Abstract
Pentoxifylline-loaded poly(ε-caprolactone) microspheres were prepared by solvent evaporation technique with different drug to carrier ratio F1 (1:3), F2 (1:4), F3 (1:5) and F4 (1:6). The microspheres were characterized for particle size, scanning electron microscopy, FT-IR study, percentage yield, drug entrapment, stability studies and for in vitro release kinetics. The shape of microspheres was found to be spherical by SEM. The size of microspheres was found to be ranging 59.3±6.3μm to 86.22±4.23 μm. Among the four drug to carrier ratio, F3 (1:5) showed maximum percentage yield of 83.34±2.46% and F2 (1:4) showed highest drug entrapment of 76.92±3.24% w/w. It was found that there was no interaction between drug and polymer by FT-IR study. No appreciable difference was observed in the extent of degradation of product during 60 d in the microspheres, which were stored at various temperatures. In the in vitro release study formulation F2 (1:4) showed 90.34% drug release at 15 h and found to be sustained. The release followed Higuchi kinetics indicating diffusion controlled drug release.
Keywords: Pentoxifylline, microspheres, poly(ε-caprolactone), biodegradable
The main objective of any drug therapy is to achieve a desire concentration of the drug in blood or tissue which is therapeutically effective and nontoxic for extended period of time, and this goal can be achieved by proper design of sustain release dosage regimen1,2. Microspheres have been widely accepted as a mean to achieve oral and parenteral controlled release3,4. The microspheres require a polymeric substance as a coating material or carrier5. A number of different substances biodegradable as well as non-biodegradable have been investigated6 for the preparation of microspheres. Of the various biodegradable polymers used for the development of sustained release formulations, poly(ε-caprolactone) has been reported to be advantageous since they are biocompatible7,8. Poly(ε-caprolactone) is aliphatic polyester polymer, suitable for controlled drug delivery due to a high permeability to many drugs and at the same time being free from toxicity9,10.
Pentoxifylline, a xanthine derivative, is an analogue of theophylline and inhibits phosphodiesterase. Pentoxifylline and its metabolites improve the flow properties of blood by decreasing its viscosity. In patient with chronic peripheral arterial disease, this increase blood flow to the affected microcirculation and enhance tissue oxygenation. Pentoxifylline has short half-life of 1.6 h and low oral availability (19±13%)11. The aim of this study was to prepare poly(ε-caprolactone) microspheres containing pentoxifylline to achieve a controlled drug release profile suitable for peroral administration.
MATERIALS AND METHODS
Pentoxifylline was obtained as a gift sample from Shreya Health Care, Aurangabad. Poly(ε-caprolactone) was obtained from Fulka Cemika, Sigma-Aldrich Chemie, Switzerland. Dichloromethane was procured from Loba Chem. Pvt. Ltd., Mumbai. All other reagents used were of analytical grade.
Preparation of microspheres:
Poly(ε-caprolactone) microspheres were prepared by solvent evaporation technique12,13. Drug to carrier ratio for different formulation was 1:3 (F1), 1:4 (F2), 1:5 (F3) and 1:6 (F4). Accurately weighed quantity of the poly(ε-caprolactone) was dissolved in 10 ml of dichloromethane than 200 mg of pentoxifylline was dissolved in this polymer phase. This solution was poured in 100 ml of liquid paraffin containing 1.3% Tween 80 and continuous stirred for 5 h at 1100 rpm. The microspheres were filtered and washed three times with 50 ml of n-hexane and dried at room temperature for 12 h. Microspheres dried at room temperature were then weighed and the yield of microspheres preparation was calculated using the formula14, Percent yield = (Amount of microspheres obtained/The theoretical amount)×100
Evaluation of the microspheres:
Pentoxifylline was extracted from the microspheres after crushing with phosphate buffer pH 7.4 and absorbance was measured using UV/Vis spectrophotometer (Shimadzu 1601, Japan) at 274 nm. Amount of pentoxifylline in the microspheres was estimated with the help of a standard graph. Particle size analysis was carried out using optical microscopy15. About 200 microspheres were selected randomly and their size was determined using optical microscope fitted with a standard micrometer scale. The surface morphology and the internal textures of microsphers were observed under a scanning electron microscope16 (Jeol JSM-5610, Japan). FT-IR spectra of pentoxifylline, and poly(ε-caprolactone) microsphere loaded with pentoxifylline were taken to check drug polymer interaction and degradation of drug during microencapsulation.
