc. Natl. Acad. Sci. USA. 1994;91:6659–6663. doi: 10.1073/pnas.91.14.6659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang L., Erlandsen H., Haavik J., Knappskog P. M., Stevens R. C. Three-dimensional structure of human tryptophan hydroxylase and its implications for the biosynthesis of the neurotransmitters serotonin and melatonin. Biochemistry. 2002;41:12569–12574. doi: 10.1021/bi026561f. [DOI] [PubMed] [Google Scholar]
- 53.McKinney J., Knappskog P. M., Haavik J. Different properties of the central and peripheral forms of human tryptophan hydroxylase. J. Neurochem. 2005;92:311–320. doi: 10.1111/j.1471-4159.2004.02850.x. [DOI] [PubMed] [Google Scholar]
- 54.Greene L. A., Tischler A. S. Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc. Natl. Acad. Sci. USA. 1976;73:2424–2428. doi: 10.1073/pnas.73.7.2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Popova N. K., Nikulina E. M., Kulikov A. V. Genetic analysis of different kinds of aggressive behavior. Behav. Genet. 1993;23:491–497. doi: 10.1007/BF01067985. [DOI] [PubMed] [Google Scholar]
- 56.Lucki I., Dalvi A., Mayorga A. J. Sensitivity to the effects of pharmacologically selective antidepressants in different strains of mice. Psychopharmacology. 2001;155:315–322. doi: 10.1007/s002130100694. [DOI] [PubMed] [Google Scholar]
- 57.Ripoll N., David D. J., Dailly E., Hascoet M., Bourin M. Antidepressant-like effects in various mice strains in the tail suspension test. Behav. Brain Res. 2003;143:193–200. doi: 10.1016/S0166-4328(03)00034-2. [DOI] [PubMed] [Google Scholar]
- 58.Wahlsten D., Metten P., Phillips T. J., Boehm S. L., Burkhart-Kasch S., Dorow J., et al. Different data from different labs: lessons from studies of gene- environment interaction. J. Neurobiol. 2003;54:283–311. doi: 10.1002/neu.10173. [DOI] [PubMed] [Google Scholar]
- 59.Kulikov A. V., Osipova D. V., Naumenko V. S., Popova N. K. Association between Tph2 gene polymorphism, brain tryptophan hydroxylase activity and aggressiveness in mouse strains. Genes Brain Behav. 2005;4:482–485. doi: 10.1111/j.1601-183X.2005.00145.x. [DOI] [PubMed] [Google Scholar]
- 60.Englander M. T., Dulawa S. C., Bhansali P., Schmauss C. How stress and fluoxetine modulate serotonin 2C receptor pre-mRNA editing. J. Neurosci. 2005;25:648–651. doi: 10.1523/JNEUROSCI.3895-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cervo L., Canetta A., Calcagno, Burbassi S., Sacchetti G., Caccia S., et al. Genotype-dependent activity of tryptophan hydroxylase-2 determines the response to citalopram in a mouse model of depression. J. Neurosci. 2005;25:8165–8172. doi: 10.1523/JNEUROSCI.1816-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Crowley J. J., Blendy J. A., Lucki I. Strain-dependent antidepressant-like effects of citalopram in the mouse tail suspension test. Psychopharmacology. 2005;183:257–264. doi: 10.1007/s00213-005-0166-5. [DOI] [PubMed] [Google Scholar]
- 63.Isles A. R., Hathway G. J., Humby T., de la Riva C., Kendrick K. M., Wilkinson L. S. An mTph2 SNP gives rise to alterations in extracellular 5-HT levels, but not in performance on a delayed-reinforcement task. Eur. J. Neurosci. 2005;22:997–1000. doi: 10.1111/j.1460-9568.2005.04265.x. [DOI] [PubMed] [Google Scholar]