Stability studies:
The microspheres were placed in screw capped glass container and stored at ambient humidity conditions, at room temperatures (27±2°), oven temperature (40±2°) and in refrigerator (5-8°) for a period of 60 d, the microspheres were analyzed for drug content17.
In vitro release studies:
The in vitro release profile of pentoxifylline from microspheres was examined in phosphate buffer pH 7.4 using the rotating paddle method (Electro Lab, Mumbai) under sink conditions18. Accurately weighed samples of microspheres were added to dissolution medium kept at 37±0.5°. At preset time intervals aliquots were withdrawn and replaced by an equal volume of dissolution medium to maintain constant volume. After suitable dilution the samples were analyzed spectrophotometrically at 274 nm19.
Kinetics of drug release:
In order to understand the mechanism and kinetics of drug release, the result of the in vitro dissolution study of microspheres were fitted with various kinetic equations, like zero order20 (percentage release vs. time), first order21 (log percentage of drug remaining to be released vs. time) and Higuchi’s model22 (Percentage drug release vs. square root of time). Correlation coefficient (r2) values were calculated for the linear curves obtained by regression analysis of the above plots.
RESULTS AND DISCUSSION
Poly(ε-caprolactone) microspheres of pentoxifylline were prepared by solvent evaporation technique. Poly(ε-caprolactone) was selected as a polymer for the preparation of microspheres due to its biodegradable and biocompatible properties. The scanning electron microphotograph of microspheres is shown in fig. 1, it indicated that microspheres were spherical and discrete. The particle size was analyzed by optical microcopy. The particle size differed due to variation in the composition of the formulation. The particle size gradually increased with increasing in the proportion of poly(ε-caprolactone). The mean particle size of the microspheres is shown in Table 1. The percentage yield and entrapment efficiency were high for all the formulations and were in the range of 79.63±2.49-83.34±2.46% and 71.96±2.94-76.92±3.24% w/w, respectively, as shown in Table 1. Among the 4 drugs to carrier ratio F3 showed maximum percentage yield of 83.34±2.46% and F2 showed highest drug entrapment of 76.92±3.24% w/w.
Fig. 1.

Scanning Electron Microphotograph of pentoxifylline loaded Poly(ε-caprolactone) microspheres
Scanning electron microphotograph of pentoxifylline loaded Poly(ε- caprolactone) microspheres was recorded at 200 X magnification to characterize shape and surface properties of the microspheres.
TABLE 1.
YIELD, DRUG ENTRAMENT AND AVERAGE PARTICLE SIZE OF PENTOXIFYLLINE LOADED POLY (ε-CAPROLACTONE) MICROSPHERES
| Formulation code | Drug: polymer | Percent yield* | Drug entrapment* % w/w | Average particle size* (μm) |
|---|---|---|---|---|
| F1 | 1 : 3 | 79.63±2.49 | 73.14±2.64 | 59.3±6.31 |
| F2 | 1 : 4 | 80.97±3.66 | 76.92±3.24 | 65.6±4.46 |
| F3 | 1 : 5 | 83.34±2.46 | 74.69±2.38 | 78.52±7.41 |
| F4 | 1 : 6 | 81.28±4.00 | 71.96±2.94 | 86.22±4.23 |
Average of three preparation ± SD
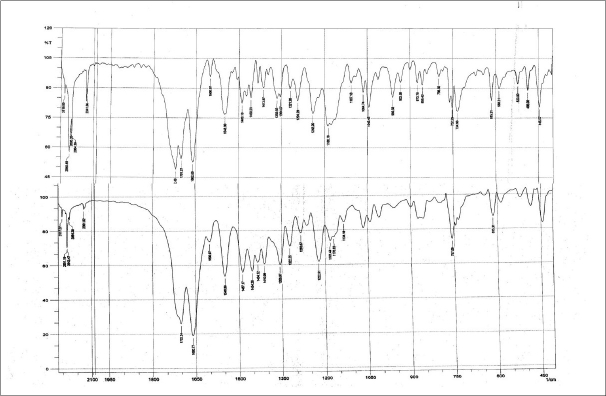
The FT-IR spectra obtained for pentoxifylline and pentoxifylline-loaded poly(ε-caprolactone) microspheres (fig. 2). The result indicated that the characteristic peaks due to pure pentoxifylline have appeared in microspheres, without any change in their position after successful encapsulation, indicating no chemical interaction between pentoxifylline and poly(ε-caprolactone) and the stability of drug during microencapsulation process. In the stability studies, no appreciable difference was observed in the extent of degradation of products during 60 d in the microspheres which were stored at various temperatures.
Fig. 2.
FT-IR Spectra obtained for pure pentoxifylline and pentoxifylline loaded poly (ε-caprolactone) microspheres
FT-IR spectra obtained for pentoxifylline and pentoxifylline loaded poly(ε-caprolactone) microspheres were recorded to check drug-polymer interaction.
The cumulative percent release of pentoxifylline from different formulations is shown in fig. 3. Pentoxifylline release from all the formulations was slow and sustained over 15 h. The drug release rate was decreasing on increasing the polymer ratio. By the end of 15h formulation F1, F2, F3 and F4 released 92.21, 90.34, 76.65 and 63.39% of loaded drug, respectively. The polymer drug ratio 1:4 (F2) showed better drug entrapment and release pattern. It controlled the drug release over 15 h and was found to be the most suitable among other formulations.
Fig. 3.
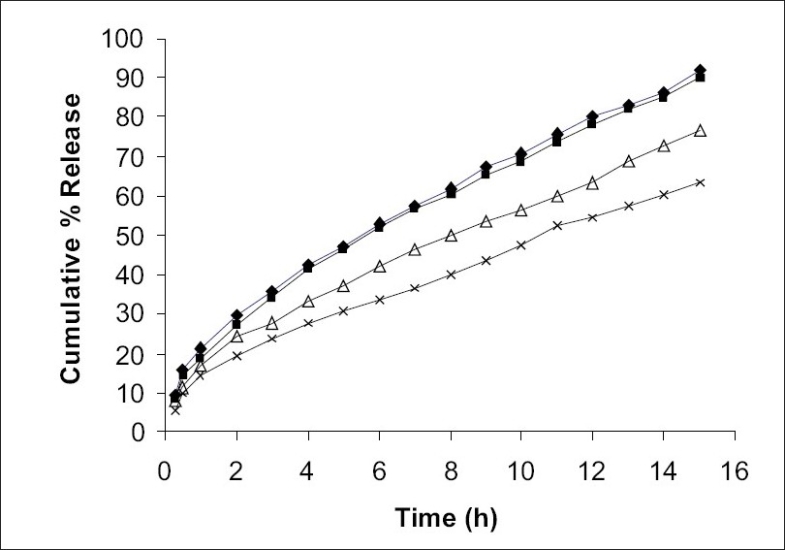
In vitro drug release of pentoxifylline from poly(ε caprolactone) microspheres
In vitro dissolution profiles of pentoxifylline from poly(ε- caprolactone) microspheres formulation F1 (-♦-), F2(-▪-), F3(-Δ-) and F4(-×-) were studied in pH 7.4 phosphate buffer over a period of 15 h.
The in vitro release data were applied to various kinetics models to predict the drug release mechanism and kinetics. The drug release mechanism from the microspheres was diffusion controlled as plots of the amount released versus square root of time (fig. 4) was found to be linear. The correlation coefficient (r2 ) was in the range of 0.978-0.987 for various formulations as shown in Table 2. When log percentage of drug remaining to be released vs. time was plotted in accordance with first order equation, straight lines were obtained (r2>0.95) indicated that drug release followed first order kinetics (fig. 5).
Fig. 4.
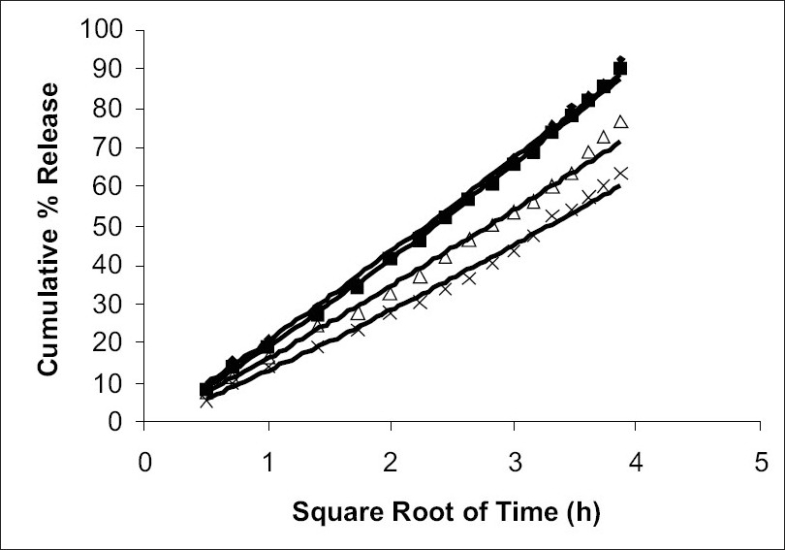
Diffusion controlled release profiles
Diffusion controlled release profiles of pentoxifylline from poly(ε- caprolactone) microspheres formulation F1(-♦-), F2(-▪-), F3(-Δ-) and F4(-×-) were studied in pH 7.4 phosphate buffer.
TABLE 2.
VALUE OF R2 FROM RELEASE DATA OF VARIOUS FORMULATIONS FOR DIFFERENT MODLES OF MECHANISMS OF DRUG RELEASE
| Model | F1 | F2 | F3 | F4 |
|---|---|---|---|---|
| Zero order | 0.853 | 0.865 | 0.883 | 0.877 |
| First order | 0.994 | 0.983 | 0.965 | 0.954 |
| Higuchi | 0.987 | 0.985 | 0.978 | 0.979 |
The values listed are the values of coefficient correlation (r2) obtained from release data of various formulations for different models of mechanism of drug release.
Fig. 5.
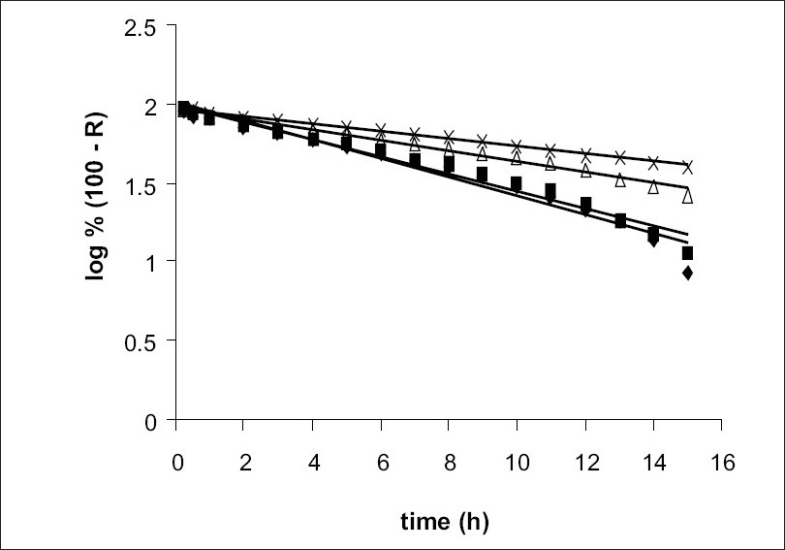
First order release profiles
First order release profiles of pentoxifylline from poly(ε-caprolactone) microspheres formulation F1(-♦-), F2(-▪-), F3(-Δ-) and F4(-×-) were studied in pH 7.4 phosphate buffer.
In present study, an attempt was made to prepare pentoxifylline microspheres using a biodegradable, biocompatible carrier, poly(ε-caprolactone) by solvent evaporation technique. The method was found to be simple and reproducible. It may be concluded from the result obtained from evaluation and performance study of microspheres that system may be useful to achieve a controlled drug release profile suitable for peroral administration and may help to reduce the dose of drug, dosing frequency and improve patient compliance.
Acknowledgments
The author would like to thank Shreya Health Care, Aurangabad for providing gift sample of Pentoxifylline.
REFERENCES
- 1.Lordi NG. Sustained release dosage forms. In: Lachman L, Libermann HA, Kanig JL, editors. Theory and practice of industrial pharmacy. 3rd ed. Mumbai: Vargheese Publishing House; 1991. p. 430. [Google Scholar]
- 2.Torrado JJ, Illum L, Devis SS. Particle size and size distribution of albumin microspheres. Int J Pharm. 1989;51:85–93. [Google Scholar]
- 3.Leung SS, Robinson JR. Parenteral product. In: Robinson JR, Lee VHL, editors. Controlled drug delivery, fundamentals and applications. 2nd ed. New York: Marcel Dekker Inc; 1987. pp. 448–50. [Google Scholar]
- 4.Freeberg S, Zhu XX. Polymer microspheres for controlled drug release. Int J Pharm. 2004;282:1–18. doi: 10.1016/j.ijpharm.2004.04.013. [DOI] [PubMed] [Google Scholar]
- 5.Upadhye K, Bakhle S, Dixit G, Wadetwar R, Deshpande S, Nagulwar V. Preparation and evaluation of gelatin micospheres containing ciproflosacin hydrochloride. Indian Drugs. 2004;41:665–9. [Google Scholar]
- 6.Vyas SP. In: Targeted and controlled drug delivery novel carrier systems. 1st ed. Khar RK, editor. New Delhi: CBS Publishers and Distributors; 2002. p. 418. [Google Scholar]
- 7.Sinha VR, Bansal K, Kaushik R, Kumriq R, Trehan A. Poly-ε-caprolactone microspheres and nanospheres; an overview. Int J Pharm. 2004;278:1–23. doi: 10.1016/j.ijpharm.2004.01.044. [DOI] [PubMed] [Google Scholar]
- 8.Alonso MJ, Garcia ML, Espina M, Valls O, Egea MA. Aceclofenac loaded poly(ε-caprolacone) nanocapsules: Effect of coadjuvants on morphometrical properties and drug entrapment. Boll Chim Farm. 2000;139:114–9. [PubMed] [Google Scholar]
- 9.Murthy RSR. Biodegradable polymers. In: Jain NK, editor. Controlled and novel drug delivery. 1st ed. New Delhi: CBS Publishers & Distributors; 1997. pp. 27–51. [Google Scholar]
- 10.Gibaud S, Gaiq A, Astier A. Slow-release melarsoprol microparticles. Int J Pharm. 2002;243:161–6. doi: 10.1016/s0378-5173(02)00278-8. [DOI] [PubMed] [Google Scholar]
- 11.Hardman JG, Limbird LE, Gilman AG, editors. Goodman and Gilman's, The pharmacological basis of therapeutics. 8th ed. Vol. II. New York: Maxwell Macmillan Publishing Corporation; 1992. p. 1699. [Google Scholar]
- 12.Anperiqdou A, Geogarakis M. Controlled release salbutamol sulphate microcapsules prepared by emulsion solvent evaporation technique and study on the release affected parameters. Int J Pharm. 1995;115:1–8. [Google Scholar]
- 13.Tsankov ST, Lambov N, Minkov E. Regulation of the tetracycline hydrochloride release from polyacrylate microspheres. Pharmazie. 1992;47:125–8. [PubMed] [Google Scholar]
- 14.Haznedar S, Dortunc B. Preparation and in vitro evaluation of Eudragit microspheres containing acetazolamide. Int J Pharm. 2004;269:131–40. doi: 10.1016/j.ijpharm.2003.09.015. [DOI] [PubMed] [Google Scholar]
- 15.Martin A, Bustamante P, Chun AHC. Physical Pharmacy. 4th ed. New Delhi: B. I. Waverty Pvt Ltd; 1991. p. 431. [Google Scholar]
- 16.Dandagi PM, Mastiolimath VS, Gabad AP, Jliger SR. Mucoadhesive microspheres of propranolol hydrochloride for nasal delivery. Indian J Pharm Sci. 2007;69:402–7. [Google Scholar]
- 17.Shabaraya AR, Narayanacharyulia R. Design and evaluation of chitosan microspheres of metoprolol tartrate for sustained release. Indian J Pharm Sci. 2003;65:250–2. [Google Scholar]
- 18.Rajesh KS, Bhowmik BB, Sa B. Effect of polyvinylpyrrolidone on physical characteristics of ketoprofen-loaded polystryrene microparticles. Indian J Pharm Sci. 2003;65:526–9. [Google Scholar]
- 19.Sant VP, Paradkar AR, Nagarsenker MS. Optimization of pentoxifylline liposomes using 24 factorial design. Indian J Pharm Sci. 2002;64:459–64. [Google Scholar]
- 20.Saparia B, Murthy RSR, Solanki A. Preparation and evaluation of chloroquine phosphate microspheres using cross-linked gelatin for long term drug delivery. Indian J Pharm Sci. 2002;64:48–52. [Google Scholar]
- 21.El-Gibaly I. Development and in vitro evaluation of novel floating chitosan microcapsules for oral use: Comparison with nonfloating chitosan microspheres. Int J Pharm. 2002;249:7–21. doi: 10.1016/s0378-5173(02)00396-4. [DOI] [PubMed] [Google Scholar]
- 22.Higuchi T. Mechanism of sustained-action medication: theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52:1145–9. doi: 10.1002/jps.2600521210. [DOI] [PubMed] [Google Scholar]